Background: Peripheral arterial compliance (PAC) is a measure of the ability of the vascular tree to dilate in response to a pressure wave. Reduced PAC is seen in patients with psychiatric diagnoses and has been associated with increased risk for stroke, myocardial infarction, and mortality. The objective of this pilot study was to identify predictors of reduced PAC in subjects with psychiatric diagnoses.
Methods: Male psychiatric subjects (N = 77) were studied in a cross-sectional study of medication effects on PAC conducted from August 2005 to February 2010. Calf and thigh compliance were modeled in separate linear regressions. The models were adjusted for age, race, smoking status, presence or absence of the metabolic syndrome, current treatment with a statin, diagnosis of schizophrenia or schizoaffective disorder, current antipsychotic treatment, and body mass index (BMI).
Results: Of the 77 subjects (mean ± SD age of 53.7 ± 8.8 years), 41 were white, 36 were black, and 27 were diagnosed with schizophrenia or schizoaffective disorder (DSM-IV criteria). Fifty participants were being treated with an antipsychotic medication, while the remaining 27 were off of antipsychotics for at least 2 months. Our model explained 27% of the variance in calf compliance. Black subjects had reduced calf compliance compared to white subjects (P = .02). Having metabolic syndrome was associated with reduced PAC at a trend level (P < .08), and BMI (P = .004) and BMI2 (P = .011) were significant predictors of calf compliance. Schizophrenia versus other psychiatric diagnoses and antipsychotic treatment were not significantly associated with calf compliance.
Conclusions: In this pilot study, significant predictors of calf compliance were race (black vs white) and BMI. PAC is a noninvasive measure that may be a predictor of cardiovascular risk in psychiatric patients. The reduced PAC seen in patients with psychiatric diagnoses does not appear to be directly related to their diagnosis or antipsychotic treatment but rather to other characteristics inherent to the subject. Future studies are warranted to better understand the pathophysiology of PAC including but not limited to inflammation in psychiatric patients.
Predictors of Calf Arterial Compliance in
Male Veterans With Psychiatric Diagnoses
ABSTRACT
Background: Peripheral arterial compliance (PAC) is a measure of the ability of the vascular tree to dilate in response to a pressure wave. Reduced PAC is seen in patients with psychiatric diagnoses and has been associated with increased risk for stroke, myocardial infarction, and mortality. The objective of this pilot study was to identify predictors of reduced PAC in subjects with psychiatric diagnoses.
Methods: Male psychiatric subjects (N = 77) were studied in a cross-sectional study of medication effects on PAC conducted from August 2005 to February 2010. Calf and thigh compliance were modeled in separate linear regressions. The models were adjusted for age, race, smoking status, presence or absence of the metabolic syndrome, current treatment with a statin, diagnosis of schizophrenia or schizoaffective disorder, current antipsychotic treatment, and body mass index (BMI).
Results: Of the 77 subjects (mean ± SD age of 53.7 ± 8.8 years), 41 were white, 36 were black, and 27 were diagnosed with schizophrenia or schizoaffective disorder (DSM-IV criteria). Fifty participants were being treated with an antipsychotic medication, while the remaining 27 were off of antipsychotics for at least 2 months. Our model explained 27% of the variance in calf compliance. Black subjects had reduced calf compliance compared to white subjects (P = .02). Having metabolic syndrome was associated with reduced PAC at a trend level (P < .08), and BMI (P = .004) and BMI2 (P = .011) were significant predictors of calf compliance. Schizophrenia versus other psychiatric diagnoses and antipsychotic treatment were not significantly associated with calf compliance.
Conclusions: In this pilot study, significant predictors of calf compliance were race (black vs white) and BMI. PAC is a noninvasive measure that may be a predictor of cardiovascular risk in psychiatric patients. The reduced PAC seen in patients with psychiatric diagnoses does not appear to be directly related to their diagnosis or antipsychotic treatment but rather to other characteristics inherent to the subject. Future studies are warranted to better understand the pathophysiology of PAC including but not limited to inflammation in psychiatric patients.
Prim Care Companion CNS Disord 2016;18(3):doi:10.4088/PCC.15m01880
© Copyright 2016 Physicians Postgraduate Press, Inc.
aClinical Research Program, Sheppard Pratt Health System, Baltimore, Maryland, and Department of Psychiatry and Behavioral Sciences, George Washington University School of Medicine and Health Sciences, Washington, DC
bBaltimore Veterans Affairs Medical Center Geriatric Research, Education, and Clinical Center, Division of Gerontology and Geriatric Medicine, University of Maryland School of Medicine, Baltimore
cMental Health Service, Atlanta Veterans Affairs Medical Center, Decatur, Georgia
dDepartment of Medicine, Emory University School of Medicine, Atlanta, Georgia
eDepartment of Psychiatry and Behavioral Sciences, Emory University School of Medicine, Atlanta, Georgia
fDepartment of Internal Medicine, Mount Sinai Beth Israel Hospital, New York, New York
gUniversity of Miami School of Medicine, Miami, Florida
*Corresponding author: Maju Mathew Koola, MD, Clinical Research Program, Sheppard Pratt Health System, 6501 N Charles St, Baltimore, MD 21204
([email protected]).
Peripheral arterial compliance (PAC) is defined as the change in vascular volume per unit change in pressure (ΔV/ΔP) and provides a measure of the vascular tree’s ability to respond to the pressure wave produced during the cardiac pumping cycle (ie, stroke volume). Reduced PAC has important clinical implications, since it is associated with atherosclerosis, stroke, and myocardial infarction.1,2 Reduced PAC is also associated with coronary and aortic lesions in asymptomatic patients as measured by angiography and magnetic resonance imaging3 and aortic atherosclerosis at necropsy.4 Reduced PAC in the thigh and calf has been associated with the presence and extent of coronary artery disease in 346 subjects undergoing coronary angiography.2 A study3 of 376 patients found similar correlations with volume of lesions in the abdominal aorta after accounting for other cardiovascular risk factors such as age, sex, diabetes mellitus, smoking, hyperlipidemia, hypertension, and obesity. Furthermore, reduced PAC is a predictor of future cardiovascular events.3 There was a significant negative correlation between PAC and the Framingham risk factor score in 122 subjects with peripheral arterial disease.5 In another study,6 blacks (N = 1,887, mean ± SD age = 59 ± 6 years, 65% women) were followed for 13 ± 4 years. Of those with reduced arterial compliance, 12% developed coronary disease, 16% developed heart failure, and 9% experienced a stroke.6 Reduced arterial compliance is also a robust predictor of mortality.7
In our previously published report of this cohort,8 subjects with psychiatric diagnoses and those on antipsychotics had reduced arterial compliance compared to healthy controls with no psychiatric diagnoses. It is known that people with psychiatric diagnoses have reduced life expectancy independent of suicide.9–12 Cardiovascular deaths are one of the most common causes of mortality in people with schizophrenia.9,11 In the same vein, those diagnosed with schizophrenia have a mean life expectancy of 51–61 years, which is about 20–25 years shorter than the general population.9–13
PAC can be assessed using the Vasogram,2,14 a device that provides a noninvasive, inexpensive, automated measurement requiring minimal operator training. Testing can be completed in 15 minutes in an office setting.15 Reduced PAC, measured with the Vasogram, has been shown to be associated with vascular disease.3 Precision studies with the Vasogram have reported high reproducibility between machines and between visits.3
Thus, arterial compliance is useful in monitoring the course of cardiovascular disease and may also be a useful way to track cardiovascular risk in psychiatric patients treated with antipsychotic medications. The objective of this pilot study was to identify factors associated with reduced PAC in subjects with psychiatric diagnoses with the ultimate aim of identifying interventions that can address the decreased life expectancy in patients diagnosed with psychiatric disease.
METHODS
Participants
Eighty-eight veterans, aged 18–70 years and receiving care for psychiatric diagnoses at the outpatient clinics of Atlanta Veterans Affairs Medical Center (Decatur, Georgia), were recruited from August 2005 through February 2010. The original study8 was designed to compare PAC in male subjects with psychiatric diagnoses (N = 63) to healthy male controls with no psychiatric diagnoses. In this secondary analysis, to examine potential predictors of thigh and calf compliance, we analyzed data from 77 subjects out of the original 88 (6 women and 5 men with missing metabolic syndrome values were excluded). All subjects were men, aged 18–70 years, who had at least 1 DSM-IV Axis I psychiatric diagnosis conferred by an outpatient psychiatrist. Exclusion criteria included weight > 300 lb (the Vasogram blood pressure cuffs were not large enough), diabetes mellitus type 1 or 2, triglycerides > 600 mg/dL, treatment with clozapine, myocardial infarction or unstable angina within the past 6 months, human immunodeficiency virus or acquired immunodeficiency syndrome, collagen vascular disease, or active DSM-IV drug or alcohol abuse or dependence within the past 3 months (use of caffeine or tobacco was allowed). We wanted to study normal physiology as much as possible; hence, diagnoses and variables that were known to affect PAC significantly were excluded.
As detailed in Table 1, the eligible subjects were divided into 1 of 3 groups utilizing criteria used in our prior study8 with the same group of subjects. Groups 1 and 2 were taking an antipsychotic medication continuously for at least 3 months (Table 1). Group 1 (n = 10) comprised subjects taking antipsychotics with lower risk for weight gain (typical antipsychotics were included in this group because of small sample size) and group 2 (n = 40) comprised subjects taking antipsychotics with higher risk for weight gain.16 Subjects in group 3 (n = 27) had received no antipsychotic (nonadherent) for at least 2 months prior to participation. Subjects signed informed consent and the Health Insurance Portability and Accountability Act (HIPAA) forms approved by the Institutional Review Board at Emory University and the Atlanta Veterans Affairs Research and Development Committee.
Study Design
In this cross-sectional study, sociodemographic variables and clinical data were collected from interviews, assessments with the subjects, and their electronic medical records. Laboratory measures were obtained from the electronic medical records. Blood pressure, height, and weight were measured at the time of Vasogram measurement.
Instrument to Measure PAC
PAC was measured at the thigh and calf with an automated, computer-controlled air plethysmograph (Vasogram, Vasocor, Inc, Charleston, South Carolina; now Soterogram, Soteria Medical, LLC, Miami, Florida). PAC measurement (both thigh and calf) took approximately 15 minutes to complete.
Determination of PAC
To calculate local arterial compliance, a representative measurement of systemic blood pressure must be obtained. The required blood pressure measurement is pulse pressure, the difference between systolic blood pressure and diastolic blood pressure. For the determination of pulse pressure, we always used the right upper arm, which most accurately represents the systemic blood pressure. The femoropopliteal arterial wall in the thigh and calf contains a distribution of elastin, collagen, and smooth muscle similar to that of the coronary arteries.17,18 Thus, thigh and calf measurements were selected as the volume component of the local arterial compliance calculation. The measurements were taken at the brachial, thigh, and calf levels with the subject in the supine position. The supine position is required to eliminate pressure variations secondary to elevation (ie, hydrostatic head). To standardize the protocol, we measured PAC on the right arm and right leg in all our participants; however, PAC could be measured on either the right or left extremities. It is important to note that none of our subjects had evidence of peripheral arterial obstruction or venous return issues in their right extremities.
The Vasogram has an air pump, calibration chamber (ie, piston), high-resolution pressure transducer, and 3 standard blood pressure cuffs, which are placed on the upper arm, thigh, and calf. The upper arm cuff measured systemic blood pressure (systolic, diastolic, and mean pressure). With the subjects in the supine position, the Vasogram measured segmental limb volume changes independently at the thigh and calf during the entire cardiac cycle. The segmental volume change in the thigh and calf were initially obtained at a pressure of 50 mm Hg. The cuff pressure automatically was increased by the device in 10–mm Hg increments, and segmental volume change measurements were obtained at each pressure until the segmental volume change was maximized. The maximum segmental volume change typically is seen when the cuff pressure is approximately mean systemic pressure (diastolic blood pressure + 1/3 pulse pressure). At each cuff pressure during early diastole, a known calibration volume (Vcal) of 0.65 mL was delivered via the piston rapidly expanding the closed cuff system. The thigh and calf measurements were taken independently. The introduction of Vcal caused the cuff pressure to rapidly change (Pcal). The maximum volume change (Max ΔV) during this sequence for the thigh and calf levels is directly related to pulse pressure. Max ΔV must be normalized to a specific pulse pressure to facilitate comparison among patients; 50 mm Hg was selected. Therefore, Max ΔV50 is calculated as follows: Max ΔV50 = Max ΔV*50/pulse pressure. Max ΔV50 is the segmental volume change normalized to a pulse pressure of 50 mm Hg. In other words, the unit of the Vmax50 is in milliliters of volume for each 50 mm Hg of pulse pressure. In this study, Max ΔV50 was used as the measure of PAC, with the unit of measurement being in milliliters. The machine automatically standardizes the volume change with each pulse wave to the 50–mm Hg value. Reduced values of PAC corresponded to reduced arterial compliance and increased atherosclerosis.2,3,15
Statistical Analyses
In this secondary analysis, log(calf) and log(thigh) compliance (measured in milliliters) were modeled in separate linear regressions. The PAC data were log transformed to normalize the distribution of the data to improve the fit of the regression analyses. The regressions contained factors of age (year), race (white vs black), smoking status (smoker vs nonsmoker), presence or absence of the metabolic syndrome (defined as having at least 3 of the following: waist circumference > 40 inches, triglycerides > 150 mg/dL, HDL < 40 mg/dL, systolic blood pressure 130 > mm Hg or > 85 mm Hg diastolic or being treated for hypertension, fasting glucose > 100 mg/dL), current treatment with a statin (yes vs no), diagnosis of schizophrenia or schizoaffective disorder (present vs absent), current antipsychotic treatment, and BMI (centered at 29.5 kg/m2 and entered in the models as BMI and BMI2 to allow for a curvilinear association between BMI and compliance). The medication status of the subjects was defined as (1) no antipsychotic medication within the prior 2 months, (2) treatment for at least 3 months with antipsychotics with a high risk of metabolic side effects (olanzapine, quetiapine, risperidone), or (3) treatment for at least 3 months with antipsychotics with a lower risk of metabolic side effects (typical antipsychotics, ziprasidone, aripiprazole). Only men were included in our analyses, because we did not have a sufficient number of women for a meaningful subset (n = 6) and women differ from men in PAC.19 Five subjects were omitted due to missing values for the metabolic syndrome factors. Because our analysis was a secondary analysis of existing data, we did not perform an ad hoc sample size estimation. All analyses were performed using SAS and R (SAS Institute Inc, Cary, North Carolina).
RESULTS
Table 1 describes the demographic and clinical variables in the sample. A total of 77 men (mean ± SD age of 53.7 ± 8.8 years) were studied. There were 41 whites and 36 blacks in the study. Twenty-seven subjects had a diagnosis of either schizophrenia or schizoaffective disorder (DSM-IV criteria). The number of subjects diagnosed with other psychiatric diagnoses included depression: n = 16, posttraumatic stress disorder (PTSD): n = 14, PTSD and anxiety: n = 2, PTSD and depression: n = 12, PTSD and schizophrenia: n = 2, bipolar disorder: n = 3, and psychosis not otherwise specified: n = 1. Fifty subjects were taking an antipsychotic and 27 were not receiving an antipsychotic medication. Of 77 participants, 36 (47%) had metabolic syndrome, 20 (26%) were on a statin, and 37 (47%) were smokers. Blacks had reduced calf PAC compared to whites (P = .02). Subjects with schizophrenia had reduced thigh PAC at a trend level (P = .06) compared to subjects with other psychiatric diagnoses. Subjects on no antipsychotic had the highest thigh PAC (4.41) compared to subjects on high-risk (3.92) and low-risk (3.45) weight gain antipsychotics, although this factor was not significant. For the 50 subjects currently treated with an antipsychotic, the duration of this treatment was not a significant predictor of thigh or calf compliance.
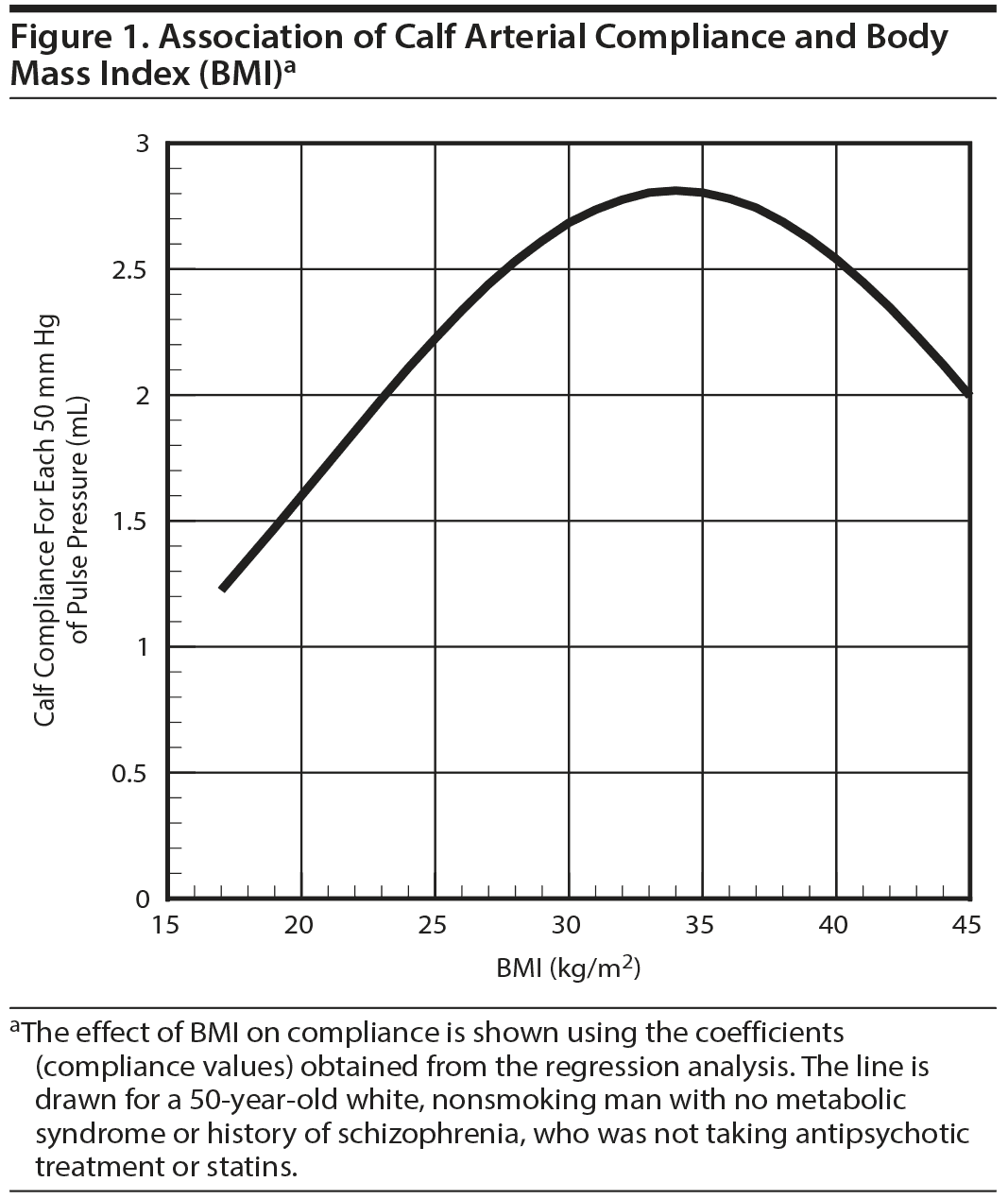
The multivariable model predicting calf compliance (Table 2) was significant (omnibus P = .0083) and predicted 27% of the variance in log calf compliance. Neither age nor age2 were predictors of log calf and log thigh compliance. Because age2 was not significant, it was dropped from the model and the model was rerun. BMI and BMI2 were significant predictors of calf compliance. Having a diagnosis of schizophrenia or being on any antipsychotic was not significantly associated with calf compliance. Similarly, use of statin therapy was not associated with compliance. The association between BMI and calf compliance was nonlinear (Figure 1). Surprisingly, compliance increased with increasing BMI up to a BMI in the mid 30-kg/m2 range and then fell as BMI continued to increase. The model did not predict thigh compliance (omnibus P = .27).
DISCUSSION
In this cross-sectional study of veterans with psychiatric diagnoses, race (white vs black) and BMI were significant predictors of arterial compliance in the calf. Having the metabolic syndrome was a borderline significant predictor of lower PAC. This secondary analyses study is, to our knowledge, the second study to investigate PAC in subjects with psychiatric diagnoses using the plethysmography technique. Using data from the same group of subjects, we previously reported8 that PAC was reduced in people with 1 or more psychiatric diagnoses, even if they were not currently being treated with antipsychotics. In this previous article,8 we compared the PAC of 63 subjects with a psychiatric diagnosis with 111 healthy controls with no psychiatric diagnoses. Also in this previous article,8 we examined differences in PAC among subjects on quetiapine or risperidone or off antipsychotics versus healthy controls. In this previous article,8 thigh and calf compliance of the 111 healthy controls with no psychiatric diagnosis were 4.7 and 2.42, respectively, which is much higher than the values in Table 1. As shown in Table 1, calf PAC was significantly reduced in smokers compared to nonsmokers, but this finding was not seen in the multivariable analyses. As shown in Table 1, subjects on no antipsychotic had higher thigh PAC compared to both subjects on high-risk and low-risk weight gain antipsychotics, although this difference was not significant. Subjects with schizophrenia had reduced thigh PAC at a trend level compared to subjects with other psychiatric diagnoses (P = .06, Table 1). However, the findings in Table 1 are limited because significant findings may not be independent of the other factors adjusted for in the multivariable analyses (Table 2). In the multivariable model predicting calf and thigh compliance, race and BMI were significant predictors of calf compliance, and metabolic syndrome was significant at a trend level (P < .08).
Vasogram is not the only way to measure arterial compliance. For instance, the pulse wave velocity (PWV) technique has also been used for the same purpose. In a study20 that measured arterial compliance using the PWV technique, arterial compliance was significantly reduced in 10 participants with schizophrenia compared to 10 age- and sex-matched healthy controls. Although PWV is considered the gold standard, it has several limitations compared to the plethysmography technique using the Vasogram device. The Vasogram accuracy has been shown to produce arterial volume changes with errors less than 0.01 mL. The measurement of arterial compliance is changed by physiologic state (pressure, flow, velocity, etc). The Vasogram procedure carefully accounts for these changes; in contrast, the PWV procedure makes no corrections for physiologic status.21 While the Vasogram measures arterial compliance directly in an arterial bed that mirrors the coronary arteries, PWV is a surrogate measure for arterial compliance from a mixture of arterial beds that are not as closely correlated to coronary atherosclerosis.21 In addition, the Vasogram procedure includes neovascularization; the PWV procedure does not.21 The concept of neovascularization has been shown to be very important with regard to the consequences of atherosclerosis. This is the phenomenon by which increasing arterial impedance secondary to atherosclerosis is compensated. The Vasogram assesses a limited but defined segment of the muscular arteries of the lower extremities. These arteries have been shown to change with improvement in compliance associated with treatments such as statins, which are known to improve vascular reactivity, ie, post ischemic dilatation in the upper extremity.22 Furthermore, the PAC as measured by a Vasogram has been shown to maintain similar values into the eighth decade of life, probably because the stiffening of elastic arteries (eg, thoracic aorta) is not relevant to this measure. PWV measured from the carotid to the groin is highly dependent on both the thoracic and abdominal aortic stiffness, and they represent 2 different types of arterial tissue. The evidence suggests that the reduced PAC measured by the Vasogram is primarily due to muscle tone, an element responsive to nitric oxide secretion and therefore to physiologic and pharmacologic mechanisms.23 However, a thoracic aorta stiffened in old age is composed of elastic tissue that is not so responsive to such changes or related interventions. The relationship to extent of vascular disease by imaging techniques (angiography and MRI) and functionality measures such as walk time on the tread mill are strongly correlated with the Vasogram measures, and, therefore, it is a powerful measure of vascular abnormalities related to both early and late atherosclerosis. In addition, PWV does not measure arterial compliance using the standard formula: Δvolume/Δpressure.
As previously stated, the Vasogram is a noninvasive, inexpensive device that provides automated measurement requiring minimal operator training. The convenience of the Vasogram is evident; the entire procedure can be completed in 15 minutes in the comfort of an office setting. In contrast, the PWV technique requires a trained professional with medical experience and an echocardiography and is more expensive. Scientific data2,3,8,14,15,19 with no professional marketing is not enough to introduce a new technology. The Soterogram, a US Food and Drug Administration–registered and cleared device, is currently available for clinical use (Soteria Medical, LLC, Miami, Florida) and may soon become the standard of care for clinical practice.
The mechanism by which reduced PAC is associated with cardiovascular pathology is not entirely clear. PAC is a predictor of cardiovascular disorders independent of hypertension.24 Several studies25,26 indicate that vascular inflammation is associated with reduced arterial compliance. Reduced arterial compliance is associated with increased activity of angiotensin II (Ang II), increased nicotinamide adenine dinucleotide phosphate oxidase activity, reduced nitric oxide activity, and increased reactive oxygen species.27–29 Ang II signaling activates transforming growth factor β (TGF-β), tumor necrosis factor α (TNF-α), interleukins (IL-1, IL-17, IL-6), and C-reactive protein (CRP), which are associated with reduced arterial compliance.29–36 In addition, high-sensitivity CRP is significantly associated with reduced arterial compliance.37–39 Finally, there is evidence to suggest that infliximab (a monoclonal antibody against TNF-α) reduces inflammation and may improve arterial compliance in patients with rheumatoid arthritis.40 Hence, there is mounting evidence that inflammation plays a significant part in the pathophysiology of reduced arterial compliance.
There is evidence that reduced PAC can be treated. A study15 of 10 patients with coronary disease with and without diabetes who were given simvastatin (40 mg/d) for 4 months demonstrated higher PAC in all 10 patients after 2 months of statin treatment. After 4 months of therapy, their PAC increased by 34%.15 At this point, we do not know how long a duration of exposure to clinical risk factors is required for a clinically meaningful effect on PAC. Only a well-designed longitudinal study can shed light on this question. There is also evidence that the use of omega-3 fatty acids increases arterial compliance in people with dyslipidemia.41 Finally, in people with rheumatoid arthritis, infliximab reduced aortic inflammation, and this effect correlated with increases in aortic compliance.40 Hence, treating the potentially reversible factors that predict reduced PAC such as metabolic syndrome and obesity (Table 2) may decrease mortality in people with psychiatric diagnoses.
While there is a high prevalence of obesity (30%) in the general population,42 people with schizophrenia have twice the prevalence of obesity compared to the general population.42–44 In a study44 of 169 subjects, 50% of women and 41% of men with schizophrenia were obese compared with 27% and 20%, respectively, in the comparison group (National Health and Nutrition Examination Survey data set with 2,404 matched individuals). The extremely high prevalence of obesity is important to note because obesity is associated with type 2 diabetes, hypertension,43,44 cardiovascular disease, and early mortality. In the United States, 42% of people with schizophrenia have a BMI ≥ 27, which places them at significant risk for insulin resistance and cardiovascular disease.45
Because of the risks involved, clinicians should consider prescribing antipsychotic medications that are less likely to cause or worsen obesity and its symptoms. Antipsychotic medications are commonly prescribed to individuals in the United States. In 2010, 1.26% of adults aged 20–64 years and 1.36% of adults aged 65–69 years in the United States were on an antipsychotic medication.46 Most antipsychotics used to treat schizophrenia and other psychiatric diagnoses cause or worsen the metabolic syndrome.47 Antipsychotics such as clozapine and olanzapine are proatherogenic.48 People with psychiatric disorders and on antipsychotics may have metabolic and inflammatory changes that we are only beginning to recognize. Some atypical antipsychotics are known to be associated with weight gain, hyperlipidemia, hyperglycemia, and type 2 diabetes.48 Since the metabolic changes associated with antipsychotic therapy are risk factors for cardiovascular disease, psychiatrists need to weigh the metabolic risks and therapeutic benefits of the various types of antipsychotics. Assessment of PAC by means of the Vasogram method may prove useful in assessing the progression of atherosclerosis during treatment with an antipsychotic medication. Larger prospective studies are warranted to investigate this potential.
To our knowledge, this is the first pilot study to examine the clinical variables that are predictors of calf arterial compliance in participants with psychiatric diagnoses. Our study has a number of limitations. For instance, our results are cross-sectional, and we lack longitudinal data. Both of these limitations can be resolved in further research on the topic. Another limitation is that the psychiatric diagnoses were established by chart review rather than a structured diagnostic interview. Because of this, the diagnoses were not verified by the research investigators outside of thorough chart reviews. In this study, we were not able to control for potential confounders such as dose and duration of antipsychotic treatment. Outpatient medication adherence was estimated by subject interview and electronic medical record documentation of the subjects having consistently obtained refills of their antipsychotic such that they had an adequate outpatient supply of medication during the treatment period. However, their adherence was inferred rather than strictly controlled as can be done in a prospective clinical study. Furthermore, we have not taken into account the duration of the mental disorder of the subjects in the project. Unlike the study in which statin treatment improved PAC,15 the current study did not detect an association between being on a statin and calf compliance. However, only 20 of our 77 subjects (25%) were on a statin, so the sample size for this subgroup of subjects may have limited our ability to detect effects of statin treatment.
CONCLUSIONS AND FUTURE DIRECTIONS
In this pilot study in male veterans, significant predictors of calf compliance were race (black vs white) and BMI. PAC is a noninvasive measure that may be a predictor of cardiovascular risk in psychiatric patients. The preliminary data from our study may be used as a source of sample size estimates for a larger study in the future. Future studies are warranted to better understand the pathophysiology of arterial compliance including but not limited to inflammation in psychiatric patients.
Submitted: August 31, 2015; accepted March 31, 2016.
Published online: June 9, 2016.
Drug names: aripiprazole (Abilify), clozapine (Clozaril, FazaClo, and others), olanzapine (Zyprexa and others), quetiapine (Seroquel and others), risperidone (Risperdal and others), simvastatin (Zocor and others), ziprasidone (Geodon and others).
Author contributions: Drs Koola, Brown, and Duncan designed the study. Drs Koola, Sorkin, and Duncan conceived the analysis plan; Dr Sorkin performed the statistical analyses. Drs Koola, Brown, Hollis, and Duncan and Mr Cuthbert were responsible for the conduct of the study. All authors interpreted the results. Dr Koola wrote the first draft of the manuscript; all authors edited the manuscript with intellectual contributions. All authors approved the final manuscript.
Potential conflicts of interest: Dr Brown has served as a consultant to Amgen, Genzyme, Sanofi, Merck, Kowa, Aegerion, and Esperion and is the editor of the Journal of Clinical Lipidology. Dr Raines is the president and chief operating officer of Soteria Medical, LLC. Dr Duncan has received grant support from Posit Science Corporation and Auspex Pharmaceuticals, Inc, Center for Translational Social Neuroscience at Emory. Drs Koola, Sorkin, and Hollis and Ms Fargotstein and Mr Cuthbert report no conflicts of interest related to the subject of this article.
Funding/support: This work was supported by VA Merit Review grant (Dr Duncan), NIDDK P30 Nutrition of Obesity Research Center of Maryland DK072488 (Dr Sorkin), National Institute on Aging P30 AG028747 (Pepper Center) (Dr Sorkin), and the Baltimore VA Geriatrics Research, Education, and Clinical Center (Dr Sorkin). The Mental Health, Research and Development, and Rehabilitation Research & Development Service Lines of the Atlanta Veterans Affairs Medical Center and Emory University contributed infrastructure support (Dr Duncan).
Role of the sponsor: The sponsors had no role in the design and conduct of the study; collection, management, analysis, and interpretation of data; or preparation, review, or approval of the manuscript.
Previous presentation: The first author (Dr Koola) presented these data at the 52nd American College of Neuropsychopharmacology meeting; December 8–12, 2013; Hollywood, Florida and the 69th Society of Biological Psychiatry meeting; May 8–10, 2014; New York, New York.
REFERENCES
1. Cohn JN. Arterial compliance to stratify cardiovascular risk: more precision in therapeutic decision making. Am J Hypertens. 2001;14(8 pt 2):258S–263S. doi:10.1016/S0895-7061(01)02154-9 PubMed
2. Willens HJ, Davis W, Herrington DM, et al. Relationship of peripheral arterial compliance and standard cardiovascular risk factors. Vasc Endovascular Surg. 2003;37(3):197–206. doi:10.1177/153857440303700307 PubMed
3. Herrington DM, Kesler K, Reiber J, et al. Arterial compliance adds to conventional risk factors for prediction of angiographic coronary artery disease. Am Heart J. 2003;146(4):662–667. doi:10.1016/S0002-8703(03)00254-0 PubMed
4. Wada T, Kodaira K, Fujishiro K, et al. Correlation of ultrasound-measured common carotid artery stiffness with pathological findings. Arterioscler Thromb. 1994;14(3):479–482. doi:10.1161/01.ATV.14.3.479 PubMed
5. Nair N, Oka RK, Waring LD, et al. Vascular compliance versus flow-mediated vasodilation: correlation with cardiovascular risk factors. Vasc Med. 2005;10(4):275–283. doi:10.1191/1358863x05vm633oa PubMed
6. Caughey MC, Loehr LR, Cheng S, et al. Associations between echocardiographic arterial compliance and incident cardiovascular disease in blacks: the ARIC study. Am J Hypertens. 2015;28(1):81–88. doi:10.1093/ajh/hpu087 PubMed
7. Papaioannou TG, Protogerou AD, Stergiopulos N, et al. Total arterial compliance estimated by a novel method and all-cause mortality in the elderly: the PROTEGER study. Age (Dordr). 2014;36(3):9661. doi:10.1007/s11357-014-9661-0 PubMed
8. Koola MM, Brown WV, Qualls C, et al. Reduced arterial compliance in patients with psychiatric diagnoses. Schizophr Res. 2012;137(1–3):251–253. doi:10.1016/j.schres.2012.02.007 PubMed
9. Newman SC, Bland RC. Mortality in a cohort of patients with schizophrenia: a record linkage study. Can J Psychiatry. 1991;36(4):239–245. PubMed
10. Harris EC, Barraclough B. Excess mortality of mental disorder. Br J Psychiatry. 1998;173(1):11–53. doi:10.1192/bjp.173.1.11 PubMed
11. Healy D, Le Noury J, Harris M, et al. Mortality in schizophrenia and related psychoses: data from two cohorts, 1875–1924 and 1994–2010. BMJ Open. 2012;2(5):e001810. doi:10.1136/bmjopen-2012-001810 PubMed
12. Colton CW, Manderscheid RW. Congruencies in increased mortality rates, years of potential life lost, and causes of death among public mental health clients in eight states. Prev Chronic Dis. 2006;3(2):A42. PubMed
13. Olfson M, Gerhard T, Huang C, et al. Premature mortality among adults with schizophrenia in the United States. JAMA Psychiatry. 2015;72(12):1172–1181. doi:10.1001/jamapsychiatry.2015.1737 PubMed
14. Herrington DM, Brown WV, Mosca L, et al. Relationship between arterial stiffness and subclinical aortic atherosclerosis. Circulation. 2004;110(4):432–437. doi:10.1161/01.CIR.0000136582.33493.CC PubMed
15. Saliashvili G, Davis WW, Harris MT, et al. Simvastatin improved arterial compliance in high-risk patients. Vasc Endovascular Surg. 2004;38(6):519–523. doi:10.1177/153857440403800605 PubMed
16. Bak M, Fransen A, Janssen J, et al. Almost all antipsychotics result in weight gain: a meta-analysis. PLoS ONE. 2014;9(4):e94112. doi:10.1371/journal.pone.0094112 PubMed
17. Cwynar M, Wojciechowska W, Kawecka-Jaszcz K, et al. Mechanisms of large artery remodeling [in Polish]. Przegl Lek. 2002;59(suppl 3):1–8. PubMed
18. Boutouyrie P. Arterial ageing and coronary artery disease: physiopathological and epidemiological data [in French]. Arch Mal Coeur Vaiss. 2006;99(spec no 4):19–24. PubMed
19. Le NA, Brown WV, Davis WW, et al. Comparison of the relation of triglyceride-rich lipoproteins and muscular artery compliance in healthy women versus healthy men. Am J Cardiol. 2005;95(9):1049–1054. doi:10.1016/j.amjcard.2004.12.057 PubMed
20. Phillips AA, Warburton DE, Flynn SW, et al. Assessment of arterial stiffness among schizophrenia-spectrum disorders using aortic pulse wave velocity and arterial compliance: a pilot study. Psychiatry Res. 2014;215(1):14–19. doi:10.1016/j.psychres.2013.10.020 PubMed
21. Torrado J, Bia D, Zócalo Y, et al. Hyperemia-related changes in arterial stiffness: comparison between pulse wave velocity and stiffness index in the vascular reactivity assessment. Int J Vasc Med. 2012;2012:490742. PubMed
22. Balk EM, Karas RH, Jordan HS, et al. Effects of statins on vascular structure and function: a systematic review. Am J Med. 2004;117(10):775–790. doi:10.1016/j.amjmed.2004.05.026 PubMed
23. Houston M, Hays L. Acute effects of an oral nitric oxide supplement on blood pressure, endothelial function, and vascular compliance in hypertensive patients. J Clin Hypertens (Greenwich). 2014;16(7):524–529. PubMed
24. Park S, Lakatta EG. Role of inflammation in the pathogenesis of arterial stiffness. Yonsei Med J. 2012;53(2):258–261. doi:10.3349/ymj.2012.53.2.258 PubMed
25. Zieman SJ, Melenovsky V, Kass DA. Mechanisms, pathophysiology, and therapy of arterial stiffness. Arterioscler Thromb Vasc Biol. 2005;25(5):932–943. doi:10.1161/01.ATV.0000160548.78317.29 PubMed
26. Lakatta EG, Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises, part II: the aging heart in health: links to heart disease. Circulation. 2003;107(2):346–354. doi:10.1161/01.CIR.0000048893.62841.F7 PubMed
27. van der Loo B, Labugger R, Skepper JN, et al. Enhanced peroxynitrite formation is associated with vascular aging. J Exp Med. 2000;192(12):1731–1744. doi:10.1084/jem.192.12.1731 PubMed
28. Csiszar A, Ungvari Z, Edwards JG, et al. Aging-induced phenotypic changes and oxidative stress impair coronary arteriolar function. Circ Res. 2002;90(11):1159–1166. doi:10.1161/01.RES.0000020401.61826.EA PubMed
29. Wang MC, Tsai WC, Chen JY, et al. Arterial stiffness correlated with cardiac remodelling in patients with chronic kidney disease. Nephrology (Carlton). 2007;12(6):591–597. doi:10.1111/j.1440-1797.2007.00826.x PubMed
30. Belmin J, Bernard C, Corman B, et al. Increased production of tumor necrosis factor and interleukin-6 by arterial wall of aged rats. Am J Physiol. 1995;268(6 pt 2):H2288–H2293. PubMed
31. Gerli R, Monti D, Bistoni O, et al. Chemokines, sTNF-Rs and sCD30 serum levels in healthy aged people and centenarians. Mech Ageing Dev. 2000;121(1–3):37–46. doi:10.1016/S0047-6374(00)00195-0 PubMed
32. Pasceri V, Willerson JT, Yeh ET. Direct proinflammatory effect of C-reactive protein on human endothelial cells. Circulation. 2000;102(18):2165–2168. doi:10.1161/01.CIR.102.18.2165 PubMed
33. Torzewski M, Rist C, Mortensen RF, et al. C-reactive protein in the arterial intima: role of C-reactive protein receptor-dependent monocyte recruitment in atherogenesis. Arterioscler Thromb Vasc Biol. 2000;20(9):2094–2099. doi:10.1161/01.ATV.20.9.2094 PubMed
34. Venugopal SK, Devaraj S, Yuhanna I, et al. Demonstration that C-reactive protein decreases eNOS expression and bioactivity in human aortic endothelial cells. Circulation. 2002;106(12):1439–1441. doi:10.1161/01.CIR.0000033116.22237.F9 PubMed
35. Csiszar A, Ungvari Z, Koller A, et al. Aging-induced proinflammatory shift in cytokine expression profile in coronary arteries. FASEB J. 2003;17(9):1183–1185. PubMed
36. Spinetti G, Wang M, Monticone R, et al. Rat aortic MCP-1 and its receptor CCR2 increase with age and alter vascular smooth muscle cell function. Arterioscler Thromb Vasc Biol. 2004;24(8):1397–1402. PubMed
37. Mattace-Raso FU, van der Cammen TJ, van der Meer IM, et al. C-reactive protein and arterial stiffness in older adults: the Rotterdam Study. Atherosclerosis. 2004;176(1):111–116. doi:10.1016/j.atherosclerosis.2004.04.014 PubMed
38. Mahmud A, Feely J. Arterial stiffness is related to systemic inflammation in essential hypertension. Hypertension. 2005;46(5):1118–1122. doi:10.1161/01.HYP.0000185463.27209.b0 PubMed
39. Nagano M, Nakamura M, Sato K, et al. Association between serum C-reactive protein levels and pulse wave velocity: a population-based cross-sectional study in a general population. Atherosclerosis. 2005;180(1):189–195. doi:10.1016/j.atherosclerosis.2004.11.019 PubMed
40. Mäki-Petäjä KM, Elkhawad M, Cheriyan J, et al. Anti-tumor necrosis factor-α therapy reduces aortic inflammation and stiffness in patients with rheumatoid arthritis. Circulation. 2012;126(21):2473–2480. doi:10.1161/CIRCULATIONAHA.112.120410 PubMed
41. Nestel P, Shige H, Pomeroy S, et al. The n-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid increase systemic arterial compliance in humans. Am J Clin Nutr. 2002;76(2):326–330. PubMed
42. Hedley AA, Ogden CL, Johnson CL, et al. Prevalence of overweight and obesity among US children, adolescents, and adults, 1999–2002. JAMA. 2004;291(23):2847–2850. doi:10.1001/jama.291.23.2847 PubMed
43. Homel P, Casey D, Allison DB. Changes in body mass index for individuals with and without schizophrenia, 1987–1996. Schizophr Res. 2002;55(3):277–284. doi:10.1016/S0920-9964(01)00256-0 PubMed
44. Dickerson FB, Brown CH, Kreyenbuhl JA, et al. Obesity among individuals with serious mental illness. Acta Psychiatr Scand. 2006;113(4):306–313. doi:10.1111/j.1600-0447.2005.00637.x PubMed
45. Fontaine KR, Heo M, Harrigan EP, et al. Estimating the consequences of antipsychotic induced weight gain on health and mortality rate. Psychiatry Res. 2001;101(3):277–288. doi:10.1016/S0165-1781(01)00234-7 PubMed
46. Olfson M, King M, Schoenbaum M. Antipsychotic treatment of adults in the United States. J Clin Psychiatry. 2015;76(10):1346–1353. doi:10.4088/JCP.15m09863 PubMed
47. Newcomer JW. Antipsychotic medications: metabolic and cardiovascular risk. J Clin Psychiatry. 2007;68(suppl 4):8–13. PubMed
48. Chaggar PS, Shaw SM, Williams SG. Effect of antipsychotic medications on glucose and lipid levels. J Clin Pharmacol. 2011;51(5):631–638. doi:10.1177/0091270010368678 PubMed
Enjoy this premium PDF as part of your membership benefits!

