Background: Effective medical and surgical management of pediatric congenital heart disease (CHD) to reduce long-term adverse neurodevelopmental outcomes is an important clinical objective in primary and specialty health care. We identify clinical predictors associated with an increased risk of 6 long-term neurodevelopmental outcomes in children with CHD compared to the general pediatric Medicaid population.
Method: South Carolina’s retrospective, 15-year Medicaid data set (January 1, 1996-December 31, 2010) for 19,947 patients aged ≤ 17 years diagnosed with ≥ 1 CHD lesions (on the basis of International Classification of Diseases, Ninth Revision, Clinical Modification codes) were compared to 19,948 patients without CHD matched on age at entry into and duration in Medicaid using logistic and Cox proportional hazards regression.
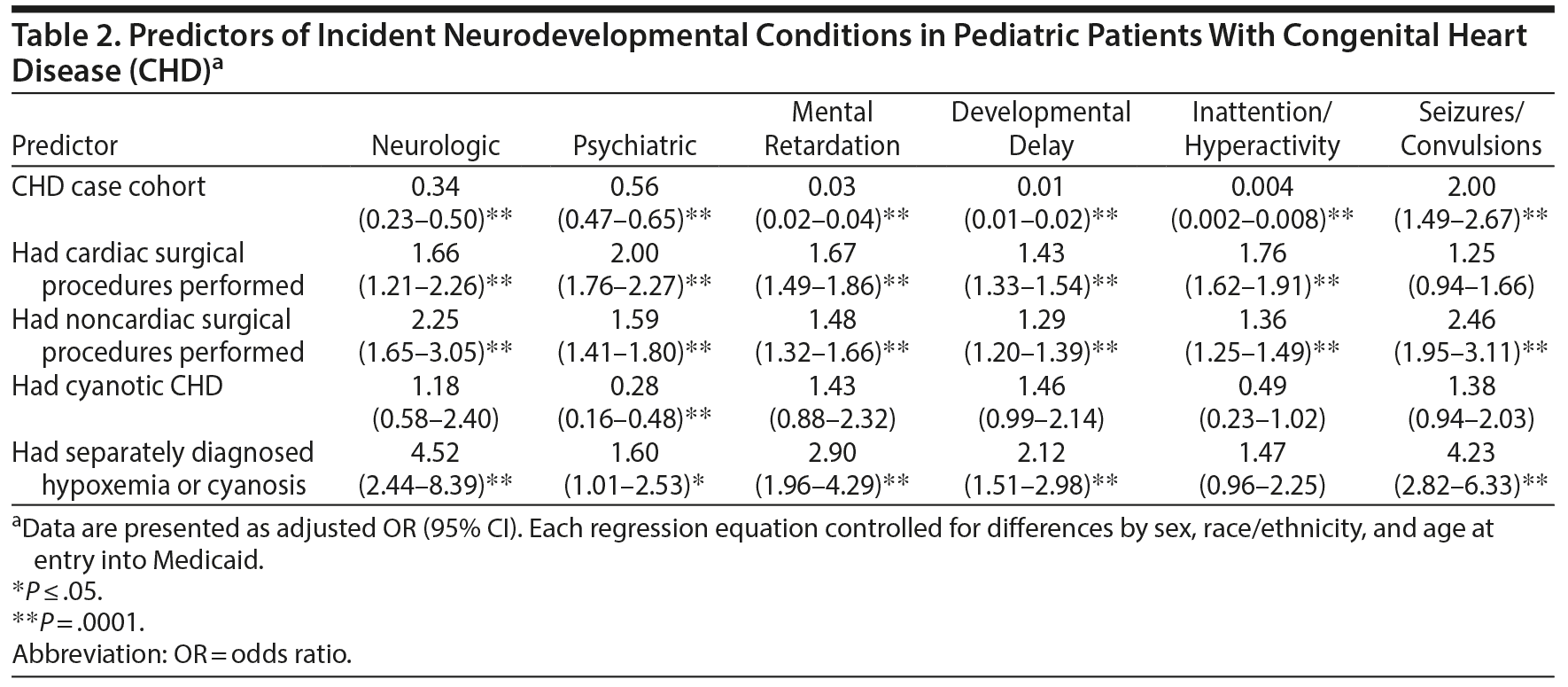
Results: The CHD cohort was significantly less likely to have incident neurologic or psychiatric disorders, mental retardation, developmental delays, or inattention/hyperactivity (adjusted odds ratios [ORs] = 0.34, 0.56, 0.03, 0.01, 0.004, respectively) but was more likely to have incident seizures (OR = 2.00) compared to controls. Exposure to both cardiac and noncardiac surgical intervention was associated with a significantly increased risk of developing neurologic or psychiatric disorders, mental retardation, developmental delays, or inattention/hyperactivity (cardiac ORs = 1.66, 2.00, 1.67, 1.43, 1.76, respectively) (noncardiac ORs = 2.25, 1.59, 1.48, 1.29, 1.36, 2.46, respectively). Any documented hypoxemia was associated with a significantly increased risk of developing 5 of the neurodevelopmental conditions (neurologic OR = 4.52, psychiatric OR = 1.60, mental retardation OR = 2.90, developmental delay OR = 2.12, seizures OR = 4.23).
Conclusion: Practitioners should maintain vigilant surveillance of all CHD patients, especially those exposed to surgical procedures or experiencing hypoxemia, to identify any neurodevelopmental issues early and address them promptly.
Long-Term Neurodevelopmental Outcomes in Children
and Adolescents With Congenital Heart Disease
ABSTRACT
Background: Effective medical and surgical management of pediatric congenital heart disease (CHD) to reduce long-term adverse neurodevelopmental outcomes is an important clinical objective in primary and specialty health care. We identify clinical predictors associated with an increased risk of 6 long-term neurodevelopmental outcomes in children with CHD compared to the general pediatric Medicaid population.
Method: South Carolina’s retrospective, 15-year Medicaid data set (January 1, 1996–December 31, 2010) for 19,947 patients aged ≤ 17 years diagnosed with ≥ 1 CHD lesions (on the basis of International Classification of Diseases, Ninth Revision, Clinical Modification codes) were compared to 19,948 patients without CHD matched on age at entry into and duration in Medicaid using logistic and Cox proportional hazards regression.
Results: The CHD cohort was significantly less likely to have incident neurologic or psychiatric disorders, mental retardation, developmental delays, or inattention/hyperactivity (adjusted odds ratios [ORs] = 0.34, 0.56, 0.03, 0.01, 0.004, respectively) but was more likely to have incident seizures (OR = 2.00) compared to controls. Exposure to both cardiac and noncardiac surgical intervention was associated with a significantly increased risk of developing neurologic or psychiatric disorders, mental retardation, developmental delays, or inattention/hyperactivity (cardiac ORs = 1.66, 2.00, 1.67, 1.43, 1.76, respectively) (noncardiac ORs = 2.25, 1.59, 1.48, 1.29, 1.36, 2.46, respectively). Any documented hypoxemia was associated with a significantly increased risk of developing 5 of the neurodevelopmental conditions (neurologic OR = 4.52, psychiatric OR = 1.60, mental retardation OR = 2.90, developmental delay OR = 2.12, seizures OR = 4.23).
Conclusion: Practitioners should maintain vigilant surveillance of all CHD patients, especially those exposed to surgical procedures or experiencing hypoxemia, to identify any neurodevelopmental issues early and address them promptly.
Prim Care Companion CNS Disord
2015;17(5):doi:10.4088/PCC.15m01842
© Copyright 2015 Physicians Postgraduate Press, Inc.
aDepartments of Neuropsychiatry and Behavioral Science and Pediatrics, University of South Carolina School of Medicine, Columbia
bDepartment of Medicine, Division of Cardiology, University of Louisville School of Medicine, Louisville, Kentucky
cDepartment of Internal Medicine, University of Virginia, Charlottesville
dDepartment of Epidemiology and Biostatistics, University of South Carolina Arnold School of Public Health, Columbia
*Corresponding author: Jeanette M. Jerrell, PhD, Department of Neuropsychiatry and Behavioral Science, University of South Carolina School of Medicine, 15 Medical Park, Ste 301, Columbia, SC 29203 ([email protected]).
Because pediatric patients with congenital heart disease (CHD) are surviving longer due to advances in pharmacologic, medical, and surgical management, the long-term noncardiac complications of these lesions have become an important topic of investigation.1,2 Patients with CHD who are surgery survivors demonstrate modestly increased rates of more “subtle” neurodevelopmental impairments by school age.3–16 However, these sequelae may be associated with myriad causes, eg, the specific type of CHD present, its severity due to cyanosis, how early the surgery is performed, the number of surgeries performed, or whether hypoxemia occurred related to anesthesia or any other cause.2–5
Substantial advances in medical and surgical management techniques during the past 2 decades have led pediatric cardiologists to speculate that modern surgical management of CHD may encumber fewer neurodevelopmental sequelae than were noted in previous studies.2,3,6 Several investigators have called for longitudinal research on the risk factors associated with neurodevelopmental outcomes and cardiac surgical management in pediatric patients with CHD.5,12,16 To our knowledge, no previous studies have identified these relative risk factors over time in a population-based investigation. Herein, we explore the incidence of various neurodevelopmental outcomes related to diagnosed CHD and surgical management in retrospective, Medicaid, population-based cohorts followed over a 15-year period in order to better understand and anticipate the potential impact of these developments on psychiatric outcomes.
METHOD
Access to health care in South Carolina is funded predominantly by public (for low-income families) or private insurance payers, which cover about 90% and 10%, respectively, of the children and adolescents residing in the state.17 Once a child is diagnosed with a serious or chronically disabling condition, including CHD, genetic syndromes, and various developmental disabilities, the family can apply for Medicaid coverage of his or her special medical needs regardless of income. Previously, the accruing treated prevalence of pediatric CHD cases within South Carolina has been documented,18 and the management of several specific lesions or complications in this population (eg, patent ductus arteriosus, atrial septal defects, or supraventricular tachycardia) has been characterized using the South Carolina Medicaid database.19–21
Study Population
Data for this study were obtained retrospectively from the South Carolina Medicaid database during a 15-year period from January 1, 1996, through December 31, 2010. Medical claims were used to identify a service encounter (inpatient, outpatient, or emergency), date of service, and the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9) diagnosis codes related to each visit. To identify cases of CHD, inclusion criteria were aged ≤ 17 years, continuous enrollment in Medicaid for 9 months in each calendar year, and at least 1 initial service encounter with an assigned diagnosis of CHD (ie, atrial septal defects; ventricular septal defect; patent ductus arteriosus; hypoplastic left heart syndrome; single/common ventricle; coarctation of the aorta; transposition of the great arteries; double outlet right ventricle; tetralogy of Fallot; Ebstein’s anomaly; atrioventricular canal/endocardial cushion defect; total anomalous pulmonary venous return; aortic arch anomaly; coronary artery anomaly; truncus arteriosus; pulmonary atresia; atrial stenosis; pulmonary stenosis; cor triatriatum; dextrocardia; tricuspid atresia, stenosis, or regurgitation; interrupted aortic arch; left superior vena cava; and pulmonary insufficiency).
Following the methods of previous epidemiologic investigators, definitions of Mitchell et al22 and Pradat et al23 were used to select CHD cases, omitting bicuspid aortic valve, mitral valve prolapse, Marfan syndrome, cardiomyopathy, chromosomal anomalies/syndromes, and congenital arrhythmia cases, since they are not considered traditionally defined CHDs in pediatric cardiology. These Medicaid data files are routinely cleaned to eliminate duplicate visit records and frequently updated before being made available for analysis. This study was approved by the University of South Carolina Institutional Review Board, Columbia, as exempt from human subject research guidelines (45 Code of Federal Regulations part 46) due to the use of deidentified, retrospective data.
From the same Medicaid population aged 0–17 years and the same time period, medical claims were also used to identify a group of pediatric patients eligible for Medicaid 9 months per calendar year who had service encounters but no diagnosis of any CHD lesion. These control cases were then matched to the CHD cohort using a propensity score technique to randomly select non-CHD individuals matched 1:1 to the CHD cohort on age at entry into the Medicaid system and total months of enrollment in Medicaid.24
Definition of Dependent and Independent Variables
In previous clinical studies of cardiac surgery survivors, neurodevelopmental sequelae were operationalized using neuropsychological testing, behavior rating scales, and information on school performance or problems. Since we are using an observational data set and do not have neuropsychological testing, rating scales, and information on school performance or problems available on the whole population of children served through Medicaid, we defined incident neurodevelopmental outcomes as conditions that would be serious enough to be diagnosed by a treating physician, ie, specific developmental delays; severe intellectual deficits diagnosed as mental retardation; neurologic, cognitive, and central nervous system symptoms diagnosed as alterations of consciousness, hallucinations, seizures/convulsions, or ischemic/cerebrovascular events; various hyperactivity or attentional deficits; and behavioral/emotional problems or any form of mental illness (eg, anxiety, depression, or psychosis).
Our analyses employed the following independent variables, which were controlled for in all statistical analyses: cardiac surgical procedures documented using the current procedural terminology codes for atrial septal defect surgical or transcatheter closure, patent ductus arteriosus repair, any cardiac valve repair, coronary artery anomalies, complex cardiac anomalies, ventricular septal defects, venous or aortic anomalies, shunting, truncus arteriosus, or pulmonary artery repairs, as well as any noncardiac surgeries documented (eg, orthopedic, otolaryngology), severe/cyanotic CHDs, hypoxemia diagnosed/documented separately from surgery or a cyanotic CHD, and any cyanosis diagnosed in the control group. Hypoxemia and cyanosis documented separately were captured for analysis because many patients had one diagnosed condition but not the other, and we wanted to control for the independent effect of these complications to the extent possible. Hypoxemia was significantly correlated with having multiple surgeries, so it may be related to surgical anesthesia, but we could not directly distinguish transcatheter interventions from open heart surgery requiring anesthesia in all of the current procedural terminology codes used.
Statistical Analyses
Preliminary descriptive analyses were performed to determine the data distribution and prevalence of these variables. Due to the low prevalence of some neurodevelopmental conditions diagnosed in the cohort, we combined these and explored the development of 6 broadly defined outcome categories using multiple variable regression analyses: any neurologic diagnoses (ie, ischemic/cerebrovascular disorders, alterations of consciousness, or hallucinations), psychiatric disorders (ie, depression, psychosis), inattention/hyperactivity disorders, specific developmental delays, mental retardation, or seizures/convulsions.
Collinearity among predictor variables for the multivariable analysis was assessed and found not to be present. Adjusted odds ratios (ORs) and associated 95% CIs are reported where adjusted OR > 1 indicates a higher risk of development of the outcome condition. P values < .05 (2-sided tests) were considered statistically significant, and all statistical analyses were performed using SAS version 9.3 (SAS Institute, Cary, North Carolina).
RESULTS
The combined cohort, consisting of 39,895 cases, was very heterogeneous. As shown in Table 1, 54% of the CHD cohort was female, and 43% was black. There was much variability in the age at CHD diagnosis, with a median of 0 months at CHD diagnosis and a mean age of 4.7 years (SD = 6.3), indicating that many CHDs were diagnosed at birth and in infants, while others were not diagnosed until later in childhood. By comparison, the control group (matched on age at entry into Medicaid, median of 0 months, and duration of eligibility/service in Medicaid, median of 68 months) was 54% male and 52% black. These individual differences were controlled for in each multiple variable regression analysis.

Neurodevelopmental outcomes diagnosed during the 15-year period were as follows: < 0.5% of the CHD cohort had inattention/hyperactivity, developmental delays or mental retardation, or neurologic disorders; 1.2% had seizures/convulsions; and 1.4% had psychiatric disorders (Table 1). In the control group, 19.8% had developmental delays, 15.1% had inattention/hyperactivity, 7.3% had mental retardation, 5.0% had psychiatric disorders (psychosis or depression), 0.7% had neurologic disorders, and 0.6% had seizures/convulsions. Although bicuspid aortic valve, mitral valve prolapse, Marfan syndrome, cardiomyopathy, chromosomal anomalies/syndromes, and congenital arrhythmia cases were omitted from the CHD cohort since they are not considered traditionally defined CHDs, they were eligible to be chosen for the control group. Marfan syndrome, Down’s syndrome, and other chromosomal anomalies/syndromes constituted 1.7% of the control group, which might explain the higher rates of developmental disabilities, inattention/hyperactivity, and mental retardation.
Cyanotic CHDs (ie, hypoplastic left heart syndrome, single/common ventricle, truncus arteriosus, transposition of the great arteries, double outlet right ventricle, tetralogy of Fallot, Ebstein’s anomaly, atrioventricular canal/endocardial cushion defect, total anomalous pulmonary venous return) were present in 9.7% of the CHD cohort. Any-cause cyanosis was diagnosed separately in only 0.4% of the control group. Any-cause hypoxemia was diagnosed separately in 2.2% of the CHD cohort and in 0.7% of the control group. Cardiac surgical intervention was required in 9.1% of the CHD cases and in 41.7% of the control cases. The relatively high number of cardiac surgeries in the control cases might again be related to the presence of cardiac disorders among individuals with bicuspid aortic valve, mitral valve prolapse, Marfan syndrome, cardiomyopathy, chromosomal anomalies/syndromes, and congenital arrhythmia as well as other acquired cardiac conditions. Noncardiac surgical intervention was required in 44.5% of the CHD cases and in 35.3% of the control cases.
Multivariate Analyses
Compared to the control group, the CHD cohort was significantly less likely to have incident neurologic or psychiatric disorders, mental retardation, developmental delays, or inattention/hyperactivity (adjusted ORs = 0.34, 0.56, 0.03, 0.01, 0.004, respectively), but was more likely to have incident seizures/convulsions (OR = 2.00) (Table 2). Having a cardiac surgical procedure was associated with an increased risk of developing neurologic or psychiatric disorders, mental retardation, developmental delays, or inattention/hyperactivity (ORs = 1.66, 2.00, 1.67, 1.43, 1.76, respectively). Exposure to a noncardiac surgical procedure was associated with a significantly increased risk of having all of the neurodevelopmental outcomes investigated (neurologic OR = 2.25, psychiatric OR = 1.59, mental retardation OR = 1.48, developmental delay = 1.29, inattention/hyperactivity = 1.36, seizures/convulsions = 2.46). Having a cyanotic CHD was associated with a lower risk of the child developing psychiatric disorders (OR = 0.28), but having separately diagnosed hypoxemia was associated with an increased risk of developing 5 of the neurodevelopmental conditions (neurologic OR = 4.52, psychiatric OR = 1.60, mental retardation OR = 2.90, developmental delay OR = 2.12, seizures/convulsions OR = 4.23).
DISCUSSION
These results provide additional empirical evidence regarding speculation that evolving surgical management of pediatric CHDs would encumber fewer adverse neurodevelopmental outcomes than were noted in previous studies.2,3,5,6,12,16 The incidence of most neurodevelopmental conditions examined in this large and heterogeneous CHD cohort was very low, while the rate of exposure to surgical intervention within the CHD cohort appears to be higher than previously reported.2,3,6,9,10,12,14,25 Furthermore, comparative analyses with a randomly selected cohort of children without CHD in the Medicaid system indicated a higher incidence in non-CHD cases of developmental delays, inattention/hyperactivity, mental retardation, and psychiatric disorders. Therefore, neurodevelopmental outcomes do not appear to be independently associated with the presence of CHD lesions, but they are associated with any-cause hypoxemia. Moreover, the rate of seizures was higher in the CHD cohort but appears to be associated with exposure to noncardiac surgical procedures.
This investigation expands the existing body of knowledge not only regarding the incidence of neurodevelopmental sequelae over time in a heterogeneous population of pediatric CHD patients, but also on the risk factors that may be significantly associated with these outcomes. Our finding of only 11 cases of diagnosed mental retardation in the CHD cohort comports with the existing literature on children with cyanotic CHDs having lower intellectual functioning, which is still within the normal range.2,6 Only the incidence of diagnosed inattention/hyperactivity and developmental delay conditions in combination, which constituted only 0.5% of the cohort, does not comport with previous clinical studies13–16 of surgery survivors, but those clinical groups were small and the incidence of these conditions may have been attributable to factors not measured in the study. Furthermore, seizures/convulsions in this cohort were significantly more likely to occur in those with cyanotic CHDs compared to those with acyanotic heart defects, which diverges from previous findings in studies8,10,12,15 reporting only subtle or no significant differences between children with cyanotic and acyanotic CHDs.
Strengths and Limitations
There are several ways in which this research cohort differs from those of previous clinical investigations. This Medicaid population-based cohort of combined CHD cases and controls was followed longitudinally in a retrospective database, so there was much greater variance in the age at CHD diagnosis and the severity of lesions than in previous clinical studies and in the timeframe observed for the emergence of the neurodevelopmental outcomes. The combined cohort was large and heterogeneous, including acyanotic and cyanotic lesions and those separately diagnosed with cyanosis and hypoxemia, and entailed considerable variability in exposure to surgery (cardiac and noncardiac) over time. The combined cohort also included a higher percentage of blacks than previous studies, except those conducted in Atlanta, Georgia.26,27 Finally, the combined cohort is representative of pediatric patients in routine care settings in the southeastern states and other small states with predominantly small-city and rural populations in terms of age, sex, racial demographics, and Medicaid eligibility.28,29
Observational studies using large databases can complement evidence generated through randomized clinical trials by assessing treatment outcomes in patients encountered in routine clinical practice, although they are more exposed to bias and are lower on the evidence hierarchy than randomized trials.30 While we tried to control for many sources of bias/error, the primary limitation of the study method is that the children and adolescents who had neurodevelopmental conditions requiring neuropsychological assessments or school functioning data for identification and those who dropped out of treatment, who were periodically ineligible for Medicaid, or who died before they were registered in Medicaid coverage are not represented in the study. We relied on physician-assigned ICD-9 codes in the Medicaid billing dataset to categorize patients, which may have contributed some degree of ascertainment bias or coding invalidity and may represent an underestimate of the incidence of outcome or related conditions. Hospital-designated surgical procedure codes were used, but we had no details available regarding, for example, exposure to anesthesia to incorporate into the research analyses. Other clinical factors not measured in this study (eg, cranial perfusion in cardiopulmonary bypass or complications from noncardiac surgical procedures, sepsis, acidosis, family genetics or history/dynamics, preterm birth status) may be associated with the subsequent development of these neuropsychiatric conditions. However, the use of Medicaid databases in epidemiologic studies has been previously validated.31 Furthermore, these results report associations and, as a result, directions of causality cannot be inferred. Finally, these results may not be generalizable to patients with referral or other access-to-care limitations or no financial coverage for their health care services.
CONCLUSION
Exposure to cardiac and noncardiac surgical procedures is associated with development of long-term neurodevelopmental sequelae in the combined CHD and control cohorts. However, the presence of CHD lesions imparts a relatively low liability for incident neurodevelopmental outcomes over time in the combined cohorts. Increasing the knowledge base about factors affecting the development of long-term adverse neurodevelopmental sequelae in children with CHD not only provides pediatric generalists and specialists with feedback about the intended/unintended outcomes of existing medical and surgical management practices, but also underscores certain strategies to optimize long-term outcomes in the high-risk group. Although we have estimated only a low incidence/risk for severe neurodevelopmental impairments among children with CHDs and surgery survivors by adolescence, previous evidence has repeatedly demonstrated a modestly increased risk of more subtle neurodevelopmental impairments requiring assessment and supportive intervention services by school age. Therefore, astute practitioners should maintain vigilant surveillance of patients with CHD, especially those exposed to surgical procedures, and any child with a documented history of hypoxemia throughout childhood and adolescence to identify neurodevelopmental issues early and address them promptly.
Submitted: May 26, 2015; accepted June 23, 2015.
Published online: October 22, 2015.
Potential conflicts of interest: None reported.
Funding/support: Funding for these analyses was provided through a Clinical Incentive Research Grant from the University of South Carolina Office of the Provost.
Role of the sponsor: The funding agency had no involvement in the conduct of the research or the reporting of these results.
Disclaimer: The views expressed do not necessarily represent those of the funding agency or official findings of the South Carolina Department of Health and Human Services (Medicaid).
Additional information: The Medicaid dataset used in these analyses is not available to other investigators due to the confidentiality restrictions imposed by the South Carolina Department of Health and Human Services.
REFERENCES
1. Earing MG. Congenital heart disease in adults. In: Kleigman RM, Stanton BF, Schor NF, et al, eds. Nelson Textbook of Pediatrics. 19th ed, chapter 428.1. Philadelphia, PA: Elsevier Saunders; 2011:1605–1609.
2. Miatton M, De Wolf D, François K, et al. Neurocognitive consequences of surgically corrected congenital heart defects: a review. Neuropsychol Rev. 2006;16(2):65–85. doi:10.1007/s11065-006-9005-7 PubMed
3. Massaro AN, El-Dib M, Glass P, et al. Factors associated with adverse neurodevelopmental outcomes in infants with congenital heart disease. Brain Dev. 2008;30(7):437–446. doi:10.1016/j.braindev.2007.12.013 PubMed
4. Donofrio MT, Massaro AN. Impact of congenital heart disease on brain development and neurodevelopmental outcome. Int J Pediatr. 2010;2010:1–13. doi:10.1155/2010/359390 PubMed
5. Morriss FH Jr, Saha S, Bell EF, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Surgery and neurodevelopmental outcome of very low birth-weight infants. JAMA Pediatr. 2014;168(8):746–754. doi:10.1001/jamapediatrics.2014.307 PubMed
6. Wickremasinghe AC, Rogers EE, Piecuch RE, et al. Neurodevelopmental outcomes following two different treatment approaches (early ligation and selective ligation) for patent ductus arteriosus. J Pediatr. 2012;161(6):1065–1072. doi:10.1016/j.jpeds.2012.05.062 PubMed
7. Miatton M, De Wolf D, François K, et al. Neurocognitive consequences of surgically corrected congenital heart defects: a review. Neuropsychol Rev. 2006;16(2):65–85. doi:10.1007/s11065-006-9005-7 PubMed
8. DiMaggio C, Sun LS, Li G. Early childhood exposure to anesthesia and risk of developmental and behavioral disorders in a sibling birth cohort. Anesth Analg. 2011;113(5):1143–1151. doi:10.1213/ANE.0b013e3182147f42 PubMed
9. Sarrechia I, Miatton M, De Wolf D, et al. Neurobehavioural functioning in school-aged children with a corrected septal heart defect. Acta Cardiol. 2013;68(1):23–30. PubMed
10. Miatton M, De Wolf D, François K, et al. Intellectual, neuropsychological, and behavioral functioning in children with tetralogy of Fallot. J Thorac Cardiovasc Surg. 2007;133(2):449–455. doi:10.1016/j.jtcvs.2006.10.006 PubMed
11. Brosig CL, Mussatto KA, Kuhn EM, et al. Neurodevelopmental outcome in preschool survivors of complex congenital heart disease: implications for clinical practice. J Pediatr Health Care. 2007;21(1):3–12. doi:10.1016/j.pedhc.2006.03.008 PubMed
12. Bellinger DC, Wypij D, Rivkin MJ, et al. Adolescents with d-transposition of the great arteries corrected with the arterial switch procedure: neuropsychological assessment and structural brain imaging. Circulation. 2011;124(12):1361–1369. doi:10.1161/CIRCULATIONAHA.111.026963 PubMed
13. Hamilton RM, Rosenthal E, Hulpke-Wette M, et al; European Network of Hyperkinetic Disorders. Cardiovascular considerations of attention-deficit/hyperactivity disorder medications: a report of the European Network on Hyperactivity Disorders work group, European Attention-Deficit/Hyperactivity Disorder Guidelines Group on attention-deficit/hyperactivity disorder drug safety meeting. Cardiol Young. 2012;22(1):63–70. doi:10.1017/S1047951111000928 PubMed
14. Shillingford AJ, Glanzman MM, Ittenbach RF, et al. Inattention, hyperactivity, and school performance in a population of school-age children with complex congenital heart disease. Pediatrics. 2008;121(4):e759–e767. doi:10.1542/peds.2007-1066 PubMed
15. Kirshbom PM, Flynn TB, Clancy RR, et al. Late neurodevelopmental outcome after repair of total anomalous pulmonary venous connection. J Thorac Cardiovasc Surg. 2005;129(5):1091–1097. doi:10.1016/j.jtcvs.2004.08.013 PubMed
16. Hövels-Gürich HH, Konrad K, Skorzenski D, et al. Attentional dysfunction in children after corrective cardiac surgery in infancy. Ann Thorac Surg. 2007;83(4):1425–1430. doi:10.1016/j.athoracsur.2006.10.069 PubMed
17. 2010 Census summary file 2, demographic profile 1 by state. American FactFinder Web site. http://factfinder2.census.gov/faces/tableservices/jsf/pages/productview.xhtml?pid=DEC_10_SF2_SF2DP1&prodType=table. Accessed February 4, 2013.
18. Shuler CO, Black GB, Jerrell JM. Population-based treated prevalence of congenital heart disease in a pediatric cohort. Pediatr Cardiol. 2013;34(3):606–611. doi:10.1007/s00246-012-0505-3 PubMed
19. Tripathi A, Black GB, Park YM, et al. Prevalence and management of patent ductus arteriosus in a pediatric Medicaid cohort. Clin Cardiol. 2013;36(9):502–506. doi:10.1002/clc.22150 PubMed
20. Shuler CO, Tripathi A, Black GB, et al. Prevalence of treatment, risk factors, and management of atrial septal defects in a pediatric Medicaid cohort. Pediatr Cardiol. 2013;34(7):1723–1728. doi:10.1007/s00246-013-0705-5 PubMed
21. Tripathi A, Black GB, Park YM, et al. Factors associated with the occurrence and treatment of supraventricular tachycardia in a pediatric congenital heart disease cohort. Pediatr Cardiol. 2014;35(2):368–373. doi:10.1007/s00246-013-0784-3 PubMed
22. Mitchell SC, Korones SB, Berendes HW. Congenital heart disease in 56,109 births: incidence and natural history. Circulation. 1971;43(3):323–332. doi:10.1161/01.CIR.43.3.323 PubMed
23. Pradat P, Francannet C, Harris JA, et al. The epidemiology of cardiovascular defects, part I: a study based on data from three large registries of congenital malformations. Pediatr Cardiol. 2003;24(3):195–221. doi:10.1007/s00246-002-9401-6 PubMed
24. Coca-Perraillon M. Local and global optimal propensity score matching. Global Forum 2007 Statistics and Data Analysis. SAS Web site. http://www2.sas.com/proceedings/forum2007/185-2007.pdf. Accessed December 3, 2011.
25. Olsen M, Sørensen HT, Hjortdal VE, et al. Congenital heart defects and developmental and other psychiatric disorders: a Danish nationwide cohort study. Circulation. 2011;124(16):1706–1712. doi:10.1161/CIRCULATIONAHA.110.002832 PubMed
26. Shuler CO, Tripathi A, Black GB, et al. Individual risk factors and complexity associated with congenital heart disease in a pediatric Medicaid cohort. South Med J. 2013;106(7):385–390. doi:10.1097/SMJ.0b013e31829bd0bc PubMed
27. Botto LD, Correa A, Erickson JD. Racial and temporal variations in the prevalence of heart defects. Pediatrics. 2001;107(3):e32. doi:10.1542/peds.107.3.e32 PubMed
28. Health insurance coverage for children under 19 years of age, at or below 200% of poverty by state, 2006–2008 and 2009–2011. Census.gov Web site. http://www.census.gov/hhes/www/hlthins/data/children/low-income.html. Accessed January 30, 2013.
29. Table D1: children under 18 and their designated parents and Table D2: children under 18 and their designated parents—characteristics of families (including region). Census.gov Web site. http://www.census.gov/hhes/socdemo/children/data/sipp/well2009/tables.html. Accessed January 30, 2013.
30. Takahashi Y, Nishida Y, Asai S. Utilization of health care databases for pharmacoepidemiology. Eur J Clin Pharmacol. 2012;68(2):123–129. doi:10.1007/s00228-011-1088-2 PubMed
31. Hennessy S, Leonard CE, Freeman CP, et al. Validation of diagnostic codes for outpatient-originating sudden cardiac death and ventricular arrhythmia in Medicaid and Medicare claims data. Pharmacoepidemiol Drug Saf. 2010;19(6):555–562. doi:10.1002/pds.1869 PubMed
Enjoy this premium PDF as part of your membership benefits!


