Lessons Learned at the Interface of Medicine and Psychiatry
The Psychiatric Consultation Service at Massachusetts General Hospital sees medical and surgical inpatients with comorbid psychiatric symptoms and conditions. During their twice-weekly rounds, Dr Stern and other members of the Consultation Service discuss diagnosis and management of hospitalized patients with complex medical or surgical problems who also demonstrate psychiatric symptoms or conditions. These discussions have given rise to rounds reports that will prove useful for clinicians practicing at the interface of medicine and psychiatry.
Prim Care Companion CNS Disord 2026;28(2):25f04094
Author affiliations are listed at the end of this article.
Have you ever been troubled by the worry and panic experienced by your dyspneic patients? Have you wondered about how your patients with dyspnea cope with persistent or episodic shortness of breath? Have you been uncertain about how best to comfort and treat your patients with dyspnea? Have you wondered whether your dyspneic patients can refuse assisted ventilation? If you have, the following case vignette and discussion should prove useful.
CASE VIGNETTE
Mr A, a 68-year-old man with interstitial lung disease (ILD), had been on 4 L/minute of oxygen at home; however, he was admitted to the hospital with 3 days of worsening dry cough and shortness of breath, with the suspicion that he had developed a pulmonary infection. He was increasingly fatigued and required 6 L of oxygen to maintain his oxygen saturation at 88%. His radiologic imaging was remarkable for bilateral lung opacities. His initial arterial blood gas (ABG) revealed a pH of 7.29 (normal range, 7.35–7.45), a PaCO2 of 48 mm Hg (normal range, 35–45), a PaO2 of 55 mm Hg (normal range, 80–100), and an HCO3-of 21 mEq/L (normal range, 22–26) with a lactate level of 2.2 mmol/L (normal range, 0.5–2.0).
He was started on broad-spectrum antibiotics (while awaiting the results of cultures and viral swabs) and high-dose steroids, and he was placed on high-flow oxygen (via a nasal cannula). Despite this, Mr A reported that he was having difficulty breathing, feeling anxious, and struggling to sleep. Psychiatry was consulted to evaluate and treat his anxiety.
On psychiatric examination, Mr A was awake, fully oriented, sitting upright in bed, and leaning slightly forward; however, he appeared anxious and uncomfortable. He was tachypneic and used his neck accessory muscles to breathe. His vital signs included a respiratory rate of 26 breaths/minute, a heart rate of 90 beats/minute, and a blood pressure of 140/85 mm Hg. He had an oxygen saturation of 91% on a high-flow oxygen mask. He said that he felt like he was not getting enough air and that he had to work hard to breathe. He worried that he would suffocate and die. Although his medical team discussed possible next steps (including intubation) with Mr A, he was not sure about whether he would agree to be intubated if it was not followed soon by extubation. He was frightened and unsure about what he wanted to do.
Laboratory testing showed an improving white blood cell count and a hemoglobin level of 11. His renal and hepatic function tests were unremarkable. His most recent ABG showed a pH of 7.46, a PaCO2 of 32 mm Hg, a PaO2 of 68 mm Hg, and a HCO3-level of 22 mEq/L.
DISCUSSION
What Is Dyspnea, How Is It Diagnosed, and What Does It Feel Like?
Dyspnea is the subjective experience of difficulty breathing reported by patients.1,2 It occurs in approximately 10% of those in the general population, with certain factors (eg, female sex, older age, cardiorespiratory diseases, an elevated body mass index, and impaired lung function) being associated with the diagnosis.3 In hospitalized patients or those in an intensive care unit (ICU) (eg, following lung transplantation), it is even more common.4,5 Some individuals describe dyspnea as a frightening sensation that mirrors that of suffocation, while others say that it feels as though they need to breathe more deeply but are unable to do so.1
Individuals with dyspnea describe their experiences differently, making it challenging to ascertain its etiology. For example, when bronchoconstriction is associated with asthma, black patients often used words that described conditions in their upper airway (eg, tight voice, tight throat), while white patients described lower chest wall and airway descriptors (eg, “It hurts to breathe,” “I am out of air”).6 Therefore, clinicians should consider how an individual’s culture or background influences their description of symptoms.6
Despite dyspnea’s prevalence and clinical significance, it is often underappreciated and underdiagnosed. Since a standard method for the assessment of dyspnea is lacking, several questionnaires have been used to assess dyspnea, although the assessment is frequently complicated by communication difficulties.3 Unfortunately, critically ill individuals who suffer from dyspnea are prone to prolonged courses of mechanical ventilation, a worse quality of life following discharge, frequent anxiety, and an increased risk of posttraumatic stress disorder (PTSD) postdischarge.7
The first steps in the evaluation of an individual with dyspnea involve asking about the symptoms, taking a thorough history, and ruling out cognitive impairment (eg, delirium), as having delirium often interferes with meaningful communication.8 Evaluators should consider what is happening when dyspnea develops (eg, whether it is during exertion, at rest, in a specific body position, or associated with emotional distress), as well as other factors (eg, anemia, deconditioning, and emotional responses).9 Certain questionnaires, including the Modified Respiratory Scale, can also assist in clarifying the history.3 The severity of dyspnea can be further characterized by using tools such as the Dyspnea Visual Analog Scale (VAS) or the Numerical Rating Scale (NRS)7; a VAS >3 or a NRS ≥4 typically indicates significant dyspnea that requires further diagnostic evaluation and a therapeutic intervention.10 Individuals with acute dyspnea often show signs of an increased work of breathing (eg, a tracheal tug, Hoover sign, nasal flaring, inspiratory laryngeal groans, sternocleidomastoid activation, recession of suprasternal fossa) or signs of respiratory distress (eg, tachypnea, activation of accessory muscles, hyperpnea).11 In patients who are unable to communicate, the Intensive Care–Respiratory Distress Observation Scale (IC-RDOS) correlates with the Dyspnea VAS.12 The IC-RDOS was further modified to assess patients who are being mechanically ventilated, known as the Mechanical Ventilation–Respiratory Distress Observation Scale.7
Once dyspnea has been identified, the emphasis shifts to identifying its etiology. Certain tools (eg, the IMPRESS Framework and the Breathing SPACE Framework) can frame a differential diagnosis and guide the ordering of laboratory tests that might refine the diagnosis.11,13
What Are the Mechanisms Behind Dyspnea?
Dyspnea involves physiological, psychological, and environmental factors.8 Moreover, disruption of each of them can lead to dyspnea. First, the motor cortex signals the intercostal and neck muscles while the brain stem signals the diaphragm via the phrenic nerve to initiate and maintain the drive to breathe.7 Simultaneously, there is an afferent feedback loop from the motor cortex to the sensory cortex (named the corollary discharge), which modulates the perception of the work of breathing.14 The mismatch between this afferent sensory feedback signaling (effort) and the activation of respiratory muscles (the actual result) is the primary driver of dyspnea, which is characterized by an increased work of breathing. Therefore, those with dyspnea report “I need to breathe harder,” “I am working hard to breathe.”
In addition, both peripheral and central chemoreceptors modulate respiratory motor activity in response to changes in arterial pH, as well as changes in oxygen and carbon dioxide (CO2) concentrations.14 The brain stem receives these afferent signals and commands the primary motor cortex to activate the respiratory muscles to maintain homeostasis. Respiratory acidosis increases signaling from the brain stem to the limbic system (comprised by the thalamus, insular cortex, and amygdala), which can trigger manifestations of anxiety.7,15 When ventilatory output fails to match or correct the chemoreceptor findings, dyspnea is characterized by air hunger (an unpleasant, unsatisfied urge to breathe). Since the limbic system regulates emotion, one’s emotional overlay is important and physiologically understandable in dyspnea.16
Finally, the stretch receptors in the lung parenchyma, airways, chest wall, and intercostal muscles (eg, the Golgi tendon organs and muscle spindles in the lung parenchyma, chest wall, and intercostal muscles; the slowly adapting and rapidly adapting stretch receptors in the smooth muscle of the proximal airways; C-fiber receptors in the distal airway mucosa near alveolar capillaries) are activated by pulmonary stretch and noxious stimuli and send signals via the vagal nerve to the brain stem.7 These signals are ultimately transmitted to the motor cortex, which leads to changes in the respiratory cycle, bronchoconstriction, and activation of the cough reflex. This might result in dyspnea that is characterized by chest tightness or constriction.
What Causes Dyspnea?
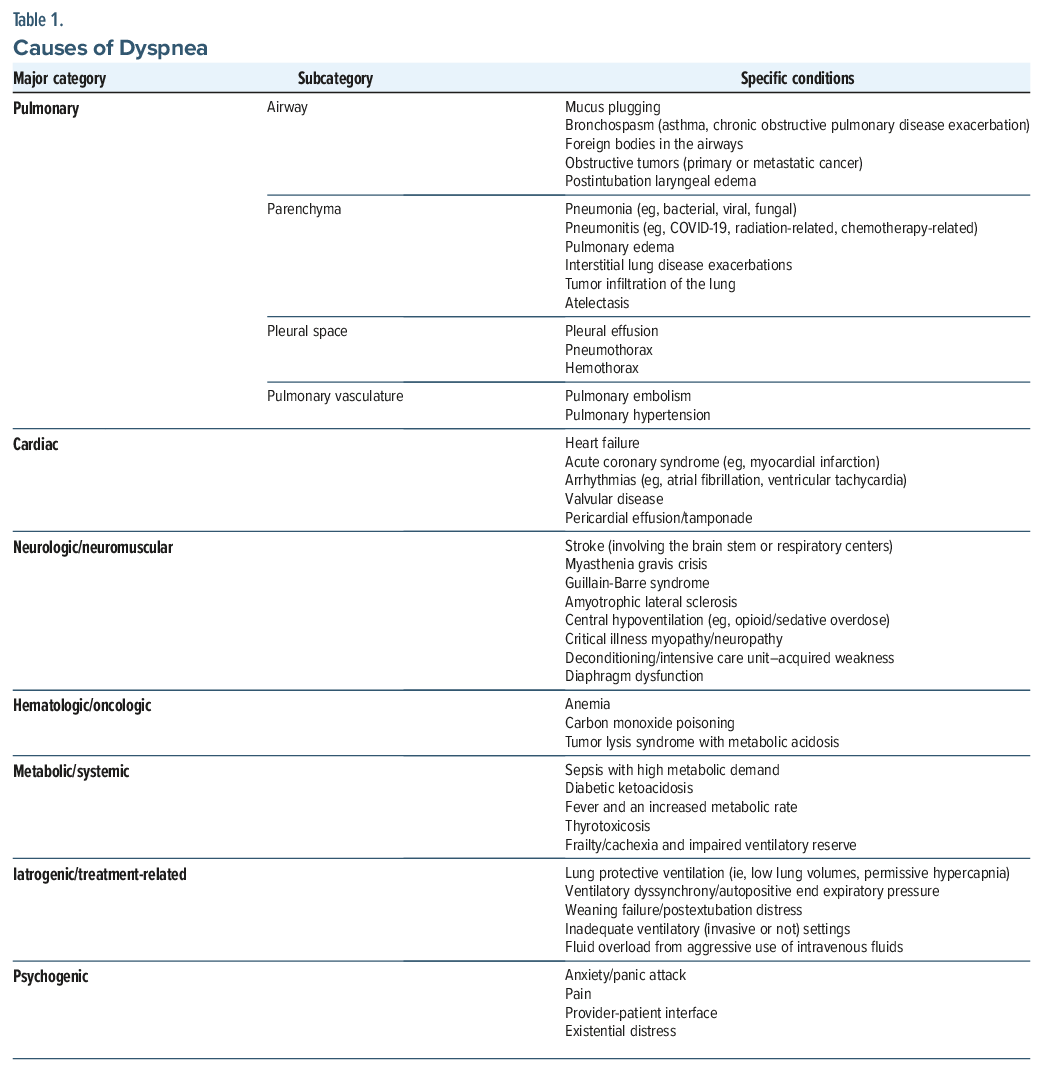
Many conditions (eg, pulmonary, cardiac, neuromuscular, hematologic/oncologic, metabolic, systemic) can contribute to, or elicit, dyspnea. However, even when underlying etiologies are maximally addressed, patients can experience persistent respiratory-derived brain suffering (mental distress related to physiologic or perceived respiratory abnormalities),8 and a multimodal approach is vital to address and reduce suffering (see Table 1 for a detailed list of etiologies for dyspnea).
Differentiating causes of dyspnea require the use of a structured approach that considers pulmonary, cardiac, and other nonpulmonary causes,17 and the way that an individual describes their shortness of breath can provide clues to its etiology (eg, “can’t get enough air” may suggest a restrictive or neuromuscular cause, while “can’t exhale” often points to obstructive diseases like asthma).18 Common physical signs and symptoms of dyspnea include rales, peripheral edema, pallor, and an irregular cardiac rhythm17,19 that can cause or worsen dyspnea by lowering cardiac output.20–23 The initial laboratory evaluation can facilitate the detection of arrhythmias or ischemic changes.9,24,25
How Can Cardiopulmonary Causes of Dyspnea Be Managed?
Effective management of dyspnea caused by cardiopulmonary conditions begins with addressing the underlying issue, as treatment is typically predicated on etiology. Identifying and treating the root cardiopulmonary causes are key to alleviating symptoms and improving outcomes.26,27 It is helpful for treatment teams to understand both the root cause of dyspnea and corresponding interventions for their patients so that they may have a full understanding of the patient’s experience, especially because different conditions may entail different psychiatric interventions for dyspnea. For example, when dyspnea is expected to be chronic and a patient has associated psychiatric symptoms, many more interventions may be considered than when the cause of dyspnea is acute and reversible. Additionally, different medical interventions may also carry psychiatric implications, such as the invasiveness of mechanical ventilation (and possible need for additional sedation) or the potentially stimulating effects of β2-agonist bronchodilators. Understanding these differential effects can help clinicians best educate and treat patients to enhance their comfort.
Cardiac arrhythmias (eg, atrial fibrillation) require a tailored approach that includes rate and rhythm control, anticoagulation (to prevent stroke), and addressing underlying triggers (like infections or heart failure).28–30 Management of asthma is often achieved with inhaled corticosteroids and bronchodilators, with escalation of doses as needed. Elderly individuals may also require lower doses and close monitoring for side effects.31,32 Pneumonia requires prompt empiric antibiotics, supportive care, and management of comorbidities.33 Anemia is managed by identifying and treating the underlying cause and administering transfusions if symptomatic or anemia is severe. Correction of reversible factors (eg, iron, B12 deficiency) is prioritized.34
For patients who are mechanically ventilated, lung-protective ventilation (LPV) is a technique designed to minimize intrapulmonary pressures and is indicated for those with dyspnea due to cardiopulmonary causes (eg, heart failure, arrhythmias, chronic obstructive pulmonary disease [COPD], asthma, pneumonia, ILD, pulmonary embolism, or anemia) when invasive mechanical ventilation is required for acute respiratory failure. This approach is recommended as the initial ventilatory strategy regardless of the presence or absence of acute respiratory distress syndrome (ARDS), due to its safety profile and its ability to minimize ventilator-induced lung injury in those with either ARDS or non-ARDS conditions.35,36 In heart failure or pulmonary edema, a higher positive end expiratory pressure may be beneficial for alveolar recruitment, but it must be balanced against the risk of hemodynamic compromise, especially in the elderly.37 However, it is important to appreciate that LPV with its low volumes and permissive hypercapnia often worsens sensations of dyspnea and might be difficult for patients to tolerate, representing an important but challenging risk-benefit analysis where clinicians must work to optimize pulmonary health while not excessively worsening subjective distress.7
What Is the Relationship Between Dyspnea and Anxiety?
Dyspnea and anxiety are closely interwoven; however, the extent of dyspnea cannot always be explained by the structural pathology associated with respiratory disorders. Moreover, breathlessness is not reliably predicted by the extent of airflow obstruction. To explain the relationship among the physiologic, psychological, and environmental factors in the phenomenology of dyspnea, a clinical model called “breathing, thinking, and functioning” was developed by the Cambridge Breathlessness Intervention Service38; it is based on cognitive and behavioral concepts of awareness of breathing, which impacts functioning and maintains a vicious cycle of increased dyspnea. Dyspnea leads to dysfunctional breathing, characterized by shorter exhalation, apical breathing, and a lack of end-expiratory pauses. The perception of “needing more air” leads to over-reliance on easily fatigued accessory muscles of respiration, and underutilization of the efficient and relatively fatigue-resistant diaphragmatic muscle, further increasing the work of breathing. This leads to a rapidly progressive sense of fear and panic that further increases the respiratory rate and respiratory demand. The unpleasant perception of dyspnea leads to restriction and avoidance of functions, which in turn result in reduced oxidative capacity, muscle fiber atrophy, and deconditioning, thereby perpetuating the cycle of difficulty in breathing.
Anxiety is frequently comorbid with dyspnea caused by respiratory-derived brain suffering.4 Air hunger evokes fear and anxiety due to physiologic processes with shared activated brain regions as well as psychological and existential connections.7,39 It is helpful to ask what patients mean by reporting anxiety or shortness of breath and how they experience it. Do they experience their anxiety more in their body (eg, difficulty breathing, restlessness) or in their mind (eg, with worry, ruminations)? Certain symptom descriptions may indicate either a more psychiatric or pulmonary etiology. For example, when a patient’s ventilatory settings are lowered and the patient is tolerating them but becomes tired and anxious, respiratory fatigue is more likely than primary anxiety. However, if anticipation of the lowering of ventilatory settings leads to anxiety as soon as the settings are lowered, worsened anxiety is likely the result of a primary anxiety disorder. However, this distinction is often not possible because dyspnea and anxiety often coexist. Some clinicians will therefore try to simply distinguish between dyspnea and dyspnea-associated anxiety (DAA) because, regardless of which came first, both symptoms are often present and each may entail different interventions.7
It is also helpful to understand that some patients are more psychologically sensitive to acute respiratory changes. For example, patients with COPD and panic disorder are sensitive to even mild variations in CO2 and arterial pH. These changes activate brain regions that are involved in ventilatory control and activation of defensive behaviors, eg, panic attacks.39 On the other hand, hyperventilation can lead to respiratory alkalosis, which can also precipitate dyspnea and panic attacks.39
Which Classes of Psychotropics Can Alleviate Dyspnea (and Dyspnea-Associated Anxiety) and How Do They Work?
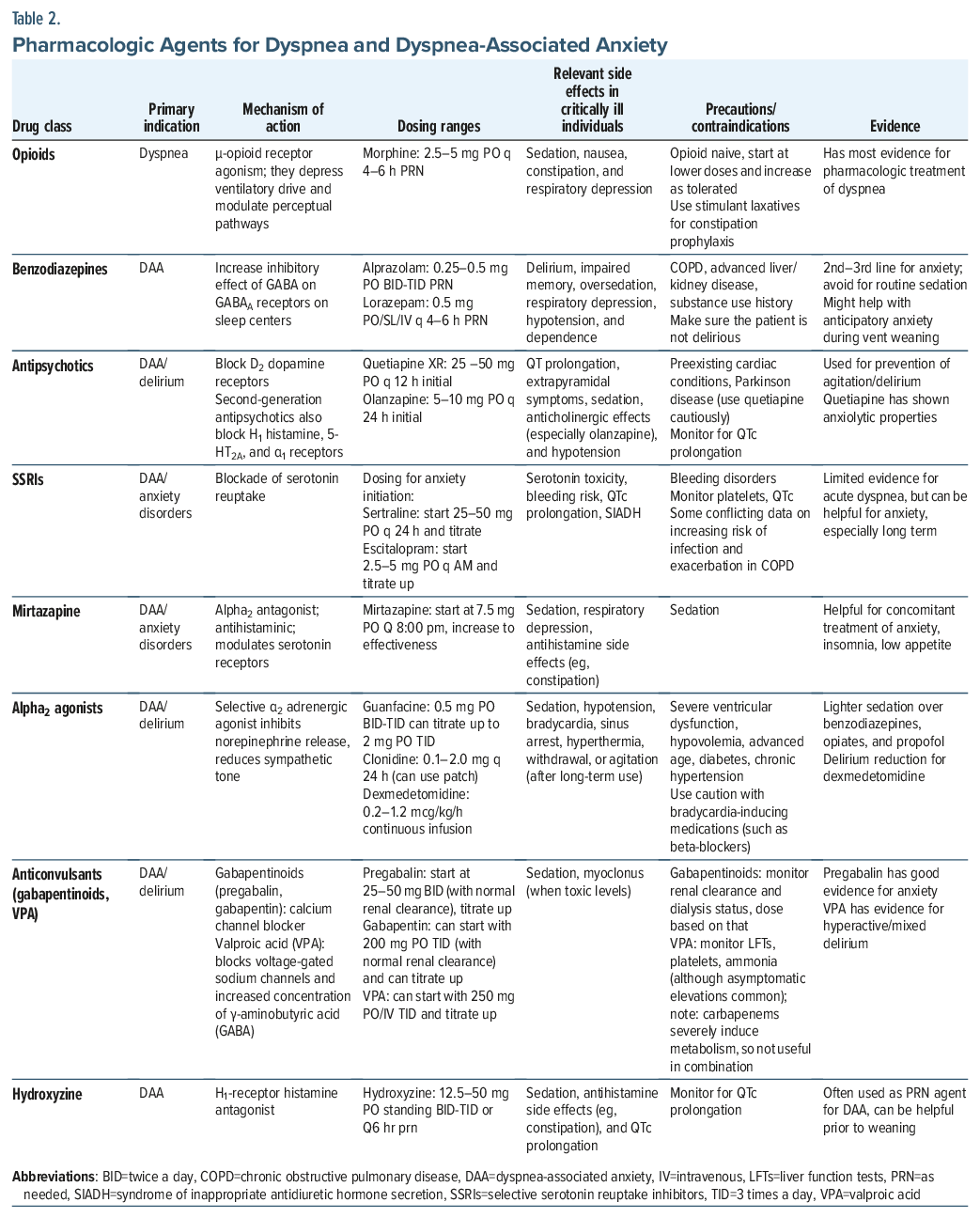
Since dyspnea is distressing and can portend clinical deterioration, it should be managed with disease-modifying interventions (eg, ventilation, transfusion) and medications (eg, diuretics for fluid overload, antibiotics for infection, β agonists and muscarinic antagonists for COPD, or oxygen for hypoxemia) in a timely fashion.40 Of note, increasing oxygen is only indicated and helpful when patients with dyspnea are hypoxemic.7 Decisions about pharmacologic interventions should consider their efficacy, tolerability, and safety41 and may be guided by tools (eg, the Beers Criteria or STOPP) and clinical judgment.42 Nonpharmacologic interventions43 (eg, cognitive-behavioral therapy [CBT], relaxation techniques, handheld fans directed towards the face [that stimulate the trigeminal nerve], music therapy, and empathic reassurance) are other important and low-risk options to consider.43,44 However, despite multiple disease-modifying interventions, many critically ill patients continue to experience dyspnea and DAA. Table 2 summarizes common pharmacologic agents used to treat dyspnea and DAA.
Opioids are the only medications with evidence to support their efficacy for relief of dyspnea and distressing “air hunger.”43 They are μ-receptor agonists that act centrally (by decreasing respiratory drive) and peripherally (by acting on pulmonary receptors) to decrease anxiety and respiratory drive.45 While myriad medications have either mixed or nonexistent evidence to treat dyspnea, they can mitigate anxiety. Benzodiazepines (BZs) (eg, midazolam, alprazolam, lorazepam) work on the γ-aminobutyric acid (GABA)A ligand-gated chloride channel that increases the inhibitory effects of GABA on sleep centers in the brain and on amygdalar fear circuitry.46 Although BZs address DAA, they can provoke delirium, drowsiness, respiratory depression, and hypotension,47 but they can be especially helpful in cases of anticipatory anxiety during weaning from ventilatory support.
Antidepressants indirectly reduce DAA. Serotonergic agents inhibit the function of the amygdala, which regulates fear and CO2 sensitivity48 and can diminish dyspnea. Time to onset of effect is an important consideration for antidepressants, limiting their indications to long-term or chronic dyspnea. 48,49
Alpha2 agonists (eg, guanfacine, dexmedetomidine, clonidine) have not been well studied as treatments for dyspnea,7 although they can attenuate sympathetic tone and hemodynamic responses, inhibit the release of norepinephrine, and terminate propagation of pain signals.50 Alpha2 agonists are often preferred over BZs given their role in the prevention and treatment of delirium in critically ill individuals.51
Antipsychotics primarily act on dopamine D2 receptors, with second-generation antipsychotics acting on 5-hydroxytryptamine receptor 2A (5-HT2A) serotonin receptors; they are used off-label for anxiety,52 and they have been used in palliative care settings for symptoms associated with dyspnea (eg, anxiety, agitation).53 Both quetiapine and olanzapine have a high affinity for 5-HT2A/AC receptors that deplete serotonin on central and peripheral chemoreceptors and might lead to hyperventilation.54 Their use should be monitored given their propensity for inducing respiratory depression and prolonging the QTc.
Hydroxyzine, an antagonist on histamine H1 receptors, calms and has an antiemetic effect on the subcortical region.55 While there is minimal evidence for the efficacy of hydroxyzine in the treatment of dyspnea in ICU patients, the US Food and Drug Administration approved its use for the treatment of generalized anxiety disorder; it can be a nonaddictive, rapid-acting, safe, and efficacious anxiolytic in critically ill individuals.7,56 Mirtazapine also has antihistaminergic activity on H1 receptors along with α2 antagonism with increased levels of serotonin and norepinephrine and 5-HT2A and 5-HT3 serotonin receptor modulation.57 Mirtazapine is used off-label for anxiety and insomnia, and it can be considered as a treatment for anxiety, especially when there is co-occurring insomnia or a diminished appetite.57
Anticonvulsants (eg, valproic acid and gabapentinoids) can mitigate anxiety, due to NMDA-antagonist activity, GABA activity, and inhibition of voltage-gated sodium-channel.58 Pregabalin, a Ca++-channel blocker, has been shown to reduce anxiety from further modulating norepinephrine and substance P.59 Good outcomes from valproic acid have been demonstrated in critical care units when used for the management of agitation and delirium,60 and it may be used as an adjunctive agent for DAA in those with agitation, delirium, or comorbid anxiety.7
How Can Psychological and Behavioral Interventions Mitigate Dyspnea?
Behavioral and psychological interventions can also manage dyspnea in conjunction with management of the underlying cardiopulmonary disease and pharmacologic strategies. Pulmonary rehabilitation is a well-studied component of the management of chronic lung disease, consisting of supervised exercise training, education, and breathing techniques.20
Pursed-lip breathing can improve breathlessness by increasing oxygenation and reducing CO2 levels,61 while diaphragmatic breathing may reduce breathlessness and improve health-related quality of life.62 Other interventions include the use of a fan that moves cool air across the face. Cold stimulation of the trigeminal nerve may lead to a decrease in the central ventilatory chemoreflex response to hypercapnia.63,64
Mindfulness, relaxation, and CBT also target dyspnea, particularly when they are associated with anxiety. CBT is a specific psychotherapy that focuses on the interplay of thoughts, emotions, and behaviors and may address catastrophic thinking and negative cognitive appraisal of dyspnea. CBT can improve anxiety, depression, and quality of life in patients with COPD65; however, there is less evidence for a direct effect of CBT on the sensation of dyspnea.66,67
Relaxation can also reduce physiological arousal and improves anxiety and inefficient breathing.68 Progressive muscle relaxation exercises have reduced dyspnea severity in patients with lung cancer69 and decreased dyspnea and anxiety levels in patients with COPD.70 Similarly, music therapy may relieve dyspnea and DAA.71
What Should a Person With Dyspnea Consider When Contemplating Being Placed On a Ventilator?
When determining code status, experiencing acute respiratory disease, or receiving treatment for a chronic respiratory disease, patients and their family members may need to decide about intubation and mechanical ventilation and the patient’s prognosis (with or without intubation). Although dyspnea is a symptom and in and of itself is not life threatening, when dyspnea occurs, it often indicates the presence of a more ominous and life-threatening condition (eg, ARDS, myocardial infarction [MI], pulmonary embolism, sepsis).11,72,73
The length and quality of life will generally be core guiding principles for patients who face these decisions. In general, intubation prolongs life, but the risks of patient-ventilator dyssynchrony, impaired communication, and increased long-term morbidity can negatively impact quality of life.74 Patients with longer life expectancies are often willing to tolerate certain types and amounts of discomfort with the goal of facilitating eventual recovery and a return to their premorbid lifestyle. In these cases, survivability and capacity for recovery can be pivotal. 75
How Can Family Members of Those With Dyspnea Cope Better and Help Their Loved One?
Quality communication is crucial to caregivers and loved ones to help them understand and build trust with providers.76 Engaging in dialog-based communication regarding dyspnea allows a common understanding and trust between the caregiver and health care provider.77
Integrating empathy and patience with caregivers can help providers understand the emotional and physical challenges being faced; in turn, this helps caregivers cope better with their own emotions.78 Educational interventions on coping with acute dyspnea (eg, relaxation techniques and CBT and resources on accessing care and support) can be helpful for caregivers.79 Caregivers can be empowered by understanding what etiologies contribute to their loved one’s dyspnea, whether it is acute or persistent, and how they can help, especially when dyspnea persists. Caregivers can reassure patients, distract them, encourage them, and help them tolerate their distress, as well as advocate for the patient when needed. Caregivers are important advocates, and they should feel empowered to communicate their loved ones’ needs and fears.
Leveraging online support groups of patients receiving critical care can also improve understanding and help better manage symptoms.80 For long-term support, directing caregivers to dyspnea support services benefits the patient and has positive effects on the caregivers by relieving the burden and improving their knowledge.81,82
What Happened to Mr A?
The psychiatric consultant diagnosed Mr A’s dyspnea and attributed it to a respiratory exacerbation in the context of ILD exacerbation and pneumonia, worsened by anemia and deconditioning. Mr A was diagnosed with DAA. He and his caregivers discussed the pathophysiology of dyspnea, and his experience was validated. The psychiatric consultant asked the pulmonary team if there were any further medical interventions that could alleviate Mr A’s dyspnea. The pulmonary team indicated that Mr A was improving with antibiotics and steroids and that instituting bilevel positive airway pressure and invasive ventilation at this time was not indicated.
Opioids were considered as a pharmacologic intervention for dyspnea, but the team wanted to avoid them for now, fearing they would suppress Mr A’s respiratory drive. The psychiatrist focused on nonpharmacological interventions for dyspnea (eg, recommending a hand-held fan to the face, slower deeper breathing, and working with a respiratory therapist) as well as embarking on pharmacologic interventions for his anxiety. Mr A was started on escitalopram (5 mg daily) for long-term management of anxiety (with the plan to raise the dose), pregabalin (25 mg by mouth 3 times/day standing) with a plan to titrate it to a therapeutic effect, and hydroxyzine (25 mg 3 times/day as needed for break-through anxiety). He was also referred to lung transplant and palliative care teams for long-term planning. Moreover, he was referred for outpatient psychiatric care for ongoing support after his hospital discharge, management of anxiety, and screening for PTSD.
Over the next several days, Mr A’s symptoms improved. He was discharged to his home on escitalopram (10 mg/day), pregabalin (50 mg 3 times/ day), and as-needed hydroxyzine. He required increased O2 support (6 L/minute) after hospital discharge.
CONCLUSION
Dyspnea is the subjective experience of difficulty breathing, as reported by patients. It is elicited by asking patients about their symptoms directly; however, in patients who are unable to communicate (eg, agitation, delirium, inability to communicate), tools are available to predict dyspnea. Once dyspnea has been identified, the emphasis shifts to clarifying its etiology. Certain tools (eg, the IMPRESS Framework and the Breathing SPACE Framework) can be used to help formulate a differential diagnosis and guide the ordering of laboratory tests that might refine the diagnosis. A variety of conditions (eg, pulmonary, cardiac, neuromuscular, hematologic/ oncologic, metabolic, systemic) can contribute to, or elicit, dyspnea. However, even when underlying causes are maximally addressed, patients can experience persistent respiratory-derived brain suffering, and a multimodal approach, which includes psychiatric consultation, is vital to address and reduce suffering. Dyspnea is a product of, and can also lead to, dysfunctional breathing, characterized by shorter exhalation, apical breathing, and a lack of end-expiratory pauses. While myriad medications (eg, opioids, BZs, selective serotonin reuptake inhibitors, α2 agonists, antipsychotics, and antiepileptic drugs) can mitigate DAA and nonpharmacologic interventions (eg, education, reassurance, relaxation, mindfulness) also reduce DAA, opioids are the only medications that can reduce sensations of dyspnea per se.
Article Information
Published Online: April 2, 2026. https://doi.org/10.4088/PCC.25f04094
© 2026 Physicians Postgraduate Press, Inc.
Submitted: September 30, 2025; accepted December 30, 2025.
To Cite: Dragonetti JD, Roy K, Cassady MK, et al. Dealing with dyspnea: strategies for patients and their family members. Prim Care Companion CNS Disord. 2026;28(2):25f04094.
Author Affiliations: Department of Psychiatry and Behavioral Medicine, Wake Forest University School of Medicine, Winston-Salem, North Carolina (Dragonetti); Department of Psychiatry, Oregon Health and Science University, Portland, Oregon (Roy); Department of Psychiatry, Brigham and Women’s Hospital, Boston, Massachusetts (Cassady); Lewis Gale Medical Center, Salem, Virginia (Braford); Psychiatric Residency, Mount Sinai Morningside/West, New York, New York (Kiriella); Department of Psychiatry, McGill University Health Care Center, Montreal, Canada (Ibrahim); Psychiatry and Behavioral Sciences, Stanford University School of Medicine, Stanford, California (Sher); Department of Psychiatry, Massachusetts General Hospital and Harvard Medical School, Boston, Massachusetts (Stern). Dragonetti, Roy, Cassady, Braford, Kiriella, and Ibrahim are co-first authors; Sher and Stern are co-senior authors.
Corresponding Author: Joseph Dragonetti, MD, Department of Psychiatry and Behavioral Medicine, Wake Forest University School of Medicine, Winston-Salem, North Carolina ([email protected]).
Financial Disclosure: None.
Funding/Support: None.
Clinical Points
- The first step in the evaluation of dyspnea involves taking a thorough history (eg, its onset, duration, and pathogenic components) and determining what is happening when dyspnea develops (eg, whether it is during exertion, at rest, in a specific body position, or associated with emotional distress).
- Concerning signs of breathlessness include an increased work of breathing (eg, a tracheal tug, Hoover sign, nasal flaring, inspiratory laryngeal groans, sternocleidomastoid activation, recession of suprasternal fossa) or signs of respiratory distress (eg, tachypnea, rapid shallow breathing, activation of accessory muscles, hyperpnea).
- Effective management of dyspnea begins with addressing the underlying issue, as treatment is typically predicated on etiology. However, pharmacologic approaches, as well as behavioral and psychological interventions (eg, mindfulness, relaxation, and cognitive-behavioral therapy), can mitigate dyspnea and dyspnea-associated anxiety.
References (82)

- Banzett RB, Lansing RW, Binks AP. Air hunger: a primal sensation and a primary element of dyspnea. Compr Physiol. 2021;11(2):1449–1483. PubMed CrossRef
- Stevens JP, Dechen T, Schwartzstein R, et al. Prevalence of dyspnea among hospitalized patients at the time of admission. J Pain Symptom Manage. 2018;56(1):15–22.e2. PubMed CrossRef
- Müller A, Mraz T, Wouters EF, et al. Prevalence of dyspnea in general adult populations: a systematic review and meta-analysis. Respir Med. 2023;218(107379):107379. PubMed CrossRef
- Schmidt M, Demoule A, Polito A, et al. Dyspnea in mechanically ventilated critically ill patients. Crit Care Med. 2011;39(9):2059–20655. PubMed
- Sato T, Tanaka S, Akazawa C, et al. Provider-documented dyspnea in intensive care unit after lung transplantation. Transpl Proc. 2022;54(8):2337–2343. PubMed CrossRef
- Mcllvaine S, Gelfand EV. Dyspnea. In: Wells B, Quintero P, Southmayd G, eds. Handbook of Inpatient Cardiology. Springer International Publishing;2020:441–456.
- Sher Y, Desai N, Sole J, et al. Dyspnea and dyspnea-associated anxiety in the ICU patient population: a narrative review for CL psychiatrists. J Acad Consult Liaison Psychiatry. 2024;65(1):54–65. PubMed CrossRef
- Demoule A, Decavele M, Antonelli M, et al. Dyspnoea in acutely ill mechanically ventilated adult patients: an ERS/ESICM statement. Intensive Care Med. 2024;50(2):159–180. PubMed CrossRef
- Berliner D, Schneider N, Welte T, et al. The differential diagnosis of dyspnea. Dtsch Arztebl Int. 2016;113(49):834–845. PubMed CrossRef
- Twaddle ML, Maxwell TL, Cassel JB, et al. Palliative care benchmarks from academic medical centers. J Palliat Med. 2007;10(1):86–98. PubMed CrossRef
- Santus P, Radovanovic D, Saad M, et al. Acute dyspnea in the emergency department: a clinical review. Intern Emerg Med. 2023;18(5):1491–1507. PubMed CrossRef
- Persichini R, Gay F, Schmidt M, et al. Diagnostic accuracy of respiratory distress observation scales as surrogates of dyspnea self-report in intensive care unit patients. Anesthesiology. 2015;123(4):830–837. PubMed CrossRef
- Sunjaya AP, Homaira N, Corcoran K, et al. Assessment and diagnosis of chronic dyspnoea: a literature review. NPJ Prim Care Respir Med. 2022;32(1):10. PubMed CrossRef
- Nishino T. Dyspnoea: underlying mechanisms and treatment. Br J Anaesth. 2011a;106(4):463–474. PubMed CrossRef
- Burki NK, Lee LY. Mechanisms of dyspnea. Chest. 2010;138(5):1196–1201. PubMed CrossRef
- Nishino T. Dyspnea and its interaction with pain. J Anesth. 2011b;25(1):157–161. PubMed CrossRef
- Wang CS, FitzGerald JM, Schulzer M, et al. Does this dyspneic patient in the emergency department have congestive heart failure?. JAMA. 2005;294(15):1944–1956. PubMed CrossRef
- Chang AS, Munson J, Gifford AH, et al. Prospective use of descriptors of dyspnea to diagnose common respiratory diseases. Chest. 2015;148(4):895–902. PubMed CrossRef
- Wahls SA. Causes and evaluation of chronic dyspnea. Am Fam Physician. 2012;86(2):173–182. PubMed
- Parshall MB, Schwartzstein RM, Adams L, et al. An official American Thoracic Society statement: update on the mechanisms, assessment, and management of dyspnea. Am J Respir Crit Care Med. 2012;185(4):435–452. PubMed CrossRef
- Karnani NG, Reisfield GM, Wilson GR. Evaluation of chronic dyspnea. Am Fam Physician. 2005;71(8):1529–1537. PubMed
- Hollenberg SM, Warner Stevenson L, Ahmad T, et al. 2019 ACC Expert consensus decision Pathway on risk assessment, management, and clinical Trajectory of patients hospitalized with heart failure: a report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2019;74(15):1966–2011. PubMed CrossRef
- Redfield MM, Borlaug BA. Heart failure with preserved ejection fraction: a review. JAMA. 2023;329(10):827–838. PubMed CrossRef
- Budhwar N, Syed Z. Chronic dyspnea: diagnosis and evaluation. Am Fam Physician. 2020;101(9):542–548. PubMed
- Gartlehner G, Wagner G, Affengruber L, et al. Point-of-care ultrasonography in patients with acute dyspnea: an evidence report for a clinical practice guideline by the American College of Physicians. Ann Intern Med. 2021;174(7):967–976. PubMed CrossRef
- Heidenreich PA, Bozkurt B, Aguilar D, et al. AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/ American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(18):e895–e1032. PubMed CrossRef
- Hollenberg SM, Stevenson LW, Ahmad T, et al. 2024 ACC Expert Consensus Decision Pathway on Clinical Assessment, Management, and Trajectory of Patients Hospitalized with Heart Failure Focused Update: A Report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2024;84(13):1241–1267. PubMed CrossRef
- Ko D, Chung MK, Evans PT, et al. Atrial fibrillation: a review. JAMA. 2025;333(4):329–342. PubMed CrossRef
- Nici L, Mammen MJ, Charbek E, et al. Pharmacologic management of chronic obstructive pulmonary disease. An official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2020;201(9):e56–e69. PubMed CrossRef
- Riley CM, Sciurba FC. Diagnosis and outpatient management of chronic obstructive pulmonary disease: a review. JAMA. 2019;321(8):786–797. PubMed CrossRef
- McCracken JL, Veeranki SP, Ameredes BT, et al. Diagnosis and management of asthma in adults: a review. JAMA. 2017;318(3):279–290. PubMed CrossRef
- Yawn BP, Han MK. Practical considerations for the diagnosis and management of asthma in older adults. Mayo Clin Proc. 2017;92(11):1697–1705. PubMed CrossRef
- Kanjee Z, Metlay JP, Moskowitz A, et al. How would you treat this patient hospitalized with community-acquired pneumonia? Grand Rounds discussion from Beth Israel Deaconess Medical Center. Ann Intern Med. 2021;174(12):1719–1726. PubMed CrossRef
- Fried TR, Fragoso CAV, Rabow MW. Caring for the older person with chronic obstructive pulmonary disease. JAMA. 2012;308(12):1254–1263. PubMed CrossRef
- Rali AS, Tran L, Balakrishna A, et al. Guide to lung-protective ventilation in cardiac patients. J Card Fail. 2024;30(6):829–837. PubMed CrossRef
- Kilickaya O, Gajic O. Initial ventilator settings for critically ill patients. Crit Care. 2013;17(2):123. PubMed CrossRef
- Demoule A, Brochard L, Dres M, et al. How to ventilate obstructive and asthmatic patients. Intensive Care Med. 2020;46(12):2436–2449. PubMed CrossRef
- Spathis A, Booth S, Moffat C, et al. The breathing, thinking, functioning clinical model: a proposal to facilitate evidence-based breathlessness management in chronic respiratory disease. NPJ Prim Care Respir Med. 2017;21(1):27.
- Sher Y. Psychiatric aspects of lung disease in critical care. Crit Care Clin. 2017;33(3):601–617. PubMed CrossRef
- Baker Rogers J, Modi P, Minteer JF. Dyspnea in Palliative Care. In: StatPearls [Internet]. StatPearls Publishing; 2025.
- Hui D, Bohlke K, Bao T, et al. Management of dyspnea in advanced cancer: ASCO guideline. J Clin Oncol. Official Journal of the American Society of Clinical Oncology. 2021;39(12):1389–1411. PubMed CrossRef
- Halli-Tierney AD, Scarbrough C, Carroll D. Polypharmacy: evaluating risks and deprescribing. Am Fam Physician. 2019;100(1):32–38. PubMed
- Banzett RB, Schwartzstein RM, Brown R. Target for today: air hunger. Am J Respir Crit Care Med. 2025.
- Bureau C, Niérat MC, Decavèle M, et al. Sensory interventions to relieve dyspnoea in critically ill mechanically ventilated patients. Eur Respir J. 2024;63(1):2202215. PubMed CrossRef
- Mahler DA. Opioids for refractory dyspnea. Expert Rev Respir Med. 2013;7(2):123–135. PubMed CrossRef
- Stahl SM. Stahl’s Essential Psychopharmacology: Prescriber’s Guide. 6th. Ed.. Cambridge University Press; 2017.
- Simon ST, Mori M, Ekström M, et al. Should benzodiazepines be used for reducing dyspnea in patients with advanced illnesses?. J Pain Symptom Management. 2023;65(3):e219–e223. PubMed CrossRef
- Higginson IJ, Bajwah S, Krajnik M, et al. Recent advances in understanding the role of antidepressants to manage breathlessness in supportive and palliative care. Curr Opin Support Palliat Care. 2025;19(2):83–94. PubMed CrossRef
- Siraj RA, Bolton CE, McKeever TM. Association between antidepressants with pneumonia exacerbation in patients with COPD: a self-controlled case series (SCCS). Thorax. 2023;79(1):50–57. PubMed CrossRef
- Fairbanks CA, Stone LS, Kitto KF, et al. alpha(2C)-Adrenergic receptors mediate spinal analgesia and adrenergic-opioid synergy. J Pharmacol Exper Ther. 2002;300(1):282–290. PubMed CrossRef
- Levenson J, Ferrando S. Special report: Prescribing psychotropic medicines for the medically ill. Psychiatr News. 2025;60(2).
- Garakani A, Buono FD, Salehi M, et al. Antipsychotic agents in anxiety disorders: an umbrella review. Acta Psychiatr Scand. 2024;149(4):295–312. PubMed CrossRef
- Dev R, Fortuno ES 3rd, Amaram-Davila JS, et al. Benefits and risks of off-label olanzapine use for symptom management in cancer patients-a case report. Ann Palliat Med. 2023;12(3):600–606. PubMed CrossRef
- Sattar SP, Gastfriend DR. Olanzapine-induced hyperventilation: case report. J Psychiatry Neurosci. 2002 Sep;27(5):360–363. PubMed CrossRef
- Ciccone CD. Hydroxyzine. In: Davis’s Drug Guide for Rehabilitation Professionals. F. A. Davis Company; 2016.
- Llorca PM, Spadone C, Sol O, et al. Efficacy and safety of hydroxyzine in the treatment of generalized anxiety disorder: a 3-month double-blind study. J Clin Psychiatry. 2002;63(11):1020–1027. PubMed CrossRef
- Jilani TN, Gibbons JR, Faizy RM, et al. Mirtazapine. In: StatPearls [Internet]. StatPearls Publishing; 2025.
- Rahman M, Awosika AO, Nguyen H. Valproic Acid. In: StatPearls [Internet]. StatPearls Publishing; 2025.
- Cardoner N, Gutiérrez-Rojas L, Saiz P, et al. Does pregabalin offer potential as a first-line therapy for generalized anxiety disorder? A meta-analysis of efficacy, safety, and cost-effectiveness. Front Pharmacol. 2025;16:1483770. PubMed CrossRef
- Crowley KE, Urben L, Hacobian G, et al. Valproic acid for the management of agitation and delirium in the intensive care setting: a retrospective analysis. Clin Ther. 2020;42(4):e65–e73. PubMed CrossRef
- Mahler DA, Selecky PA, Harrod CG, et al. American College of Chest Physicians consensus statement on the management of dyspnea in patients with advanced lung or heart disease. Chest. 2010;137(3):674–691. PubMed CrossRef
- Burge AT, Gadowski AM, Jones A, et al. Breathing techniques to reduce symptoms in people with serious respiratory illness: a systematic review. Eur Respir Rev. 2024;33(174):240012. PubMed CrossRef
- Aucoin R, Lewthwaite H, Ekström M, et al. Impact of trigeminal and/or olfactory nerve stimulation on measures of inspiratory neural drive: implications for breathlessness. Respir Physiol Neurobiol. 2023;311:104035. PubMed CrossRef
- Kako J, Kobayashi M, Oosono Y, et al. Immediate effect of fan therapy in terminal cancer with dyspnea at rest: a meta-analysis. Am J Hosp Palliat Care. 2020;37(4):294–299. PubMed CrossRef
- Ma RC, Yin YY, Wang YQ, et al. Effectiveness of cognitive behavioural therapy for chronic obstructive pulmonary disease patients: a systematic review and meta-analysis. Complement Ther Clin Pract. 2020;38:101071. PubMed CrossRef
- Williams MT, Cafarella P, Paquet C, et al. Cognitive behavioral therapy for management of dyspnea: a pilot study. Respir Care. 2015;60(9):1303–1313. PubMed CrossRef
- Livermore N, Dimitri A, Sharpe L, et al. Cognitive behaviour therapy reduces dyspnoea ratings in patients with chronic obstructive pulmonary disease (COPD). Respir Physiol Neurobiol. 2015;216:35–42. PubMed CrossRef
- von Leupoldt A, Fritzsche A, Trueba AF, et al. Behavioral medicine approaches to chronic obstructive pulmonary disease. Ann Behav Med. 2012;44(1):52–65. PubMed CrossRef
- Turan GB, Özer Z, Sariköse A. The effects of progressive muscle relaxation exercise applied to lung cancer patients receiving chemotherapy on dyspnea, pain and sleep quality: a randomized controlled trial. Eur J Oncol Nurs. 2024;70:102580. PubMed CrossRef
- Kılıç Z, Karadağ S, Tutar N. The effect of progressive relaxation exercises on dyspnea and anxiety levels in individuals with COPD: a randomized controlled trial. Holist Nurs Pract. 2023;37(1):E14–E23. PubMed CrossRef
- Huang J, Yuan X, Zhang N, et al. Music therapy in adults with COPD. Respir Care. 2021;66(3):501–509. PubMed CrossRef
- Hashmi MF, Modi P, Basit H, et al. Dyspnea. In: StatPearls. StatPearls Publishing; 2025. https://www.ncbi.nlm.nih.gov/books/NBK499965/. Accessed May 29, 2025
- Hale ZE, Singhal A, Hsia RY. Causes of shortness of breath in the acute patient: a national study. Acad Emerg Med. 2018;25(11):1227–1234. PubMed CrossRef
- Pham T, Brocgard LJ, Slutsky AS, et al. Mechanical ventilation: state of the art. Mayo Clin Proc. 2017;92(9):1382–1400. PubMed
- Elwyn G, Frosch D, Thomson R, et al. Shared decision making: a model for clinical practice. J Gen Intern Med. 2012;27(10):1361–1367. PubMed CrossRef
- Bove DG, Zakrisson AB, Midtgaard J, et al. Undefined and unpredictable responsibility: a focus group study of the experiences of informal caregiver spouses of patients with severe COPD. J Clin Nurs. 2016;25(3-4):483–493. PubMed CrossRef
- Strang S, Fährn J, Strang P, et al. Support to informal caregivers of patients with severe chronic obstructive pulmonary disease: a qualitative study of caregivers’ and professionals’ experiences in Swedish hospitals. BMJ Open. 2019;9(8):e028720. PubMed CrossRef
- Farquhar M, Penfold C, Benson J, et al. Six key topics informal carers of patients with breathlessness in advanced disease want to learn about and why: MRC phase I study to inform an educational intervention. PloS One. 2017;12(5):e0177081. PubMed CrossRef
- Barnes S, Ewing G, Farquhar M. What do informal carers of people living with breathlessness in advanced disease what to learn about ‘what to expect in the future. 2019. https://doi.org/10.1136/SPCARE-2019-
- Lassen-Greene CL, Nordness M, Kiehl A, et al. Peer support group for intensive care unit survivors: perceptions on supportive recovery in the era of social distancing. Ann Am Thorac Soc. 2021;18(1):177–182. PubMed CrossRef
- Blütgen S, Pralong A, Wilharm C, et al. BreathCarer: informal carers of patients with chronic breathlessness: a mixed-methods systematic review of burden, needs, coping, and support interventions. BMC Palliat Care. 2025;24(1):33. PubMed CrossRef
- Marques A. Pulmonary rehabilitation and family/friend caregivers: the hidden reciprocal relationship improving outcomes in chronic respiratory diseases. Expert Rev Respir Med. 2024;18(10):745–757. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!