Objective: To assess the clinical course of patients with methamphetamine-induced psychotic disorder (MIPD) and any possible predictors of the clinical course in a 3-month follow-up.
Methods: This prospective cohort study included 50 patients (7 female, 43 male) with MIPD and was performed from September 2014 to October 2015. Patients were assessed during hospitalization and in a follow-up visit 3 months later. Diagnoses were made using interviews based on the Structured Clinical Interview for DSM-IV Axis I Disorders. Positive, negative, manic, and depressive symptoms were the main outcome measures that were assessed using the Scale for the Assessment of Positive Symptoms, Scale for the Assessment of Negative Symptoms, Young Mania Rating Scale, and Hamilton Depression Rating Scale, respectively. Paired t test and regression analysis were used to analyze the data.
Results: Forty-six patients (92%) were reassessed at follow-up. More than half of the patients relapsed to methamphetamine use, did not adhere to treatment, and were functionally impaired. Positive, negative, and manic symptoms, but not depressive symptoms, improved in abstinent patients (P < .001, P = .001, P < .001, and P = .395, respectively). The best predictor of depressive and negative symptoms at follow-up was the patients’ respective baseline scores; but positive and manic symptoms were best predicted by reuse of methamphetamine during follow-up.
Conclusions: Various symptom categories do not always change in the same direction during the course of the disorder, especially depressive symptoms that do not improve with abstinence but aggravate with frequency of methamphetamine use. Negative symptoms at baseline also seem to have a possible role in prediction of methamphetamine reuse in patients with MIPD. Physicians should be advised to independently address all of the symptom categories of their patients with MIPD at each follow-up visit.
Clinical Course of Methamphetamine-Induced Psychotic Disorder in a 3-Month Follow-Up
Abstract
Objective: To assess the clinical course of patients with methamphetamine-induced psychotic disorder (MIPD) and any possible predictors of the clinical course in a 3-month follow-up.
Methods: This prospective cohort study included 50 patients (7 female, 43 male) with MIPD and was performed from September 2014 to October 2015. Patients were assessed during hospitalization and in a follow-up visit 3 months later. Diagnoses were made using interviews based on the Structured Clinical Interview for DSM-IV Axis I Disorders. Positive, negative, manic, and depressive symptoms were the main outcome measures that were assessed using the Scale for the Assessment of Positive Symptoms, Scale for the Assessment of Negative Symptoms, Young Mania Rating Scale, and Hamilton Depression Rating Scale, respectively. Paired t test and regression analysis were used to analyze the data.
Results: Forty-six patients (92%) were reassessed at follow-up. More than half of the patients relapsed to methamphetamine use, did not adhere to treatment, and were functionally impaired. Positive, negative, and manic symptoms, but not depressive symptoms, improved in abstinent patients (P < .001, P = .001, P < .001, and P = .395, respectively). The best predictor of depressive and negative symptoms at follow-up was the patients’ respective baseline scores; but positive and manic symptoms were best predicted by reuse of methamphetamine during follow-up.
Conclusions: Various symptom categories do not always change in the same direction during the course of the disorder, especially depressive symptoms that do not improve with abstinence but aggravate with frequency of methamphetamine use. Negative symptoms at baseline also seem to have a possible role in prediction of methamphetamine reuse in patients with MIPD. Physicians should be advised to independently address all of the symptom categories of their patients with MIPD at each follow-up visit.
Prim Care Companion CNS Disord 2016;18(6):doi:10.4088/PCC.16m02002
© Copyright 2016 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Iran University of Medical Sciences, Tehran, Iran
bMental Health Research Center, Tehran Institute of Psychiatry–School of Behavioral Sciences and Mental Health, Iran University of Medical Sciences, Tehran, Iran
*Corresponding author: Seyed Vahid Shariat, Mental Health Research Center, Tehran Institute of Psychiatry–School of Behavioral Sciences and Mental Health, Mansouri Lane, Niayesh Street, Sattarkhan Ave, Tehran, Iran ([email protected]).
Although overall drug use has remained stable globally during recent years, amphetamine use continues to increase in some regions of the world.1 Iran is one of the regions that has encountered an unexpectedly large increase in the number of users of amphetamines during the last decade and currently is facing an epidemic of methamphetamine use.2,3 The last national epidemiologic study4 performed on a representative sample of 7,886 Iranians reported a 1-year prevalence of 2.8% (95% CI, 2.3%–3.3%) for any substance and alcohol use disorder; stimulants ranked fourth after opiates, alcohol, and cannabis, respectively. However, the high number of morbidities associated with amphetamine use disorder, including psychotic disorder, mood disorders, sexual disorders, functional impairment, cognitive dysfunction, cerebrovascular disorders, and increased every-cause mortality,5–7 makes methamphetamine use one of the most prominent substance-related health issues in Iran.
Methamphetamine-induced psychotic disorder (MIPD) is an acute psychotic episode with a clinical presentation that resembles schizophrenia and generally needs urgent treatment.8 Sometimes, the psychotic episode is severe enough to mandate a visit to the emergency department and hospitalization.9 Therefore, psychiatric emergency departments and wards have been flooded by patients with MIPD in recent years, limiting access for other patients with mental disorders to inpatient services.10
Surprisingly, little evidence is available about the clinical course of patients with MIPD following discharge from the psychiatric hospital, including the rate of reuse and possible predictors of clinical outcome. Yeh et al11 followed 21 inpatients with MIPD for 6 months and reported that 8 of 17 interviewed patients admitted use of methamphetamine. Another study12 in Thailand evaluated the long-term outcome of patients with MIPD after their first hospitalization. They reinterviewed 449 patients about 6–7 years later and reported that more than half of them had experienced psychosis relapse and 39.2% had been rehospitalized. A study13 in Iran reported a 1-year follow-up of patients with MIPD and suggested that MIPD is a relatively stable diagnosis, and the 1-year course of these patients is more similar to affective than nonaffective psychoses.
The clinical course of MIPD needs to be further scrutinized in prospective studies, paying special attention to severity of both psychotic and mood symptoms. Clinical symptoms of the patients with MIPD are important not only in designing pharmacologic and nonpharmacologic treatment plans, but also theoretically might affect important course modifiers like compliance with treatment and reuse of methamphetamine.
Therefore, this study assessed the clinical course of a sample of patients with MIPD and possible predictors of the clinical course during a 3-month follow-up. We also assessed the rate of methamphetamine reuse in the follow-up period as a major determinant of clinical course.14
METHODS
Participants
This was a prospective cohort study performed from September 2014 to October 2015. The subjects were 50 patients with MIPD (DSM-IV criteria) hospitalized in Iran Psychiatric Hospital, Tehran, Iran. The patients were recruited via convenience sampling. Once or twice a week, a third-year psychiatry resident (S.J.) visited all of the patients with a diagnosis of MIPD who had been recently admitted to the hospital. She performed the interviews during hospitalization using the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I)15,16 to make the diagnosis and check for the inclusion and exclusion criteria. The SCID-I was given to all patients as a semi-structured interview using the specified questions in the manual.
The inclusion criteria were being diagnosed with MIPD by SCID-I interview and signing the informed consent. The exclusion criteria were unwillingness to participate in the study and presence of other brain disorders including tumors, structural brain disorders, and other psychotic disorders not related to methamphetamine use. The study was approved by the Iran University of Medical Sciences Ethics Committee.
Assessments
All of the selected patients answered a set of questions about demographic data (age, sex, marital status, job, education), methamphetamine abuse (age at first abuse, duration of abuse, lifetime history of abusing other substances including cannabis, opium, etc), and clinical history (the number of psychiatric hospitalizations, history of suicide attempt, history of legal problems). Recent use of methamphetamine was assessed for the last 3 months prior to admission on the basis of patients’ self-report. Frequency of use was rated on a 3-point Likert scale including never, occasional use (1 or 2 times in 3 months or 1 to 3 times a month), and frequent use (1 to 4 times a week or 5 to 7 times a week).
Clinical symptoms were also assessed using the following 4 scales: Scale for the Assessment of Negative Symptoms (SANS),17 Scale for the Assessment of Positive Symptoms (SAPS),18 Young Mania Rating Scale (YMRS),19 and Hamilton Depression Rating Scale (HDRS).20 The Family History Screen (FHS)21 was also used to elicit the family history of the subjects. The scales are described as follows:
SANS: A rating scale that is used to assess severity of negative symptoms. The SANS has 5 domains, and each domain is rated from 0 (absent) to 5 (severe); so, the total score could range from 0 to 25.
SAPS: A rating scale that is used to assess positive symptoms. This scale has 4 domains, and each domain is rated from 0 (absent) to 5 (severe); so, the total score could range from 0 to 20. Favorable reliability and validity has been reported in administration of this scale in an Iranian population.22
YMRS: An 11-item multiple-choice questionnaire for measuring the severity of mania, with total scores ranging from 0 to 60. Higher scores indicate greater severity of mania. The Persian version of the scale has acceptable reliability and validity23 and interrater reliability.24
HDRS: A questionnaire for rating the severity of depression. The 7-item version of the scale was used in this study. A study25 has shown that the 7-item version is as effective as the 17-item version. Six items are scored from 0 to 4 and 1 item from 0 to 2, with a possible total score of 0 to 26.
FHS: A brief screen for collecting information about family history of 15 psychiatric disorders and suicidal behavior in first-degree relatives of patients with mental disorders. The FHS has acceptable validity and reliability.26
Follow-Up Assessment
The patients were followed for 3 months after discharge from the hospital. The patients were invited to the hospital, and another face-to-face interview was performed. Items that were assessed in the follow-up visit included job status, marital status, relapse of methamphetamine and other substance use, clinical evaluation (SANS, SAPS, YMRS, and HDRS), medication use, readmission, attendance for outpatient visits, suicide attempt, and legal problems.
Statistical Analysis
Data analysis was performed with SPSS software version 16. Descriptive analysis and frequency of demographic variable were calculated. Comparisons between 2 measurements (admission and follow-up) were carried out using paired t tests. We also performed 4 multiple regression analyses to find the possible predictors of the 4 main outcome measures including the SANS, SAPS, HDRS, and YMRS. Baseline scores of each scale and age at first methamphetamine abuse, frequency of recent use of methamphetamine, reuse of methamphetamine during follow-up, number of previous admissions, and number of previous suicide attempts were included as possible predictors in each analysis. Furthermore, an ordinal regression analysis was performed to see if variables in the initial assessment could predict reuse of methamphetamine in the follow-up. A P value < .05 was considered significant for all of the tests.
RESULTS
A total of 50 patients (7 female, 43 male) with MIPD were included in this study. The mean ± SD age of the participants was 34.64 ± 8.18 years, and 41 (82%) were unemployed (Table 1). Only 9 patients (18%) were married. The mean ± SD age at first methamphetamine abuse was 28.78 ± 9.15 years. All of the patients were current users of methamphetamine and met the criteria for current dependence. Frequency of recent 3-month use of methamphetamine is provided in Table 2. All of the patients had a history of frequent use of other substances, with the most prevalent substances being nicotine: 50 (100%), opium: 37 (74%), and cannabis: 27 (54%). Also, 36 patients (72%) occasionally used alcohol as well, and 1 patient (2%) used hallucinogens (Table 2).
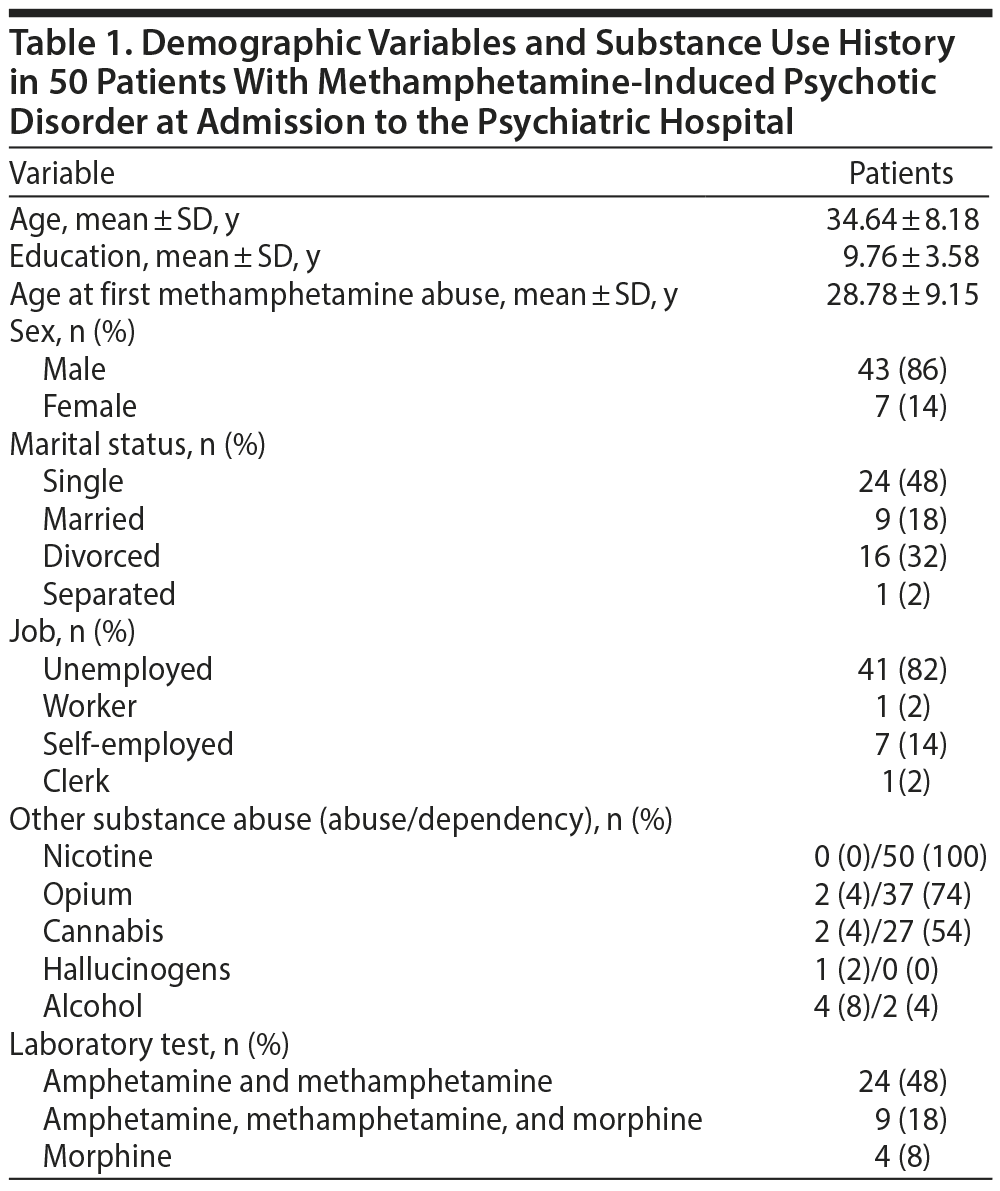
Eighteen patients (36%) had a positive family history of first-degree relatives with psychiatric disorders—a total of 28 relatives—which included opioid dependency in 10 (35.8%), amphetamine dependency in 8 (28.5%), bipolar disorder in 3 (10.7%), alcohol dependency in 2 (7.1%), major depressive disorder in 2 (7.1%), primary psychotic disorder in 1 (3.5%), obsessive-compulsive disorder in 1 (3.5%), and suicide attempt in 1 (3.5%). All of the patients received antipsychotic medications as well as adjunctive medications as needed. However, we did not register the medications in the study.
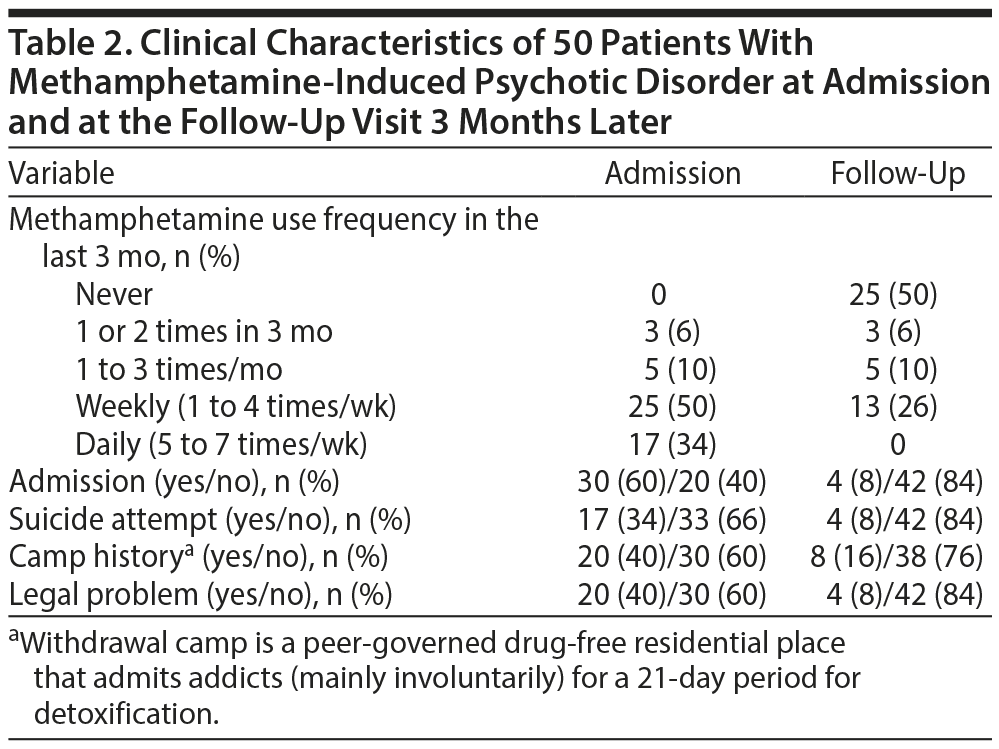
Three months later, 46 patients (92%) were reassessed, 4 patients (8%) were lost to follow-up, and 29 subjects (63%) were visited. Only 25 patients (50%) were regularly taking their medications in the follow-up, 2 patients (4%) irregularly used medications, and 19 patients (38%) had discontinued their medications. Twelve (24%) patients were working, and others were unemployed.
The clinical symptoms of the subjects were significantly different in the follow-up compared to the baseline assessment. The positive psychotic symptoms and manic symptoms decreased (SAPS mean difference = −4.04, P < .001; YMRS mean difference = −3.83, P <.001), but depressive symptoms increased (HDRS mean difference = 0.75, P = .01) 3 months after the baseline assessment. However, negative symptoms did not statistically change in the follow-up (SANS mean difference = −0.4, P = .1) (Figure 1).
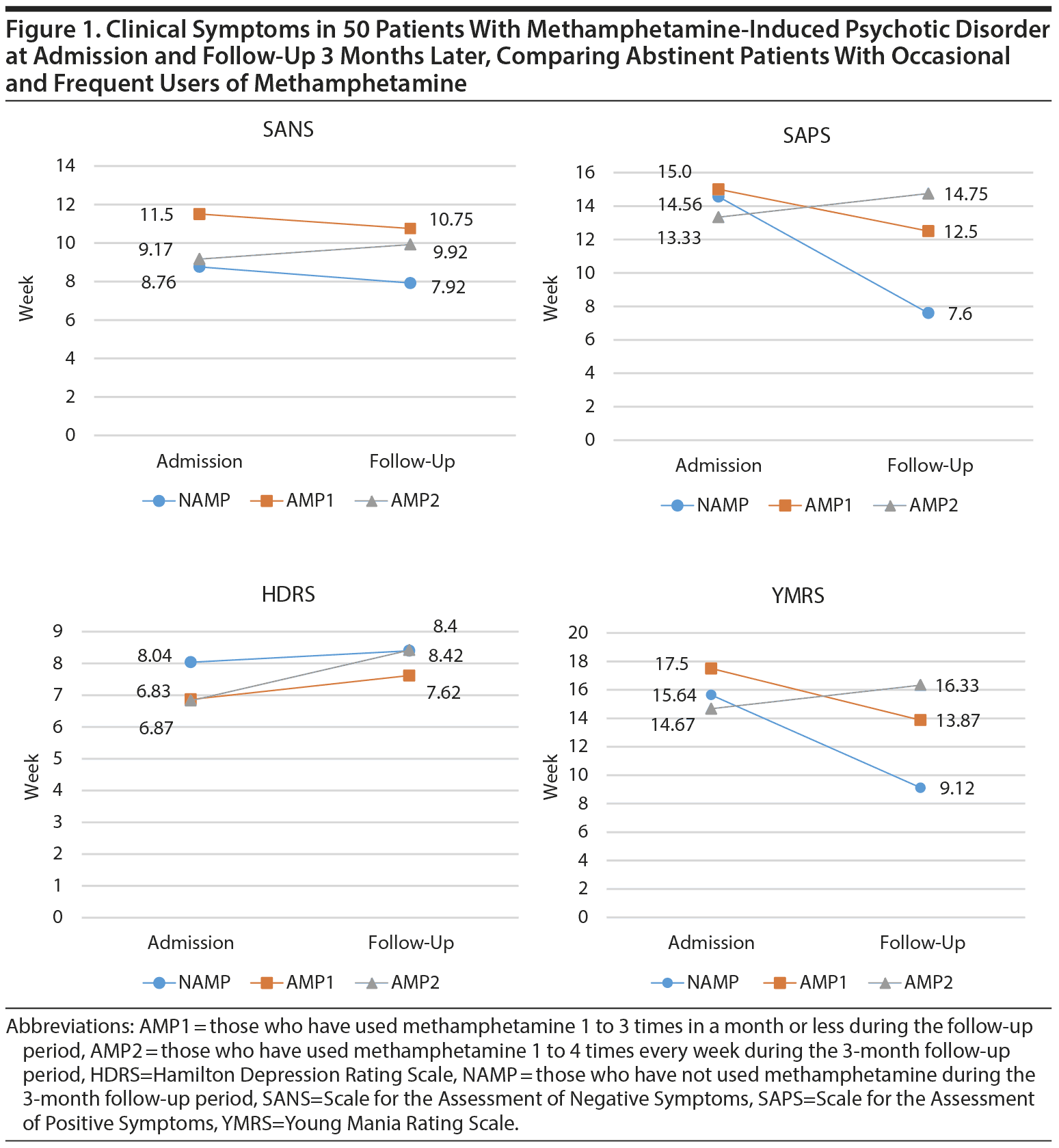
We also compared the clinical characteristics of the patients who had relapsed into occasional use of methamphetamine (1 to 3 times a month or less during 3 months of follow-up), patients who had relapsed into regular use of methamphetamine, or those who remained abstinent at the follow-up (Figure 1). The patients who had relapsed to methamphetamine use did not show improvement in positive, negative, and manic symptoms in contrast to those who were abstinent. Positive symptoms significantly increased in those with regular use of methamphetamine during follow-up, and although depressive symptoms did not improve in the abstinent patients, these symptoms worsened in those with regular use (abstinent patients: SANS, P = .001; SAPS, P < .001; HDRS, P = .395; YMRS, P < .001; occasional users: SANS, P = .22; SAPS, P < .153; HDRS, P = .08; YMRS, P < .194; frequent users: SANS, P = .24; SAPS, P < .37; HDRS, P = .009; YMRS, P < .299).
We also performed 4 regression analyses to find out which variables could potentially predict the 4 main outcome variables (ie, SANS, SAPS, HDRS, and YMRS) at follow-up. Baseline SANS score (B = 0.9, SE = 0.084, β = 0.81, P < .001) and reuse of methamphetamine during the follow-up period (B = 0.5, SE = 0.17, β = 0.23, P = .005) could predict the final SANS score. However, the only significant predictor of SAPS score in the follow-up was reuse of methamphetamine (B = 2.4, SE = 0.45, β = 0.68, P < .001). For the HDRS, again, baseline HDRS scores (B = 0.64, SE = 0.1, β = 0.76, P < .001) and reuse of methamphetamine (B = 0.38, SE = 0.16, β = 0.23, P = .046) were significant predictors of depression severity after 3 months of follow-up. Finally, only reuse of methamphetamine could significantly predict the YMRS score in the follow-up (B = 2.43, SE = 0.53, β = 0.6, P < .001).
Considering the importance of reuse of methamphetamine in the course of symptoms, as can be observed in Figure 1, we performed an ordinal regression analysis to see which of the clinical features or substance-related variables could reliably predict the relapse to methamphetamine use 3 months later. In this regression, we included relapse to methamphetamine use as the dependent variable (being abstinent, occasional use, regular use), frequency of recent methamphetamine use as factor (occasional use, regular use, daily use), and the SANS, SAPS, HDRS, and YMRS as covariates.
In this regression analysis, only the SANS score could significantly predict the outcome variable (relapse) (Wald χ2 = 5.58; P = .028; 95% CI, 0.034–0.369). Thus, having a higher score of negative symptoms at baseline was associated with an increased risk of relapse of methamphetamine use. Other variables (SAPS, HDRS, YMRS, and recent use frequency) could not reliably predict the relapse.
DISCUSSION
Few data exist regarding the natural course of patients with MIPD early after discharge from a psychiatric hospital. The current study showed that after 3 months of follow-up, about half of the patients with MIPD relapsed to methamphetamine use, and a sizable proportion of them were faced with difficult situations including legal problems, readmission to a psychiatric hospital or a withdrawal camp (a peer-governed drug-free residential place that admits addicts, mainly involuntarily, for a 21-day period for detoxification), unemployment, and suicide attempt.
Other studies12,27 have also shown that patients with MIPD encounter negative social, medical, and mental consequences during follow-up. For instance, 1 study12 followed 449 patients for about 6 to 7 years and showed more than half of the patients experienced a relapse of psychosis, and 39.2% of them needed rehospitalization. Furthermore, 22.3% showed some degree of suicidality at the time of interview, and 8.2% of the patients had died.12 In another study27 that followed 526 patients with methamphetamine dependence, those who had a history of psychotic disorder reported a higher rate of suicide attempts and a 2 times higher chance for hospitalization.
The current study also showed that the patients’ manic and positive, but not negative, symptoms significantly improved and depressive symptoms deteriorated in the follow-up. We also found that the clinical course of the patients was very much affected by whether they remained abstinent or relapsed into occasional or frequent use of methamphetamine. As it could be predicted, positive and negative symptoms as well as manic symptoms improved in abstinent but not in relapsed patients. Frequent users of methamphetamine even showed a significant increase of positive symptoms and nonsignificant increases in the scores of manic and negative symptoms. Other studies have also shown that psychotic and manic symptoms improve with abstinence, and there is a clear dose-response effect between days of methamphetamine use and severity of psychotic symptoms.28,29 A recent study by McKetin et al30 suggested that methamphetamine use is associated with exacerbation of positive psychotic and affective symptoms but not negative symptoms of schizophrenia in methamphetamine users. However, some other studies31,32 have reported negative symptoms in patients with MIPD as well. On the other hand, depressive symptoms showed a totally different course from other symptoms. Neither group showed an improvement, but the situation was worse in users and more severe in frequent users. Our results are in line with previous findings29,33 that have shown that depression increases after initiation of amphetamine use, and adverse psychological symptoms are associated with higher frequency of substance use. Moreover, the lack of change of depressive symptoms in the abstinent patients in our study could be referred to protracted withdrawal symptoms or real permanence or relapse of depressive symptoms. Although depressed mood, anhedonia, irritability, and lack of concentration have been reported during withdrawal of stimulants, these symptoms are generally mild and resolve in a few days.34,35 Therefore, the observed depressive symptoms do not seem to be related to withdrawal symptoms.
Moreover, presence of major depression has been reported in a subgroup of amphetamine users several months after withdrawal, which has been associated with a lower level of excretion of a norepinephrine metabolite, 3-methoxy-4-hydroxyphenylglycol (MHPG).36 Chronic methamphetamine use is also associated with changes in dopamine, glutamate, and γ-aminobutyric acid neurotransmission,37 all of which have been implicated in the neurobiology of depressive disorders.38 Furthermore, hyperactivity of the hypothalamic-pituitary-adrenal axis has been proposed as a possible mechanism of induction of depression in methamphetamine users.39
The current study also suggested that reuse of methamphetamine in the follow-up could significantly predict all of the assessed clinical symptoms including manic, depressive, and positive and negative psychotic symptoms. This prediction is much stronger for positive psychotic and manic symptoms but not so strong for depressive and negative psychotic symptoms, in which their respective baseline values are better predictors. This finding suggests that, although depressive and negative psychotic symptoms in patients with MIPD are affected to some degree by the reuse of methamphetamine, this effect is not so large. Severity of baseline levels of depression and negative symptoms seems to be the most important predictor of the severity of these symptoms in the follow-up.
As a very influential variable in the clinical outcome of patients with MIPD, we need to know the possible predictors of reuse of methamphetamine. Interestingly, the only baseline variable that could reliably predict relapse in the patients with MIPD to reuse of methamphetamine was the score of negative symptoms. This finding means that the higher a patient’s negative symptoms, the higher his or her risk of relapse to methamphetamine use. To our knowledge, this is the first time that negative symptoms have been shown to be related to risk of relapse of substance use. In a study on retention of methamphetamine users in treatment, Kobayashi et al40 showed that from among 4 potential predictors (age at initial assessment, presence of hallucinations, receiving public assistance, and history of incarceration), only history of incarceration could significantly predict treatment retention.
But why would negative symptoms increase the risk of methamphetamine reuse? We can use the extensive literature on schizophrenia and other psychotic disorders to find possible explanations. Ziedonis et al41 reported that using stimulants may improve negative symptoms and uncomfortable feelings that psychotic patients experience. However, Gregg et al42 suggested that still a consistent association could not be confirmed between schizophrenia and substance use. For example, 2 reviews have found contrasting results; 1 study43 reported that patients with schizophrenia prefer substances that would reduce their negative symptoms, but the other review44 showed that patients with comorbid schizophrenia and substance use had more positive than negative symptoms compared to noncomorbid patients. It is also important to note that the latter review44 did not separate the patients who used activating drugs (cocaine, amphetamines, and cannabis) from those who used sedatives (opiates and alcohol). This review also notes that the reason for the inconsistency in its findings with other studies might be due to selection of less socially impaired patients that could more easily find substances for their use. This article also mentioned that “if SUD [substance use disorder] is driven by self-medication, it may be aimed particularly toward alleviating negative symptoms.”44(p254) The same reasoning could be applicable to patients with MIPD and justify the observed association of negative symptoms with relapse of methamphetamine use 3 months later.
Limitations
The sample size of our study was relatively small and selected only from 1 psychiatric hospital in Tehran. Although this is a referral hospital that receives patients from all over Iran, our sample could not be representative of all of the patients with MIPD, especially those who do not need hospitalization and are treated in an outpatient setting. Furthermore, we only followed the patients for 3 months; longer follow-up might provide more information about the long-term outcome of these patients. The other limitation of the study was the lack of a control group. We also did not register the medications that the patients used during the study and could not control the possible effects that this could have on the results of the study. Comparison of the patients with MIPD with those with other psychotic disorders including schizophrenia could also be informative.
CONCLUSION
More than half of the patients relapsed into methamphetamine use, did not adhere to treatment, and were functionally impaired. Although abstinent patients improved in most of the clinical scales (positive, negative, and manic symptoms) 3 months later, depressive symptoms did not improve. On the other hand, patients who reused methamphetamine, especially frequent users of methamphetamine, continued to experience symptoms or even significantly deteriorated in the follow-up assessment.
Severity of negative symptoms at initial assessment was the only clinical variable that could predict reuse of methamphetamine 3 months later in the patients with MIPD. The current study underscores the importance of depressive and negative symptoms in patients with MIPD. Depressive symptoms indicated a different clinical course from other symptom categories. Negative symptoms also turned out to have a possible role in relapse prediction in the patients with MIPD. Therefore, physicians should be advised to independently address all of the symptom categories of patients with MIPD, including depressive and negative psychotic symptoms, at each follow-up visit.
Submitted: May 31, 2016; accepted August 17, 2016.
Published online: November 3, 2016.
Potential conflicts of interest: None.
Funding/support: None.
Previous presentation: None.
Acknowledgments: The authors thank the staff of Iran Psychiatric Hospital for their help in managing patient visits and the patients who participated in the study. The study was performed as the thesis of Dr Javadian.
REFERENCES
1. World Drug Report 2015, United Nations publication, Sales No. E.15.XI.6. United Nations Office on Drugs and Crime Web site. https://www.unodc.org/documents/wdr2015/World_Drug_Report_2015.pdf. Accessed August 30, 2016.
2. Alam Mehrjerdi Z. Crystal in Iran: methamphetamine or heroin kerack. Daru. 2013;21(1):22. PubMed doi:10.1186/2008-2231-21-22
3. Shariat SV, Elahi A. Symptoms and course of psychosis after methamphetamine abuse: one-year follow-up of a case. Prim Care Companion J Clin Psychiatry. 2010;12(5):e1–e2. PubMed
4. Sharifi V, Amin-Esmaeili M, Hajebi A, et al. Twelve-month prevalence and correlates of psychiatric disorders in Iran: the Iranian Mental Health Survey, 2011. Arch Iran Med. 2015;18(2):76–84. PubMed
5. Zorick TS, Rad D, Rim C, et al. An overview of methamphetamine induced psychotic syndromes. Addict Disord Their Treat. 2008;7:143–156. doi:10.1097/ADT.0b013e318066d5e0
6. Scott JC, Woods SP, Matt GE, et al. Neurocognitive effects of methamphetamine: a critical review and meta-analysis. Neuropsychol Rev. 2007;17(3):275–297. PubMed doi:10.1007/s11065-007-9031-0
7. Cruickshank CC, Dyer KR. A review of the clinical pharmacology of methamphetamine. Addiction. 2009;104(7):1085–1099. PubMed doi:10.1111/j.1360-0443.2009.02564.x
8. Medhus S, Rognli EB, Gossop M, et al. Amphetamine-induced psychosis: transition to schizophrenia and mortality in a small prospective sample. Am J Addict. 2015;24(7):586–589. PubMed doi:10.1111/ajad.12274
9. Toles M, Jiang C, Goebert D, et al. Methamphetamine in emergency psychiatry. Addict Disord Their Treat. 2006;5(4):191–199. doi:10.1097/01.adt.0000210076.53993.05
10. Fasihpour B, Molavi S, Shariat SV. Clinical features of inpatients with methamphetamine-induced psychosis. J Ment Health. 2013;22(4):341–349. PubMed doi:10.3109/09638237.2012.745184
11. Yeh HS, Lee YC, Sun HJ, et al. Six months follow-up of patients with methamphetamine psychosis. Zhonghua Yi Xue Za Zhi (Taipei). 2001;64(7):388–394. PubMed
12. Kittirattanapaiboon P, Mahatnirunkul S, Booncharoen H, et al. Long-term outcomes in methamphetamine psychosis patients after first hospitalisation. Drug Alcohol Rev. 2010;29(4):456–461. PubMed doi:10.1111/j.1465-3362.2010.00196.x
13. Hajebi A, Shrifi V, Amini H, et al. Course and Outcome of Crystal Methamphetamine Induced Psychotic Disorder Compared With Other Primary Psychotic Disorders (Affective and Non-Affective) in Roozbeh and Iran Psychiatric Hospitals in a One Year Follow Up, 2013. Tehran, Iran; Tehran University of Medical Sciences, Mental Health Research Center; 2013.
14. Glasner-Edwards S, Mooney LJ. Methamphetamine psychosis: epidemiology and management. CNS Drugs. 2014;28(12):1115–1126. PubMed doi:10.1007/s40263-014-0209-8
15. First MB, et al. Structured Clinical Interview for DSM-IV Axis I Disorders, Clinician Version (SCID-CV). Washington, DC: American Psychiatric Press, Inc; 1996.
16. Sharifi V, Assadi SM, Mohammadi MR, et al. A Persian translation of the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition: psychometric properties. Compr Psychiatry. 2009;50(1):86–91. PubMed doi:10.1016/j.comppsych.2008.04.004
17. Andreasen N. Scale for the Assessment of Negative Symptoms (SANS). Iowa City: University of Iowa; 1983.
18. Andreasen N. Scale for the Assessment of Positive Symptoms (SAPS). Iowa City: University of Iowa; 1984.
19. Young RC, Biggs JT, Ziegler VE, et al. A rating scale for mania: reliability, validity and sensitivity. Br J Psychiatry. 1978;133:429–435. PubMed doi:10.1192/bjp.133.5.429
20. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56–62. PubMed doi:10.1136/jnnp.23.1.56
21. Weissman MM, Wickramaratne P, Adams P, et al. Brief screening for family psychiatric history: the Family History Screen. Arch Gen Psychiatry. 2000;57(7):675–682. PubMed doi:10.1001/archpsyc.57.7.675
22. Alaghband-Rad J, Boroumand M, Amini H, et al. Non-affective acute remitting psychosis: a preliminary report from Iran. Acta Psychiatr Scand. 2006;113(2):96–101. PubMed doi:10.1111/j.1600-0447.2005.00658.x
23. Barekatain M, et al. Standardization, reliability and validity of the Young Mania Rating Scale. Psychology. 2007;11(2):150–166.
24. Shabani A, Taheri A, Azadforouz S, et al. Bipolar Disorder Patients Follow-up (BDPF): methods and materials. J Res Med Sci. 2010;15(4):229–234. PubMed
25. McIntyre R, Kennedy S, Bagby RM, et al. Assessing full remission. J Psychiatry Neurosci. 2002;27(4):235–239. PubMed
26. Milne BJ, Caspi A, Crump R, et al. The validity of the Family History Screen for assessing family history of mental disorders. Am J Med Genet B Neuropsychiatr Genet. 2009;150B(1):41–49. PubMed doi:10.1002/ajmg.b.30764
27. Glasner-Edwards S, Mooney LJ, Marinelli-Casey P, et al; Methamphetamine Treatment Project Corporate Authors. Clinical course and outcomes of methamphetamine-dependent adults with psychosis. J Subst Abuse Treat. 2008;35(4):445–450. PubMed doi:10.1016/j.jsat.2007.12.004
28. McKetin R, Lubman DI, Baker AL, et al. Dose-related psychotic symptoms in chronic methamphetamine users: evidence from a prospective longitudinal study. JAMA Psychiatry. 2013;70(3):319–324. PubMed doi:10.1001/jamapsychiatry.2013.283
29. Hall W, Hando J, Darke S, et al. Psychological morbidity and route of administration among amphetamine users in Sydney, Australia. Addiction. 1996;91(1):81–87. PubMed doi:10.1111/j.1360-0443.1996.tb03163.x
30. McKetin R, Dawe S, Burns RA, et al. The profile of psychiatric symptoms exacerbated by methamphetamine use. Drug Alcohol Depend. 2016;161:104–109. PubMed doi:10.1016/j.drugalcdep.2016.01.018
31. Akiyama K. Longitudinal clinical course following pharmacological treatment of methamphetamine psychosis which persists after long-term abstinence. Ann N Y Acad Sci. 2006;1074:125–134. PubMed doi:10.1196/annals.1369.012
32. Srisurapanont M, Ali R, Marsden J, et al. Psychotic symptoms in methamphetamine psychotic in-patients. Int J Neuropsychopharmacol. 2003;6(4):347–352. PubMed doi:10.1017/S1461145703003675
33. Glasner-Edwards S, Marinelli-Casey P, Hillhouse M, et al; Methamphetamine Treatment Project Corporate Authors. Depression among methamphetamine users: association with outcomes from the Methamphetamine Treatment Project at 3-year follow-up. J Nerv Ment Dis. 2009;197(4):225–231. PubMed doi:10.1097/NMD.0b013e31819db6fe
34. Newton TF, Kalechstein AD, Duran S, et al. Methamphetamine abstinence syndrome: preliminary findings. Am J Addict. 2004;13(3):248–255. PubMed doi:10.1080/10550490490459915
35. Cantwell B, McBride AJ. Self detoxication by amphetamine dependent patients: a pilot study. Drug Alcohol Depend. 1998;49(2):157–163. PubMed doi:10.1016/S0376-8716(97)00160-9
36. Watson R, Hartmann E, Schildkraut JJ. Amphetamine withdrawal: affective state, sleep patterns, and MHPG excretion. Am J Psychiatry. 1972;129(3):263–269. PubMed doi:10.1176/ajp.129.3.263
37. Hsieh JH, Stein DJ, Howells FM. The neurobiology of methamphetamine induced psychosis. Front Hum Neurosci. 2014;8:537. PubMed doi:10.3389/fnhum.2014.00537
38. Kalia M. Neurobiological basis of depression: an update. Metabolism. 2005;54(suppl 1):24–27. PubMed doi:10.1016/j.metabol.2005.01.009
39. Zuloaga DG, Jacobskind JS, Raber J. Methamphetamine and the hypothalamic-pituitary-adrenal axis. Front Neurosci. 2015;9:178. PubMed doi:10.3389/fnins.2015.00178
40. Kobayashi O, Matsumoto T, Otsuki M, et al. Profiles associated with treatment retention in Japanese patients with methamphetamine use disorder: preliminary survey. Psychiatry Clin Neurosci. 2008;62(5):526–532. PubMed doi:10.1111/j.1440-1819.2008.01845.x
41. Ziedonis D, Bizamcer AN, Steinberg ML, et al. Co-occuring addiction and psychotic disorders. In: Graham AW, Schultz TK, Mayo-Smith MF, et al, eds. Principles of Addiction Medicine. 3rd ed. Philadelphia, PA: Lippincot Williams & Wilkins; 2003:1297–1320.
42. Gregg L, Barrowclough C, Haddock G. Reasons for increased substance use in psychosis. Clin Psychol Rev. 2007;27(4):494–510. PubMed doi:10.1016/j.cpr.2006.09.004
43. Schneier FR, Siris SG. A review of psychoactive substance use and abuse in schizophrenia: patterns of drug choice. J Nerv Ment Dis. 1987;175(11):641–652. PubMed doi:10.1097/00005053-198711000-00001
44. Talamo A, Centorrino F, Tondo L, et al. Comorbid substance-use in schizophrenia: relation to positive and negative symptoms. Schizophr Res. 2006;86(1–3):251–255. PubMed doi:10.1016/j.schres.2006.04.004
Enjoy this premium PDF as part of your membership benefits!


