Objective: To study cases of lithium neurotoxicity (LN), both reversible (RLN) and irreversible (ILN), due to second-generation antipsychotics (SGAs) combined with lithium.
Data Sources: A comprehen’ ‹sive search was conducted in MEDLINE, PsycINFO, EMBASE, and the Cochrane Library from their inception to July 31, 2017.
Study Selection: Free terms and MeSH headings were combined as follows: [(lithium) AND (adverse events OR safety OR side effects OR neurotoxicity OR neurologic manifestations OR central nervous system) AND (risperidone OR paliperidone OR olanzapine OR aripiprazole OR clozapine OR quetiapine OR ziprasidone OR amisulpride OR asenapine OR lurasidone OR iloperidone)]. Only English-language articles reporting about LN due to SGAs combined with lithium were selected.
Data Extraction: The age, sex, diagnostic categories, clinical features, lithium doses, serum lithium levels, antipsychotic dosages, causative factors, and preventive measures of 11 cases of LN (8 RLN and 3 ILN) due to the lithium and SGA combination were extracted.
Data Synthesis: Forty-five percent of patients were aged > 60 years. The diagnostic categories were schizoaffective disorders, bipolar disorders, and schizophrenia. Cases of RLN presented as an acute brain syndrome, which recovered completely. Cases of ILN presented as a chronic brain syndrome and only partially recovered. The lithium doses in 9 cases were < 1,200 mg/d. The serum lithium levels in 2 cases in each of the groups were > 1.7 mEq/L. The SGAs implicated were clozapine, risperidone, aripiprazole, and quetiapine. One patient with RLN received 2 different first-generation antipsychotics and 1 patient with ILN received 2 different SGAs with lithium. Both groups had patients with medical comorbidities who were taking prescription medication.
Conclusion: LN, both reversible and irreversible, due to SGAs combined with lithium presents with certain causative factors and a clinical profile. Early detection and prompt management will help prevent LN.’ ‹
This CME activity is expired. For more CME activities, visit cme.psychiatrist.com.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
CME Background
Articles are selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME articles on a variety of topics throughout each volume. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the article, correctly answer the questions in the Posttest, and complete the Evaluation. A $10 processing fee will apply.
CME Objective
After studying this article, you should be able to:
- Increase prevention and early detection of lithium neurotoxicity
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1.0 hour of Category I credit for completing this program.
Release, Expiration, and Review Dates
This educational activity was published in June 2019 and is eligible for AMA PRA Category 1 Creditâ„¢ through June 30, 2021. The latest review of this material was June 2019.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Larry Culpepper, MD, MPH, Editor in Chief, has been a consultant for Alkermes, Harmony Biosciences, Merck, Shire, Supernus, and Sunovion. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears at the end of the article.
ABSTRACT
Objective: To study cases of lithium neurotoxicity (LN), both reversible (RLN) and irreversible (ILN), due to second-generation antipsychotics (SGAs) combined with lithium.
Data Sources: A comprehensive search was conducted in MEDLINE, PsycINFO, EMBASE, and the Cochrane Library from their inception to July 31, 2017.
Study Selection: Free terms and MeSH headings were combined as follows: [(lithium) AND (adverse events OR safety OR side effects OR neurotoxicity OR neurologic manifestations OR central nervous system) AND (risperidone OR paliperidone OR olanzapine OR aripiprazole OR clozapine OR quetiapine OR ziprasidone OR amisulpride OR asenapine OR lurasidone OR iloperidone)]. Only English-language articles reporting about LN due to SGAs combined with lithium were selected.
Data Extraction: The age, sex, diagnostic categories, clinical features, lithium doses, serum lithium levels, antipsychotic dosages, causative factors, and preventive measures of 11 cases of LN (8 RLN and 3 ILN) due to the lithium and SGA combination were extracted.
Data Synthesis: Forty-five percent of patients were aged > 60 years. The diagnostic categories were schizoaffective disorders, bipolar disorders, and schizophrenia. Cases of RLN presented as an acute brain syndrome, which recovered completely. Cases of ILN presented as a chronic brain syndrome and only partially recovered. The lithium doses in 9 cases were < 1,200 mg/d. The serum lithium levels in 2 cases in each of the groups were > 1.7 mEq/L. The SGAs implicated were clozapine, risperidone, aripiprazole, and quetiapine. One patient with RLN received 2 different first-generation antipsychotics and 1 patient with ILN received 2 different SGAs with lithium. Both groups had patients with medical comorbidities who were taking prescription medication.
Conclusion: LN, both reversible and irreversible, due to SGAs combined with lithium presents with certain causative factors and a clinical profile. Early detection and prompt management will help prevent LN.
Prim Care Companion CNS Disord 2019;21(3):17r02225
To cite: Netto I, Phutane VH, Ravindran B. Lithium neurotoxicity due to second-generation antipsychotics combined with lithium: a systematic review. Prim Care Companion CNS Disord. 2019;21(3):17r02225.
To share: https://doi.org/10.4088/PCC.17r02225
© Copyright 2019 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Byramjee Jeejeebhoy Medical College and Sassoon General Hospital, Pune, Maharashtra, India
bParmar Plaza Clinic, Wanowrie, Pune, Maharashtra, India
cDepartment of Rural Health, Melbourne Medical School, The University of Melbourne, Melbourne, Australia
dDepartment of Psychiatry, Goulburn Valley Area Mental Health Service, Shepparton, Victoria, Australia
eHunter New England Health Services, Mental Health Centre, The Mater Hospital, Waratah, New South Wales, Australia
*Corresponding author: Ivan Netto, MBBS, MD, E-10 Parmar Plaza Clinic, Parmar Plaza, Shivarkar Rd, Pune 411001, Maharashtra, India ([email protected]).
Lithium neurotoxicity (LN) may be of 2 types: reversible (RLN) or irreversible (ILN). Most authors define RLN as a complete recovery 2 months after an episode of lithium toxicity, whereas those patients who recover partially or with permanent neurologic sequelae are classified as having ILN.1-3
Lithium toxicity may be of the following subtypes: acute, acute-on-chronic, or chronic. Lithium has a narrow therapeutic range, and patients vary in their response to therapeutic dosing.4,5 In lithium-naive patients, acute toxicity caused by accidental or intentional overdose presents with gastrointestinal and cardiac symptoms, and neurologic symptoms develop later.4,5 Acute-on-chronic toxicity occurs with acute overdose in patients on long-term lithium treatment, and the presentation is a more severe form of acute toxicity.4,5 Chronic toxicity occurs insidiously during long-term lithium treatment when serum lithium levels may be in the therapeutic range. These patients develop mainly neurologic symptoms and sometimes only partially recover.4,5 The distinction between acute-on-chronic and chronic toxicity is often not so clear and difficult to establish.4,5 The Amdisen Scale5-7 is a useful tool for the assessment of the severity of lithium toxicity.

- Treatments for schizophrenia and bipolar disorders are now converging, and current evidence supports combining 2 drugs with 2 different mechanisms of action; however, patients receiving lithium and second-generation antipsychotics (SGAs) together can develop lithium neurotoxicity.
- High-risk groups on the lithium and SGA combination are elderly patients, those with medical comorbidities, and those on other prescription medication.
- Clinicians can help potentially prevent lithium neurotoxicity by reducing the dose of lithium and SGA when both are prescribed together along with early detection, prompt management, careful prescribing, and psychoeducation of patients and caregivers.
Schou8 was the pioneering researcher to report LN and its long-lasting neurologic sequelae. Lithium toxicity is common in the elderly, with an annual incidence of 1.5%.9 In 1.3%-4% of these cases, there is also acute kidney damage and renal toxicity, which usually resolve within 12 months, but some renal impairment may be more sustained.9-11
Some of the proposed risk factors for LN are nephrogenic diabetes insipidus, age > 50 years, thyroid dysfunction, and impaired creatinine clearance.12 Dehydration and volume depletion may cause lithium toxicity due to increased sodium and lithium reabsorption.13,14 Medications such as angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, thiazide diuretics, and nonsteroidal anti-inflammatory drugs increase renal sodium reabsorption and raise serum lithium levels.13-15 Cardiovascular disease such as congestive cardiac failure may also precipitate lithium toxicity by decreasing lithium delivery to the kidneys, the only site of its excretion.13
As the evidence for the use of lithium increases, paradoxically, there is a decrease in its use.16 As there is a delay in the onset of the therapeutic effects of lithium, especially in agitated patients, lithium is sometimes combined with antipsychotics and anticonvulsants.17-24 In the past, lithium was combined with first-generation antipsychotics (FGAs).1-3 However, recently, lithium is more commonly combined with second-generation antipsychotics (SGAs).23,24 This combination has become an area of caution and concern.
In reviews published on RLN and ILN, most authors1-3 based their opinions on individual case reports and hospital records. However, to our knowledge, no review describes the clinical presentation of both RLN and ILN due to the combination of lithium and SGAs. Hence, we studied the clinical presentation of cases of LN (reversible and irreversible) due to the SGA and lithium combination. We also studied the causes of and preventive measures to avoid LN. This review has important clinical implications for psychiatric management, training of mental health professionals, and further research for patients receiving a combination of lithium and SGAs.
METHODS
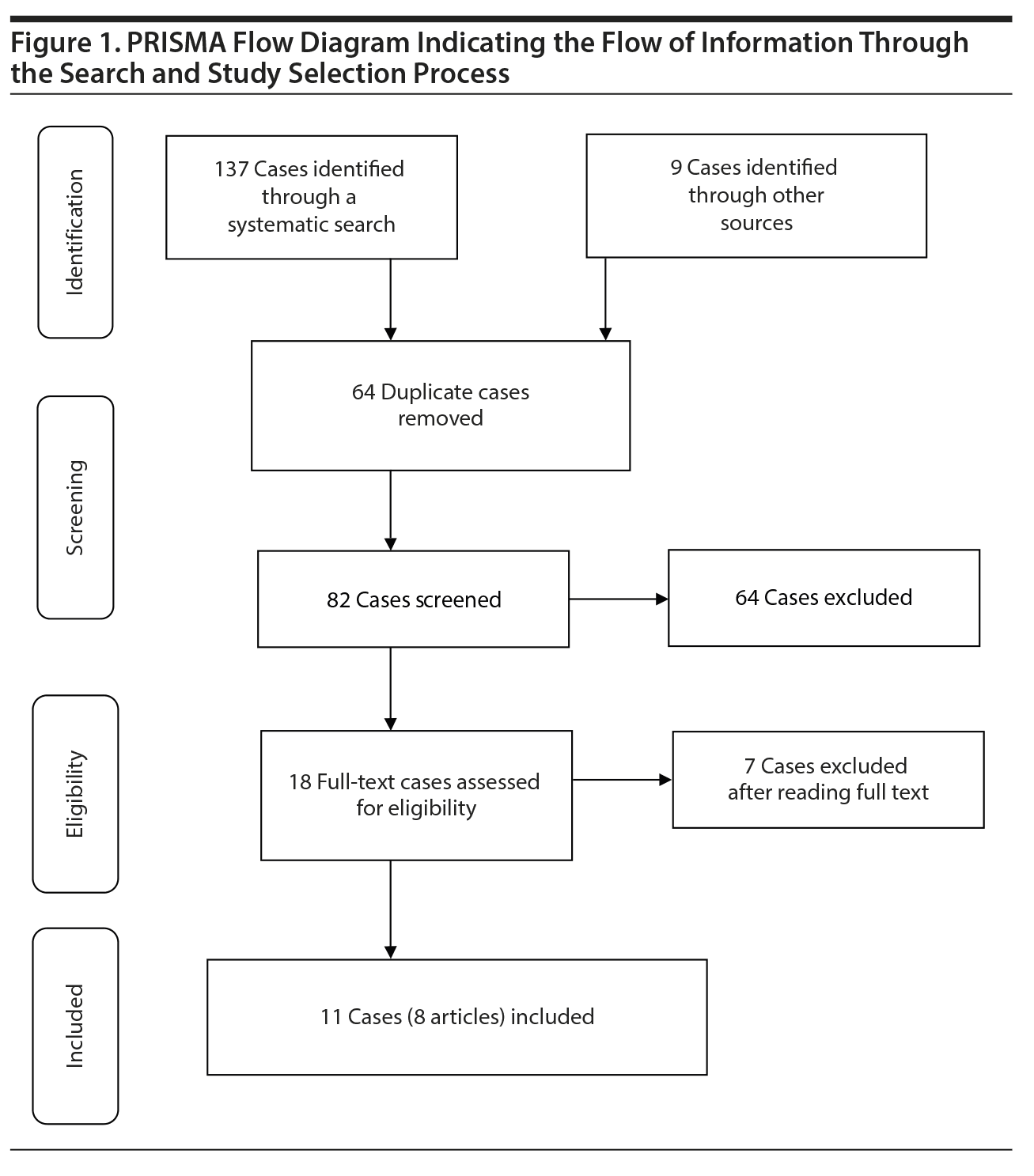
Studies were identified from a comprehensive search of the electronic databases in MEDLINE, PsycINFO, EMBASE, and Cochrane Library from their inception to July 31, 2017. We combined free terms and MeSH headings as follows: [(lithium) AND (adverse events OR safety OR side effects OR neurotoxicity OR neurologic manifestations OR central nervous system) AND (risperidone OR paliperidone OR olanzapine OR aripiprazole OR clozapine OR quetiapine OR ziprasidone OR amisulpride OR asenapine OR lurasidone OR iloperidone)]. The search was limited to English-language human studies. Further studies were collected through manual searches, discussion with experts in the field, and review of references of relevant articles. The Preferred Reporting Items for Systematic Reviews and Meta-Analysis25 flow diagram indicates the flow of information through search and study selection processes (Figure 1).
Two reviewers (I.N. and V.H.P.) independently searched and extracted the data. Disagreements were resolved through discussion. Only articles reporting on LN due to SGAs combined with lithium were selected. All cases of LN in adults, irrespective of their age, sex, ethnicity, functioning level, and inpatient or outpatient status, were included. Cases of LN due to SGAs combined with lithium with insufficient data and no clear outcome of reversibility or partial recovery from LN and cases reporting minor neurologic side effects of lithium were excluded.
The data from the included studies were extracted according to the following criteria: author, year of publication, age, sex, diagnostic categories, lithium dosages, serum lithium levels, investigations, clinical features (physical and mental), and causes and preventive measures. The results obtained were tabulated.
RESULTS
A total of 137 studies were retrieved through a systematic search. Nine other studies were selected from the references of those studies. A total of 82 articles remained after removing duplicates, which were also screened. Of the 82 articles, 64 did not meet the eligibility criteria. The remaining 18 articles were assessed for eligibility, but 7 were excluded after scrutinizing the full text of each of the 18 articles. A total of 8 studies5,26-32 with 11 cases of LN due to the lithium and SGA combination were included in the systematic review.
Of the 11 cases studied, 8 were cases of RLN and 3 were cases of ILN. Of the 8 cases of RLN, there were 4 case reports of individual patients and 1 case series of 4 patients.26-30 Of the 3 cases of ILN, all were reports of individual cases.5,31,32 None of the studies reported receiving any grants or financial support.
Year of Publication
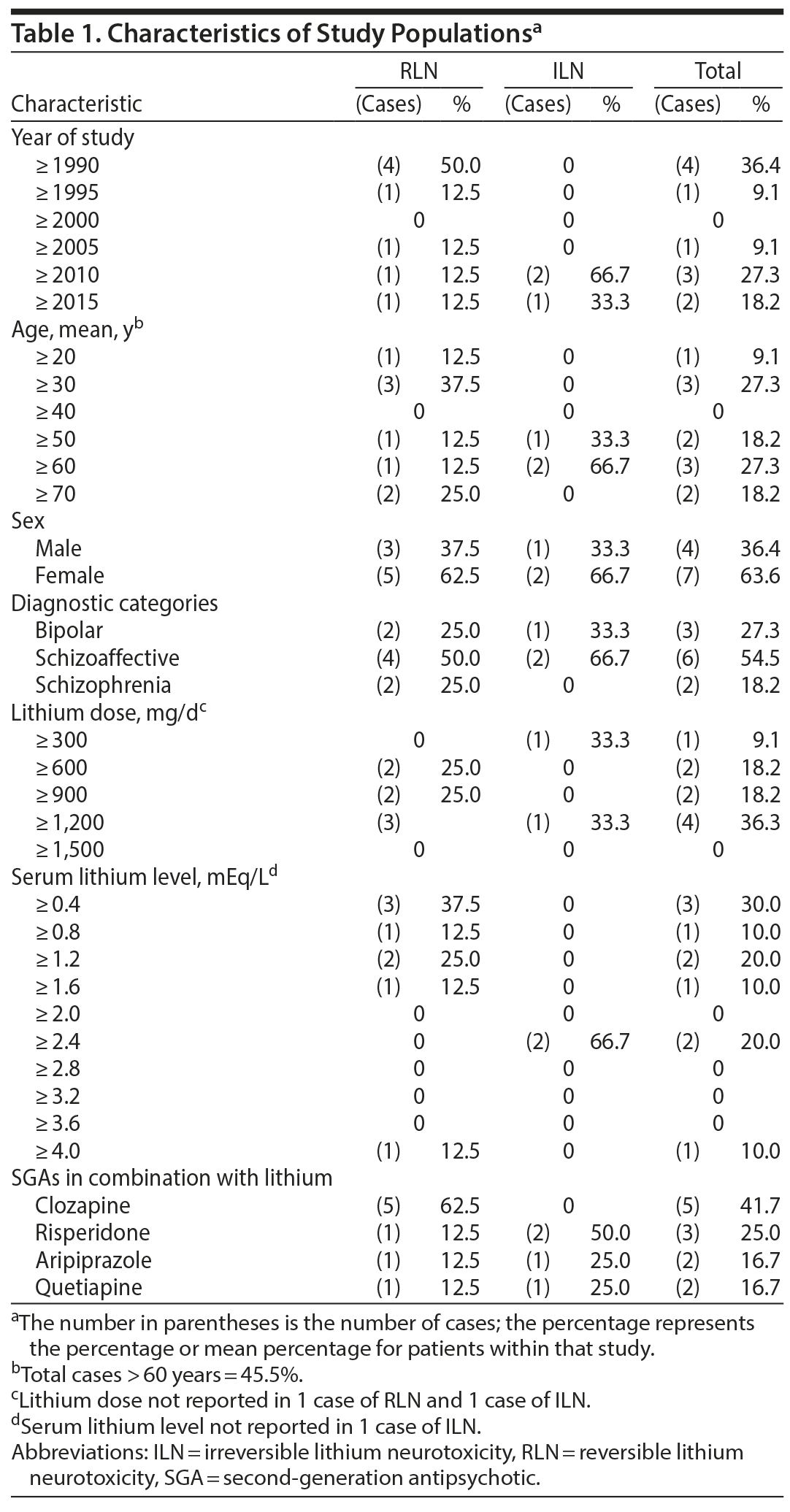
RLN. Eight cases were published during the period 1992-2015.26-30
ILN. Three cases were published during the period 2010-20145,31,32 (Tables 1-3).
Age
RLN. The age of patients ranged from 27 to 78 years. Of 8 cases,28-29 2 were elderly patients aged 75 and 78 years.
ILN. The age of patients ranged from 56 to 69 years.5,31,32 Of 5 cases, 45.5% were aged ≥ 60 years (Tables 1 – 3).
Natural aging increases the risk for lithium toxicity. Due to decreased estimated glomerular filtration rate, the lithium dose required to achieve a given therapeutic serum level may decrease 3 times between the ages of 40 and 95 years.11
Sex
RLN. The RLN group included 3 men and 5 women.26-30
ILN. The ILN group included 1 man and 2 women.
Thus, of a total of 11 cases of LN, 7 were women and 4 were men5,31,32 (Tables 1-3). Two reviews have also reported a greater number of female cases of LN in the literature.1,3
Diagnostic Categories
RLN. Of the 8 cases of RLN, 2 had bipolar disorders, 4 had schizoaffective disorder, and 2 had schizophrenia.26-30
ILN. Of the 3 cases of ILN, 1 had a bipolar disorder and 2 had schizoaffective disorders5,31,32 (Tables 1-3).
Diagnostic classification was reported in only 1 study,32 which used the ICD-10 diagnostic classification. Psychiatric diagnosis does not seem to be a factor related to the development of lithium toxicity.1-3
Clinical Features: Physical
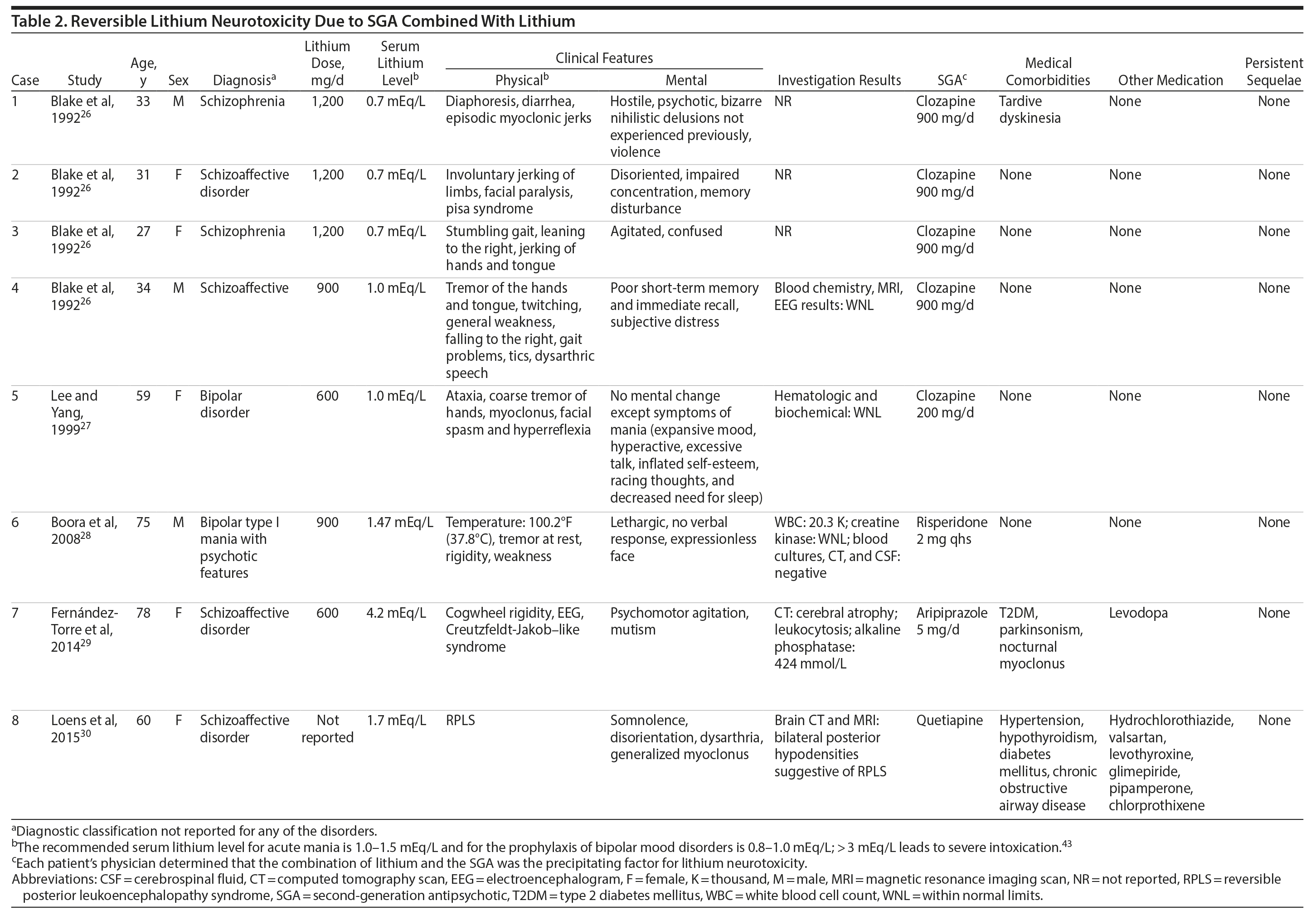
RLN. Cases of RLN presented with pyramidal, extrapyramidal, and cerebellar signs, and patients recovered completely.26-30 One patient developed reversible posterior leukoencephalopathy syndrome (RPLS; Table 2).30
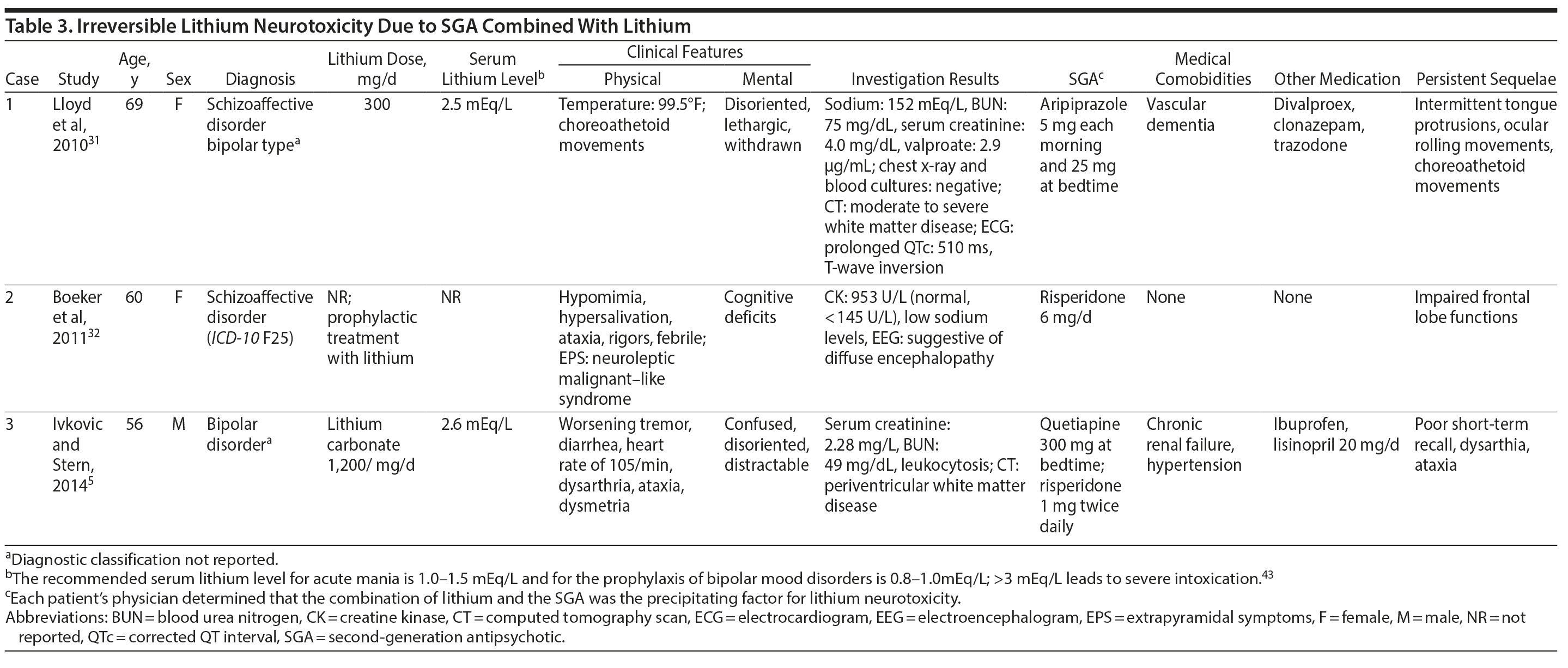
ILN. Cases of ILN presented with pyramidal signs, extrapyramidal signs, cerebellar signs, choreoathetoid movements, Creutzfeldt-Jakob-like syndrome, neuroleptic malignant-like syndrome, cognitive deficits, ocular signs, and impaired frontal lobe functions, and patients recovered partially5,31,32 (Table 3).
Cerebellar signs have been reported by some authors in cases of RLN.1-3 One author8 maintained that cerebellar signs do not occur in acute lithium toxicity. Cases presenting as a serotonin-like syndrome, neuroleptic malignant syndrome, pseudotumor cerebri, or Creutzfeldt-Jakob-like syndrome have also been reported.33-40
Clinical Features: Mental
RLN. Most cases of RLN presented as an acute organic brain syndrome (delirium), and patients later recovered completely.26-30
ILN. Cases of ILN presented initially as an acute brain syndrome, but patients later developed a chronic brain syndrome and only partially recovered from LN5,31,32 (Tables 1-3). Similar findings have also been found in other studies.1-3
Investigations
RLN. Five cases of RLN showed laboratory investigations within normal limits.26-30 In 3 cases, no laboratory results were reported (Table 2).26
ILN. Cases of ILN showed abnormal hemogram, renal function, electrocardiogram, electroencephalogram (EEG), and computed tomography findings.5,31,32
One patient’s EEG findings showed Creutzfeldt-Jakob-like syndrome,29 and another showed RPLS on magnetic resonance imaging30 (Table 3). Abnormal EEG findings are important indicators of early LN according to some authors.41
Lithium Dosage
RLN. The doses of lithium ranged from 600-1,200 mg/d (Table 2).26-30
ILN. The doses of lithium ranged from 300-1,200 mg/d (Table 3).
Most cases had lithium dosages < 1,200 mg/d in both groups.5,31,32 In 1 case each of RLN and ILN, the dose was not reported.30,32 Neuroleptic-induced LN is dose dependent and has been highlighted by some authors.42
Serum Lithium Levels
RLN. The serum lithium levels ranged from 0.7-4.2 mEq/L. In 6 cases of RLN, serum lithium levels were < 1.5 mEq/L, but in 2 cases they were 1.7 and 4.2 mEq/L (Table 2).26-30
ILN. The serum lithium levels in 2 cases were 2.5 and 2.6 mEq/L, and in 1 case the level was not reported (Table 3).
Generally, serum lithium levels of 1.0-1.5 mEq/L have been recommended for the treatment of acute manic episodes and levels of 0.8-1.0 mEq/L for the prophylaxis of bipolar mood disorders.43 Severe intoxication occurs at serum lithium levels > 3 mEq/L, and serum lithium levels > 5 mEq/L can be fatal.44
In the age group ≥ 60 years, the serum lithium levels were 1.47, 1.7, and 4.2 mEq/L in cases of RLN and 2.5 mEq/L in a case of ILN.28-31 In 1 case of ILN, the serum lithium level was not reported.32 Three patients had medical comorbidities and were on prescription medication.29-31 In the age group < 60 years, in cases of RLN, 3 had a serum lithium level of 0.7 mEq/L26 and 2 had a level of 1.0 mEq/L,26,27 and in a case of ILN, the level was 2.6 mEq/L.5 One author11 has recommended monitoring of serum lithium levels at 3-month intervals in the elderly due to the high risk for development of LN. The lithium toxicity occurring at low or therapeutic serum lithium levels has been referred to as paradoxical or pathological LN.43,45,46
Dosages of SGA Medications
All patients received oral SGA medication.
RLN. Clozapine dosages were 900 mg/d in 4 cases26 and 200 mg/d in another.27 In 1 case,28 the risperidone dose was 2 mg/d. In another case,29 the aripiprazole dose was 5 mg/d. The dose of quetiapine was not reported in 1 case (Table 2).30
ILN. One patient received aripiprazole 30 mg/d,31 1 patient received risperidone 6 mg/d,32and 1 patient received an SGA combination of risperidone 2 mg/d and quetiapine 300 mg/d (Table 3).5 Some authors42 have emphasized that the dosage of the antipsychotic, not the serum lithium level or lithium dose, predicts neurotoxicity.
Medical Comorbidities
RLN. The RLN group had 3 cases with medical comorbidities. One patient had tardive dyskinesia,26 1 had parkinsonism and diabetes,29 and 1 had hypertension, hypothyroidism, diabetes mellitus, and chronic obstructive airway disease30 (Table 2).
ILN. One patient with ILN had vascular dementia, fever, and dehydration31 and another had chronic renal failure5 (Table 3).
It has been cautioned that medical comorbidities such as thyroid dysfunction, diabetes insipidus, dehydration, renal disease, cardiovascular disease, and preexisting central nervous system disease may lead to LN.5-15
Other Medication
RLN. One patient received levodopa29 and another hydrochlorothiazide, valsartan, levothyroxine, and glimepiride30 along with the FGAs pipamperone and chlorprothixene (Table 2).
ILN. One patient received divalproex, clonazepam, and trazodone31 and another received ibuprofen and lisinopril5 (Table 3).
The risk factors for LN include medications commonly prescribed, such as nonsteroidal anti-inflammatory drugs, diuretics, angiotensin-converting enzyme inhibitors, and angiotensin II receptor blockers, which have been reported in some studies.12-15
Preventive Measures
Various measures to prevent LN due to the lithium and SGA combination have been suggested in light of clinical findings. When combining lithium with SGAs, a decrease in the dose of the antipsychotic as well as a lower dose of lithium is recommended.27,32 A special caution in the elderly is advised. In addition to serum lithium level, renal function monitoring has also been recommended in the elderly.9-12 Some authors5 have recommended that clear guidelines should be made available regarding the use of renal dialysis and other procedures in the management of LN.
DISCUSSION
This review is the first to examine the clinical presentation of cases of LN, both reversible and irreversible, due to SGAs combined with lithium. A total of 11 cases of LN due to lithium plus SGA combinations were evaluated. Eight were reversible and 3 irreversible.5,26-32
The most important hypotheses for neurotoxicity due to the lithium and SGA combination is increased dopamine receptor blockage. Both FGAs and SGAs cause postsynaptic blockade of brain dopamine D2 receptors with the exception of aripiprazole, which is a D2 receptor partial agonist.47 Most SGAs differ from FGAs pharmacologically in that serotonin 5-HT2 receptor binding exceeds their affinity for dopamine D2 receptors, whereas in FGAs this is generally not the case.48 SGAs also have loose D2 receptor binding, rapid dissociation rates, and preferential binding of drugs to receptors in limbic and cortical brain regions rather than striatal areas.49,50 The interaction of the serotonergic effects of lithium and SGAs has also been postulated as a potential mechanism for LN.26 Lithium appears to affect multiple neurotransmitter systems and inhibits postsynaptic dopamine release. Some authors have also highlighted “the two-pronged effect” of lithium and SGAs when combined together, resulting in profound dopamine hypofunctionality as a causative mechanism for LN.32
Similarities and Differences in Cases of RLN and ILN Due to SGAs
RLN and ILN have no specific association with age or sex distribution. RLN was seen in schizophrenia and bipolar and schizoaffective disorders, whereas ILN was seen only in bipolar and schizoaffective disorders. Patients with RLN developed an acute brain syndrome and recovered completely, and patients with ILN developed a chronic brain syndrome with partial recovery. Cerebellar signs in RLN recovered completely, whereas those of ILN did not recover. One patient with RLN developed Creutzfeldt-Jakob-like syndrome and another RPLS.29,30 Investigations in cases of RLN were normal, whereas those in ILN showed abnormalities. In both groups, the lithium doses in all cases of LN were less than 1,200 mg/d (lithium dosage was not reported in 2 cases). Serum lithium levels in 2 cases of RLN were 1.7 and 4.2 mEq/L and in 2 cases of ILN were 2.5 and 2.6 mEq/L. One RLN patient received 2 different FGAs along with the lithium and SGA combination.30 One ILN patient had received a combination of 2 different SGAs along with lithium.5 Both groups had cases with medical comorbidities who were also receiving other prescription medication that was a contributing factor for the development of LN.
Limitations
The data were secondary and heterogeneous in nature. A few studies did not report certain data such as lithium doses and serum lithium levels. The most significant serum lithium level with a temporal relationship to the onset of lithium toxicity was chosen in some studies that reported different serum lithium levels.
CONCLUSIONS
The following clinical profile emerges after evaluating 11 cases of LN, of which 8 were cases of RLN and 3 of ILN due to the lithium and SGA combination. Forty-five percent of cases were aged ≥ 60 years. There were more female cases compared to males. The diagnostic categories in both RLN and ILN were schizoaffective disorders and bipolar disorders, but 2 patients with RLN had schizophrenia. None of the cases were of the acute subtype with an acute onset due to an accidental or intentional overdose, but all were acute-on-chronic or chronic subtypes with an insidious or gradual onset. The clinical features in cases of RLN were those of an acute brain syndrome with pyramidal, extrapyramidal, and cerebellar signs that recovered completely. Cases of ILN showed signs of a chronic brain syndrome with choreoathetosis, a Creutzfeldt-Jakob-like syndrome, and a neuroleptic malignant-like syndrome and only partially recovered. The lithium doses in all (except 2 cases for which a lithium dose was not reported) were less than 1,200 mg/d. Serum lithium levels in 2 cases of RLN were 1.7 and 4.2 mEq/L and in 2 cases of ILN were 2.5 and 2.6 mEq/L; the rest (except 1 that did not report the serum lithium level) were 1.5 mEq/L. The SGAs used in combination with lithium and responsible for LN were clozapine, risperidone, aripiprazole, and quetiapine. One patient with RLN received 2 different FGAs in addition to the lithium and SGA combination. One patient with ILN received 2 different SGAs with lithium. One patient with RLN had tardive dyskinesia, a second parkinsonism and diabetes, and a third hypertension, hypothyroidism, diabetes mellitus, and chronic obstructive airway disease. One patient with ILN had vascular dementia, fever, and dehydration and another chronic renal failure. One RLN patient received levodopa and another hydrochlorothiazide, valsartan, levothyroxine, glimepiride, pipamperone, and chlorprothixene. One ILN patient received divalproex, clonazepam, and trazodone, while another received ibuprofen and lisinopril. Diagnostic classifications were not reported in all but 1 case. Lithium doses in 2 cases, serum lithium levels in 1 case, and the SGA dose in 1 case were not reported.
Clinical Implications
This review helps to gain a better understanding of LN, both reversible and irreversible, due to the lithium and SGA combination. A ban on the use of the combination has not been recommended but caution while prescribing lithium and SGAs together is urged. A reduction in the doses of both lithium and SGAs along with close monitoring has been suggested. Potential high-risk groups for developing LN on the lithium and SGA combination such as elderly patients, patients with medical comorbidities, and those on prescription medication should be monitored carefully. This review will help in the prevention of LN through early detection, close monitoring, care in combining other drugs with the lithium and SGA combination, prompt management of LN, and proper psychoeducation of patients and caregivers.
Submitted: September 23, 2017; accepted February 5, 2019.
Published online: June 20, 2019.
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Financial disclosure: Drs Netto, Phutane, and Ravindran have no personal affiliations or financial relationships with any commercial interest to disclose relative to this article.
Funding/support: None.
This CME activity is expired. For more CME activities, visit cme.psychiatrist.com.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1.Kores B, Lader MH. Irreversible lithium neurotoxicity: an overview. Clin Neuropharmacol. 1997;20(4):283-299. PubMed CrossRef
2.Adityanjee RJ. The syndrome of irreversible lithium-effectuated neurotoxicity (SILENT). Pharmacopsychiatry. 1989;22(2):81-83. PubMed CrossRef
3.Netto I, Phutane VH. Reversible lithium neurotoxicity: review of the literature. Prim Care Companion CNS Disord. 2012;14(1):doi:10.4088/PCC.11r01197. PubMed CrossRef
4.Katz RB, Packer CD. Lithium toxicity presenting as transient transcortical motor aphasia: a case report. Psychosomatics. 2014;55(1):87-91. PubMed CrossRef
5.Ivkovic A, Stern TA. Lithium-induced neurotoxicity: clinical presentations, pathophysiology, and treatment. Psychosomatics. 2014;55(3):296-302. PubMed CrossRef
6.Hansen HE, Amdisen A. Lithium intoxication: report of 23 cases and review of 100 cases from the literature. Q J Med. 1978;47(186):123-144. PubMed
7.Waring WS, Laing WJ, Good AM, et al. Pattern of lithium exposure predicts poisoning severity: evaluation of referrals to a regional poisons unit. QJM. 2007;100(5):271-276. PubMed CrossRef
8.Schou M. Long-lasting neurological sequelae after lithium intoxication. Acta Psychiatr Scand. 1984;70(6):594-602. PubMed CrossRef
9.Juurlink DN, Mamdani MM, Kopp A, et al. Drug-induced lithium toxicity in the elderly: a population-based study. J Am Geriatr Soc. 2004;52(5):794-798. PubMed CrossRef
10. Chen KP, Shen WW, Lu ML. Implication of serum concentration monitoring in patients with lithium intoxication. Psychiatry Clin Neurosci. 2004;58(1):25-29. PubMed CrossRef
11. Rej S, Shulman K, Herrmann N, et al. Prevalence and correlates of renal disease in older lithium users: a population-based study. Am J Geriatr Psychiatry. 2014;22(11):1075-1082. PubMed CrossRef
12. Oakley PW, Whyte IM, Carter GL. Lithium toxicity: an iatrogenic problem in susceptible individuals. Aust N Z J Psychiatry. 2001;35(6):833-840. PubMed CrossRef
13. Grandjean EM, Aubry JM. Lithium: updated human knowledge using an evidence-based approach, part II: clinical pharmacology and therapeutic monitoring. CNS Drugs. 2009;23(4):331-349. PubMed CrossRef
14. Nagaraja D, Taly AB, Sahu RN, et al. Permanent neurological sequelae due to lithium toxicity. Clin Neurol Neurosurg. 1987;89(1):31-34. PubMed CrossRef
15. Freeman MP, Freeman SA. Lithium: clinical considerations in internal medicine. Am J Med. 2006;119(6):478-481. PubMed CrossRef
16. Young AH, Hammond JM. Lithium in mood disorders: increasing evidence base, declining use? Br J Psychiatry. 2007;191(6):474-476. PubMed CrossRef
17. Fieve RR. Lithium therapy at the millennium: a revolutionary drug used for 50 years faces competing options and possible demise. Bipolar Disord. 1999;1(2):67-70. PubMed CrossRef
18. Goodwin FK. Rationale for long-term treatment of bipolar disorder and evidence for long-term lithium treatment. J Clin Psychiatry. 2002;63(suppl 10):5-12. PubMed
19. Macritchie K, Geddes JR, Scott J, et al. Valproate for acute mood episodes in bipolar disorder. Cochrane Database Syst Rev. 2003;1(1):CD004052. PubMed
20. Emilien G, Maloteaux JM, Seghers A, et al. Lithium compared to valproic acid and carbamazepine in the treatment of mania: a statistical meta-analysis. Eur Neuropsychopharmacol. 1996;6(3):245-252. PubMed CrossRef
21. Goodwin GM, Bowden CL, Calabrese JR, et al. A pooled analysis of 2 placebo-controlled 18-month trials of lamotrigine and lithium maintenance in bipolar I disorder. J Clin Psychiatry. 2004;65(3):432-441. PubMed CrossRef
22. McIntyre RS, Mancini DA, McCann S, et al. Topiramate versus bupropion SR when added to mood stabilizer therapy for the depressive phase of bipolar disorder: a preliminary single-blind study. Bipolar Disord. 2002;4(3):207-213. PubMed CrossRef
23. Vieta E, Goikolea JM. Atypical antipsychotics: newer options for mania and maintenance therapy. Bipolar Disord. 2005;7(suppl 4):21-33. PubMed CrossRef
24. Yatham LN. Atypical antipsychotics for bipolar disorder. Psychiatr Clin North Am. 2005;28(2):325-347. PubMed CrossRef
25. Moher D, Liberati A, Tetzlaff J, et al; PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. PubMed CrossRef
26. Blake LM, Marks RC, Luchins DJ. Reversible neurologic symptoms with clozapine and lithium. J Clin Psychopharmacol. 1992;12(4):297-299. PubMed CrossRef
27. Lee SH, Yang YY. Reversible neurotoxicity induced by a combination of clozapine and lithium: a case report. Zhonghua Yi Xue Za Zhi (Taipei). 1999;62(3):184-187. PubMed
28. Boora K, Xu J, Hyatt J. Encephalopathy with combined lithium-risperidone administration. Acta Psychiatr Scand. 2008;117(5):394-395, discussion 396. PubMed CrossRef
29. Fernסndez-Torre JL, Oterino A, Marcelino-Salas K, et al. Creutzfeldt-Jakob-like syndrome secondary to severe lithium intoxication: a detailed follow-up electroencephalographic study. Clin Neurophysiol. 2014;125(11):2315-2317. PubMed CrossRef
30. Loens S, Conzen J, Welte GS, et al. Reversible posterior leukoencephalopathy syndrome after withdrawal of antipsychotic medication in the context of lithium intoxication. Gen Hosp Psychiatry. 2015;37(3):274.e3-274.e5. PubMed CrossRef
31. Lloyd RB, Perkins RE, Schwartz AC. Choreoathetosis in the setting of lithium toxicity. Psychosomatics. 2010;51(6):529-531. PubMed CrossRef
32. Boeker H, Seidl A, Schopper C. Neurotoxicity related to combined treatment with lithium, antidepressants and atypical antipsychotics. Schweiz Arch Neurol Psychiatr. 2011;162(6):72-76.
33. Muly EC, McDonald W, Steffens D, et al. Serotonin syndrome produced by a combination of fluoxetine and lithium. Am J Psychiatry. 1993;150(10):1565. PubMed CrossRef
34. Rosenberg PB, Pearlman CA. NMS-Like syndrome with a lithium/doxepin combination. J Clin Psychopharmacol. 1991;11(1):75-76. PubMed CrossRef
35. Smith SJ, Kocen RS. A Creutzfeldt-Jakob-like syndrome due to lithium toxicity. J Neurol Neurosurg Psychiatry. 1988;51(1):120-123. PubMed CrossRef
36. Broussolle E, Setiey A, Moene Y, et al. Reversible Creutzfeldt-Jakob-like syndrome induced by lithium plus levodopa treatment. J Neurol Neurosurg Psychiatry. 1989;52(5):686-687. PubMed CrossRef
37. Finelli PF. Drug-induced Creutzfeldt-Jakob-like syndrome. J Psychiatry Neurosci. 1992;17(3):103-105. PubMed
38. Waldman AJA. A Creutzfeldt-Jakob-like syndrome due to polypharmacy. South Med J. 1990;83(12):1493. PubMed CrossRef
39. Primavera A, Brusa G, Poeta MG. A Creutzfeldt-Jakob-like syndrome due to lithium toxicity. J Neurol Neurosurg Psychiatry. 1989;52(3):423. PubMed CrossRef
40. Saul RF, Hamburger HA, Selhorst JB. Pseudotumor cerebri secondary to lithium carbonate. JAMA. 1985;253(19):2869-2870. PubMed CrossRef
41. Spring GK. Neurotoxicity with combined use of lithium and thioridazine. J Clin Psychiatry. 1979;40(3):135-138. PubMed
42. Miller F, Menninger J. Lithium-neuroleptic neurotoxicity is dose dependent. J Clin Psychopharmacol. 1987;7(2):89-91. PubMed
43. Spiers J, Hirsch SR. Severe lithium toxicity with ‘ normal’ serum concentrations. Br Med J. 1978;1(6116):815-816. PubMed CrossRef
44. Strayhorn JM Jr, Nash JL. Severe neurotoxicity despite “therapeutic” serum lithium levels. Dis Nerv Syst. 1977;38(2):107-111. PubMed
45. Gangadhar BN, Subhash MN, Umapathy C, et al. Lithium toxicity at therapeutic serum levels. Br J Psychiatry. 1993;163(05):695. PubMed CrossRef
46. Bell AJ, Cole A, Eccleston D, et al. Lithium neurotoxicity at normal therapeutic levels. Br J Psychiatry. 1993;162(05):689-692. PubMed CrossRef
47. Seeman P. Dopamine receptors and the dopamine hypothesis of schizophrenia. Synapse. 1987;1(2):133-152. PubMed CrossRef
48. Kapur S, Zipursky R, Jones C, et al. Relationship between dopamine D(2) occupancy, clinical response, and side effects: a double-blind PET study of first-episode schizophrenia. Am J Psychiatry. 2000;157(4):514-520. PubMed CrossRef
49. Meltzer HY. What’s atypical about atypical antipsychotic drugs? Curr Opin Pharmacol. 2004;4(1):53-57. PubMed CrossRef
50. Seeman P. Atypical antipsychotics: mechanism of action. Can J Psychiatry. 2002;47(1):27-38. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!