Intoxications Associated With Agitation, Tachycardia, Hypertension, and Fever: Differential Diagnosis,
Evaluation, and Management
Christopher J. Keary, MD; Shamim H. Nejad, MD; J. J. Rasimas, MD, PhD; and Theodore A. Stern, MD
Have you ever wondered what prompts a patient to become suddenly and severely agitated, with prominent elevations in temperature, heart rate, and blood pressure? Have you been uncertain about how best to work up the etiology of aberrations in vital signs, laboratory results, behavior, and cognition? Have you been challenged by how to initiate safe and effective treatment in patients who have similar signs and symptoms?
If you have, then the following case vignette and discussion should serve as a stimulus for further inquiry into these and other questions.
CASE VIGNETTE
Mr A, a 25-year-old man, was brought to the emergency department by police after he was found running along train tracks; he was instigating a fight with the transit police, demanding that they “Take me out for good!” The police brought Mr A to the emergency department wherein he was noted to be confused, diaphoretic, and warm to the touch. Although no identification was found on his person, police did find a pipe and an unlabeled packet with a substance in it that Mr A admitted to using; he identified it as “bath salts” (Figure 1).

In the emergency department, Mr A’s initial vital signs were heart rate of 180 beats/min, blood pressure of 230/110 mm Hg, temperature of 103.7°F (39.8°C), and respiratory rate of 48 breaths/min (with an oxygen saturation of 97% on room air). He was screaming and spitting at staff. Security was called, and Mr A was placed in 4-point locked leather restraints. Eighteen-gauge intravenous (IV) needles were placed in the antecubital fossas of both arms, and 5 mg of haloperidol and 2 mg of lorazepam were administered intravenously. One liter of normal saline was started and “stat” laboratory values were drawn.
Initial laboratory results were significant for an anion gap metabolic acidosis, mild hypokalemia, an elevated creatinine level, a transaminitis (with normal bilirubin studies), and mild leukocytosis (Table 1). Urinalysis, creatine phosphokinase, and troponin studies were within normal limits. Interestingly, a full serum and urine toxicology screen was reported as negative. Meanwhile, Mr A remained agitated; he was treated with rectal acetaminophen and repeat dosages of haloperidol (5 mg) and lorazepam
(2 mg). Fifteen minutes later, Mr A remained highly agitated (singing loudly and cursing at staff), struggling against his restraints, such that he developed blistering around his ankles. Follow-up vital signs revealed a temperature of 101.6°F (38.7°C), a heart rate of 130 beats/min, and a blood pressure of 221/95 mm Hg. To maintain adequate sedation while protecting his airway, Mr A was intubated. He was induced with 100 mg of IV lidocaine, 20 mg of IV etomidate, and 70 mg of IV rocuronium, and propofol and midazolam drips were started. Blood cultures and an arterial blood gas were obtained, as well as a urinalysis/urine culture and a lumbar puncture. Mr A was admitted to the medical intensive care unit.
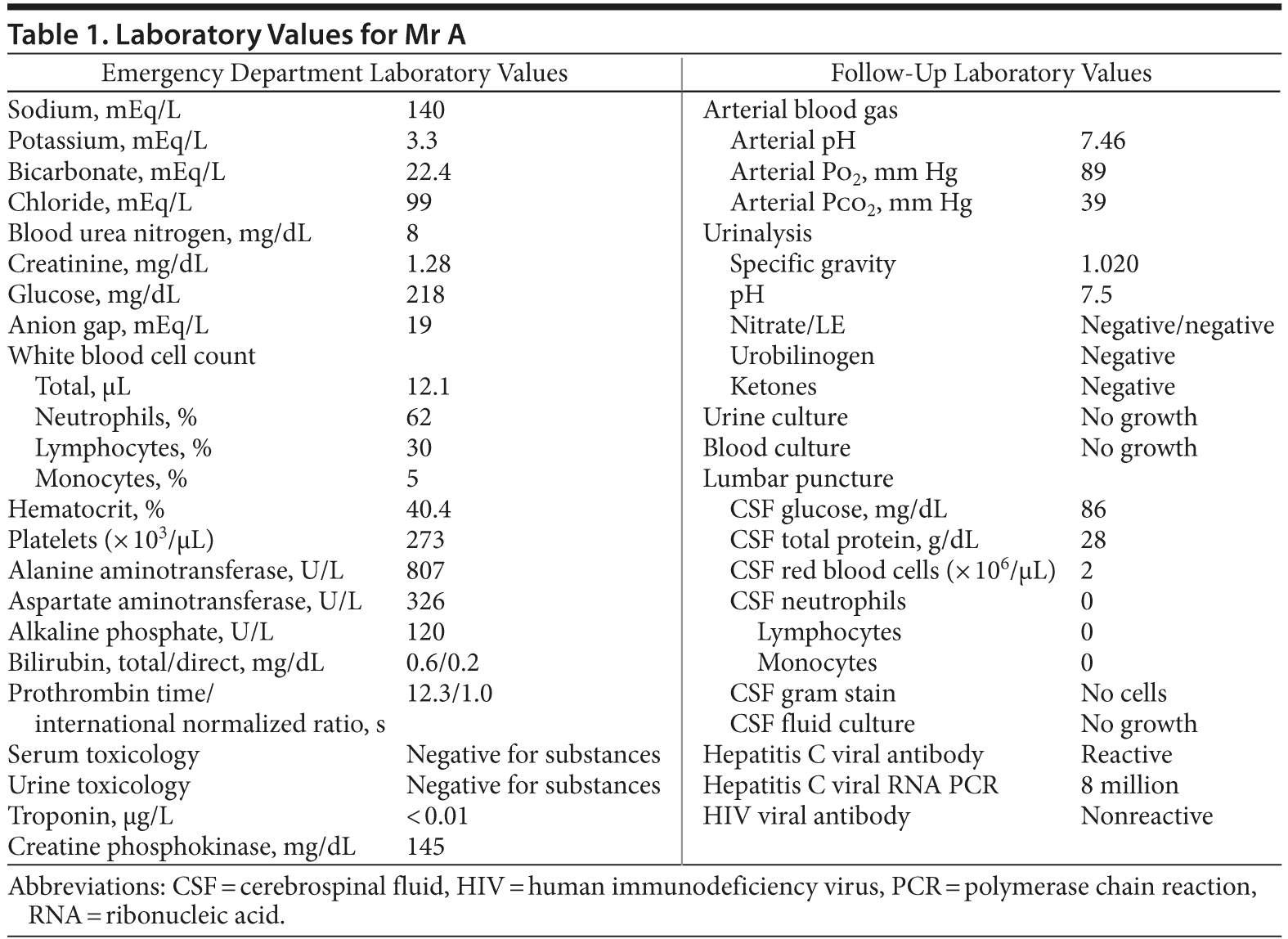
Mr A remained in the medical intensive care unit for the next 3 days. His cultures, urinalysis, and arterial blood gas levels were within normal limits, as were the results of his lumbar puncture. Although the emergency department physical examination was limited by agitation, a follow-up neurologic examination revealed normal muscle tone and reflexes; there was no clonus. Imaging revealed a normal chest X-ray and a brain magnetic resonance image with slight hypoattenuation near the frontal horn of the left lateral ventricle. On the third hospital day, Mr A was extubated; he complained of persistent anxiety and “restlessness” and was constantly pacing. On the evening after his extubation, he reported hearing noises “like a train right outside my room.”
WHAT IS THE DIFFERENTIAL DIAGNOSIS
FOR THE SUDDEN ONSET OF AGITATION, HYPERDYNAMIC VITAL SIGNS, AND FEVER?
While not all patients with agitation or aggression have an altered mental status, the combination of agitation, autonomic instability, and fever is almost always secondary to a medical etiology. Broadly viewed, the causes include substance intoxications, toxidromes (medication-related syndromes), and medical conditions (Table 2).
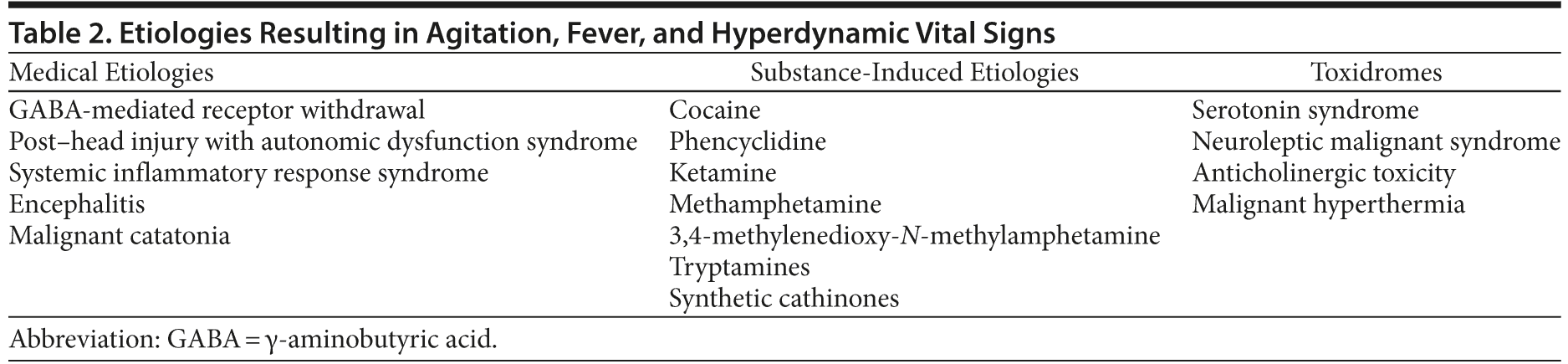
Medical Conditions
A variety of medical conditions may lead to the triad of signs and symptoms described above. One of the most common is a γ-aminobutyric acid (GABA)–mediated receptor withdrawal syndrome. Long-term exposure to GABAA agents (such as benzodiazepines, barbiturates, ethanol) results in several adaptive changes in neurotransmitter systems (including those on GABA receptors and glutamate receptors) and in noradrenergic activity.1 Chronic exposure to GABAA agents or drugs results in reduced GABA activity and increased glutamatergic action.2 In the setting of an abrupt discontinuation of these agents, the decrease in GABA activity leads to restlessness, anxiety, panic, tremor, nausea, and malaise. In addition, neuroadaptive changes result in an unopposed hyperglutamatergic state and neuroexcitability that activates the locus ceruleus and causes the release of norepinephrine. This release of norepinephrine results in tachycardia, hypertension, diaphoresis, tremor, and fever.3 The hyperglutamatergic state activates dopamine release via the mesolimbic tract and leads to the development of paranoia, visual and tactile hallucinations, confusion, and agitation. The onset, timing, and severity of symptoms depend on the medication or drug that the patient is withdrawing from, the duration and severity of its use, and the baseline physiologic reserve of the patient.
An autonomic dysfunction syndrome can develop with a traumatic brain injury, hydrocephalus, brain tumor, subarachnoid hemorrhage, or intracerebral hemorrhage. In autonomic dysfunction syndrome, altered autonomic activity results in hypertension, fever, tachycardia, tachypnea, and pupillary dilation (often in association with an encephalopathy). The cause of autonomic dysfunction syndrome is dysregulation of the autonomic nervous system from injury to one or more parts of the central nervous system.4–6 Cortical areas that influence activity of the hypothalamus include the orbitofrontal and the anterior temporal and insular regions. Subcortical areas that influence the hypothalamus include the amygdala, the periaqueductal gray, the nucleus of the tractus solitarius, the cerebellar uvula, and the cerebellar vermis. Damage to these areas releases control of vegetative functions and results in dysregulation of overall autonomic balance. The orbitofrontal region is believed to promote parasympathetic activity and to inhibit sympathetic activity. When damaged, dysregulation may cause cortically provoked release of adrenomedullary catecholamines during episodes of autonomic dysfunction, resulting in elevated blood pressure, tachycardia, and tachypnea.7
Systemic inflammatory response syndrome (SIRS) is a condition related to systemic inflammation, organ dysfunction, and organ failure. It is a subset of a cytokine storm, in which there is abnormal regulation of various proinflammatory mediators. SIRS is also closely related to sepsis, in which a patient satisfies criteria for SIRS and has a suspected or proven infection. The American College of Chest Physicians and the Society of Critical Care Medicine define SIRS as a condition having 2 or more of the following parameters: fever > 100.4°F (38.0°C) or < 96.8°F (36.0°C), a heart rate > 90 beats/min, a respiratory rate > 20 breaths/min, an arterial carbon dioxide tension < 32 mm Hg, and an abnormal white blood cell count (> 12,000/μL or < 4,000/μL or > 10% immature [band] forms).8 The condition almost always is accompanied by encephalopathy, and altered mental status is often the first symptom noted prior to fever or the development of hyperdynamic vital signs. The causes of SIRS are broadly classified as infectious or noninfectious. When SIRS is due to an infection, it is considered sepsis. Common noninfectious causes of SIRS include trauma, burns, pancreatitis, ischemia, and hemorrhage.
Encephalitis (an inflammation of the brain parenchyma) may also present with neuropsychiatric disturbances
(eg, severe headache, fever, altered mental status, confusion, agitation, personality changes, seizures, hallucinations, problems with speech or hearing). While bacterial, fungal, and autoimmune disorders may produce encephalitis, most cases are viral in origin, with herpes simplex virus being the most common cause.9 Certain paraneoplastic causes of limbic encephalitis (such as N-methyl-d-aspartate receptor encephalitis) may have prominent neuropsychiatric symptoms (with initial complaints of anxiety and mood changes, then progressing to severe behavioral and personality disturbances, delusions, paranoid ideation, and hallucinations). Patients may become markedly agitated, with autonomic dysfunction (often leading to intubation and mechanical ventilation given poor response to medications administered for the control of agitation).10
Malignant catatonia is a rare and potentially fatal neuropsychiatric syndrome associated with both medical and psychiatric conditions. The clinical presentation typically begins with a nonspecific prodrome (characterized by insomnia and mood changes) that lasts up to 2 weeks. This presentation is usually followed in rapid order by severe anxiety, delusions, hallucinations, and agitation.11 Motoric symptoms include rigidity, waxy flexibility, mitgehen, gegenhalten, and severe nonpurposeful hyperkinetic movements. Autonomic disturbances (including high fever, tachycardia, labile blood pressure, urinary retention or incontinence, constipation, and acrocyanosis) are also present.11
Endocrine-induced causes of agitation, autonomic instability, and fever are less common and are often overlooked. Thyrotoxic crisis, or thyroid storm, is a rare and potentially life-threatening exacerbation of hyperthyroidism seen most often with acute medical illness, surgical intervention on the thyroid, or recent administration of radioactive iodine to a patient with untreated hyperthyroidism. Sudden surges in thyroid hormone result in overactivation of thermogenic and metabolic feedback loops and can result in mortality via cardiac failure, arrhythmia, or hyperthermia. Pheochromocytomas (tumors derived from the sympathetic ganglia) can result in increased sympathetic tone leading primarily to hypertension and tachycardia. While sweating and panic attacks are common, fever and delirious agitation are not; presence of these manifestations warrants creation of an expanded differential.
Substance Intoxication: Etiologies
Multiple drugs of abuse (eg, phencyclidine; ketamine; cocaine; methamphetamines; 3,4-methylenedioxy-N-methylamphetamine [MDMA]; tryptamines; psilocybin; and lysergic acid diethylamide, in addition to synthetic cathinones or bath salts) can contribute to a constellation of fever, autonomic dysfunction, psychosis, and agitation. Broadly viewed, these drugs have effects on dopaminergic, noradrenergic, and serotonergic activity.
Most currently employed routine drug screens do not detect synthetic cathinones. Consequently, these drugs appeal to some substance abusers who are subject to mandatory drug testing. While synthetic cathinones go undetected by routine screens, some hospitals and commercial drug-testing laboratories are beginning to offer specialized synthetic cathinone testing (with both serum and urine toxicology screening). At many institutions, these laboratory studies are sent out for processing, thereby delaying receipt of results and decreasing their usefulness in acute care settings.12
Toxidromes
Several toxidromes (eg, serotonin syndrome, anticholinergic toxicity, neuroleptic malignant syndrome, malignant hyperthermia) should be considered in the patient with hyperdynamic vital signs, altered mental status, and fever.13
While the diagnosis of malignant hyperthermia is clearer following the administration of inhalational anesthetics, the recognition of other toxidromes, particularly in the patient with an altered sensorium, may not be as simple. The recent administration of medications and/or drugs of abuse with serotonergic activity may induce a serotonin syndrome, characterized by encephalopathy, hyperreflexia,
myoclonus, diaphoresis, and mydriasis. Neuroleptic malignant syndrome, a medication-induced form of malignant catatonia, is associated with mental status changes, “lead-pipe” rigidity, and autonomic instability. Medications or drugs of abuse with significant anticholinergic properties may lead to anticholinergic excess (typically with the classic triad of altered mental status, dry mucous membranes, and mydriasis).13
WHAT TOXIDROMES AND INTOXICATIONS
CAN PRESENT WITH FEVER, ALTERED MENTATION, AND AUTONOMIC INSTABILITY?
While the differential diagnosis for this syndrome is broad, a toxicologic cause is most likely when the onset is sudden. Assessment of each of these elements is useful.
Fever
A pathophysiologic distinction between fever and hyperthermia may guide consideration of etiologies. Fever depends on the alteration of the hypothalamic set point, while hyperthermia describes an imbalance between heat-generating and heat-dissipating mechanisms.14 In the absence of an inflammatory process or trauma that can alter hypothalamic function directly, assessment of an elevated temperature may turn to hyperthermic causes, many of which are toxicologic. Adrenergic excess, a consequence of toxicity from psychostimulants, serotonergic agents, and withdrawal from sedatives and ethanol, elevates metabolic indices and enhances neuromuscular activity (such that heat is generated). When endogenous cooling mechanisms are overwhelmed by the effects of monoamines on the cardiovascular and neuromuscular systems, the result can be an elevated body temperature. Excess heat generation through sustained muscle contraction coupled with hypothalamic dysfunction is the driving force behind the high temperatures typical of neuroleptic malignant syndrome.15 Inadequate dissipation is the core regulation problem in anticholinergic excess, wherein secretory mechanisms are impaired. Since there is no primary driving force for heat generation in anticholinergic delirium, fever is mild and may be absent if environmental factors are not at play.
Several acute mental status changes are suggestive of toxicologically mediated delirium (eg, the energy and volume of behavioral outputs). Abundant and loud speech coupled with a congruent excess of physical movements implies neural stimulation (eg, with intoxication from phencyclidine, serotonergic agents, phenethylamines, other ephedrine derivatives). Afflicted individuals will often display a wide-eyed gaze (that belies fear) and vigilance for visual inputs of all kinds—real, illusory, and hallucinatory. Although agitated, those suffering from delirious withdrawal from sedatives or ethanol typically do not move or speak as quickly or as voluminously. They will make many sounds and movements that indicate discomfort, but they tend to lack an obvious purpose that stimulant-toxic individuals demonstrate. Patients with neuroleptic malignant syndrome will also be slow thinking. In contrast to the hyperdynamic state of the muscles and cardiovascular system, nervous system (both central and peripheral) activity is depressed.15 Responses are typically slow and speech is full of confused, heavy effort. Patients with anticholinergic excess, by contrast, will mutter rapidly, but their speech is also difficult to understand (the low amplitude and high frequency of lip movements in these patients frequently mirror the movements of their fingers). Such individuals’ consciousness waxes and wanes; when left unstimulated, they will drift off to sleep, but when roused, they are fidgety and do not calm with verbal interventions. Of note, a heart rate increase of > 20 beats/min can be seen as an individual with anticholinergic excess switches from quiet stupor to delirious arousal. This link between mental activity and cardiovascular indices propels our discussion to the last part of the differential diagnostic triad.
Autonomic Instability
Autonomic instability implies lability in the measures of cardiac output and neuromuscular regulation, though this term is most often used to describe hyperdynamic states. Careful attention to the pattern of derangements can help to hone in on particular toxicologic causes.16 As mentioned above, patients with a head injury will often display autonomic asynchrony, with high blood pressure and a low heart rate, whereas most toxidromes drive rate and pressure congruently. Stimulants (like the synthetic cathinones) can cause extreme elevation of both parameters. Agents that are primarily serotonergic also elevate both parameters, though less severely.16 Patients with anticholinergic excess typically show milder elevations of both measures; decreased vagal activity often directly increases heart rate, whereas blood pressure elevates secondary to agitation. As mentioned above, heart rate reactivity in response to external stimulation is common.16 Withdrawal from sedatives and ethanol sometimes leads to more true autonomic instability with wider numerical fluctuations, and cardiovascular indices may remain elevated even when neurologic status has improved. Such autonomic instability is evident in cases of neuroleptic malignant syndrome, a state that has been described as a form of malignant catatonia in which patients may be virtually unresponsive but have life-threatening shifts in autonomic measures.17 In most instances of toxin-mediated cardiovascular changes, however, mental speed and activity tend to roughly parallel the degree of stimulation to autonomic and peripheral nervous systems (as reflected in the blood pressure, heart rate, motor display, muscle tone, and deep tendon reflex activity).18
WHAT ARE BATH SALTS
AND HOW DO SUCH INTOXICATIONS PRESENT?
Psychoactive bath salts (alternatively called “plant food,” “potpourri,” or “incense” to skirt legislative action) are increasingly used drugs of abuse; most contain synthetic cathinones, a class of central nervous system stimulants that belong to the phenethylamine family. Absorbed after oral, intranasal, IV, or rectal routes of administration, these substances most often are prepared as a white powder or as small crystals; they are advertised as alertness enhancers, aphrodisiacs, or legal cocaine replacements. Use of synthetic cathinones (such as the well-studied agent mephedrone) was first observed in 2007 among night club attendees in Europe, and these agents have become increasingly recognized as dangerous drugs of abuse.19 Evidence for their increasing prevalence in the United States has been reflected by more frequent calls to poison control centers, by nationwide forensic laboratory reports, and by publications regarding emergency department visits.20–22 Results from these publications suggest that users of synthetic cathinones tend to be young males often with comorbid substance use disorders. This emerging evidence resulted in an emergency federal order in the Fall of 2011 to categorize the 3 major synthetic cathinones as schedule 1 agents.23
Synthetic cathinones represent the most common ingredients in psychoactive bath salts and are thought to be the mediators of the stimulatory effects for which the drug is used. Naturally occurring cathinones exist in the leaves of khat, a flowering plant grown in the Horn of Africa and on the Arabian Peninsula; it is highly sought after for its stimulant properties. This class of compounds, belonging to the phenethylamine family, contains common stimulants (such as amphetamine, methamphetamine, MDMA) that mediate their stimulant effects through blockade of epinephrine and norepinephrine reuptake.24 Pharmacologic modification (of side groups to the basic structure) can also confer varying degrees of direct and indirect dopaminergic and serotonergic activity. Independent studies (in rodent models) of the most commonly found cathinones in psychoactive bath salts have detected increases in peak dopamine and serotonin levels, as well as norepinephrine levels consistent with a sympathomimetic side effect profile.25–27
Although epidemiologic studies of emergency department patients intoxicated with psychoactive bath salts have allowed for the characterization of some of their active compounds such as mephedrone, methylone, and methylenedioxypyrovalerone (MDPV), the contents of these synthetically manufactured bath salts vary. A 2011 study of bath salts purchased online found that 6 of 7 compounds contained additives (such as caffeine or synthetic drugs of the piperazine class), as well as a number of other agents (eg, MDMA, lidocaine, methadone, zolpidem).28 Moreover, a 2010 study29 of bath salts purchased online from the same source at 3 separate points in time found significant variation in the concentration of the psychoactive compounds (with some products containing no detectable psychoactive molecules).
Animal and human studies on mephedrone, have provided convincing evidence for the abuse potential of synthetic cathinones. Studies of mephedrone usage by rodents revealed a preference for self-administration of the drug (with releases of dopamine in striatal pathways following drug administration that approached the potency of methamphetamine).30 In a structured telephone survey of 947 polysubstance users in the United Kingdom, numerous subjects described bingeing on mephedrone, and 20% met criteria for mephedrone dependence by DSM-IV criteria.31,32 More than half of mephedrone users developed tolerance after continued use31 and withdrawal symptoms (eg, tiredness, nasal congestion, difficulty concentrating, depression/anxiety, irritability, continued cravings to use) after abstinence. Comparable research on the addictive potential of other synthetic cathinones, such as MDPV, is still required; however, given the frequency of mephedrone use in the United States, it is reasonable to expect analogous human psychobehavioral patterns.
The signs and symptoms of bath salt intoxication reflect a known neurochemical profile: adrenergic excess (eg, tachycardia, sweating, hypertension, tachycardia, mydriasis, hyperthermia), dopaminergic excess (eg, psychosis, confusion), and serotonergic excess (eg, myoclonus, nausea, agitation, confusion). Adverse side effects are commonly reported with bath salt usage and include psychiatric (eg, hallucinations, paranoia, agitation/aggression), neurologic (eg, myoclonus, confusion), and cardiac (eg, tachycardia, hypertension, chest pain) effects.19 Agitation is the adverse reaction that most often results in emergency department presentation, and it is often exacerbated by concurrent usage of other drugs of abuse (eg, alcohol, cocaine). Recently, dramatic examples of disorganized and agitated behavior (such as aggression or self-injury) have received notoriety in the popular press due to injury and loss of life. Several studies have described delusions of persecution and auditory hallucinations from binge usage even in those without a history of psychosis.33–35 Psychosis tends to remit between 1–5 days after use, but it can be protracted.
Evidence of organ damage and other life-threatening sequelae of bath salts use is emerging. Renal failure can occur in a pattern suggestive of acute tubular necrosis and hypothesized to be mediated via severe renal tubular vasospasm and elevated creatine phosphokinase.36 Systemic monoamine excess can result in seizure activity or an anion gap metabolic acidosis that reflects excessive anaerobic metabolism. Three case reports have noted severe hyponatremia associated with mephedrone usage, with 2 cases resulting in cerebral edema and death.37 One case report of isolated MDPV exposure resulted in fulminant hepatic failure, among other injured organs, and disseminated intravascular coagulation.38 Similarly, a death related to isolated MDPV usage (thought to be mediated by an excited delirium followed by a pulseless electrical activity arrest, multiorgan failure, and anoxic brain injury) has also been reported.39 The majority of deaths related to the use
of synthetic cathinone usage have been due to aggression/self-harm in the setting of severe agitation and psychosis.
Diagnosis of exposure to synthetic cathinones can be a challenge, as routine hospital enzyme-linked immunosorbent assay–based drug screening performed in most emergency departments will not detect these agents. Moreover, synthetic cathinones may result in a false-positive methamphetamine screen. Gas chromatography–mass spectroscopy screening remains the gold standard for detection of active molecules in samples of urine and serum and would allow for a definitive diagnosis. As with most toxic presentations, however, laboratory testing is of limited diagnostic value due to reporting delays; prompt recognition of the clinical toxidrome and aggressive management are of highest importance.
HOW SHOULD FEVER, ALTERED MENTATION, AND AUTONOMIC INSTABILITY INDUCED BY INTOXICATIONS AND TOXIDROMES BE MANAGED?
When a patient displays these toxidromic features, the most important intervention is rapid and adequate sedation, with benzodiazepines representing the core class of agents suited to that purpose. These agents are the drugs of choice because they decrease excesses in heart rate, blood pressure, neural stimulation, and muscular activity—all with a wide margin of safety. Contrary to popular belief, unless used in combination with other potent sedatives, benzodiazepines do not produce dangerous decreases in blood pressure, heart rate, or respiratory drive. They can usually be dosed such that airway protection can be preserved while adequate sedation for safe and effective treatment is achieved. Without IV access in place, lorazepam and midazolam are useful intramuscular medications for initial treatment. Once an IV is in place, diazepam is effective for rapid titration, because the full effect of each dose occurs within 5 minutes, thus allowing for repeat dosing without “overshooting”—a problem with lorazepam, whose peak sedative efficacy can be delayed up to 30 minutes after an IV dose. With a set of elevated vital signs and an agitated toxic presentation, benzodiazepines should be given aggressively until the patient achieves a mildly sedated state (calm), which can then be maintained with as-needed doses of lorazepam thereafter. This intervention will treat hypertension, reduce tachycardia, prevent seizures, protect against physical violence, and reduce muscle hyperactivity that drives fever and leads to rhabdomyolysis and renal failure. In support of this pharmacologic cornerstone of care, use of high-rate IV fluid therapy and external cooling measures should also be implemented. The combination of sedation, fluids, and cooling will reverse metabolic acidosis and protect from further muscular and hepatorenal injury. If escalating doses of benzodiazepines are ineffective (see Gold et al40 for a protocol involving high-dose diazepam in severe cases of ethanol withdrawal), augmentation with barbiturates or propofol may be necessary. Sedatives are preferable to physical restraints, as patients may suffer from morbidity and even mortality from ongoing hyperthermia and muscle breakdown if allowed to struggle instead of being calmed through adequate treatment. This management plan is essentially the toxicologic management of serotonin syndrome—a diagnosis for which most patients with bath salt toxicity will meet the criteria.41,42
In the latter stages of medical hospitalization for bath salt toxicity, once the autonomic storm has abated and reflexes and muscle tone have normalized, there may be a role for the use of antipsychotic medications. The synthetic cathinones are becoming notorious for their tendency to cause psychosis, dysphoria, and irritable unrest that extends beyond the duration of somatic toxicity. It is not uncommon for patients to recover physically and not require benzodiazepines to manage autonomic and neuromuscular symptoms, but also suffer from paranoia and hallucinosis that responds best to dopamine blockade. This feature of bath salts makes caring for those who have used them challenging. Psychiatric treatment centers may be reluctant to care for these individuals even after physical manifestations of toxicity have resolved. It is therefore important for physicians and caregivers of different specialties to be familiar with the presentation of this toxidrome and to work collaboratively to manage different phases of illness caused by these novel drugs of abuse.
CLINICAL VIGNETTE CONTINUED
Mr A gave an expanded history of the events leading up to his admission. He reported having bipolar disorder and a long history of substance use (eg, opiates, cocaine, cannabis) with resulting hepatitis C infection. He had been living at a residential drug treatment program for several months wherein he was subjected to intermittent (random) urine drug testing. He learned about psychoactive bath salts from others staying there who used bath salts together on a regular basis and avoided “dirty urines.” He described having inhaled bath salts 3 times over the preceding week without unwanted effects. Regarding his latest drug ingestion, he said:
I remember running down the street and feeling like everyone was staring at me. . . . I thought, “This is never going to go away; I’ve got to kill myself. . . .” That’s why I was on the train tracks. . . . When the cops showed up I thought that if I went for their gun they would kill me. I remember fighting like an animal. I think I would have killed someone if there hadn’t been so many of them.
Mr A had no history of psychosis or current mood symptoms; anxiety was his main complaint. He was started on lorazepam (1 mg by mouth every 6 hours as needed for anxiety/agitation) with good effect. Lorazepam was tapered and discontinued before his discharge on hospital day 4. He was also started on gabapentin (300 mg 3 times/day) to target anxiety. No further psychosis was observed during his admission; he remained afebrile and hemodynamically stable. Elevated transaminase levels were attributed to a combination of hepatitis C infection and potential direct toxic effect of ingestion. He was discharged to his residential drug treatment program.
CONCLUSION
The above case and discussion illustrates how a clinical triad of delirious agitation, autonomic instability, and hyperthermia (induced by sympathomimetic drug intoxication, specifically bath salts) can develop. While the above clinical triad can also be induced by a variety of toxidromes and urgent medical conditions, clinicians should be aware of the possibility of substance-induced etiologies even in the absence of a reliable history or clear toxicologic data. The emergence of synthetic cathinones under the street name bath salts represents an increasingly prevalent drug class of abuse, with observational studies and animal experiment data suggesting high addiction potential. These substances can carry severe psychiatric (agitation, psychosis), neurologic (confusion), and cardiac (autonomic instability) sequelae, as well as risk for organ injury and death. Achieving rapid and effective sedation represents the most urgent intervention along with evaluation for evidence of renal or hepatic injury and the need for external cooling measures. Psychiatric complications from synthetic cathinone intoxication, including hallucinations and delusions of persecution, can be prolonged, necessitating management with antipsychotic medications or psychiatric hospitalization once the patient is medically stabilized.
Acknowledgment: The authors acknowledge J. Ward Donovan, MD, Penn State/PinnacleHealth Toxicology Program, Harrisburg, Pennsylvania, for contributions from clinical expertise related to his work in medical toxicology and for education of the author (J.J.R.) through bedside care
of poisoned patients. Dr Donovan has given written permission for mention of his input and reports no relevant conflicts of interest.
REFERENCES
1. Addolorato G, Leggio L, Abenavoli L, et al; Alcoholism Treatment Study Group. Neurobiochemical and clinical aspects of craving in alcohol addiction: a review. Addict Behav. 2005;30(6):1209–1224. doi:10.1016/j.addbeh.2004.12.011 PubMed
2. Littleton J. Neurochemical mechanisms underlying alcohol withdrawal. Alcohol Health Res World. 1998;22(1):13–24. PubMed
3. Clapp P, Bhave SV, Hoffman PL. How adaptation of the brain to alcohol
leads to dependence: a pharmacological perspective. Alcohol Res Health. 2008;31(4):310–339. PubMed
4. Baguley IJ, Heriseanu RE, Cameron ID, et al. A critical review of the pathophysiology of dysautonomia following traumatic brain injury.
Neurocrit Care. 2008;8(2):293–300. doi:10.1007/s12028-007-9021-3 PubMed
5. Baguley IJ, Heriseanu RE, Nott MT, et al. Dysautonomia after severe traumatic brain injury: evidence of persisting overresponsiveness to afferent stimuli. Am J Phys Med Rehabil. 2009;88(8):615–622. doi:10.1097/PHM.0b013e3181aeab96 PubMed
6. Srinivasan S, Lim CC, Thirugnanam U. Paroxysmal autonomic instability with dystonia. Clin Auton Res. 2007;17(6):378–381. doi:10.1007/s10286-007-0428-x PubMed
7. Blackman JA, Patrick PD, Buck ML, et al. Paroxysmal autonomic instability with dystonia after brain injury. Arch Neurol. 2004;61(3):321–328. doi:10.1001/archneur.61.3.321 PubMed
8. Bone RC, Balk RA, Cerra FB, et al; ACCP/SCCM Consensus Conference Committee. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. 1992. Chest. 2009;136(suppl 5):e28. PubMed
9. Nejad SH, Roffman JL, Kueppenbender K, et al. Patients with neurologic conditions II: movement disorders, multiple sclerosis, and other neurologic conditions. In: Stern TA, Fricchione GL, Cassem EH, et al, eds. Massachusetts General Hospital Handbook of General Hospital Psychiatry. 6th ed. Philadelphia, PA: Saunders Elsevier; 2010:255–272. doi:10.1016/B978-1-4377-1927-7.00020-0
10. Dalmau J, Tüzün E, Wu HY, et al. Paraneoplastic anti-N-methyl-d-aspartate receptor encephalitis associated with ovarian teratoma. Ann Neurol. 2007;61(1):25–36. doi:10.1002/ana.21050 PubMed
11. Fink M, Taylor MA. The many varieties of catatonia. Eur Arch Psychiatry Clin Neurosci. 2001;251(suppl 1):I8–I13. doi:10.1007/PL00014200 PubMed
12. Synthetic Cathinones (Bath Salts): An Emerging Domestic Threat.
US Dept of Justice Web site. http://www.justice.gov/archive/ndic/pubs44/44571/44571p.pdf. Updated June 26, 2012. Accessed
February 1, 2013.
13. Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med. 2005;352(11):
1112–1120. PubMed doi:10.1056/NEJMra041867
14. McAllen KJ, Schwartz DR. Adverse drug reactions resulting in hyperthermia in the intensive care unit. Crit Care Med. 2010;38(suppl 6):S244–S252. doi:10.1097/CCM.0b013e3181dda0d4 PubMed
15. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome.
Am J Psychiatry. 2007;164(6):870–876. doi:10.1176/appi.ajp.164.6.870 PubMed
16. Donovan JW, Burkhart KK, Brent J. General management of the critically poisoned patient. In: Brent J, Wallace KL, Burkhart KK, et al, eds. Critical Care Toxicology: Diagnosis and Management of the Critically Poisoned Patient. Philadelphia: Elsevier Mosby; 2005:1–11.
17. Philbrick KL, Rummans TA. Malignant catatonia. J Neuropsychiatry Clin Neurosci. 1994;6(1):1–13. PubMed
18. Flomenbaum NE, Goldfrank LR, Hoffman RS, et al. Principles of managing the poisoned or overdosed patient. In: Flomenbaum NE, Goldfrank LR, Hoffman RS, et al, eds. Goldfrank’s Toxicologic Emergencies. 8th ed.
New York: McGraw-Hill; 2006:42–50.
19. Winstock AR, Mitcheson LR, Deluca P, et al. Mephedrone, new kid for the chop? Addiction. 2011;106(1):154–161. PubMed doi:10.1111/j.1360-0443.2010.03130.x
20. Spiller HA, Ryan ML, Weston RG, et al. Clinical experience with and analytical confirmation of “bath salts” and “legal highs” (synthetic cathinones) in the United States. Clin Toxicol (Phila). 2011;49(6):499–505. doi:10.3109/15563650.2011.590812 PubMed
21. Benzie F, Hekman K, Cameron L, et al. Emergency department visits after use of a drug sold as “bath salts”: Michigan, November 13, 2010–March 31, 2011. MMWR. 2011;60(19):624–627.
22. Forrester MB. Synthetic cathinone exposures reported to Texas poison centers. Am J Drug Alcohol Abuse. 2012;38(6):609–615. doi:10.3109/00952990.2012.677890 PubMed
23. US Drug Enforcement Administration. Schedules of controlled substances: temporary placement of three synthetic cathinones into schedule 1.
Fed Regist. 2011;7(6):65371–65375.
24. Hill SL, Thomas SHL. Clinical toxicology of newer recreational drugs.
Clin Toxicol. 2011;49(8):705–719. doi:10.3109/15563650.2011.615318 PubMed
25. Kehr J, Ichinose F, Yoshitake S, et al. Mephedrone, compared with MDMA (Ecstasy) and amphetamine, rapidly increases both dopamine and 5-HT levels in nucleus accumbens of awake rats. Br J Pharmacol. 2011;164(8):
1949–1958. doi:10.1111/j.1476-5381.2011.01499.x PubMed
26. Kriikku P, Wilhelm L, Schwarz O, et al. New designer drug of abuse: 3,4-methylenedioxypyrovalerone (MDPV): findings from apprehended drivers in Finland. Forensic Sci Int. 2011;210(1–3):195–200. doi:10.1016/j.forsciint.2011.03.015 PubMed
27. Ross EA, Watson M, Goldberger B. “Bath salts” intoxication.
N Engl J Med. 2011;365(10):967–968. doi:10.1056/NEJMc1107097 PubMed
28. Baron M, Elie M, Elie L. An analysis of legal highs: do they contain
what it says on the tin? Drug Test Anal. 2011;3(9):576–581. doi:10.1002/dta.274 PubMed
29. Davies S, Wood DM, Smith G, et al. Purchasing “legal highs” on the Internet: is there consistency in what you get? QJM. 2010;103(7):489–493. doi:10.1093/qjmed/hcq056 PubMed
30. Hadlock GC, Webb KM, McFadden LM, et al. 4-Methylmethcathinone (mephedrone): neuropharmacological effects of a designer stimulant of abuse. J Pharmacol Exp Ther. 2011;339(2):530–536. doi:10.1124/jpet.111.184119 PubMed
31. Winstock A, Mitcheson L, Ramsey J, et al. Mephedrone: use, subjective
effects and health risks. Addiction. 2011;106(11):1991–1996. doi:10.1111/j.1360-0443.2011.03502.x PubMed
32. American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders, Fourth Edition, Text Revision. Washington, DC:
American Psychiatric Association; 2000.
33. Penders TM, Gestring RE, Vilensky DA. Intoxication delirium following use of synthetic cathinone derivatives. Am J Drug Alcohol Abuse. 2012;38(6):
616–617. doi:10.3109/00952990.2012.694535 PubMed
34. Antonowicz JL, Metzger AK, Ramanujam SL. Paranoid psychosis induced
by consumption of methylenedioxypyrovalerone: two cases. Gen Hosp Psychiatry. 2011;33(6):640, e5–e6. PubMed
35. Goshgarian AM, Benford DM, Caplan JP. Bath salt abuse: neuropsychiatric effects of cathinone derivatives. Psychosomatics. 2011;52(6):593–594. PubMed
36. Adebamiro A, Perazella MA. Recurrent acute kidney injury following bath salts intoxication. Am J Kidney Dis. 2012;59(2):273–275. doi:10.1053/j.ajkd.2011.10.012 PubMed
37. Prosser JM, Nelson LS. The toxicology of bath salts: a review of synthetic cathinones. J Med Toxicol. 2012;8(1):33–42. doi:10.1007/s13181-011-0193-z PubMed
38. Borek HA, Holstege CP. Hyperthermia and multiorgan failure after abuse of “bath salts” containing 3,4-methylenedioxypyrovalerone. Ann Emerg Med. 2012;60(1):103–105. doi:10.1016/j.annemergmed.2012.01.005 PubMed
39. Murray BL, Murphy CM, Beuhler MC. Death following recreational use of designer drug “bath salts” containing 3,4-methylenedioxypyrovalerone (MDPV). J Med Toxicol. 2012;8(1):69–75. doi:10.1007/s13181-011-0196-9 PubMed
40. Gold JA, Rimal B, Nolan A, et al. A strategy of escalating doses of benzodiazepines and phenobarbital administration reduces the need for mechanical ventilation in delirium tremens. Crit Care Med. 2007;35(3):
724–730. doi:10.1097/01.CCM.0000256841.28351.80 PubMed
41. Dunkley EJ, Isbister GK, Sibbritt D, et al. The Hunter Serotonin Toxicity Criteria: simple and accurate diagnostic decision rules for serotonin toxicity. QJM. 2003;96(9):635–642. doi:10.1093/qjmed/hcg109 PubMed
42. Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med. 2005;352(11):1112–1120. doi:10.1056/NEJMra041867 PubMed
LESSONS LEARNED AT THE INTERFACE
OF MEDICINE AND PSYCHIATRY
The Psychiatric Consultation Service at Massachusetts General Hospital sees medical and surgical inpatients with comorbid psychiatric symptoms and conditions. Such consultations require the integration of medical and psychiatric knowledge. During their twice-weekly rounds,
Dr Stern and other members of the Consultation Service discuss the diagnosis and management of conditions confronted. These discussions have given rise to rounds reports that will prove
useful for clinicians practicing at the interface
of medicine and psychiatry.
Dr Keary is a resident in psychiatry at Massachusetts General Hospital, Boston, and McLean Hospital, Belmont, Massachusetts, and is a clinical fellow in psychiatry at Harvard Medical School, Boston, Massachusetts. Dr Nejad is an attending physician on the Psychiatric Consultation Service and the director of the Burns and Trauma Psychiatry Consultation at Massachusetts General Hospital, Boston, and an assistant professor of psychiatry at Harvard Medical School, Boston, Massachusetts.
Dr Rasimas is an intramural staff clinician in the Experimental Therapeutics and Pathophysiology Branch of the National Institute of Mental Health, Bethesda, Maryland, and an associate professor of psychiatry and emergency medicine at Penn State College of Medicine, Hershey, Pennsylvania. Dr Stern is chief of the Psychiatric Consultation Service at Massachusetts General Hospital, Boston, and a professor of psychiatry at Harvard Medical School, Boston, Massachusetts.
Dr Stern is an employee of the Academy of Psychosomatic Medicine, has served on the speaker’s board of Reed Elsevier, is a stock shareholder in WiFiMD (Tablet PC), and has received royalties from Mosby/Elsevier and McGraw Hill. Drs Keary, Nejad, and Rasimas report no conflicts of interest related to the subject of this article.
Prim Care Companion CNS Disord 2013;15(3):doi:10.4088/PCC.12f01459
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: August 21, 2012; accepted December 12, 2012.
Published online: May 16, 2013.
Funding/support: None reported.
Corresponding author: Theodore A. Stern, MD, Department of Psychiatry, Massachusetts General Hospital, Fruit St, WRN 605, Boston, MA 02114.








