Objective: To observe (1) the proportion and nature of behavioral and psychological symptoms of dementia (BPSD) in patients treated at a geriatric psychiatry ward in Germany over a time span of 1 year and (2) the use and effect of various pharmacologic interventions in managing BPSD in a real-world scenario.
Methods: This study was a naturalistic, retrospective analysis. Between May 2015 and May 2016, 437 patients aged 65 to 100 years with various psychiatric diseases (including dementia, schizophrenia, and depression) were admitted to a geriatric psychiatry unit in Germany. Their medical charts were reviewed to identify patients with dementia who presented with BPSD according to DSM-5 criteria. Since a decline in behavioral organization is a typical clinical manifestation of delirium, delirious patients with behavioral symptoms at admission were also included.
Results: Of 437 patients, 74 (16.9%) showed severe behavioral symptoms that were categorized into 6 groups: aggression, screaming, wandering, food refusal, self-harm, and a combination of both behavioral and psychological symptoms. Overall, 65 patients (87.5%) showed improvement in their behavior regardless of the pharmacotherapy they were receiving.
Conclusions: BPSD is one of the main reasons for admission to a geriatric psychiatry unit. Most of the patients with BPSD showed an improvement of behavioral symptoms with pharmacotherapy. Nonetheless, a clear correlation between a specific pharmacologic agent and behavioral improvement could not be established. A multimodal concept that involves both pharmacologic and nonpharmacologic approaches in managing BPSD should be the focus of future research.
Prevalence and Pharmacotherapy of Behavioral
and Psychological Symptoms of Dementia
in a Geriatric Psychiatry Unit:
A Retrospective Analysis
ABSTRACT
Objective: To observe (1) the proportion and nature of behavioral and psychological symptoms of dementia (BPSD) in patients treated at a geriatric psychiatry ward in Germany over a time span of 1 year and (2) the use and effect of various pharmacologic interventions in managing BPSD in a real-world scenario.
Methods: This study was a naturalistic, retrospective analysis. Between May 2015 and May 2016, 437 patients aged 65 to 100 years with various psychiatric diseases (including dementia, schizophrenia, and depression) were admitted to a geriatric psychiatry unit in Germany. Their medical charts were reviewed to identify patients with dementia who presented with BPSD according to DSM-5 criteria. Since a decline in behavioral organization is a typical clinical manifestation of delirium, delirious patients with behavioral symptoms at admission were also included.
Results: Of 437 patients, 74 (16.9%) showed severe behavioral symptoms that were categorized into 6 groups: aggression, screaming, wandering, food refusal, self-harm, and a combination of both behavioral and psychological symptoms. Overall, 65 patients (87.5%) showed improvement in their behavior regardless of the pharmacotherapy they were receiving.
Conclusions: BPSD is one of the main reasons for admission to a geriatric psychiatry unit. Most of the patients with BPSD showed an improvement of behavioral symptoms with pharmacotherapy. Nonetheless, a clear correlation between a specific pharmacologic agent and behavioral improvement could not be established. A multimodal concept that involves both pharmacologic and nonpharmacologic approaches in managing BPSD should be the focus of future research.
Prim Care Companion CNS Disord 2017;19(4):17m02137
https://doi.org/10.4088/PCC.17m02137
© Copyright 2017 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry and Psychotherapy, Charité—Campus Benjamin Franklin, Berlin, Germany
bDepartment of Psychiatry and Psychotherapy, Friedrich von Bodelschwingh-Klinik, Berlin, Germany
*Corresponding author: Arnim Quante, MD, Department of Psychiatry and Psychotherapy, Friedrich von Bodelschwingh-Klinik, Landhausstrasse 33-35, 10787 Berlin, Germany ([email protected]).
While Alzheimer’s disease and other forms of dementia are mainly characterized by cognitive impairment, the term behavioral and psychological symptoms of dementia (BPSD) encompasses a broad range of symptoms of behavior, thought, or mood including verbal and physical aggression, agitation, psychotic symptoms, and wandering.1,2 At least 1 of these noncognitive symptoms will manifest among nearly all patients with dementia during the course of the illness,3 or, as Steinberg et al4 maintain, over a 5-year period more than 90% of the patients develop at least 1 BPSD. BPSD has also been linked to poor functional prognosis, high mortality and morbidity, and rapid progression of the disease,5,6 further adding to the management difficulties that BPSD imposes on caregivers, nursing home residents, and the patients themselves. Multiple studies7–10 suggest that caregivers of BPSD patients are more prone to stress, depression, and generally poorer physical health compared to other caregivers, making BPSD a leading cause of premature institutionalization. While it is clear that early institutionalization of the patient can lead to a rapid improvement of the caregiver’s physical and psychological well-being, the impact it has on the patient remains questionable, especially considering that relocation to a nursing home has been related to immense stress and a decline in health.11,12 Other sources13 argue that there is a more frequent occurrence and a greater severity of BPSD among noninstitutionalized patients, thus maintaining that institutions provide a safer and more stable environment that helps moderate the symptoms.
However, with regard to therapy, it should be kept in mind that there is a lack of solid understanding of the complex etiology of BPSD, which strengthens the difficulties in approaching the patient. A nonpharmacologic approach offers a wide spectrum of supportive behavioral, social, environmental, and psychological interventions for both the patient and the caregivers or family. Examples of nonpharmacologic strategies (besides aromatherapy, light and music therapy, and acupuncture) include caregiver education, support and behavioral training, cognitive training and rehabilitation, reminiscence therapy, and validation therapy (tackling unresolved conflicts).14 Nevertheless, most of these strategies are barely practiced in clinical or standard care. We have found that physicians show limited familiarity and knowledge of these approaches, although different guidelines15,16 recommend the nonpharmacologic approach as first-line therapy and urge limiting antipsychotic medications to emergency cases or when other psychosocial interventions have proved ineffective. However, according to Kales et al,14 pharmacologic interventions are preferred over nonpharmacologic approaches due to vague guidelines and the extensive range of nonpharmacologic alternatives. Indeed, the heterogeneity and uncertainty about the effectiveness and methods of nonpharmacologic therapy deter providers from implementation. The lack of adequate human resources could also be a reason for not implementing these strategies.
Nevertheless, many patients do not respond to medication trials, responses are sometimes idiosyncratic, and the mortality risk is considerable.17 A good example would be the atypical antipsychotics (risperidone, quetiapine, and olanzapine). According to Maust et al,18 the effect of atypical antipsychotics on mortality risk in elderly patients with dementia is greater than previously thought. The study18 further shows that the mortality risk increases with the antipsychotic dosage.
The aim of this article is to study the proportion of behavioral and psychological symptoms in patients with dementia in a geriatric psychiatry unit in Germany and to further identify the types of symptoms. Additionally, we examine real-world pharmacologic interventions and evaluate their effectiveness in managing BPSD.
METHODS
This study was a naturalistic, retrospective analysis. Between May 2015 and May 2016, 437 patients aged 65–100 years with various psychiatric diseases (including dementia, schizophrenia, and depression) were admitted to a geriatric psychiatry unit in Germany. Their medical charts were reviewed to identify patients with dementia who presented with BPSD according to DSM-5 criteria. Since a decline in behavioral organization is a typical clinical manifestation of delirium as well, delirious patients with behavioral symptoms at admission were also included in the study. Mood symptoms, like depression or apathy, were not thoroughly observed since they are not considered to be an acute complication for the patient or the surroundings and therefore do not require intensive nursing care. Medical records of patients with severe BPSD were further reviewed to identify the most common symptoms and the pharmacologic interventions given. In most cases, pharmacotherapy was started after an observational phase of 1 or 2 days. If patients presented side effects, a dose decrease was attempted. Most of the patients continued medication throughout the hospital admission. According to guidelines,19 medication was started at very low dosages. Usually, medication was not stopped if BPSD resolved, but a reevaluation was recommended. A rescue medication (medication on demand) was provided for all patients. In nearly all cases, no changes of medications (except dose changes) were made until discharge. In cases when a medication was infective, a combination of 2 medications was provided.
This article evaluates only the effect of psychiatric medications used to improve behavioral organization: antidementives, antipsychotics, anticonvulsants, benzodiazepines, and antidepressants. We did not control the impact of nonpharmacologic therapies such as ergotherapy and music therapy, which are offered to all patients irrespective of the diagnosis.
All data were collected anonymously from the medical records only. Also, since this is a retrospective, naturalistic observation, an authorization by the ethics committee was not required, and informed consent was not necessary (as stated by the Ethics Committee of the Charité—University Medicine Berlin). The data were analyzed by descriptive statistical methods using means and standard deviations. Statistical analyses were performed with SPSS for Windows, version 21.
RESULTS
Demographics
Altogether, 74 of 437 inpatients (16.9%) had BPSD. The sample of 74 patients includes both male and female patients divided into 4 age categories as shown in Table 1. As expected, the number of female patients (n = 47) with BPSD was almost twice as high as that of male patients (n = 27). No major sex bias regarding behavioral disturbances was recognized. We detected no sex or age difference or an influence in the etiology of dementia—as far as a sufficient diagnosis could provide—in the response to medication. The mean duration of hospital stay was 18.3 days.
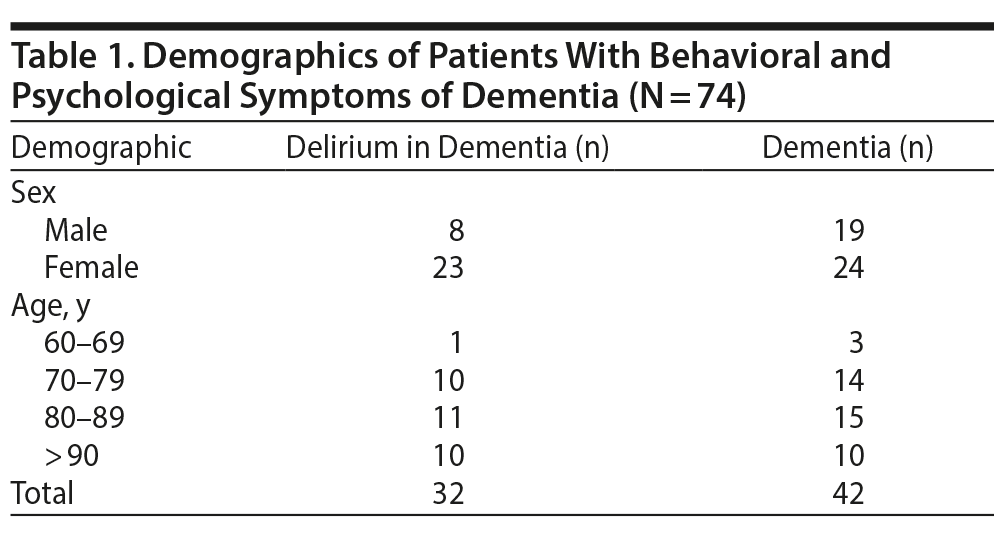
Within the sample, 2 main diagnoses were identified: dementia and delirium in dementia. They often share the same symptoms, making it difficult to differentiate between the 2 illnesses.
Proportion of Behavioral Symptoms
Five different isolated behavioral and psychological symptoms were identified: aggression, screaming, wandering, food refusal, and self-harm. However, 22.97% (n = 17) of the patients exhibited a combination of more than 1 behavioral disturbance. The most frequent symptom was aggression (which quite often was both verbal and physical) with 56.76% (n = 42), and the least common symptom was self-harm (patient beat his head on the wall) with only 1.35% (n = 1). Figure 1 shows the prevalence of these symptoms.
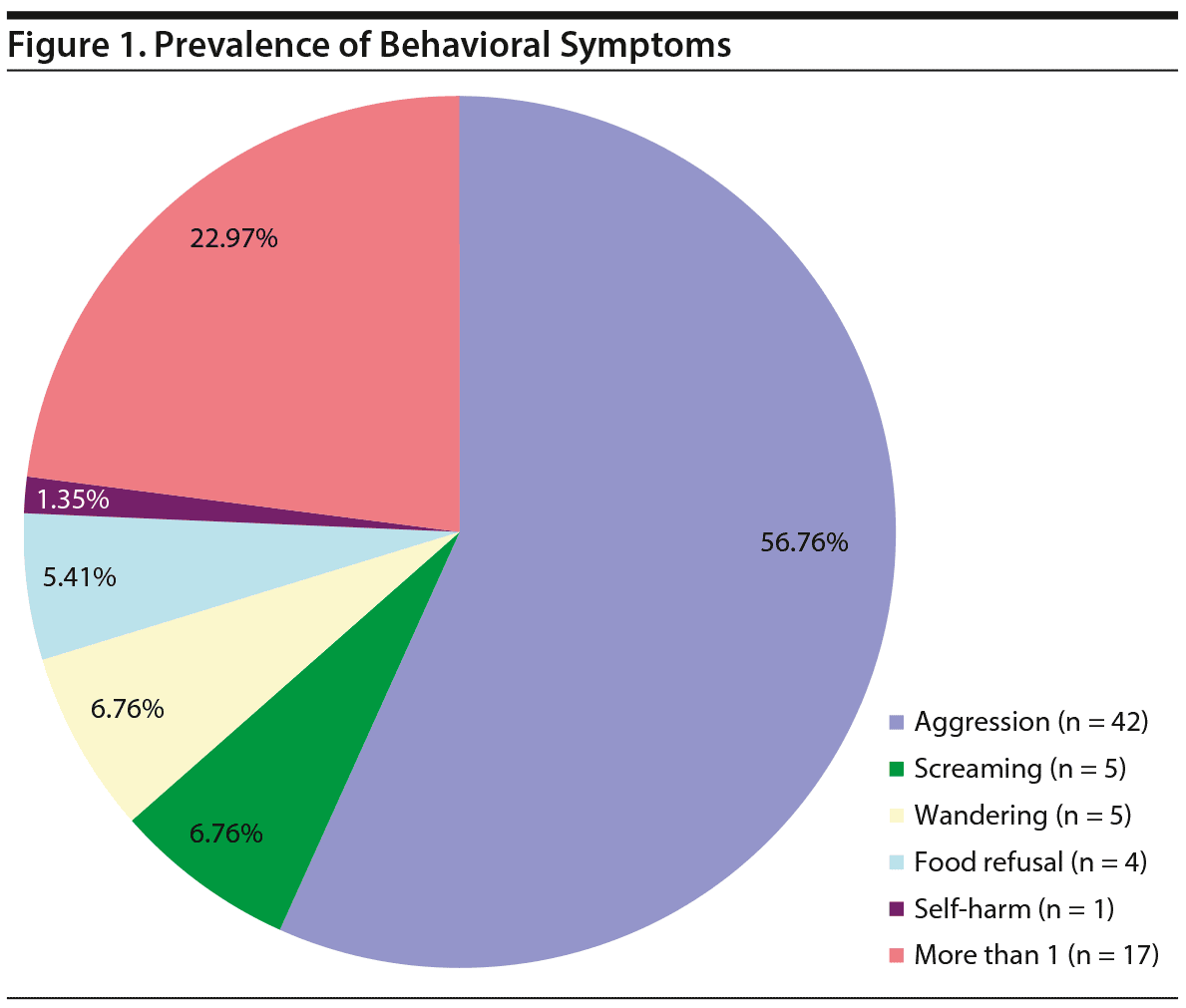
Patients who were prone to psychological symptoms, like hallucination, depressive episodes, or apathy, additionally manifest at least 1 of the 5 behavioral symptoms depicted in Figure 1. There was no case of isolated psychological or mood symptoms; hence, all are included in the “more than 1” category.
Pharmacologic Intervention
and Behavioral Improvement
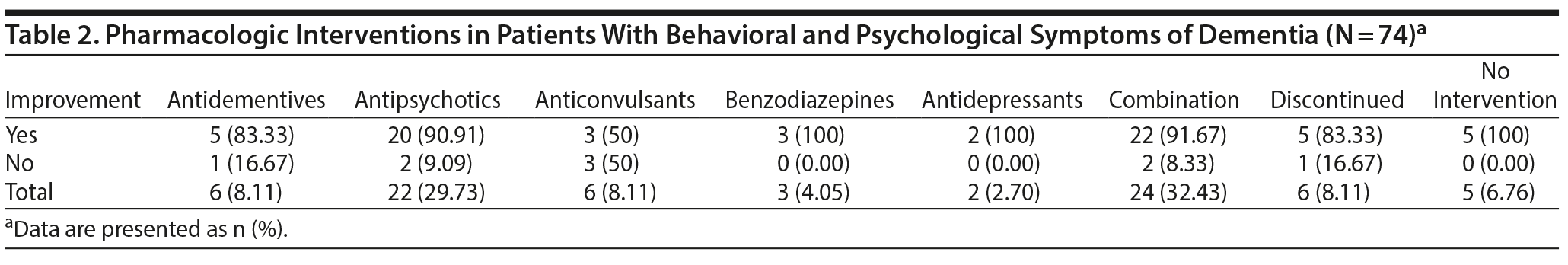
Of the patients who received pharmacotherapy, 32.43% were treated with more than 1 pharmacologic group of drugs. Most of the patients (29.73%) were treated with antipsychotics such as risperidone, quetiapine, haloperidol, or melperone. The least-administered drugs were the antidepressants (escitalopram, mirtazapine, doxepine, and venlafaxine) with 2.70% (2 patients).
No pharmacologic intervention was assigned for 5 of the patients (6.76%), and all of them showed improvement. In another group of 6 patients (8.11%), the prescribed psychiatric medications were discontinued, and only 1 of them did not experience recovery. Two patients experienced side effects due to initial medication; in these cases, the medication regimen was changed. Fortunately, the majority of patients (87.50%) showed improvement with or without a pharmacologic intervention.
Table 2 shows the symptomatic improvement in relation to the given pharmacotherapy and frequency of prescription of the drugs. All patients treated with benzodiazepines and antidepressants and all patients who received no pharmacologic intervention showed 100% improvement. A large percentage (90.91%) of patients who were given antipsychotics, the most-prescribed drug in the patient sample, also showed improvement in behavioral organization. The same applies to patients who were treated with a combination of drugs. As for the patients who received an anticonvulsive therapy, in 50% of them, the symptoms persisted.
DISCUSSION
A relatively high proportion of patients (16.9%) in a geriatric psychiatry unit had BPSD in a 1-year period. Aggression was the most frequent symptom, possibly because it is the most challenging, especially in nursing home units. As far as we know, the prevalence of BPSD in a geriatric psychiatry unit has not been examined, although the mean prevalence of behavioral and psychological symptoms in people with dementia is about 46%.20 In this naturalistic study, BPSD was one of the main reasons for admission to a geriatric psychiatry unit, as management of the symptoms and pharmacotherapy is challenging.
Of the 74 patients with BPSD, 31 were diagnosed with delirium in dementia. Delirium in dementia is associated with a poor prognosis in terms of a decline in cognitive function and must be identified and treated as soon as possible.16 Some typical causes of delirium in demented individuals would be, for example, an infection, dehydration, withdrawal, or even adverse effects of certain medications for treating comorbidities. However, because these causes of delirium could be identified, treating the symptoms by targeting these causes could be successful. Furthermore, delirium in dementia has been successfully treated with both typical and atypical antipsychotics; however, multiple studies21–23 have shown that atypical antipsychotics (risperidone, olanzapine, and quetiapine) can be more effective and cause less extrapyramidal side effects. Hallucinations, a very common symptom of delirium, can also be well managed with atypical antipsychotics.24
Concerning pharmacotherapy, our results indicate that the majority of patients (n = 65) showed improvement in behavior regardless of the pharmacotherapy administered, demonstrating the importance of medication in the treatment of BPSD. The most-prescribed drugs, antipsychotics and antidementives, correspond with the German S3 guidelines,16 which support the use of cholinesterase inhibitors in patients with mild to moderate Alzheimer’s disease and memantine in patients with moderate to severe Alzheimer’s disease (but not with frontotemportal dementia).
This study demonstrated no correlation between any specific off-label drug used to treat BPSD and symptomatic improvement, since the majority of the patients showed an improvement in their behavioral organization regardless of the medication they were administered. However, pharmacotherapy is associated with a higher mortality risk. In this study, all patients, in addition to receiving pharmacologic treatment, were also exposed to various nonpharmacologic therapies, a multimodal concept in managing behavior. Also, the importance of family members or caregivers cannot be overlooked. This multimodal concept and caregiver involvement should be the focus of future research. This study shows positive effects of combined therapies on behavioral symptoms in a real-world scenario, which indicates that acute treatment of BPSD is not impossible. Next, we discuss the findings of other studies regarding conventional and atypical antipsychotics, antidementives, and anticonvulsants in treating BPSD.
Antipsychotics
In this study, antipsychotics (mostly atypical) were the most-prescribed drugs for treatment of BPSD. Conventional antipsychotics were predominantly prescribed until the 1990s when atypical antipsychotics proved to be more effective and to generate fewer side effects. Despite a lack of sufficient scientific evidence confirming the efficiency of the atypical agents, multiple studies—including the data from our study—demonstrate that atypical antipsychotics can be beneficial for behavioral symptoms. For instance, a randomized, placebo-controlled trial25 showed that demented patients treated with low-dose risperidone had significant improvement in aggression and psychotic symptoms. Another double-blind, randomized, placebo-controlled trial26 that examined the effect of olanzapine in treating both behavioral and psychotic symptoms in dementia showed that a dose of olanzapine 5–10 mg daily is significantly more effective than placebo. Also, in a meta-analysis,27 quetiapine was found to be superior over placebo for treatment of BPSD, even when improvement was of small magnitude. Data for aripiprazole and clozapine are rare and inconclusive.28 In a small comparative study29 of aripiprazole versus risperidone in patients with BPSD, no significant difference could be detected. Both aripiprazole and risperidone were effective, but aripiprazole showed fewer side effects such as weight gain.29
However, according to Schneider and colleagues,30 the effect of atypical antipsychotics is inconsistent and not superior to placebo. The study30 also maintains that the side effects of the drug offset its benefits. Evidence31,32 shows that risperidone and olanzapine are strongly linked to extrapyramidal and cerebrovascular side effects, similar to the conventional agents, despite their efficacy in managing BPSD. Consequently, according to Ballard and Waite,32 neither risperidone nor olanzapine should be used routinely to treat BPSD and preferably should be limited to emergency cases or significant distress. Nevertheless, Lopez et al33 interestingly suggest that the high mortality rate among individuals treated with both conventional and atypical antipsychotics is a result of the clinical presence of psychosis and agitation—which also leads to a higher risk of premature institutionalization—and not due to the therapy itself.
Anticonvulsants
In a retrospective chart review34 of the treatment of aggression in dementia with gabapentin, the symptoms of the majority of the patients became effectively milder. On the other hand, according to a systematic review32 of articles published from 1966 to 2004, including the Cochrane Database of Systematic Reviews, 3 randomized controlled trials investigating valproate showed no efficacy in improving behavioral symptoms, and it was further associated with more adverse effects than placebo. An inconsistency in the results of our study was also observed, since only 50% of the patients treated with just anticonvulsants (valproic acid or gabapentin) showed improvement in their behavior. Carbamazepine was not prescribed to any of the patients, although it could be useful in reducing agitation and aggression.35
Antidementives
The most commonly prescribed antidementives were rivastigmine (cholinesterase inhibitors) and memantine. In their systematic meta-analysis, Maidment and colleagues36 maintain that memantine may be beneficial in managing BPSD; however, due to major limitations, the clinical effect of the drug is not clear or significant. Another systematic review35 shows that in 2 randomized controlled trials, the role of mementine in treating BPSD is conflicting. The same review,35 however, also asserts that in 2 meta-analyses and 6 randomized controlled trials cholinesterase inhibitors are mildly but statistically significantly effective. In our study, 5 of 6 patients treated with only antidementives (using rivastigmine as a transdermal patch) showed symptomatic improvement.
Antidepressants
In the Citalopram for Agitation in Alzheimer’s Disease Study (CitAD) study,37 citalopram 30 mg significantly reduced agitation compared to placebo. On the other hand, cognitive and cardiac adverse events were significantly higher in the citalopram group. In another small study,38 the antidepressants venlafaxine, sertraline, and desipramine were examined only in depressed patients with Alzheimer’s disease. In this study,38 sertraline was associated with superior effectiveness in relation to depressive, cognitive, and behavioral symptoms. Altogether, there is no strong evidence that warrants the use of antidepressants in BPSD. In cases of agitation, the use of citalopram can be useful, but adverse effects may limit its practical application. Despite the evidence37 for use of selective serotonin reuptake inhibitors in the treatment of agitation in patients with Alzheimer’s disease, antidepressants were not frequently used in our population. The reason for this is not totally clear. One reason might be insufficient experiences with antidepressants for rapid improvements of BPSD by the geriatric psychiatry unit physicians on one hand and possible side effects on the other.
LIMITATIONS
The patient sample of this study is small, while the variety of pharmacologic interventions is too broad. There was 100% improvement among patients who received monotherapy with antidepressants or benzodiazepines, or even those who were not treated pharmacologically at all, but the number of patients who received these therapies is too small (2, 3, and 5, respectively) to draw a significant connection between therapy and a symptomatic recovery. In other words, the small sample cannot be seen as representative of the general population.
More importantly, all patients included in this study additionally received nonpharmacologic treatment, predominantly ergotherapy and physiotherapy but also music therapy; however, their role in managing behavior was not considered in the study. Thus, the symptomatic improvement of the 65 patients could be accredited to the nonpharmacologic therapies they received, which weakens any possible link between a drug and behavioral recovery.
Most of the patients were discharged only when improved. Therefore, the positive outcome might be an artifact. Moreover, we did not control if the number of staff members had an influence on the outcome. We also did not control for medication initiated prior to admission.
Furthermore, examining behavior as a symptom can be conflicting, as it cannot be quantified or observed without bias. How do we confine the definition of aggression and its occurrences? Approximately 30 scales for measuring behavior have been developed, but the most functional and reliable are the Cohen-Mansfield Agitation Inventory15 and Neuropsychiatric Inventory Nursing Home Version scales.39,40 Of 74 patients, unfortunately, only 12 Mini-Mental State Examination41 results were documented in the clinical records. The Cohen-Mansfield Agitation Inventory or other useful scales for measuring behavior were not documented. In other words, the improvement in behavior was not objectively quantified but rather evaluated independently by the treating doctor. This factor, of course, leaves room for bias.
CONCLUSIONS
One of the main reasons for admission to a geriatric psychiatry unit is BPSD. Most of the patients with BPSD showed an improvement of behavioral symptoms with pharmacotherapy. Nonetheless, a clear correlation between a specific pharmacologic agent and a behavioral improvement could not be established. A multimodal concept that involves both pharmacologic and nonpharmacologic approaches in managing BPSD should be the focus of future research.
Submitted: March 14, 2017; accepted April 20, 2017.
Published online: July 27, 2017.
Potential conflicts of interest: Dr Quante has received speaker honoraria from Schwabe, Servier, and Janssen. Dr Sulejmani has no conflict of interest related to the subject of this article.
Funding/support: None.
REFERENCES
1. Finkel SI, Costa e Silva J, Cohen G, et al. Behavioral and psychological signs and symptoms of dementia: a consensus statement on current knowledge and implications for research and treatment. Int Psychogeriatr. 1996;8(suppl 3):497–500. PubMed doi:10.1017/S1041610297003943
2. Liperoti R, Pedone C, Corsonello A. Antipsychotics for the treatment of behavioral and psychological symptoms of dementia (BPSD). Curr Neuropharmacol. 2008;6(2):117–124. PubMed doi:10.2174/157015908784533860
3. Lyketsos CG, Carrillo MC, Ryan JM, et al. Neuropsychiatric symptoms in Alzheimer’s disease. Alzheimers Dement. 2011;7(5):532–539. PubMed doi:10.1016/j.jalz.2011.05.2410
4. Steinberg M, Shao H, Zandi P, et al; Cache County Investigators. Point and 5-year period prevalence of neuropsychiatric symptoms in dementia: the Cache County Study. Int J Geriatr Psychiatry. 2008;23(2):170–177. PubMed doi:10.1002/gps.1858
5. Levy ML, Cummings JL, Fairbanks LA, et al. Longitudinal assessment of symptoms of depression, agitation, and psychosis in 181 patients with Alzheimer’s disease. Am J Psychiatry. 1996;153(11):1438–1443. PubMed doi:10.1176/ajp.153.11.1438
6. Beeri MS, Werner P, Davidson M, et al. The cost of behavioral and psychological symptoms of dementia (BPSD) in community dwelling Alzheimer’s disease patients. Int J Geriatr Psychiatry. 2002;17(5):403–408. PubMed doi:10.1002/gps.490
7. Ballard CG, Gauthier S, Cummings JL, et al. Management of agitation and aggression associated with Alzheimer disease. Nat Rev Neurol. 2009;5(5):245–255. PubMed doi:10.1038/nrneurol.2009.39
8. de Vugt ME, Stevens F, Aalten P, et al. Do caregiver management strategies influence patient behaviour in dementia? Int J Geriatr Psychiatry. 2004;19(1):85–92. PubMed doi:10.1002/gps.1044
9. Cohen CA, Gold DP, Shulman KI, et al. Factors determining the decision to institutionalize dementing individuals: a prospective study. Gerontologist. 1993;33(6):714–720. PubMed doi:10.1093/geront/33.6.714
10. Hébert R, Dubois MF, Wolfson C, et al. Factors associated with long-term institutionalization of older people with dementia: data from the Canadian Study of Health and Aging. J Gerontol A Biol Sci Med Sci. 2001;56(11):M693–M699. PubMed doi:10.1093/gerona/56.11.M693
11. Grant I, Adler KA, Patterson TL, et al. Health consequences of Alzheimer’s caregiving transitions: effects of placement and bereavement. Psychosom Med. 2002;64(3):477–486. PubMed doi:10.1097/00006842-200205000-00012
12. Gaugler JE, Mittelman MS, Hepburn K, et al. Clinically significant changes in burden and depression among dementia caregivers following nursing home admission. BMC Med. 2010;8:85. PubMed doi:10.1186/1741-7015-8-85
13. Cheng TW, Chen TF, Yip PK, et al. Comparison of behavioral and psychological symptoms of Alzheimer’s disease among institution residents and memory clinic outpatients. Int Psychogeriatr. 2009;21(6):1134–1141. PubMed doi:10.1017/S1041610209990767
14. Kales HC, Gitlin LN, Lyketsos CG. Assessment and management of behavioral and psychological symptoms of dementia. BMJ. 2015;350:h369. PubMed doi:10.1136/bmj.h369
15. Cohen-Mansfield J, Juravel-Jaffe A, Cohen A, et al. Physicians’ practice and familiarity with treatment for agitation associated with dementia in Israeli nursing homes. Int Psychogeriatr. 2013;25(2):236–244. PubMed doi:10.1017/S104161021200172X
16. Deutsche Gesellschaft für Psychiatrie. Psychotherapie und Psychosomatik. S3 Leitlinie‘Demenzen’ website. https://www.dgppn.de/fileadmin/user_upload/_medien/download/pdf/kurzversion-leitlinien/REV_S3-leiltlinie-demenzen.pdf. Updated November, 2015. Accessed September 30, 2016.
17. Casey DA. Pharmacotherapy of neuropsychiatric symptoms of dementia. P&T. 2015;40(4):284–287. PubMed
18. Maust DT, Kim HM, Seyfried LS, et al. Antipsychotics, other psychotropics, and the risk of death in patients with dementia: number needed to harm. JAMA Psychiatry. 2015;72(5):438–445. PubMed doi:10.1001/jamapsychiatry.2014.3018
19. Reus VI, Fochtmann LJ, Eyler AE, et al. The American Psychiatric Association Practice Guideline on the Use of Antipsychotics to Treat Agitation or Psychosis in Patients With Dementia. Am J Psychiatry. 2016;173(5):543–546. doi:10.1176/appi.ajp.2015.173501
20. Devshi R, Shaw S, Elliott-King J, et al. Prevalence of behavioural and psychological symptoms of dementia in individuals with learning disabilities. Diagnostics (Basel). 2015;5(4):564–576. doi:10.3390/diagnostics5040564
21. Boettger S, Breitbart W. Atypical antipsychotics in the management of delirium: a review of the empirical literature. Palliat Support Care. 2005;3(3):227–237. PubMed doi:10.1017/S1478951505050352
22. van Iersel MB, Zuidema SU, Koopmans RT, et al. Antipsychotics for behavioural and psychological problems in elderly people with dementia: a systematic review of adverse events. Drugs Aging. 2005;22(10):845–858. doi:10.2165/00002512-200522100-00004
23. Yoon HJ, Park KM, Choi WJ, et al. Efficacy and safety of haloperidol versus atypical antipsychotic medications in the treatment of delirium. BMC Psychiatry. 2013;13:240. doi:10.1186/1471-244X-13-240
24. Johnsen E, Sinkeviciute I, Løberg E-M, et al. Hallucinations in acutely admitted patients with psychosis, and effectiveness of risperidone, olanzapine, quetiapine, and ziprasidone: a pragmatic, randomized study. BMC Psychiatry. 2013;13:241. PubMed doi:10.1186/1471-244X-13-241
25. Brodaty H, Ames D, Snowdon J, et al. A randomized placebo-controlled trial of risperidone for the treatment of aggression, agitation, and psychosis of dementia. J Clin Psychiatry. 2003;64(2):134–143. PubMed doi:10.4088/JCP.v64n0205
26. Street JS, Clark WS, Gannon KS, et al; The HGEU Study Group. Olanzapine treatment of psychotic and behavioral symptoms in patients with Alzheimer disease in nursing care facilities: a double-blind, randomized, placebo-controlled trial. Arch Gen Psychiatry. 2000;57(10):968–976. PubMed doi:10.1001/archpsyc.57.10.968
27. Cheung G, Stapelberg J. Quetiapine for the treatment of behavioural and psychological symptoms of dementia (BPSD): a meta-analysis of randomised placebo-controlled trials. N Z Med J. 2011;124(1336):39–50. PubMed
28. Tariot PN, Profenno LA, Ismail MS. Efficacy of atypical antipsychotics in elderly patients with dementia. J Clin Psychiatry. 2004;65(suppl 11):11–15. PubMed
29. Luo KY, Fu HB. Comparison of efficacy and safety of aripiprazole and risperidone in the treatment of behavioural and psychological symptoms of dementia. AMM. 2014;595:258–262. doi:10.4028/www.scientific.net/AMM.595.258
30. Schneider LS, Tariot PN, Dagerman KS, et al; CATIE-AD Study Group. Effectiveness of atypical antipsychotic drugs in patients with Alzheimer’s disease. N Engl J Med. 2006;355(15):1525–1538. PubMed doi:10.1056/NEJMoa061240
31. Herrmann N, Lanctôt KL. Atypical antipsychotics for neuropsychiatric symptoms of dementia: malignant or maligned? Drug Saf. 2006;29(10):833–843. doi:10.2165/00002018-200629100-00002
32. Ballard C, Waite J. The effectiveness of atypical antipsychotics for the treatment of aggression and psychosis in Alzheimer’s disease. Cochrane Database Syst Rev. 2006;(1):CD003476. PubMed
33. Lopez OL, Becker JT, Chang YF, et al. The long-term effects of conventional and atypical antipsychotics in patients with probable Alzheimer’s disease. Am J Psychiatry. 2013;170(9):1051–1058. PubMed doi:10.1176/appi.ajp.2013.12081046
34. Hawkins JW, Tinklenberg JR, Sheikh JI, et al. A retrospective chart review of gabapentin for the treatment of aggressive and agitated behavior in patients with dementias. Am J Geriatr Psychiatry. 2000;8(3):221–225. PubMed doi:10.1097/00019442-200008000-00007
35. Sink KM, Holden KF, Yaffe K. Pharmacological treatment of neuropsychiatric symptoms of dementia: a review of the evidence. JAMA. 2005;293(5):596–608. PubMed doi:10.1001/jama.293.5.596
36. Maidment ID, Fox CG, Boustani M, et al. Efficacy of memantine on behavioral and psychological symptoms related to dementia: a systematic meta-analysis. Ann Pharmacother. 2008;42(1):32–38. PubMed doi:10.1345/aph.1K372
37. Porsteinsson AP, Drye LT, Pollock BG, et al; CitAD Research Group. Effect of citalopram on agitation in Alzheimer disease: the CitAD randomized clinical trial. JAMA. 2014;311(7):682–691. PubMed doi:10.1001/jama.2014.93
38. Mokhber N, Abdollahian E, Soltanifar A, et al. Comparison of sertraline, venlafaxine and desipramine effects on depression, cognition and the daily living activities in Alzheimer patients. Pharmacopsychiatry. 2014;47(4–5):131–140. PubMed
39. Weiner MF, Koss E, Wild KV, et al. Measures of psychiatric symptoms in Alzheimer patients: a review. Alzheimer Dis Assoc Disord. 1996;10(1):20–30. PubMed
40. De Deyn PP, Wirshing WC. Scales to assess efficacy and safety of pharmacologic agents in the treatment of behavioral and psychological symptoms of dementia. J Clin Psychiatry. 2001;62(suppl 21):19–22. PubMed
41. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–198. doi:10.1016/0022-3956(75)90026-6
Enjoy this premium PDF as part of your membership benefits!


