Transdermal Patches for the Treatment of Neurologic Conditions in Elderly Patients: A Review

ABSTRACT
Objective: The mode of drug delivery can be an important consideration in optimizing drug therapy, as it can affect treatment compliance and outcomes. It is particularly important to develop optimal drug formulations for chronic diseases or conditions in the elderly for which treatment compliance is known to be low. In this review, the features and benefits of transdermal formulations for treating neurologic conditions in elderly patients are described.
Data Sources: English-language articles were identified by searching MEDLINE in November 2010 (there were no search parameters on date of publication) using the search terms transdermal patch, transdermal system, neurology, rivastigmine, rotigotine, selegiline, lidocaine, capsaicin, compliance, and neuropathic pain.
Data Selection: Articles describing the development, use, efficacy, and safety of licensed transdermal patch treatments for neurologic conditions that affect the elderly were included.
Data Extraction: The features of transdermal systems and comparisons between transdermal and oral formulations for the treatment of specific neurologic conditions in elderly patients were reviewed.
Data Synthesis: There are 5 transdermal patch systems currently available for neurologic conditions in adults: rivastigmine, rotigotine, selegiline, lidocaine, and capsaicin. These are all modern formulations in matrix patches, developed to provide appropriate drug dosage in an acceptable and well-tolerated form.
Conclusions: Transdermal patches can offer benefits to patients over oral formulations in terms of ease of use, simple treatment regimens, avoidance of the first-pass effect, and avoidance of high maximum plasma concentrations with rapid changes in drug levels, without the invasive procedures associated with intravenous treatment.
Prim Care Companion CNS Disord 2011;13(6):doi:10.4088/PCC.11r01149
© Copyright 2011 Physicians Postgraduate Press, Inc.
Submitted: January 25, 2011; accepted May 18, 2011.
Published online: December 08, 2011.
Corresponding author: Martin R. Farlow, MD, Department of Neurology, Clinical Bldg, Room 291, 541 Clinical Dr, Indiana University School of Medicine, Indianapolis, IN 46202 ([email protected]).
The mode of drug delivery can be an important consideration in prescribing drugs, and different routes may benefit the pharmacokinetic profile of the drug or the disease or condition. Transdermal patch delivery systems have been developed for several drugs used in the treatment of neurologic conditions and may provide practical and pharmacokinetic advantages over oral drug administration.1 Drug formulation can also affect the acceptability of treatment to the patient in terms of ease of use and tolerability. These factors can in turn greatly affect treatment compliance and hence treatment outcomes. It is particularly important to develop appropriate treatment formulations and modes of therapy for diseases or conditions that mainly affect the elderly, as they are more likely to have several coexisting conditions and to be taking multiple concomitant medications. In this review, we describe the features and benefits of transdermal formulations for treating neurologic conditions in elderly patients.
Method
In November 2010 (there were no search parameters on date of publication), English-language articles describing the development, use, efficacy, and safety of licensed transdermal patch treatments for neurologic conditions that affect the elderly were identified by searching MEDLINE. The following search terms were used: transdermal patch, transdermal system, neurology, rivastigmine, rotigotine, selegiline, lidocaine, capsaicin, compliance, and neuropathic pain.
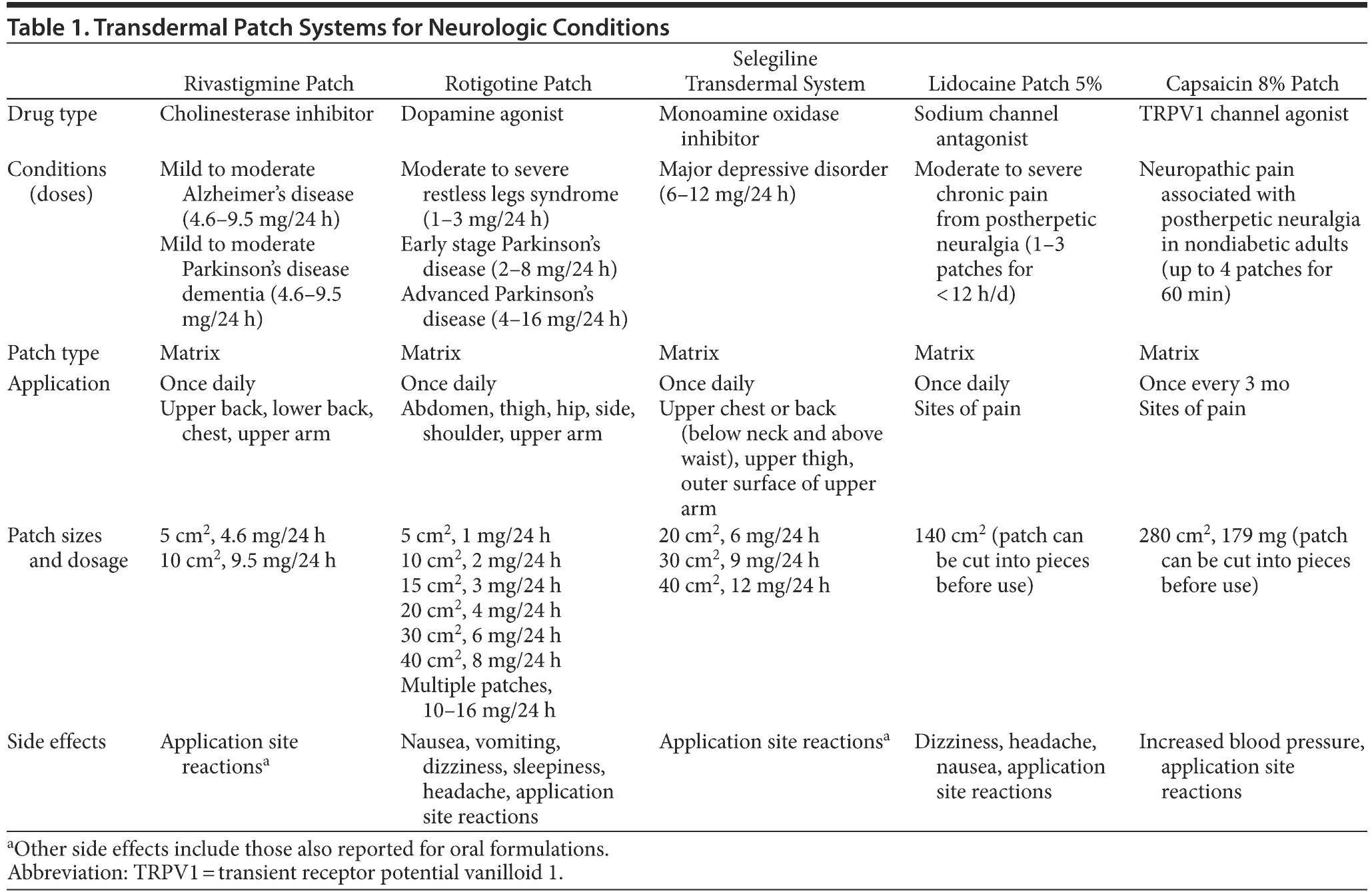
We discuss the use of the 5 transdermal patch delivery systems available for the treatment of neurologic conditions in adult populations. These include the rivastigmine patch for the treatment of mild to moderate Alzheimer’s disease and Parkinson’s disease dementia,2 the rotigotine patch for the treatment of Parkinson’s disease and restless legs syndrome,3 the selegiline transdermal system for the treatment of major depressive disorder,4 and the lidocaine 5%5 patch and capsaicin 8%6 patch for the relief of pain from postherpetic neuralgia (Table 1).
Transdermal Formulations
Desirable Features of a Transdermal Patch
Modern transdermal patch systems are developed to provide appropriate drug dosage in an easy-to-use formulation that is acceptable to the patient and the caregiver. To achieve these goals, the matrix-type patch is frequently employed, as these patches are small and thin compared to the older and less discreet reservoir-type patches and adhere better to the skin. The matrix system comprises 4 main components: a colored backing layer; an acrylic matrix containing the drug, antioxidants, and an acrylic polymer mixture; a silicone matrix adhesive layer; and a release liner that is resistant to humidity and to the drugs within the preparation. Matrix patches are also generally well tolerated as, unlike many reservoir patches, the drug is not contained in an alcohol solution known to irritate the skin.7 The 5 patches discussed in this article are all matrix-type patches (Table 1).2-6

- Transdermal patches have a number of advantages over oral formulations.
- The benefits of transdermal administration may increase compliance in elderly patients with chronic conditions such as Alzheimer’s disease and Parkinson’s disease.
- There is the potential to extend the use of transdermal patches to new medications and indications.
An important feature of transdermal patches is application site tolerability. In general, matrix-type patches have been shown to be well tolerated, even in elderly populations, with the majority of skin reactions being mild to moderate in severity and transient.8-11 Advice to limit or manage skin tolerability when using transdermal patches can be found in the prescribing information for these medications or in Ale et al.8 In summary, patches should be applied to clean and undamaged skin. They should be removed carefully, and the site of application should be rotated.8
The use of transdermal patches is convenient to patients, as the patches are often only applied once daily, while oral medications may need to be taken several times a day to maintain adequate drug levels. Transdermal patches also offer an alternative mode of drug delivery for oral medications that cannot be crushed or chewed. Additionally, patches can be used to provide the drug over longer periods of time such as a week. This type of formulation may reduce the medication burden on patients, but patients may find it more difficult to remember to replace their patch compared with a daily routine. Also, more frequent application site reactions may affect the tolerability of weekly formulations, as the risk of skin reactions increases with the size of the patch and the duration of contact with the skin.12
What Are the Potential Advantages and Challenges of Transdermal Patches Versus Oral Formulations?
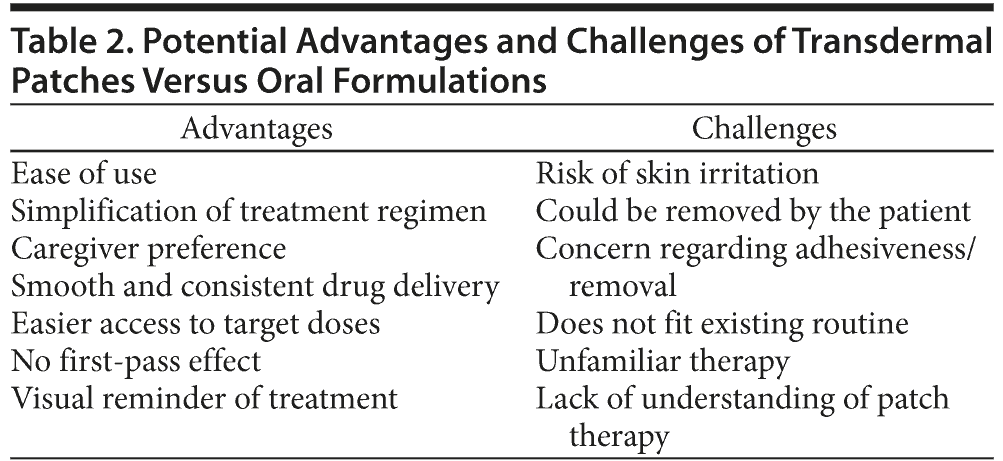
Transdermal patch formulations are most often developed to optimize drug delivery, efficacy, and tolerability and are particularly appropriate for those drugs with a short half-life, poor oral absorption, or low tolerability of the oral formulation. Transdermal patch formulations provide a noninvasive technique to deliver a steady supply of drug molecules directly into the circulation, avoiding the first-pass effect (Table 2). However, the use of transdermal systems of delivery is restricted to those drugs able to penetrate the skin and enter the blood system, although new technological developments may extend the range of drugs appropriate for transdermal delivery.
Drug is delivered directly to the circulatory system. Transdermal patches deliver drugs directly into the circulatory system, bypassing the gastrointestinal system and avoiding the hepatic first-pass effect. This delivery system is advantageous for the treatment of depression with monoamine oxidase inhibitors (MAOIs). Although selective for MAO-B, selegiline also inhibits MAO-A; therefore, the clinical use of oral formulations is limited by the risk of drug-food interactions due to inhibition of the MAO-A enzyme in the intestine and liver.13 This inhibition can lead to dietary tyramine entering the circulatory system, stimulating the release of norepinephrine and causing hypertensive crisis.13 Consequently, oral MAOIs require strict dietary restrictions to limit tyramine intake.13
The selegiline transdermal system delivers selegiline to the systemic circulation, avoiding direct inhibition of MAO-A in the intestine and the first-pass effect in the liver.14 The 6-mg/24-hour selegiline patch has been approved in the United States for the treatment of major depressive disorder without the need for dietary restriction.4 Dietary restriction is currently required for the 9-mg/24-hour and 12-mg/24-hour doses, as the safety and tolerability data for these doses are more limited.4
Rotigotine is a dopamine agonist administered by transdermal patch and is currently approved for the treatment of early and advanced stage Parkinson’s disease and restless legs syndrome in Europe.15 Rotigotine is only available as a transdermal patch formulation due to an extensive first-pass effect and low bioavailability when taken orally.16 As a transdermal formulation, the rotigotine patch can also offer a noninvasive alternative for patients temporarily or permanently unable to take oral formulations. For example, the patch delivery system may also be useful for the management of parkinsonian symptoms in the perioperative period when patients with Parkinson’s disease are unable to adhere to an oral regimen of dopamine agonists.17 The feasibility of replacing a patient’s usual treatment regimen with the rotigotine patch for surgery was demonstrated in a small open-label study of patients with Parkinson’s disease who needed surgery under general anesthesia.17
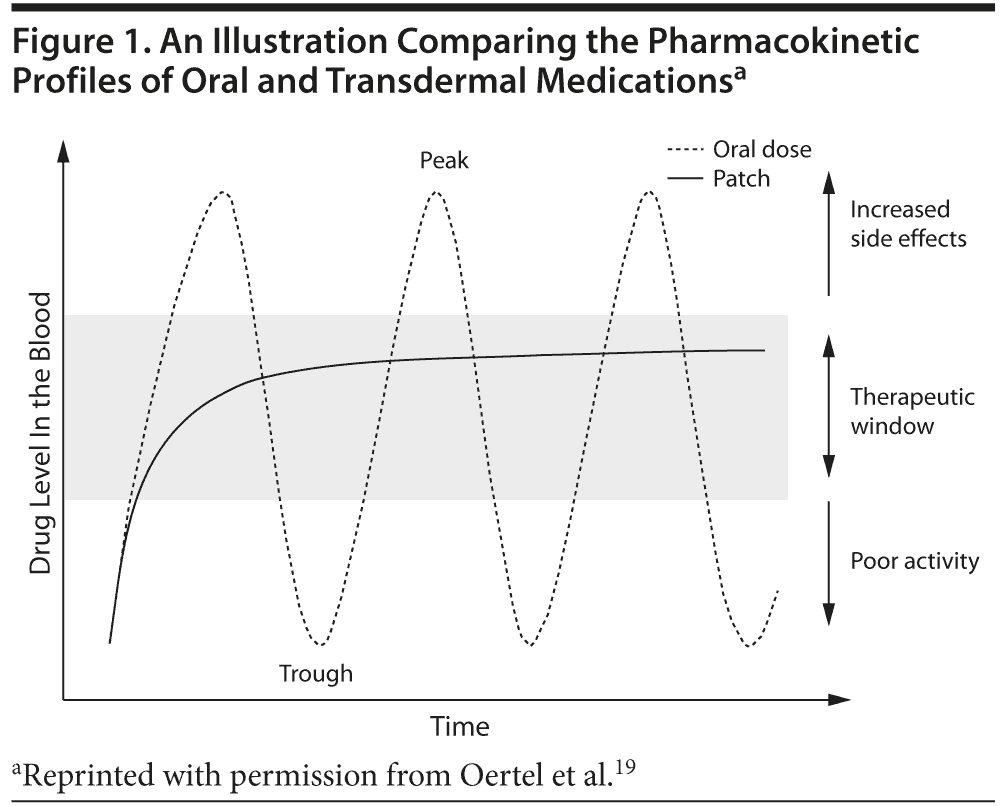
Continuous drug delivery. A major advantage of transdermal patches over oral formulations is their continuous delivery of drug, resulting in steady plasma concentrations with lower maximum plasma levels (Cmax) (Figure 1).18,19 This steady and continuous drug delivery can reduce tolerability issues associated with fluctuations in plasma concentrations and can also increase the number of patients able to achieve therapeutic doses. Both of these factors are beneficial, especially to elderly patients, who may be more sensitive to fluctuating plasma levels due to reduced hepatic and renal functioning, other health conditions, and use of concomitant medications. The smooth drug delivery of transdermal patches may increase treatment compliance by decreasing adverse events and improving the tolerability of therapeutic doses, as adverse events and perceived lack of efficacy have both been reported as reasons for noncompliance that are relevant to elderly populations with chronic conditions.20
Rivastigmine is a cholinesterase inhibitor used for the treatment of mild to moderate Alzheimer’s disease and Parkinson’s disease dementia2,21 and is available in both oral and transdermal formulations. With the oral rivastigmine capsule formulation, both the high Cmax and the short time from administration to Cmax are associated with centrally induced cholinergic side effects such as nausea and vomiting.22 Since the tolerability of rivastigmine capsules was improved when the same daily dosage was administered over 3 doses rather than 2,23 the rivastigmine patch was developed with the aim of providing improved patient tolerability. The 9.5-mg/24-hour rivastigmine patch provides comparable drug exposure to 12-mg/day rivastigmine capsules,22 but with lower fluctuations in plasma concentrations.22,24 Variation between patients in terms of Cmax and drug exposure is also reduced with the rivastigmine patch compared with capsules, demonstrating the improved reliability of the transdermal route.25 In a large clinical trial comparing the rivastigmine patch with capsules and placebo, the 9.5-mg/24-hour rivastigmine patch was associated with 3 times fewer reports of nausea and vomiting compared with 12-mg/day rivastigmine capsules.26 This improved tolerability profile allows easier access to the target therapeutic dose, with 95.9% of patients assigned to the rivastigmine patch achieving the 9.5-mg/24-hour dose compared with 64.6% of patients reaching the 12-mg/day dose with rivastigmine capsules.27
Transdermal patch formulations of dopamine agonists, rather than oral formulations, may be preferable for the treatment of patients with Parkinson’s disease, as pulsatile dopaminergic stimulation may lead to dyskinesias and other motor complications.28 Hence, the smooth and continuous delivery associated with transdermal patches may limit the development of side effects. Rotigotine is currently the only dopamine agonist available as a transdermal formulation and has been shown to provide clinical efficacy and tolerability similar to orally administered nonergot dopamine agonists.29,30 Further research is needed to determine whether transdermal delivery reduces the occurrence of motor complications.29 The rotigotine patch is also indicated for the treatment of restless legs syndrome and has been shown to significantly reduce symptoms relative to placebo31,32 and to provide sustained efficacy over a period of 2 years.33 Continuous drug delivery throughout both the day and night is thought to benefit patients with this condition.
Simplification of treatment regimen. Transdermal patch formulations can simplify drug treatment schedules, especially for those drugs that are rapidly metabolized or excreted from the body, as one patch can replace several doses of oral medication. Simplification of treatment regimens may help patients adhere to a course of drug therapy and receive maximum benefit from the treatment. In the case of elderly patients with long-term neurologic conditions, noncompliance to treatment is a recognized issue.20 These patients are often taking multiple medications, and, so, any reduction in the frequency of dosing may increase compliance and may also reduce the burden on their caregivers.20,34
Rivastigmine is an example of a drug with a simpler treatment regimen for the transdermal formulation than the oral formulation because of the short elimination half-life of oral rivastigmine and the gastrointestinal tolerability problems associated with high plasma levels.35 While oral rivastigmine is administered twice daily, controlled drug release over 24 hours by the rivastigmine patch allows therapeutic plasma concentrations to be maintained with a single daily application.22 There are also fewer steps in the titration schedule for the rivastigmine patch compared with rivastigmine capsules, further simplifying the treatment regimen. Transdermal rivastigmine is initiated at 4.6 mg/24 hours and can then be increased to the target therapeutic dose of 9.5 mg/24 hours after 4 weeks, while the comparable 12-mg/day rivastigmine capsule dose can only be reached 8 weeks later, after 3 dose increases and 12 weeks of treatment.2,22 A much larger percentage of patients achieve optimal 9.5-mg/24-hour dosing by rivastigmine patch than by the comparable 12-mg/day oral rivastigmine capsules.26 At the end of a 6-month clinical trial comparing rivastigmine patch, rivastigmine capsules, and placebo in patients with Alzheimer’s disease, 72% of caregivers preferred the rivastigmine patch to rivastigmine capsules.36 Ease of following the dosing schedule was the most common reason provided for preferring transdermal patch delivery.36
Local relief of neuropathic pain. Transdermal patches can also be used to supply drugs locally at the application site. Local absorption can be beneficial when systemic exposure is associated with adverse events or the risk of drug interactions. The lidocaine 5% patch and the capsaicin 8% patch are both approved for the local management of pain associated with postherpetic neuralgia.5,6 Transdermal application allows the treatment to be targeted to the area of pain and also limits exposure to the drug. Lidocaine and capsaicin patches are both associated with low systemic exposure5,37,38 and so may relieve pain with minimal risk of system toxicity.1 These patches may be suitable for elderly patients who are likely to also be taking other medications, as the other treatment options for neuropathic pain (eg, tricyclic antidepressants, gabapentinoids, selective norepinephrine reuptake inhibitors, sodium channel blockers, and opioid agonists) are associated with systemic adverse events and the risk of drug interactions.1,39,40 The capsaicin patch is applied by a doctor to the area of pain for 60 minutes every 3 months.6 Although application of the capsaicin patch is associated with temporary redness and pain, the simple treatment regimen and sustained pain relief may help maintain treatment compliance.41,42
Practical advantages of transdermal delivery. Transdermal patch systems are simple to use and can usually be applied independently of meals. One particular advantage is the physical presence of the patch on the skin. This presence can provide visual reassurance to the patient or caregiver that the medication has been properly administered.20 In a survey of caregivers, ease of use of transdermal patches was a common reason for preferring the patch to capsule medication.36
Challenges of transdermal delivery. Despite recent advances in patch technology, skin application site reactions may still occur with transdermal medications. The majority of application site reactions that occur with modern transdermal medications are mild in severity and resolve spontaneously after patch removal.8 Often, the potential advantages of using transdermal medications over oral forms outweigh minor skin reactions. Preventative and palliative measures are required to limit application site reactions; patches should be applied to clean, dry, unbroken skin, and regular rotation of the application site is necessary to prevent cumulative irritant contact dermatitis.8
CONCLUSIONS
Transdermal patches are an important method of drug delivery that can offer benefits to patients over oral formulations in terms of ease of use, simple treatment regimens, avoidance of the first-pass effect, and avoidance of peak dose effects. These benefits may be particularly relevant for treatment compliance and tolerability in elderly patients with chronic conditions including Alzheimer’s disease and Parkinson’s disease. Looking forward, new technologies have the potential to extend the use of transdermal patches to new medications and indications.
Drug names: capsaicin (Qutenza), lidocaine (Lidoderm), rivastigmine (Exelon), selegiline transdermal system (EMSAM).
Author affiliations: Department of Neurology, Indiana University School of Medicine, Indianapolis (Dr Farlow); and Novartis Pharmaceuticals Corporation, East Hanover, New Jersey (Dr Somogyi).
Potential conflicts of interest: Dr Farlow has received grant/research support from Bristol-Myers Squibb, Danone, Elan, Eli Lilly, Novartis, OctaPharma, Pfizer, Sanofi-Aventis, and Sonexa; has served as a consultant to and received honoraria from Accera, Astellas, Baxter, Bayer, Bristol-Myers Squibb, Eisai, GE Healthcare, Medavante, Medivation, Merck, Novartis, Pfizer, Prana Biotechnology, QR Pharma, Sanofi-Aventis, and Toyama Chemical; and has served on the speakers boards for Eisai Medical Research, Forest, Novartis, and Pfizer. Dr Somogyi is an employee of Novartis Pharmaceuticals Corporation.
Funding/support: This review was supported by Novartis Pharmaceuticals Corporation, East Hanover, New Jersey. Alpha-Plus Medical Communications Ltd (Oxford, United Kingdom) provided editorial and administrative support with the production of this manuscript, which was funded by Novartis Pharmaceuticals Corporation.
REFERENCES
1. Priano L, Gasco MR, Mauro A. Transdermal treatment options for neurological disorders: impact on the elderly. Drugs Aging. 2006;23(5):357-375. PubMed doi:10.2165/00002512-200623050-00001
2. Exelon Patch [package insert]. East Hanover, NJ: Novartis Pharmaceuticals; 2000. http://www.pharma.us.novartis.com/product/pi/pdf/exelonpatch.pdf. Accessed September 28, 2011.
3. Neupro [summary of product characteristics]. London, UK: European Medicines Agency (EMA); 2006. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000626/WC500026397.pdf. Accessed September 28, 2011.
4. EMSAM (selegiline transdermal system) [package insert]. Napa, CA: Dey, LP; 2009. http://www.emsam.com/pi_emsam.pdf. Accessed September 28, 2011.
5. Lidoderm [package insert]. Chadds Ford, PA: Endo Pharmaceuticals Inc; 2010. http://www.lidoderm.com/pdf/lidoderm_pack_insert.pdf. Accessed September 28, 2011.
6. Qutenza [package insert]. San Mateo, CA: NeurogesX, Inc; 2009. http://www.qutenza.com/_docs/qutenza_full_PI_.pdf. Accessed September 28, 2011.
7. Winblad B, Machado JC. Use of rivastigmine transdermal patch in the treatment of Alzheimer’s disease. Expert Opin Drug Deliv. 2008;5(12):1377-1386. PubMed doi:10.1517/17425240802542690
8. Ale I, Lachapelle JM, Maibach HI. Skin tolerability associated with transdermal drug delivery systems: an overview. Adv Ther. 2009;26(10):920-935. PubMed doi:10.1007/s12325-009-0075-9
9. Grossberg G, Sadowsky C, Förstl H, et al. Safety and tolerability of the rivastigmine patch: results of a 28-week open-label extension. Alzheimer Dis Assoc Disord. 2009;23(2):158-164. PubMed doi:10.1097/WAD.0b013e31818b1c2c
10. LeWitt PA, Lyons KE, Pahwa R; SP 650 Study Group. Advanced Parkinson disease treated with rotigotine transdermal system: PREFER study. Neurology. 2007;68(16):1262-1267. PubMed doi:10.1212/01.wnl.0000259516.61938.bb
11. Amsterdam JD, Bodkin JA. Selegiline transdermal system in the prevention of relapse of major depressive disorder: a 52-week, double-blind, placebo-substitution, parallel-group clinical trial. J Clin Psychopharmacol. 2006;26(6):579-586. PubMed doi:10.1097/01.jcp.0000239794.37073.70
12. Hurkmans JF, Boddé HE, Van Driel LM, et al. Skin irritation caused by transdermal drug delivery systems during long-term (5 days) application. Br J Dermatol. 1985;112(4):461-467. PubMed doi:10.1111/j.1365-2133.1985.tb02321.x
13. Jessen L, Kovalick LJ, Azzaro AJ. The selegiline transdermal system (EMSAM): a therapeutic option for the treatment of major depressive disorder. P T. 2008;33(4):212-246. PubMed
14. Culpepper L, Kovalick LJ. A review of the literature on the selegiline transdermal system: an effective and well-tolerated monoamine oxidase inhibitor for the treatment of depression. Prim Care Companion J Clin Psychiatry. 2008;10(1):25-30. PubMed doi:10.4088/PCC.v10n0105
15. Neupro. European Public Assessment Report Summary. London, UK: European Medicines Agency; 2010. http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/000626/human_med_000926.jsp&murl=menus/medicines/
medicines.jsp&jsenabled=true. Accessed September 28, 2011.
16. Bunten S, Happe S. Rotigotine transdermal system: a short review. Neuropsychiatr Dis Treat. 2006;2(4):421-426. PubMed doi:10.2147/nedt.2006.2.4.421
17. W×¼llner U, Kassubek J, Odin P, et al; NEUPOS Study Group. Transdermal rotigotine for the perioperative management of Parkinson’s disease. J Neural Transm. 2010;117(7):855-859. PubMed doi:10.1007/s00702-010-0425-4
18. Cevc G. Drug delivery across the skin. Expert Opin Investig Drugs. 1997;6(12):1887-1937. PubMed doi:10.1517/13543784.6.12.1887
19. Oertel W, Ross JS, Eggert K, et al. Rationale for transdermal drug administration in Alzheimer disease. Neurology. 2007; 69(4):S4-S9. PubMed
20. Small G, Dubois B. A review of compliance to treatment in Alzheimer’s disease: potential benefits of a transdermal patch. Curr Med Res Opin. 2007;23(11):2705-2713. PubMed doi:10.1185/030079907X233403
21. Exelon Patch [summary of product characteristics]. London, UK: European Medicines Agency (EMA); 2006. http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/000169/human_med_000777.jsp&murl=menus/medicines/
medicines.jsp&mid=WC0b01ac058001d125#. Accessed September 28, 2011.
22. Lefרvre G, SÄ™dek G, Jhee SS, et al. Pharmacokinetics and pharmacodynamics of the novel daily rivastigmine transdermal patch compared with twice-daily capsules in Alzheimer’s disease patients. Clin Pharmacol Ther. 2008;83(1):106-114. PubMed doi:10.1038/sj.clpt.6100242
23. Feldman HH, Lane R; Study 304 Group. Rivastigmine: a placebo controlled trial of twice daily and three times daily regimens in patients with Alzheimer’s disease.J Neurol Neurosurg Psychiatry. 2007;78:1056-1063. PubMed doi:10.1136/jnnp.2006.099424
24. Shua-Haim J, Smith J, Picard F, et al. Steady-state pharmacokinetics of rivastigmine in patients with mild to moderate Alzheimer’s disease not affected by co-administration of memantine: an open-label, crossover, single-centre study. Clin Drug Investig. 2008;28(6):361-374. PubMed doi:10.2165/00044011-200828060-00004
25. Mercier F, Lefרvre G, Huang HL, et al. Rivastigmine exposure provided by a transdermal patch versus capsules. Curr Med Res Opin. 2007;23(12):3199-3204. PubMed doi:10.1185/030079908X253438
26. Winblad B, Cummings J, Andreasen N, et al. A six-month double-blind, randomized, placebo-controlled study of a transdermal patch in Alzheimer’s disease: rivastigmine patch versus capsule. Int J Geriatr Psychiatry. 2007;22(5):456-467. PubMed doi:10.1002/gps.1788
27. Kurz A, Farlow M, Lefרvre G. Pharmacokinetics of a novel transdermal rivastigmine patch for the treatment of Alzheimer’s disease: a review. Int J Clin Pract. 2009;63(5):799-805. PubMed doi:10.1111/j.1742-1241.2009.02052.x
28. Olanow CW, Obeso JA, Stocchi F. Continuous dopamine-receptor treatment of Parkinson’s disease: scientific rationale and clinical implications. Lancet Neurol. 2006;5(8):677-687. PubMed doi:10.1016/S1474-4422(06)70521-X
29. Splinter MY. Rotigotine: transdermal dopamine agonist treatment of Parkinson’s disease and restless legs syndrome. Ann Pharmacother. 2007;41(2):285-295. PubMed doi:10.1345/aph.1H113
30. Giladi N, Boroojerdi B, Korczyn AD, et al; SP513 investigators. Rotigotine transdermal patch in early Parkinson’s disease: a randomized, double-blind, controlled study versus placebo and ropinirole. Mov Disord. 2007;22(16):2398-2404. PubMed doi:10.1002/mds.21741
31. Oertel WH, Benes H, Garcia-Borreguero D, et al. Rotigotine transdermal patch in moderate to severe idiopathic restless legs syndrome: a randomized, placebo-controlled polysomnographic study. Sleep Med. 2010;11(9):848-856. PubMed doi:10.1016/j.sleep.2010.02.014
32. Hening WA, Allen RP, Ondo WG, et al; SP792 Study Group. Rotigotine improves restless legs syndrome: a 6-month randomized, double-blind, placebo-controlled trial in the United States. Mov Disord. 2010;25(11):1675-1683. PubMed doi:10.1002/mds.23157
33. Högl B, Oertel WH, Stiasny-Kolster K, et al. Treatment of moderate to severe restless legs syndrome: 2-year safety and efficacy of rotigotine transdermal patch. BMC Neurol. 2010;10(1):86. PubMed doi:10.1186/1471-2377-10-86
34. Claxton AJ, Cramer J, Pierce C. A systematic review of the associations between dose regimens and medication compliance. Clin Ther. 2001;23(8):1296-1310. PubMed doi:10.1016/S0149-2918(01)80109-0
35. Imbimbo BP. Pharmacodynamic-tolerability relationships of cholinesterase inhibitors for Alzheimer’s disease. CNS Drugs. 2001;15(5):375-390. PubMed doi:10.2165/00023210-200115050-00004
36. Winblad B, Kawata AK, Beusterien KM, et al. Caregiver preference for rivastigmine patch relative to capsules for treatment of probable Alzheimer’s disease. Int J Geriatr Psychiatry. 2007;22(5):485-491. PubMed doi:10.1002/gps.1806
37. Campbell BJ, Rowbotham M, Davies PS, et al. Systemic absorption of topical lidocaine in normal volunteers, patients with post-herpetic neuralgia, and patients with acute herpes zoster. J Pharm Sci. 2002;91(5):1343-1350. PubMed doi:10.1002/jps.10133
38. Babbar S, Marier JF, Mouksassi MS, et al. Pharmacokinetic analysis of capsaicin after topical administration of a high-concentration capsaicin patch to patients with peripheral neuropathic pain. Ther Drug Monit. 2009;31(4):502-510. PubMed doi:10.1097/FTD.0b013e3181a8b200
39. Haanpää ML, Gourlay GK, Kent JL, et al. Treatment considerations for patients with neuropathic pain and other medical comorbidities. Mayo Clin Proc. 2010;85(suppl 3):S15-S25. PubMed doi:10.4065/mcp.2009.0645
40. Baron R, Mayoral V, Leijon G, et al. 5% lidocaine medicated plaster versus pregabalin in post-herpetic neuralgia and diabetic polyneuropathy: an open-label, non-inferiority two-stage RCT study. Curr Med Res Opin. 2009;25(7):1663-1676. PubMed doi:10.1185/03007990903047880
41. Webster LR, Tark M, Rauck R, et al. Effect of duration of postherpetic neuralgia on efficacy analyses in a multicenter, randomized, controlled study of NGX-4010, an 8% capsaicin patch evaluated for the treatment of postherpetic neuralgia. BMC Neurol. 2010;10(1):92. PubMed doi:10.1186/1471-2377-10-92
42. Backonja M, Wallace MS, Blonsky ER, et al; NGX-4010 C116 Study Group. NGX-4010, a high-concentration capsaicin patch, for the treatment of postherpetic neuralgia: a randomised, double-blind study. Lancet Neurol. 2008;7(12):1106-1112. PubMed doi:10.1016/S1474-4422(08)70228-X