Objective: To review currently available formulations and emphasize unmet needs in the pharmacologic management of attention-deficit/hyperactivity disorder (ADHD).
Data Sources: Publications and clinical trials were identified through PubMed and ClinicalTrials.gov, respectively. A Web-based search identified prescribing information for approved agents for treating ADHD, along with relevant guidelines and diagnostic criteria.
Study Selection: The following search terms were used: (1) ADHD or attention-deficit/hyperactivity disorder or ADD or attention deficit hyperactivity disorder and/or (2) amphetamine or methylphenidate or atomoxetine or guanfacine or clonidine. Additional searches were performed using product brand names, and clinical trial was applied as a filter. Relevant studies were only included if published in English-language peer-reviewed journals and if involved agents were currently available (or pursuing approval) in the United States. Reviews of literature prior to 2005 and from 2005 to 2008 have been published previously; therefore, the present search focused on studies published from January 2009 through May 2016. In addition, reference lists of review articles and relevant studies were also examined to help identify additional studies.
Data Extraction: A total of 578 publications were identified from the PubMed search, from which 426 publications were initially excluded based on review of the title and abstract. Reasons for exclusion included a focus on comorbid disorders, specific subpopulations, endpoints unrelated to improving ADHD symptomatology (eg, executive function, cognition, substance use), or quality of life measures. A more thorough assessment of the remaining citations, including publications and prescribing information, produced the final 219.
Results: Pharmacotherapy with stimulant and nonstimulant options is the most common approach for treating ADHD in adults and children. Stimulants are mostly formulated as either tablets or capsules; however, the newer generation includes transdermal patches, oral suspensions (liquids), chewable tablets, and orally disintegrating tablets. Nonstimulants are available in oral capsule (atomoxetine) and tablet (guanfacine and clonidine) formulations.
Conclusions: Despite the broad range of treatment options currently available, nonadherence remains a significant problem in ADHD. While evidence is currently lacking, the availability of new formulations may improve adherence.
Current and Investigational Medication Delivery Systems
for Treating Attention-Deficit/Hyperactivity Disorder
ABSTRACT
Objective: To review currently available formulations and emphasize unmet needs in the pharmacologic management of attention-deficit/hyperactivity disorder (ADHD).
Data Sources: Publications and clinical trials were identified through PubMed and ClinicalTrials.gov, respectively. A Web-based search identified prescribing information for approved agents for treating ADHD, along with relevant guidelines and diagnostic criteria.
Study Selection: The following search terms were used: (1) ADHD or attention-deficit/hyperactivity disorder or ADD or attention deficit hyperactivity disorder and/or (2) amphetamine or methylphenidate or atomoxetine or guanfacine or clonidine. Additional searches were performed using product brand names, and clinical trial was applied as a filter. Relevant studies were only included if published in English-language peer-reviewed journals and if involved agents were currently available (or pursuing approval) in the United States. Reviews of literature prior to 2005 and from 2005 to 2008 have been published previously; therefore, the present search focused on studies published from January 2009 through May 2016. In addition, reference lists of review articles and relevant studies were also examined to help identify additional studies.
Data Extraction: A total of 578 publications were identified from the PubMed search, from which 426 publications were initially excluded based on review of the title and abstract. Reasons for exclusion included a focus on comorbid disorders, specific subpopulations, endpoints unrelated to improving ADHD symptomatology (eg, executive function, cognition, substance use), or quality of life measures. A more thorough assessment of the remaining citations, including publications and prescribing information, produced the final 219.
Results: Pharmacotherapy with stimulant and nonstimulant options is the most common approach for treating ADHD in adults and children. Stimulants are mostly formulated as either tablets or capsules; however, the newer generation includes transdermal patches, oral suspensions (liquids), chewable tablets, and orally disintegrating tablets. Nonstimulants are available in oral capsule (atomoxetine) and tablet (guanfacine and clonidine) formulations.
Conclusions: Despite the broad range of treatment options currently available, nonadherence remains a significant problem in ADHD. While evidence is currently lacking, the availability of new formulations may improve adherence.
Prim Care Companion CNS Disord 2016;18(4):doi:10.4088/PCC.16r01979
© Copyright 2016 Physicians Postgraduate Press, Inc
aDepartment of Psychiatry at Texas Tech Health Sciences Center Medical School, Midland, Texas
bHouston Clinical Trials, Houston, Texas
*Corresponding author: Rakesh Jain, MD, MPH, 2500 W William Cannon Dr, Suite 505, Austin, TX 78745 ([email protected]).
Attention-deficit/hyperactivity disorder (ADHD), which is characterized by impairments in maintaining sustained attention, modulating activity level, and exercising impulse control,1 is the most prevalent neurobehavioral disorder in children.2 A study3 conducted in 2011 estimated that 11% of US school-aged children had ever received a diagnosis of ADHD. In addition, although ADHD symptoms tend to decline with age,4 symptoms and associated impairment persist into adulthood in approximately 50% of cases,5 affecting at least 4% of the adults in the United States.6
While there has been a reported 42% increase in ADHD diagnoses in children since 2003 in the United States, it is unclear if the true prevalence is increasing or if the increased rate reflects overdiagnosis or more disease awareness as a result of the more inclusive guidelines put forth in 2013 with the introduction of the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5).3,7–9 Despite increasing incidence rates, ADHD remains underdiagnosed in some patient populations including children and adults in the United States.10–12 Additionally, the prevalence of ADHD and its subtypes reportedly varies by the socioeconomic and racial and ethnic composition of the population.11,12
ADHD symptoms are often associated with considerable functional impairment. In children and adolescents, symptoms can negatively affect family life, academic achievement, and overall conduct.13–15 Similarly, adults with ADHD tend to have lower educational attainment and higher rates of arrests, traffic violations, unemployment, and divorce.16,17 The annual incremental costs of ADHD have been estimated to range from $143 billion to $266 billion in 2010 US dollars.18
The primary treatment goals for patients with ADHD are improving symptoms and functionality and removing behavioral obstacles through behavioral therapy and pharmacologic agents.19 Behavioral therapies are the first-line option recommended for preschool-aged children, while pharmacologic agents are often the first-line option for school-aged children and adults.2,20 Although most patients exhibit a clinical response to stimulant therapy, some patients respond differently in terms of efficacy or tolerability to the different classes of medication.21 A priori predictors of treatment response would be invaluable,22 but few reliable predictors of response are available, and most therapeutic decisions are based on trial-and-error.23
Currently, more than 20 different pharmacologic options exist from which providers may choose in everyday clinical practice, and there are numerous other stimulant and nonstimulant options in various stages of clinical development.24,25 This systematic review summarizes recent phase 2 through 4 clinical trials of pharmacologic ADHD treatments.
METHOD
Publications and clinical trials were identified through PubMed and ClinicalTrials.gov, respectively. The following search terms were used: (1) ADHD or attention-deficit/hyperactivity disorder or ADD or attention deficit hyperactivity disorder and/or (2) amphetamine or methylphenidate or atomoxetine or guanfacine or clonidine. Additional searches were performed using product brand names, and clinical trial was applied as a filter.
Figure 1 shows the design for the systematic search strategy that was used to identify relevant studies. Relevant studies involved any age group (children, adolescents, or adults) and agents currently available (or pursuing approval) in the United States. Because similar reviews of literature prior to 200526 and from 2005 to 200824 have been published, the present search focused only on studies completed or published in English-language peer-reviewed journals between January 2009 and May 2016. In addition, reference lists of review articles and relevant studies were also examined to help identify additional studies.
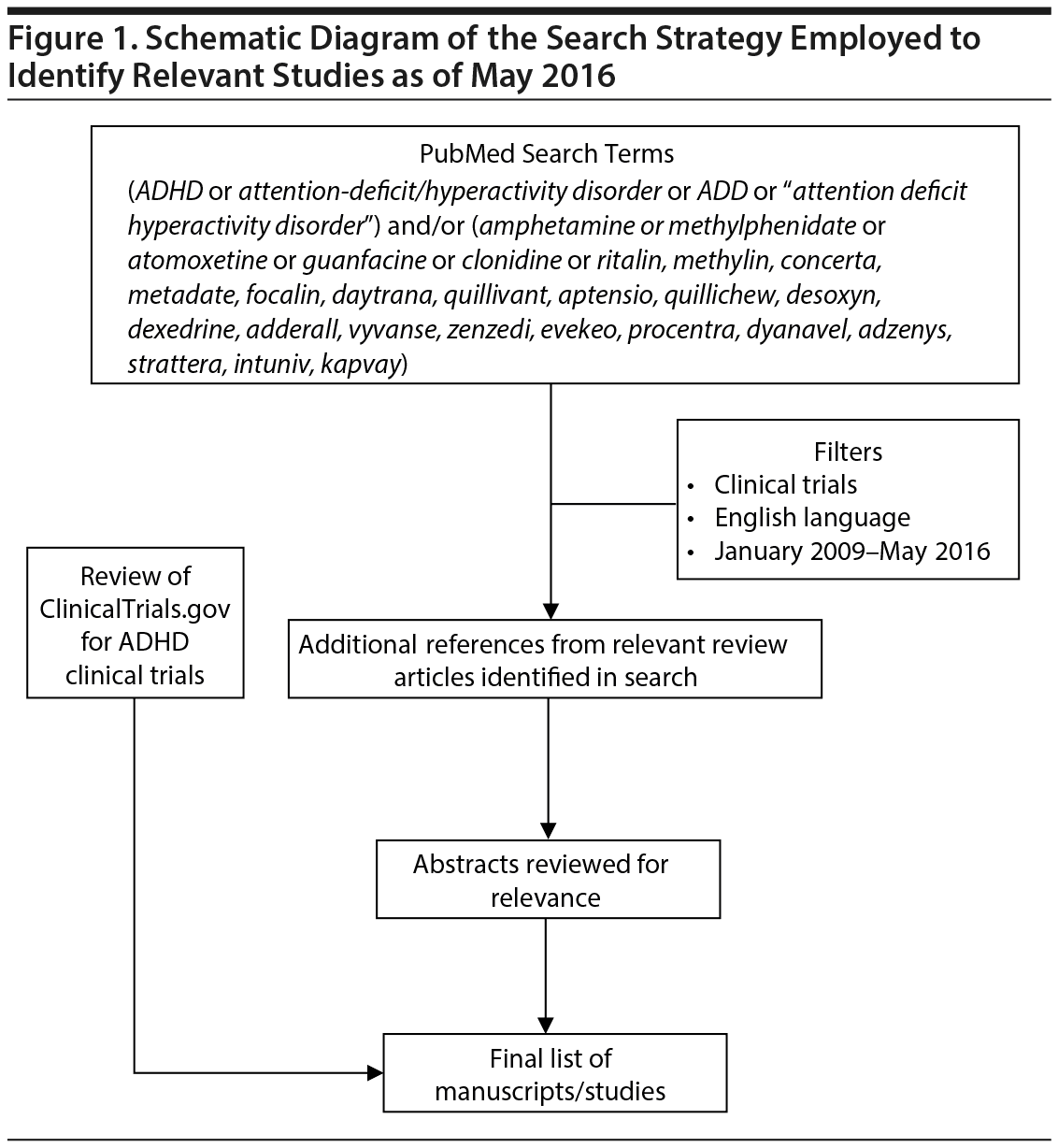
RESULTS
A total of 578 publications (from January 2009 to May 2016) were identified from the PubMed search, from which 426 publications were initially excluded based on review of the title and abstract. Reasons for exclusion included a focus on comorbid disorders, specific subpopulations, endpoints unrelated to improving ADHD symptomatology (eg, executive function, cognition, substance use), or quality of life measures. We also used a Web-based search to identify prescribing information for approved agents for treating ADHD, along with relevant guidelines and diagnostic criteria. A more thorough assessment of the remaining citations produced the final 219.1–219
Stimulants
Stimulants including methylphenidate and dexmethylphenidate and amphetamine derivatives (ie, mixed amphetamine salts, lisdexamfetamine, and dextroamphetamine) are considered first-line therapeutic options for adults and children with ADHD.27,28 Short-term use of these agents reduces symptoms, including short attention span, distractibility, impulsive behavior, hyperactivity, and restlessness, in addition to improving vigilance, cognition, reaction time, response time, response inhibition, and short-term memory.29 In general, stimulants offer greater effectiveness than nonstimulant options,30 and approximately 70% of patients will experience clinical improvement with their initial stimulant treatment.31,32 Stimulants have been shown to also improve performance in some individuals without ADHD.33
Overall, side effects with stimulants tend to be mild to moderate in severity and can be managed by reducing the dose or by modifying the medication administration time.29 Commonly observed side effects, including insomnia, anorexia, nausea, decreased appetite, weight loss, headache, increased blood pressure, elevated heart rate, abdominal pain, irritability, and mood lability, usually emerge soon after beginning treatment and generally subside over time.29 Although there is concern that use of stimulants in children may stunt growth, the results of a recent longitudinal study34 suggest that stimulants do not cause significant changes in growth or adult height. However, in rare instances, stimulants have been associated with more serious side effects including seizures, hypertension, psychosis, and hepatotoxicity.29 In general, stimulants can be used safely in children and adults.35,36
ADHD is thought to result from dopaminergic and noradrenergic imbalances that result in inefficient information processing by pyramidal neurons in the prefrontal cortex.37 Although the exact mechanisms of the therapeutic action of stimulants in ADHD are incompletely understood, it has been hypothesized that they work by increasing extraneuronal catecholamine concentrations in the prefrontal cortex.37,38 Both classes of stimulants (methylphenidate and amphetamine derivatives) increase synaptic dopamine and norepinephrine transmission.38 Dopamine may decrease background noise associated with extraneous input that contributes to inattentive symptoms, and norepinephrine may increase the signal gain associated with task-relevant information.37
Most short-acting stimulant formulations have a duration of action ranging from 3 to 6 hours,29,31,39 which may necessitate multiple doses to manage ADHD symptoms throughout the day. Short-acting formulations are associated with decreased adherence, potential embarrassment or stigma for individuals taking doses during the daytime (eg, during school or work hours), difficulties with storing and handling controlled substances, and increased potential for diversion.39 In order to help overcome some of these drawbacks, numerous long-acting stimulant formulations have been developed, with durations of effect of up to 12 hours.30,40,41 Included among the long-acting stimulant options are multiple controlled-release formulations that combine a rapid onset of action with extended coverage throughout the day (ie, multiphasic release).39 More recent advances with stimulant formulations, including transdermal patches, oral suspensions (liquids), chewable tablets, and orally disintegrating tablets (ODTs), provide patients with alternatives if they cannot or prefer not to swallow tablets or capsules (or prefer not to open and sprinkle capsule contents on food) without compromising efficacy or duration of effect.42
Although there have been no direct comparisons between the abuse potential of short- and long-acting stimulants, some of the characteristics of long-acting stimulants suggest that they could reduce the likelihood of diversion. For instance, the once-daily administration of long-acting stimulants could reduce the supply of medication at schools needed for intraday dosing.39 In addition, controlled release of stimulants may result in less euphoria43,44 because the reinforcing effects of drugs are related to the rate of delivery to the brain.45 Lastly, the long-acting stimulant lisdexamfetamine is a prodrug that must be metabolized to dextroamphetamine in the bloodstream for a therapeutic effect. There was no difference in the pharmacokinetic (PK) profile of d-amphetamine when 50-mg lisdexamfetamine was administered orally or intranasally in a study46 of healthy adults; these attributes may make lisdexamfetamine less attractive to abusers than immediate-release dextroamphetamine.46–48
Methylphenidate. Methylphenidate was first synthesized more than 70 years ago,49 and its efficacy and safety in patients with ADHD have been studied extensively. Numerous methylphenidate formulations of d– isomers (ie, dexmethylphenidate) or racemic mixtures of both d– and l– isomers, including liquids (both solution and suspension), chewable tablets, oral tablets, capsules, an oral disintegrating tablet, and a transdermal patch, are currently approved or in late stages of development (Table 1).
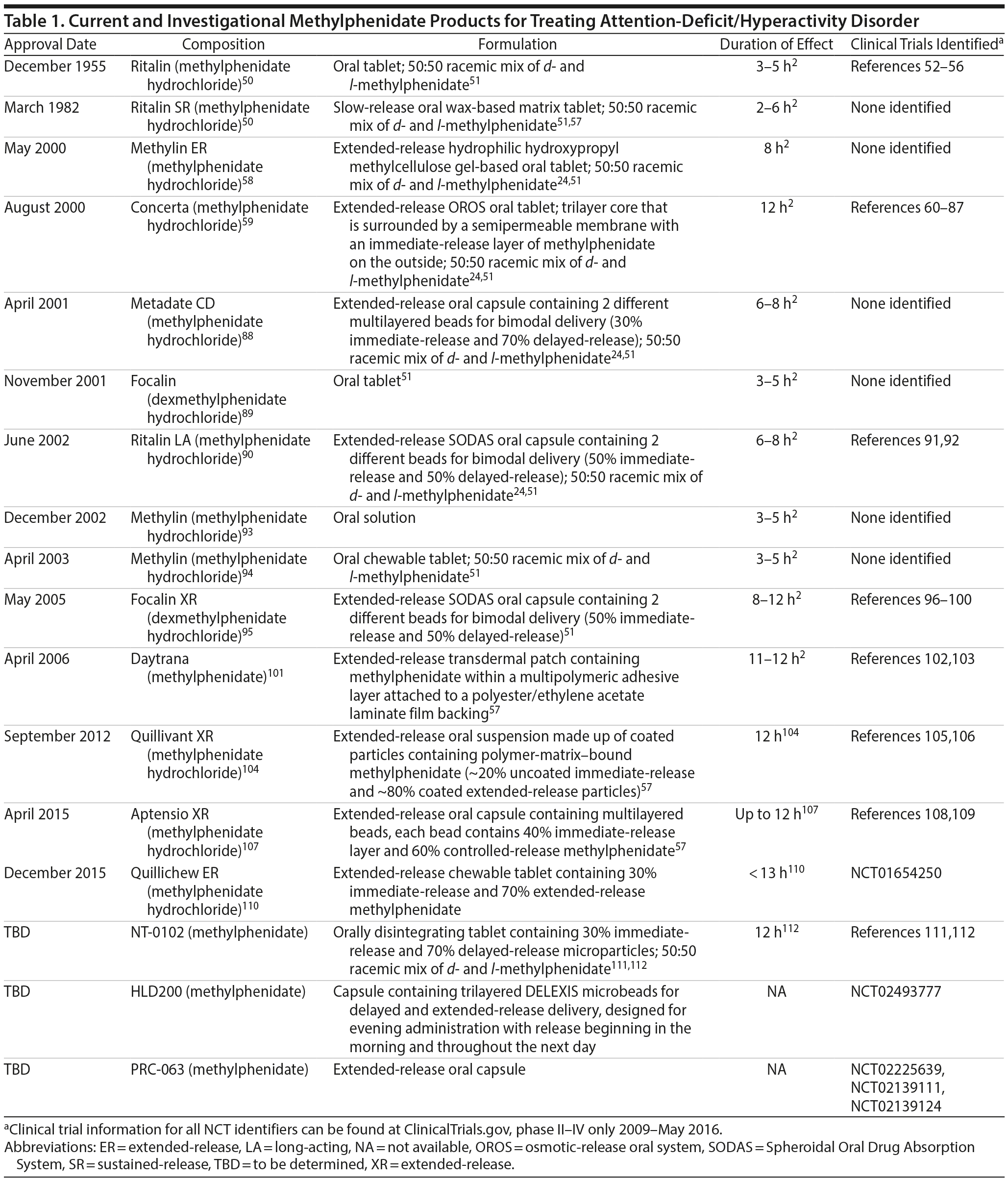
Short-acting methylphenidate formulations, including oral tablets, chewable tablets, and solutions, require frequent dosing to control symptoms throughout the day and produce large peak/trough effects. Nevertheless, these agents may be useful when an additional dose is needed later in the day to supplement a once-daily dose or for those patients who wish to have greater flexibility in dosing.29 Long-acting methylphenidate formulations have a duration of action of 8 to 12 hours.2 The first-generation long-acting methylphenidate formulation (Ritalin SR, Novartis Pharmaceuticals, East Hanover, New Jersey50) used a wax matrix as the modified-release technology to provide a duration of up to 8 hours. However, Ritalin SR has a variable duration of action due to an inconsistent absorption profile of methylphenidate from the wax matrix and is not widely used in clinical practice.113–116
Subsequently, several controlled-release methylphenidate formulations have been developed with a multiphasic release profile. The osmotically controlled-release (OROS) oral methylphenidate tablet formulation (Concerta, Janssen Pharmaceuticals, Titusville, New Jersey59) is designed to deliver methylphenidate in a controlled manner for approximately 10 to 12 hours.117 The OROS formulation does not allow the pill to be crushed, which may present challenges for patients who have difficulty taking oral medications.24 However, it may also reduce the risk of diversion and abuse because it cannot be easily manipulated for more rapid absorption.29,39 Metadate CD (UCB, Smyrna, Georgia88) is a capsule formulation that includes a mixture of immediate- and extended-release beads (30% immediate-release to 70% extended-release ratio). Metadate CD delivers methylphenidate through a 2-phase drug release providing symptom control through ~8 hours.24,88,118 Aptensio XR (Rhodes Pharmaceuticals, Coventry, Rhode Island107) is another capsule formulation; however, it contains multilayer beads (40% as immediate- and 60% extended-release). After administration, methylphenidate concentrations reach an initial maximum at ~2 hours, followed by a second peak ~4 to 6 hours after the first, offering a 12-hour duration of action.108 Other controlled-release capsule formulations (Ritalin LA, Novartis Pharmaceuticals, East Hanover, New Jersey90; Focalin XR, Novartis Pharmaceuticals, East Hanover, New Jersey95) use the spheroidal oral drug absorption system, which involves 50% immediate-release and 50% extended-release beads covered by a polymer coating.24 The capsules containing the controlled-release beads may be opened, allowing for the beads to be sprinkled over certain foods.24 Additionally, 2 new capsule formulations are in clinical development. The first, HLD200, is composed of trilayered DELEXIS microbeads that deliver methylphenidate after an ~8 to 9–hour delay, with a ~17-hour time to maximum plasma concentration, intended for administration in the evening for next-day symptom coverage (ClinicalTrials.gov: NCT02493777, NCT02255513, and NCT01907360, Ironshore [America] Inc, Camana Bay, Cayman Islands, subsidiary of Highland Therapeutics, Berwyn, Pennsylvania.). The second is PRC-063, an extended-release methylphenidate formulation currently being evaluated in adolescents and adults. Available clinical trial data indicate that efficacy assessments to 16 hours after administration and doses up to 100 mg have been evaluated (ClinicalTrials.gov: NCT02225639, NCT02139111, NCT02139124, Rhodes Pharmaceuticals, LP, Coventry, Rhode Island).25,57
Although there are numerous long-acting methylphenidate formulations, most are available only in oral capsule or tablet formulations, which, as previously stated, can present problems for children and adults who prefer not to swallow or have reported difficulty swallowing tablets.119 To that end, many alternative formulations have been developed (eg, transdermal patch, oral solution, oral suspension, chewable tablet) or are in clinical development (eg, long-acting chewable tablet [NWP09, ClinicalTrials.gov: NCT01654250] and a methylphenidate extended-release ODT [XR-ODT] formulation [NT-0102, ClinicalTrials.gov: NCT01835548]).25,111,112
With the methylphenidate transdermal patch system (Daytrana, Shire, Wayne, Pennsylvania101), absorption is unaffected by meals or first-pass metabolism.120 However, considerable intraindividual variability in absorption has been observed.121 Nevertheless, the transdermal patch may offer a lower risk for abuse and diversion120 and allow for greater flexibility in the length of dosing.122 The patch is intended to be worn for 9 hours; it is applied 2 hours before the effects are needed, and the effects persist for up to 3 hours after the patch is removed.
The extended-release oral suspension of methylphenidate (Quillivant XR, NextWave Pharmaceuticals, Inc, Cupertino, California) offers once-daily dosing104 with a liquid formulation, which may be especially suitable for those who have difficulty swallowing tablets or capsules. However, because this product is a suspension, vigorous shaking of the bottle is recommended before each dose to ensure that the proper amount is administered.104 Recently, clinical studies have evaluated the safety and efficacy of the extended-release chewable tablet formulation (Quillichew ER [NWP09], Pfizer Inc, New York, New York; ClinicalTrials.gov: NCT01654250). This formulation was approved by the US Food and Drug Administration (FDA) in December 2015.
NT-0102 is composed of a racemic mixture of d-:l- methylphenidate and is the first extended-release methylphenidate ODT. The XR-ODT formulation is made possible through ion exchange resin technology. When the methylphenidate salt is dissolved in the presence of the exchange resin during the manufacturing process, the positively charged mobile Na+ ion of the exchange resin is replaced by the positively charged methylphenidate molecule, resulting in stable methylphenidate microparticles. Methylphenidate microparticles are either coated (extended-release) or uncoated (immediate-release) and compressed into ODTs containing ~30% immediate-release methylphenidate and ~70% extended-release methylphenidate. This methylphenidate XR-ODT formulation remains under regulatory review; however, it has demonstrated a PK profile consistent with once-daily dosing and has shown efficacy in controlling symptoms in children with ADHD through 12 hours in a laboratory classroom setting.25,57,111,112
Amphetamine derivatives. Amphetamine was discovered more than 100 years ago and has been used effectively to treat ADHD for nearly 80 years.48 Some amphetamine formulations currently available or in development are comprised solely of the d– isomer (ie, dextroamphetamine), and others are comprised of the racemic mixture of both d– and l– isomers, along with other formulations containing methamphetamine (Desoxyn, Recordati Rare Diseases Inc, Lebanon, New Jersey123) or a mixture of various amphetamine salts (both d– and l– isomers) (Table 2). Short-acting amphetamine derivative products are available as oral tablets or as an oral solution, whereas long-acting products currently available or in late stages of clinical development include oral capsules, an oral suspension, a transdermal patch, and an ODT.
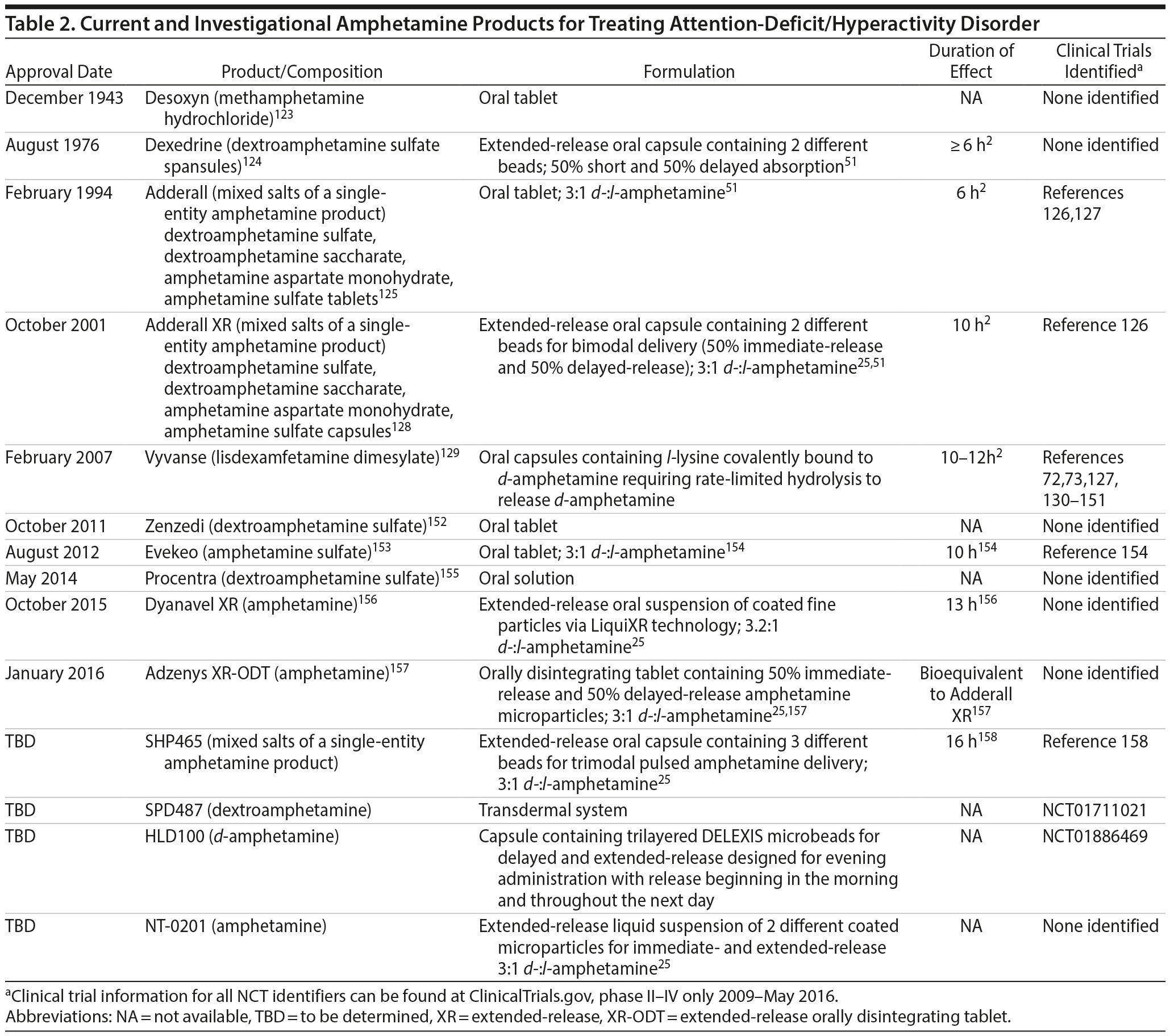
Dextroamphetamine is available in a longer-acting capsule formulation containing sustained-release beads (Dexedrine Spansule, Amedra Pharmaceuticals, Horsham, Pennsylvania124) with an 8-hour duration of action.24 Lisdexamfetamine (Vyvanse, Shire, Wayne, Pennsylvania129), a prodrug of d-amphetamine,159 is another long-acting agent formulated as an oral capsule with a duration of action of up to 13 hours in children and 14 hours in adults with ADHD. Lisdexamfetamine capsules can be opened and the contents dissolved in yogurt, water, or orange juice if a patient has difficulty swallowing the capsules.129 The extended-release oral suspension formulation (Dyanavel XR, Tris Pharma, Monmouth Junction, New Jersey156), approved in October 2015, contains a mixture of d– and l-amphetamine and has a duration of effect of up to 13 hours. The XR-ODT amphetamine formulation tablet (Adzenys XR-ODT [NT-0202], Neos Therapeutics, Grand Prairie, Texas157), which contains a mixture of d– and l-amphetamine and disintegrates in the mouth without water, contains ~50% immediate-release microparticles and ~50% extended-release microparticles, and received approval in January 2016.25 This extended-release ODT formulation of amphetamine has a PK profile consistent with once-daily dosing and is bioequivalent to Adderall XR (Shire, Wayne, PA128).25,157 A new long-acting liquid formulation (NT-0201) containing a mixture of d– and l-amphetamine is also in development and relies on similar technology as NT-0202.25 The transdermal patch (SPD487) long-acting amphetamine formulation is currently in late stages of clinical development. Additionally, a new capsule formulation similar to HLD200 is in development. HLD100 is also composed of trilayered DELEXIS microbeads and is intended for administration in the evening for next-day symptom coverage; however, this formulation contains delayed- and extended-release amphetamine (ClinicalTrials.gov: NCT01886469, Ironshore Pharmaceuticals and Development, Inc).25 The mixed amphetamine salt products are available in a short-acting tablet formulation (Adderall, Teva Pharmaceuticals, Horsham, Pennsylvania125) and a long-acting capsule formulation (Adderall XR). The long-acting capsule formulation contains an equal number of immediate-release and extended-release beads.24 The duration of action of the long-acting formulation is 10 to 12 hours.24 A triple-bead mixed amphetamine salts formulation SPD465/SHP465 (now referred to as SHP465), with a duration of action of up to 16 hours, is under consideration by the FDA for an indication in adults with ADHD.25,158
Nonstimulants
Nonstimulant treatments currently available for ADHD are available in oral capsule (atomoxetine) and tablet (guanfacine and clonidine) formulations, although additional agents are being investigated (Table 3). Along with stimulants, atomoxetine is considered appropriate first-line therapy for adults; however, there is insufficient evidence to recommend the use of clonidine or guanfacine for routine treatment in adults.20 For children, nonstimulants are generally second-line options, unless there are circumstances that warrant their consideration.2 Extended-release formulations of guanfacine and clonidine are also approved for use as monotherapy and as adjunctive therapy to stimulants in children and adolescents with ADHD.2,182,192
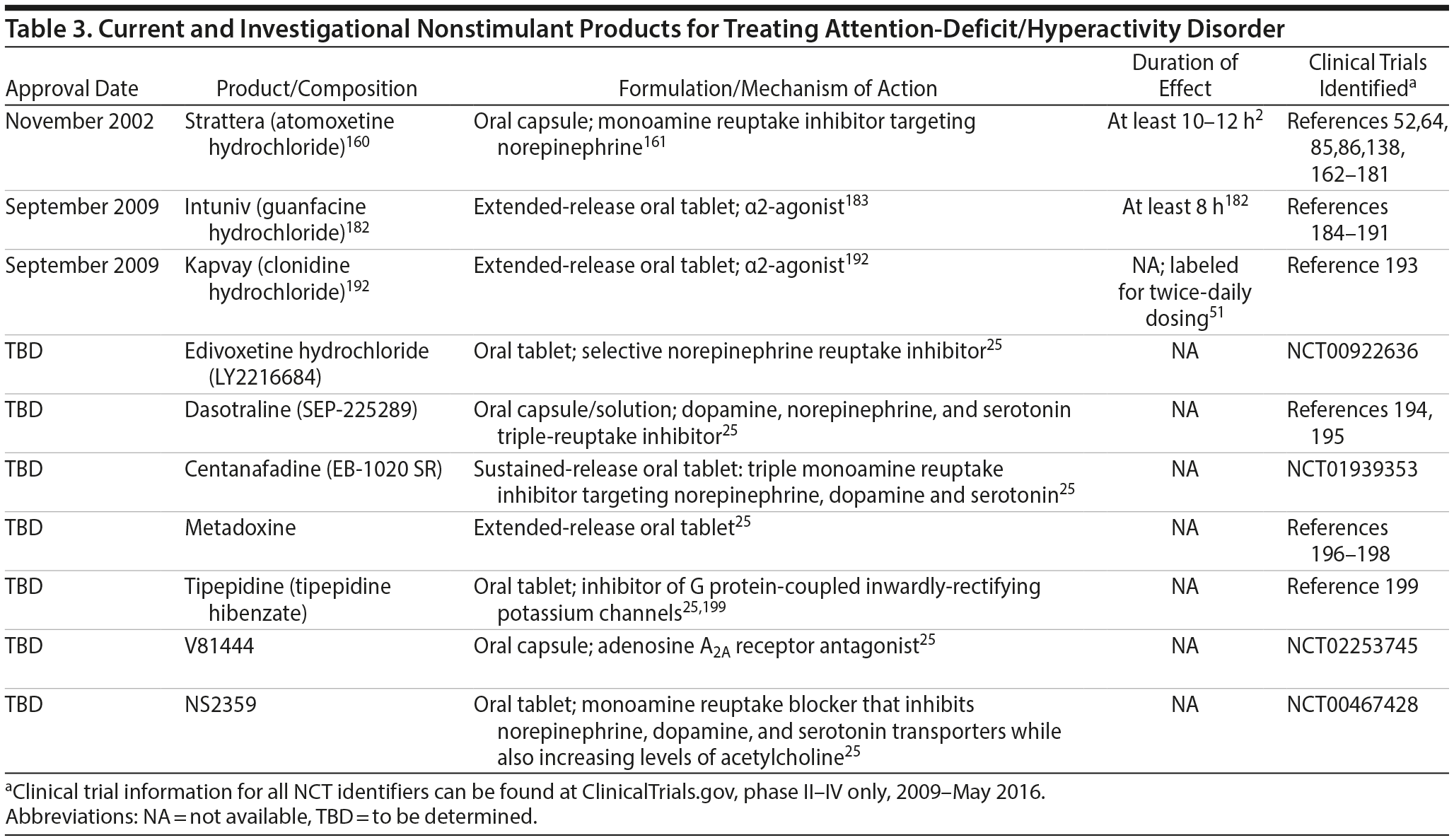
Atomoxetine inhibits presynaptic norepinephrine reuptake, thereby increasing synaptic concentrations of this neurotransmitter.38 Atomoxetine has minimal effects on other neurotransmitters.38 However, stimulation of norepinephrine increases synaptic dopamine concentrations in the prefrontal cortex, which may help explain the effects of atomoxetine in controlling ADHD symptoms.38 Atomoxetine’s onset of action is slower than that of stimulants; several weeks of treatment are typically needed to observe maximal treatment effect.29 Side effects in adults include dry mouth, insomnia, nausea, decreased appetite, constipation, decreased libido, dizziness, erectile dysfunction, sweating, and dysuria.200 In children, side effects include somnolence, gastrointestinal upset/nausea, and reduced appetite.19 Like stimulants, atomoxetine increases heart rate and blood pressure in the short term; however, it has not been associated with sudden cardiac death.201
Guanfacine and clonidine are α-2 adrenergic agonists that modulate presynaptic and postsynaptic noradrenergic activity, producing physiologic effects on the prefrontal cortex that affect the pathophysiology of ADHD.38 Guanfacine may be helpful if risk for abuse/diversion is high,29 whereas clonidine may be particularly useful in patients with comorbid Tourette’s syndrome or other tic disorders.202,203 The most common side effects of guanfacine include somnolence, fatigue, bradycardia, and hypotension. Although they tend to diminish over time,204 somnolence and sedation are the leading causes of discontinuing guanfacine treatment205; however, guanfacine is less sedating than clonidine.29 Common side effects of clonidine include dry mouth, sedation, somnolence, dizziness, and constipation.29
Extended-release guanfacine and clonidine formulations preclude some side effects associated with plasma-concentration fluctuations inherent to the immediate-release formulations.193 Extended-release (long-acting) guanfacine hydrochloride (Intuniv, Shire USA, Inc, Wayne, Pennsylvania182) has a ≥ 8-hour duration of effect based on the Conners’ Parent Rating Scale–Revised: Short Form and the Conners’ Teacher Rating Scale–Revised: Short Form.206,207 This matrix tablet formulation may help control symptoms of ADHD in patients 6 to 17 years of age but has not been approved for adults with ADHD.184–191,207 Extended-release clonidine hydrochloride (Kapvay, Shionogi Pharma, Inc, Atlanta, Georgia192) was designed to delay active drug absorption in order to decrease peak-to-trough plasma concentration differences, and can be dosed up to twice daily.193 A significant reduction in ADHD-Rating Scale-IV total score was observed in a study of children and adolescents with ADHD taking extended-release clonidine hydrochloride versus placebo. The effect size was 0.71 for 0.2 mg/d and 0.77 for 0.4 mg/d.193 Additionally, 0.1 to 0.4 mg/d extended-release clonidine as an adjuvant therapy to a stimulant has shown increased efficacy versus stimulant alone in children and adolescents who had an inadequate response to their baseline stimulant treatment.208 The potential advantages of guanfacine over clonidine include fewer hypotensive and sedative effects and a longer half-life. Both agents have been suggested to be useful for ADHD patients taking stimulants and with comorbid sleep problems, tics, or Tourette’s syndrome.19
SUMMARY DISCUSSION: SELECTING THERAPY
FOR INDIVIDUALS WITH ADHD
Management of ADHD should be individualized whenever possible.22 Although certain patients may respond preferentially to either methylphenidate or amphetamine derivatives,209 the overall response rate to stimulants is high. Approximately two-thirds of patients will respond to either methylphenidate or amphetamine derivatives; this rate increases to ~90% after trying agents from both stimulant classes.31,209 The high response rate to stimulants bodes well for patients, considering that onset of the therapeutic effect is essentially immediate.2 Doses can be titrated frequently (every 3–7 days),2 and usually treatment can be optimized within a few weeks. For the 10% to 30% of patients who do not respond to stimulant treatment or experience intolerable side effects, treatment with nonstimulant options may be warranted as monotherapy or adjunctive therapy in combination with stimulants.2,29,210 Factors to consider when selecting therapy include the patient’s age, comorbid symptoms, treatment history (and response), and the patient or parental/caregiver attitudes.31,211 In addition, on the basis of different patient lifestyles, different formulations may be preferential. For instance, individuals with ADHD, especially adults with ADHD, who require extended symptom control later in the day (eg, at the end of an 8-hour work day) may benefit from a long-acting formulation.2,24 Similarly, ensuring coverage for > 12 hours may be helpful for patients requiring symptom control for afternoon and evening activities.2,200
Poor adherence to ADHD treatments is common, which is a considerable unmet need in this therapeutic area, given that ADHD persists for life in many patients. Despite the broad range of treatment options currently available, nonadherence rates range from 13% to 64%.212 Ineffectiveness and unwanted side effects are the most common reasons for discontinuing treatment, and dosing convenience may be another significant factor in adherence.213 Other approaches to dosage administration (eg, opening certain formulations and sprinkling the contents on food or dissolving in water) may improve acceptance. Many novel formulations that circumvent problems associated with swallowing tablets or capsules are currently available (eg, transdermal patch, oral solution, oral suspension, and chewable tablet), while others are in late stages of clinical development or have recently received FDA approval (eg, long-acting chewable tablet and long-acting XR-ODTs). Interestingly, recent evidence suggests that adherence and persistence improve when using long-acting agents (vs short-acting) and stimulants (vs nonstimulants).213,214 However, a majority of extended-release stimulants are formulated as solid tablets or capsules, and difficulty swallowing tablets or capsules is known to affect adults as well as children.215 In one survey, fewer than 50% of parents reported that their children could easily swallow a large pill or a small or large capsule.216 Similarly, in a survey of adult patients, roughly 37% of adults reported difficulty swallowing pills or capsules of which the majority reported always or sometimes experiencing these difficulties.217 Studies also estimate that 3% of children and adolescents with ADHD discontinue their medication due to swallowing difficulties, and expert opinions report that this rate may be even higher (8%–9%).213 These results warrant future efforts to fully determine the role of formulations on treatment adherence in ADHD patients of all ages. The development of formulations that are acceptable to both pediatric and adult patients will be crucial to improving adherence.2,31
Another challenge with the development of new ADHD formulations (particularly controlled-release formulations) pertains to the criteria defining bioequivalence for generic products. According to the FDA, regulatory bioequivalence is established if the 90% confidence intervals for the ratios (test product to reference product) of various PK parameters fall between 80% and 125%.218 Therapeutic substitution is permitted for products that are both pharmaceutically equivalent and bioequivalent.219 However, experience with certain generic versions of Concerta shows that bioequivalence may not universally translate to therapeutic equivalence. These generic versions of Concerta do not use the OROS technology, and after reviewing data, the FDA changed their rating from AB (therapeutically equivalent) to BX (insufficient data to confirm bioequivalence).219
In conclusion, long-acting stimulants are a first-line treatment option for adults and children with ADHD, and a variety of distinct formulations are currently available. Many long-acting stimulant agents were initially formulated as oral tablets or capsules, whereas more recently approved agents (and those in late stages of clinical development) include long-acting liquid, long-acting chewable tablets, and long-acting ODTs. Despite the current landscape, which includes more than 20 options (stimulants and nonstimulants), adherence and patient outcomes in ADHD remain far from optimal. New formulations enabling further treatment individualization may improve adherence and overall treatment outcomes.
Submitted: May 3, 2016; accepted June 21, 2016.
Published online: August 18, 2016.
Drug names: amphetamine (Dyanavel and Adzenys), atomoxetine (Strattera and others), clonidine (Catapres and others), dexmethylphenidate (Focalin and others), dextroamphetamine (Dexedrine and others), guanfacine (Tenex and others), lisdexamfetamine (Vyvanse), methamphetamine (Desoxyn and others), methylphenidate (Ritalin and others), mixed amphetamine salts (Adderall).
Potential conflicts of interest: Dr Jain has received research support from Eli Lilly and Shire and has served on the speakers bureaus of Eli Lilly, Shire, Rhodes, Neos Therapeutics, Tris Pharma, and Pfizer. Dr Katic has received research support from Alcobra, Allergan, Alkermes, Eli Lilly, Forest, Lundbeck, Medgenics, Noven, Purdue, Shire, Sunovion, and Supernus and has served on the speakers bureaus of Neos Therapeutics and Shire.
Funding/support: Funding for this systematic review was provided through a research grant from Neos Therapeutics, Inc, Grand Prairie, Texas.
Role of the sponsors: The sponsors had no role in the design, analysis, interpretation, or publication of this study.
Acknowledgments: Medical writing and editorial support was provided by Kevin Jarvis, PhD, and Jennifer Tyson, PhD, of AlphaBioCom, LLC (King of Prussia, Pennsylvania) and was funded by Neos Therapeutics, Inc (Grand Prairie, Texas). Drs Jarvis and Tyson report no other conflicts of interest related to the subject of this article.
REFERENCES
1. American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders. Fifth Edition. Washington, DC: American Psychiatric Association; 2013.
2. Wolraich M, Brown L, Brown RT, et al; Steering Committee on Quality Improvement and Management. ADHD: clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics. 2011;128(5):1007–1022. doi:10.1542/peds.2011-2654 PubMed
3. Visser SN, Danielson ML, Bitsko RH, et al. Trends in the parent-report of health care provider–diagnosed and medicated attention-deficit/hyperactivity disorder: United States, 2003–2011. J Am Acad Child Adolesc Psychiatry. 2014;53(1):34–46, e2. doi:10.1016/j.jaac.2013.09.001 PubMed
4. Faraone SV, Biederman J, Mick E. The age-dependent decline of attention deficit hyperactivity disorder: a meta-analysis of follow-up studies. Psychol Med. 2006;36(2):159–165. doi:10.1017/S003329170500471X PubMed
5. Biederman J, Faraone SV. Attention-deficit hyperactivity disorder. Lancet. 2005;366(9481):237–248. doi:10.1016/S0140-6736(05)66915-2 PubMed
6. Kessler RC, Adler L, Barkley R, et al. The prevalence and correlates of adult ADHD in the United States: results from the National Comorbidity Survey Replication. Am J Psychiatry. 2006;163(4):716–723. doi:10.1176/ajp.2006.163.4.716 PubMed
7. Polanczyk GV, Willcutt EG, Salum GA, et al. ADHD prevalence estimates across three decades: an updated systematic review and meta-regression analysis. Int J Epidemiol. 2014;43(2):434–442. doi:10.1093/ije/dyt261 PubMed
8. Matte B, Anselmi L, Salum GA, et al. ADHD in DSM-5: a field trial in a large, representative sample of 18- to 19-year-old adults. Psychol Med. 2015;45(2):361–373. doi:10.1017/S0033291714001470 PubMed
9. Dunlop AJ, Newman LK. ADHD and psychostimulants—overdiagnosis and overprescription. Med J Aust. 2016;204(4):139. doi:10.5694/mja15.01387 PubMed
10. Ginsberg Y, Quintero J, Anand E, et al. Underdiagnosis of attention-deficit/hyperactivity disorder in adult patients: a review of the literature. Prim Care Companion CNS Disord. 2014;16(3): doi:10.4088/PCC.13r01600. PubMed
11. Froehlich TE, Lanphear BP, Epstein JN, et al. Prevalence, recognition, and treatment of attention-deficit/hyperactivity disorder in a national sample of US children. Arch Pediatr Adolesc Med. 2007;161(9):857–864. doi:10.1001/archpedi.161.9.857 PubMed
12. Cuffe SP, Moore CG, McKeown RE. Prevalence and correlates of ADHD symptoms in the national health interview survey. J Atten Disord. 2005;9(2):392–401. doi:10.1177/1087054705280413 PubMed
13. Barkley RA, Fischer M, Smallish L, et al. Young adult outcome of hyperactive children: adaptive functioning in major life activities. J Am Acad Child Adolesc Psychiatry. 2006;45(2):192–202. doi:10.1097/01.chi.0000189134.97436.e2 PubMed
14. Klassen AF, Miller A, Fine S. Health-related quality of life in children and adolescents who have a diagnosis of attention-deficit/hyperactivity disorder. Pediatrics. 2004;114(5):e541–e547. doi:10.1542/peds.2004-0844 PubMed
15. Sawyer MG, Whaites L, Rey JM, et al. Health-related quality of life of children and adolescents with mental disorders. J Am Acad Child Adolesc Psychiatry. 2002;41(5):530–537. doi:10.1097/00004583-200205000-00010 PubMed
16. Able SL, Johnston JA, Adler LA, et al. Functional and psychosocial impairment in adults with undiagnosed ADHD. Psychol Med. 2007;37(1):97–107. doi:10.1017/S0033291706008713 PubMed
17. Biederman J, Faraone SV, Spencer TJ, et al. Functional impairments in adults with self-reports of diagnosed ADHD: a controlled study of 1001 adults in the community. J Clin Psychiatry. 2006;67(4):524–540. doi:10.4088/JCP.v67n0403 PubMed
18. Doshi JA, Hodgkins P, Kahle J, et al. Economic impact of childhood and adult attention-deficit/hyperactivity disorder in the United States. J Am Acad Child Adolesc Psychiatry. 2012;51(10):990–1002, e2. doi:10.1016/j.jaac.2012.07.008 PubMed
19. Felt BT, Biermann B, Christner JG, et al. Diagnosis and management of ADHD in children. Am Fam Physician. 2014;90(7):456–464. PubMed
20. Post RE, Kurlansik SL. Diagnosis and management of adult attention-deficit/hyperactivity disorder. Am Fam Physician. 2012;85(9):890–896. PubMed
21. McGough JJ. Attention-deficit/hyperactivity disorder pharmacogenomics. Biol Psychiatry. 2005;57(11):1367–1373. doi:10.1016/j.biopsych.2004.10.021 PubMed
22. Hodgkins P, Dittmann RW, Sorooshian S, et al. Individual treatment response in attention-deficit/hyperactivity disorder: broadening perspectives and improving assessments. Expert Rev Neurother. 2013;13(4):425–433. doi:10.1586/ern.13.31 PubMed
23. Stein MA, McGough JJ. The pharmacogenomic era: promise for personalizing attention deficit hyperactivity disorder therapy. Child Adolesc Psychiatr Clin N Am. 2008;17(2):475–490, xi–xii. doi:10.1016/j.chc.2007.11.009 PubMed
24. Chavez B, Sopko MA Jr, Ehret MJ, et al. An update on central nervous system stimulant formulations in children and adolescents with attention-deficit/hyperactivity disorder. Ann Pharmacother. 2009;43(6):1084–1095. doi:10.1345/aph.1L523 PubMed
25. Childress A, Tran C. Current investigational drugs for the treatment of attention-deficit/hyperactivity disorder. Expert Opin Investig Drugs. 2016;25(4):463–474. doi:10.1517/13543784.2016.1147558 PubMed
26. Prince JB. Pharmacotherapy of attention-deficit hyperactivity disorder in children and adolescents: update on new stimulant preparations, atomoxetine, and novel treatments. Child Adolesc Psychiatr Clin N Am. 2006;15(1):13–50. doi:10.1016/j.chc.2005.08.002 PubMed
27. Stevens JR, Wilens TE, Stern TA. Using stimulants for attention-deficit/hyperactivity disorder: clinical approaches and challenges. Prim Care Companion CNS Disord. 2013;15(2): doi:10.4088/PCC.12f01472. PubMed
28. Wilens TE, Spencer TJ. Understanding attention-deficit/hyperactivity disorder from childhood to adulthood. Postgrad Med. 2010;122(5):97–109. doi:10.3810/pgm.2010.09.2206 PubMed
29. Kolar D, Keller A, Golfinopoulos M, et al. Treatment of adults with attention-deficit/hyperactivity disorder. Neuropsychiatr Dis Treat. 2008;4(2):389–403. PubMed
30. Faraone SV. Using meta-analysis to compare the efficacy of medications for attention-deficit/hyperactivity disorder in youths. P T. 2009;34(12):678–694. PubMed
31. Hodgkins P, Shaw M, Coghill D, et al. Amfetamine and methylphenidate medications for attention-deficit/hyperactivity disorder: complementary treatment options. Eur Child Adolesc Psychiatry. 2012;21(9):477–492. doi:10.1007/s00787-012-0286-5 PubMed
32. Spencer T, Biederman J, Wilens T, et al. Pharmacotherapy of attention-deficit hyperactivity disorder across the life cycle. J Am Acad Child Adolesc Psychiatry. 1996;35(4):409–432. doi:10.1097/00004583-199604000-00008 PubMed
33. Lakhan SE, Kirchgessner A. Prescription stimulants in individuals with and without attention deficit hyperactivity disorder: misuse, cognitive impact, and adverse effects. Brain Behav. 2012;2(5):661–677. doi:10.1002/brb3.78 PubMed
34. Harstad EB, Weaver AL, Katusic SK, et al. ADHD, stimulant treatment, and growth: a longitudinal study. Pediatrics. 2014;134(4):e935–e944. doi:10.1542/peds.2014-0428 PubMed
35. Martinez-Raga J, Knecht C, Szerman N, et al. Risk of serious cardiovascular problems with medications for attention-deficit hyperactivity disorder. CNS Drugs. 2013;27(1):15–30. doi:10.1007/s40263-012-0019-9 PubMed
36. Ken Mehta S, Jacobs I. Stimulants for kids with ADHD—how to proceed safely. J Fam Pract. 2014;63(2):E1–E5. PubMed
37. Stahl SM. Mechanism of action of stimulants in attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2010;71(1):12–13. doi:10.4088/JCP.09bs05890pur PubMed
38. Wilens TE. Mechanism of action of agents used in attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2006;67(suppl 8):32–38. PubMed
39. López FA, Leroux JR. Long-acting stimulants for treatment of attention-deficit/hyperactivity disorder: a focus on extended-release formulations and the prodrug lisdexamfetamine dimesylate to address continuing clinical challenges. Atten Defic Hyperact Disord. 2013;5(3):249–265. doi:10.1007/s12402-013-0106-x PubMed
40. Faraone SV, Biederman J, Spencer TJ, et al. Comparing the efficacy of medications for ADHD using meta-analysis. MedGenMed. 2006;8(4):4. PubMed
41. Faraone SV, Buitelaar J. Comparing the efficacy of stimulants for ADHD in children and adolescents using meta-analysis. Eur Child Adolesc Psychiatry. 2010;19(4):353–364. doi:10.1007/s00787-009-0054-3 PubMed
42. Childress A, Sallee FR. The use of methylphenidate hydrochloride extended-release oral suspension for the treatment of ADHD. Expert Rev Neurother. 2013;13(9):979–988. doi:10.1586/14737175.2013.833002 PubMed
43. Biederman J, Mick E, Surman C, et al. A randomized, placebo-controlled trial of OROS methylphenidate in adults with attention-deficit/hyperactivity disorder. Biol Psychiatry. 2006;59(9):829–835. doi:10.1016/j.biopsych.2005.09.011 PubMed
44. Kollins SH, MacDonald EK, Rush CR. Assessing the abuse potential of methylphenidate in nonhuman and human subjects: a review. Pharmacol Biochem Behav. 2001;68(3):611–627. doi:10.1016/S0091-3057(01)00464-6 PubMed
45. Volkow ND, Swanson JM. Variables that affect the clinical use and abuse of methylphenidate in the treatment of ADHD. Am J Psychiatry. 2003;160(11):1909–1918. doi:10.1176/appi.ajp.160.11.1909 PubMed
46. Ermer JC, Dennis K, Haffey MB, et al. Intranasal versus oral administration of lisdexamfetamine dimesylate: a randomized, open-label, two-period, crossover, single-dose, single-centre pharmacokinetic study in healthy adult men. Clin Drug Investig. 2011;31(6):357–370. doi:10.2165/11588190-000000000-00000 PubMed
47. Ermer JC, Pennick M, Frick G. Lisdexamfetamine dimesylate: prodrug delivery, amphetamine exposure and duration of efficacy. Clin Drug Investig. 2016;36(5):341–356. doi:10.1007/s40261-015-0354-y PubMed
48. Heal DJ, Smith SL, Gosden J, et al. Amphetamine, past and present—a pharmacological and clinical perspective. J Psychopharmacol. 2013;27(6):479–496. doi:10.1177/0269881113482532 PubMed
49. Lange KW, Reichl S, Lange KM, et al. The history of attention deficit hyperactivity disorder. Atten Defic Hyperact Disord. 2010;2(4):241–255. doi:10.1007/s12402-010-0045-8 PubMed
50. Ritalin and Ritalin SR [package insert]. Hydrochloride methylphenidate hydrochloride tablets and sustained release tablets. East Hanover, NJ: Novartis Pharmaceuticals; 2013.
51. Stern AT, Fava M, Wilens TE, et al. Massachusetts General Hospital Psychopharmacology and Neurotherapeutics. 1st Har/Psc edition. London, UK: Elsevier; 2015.
52. Garg J, Arun P, Chavan BS. Comparative short term efficacy and tolerability of methylphenidate and atomoxetine in attention deficit hyperactivity disorder. Indian Pediatr. 2014;51(7):550–554. doi:10.1007/s13312-014-0445-5 PubMed
53. Ni HC, Shang CY, Gau SS, et al. A head-to-head randomized clinical trial of methylphenidate and atomoxetine treatment for executive function in adults with attention-deficit hyperactivity disorder. Int J Neuropsychopharmacol. 2013;16(9):1959–1973. doi:10.1017/S1461145713000357 PubMed
54. Ramtvedt BE, Aabech HS, Sundet K. Minimizing adverse events while maintaining clinical improvement in a pediatric attention-deficit/hyperactivity disorder crossover trial with dextroamphetamine and methylphenidate. J Child Adolesc Psychopharmacol. 2014;24(3):130–139. doi:10.1089/cap.2013.0114 PubMed
55. Solanto M, Newcorn J, Vail L, et al. Stimulant drug response in the predominantly inattentive and combined subtypes of attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2009;19(6):663–671. doi:10.1089/cap.2009.0033 PubMed
56. Wender PH, Reimherr FW, Marchant BK, et al. A one year trial of methylphenidate in the treatment of ADHD. J Atten Disord. 2011;15(1):36–45. doi:10.1177/1087054709356188 PubMed
57. Childress AC. Methylphenidate HCL for the treatment of ADHD in children and adolescents. Expert Opin Pharmacother. 2016;17(8):1171–1178. doi:10.1080/14656566.2016.1182986 PubMed
58. Methylin ER [package insert]. Methylphenidate hydrochloride extended-release tablets. Hazelwood, MO: Mallinckrodt; 2015.
59. Concerta [package insert]. Methylphenidate HCl extended-release tablets. Titusville, NJ: Janssen Pharmaceuticals; 2015.
60. Adler LA, Orman C, Starr HL, et al. Long-term safety of OROS methylphenidate in adults with attention-deficit/hyperactivity disorder: an open-label, dose-titration, 1-year study. J Clin Psychopharmacol. 2011;31(1):108–114. doi:10.1097/JCP.0b013e318203ea0a PubMed
61. Adler LA, Zimmerman B, Starr HL, et al. Efficacy and safety of OROS methylphenidate in adults with attention-deficit/hyperactivity disorder: a randomized, placebo-controlled, double-blind, parallel group, dose-escalation study. J Clin Psychopharmacol. 2009;29(3):239–247. doi:10.1097/JCP.0b013e3181a390ce PubMed
62. Armstrong RB, Damaraju CV, Ascher S, et al. Time course of treatment effect of OROS methylphenidate in children with ADHD. J Atten Disord. 2012;16(8):697–705. doi:10.1177/1087054711425772 PubMed
63. Banaschewski T, Soutullo C, Lecendreux M, et al. Health-related quality of life and functional outcomes from a randomized, controlled study of lisdexamfetamine dimesylate in children and adolescents with attention deficit hyperactivity disorder. CNS Drugs. 2013;27(10):829–840. doi:10.1007/s40263-013-0095-5 PubMed
64. Bédard AC, Stein MA, Halperin JM, et al. Differential impact of methylphenidate and atomoxetine on sustained attention in youth with attention-deficit/hyperactivity disorder. J Child Psychol Psychiatry. 2015;56(1):40–48. doi:10.1111/jcpp.12272 PubMed
65. Biederman J, Mick E, Fried R, et al. Are stimulants effective in the treatment of executive function deficits? results from a randomized double blind study of OROS-methylphenidate in adults with ADHD. Eur Neuropsychopharmacol. 2011;21(7):508–515. doi:10.1016/j.euroneuro.2010.11.005 PubMed
66. Biederman J, Mick E, Surman C, et al. A randomized, 3-phase, 34-week, double-blind, long-term efficacy study of osmotic-release oral system-methylphenidate in adults with attention-deficit/hyperactivity disorder. J Clin Psychopharmacol. 2010;30(5):549–553. doi:10.1097/JCP.0b013e3181ee84a7 PubMed
67. Blum NJ, Jawad AF, Clarke AT, et al. Effect of osmotic-release oral system methylphenidate on different domains of attention and executive functioning in children with attention-deficit-hyperactivity disorder. Dev Med Child Neurol. 2011;53(9):843–849. doi:10.1111/j.1469-8749.2011.03944.x PubMed
68. Bron TI, Bijlenga D, Boonstra AM, et al. OROS-methylphenidate efficacy on specific executive functioning deficits in adults with ADHD: a randomized, placebo-controlled cross-over study. Eur Neuropsychopharmacol. 2014;24(4):519–528. doi:10.1016/j.euroneuro.2014.01.007 PubMed
69. Buitelaar JK, Casas M, Philipsen A, et al. Functional improvement and correlations with symptomatic improvement in adults with attention deficit hyperactivity disorder receiving long-acting methylphenidate. Psychol Med. 2012;42(1):195–204. doi:10.1017/S0033291711000845 PubMed
70. Casas M, Rösler M, Sandra Kooij JJ, et al. Efficacy and safety of prolonged-release OROS methylphenidate in adults with attention deficit/hyperactivity disorder: a 13-week, randomized, double-blind, placebo-controlled, fixed-dose study. World J Biol Psychiatry. 2013;14(4):268–281. doi:10.3109/15622975.2011.600333 PubMed
71. Chou WJ, Chen SJ, Chen YS, et al. Remission in children and adolescents diagnosed with attention-deficit/hyperactivity disorder via an effective and tolerable titration scheme for osmotic release oral system methylphenidate. J Child Adolesc Psychopharmacol. 2012;22(3):215–225. doi:10.1089/cap.2011.0006 PubMed
72. Coghill D, Banaschewski T, Lecendreux M, et al. European, randomized, phase 3 study of lisdexamfetamine dimesylate in children and adolescents with attention-deficit/hyperactivity disorder. Eur Neuropsychopharmacol. 2013;23(10):1208–1218. doi:10.1016/j.euroneuro.2012.11.012 PubMed
73. Coghill DR, Banaschewski T, Lecendreux M, et al. Efficacy of lisdexamfetamine dimesylate throughout the day in children and adolescents with attention-deficit/hyperactivity disorder: results from a randomized, controlled trial. Eur Child Adolesc Psychiatry. 2014;23(2):61–68. doi:10.1007/s00787-013-0421-y PubMed
74. Döpfner M, Ose C, Fischer R, et al. Comparison of the efficacy of two different modified release methylphenidate preparations for children and adolescents with attention-deficit/hyperactivity disorder in a natural setting: comparison of the efficacy of Medikinet retard and Concerta—a randomized, controlled, double-blind multicenter clinical crossover trial. J Child Adolesc Psychopharmacol. 2011;21(5):445–454. doi:10.1089/cap.2010.0082 PubMed
75. Gerwe M, Stollhoff K, Mossakowski J, et al. Tolerability and effects of OROS MPH (Concerta) on functioning, severity of disease and quality of life in children and adolescents with ADHD: results from a prospective, non-interventional trial. Atten Defic Hyperact Disord. 2009;1(2):175–186. doi:10.1007/s12402-009-0010-6 PubMed
76. Kordon A, Stollhoff K, Niederkirchner K, et al. Exploring the impact of once-daily OROS methylphenidate (MPH) on symptoms and quality of life in children and adolescents with ADHD transitioning from immediate-release MPH. Postgrad Med. 2011;123(5):27–38. doi:10.3810/pgm.2011.09.2457 PubMed
77. Marchant BK, Reimherr FW, Halls C, et al. OROS methylphenidate in the treatment of adults with ADHD: a 6-month, open-label, follow-up study. Ann Clin Psychiatry. 2010;22(3):196–204. PubMed
78. Murray DW, Childress A, Giblin J, et al. Effects of OROS methylphenidate on academic, behavioral, and cognitive tasks in children 9 to 12 years of age with attention-deficit/hyperactivity disorder. Clin Pediatr (Phila). 2011;50(4):308–320. doi:10.1177/0009922810394832 PubMed
79. Newcorn JH, Stein MA, Cooper KM. Dose-response characteristics in adolescents with attention-deficit/hyperactivity disorder treated with OROS methylphenidate in a 4-week, open-label, dose-titration study. J Child Adolesc Psychopharmacol. 2010;20(3):187–196. doi:10.1089/cap.2009.0102 PubMed
80. Rösler M, Ginsberg Y, Arngrim T, et al. Correlation of symptomatic improvements with functional improvements and patient-reported outcomes in adults with attention-deficit/hyperactivity disorder treated with OROS methylphenidate. World J Biol Psychiatry. 2013;14(4):282–290. doi:10.3109/15622975.2011.571283 PubMed
81. Spencer TJ, Mick E, Surman CB, et al. A randomized, single-blind, substitution study of OROS methylphenidate (Concerta) in ADHD adults receiving immediate release methylphenidate. J Atten Disord. 2011;15(4):286–294. doi:10.1177/1087054710367880 PubMed
82. Takahashi N, Koh T, Tominaga Y, et al. A randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of osmotic-controlled release oral delivery system methylphenidate HCl in adults with attention-deficit/hyperactivity disorder in Japan. World J Biol Psychiatry. 2014;15(6):488–498. doi:10.3109/15622975.2013.868925 PubMed
83. Wigal SB, Wigal T, Schuck S, et al. Academic, behavioral, and cognitive effects of OROS methylphenidate on older children with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2011;21(2):121–131. doi:10.1089/cap.2010.0047 PubMed
84. Wolff C, Alfred A, Lindermüller A, et al. Effect of transitioning from extended-release methylphenidate onto osmotic, controlled-release methylphenidate in children/adolescents with ADHD: results of a 3-month non-interventional study. Curr Med Res Opin. 2011;27(suppl 2):35–44. doi:10.1185/03007995.2011.601733 PubMed
85. Yang L, Cao Q, Shuai L, et al. Comparative study of OROS-MPH and atomoxetine on executive function improvement in ADHD: a randomized controlled trial. Int J Neuropsychopharmacol. 2012;15(1):15–26. doi:10.1017/S1461145711001490 PubMed
86. Yildiz O, Sismanlar SG, Memik NC, et al. Atomoxetine and methylphenidate treatment in children with ADHD: the efficacy, tolerability and effects on executive functions. Child Psychiatry Hum Dev. 2011;42(3):257–269. doi:10.1007/s10578-010-0212-3 PubMed
87. Zheng Y, Wang YF, Qin J, et al. Prospective, naturalistic study of open-label OROS methylphenidate treatment in Chinese school-aged children with attention-deficit/hyperactivity disorder. Chin Med J (Engl). 2011;124(20):3269–3274. PubMed
88. Metadate CD [package insert]. Methylphenidate HCl extended-release capsules. Smyrna, GA: UCB, Inc; 2014.
89. Focalin [package insert]. Dexmethylphenidate hydrochloride tablets. East Hanover, NJ: Novartis Pharmaceuticals; 2015.
90. Ritalin LA [package insert]. Methylphenidate hydrochloride extended-release capsules. East Hanover, NJ: Novartis Pharmaceuticals; 2013.
91. Ginsberg Y, Arngrim T, Philipsen A, et al. Long-term (1 year) safety and efficacy of methylphenidate modified-release long-acting formulation (MPH-LA) in adults with attention-deficit hyperactivity disorder: a 26-week, flexible-dose, open-label extension to a 40-week, double-blind, randomised, placebo-controlled core study. CNS Drugs. 2014;28(10):951–962. doi:10.1007/s40263-014-0180-4 PubMed
92. Schulz E, Fleischhaker C, Hennighausen K, et al. A randomized, rater-blinded, crossover study comparing the clinical efficacy of Ritalin LA (methylphenidate) treatment in children with attention-deficit hyperactivity disorder under different breakfast conditions over 2 weeks. Atten Defic Hyperact Disord. 2010;2(3):133–138. doi:10.1007/s12402-010-0031-1 PubMed
93. Methylin oral solution [package insert]. Methylphenidate hydrochloride oral solution. Hazelwood, MO: Mallinckrodt; 2013.
94. Methylin chewable tablets [package insert]. Methylphenidate hydrochloride chewable tablets. Hazelwood, MO: Mallinckrodt; 2013.
95. Focalin XR [package insert]. Dexmethylphenidate hydrochloride extended-release capsules. East Hanover, NJ: Novartis Pharmaceuticals; 2015.
96. Adler LA, Spencer T, McGough JJ, et al. Long-term effectiveness and safety of dexmethylphenidate extended-release capsules in adult ADHD. J Atten Disord. 2009;12(5):449–459. doi:10.1177/1087054708320397 PubMed
97. Brams M, Turnbow J, Pestreich L, et al. A randomized, double-blind study of 30 versus 20 mg dexmethylphenidate extended-release in children with attention-deficit/hyperactivity
disorder: late-day symptom control. J Clin Psychopharmacol. 2012;32(5):637–644. doi:10.1097/JCP.0b013e3182677825 PubMed
98. Childress AC, Spencer T, Lopez F, et al. Efficacy and safety of dexmethylphenidate extended-release capsules administered once daily to children with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2009;19(4):351–361. doi:10.1089/cap.2009.0007 PubMed
99. Silva RR, Brams M, McCague K, et al. Extended-release dexmethylphenidate 30 mg/d versus 20 mg/d: duration of attention, behavior, and performance benefits in children with attention-deficit/hyperactivity disorder. Clin Neuropharmacol. 2013;36(4):117–121. doi:10.1097/WNF.0b013e31829aa92c PubMed
100. Stein MA, Waldman ID, Charney E, et al. Dose effects and comparative effectiveness of extended release dexmethylphenidate and mixed amphetamine salts. J Child Adolesc Psychopharmacol. 2011;21(6):581–588. doi:10.1089/cap.2011.0018 PubMed
101. Daytrana [package insert]. Methylphenidate transdermal system. Miami, FL: Noven Therapeutics; 2013.
102. Findling RL, Katic A, Rubin R, et al. A 6-month, open-label, extension study of the tolerability and effectiveness of the methylphenidate transdermal system in adolescents diagnosed with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2010;20(5):365–375. doi:10.1089/cap.2009.0122 PubMed
103. Wilens TE, Hammerness P, Martelon M, et al. A controlled trial of the methylphenidate transdermal system on before-school functioning in children with attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2010;71(5):548–556. doi:10.4088/JCP.09m05779pur PubMed
104. Quillivant XR [package insert]. QUILLIVANT XR- methylphenidate hydrochloride powder, for suspension. Cupertino, CA: NextWave Pharmaceuticals, Inc; 2012.
105. Wigal SB, Childress AC, Belden HW, et al. NWP06, an extended-release oral suspension of methylphenidate, improved attention-deficit/hyperactivity disorder symptoms compared with placebo in a laboratory classroom study. J Child Adolesc Psychopharmacol. 2013;23(1):3–10. doi:10.1089/cap.2012.0073 PubMed
106. Robb AS, Findling RL, Childress AC, et al. Efficacy, safety, and tolerability of a novel methylphenidate extended-release oral suspension (MEROS) in ADHD [published online ahead of print May 29, 2014]. J Atten Disord. doi:10.1177/1087054714533191 PubMed
107. Aptensio XR [package insert]. Methylphenidate hydrochloride extended-release capsules. Coventry, RI: Rhodes Pharmaceuticals LP; 2015.
108. Wigal SB, Greenhill LL, Nordbrock E, et al. A randomized placebo-controlled double-blind study evaluating the time course of response to methylphenidate hydrochloride extended-release capsules in children with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2014;24(10):562–569. doi:10.1089/cap.2014.0100 PubMed
109. Wigal SB, Nordbrock E, Adjei AL, et al. Efficacy of methylphenidate hydrochloride extended-release capsules (Aptensio XR) in children and adolescents with attention-deficit/hyperactivity disorder: a phase iii, randomized, double-blind study. CNS Drugs. 2015;29(4):331–340. doi:10.1007/s40263-015-0241-3 PubMed
110. Quillichew ER [package insert]. Methylphenidate hydrochloride extended-release chewable tablets. Monmouth Junction, NJ: Tris Pharma, Inc; 2015.
111. Childress A, Newcorn J, Stark JG, et al. A single-dose, single-period pharmacokinetic assessment of an extended-release orally disintegrating tablet of methylphenidate in children and adolescents with attention-deficit/hyperactivity disorder [published online ahead of print May 26, 2016]. J Child Adolesc Psychopharmacol. PubMed
112. Childress AC, Kollins SH, Cutler AJ, et al. Efficacy, safety, and tolerability of an extended-release orally disintegrating methylphenidate tablet in children 6-12 years of age with attention-deficit/hyperactivity disorder in the laboratory classroom setting [published online ahead of print May 16, 2016]. J Child Adolesc Psychopharmacol. PubMed
113. Coghill D, Seth S. Osmotic, controlled-release methylphenidate for the treatment of ADHD. Expert Opin Pharmacother. 2006;7(15):
2119–2138. doi:10.1517/14656566.7.15.2119 PubMed
114. Ritalin/Ritalin SR/Ritalin LA (methylphenidate hydrochloride) Data Sheet. 2016. Novartis New Zealand Limited. http://www.medsafe.govt.nz/profs/datasheet/r/RitalintabSRtabLAtab.pdf. Accessed June 28, 2016.
115. Swanson JM, Volkow ND. Psychopharmacology: concepts and opinions about the use of stimulant medications. J Child Psychol Psychiatry. 2009;50(1-2):
180–193. doi:10.1111/j.1469-7610.2008.02062.x PubMed
116. Canadian Attention Deficit Hyperactivity Disorder Resource Alliance (CADDRA) Guidelines. Chapter 7: Pharmacological Treatment of ADHD. CADDRA Web site. http://www.caddra.ca. Accessed May 2016.
117. Modi NB, Lindemulder B, Gupta SK. Single- and multiple-dose pharmacokinetics of an oral once-a-day osmotic controlled-release OROS (methylphenidate HCl) formulation. J Clin Pharmacol. 2000;40(4):379–388. doi:10.1177/00912700022009080 PubMed
118. Swanson JM, Wigal SB, Wigal T, et al; COMACS Study Group. A comparison of once-daily extended-release methylphenidate formulations in children with attention-deficit/hyperactivity disorder in the laboratory school (the Comacs Study). Pediatrics. 2004;113(3 pt 1):e206–e216. doi:10.1542/peds.113.3.e206 PubMed
119. Beck MH, Cataldo M, Slifer KJ, et al. Teaching children with attention deficit hyperactivity disorder (ADHD) and autistic disorder (AD) how to swallow pills. Clin Pediatr (Phila). 2005;44(6):515–526. doi:10.1177/000992280504400608 PubMed
120. Patrick KS, Straughn AB, Perkins JS, et al. Evolution of stimulants to treat ADHD: transdermal methylphenidate. Hum Psychopharmacol. 2009;24(1):1–17. doi:10.1002/hup.992 PubMed
121. Ermer JC, Adeyi BA, Pucci ML. Pharmacokinetic variability of long-acting stimulants in the treatment of children and adults with attention-deficit hyperactivity disorder. CNS Drugs. 2010;24(12):1009–1025. doi:10.2165/11539410-000000000-00000 PubMed
122. Wilens TE, Boellner SW, López FA, et al. Varying the wear time of the methylphenidate transdermal system in children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2008;47(6):700–708. doi:10.1097/CHI.0b013e31816bffdf PubMed
123. Desoxyn XR [package insert]. Methamphetamine hydrochloride tablet. Lebanon, NJ: Recordati Rare Diseases Inc; 2015.
124. Dexedrine Spansule [package insert]. Dextroamphetamine sulfate sustained release capsules. Horsham, PA: Amedra Pharmaceuticals; 2015.
125. Adderall [package insert]. Dextroamphetamine saccharate, amphetamine aspartate, dextroamphetamine sulfate and amphetamine sulfate tablets. Horsham, PA: Teva Pharmaceuticals; 2015.
126. Adler LA, Lynch LR, Shaw DM, et al. Medication adherence and symptom reduction in adults treated with mixed amphetamine salts in a randomized crossover study. Postgrad Med. 2011;123(5):71–79. doi:10.3810/pgm.2011.09.2461 PubMed
127. Martin PT, Corcoran M, Zhang P, et al. Randomized, double-blind, placebo-controlled, crossover study of the effects of lisdexamfetamine dimesylate and mixed amphetamine salts on cognition throughout the day in adults with attention-deficit/hyperactivity disorder. Clin Drug Investig. 2014;34(2):147–157. doi:10.1007/s40261-013-0156-z PubMed
128. Adderall XR [package insert]. Dextroamphetamine saccharate, amphetamine aspartate, dextroamphetamine sulfate and amphetamine sulfate capsules. Wayne, PA: Shire; 2015.
129. Vyvanse [package insert]. Lisdexamfetamine dimesylate. Wayne, PA: Shire; 2015.
130. Adler LA, Dirks B, Deas P, et al. Self-Reported quality of life in adults with attention-deficit/hyperactivity disorder and executive function impairment treated with lisdexamfetamine dimesylate: a randomized, double-blind, multicenter, placebo-controlled, parallel-group study. BMC Psychiatry. 2013;13(1):253. doi:10.1186/1471-244X-13-253 PubMed
131. Adler LA, Dirks B, Deas PF, et al. Lisdexamfetamine dimesylate in adults with attention-deficit/ hyperactivity disorder who report clinically significant impairment in executive function: results from a randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2013;74(7):694–702. doi:10.4088/JCP.12m08144 PubMed
132. Babcock T, Dirks B, Adeyi B, et al. Efficacy of lisdexamfetamine dimesylate in adults with attention-deficit/hyperactivity disorder previously treated with amphetamines: analyses from a randomized, double-blind, multicenter, placebo-controlled titration study. BMC Pharmacol Toxicol. 2012;13(1):18. doi:10.1186/2050-6511-13-18 PubMed
133. Banaschewski T, Johnson M, Lecendreux M, et al. Health-related quality of life and functional outcomes from a randomized-withdrawal study of long-term lisdexamfetamine dimesylate treatment in children and adolescents with attention-deficit/hyperactivity disorder. CNS Drugs. 2014;28(12):1191–1203. doi:10.1007/s40263-014-0193-z PubMed
134. Brams M, Weisler R, Findling RL, et al. Maintenance of efficacy of lisdexamfetamine dimesylate in adults with attention-deficit/hyperactivity disorder: randomized withdrawal design. J Clin Psychiatry. 2012;73(7):977–983. doi:10.4088/JCP.11m07430 PubMed
135. Brown TE, Brams M, Gao J, et al. Open-label administration of lisdexamfetamine dimesylate improves executive function impairments and symptoms of attention-deficit/hyperactivity disorder in adults. Postgrad Med. 2010;122(5):7–17. doi:10.3810/pgm.2010.09.2196 PubMed
136. Coghill DR, Banaschewski T, Lecendreux M, et al. Maintenance of efficacy of lisdexamfetamine dimesylate in children and adolescents with attention-deficit/hyperactivity disorder: randomized-withdrawal study design. J Am Acad Child Adolesc Psychiatry. 2014;53(6):647–657, e1. doi:10.1016/j.jaac.2014.01.017 PubMed
137. Dittmann RW, Cardo E, Nagy P, et al. Treatment response and remission in a double-blind, randomized, head-to-head study of lisdexamfetamine dimesylate and atomoxetine in children and adolescents with attention-deficit hyperactivity disorder. CNS Drugs. 2014;28(11):1059–1069. doi:10.1007/s40263-014-0188-9 PubMed
138. Dittmann RW, Cardo E, Nagy P, et al. Efficacy and safety of lisdexamfetamine dimesylate and atomoxetine in the treatment of attention-deficit/hyperactivity disorder: a head-to-head, randomized, double-blind, phase IIIb study. CNS Drugs. 2013;27(12):1081–1092. doi:10.1007/s40263-013-0104-8 PubMed
139. Dupaul GJ, Weyandt LL, Rossi JS, et al. Double-blind, placebo-controlled, crossover study of the efficacy and safety of lisdexamfetamine dimesylate in college students with ADHD. J Atten Disord. 2012;16(3):202–220. doi:10.1177/1087054711427299 PubMed
140. Faraone SV, Spencer TJ, Kollins SH, et al. Dose response effects of lisdexamfetamine dimesylate treatment in adults with ADHD: an exploratory study. J Atten Disord. 2012;16(2):118–127. doi:10.1177/1087054711403716 PubMed
141. Findling RL, Adeyi B, Dirks B, et al. Parent-reported executive function behaviors and clinician ratings of attention-deficit/hyperactivity disorder symptoms in children treated with lisdexamfetamine dimesylate. J Child Adolesc Psychopharmacol. 2013;23(1):28–35. doi:10.1089/cap.2011.0120 PubMed
142. Findling RL, Childress AC, Cutler AJ, et al. Efficacy and safety of lisdexamfetamine dimesylate in adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2011;50(4):395–405. doi:10.1016/j.jaac.2011.01.007 PubMed
143. Findling RL, Cutler AJ, Saylor K, et al. A long-term open-label safety and effectiveness trial of lisdexamfetamine dimesylate in adolescents with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2013;23(1):11–21. doi:10.1089/cap.2011.0088 PubMed
144. Findling RL, Ginsberg LD, Jain R, et al. Effectiveness, safety, and tolerability of lisdexamfetamine dimesylate in children with attention-deficit/hyperactivity disorder: an open-label, dose-optimization study. J Child Adolesc Psychopharmacol. 2009;19(6):649–662. doi:10.1089/cap.2008.0165 PubMed
145. Ginsberg L, Katic A, Adeyi B, et al. Long-term treatment outcomes with lisdexamfetamine dimesylate for adults with attention-deficit/hyperactivity disorder stratified by baseline severity. Curr Med Res Opin. 2011;27(6):
1097–1107. doi:10.1185/03007995.2011.567256 PubMed
146. Jain R, Babcock T, Burtea T, et al. Efficacy and safety of lisdexamfetamine dimesylate in children with attention-deficit/hyperactivity disorder and recent methylphenidate use. Adv Ther. 2013;30(5):472–486. doi:10.1007/s12325-013-0027-2 PubMed
147. Mattingly GW, Weisler RH, Young J, et al. Clinical response and symptomatic remission in short- and long-term trials of lisdexamfetamine dimesylate in adults with attention-deficit/hyperactivity disorder. BMC Psychiatry. 2013;13(1):39. doi:10.1186/1471-244X-13-39 PubMed
148. Turgay A, Ginsberg L, Sarkis E, et al. Executive function deficits in children with attention-deficit/hyperactivity disorder and improvement with lisdexamfetamine dimesylate in an open-label study. J Child Adolesc Psychopharmacol. 2010;20(6):
503–511. doi:10.1089/cap.2009.0110 PubMed
149. Weisler R, Young J, Mattingly G, et al; 304 Study Group. Long-term safety and effectiveness of lisdexamfetamine dimesylate in adults with attention-deficit/ hyperactivity disorder. CNS Spectr. 2009;14(10):573–585. PubMed
150. Wigal T, Brams M, Gasior M, et al. Effect size of lisdexamfetamine dimesylate in adults with attention-deficit/hyperactivity disorder. Postgrad Med. 2011;123(2):169–176. doi:10.3810/pgm.2011.03.2275 PubMed
151. Wigal T, Brams M, Gasior M, et al; 316 Study Group. Randomized, double-blind, placebo-controlled, crossover study of the efficacy and safety of lisdexamfetamine dimesylate in adults with attention-deficit/hyperactivity disorder: novel findings using a simulated adult workplace environment design. Behav Brain Funct. 2010;6(1):34. doi:10.1186/1744-9081-6-34 PubMed
152. Zenzedi [package insert]. Dextroamphetamine sulfate. Atlanta, GA: Arbor Pharmaceuticals; 2014.
153. Evekeo [package insert]. Amphetamine sulfate tablets. Atlanta, GA: Arbor Pharmaceuticals; 2015.
154. Childress AC, Brams M, Cutler AJ, et al. The efficacy and safety of evekeo, racemic amphetamine sulfate, for treatment of attention-deficit/hyperactivity disorder symptoms: a multicenter, dose-optimized, double-blind, randomized, placebo-controlled crossover laboratory classroom study. J Child Adolesc Psychopharmacol. 2015;25(5):402–414. doi:10.1089/cap.2014.0176 PubMed
155. Procentra [package insert]. Dextroamphetamine sulfate solution. Keyport, KY: Independence Pharmaceuticals; 2015.
156. Dyanavel XR [package insert]. Amphetamine extended-release oral suspension. Monmouth Junction, NJ: Tris Pharma, Inc; 2015.
157. Adzenys XR-ODT. [package insert]. Amphetamine extended-release orally disintegrating tablets. Grand Prairie, TX: Neos Therapeutics; 2016.
158. Spencer TJ, Adler LA, Weisler RH, et al. Triple-bead mixed amphetamine salts (SPD465), a novel, enhanced extended-release amphetamine formulation for the treatment of adults with ADHD: a randomized, double-blind, multicenter, placebo-controlled study. J Clin Psychiatry. 2008;69(9):1437–1448. doi:10.4088/JCP.v69n0911 PubMed
159. Pennick M. Absorption of lisdexamfetamine dimesylate and its enzymatic conversion to d-amphetamine. Neuropsychiatr Dis Treat. 2010;6:317–327. doi:10.2147/NDT.S9749 PubMed
160. Strattera [package insert]. Atomoxetine. Indianapolis, IN: Lilly USA; 2002.
161. Vaughan B, Fegert J, Kratochvil CJ. Update on atomoxetine in the treatment of attention-deficit/hyperactivity disorder. Expert Opin Pharmacother. 2009;10(4):669–676. doi:10.1517/14656560902762873 PubMed
162. Adler L, Tanaka Y, Williams D, et al. Executive function in adults with attention-deficit/hyperactivity disorder during treatment with atomoxetine in a randomized, placebo-controlled, withdrawal study. J Clin Psychopharmacol. 2014;34(4):461–466. doi:10.1097/JCP.0000000000000138 PubMed
163. Adler LA, Spencer T, Brown TE, et al. Once-daily atomoxetine for adult attention-deficit/hyperactivity disorder: a 6-month, double-blind trial. J Clin Psychopharmacol. 2009;29(1):44–50. doi:10.1097/JCP.0b013e318192e4a0 PubMed
164. Block SL, Kelsey D, Coury D, et al. Once-daily atomoxetine for treating pediatric attention-deficit/hyperactivity disorder: comparison of morning and evening dosing. Clin Pediatr (Phila). 2009;48(7):723–733. doi:10.1177/0009922809335321 PubMed
165. Durell TM, Adler LA, Williams DW, et al. Atomoxetine treatment of attention-deficit/hyperactivity disorder in young adults with assessment of functional outcomes: a randomized, double-blind, placebo-controlled clinical trial. J Clin Psychopharmacol. 2013;33(1):45–54. doi:10.1097/JCP.0b013e31827d8a23 PubMed
166. Durell TM, Pumariega AJ, Rothe EM, et al. Effects of open-label atomoxetine on African-American and Caucasian pediatric outpatients with attention-deficit/hyperactivity disorder. Ann Clin Psychiatry. 2009;21(1):26–37. PubMed
167. Fuentes J, Danckaerts M, Cardo E, et al. Long-term quality-of-life and functioning comparison of atomoxetine versus other standard treatment in pediatric attention-deficit/hyperactivity disorder. J Clin Psychopharmacol. 2013;33(6):766–774. doi:10.1097/JCP.0b013e31829c762b PubMed
168. Hammerness P, Doyle R, Kotarski M, et al. Atomoxetine in children with attention-deficit hyperactivity disorder with prior stimulant therapy: a prospective open-label study. Eur Child Adolesc Psychiatry. 2009;18(8):493–498. doi:10.1007/s00787-009-0017-8 PubMed
169. Hirata Y, Goto T, Takita Y, et al. Long-term safety and tolerability of atomoxetine in Japanese adults with attention deficit hyperactivity disorder. Asia Pac Psychiatry. 2014;6(3):292–301. doi:10.1111/appy.12119 PubMed
170. Johnson M, Cederlund M, Råstam M, et al. Open-label trial of atomoxetine hydrochloride in adults with ADHD. J Atten Disord. 2010;13(5):539–545. doi:10.1177/1087054709332372 PubMed
171. Kratochvil CJ, Vaughan BS, Stoner JA, et al. A double-blind, placebo-controlled study of atomoxetine in young children with ADHD. Pediatrics. 2011;127(4):e862–e868. doi:10.1542/peds.2010-0825 PubMed
172. Martenyi F, Zavadenko NN, Jarkova NB, et al. Atomoxetine in children and adolescents with attention-deficit/hyperactivity disorder: a 6-week, randomized, placebo-controlled, double-blind trial in Russia. Eur Child Adolesc Psychiatry. 2010;19(1):57–66. doi:10.1007/s00787-009-0042-7 PubMed
173. Montoya A, Hervas A, Cardo E, et al. Evaluation of atomoxetine for first-line treatment of newly diagnosed, treatment-naïve children and adolescents with attention deficit/hyperactivity disorder. Curr Med Res Opin. 2009;25(11):2745–2754. doi:10.1185/03007990903316152 PubMed
174. Sobanski E, Sabljic D, Alm B, et al. A randomized, waiting list-controlled 12-week trial of atomoxetine in adults with ADHD. Pharmacopsychiatry. 2012;45(3):100–107. doi:10.1055/s-0031-1291319 PubMed
175. Surman C, Hammerness P, Petty C, et al. Atomoxetine in the treatment of adults with subthreshold and/or late onset attention-deficit hyperactivity disorder-not otherwise specified (ADHD-NOS): a prospective open-label 6-week study. CNS Neurosci Ther. 2010;16(1):6–12. doi:10.1111/j.1755-5949.2009.00124.x PubMed
176. Takahashi M, Takita Y, Yamazaki K, et al. A randomized, double-blind, placebo-controlled study of atomoxetine in Japanese children and adolescents with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2009;19(4):341–350. doi:10.1089/cap.2008.0154 PubMed
177. Waxmonsky JG, Waschbusch DA, Akinnusi O, et al. A comparison of atomoxetine administered as once versus twice daily dosing on the school and home functioning of children with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2011;21(1):21–32. doi:10.1089/cap.2010.0042 PubMed
178. Wehmeier PM, Schacht A, Ulberstad F, et al. Does atomoxetine improve executive function, inhibitory control, and hyperactivity? results from a placebo-controlled trial using quantitative measurement technology. J Clin Psychopharmacol. 2012;32(5):653–660. doi:10.1097/JCP.0b013e318267c304 PubMed
179. Wehmeier PM, Schacht A, Wolff C, et al. Neuropsychological outcomes across the day in children with attention-deficit/hyperactivity disorder treated with atomoxetine: results from a placebo-controlled study using a computer-based continuous performance test combined with an infra-red motion-tracking device. J Child Adolesc Psychopharmacol. 2011;21(5):
433–444. doi:10.1089/cap.2010.0142 PubMed
180. Wietecha LA, Williams DW, Herbert M, et al. Atomoxetine treatment in adolescents with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2009;19(6):719–730. doi:10.1089/cap.2008.074 PubMed
181. Young JL, Sarkis E, Qiao M, et al. Once-daily treatment with atomoxetine in adults with attention-deficit/hyperactivity disorder: a 24-week, randomized, double-blind, placebo-controlled trial. Clin Neuropharmacol. 2011;34(2):51–60. PubMed
182. Intuniv [package insert]. Guanfacine extended-release tablets. Wayne, PA: Shire USA, Inc; 2013.
183. Strange BC. Once-daily treatment of ADHD with guanfacine: patient implications. Neuropsychiatr Dis Treat. 2008;4(3):499–506. doi:10.2147/NDT.S1711 PubMed
184. Hervas A, Huss M, Johnson M, et al. Efficacy and safety of extended-release guanfacine hydrochloride in children and adolescents with attention-deficit/hyperactivity disorder: a randomized, controlled, phase III trial. Eur Neuropsychopharmacol. 2014;24(12):1861–1872. doi:10.1016/j.euroneuro.2014.09.014 PubMed
185. Newcorn JH, Stein MA, Childress AC, et al. Randomized, double-blind trial of guanfacine extended release in children with attention-deficit/hyperactivity disorder: morning or evening administration. J Am Acad Child Adolesc Psychiatry. 2013;52(9):921–930. doi:10.1016/j.jaac.2013.06.006 PubMed
186. Sallee FR, Kollins SH, Wigal TL. Efficacy of guanfacine extended release in the treatment of combined and inattentive only subtypes of attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2012;22(3):206–214. doi:10.1089/cap.2010.0135 PubMed
187. Sallee FR, Lyne A, Wigal T, et al. Long-term safety and efficacy of guanfacine extended release in children and adolescents with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2009;19(3):215–226. doi:10.1089/cap.2008.0080 PubMed
188. Sallee FR, McGough J, Wigal T, et al; SPD503 STUDY GROUP. Guanfacine extended release in children and adolescents with attention-deficit/hyperactivity disorder: a placebo-controlled trial. J Am Acad Child Adolesc Psychiatry. 2009;48(2):155–165. doi:10.1097/CHI.0b013e318191769e PubMed
189. Spencer TJ, Greenbaum M, Ginsberg LD, et al. Safety and effectiveness of coadministration of guanfacine extended release and psychostimulants in children and adolescents with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2009;19(5):501–510. doi:10.1089/cap.2008.0152 PubMed
190. Wilens TE, Bukstein O, Brams M, et al. A controlled trial of extended-release guanfacine and psychostimulants for attention-deficit/hyperactivity disorder.
J Am Acad Child Adolesc Psychiatry. 2012;51(1):74–85, e2. doi:10.1016/j.jaac.2011.10.012 PubMed
191. Young J, Rugino T, Dammerman R, et al. Efficacy of guanfacine extended release assessed during the morning, afternoon, and evening using a modified Conners’ Parent Rating Scale-revised: Short Form. J Child Adolesc Psychopharmacol. 2014;24(8):
435–441. doi:10.1089/cap.2013.0134 PubMed
192. Kapvay [package insert]. Clonidine hydrochloride extended-release tablets. Atlanta, GA: Shionogi Pharma; 2010.
193. Jain R, Segal S, Kollins SH, et al. Clonidine extended-release tablets for pediatric patients with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2011;50(2):171–179. doi:10.1016/j.jaac.2010.11.005 PubMed
194. Hopkins SC, Sunkaraneni S, Skende E, et al. Pharmacokinetics and exposure-response relationships of dasotraline in the treatment of attention-deficit/hyperactivity disorder in adults. Clin Drug Investig. 2016;36(2):137–146. doi:10.1007/s40261-015-0358-7 PubMed
195. Koblan KS, Hopkins SC, Sarma K, et al. Dasotraline for the treatment of attention-deficit/hyperactivity disorder: a randomized, double-blind, placebo-controlled, proof-of-concept trial in adults. Neuropsychopharmacology. 2015;40(12):
2745–2752. doi:10.1038/npp.2015.124 PubMed
196. Manor I, Rubin J, Daniely Y, et al. Attention benefits after a single dose of metadoxine extended release in adults with predominantly inattentive ADHD. Postgrad Med. 2014;126(5):7–16. doi:10.3810/pgm.2014.09.2795 PubMed
197. Manor I, Newcorn JH, Faraone SV, et al. Efficacy of metadoxine extended release in patients with predominantly inattentive subtype attention-deficit/hyperactivity disorder. Postgrad Med. 2013;125(4):
181–190. doi:10.3810/pgm.2013.07.2689 PubMed
198. Manor I, Ben-Hayun R, Aharon-Peretz J, et al. A randomized, double-blind, placebo-controlled, multicenter study evaluating the efficacy, safety, and tolerability of extended-release metadoxine in adults with attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2012;73(12):
1517–1523. doi:10.4088/JCP.12m07767 PubMed
199. Sasaki T, Hashimoto K, Tachibana M, et al. Tipepidine in children with attention deficit/hyperactivity disorder: a 4-week, open-label, preliminary study. Neuropsychiatr Dis Treat. 2014;10:147–151. doi:10.2147/NDT.S58480 PubMed
200. Weiss MD, Weiss JR. A guide to the treatment of adults with ADHD. J Clin Psychiatry. 2004;65(suppl 3):27–37. PubMed
201. Stiefel G, Besag FM. Cardiovascular effects of methylphenidate, amphetamines and atomoxetine in the treatment of attention-deficit hyperactivity disorder. Drug Saf. 2010;33(10):821–842. doi:10.2165/11536380-000000000-00000 PubMed
202. Robertson MM. Attention deficit hyperactivity disorder, tics and Tourette’s syndrome: the relationship and treatment implications: a commentary. Eur Child Adolesc Psychiatry. 2006;15(1):1–11. doi:10.1007/s00787-006-0505-z PubMed
203. Steingard R, Biederman J, Spencer T, et al. Comparison of clonidine response in the treatment of attention-deficit hyperactivity disorder with and without comorbid tic disorders. J Am Acad Child Adolesc Psychiatry. 1993;32(2):350–353. doi:10.1097/00004583-199303000-00016 PubMed
204. Faraone SV, Glatt SJ. Effects of extended-release guanfacine on ADHD symptoms and sedation-related adverse events in children with ADHD. J Atten Disord. 2010;13(5):
532–538. doi:10.1177/1087054709332472 PubMed
205. Bello NT. Clinical utility of guanfacine extended release in the treatment of ADHD in children and adolescents. Patient Prefer Adherence. 2015;9:877–885. doi:10.2147/PPA.S73167 PubMed
206. Biederman J, Melmed RD, Patel A, et al; SPD503 Study Group. A randomized, double-blind, placebo-controlled study of guanfacine extended release in children and adolescents with attention-deficit/hyperactivity disorder. Pediatrics. 2008;121(1):e73–e84. doi:10.1542/peds.2006-3695 PubMed
207. Cruz MP. Guanfacine extended-release tablets (Intuniv), a nonstimulant selective Alpha(2A)-Adrenergic receptor agonist for attention-deficit/hyperactivity disorder. P T. 2010;35(8):448–451. PubMed
208. Kollins SH, Jain R, Brams M, et al. Clonidine extended-release tablets as add-on therapy to psychostimulants in children and adolescents with ADHD. Pediatrics. 2011;127(6):e1406–e1413. doi:10.1542/peds.2010-1260 PubMed
209. Arnold LE. Methylphenidate vs amphetamine: comparative review. J Atten Disord. 2000;3(4):200–211. doi:10.1177/108705470000300403
210. Childress AC, Sallee FR. Attention-deficit/hyperactivity disorder with inadequate response to stimulants: approaches to management. CNS Drugs. 2014;28(2):121–129. doi:10.1007/s40263-013-0130-6 PubMed
211. Setyawan J, Yang H, Cheng D, et al. Developing a risk score to guide individualized treatment selection in attention deficit/hyperactivity disorder. Value Health. 2015;18(6):824–831. doi:10.1016/j.jval.2015.06.005 PubMed
212. Adler LD, Nierenberg AA. Review of medication adherence in children and adults with ADHD. Postgrad Med. 2010;122(1):
184–191. doi:10.3810/pgm.2010.01.2112 PubMed
213. Gajria K, Lu M, Sikirica V, et al. Adherence, persistence, and medication discontinuation in patients with attention-deficit/hyperactivity disorder – a systematic literature review. Neuropsychiatr Dis Treat. 2014;10:1543–1569. PubMed
214. Ahmed R, Aslani P. Attention-deficit/hyperactivity disorder: an update on medication adherence and persistence in children, adolescents and adults. Expert Rev Pharmacoecon Outcomes Res. 2013;13(6):
791–815. doi:10.1586/14737167.2013.841544 PubMed
215. Bhosle M, Benner JS, Dekoven M, et al. Difficult to swallow: patient preferences for alternative valproate pharmaceutical formulations. Patient Prefer Adherence. 2009;3:161–171. doi:10.2147/PPA.S5691 PubMed
216. Polaha J, Dalton WT 3rd, Lancaster BM. Parental report of medication acceptance among youth: implications for everyday practice. South Med J. 2008;101(11):1106–1112. doi:10.1097/SMJ.0b013e318180645d PubMed
217. Schiele JT, Quinzler R, Klimm HD, et al. Difficulties swallowing solid oral dosage forms in a general practice population: prevalence, causes, and relationship to dosage forms. Eur J Clin Pharmacol. 2013;69(4):937–948. doi:10.1007/s00228-012-1417-0 PubMed
218. FDA Center for Drug Evaluation and Research. Guidance for industry: bioavailability and bioequivalence studies for orally administered drug products—general considerations. FDA Web site. http://www.fda.gov/ohrms/dockets/ac/03/briefing/3995B1_07_GFI-BioAvail-BioEquiv.pdf. Accessed December 7, 2015.
219. Lally MD, Kral MC, Boan AD. Not All generic Concerta is created equal: comparison of OROS versus non-OROS for the treatment of ADHD. Clin Pediatr (Phila). 2015. doi:10.1177/0009922815611647 PubMed
Enjoy this premium PDF as part of your membership benefits!


