Because this piece does not have an abstract, we have provided for your benefit the first 3 sentences of the full text.
To the Editor: Interventions in patients with schizophrenia and significant weight loss are not well studied. Here, we discuss a patient with a history of schizophrenia presenting with very limited intake of sustenance and a 40-lb weight loss. During his hospitalization, medications studied for weight gain in patients with an Axis III diagnosis were used, but there was no clinically significant benefit.
Psychostimulant Use in Schizophrenia for Treatment of Negative Symptoms and Weight Loss
To the Editor: Interventions in patients with schizophrenia and significant weight loss are not well studied. Here, we discuss a patient with a history of schizophrenia presenting with very limited intake of sustenance and a 40-lb weight loss. During his hospitalization, medications studied for weight gain in patients with an Axis III diagnosis were used, but there was no clinically significant benefit. We discuss our observations in the context of the literature findings.
Use of psychostimulants in patients with schizophrenia has been reported for the purposes of reducing weight gain caused by atypical antipsychotics1,2 and treating the negative symptoms of schizophrenia.3,4 A recent systematic review4 found that using modafinil appears to have a small effect on reducing negative symptoms in patients with schizophrenia. In patients receiving clozapine for schizophrenia, an 8-week double-blind, placebo-controlled study found that patients taking modafinil had less weight gain than a placebo group (mean change in weight = −0.84 kg and +0.1 kg, respectively; effect size = 0.622).1 Considerable evidence suggests that patients with schizophrenia are at increased risk for developing metabolic syndrome secondary to high caloric intake and poor dietary choices,5 which is further aggravated by the weight-gaining effects of atypical antipsychotics.6,7 However, there are far fewer cases in the literature characterizing schizophrenic patients with poor food intake. Currently, a lack of consensus on clinical guidelines for managing schizophrenic patients with poor food intake makes management of these patients challenging.
Case report. A 35-year old man, Mr A, was brought to the emergency department for trauma sustained from his car plunging into a divider at 80 miles per hour with the intent to kill himself. He had a history of schizophrenia (DSM-IV) and suffered from ongoing delusions that led to his suicide attempt. He was diagnosed when he was in his late teens and soon after was placed on disability and taken into the care of his family. He had a history of only partial response to medications, and as a result, displayed prominent negative symptoms.
One year before his admission, his family reported that Mr A was primarily occupied with his computer; it was discovered he was posting his delusional thoughts on conspiracy websites, which resulted in his being kicked off the server and he had to create a new ID several times. In the months leading to his hospitalization, one delusion emerged at the forefront: he believed his mind could create a powerful earthquake that would kill his family. As a result of this delusion, he created barriers so his "mind waves" would not leave the house. Eventually, out of desperation, he realized that the only way to save his family was to kill himself.
After his admission to the psychiatric hospital, his family informed us that he received several objects he had purchased online, including a knife and a noose.
During the first weeks of hospitalization, Mr A was dysphoric, asking that we kill him, as it was his right to decide whether he should live or not. Due to his refusal to eat, he often required intravenous fluids. His refusal to eat had continued during hospitalization. The clinical impression was that his low weight and the diet course were components of his core psychosis. For the first part of hospitalization, he did not eat because of his desire to die. Later, he started believing that he was unable to eat because he was "full" without other associated explanations.
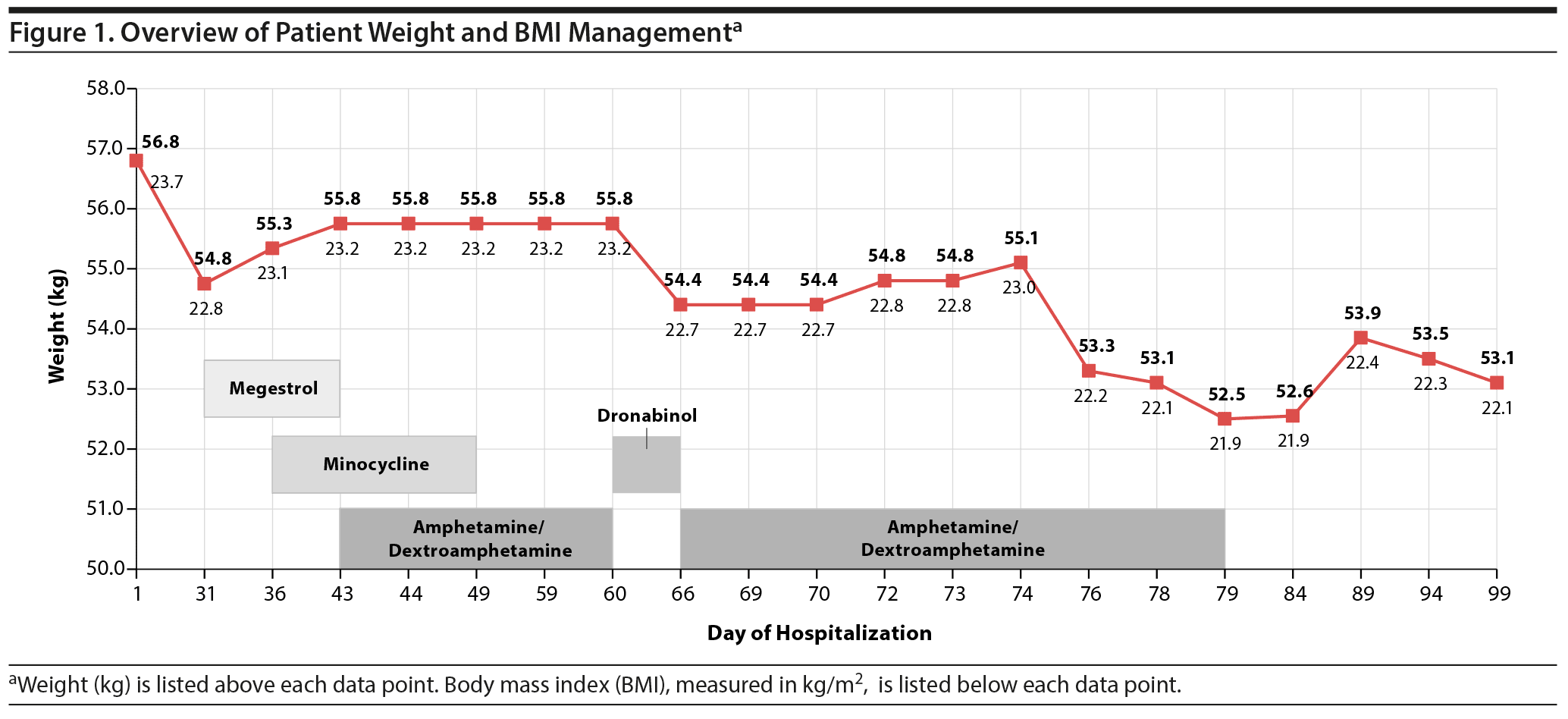
Mr A was started on several second-generation antipsychotic medications at maximum doses, but these still failed to reduce his symptoms. As a result, only lithium was continued and clozapine was started. These 2 medications were tolerated well, and as such, he was kept on this medication regimen because his suicidal thoughts resolved (Figure 1). His sustenance intake also seemed to have mildly improved on this medication regimen evidenced by his stabilized weight. Mr A appeared to have returned to baseline—he was not suicidal and was not found to be screaming at staff members to kill him. He did continue to limit his sustenance intake exclusively to Ensure Clear or Magic Cup, as he stated his preference was to eat dessert type of foods and he was too full to eat anything else.
We describe the case of a 35-year-old man with schizophrenia who had only partial response to antipsychotic medications including clozapine. Electroconvulsive therapy was not an option because of insurance and legal considerations. Thus, the treatment team was left with limited management options. Despite improvements in the acute presentation of the patient, he continued to display significant psychomotor retardation and other negative symptoms. Poor food intake was a major problem, as the patient would consistently refuse meals due to lack of appetite. Currently, there is no clinical consensus on the appropriate management of schizophrenic patients who have poor food intake. Although the use of megestrol for appetite stimulation has been used with some success in cases of HIV, cancer, and geriatric cachexia,8,9 its efficacy has not been clearly described in schizophrenic patients with poor appetite. In our 12-day trial, the use of megestrol appeared to promote some weight gain, though the patient still endorsed a poor appetite. The addition of minocycline, a tetracycline with described benefits in treatment-refractory schizophrenia,10 did not appear to improve negative symptoms or promote weight gain. Dronabinol has also been described to have appetite-stimulation effects,11 though in our short trial of dronabinol, our patient actually experienced weight loss. Due to the lack of considerable improvement in food intake while taking other appetite stimulants, our patient was given amphetamine/dextroamphetamine based on the potential for psychostimulants to improve negative symptoms of schizophrenia.4 Although dextroamphetamine is sometimes used as an appetite suppressant, we hypothesized that using it to treat the prominent negative symptoms in our patient may have a secondary impact of stimulating appetite as a result of increased motivation and psychomotor activity.
References
1. Henderson DC, Freudenreich O, Borba CPC, et al. Effects of modafinil on weight, glucose and lipid metabolism in clozapine-treated patients with schizophrenia. Schizophr Res. 2011;130(1-3):53-56. PubMed doi:10.1016/j.schres.2011.04.009
2. Roerig JL, Steffen KJ, Mitchell JE, et al. An exploration of the effect of modafinil on olanzapine associated weight gain in normal human subjects. Biol Psychiatry. 2009;65(7):607-613. PubMed doi:10.1016/j.biopsych.2008.10.037
3. Lindenmayer J-P, Nasrallah H, Pucci M, et al. A systematic review of psychostimulant treatment of negative symptoms of schizophrenia: challenges and therapeutic opportunities. Schizophr Res. 2013;147(2-3):241-252. PubMed doi:10.1016/j.schres.2013.03.019
4. Andrade C, Kisely S, Monteiro I, et al. Antipsychotic augmentation with modafinil or armodafinil for negative symptoms of schizophrenia: systematic review and meta-analysis of randomized controlled trials. J Psychiatr Res. 2015;60:14-21. PubMed doi:10.1016/j.jpsychires.2014.09.013
5. Dipasquale S, Pariante CM, Dazzan P, et al. The dietary pattern of patients with schizophrenia: a systematic review. J Psychiatr Res. 2013;47(2):197-207. PubMed doi:10.1016/j.jpsychires.2012.10.005
6. Cuerda C, Velasco C, Merchסn-Naranjo J, et al. The effects of second-generation antipsychotics on food intake, resting energy expenditure and physical activity. Eur J Clin Nutr. 2014;68(2):146-152. PubMed doi:10.1038/ejcn.2013.253
7. Gothelf D, Falk B, Singer P, et al. Weight gain associated with increased food intake and low habitual activity levels in male adolescent schizophrenic inpatients treated with olanzapine. Am J Psychiatry. 2002;159(6):1055-1057. PubMed doi:10.1176/appi.ajp.159.6.1055
8. Yeh SS, Schuster MW. Megestrol acetate in cachexia and anorexia. Int J Nanomedicine. 2006;1(4):411-416. PubMed doi:10.2147/nano.2006.1.4.411
9. Yeh SS, Wu SY, Lee TP, et al. Improvement in quality-of-life measures and stimulation of weight gain after treatment with megestrol acetate oral suspension in geriatric cachexia: results of a double-blind, placebo-controlled study. J Am Geriatr Soc. 2000;48(5):485-492. PubMed doi:10.1111/j.1532-5415.2000.tb04993.x
10. Kelly DL, Sullivan KM, McEvoy JP, et al. Adjunctive minocycline in clozapine-treated schizophrenia patients with persistent symptoms. J Clin Psychopharmacol. 2015;35(4):374-381. PubMed
11. Badowski ME, Perez SE. Clinical utility of dronabinol in the treatment of weight loss associated with HIV and AIDS. HIV AIDS (Auckl). 2016;8:37-45. PubMed
aSchool of Medicine, University of California, Irvine
bDepartment of Psychiatry, University of California, Irvine
Potential conflicts of interest: The authors report no conflicts of interest.
Funding/support: None.
Patient consent: The patient provided written consent to publish his case, and information has been de-identified to protect anonymity.
Published online: July 27, 2017.
Prim Care Companion CNS Disord 2017;19(4):17l02093
https://doi.org/10.4088/PCC.17l02093
© Copyright 2017 Physicians Postgraduate Press, Inc.
Enjoy this premium PDF as part of your membership benefits!