Objective: To investigate the effect of early versus late versus no antipsychotic administration on intensive care unit (ICU) delirium.
Methods: This retrospective cohort study was conducted in 2 adult medical ICUs at a single tertiary care center in Boston, Massachusetts, from October 1, 2015, to May 31, 2016. The study included 322 patients stratified into those who first received antipsychotics < 48 hours after first positive or unscorable (due to sedation) modified Confusion Assessment Method (CAM-ICU-m) (early), > 48 hours after first positive or unscorable CAM-ICU-m (late), and never received antipsychotics. Primary outcomes were hours alive without delirium or coma and likelihood of delirium-coma resolution. Secondary outcomes included ventilator-free hours, likelihood of extubation, and 10-day mortality. In post hoc exploratory analyses, outcomes were reanalyzed excluding comatose patients.
Results: Mean ± SD delirium-coma-free hours were 63 ± 87 for patients who received antipsychotics early, 66 ± 92 for those who received antipsychotics late, and 89 ± 107 for those who never received antipsychotics (P = .71). Antipsychotic exposure did not impact delirium-coma resolution. Mean ventilator-free hours were 103 ± 87 for patients who received antipsychotics early, 90 ± 83 for those who received antipsychotics late, and 89 ± 101 for patients who never received antipsychotics (P = .11). The hazard ratio (HR) for 10-day mortality among patients who received antipsychotics early was 0.68 (95% CI, 0.37-1.22) and 0.30 (95% CI, 0.10-0.88) for those who received antipsychotics late compared to those who never received antipsychotics (P = .03). After excluding comatose patients, the effect of antipsychotics on 10-day mortality was no longer observed (early HR = 0.57, 95% CI, 0.30-1.07; late HR = 0.57, 95% CI, 0.28-1.18; never HR = 1 [reference]; P = .14).
Conclusion: Antipsychotics were not associated with changes in delirium-coma-free hours or ventilator-free hours.
A Comparison of Early, Late, and No Treatment of
Intensive Care Unit Delirium With Antipsychotics:
A Retrospective Cohort Study
ABSTRACT
Objective: To investigate the effect of early versus late versus no antipsychotic administration on intensive care unit (ICU) delirium.
Methods: This retrospective cohort study was conducted in 2 adult medical ICUs at a single tertiary care center in Boston, Massachusetts, from October 1, 2015, to May 31, 2016. The study included 322 patients stratified into those who first received antipsychotics < 48 hours after first positive or unscorable (due to sedation) modified Confusion Assessment Method (CAM-ICU-m) (early), > 48 hours after first positive or unscorable CAM-ICU-m (late), and never received antipsychotics. Primary outcomes were hours alive without delirium or coma and likelihood of delirium-coma resolution. Secondary outcomes included ventilator-free hours, likelihood of extubation, and 10-day mortality. In post hoc exploratory analyses, outcomes were reanalyzed excluding comatose patients.
Results: Mean ± SD delirium-coma–free hours were 63 ± 87 for patients who received antipsychotics early, 66 ± 92 for those who received antipsychotics late, and 89 ± 107 for those who never received antipsychotics (P = .71). Antipsychotic exposure did not impact delirium-coma resolution. Mean ventilator-free hours were 103 ± 87 for patients who received antipsychotics early, 90 ± 83 for those who received antipsychotics late, and 89 ± 101 for patients who never received antipsychotics (P = .11). The hazard ratio (HR) for 10-day mortality among patients who received antipsychotics early was 0.68 (95% CI, 0.37–1.22) and 0.30 (95% CI, 0.10–0.88) for those who received antipsychotics late compared to those who never received antipsychotics (P = .03). After excluding comatose patients, the effect of antipsychotics on 10-day mortality was no longer observed (early HR = 0.57, 95% CI, 0.30–1.07; late HR = 0.57, 95% CI, 0.28–1.18; never HR = 1 [reference]; P = .14).
Conclusion: Antipsychotics were not associated with changes in delirium-coma–free hours or ventilator-free hours.
Prim Care Companion CNS Disord 2018;20(6):18m02320
To cite: Thom RP, Bui MP, Rosner B, et al. A comparison of early, late, and no treatment of intensive care unit delirium with antipsychotics: a retrospective cohort study. Prim Care Companion CNS Disord. 2018;20(6):18m02320.
To share: https://doi.org/10.4088/PCC.18m02320
© Copyright 2018 Physicians Postgraduate Press, Inc.
aBrigham and Women’s Hospital, Boston, Massachusetts
bBeth Israel Deaconess Medical Center, Boston, Massachusetts
cDepartment of Biostatistics, Harvard School of Public Health, Boston, Massachussets
dDepartment of Population Medicine, Harvard Medical School and Harvard Pilgrim Health Care Institute, Boston, Massachusetts
*Corresponding author: Robyn P. Thom, MD, 60 Fenwood Rd, Dept. of Psychiatry, Boston, MA 02115 ([email protected]).
Delirium, the syndrome of acute brain dysfunction caused by an underlying medical illness or toxic exposure, occurs in up to 87% of intensive care unit (ICU) patients.1 According to DSM-5, delirium is characterized by the acute onset of disturbance in attention, awareness, and cognition, which fluctuates in severity throughout the day.2 ICU patients are extremely vulnerable to developing delirium.3 The 2018 pain, agitation, and delirium guidelines of the Society for Critical Care Medicine4 recommend a multicomponent, nonpharmacologic intervention focused on reducing modifiable risk factors for delirium but do not advocate routine use of specific pharmacologic agents for the treatment of delirium.
While antipsychotics are often used to treat delirium symptoms such as agitation, hallucinations, paranoia, and sleep disturbances, data on the impact of antipsychotics on ICU delirium are mixed. Three randomized, placebo-controlled studies have assessed the efficacy of antipsychotics to treat ICU delirium. One trial5 found that antipsychotic use reduced delirium duration, while the other 2 did not.6,7 A fourth randomized controlled trial8 (RCT) examined the use of antipsychotics to prevent delirium in ICU patients. This trial8 reported that prophylaxis with haloperidol reduced the incidence of delirium, suggesting that timing of antipsychotic intervention may be an important factor for efficacy. Little is known, however, about the effect of the timing of antipsychotic exposure on ICU delirium treatment outcomes.
The aim of this study was to compare outcomes of critically ill delirious patients treated with early antipsychotics (administered within 48 hours of altered mental status), late antipsychotics (administered after more than 48 hours of altered mental status), or no antipsychotics. The primary outcomes of interest were delirium-coma–free hours and likelihood of delirium-coma resolution within a predefined 10-day surveillance period. Secondary outcomes included ventilator-free hours, likelihood of extubation during the predefined 10-day surveillance period, total amount of antipsychotic administered, likelihood of discharge on an antipsychotic, and 10-day mortality.
METHODS
Design and Setting
This was a retrospective cohort study in 2 adult medical ICUs at a single tertiary care center in Boston, Massachusetts, from October 1, 2015, to May 31, 2016. The research ethics board approved the study. Patient care decisions were at the discretion of the ICU teams. One of the 2 ICUs had an embedded psychiatrist who rounded each weekday with the ICU teams and provided consultation wherever necessary, addressing issues such as delirium, substance withdrawal, mood and anxiety symptoms, and capacity evaluations. Hospital protocol requires nurses to document modified Confusion Assessment Method9 (CAM-ICU-m) scores in the electronic health record at least once every 8 hours. The modified version used at this facility includes all components of the CAM-ICU-m; however, data are gathered in a different order.
Participants
All patients admitted during the study period were screened for inclusion. The inclusion criteria were at least 1 positive CAM-ICU-m score during the ICU stay. Exclusion criteria included antipsychotic use prior to hospital admission, alcohol or substance withdrawal, missing CAM-ICU-m data (recorded less than every 8 hours), or developmental delay. No other inclusion or exclusion criteria were applied.
Measurements and Outcomes
Data were collected retrospectively from the electronic health record. The following data were collected: baseline demographics, discharge diagnosis, mechanical ventilation status, presence of an embedded psychiatrist, CAM-ICU-m score, maximum absolute value of the Richmond Agitation-Sedation Scale10 (RASS) score in the first 24 hours of altered mental status, amount of antipsychotic administered, benzodiazepine exposure, continuation of antipsychotic at discharge, and adverse events including death, extrapyramidal symptoms, and torsades de pointes. Acute Physiology and Chronic Health Evaluation11 (APACHE II) scores were calculated using electronic health record data. RASS scores were categorized as 0, positive (> 0), negative (−1, −2, or −3), or coma (−4 or −5).
The follow-up period was defined as 240 hours (10 days × 24 hours) after the first non-negative CAM-ICU-m score (either unable to assess due to deep sedation or positive). The decision to group delirium and coma was modeled after the methodologies of the HOPE-ICU7 and MIND6 studies, the only 2 RCTs that have prospectively studied the effect of antipsychotics on the treatment of ICU delirium in mechanically ventilated and comatose patients. By grouping delirium and coma, the impact of antipsychotics on achieving “normal” brain function once altered mental status occurs can be assessed. Additionally, coma is an independent risk factor for subsequent delirium wherein > 50% of comatose patients develop subsequent delirium.12 Thus, grouping delirium and coma at the initial time of surveillance allows us to assess the effect of very early antipsychotic intervention on a population at high risk for developing delirium. In post hoc exploratory analyses, we excluded comatose patients (RASS score: −4 or −5) and subsequently compared all outcomes within the remaining noncomatose group.
In our health care center, nurses continue to regularly document CAM-ICU-m scores in the electronic health record after patients leave the ICU to receive ongoing care on medical wards. Delirium-coma resolution was defined as a minimum of 48 hours of negative CAM-ICU-m scores. Early antipsychotic administration was defined as administration of an antipsychotic medication (haloperidol, risperidone, quetiapine, olanzapine, aripiprazole, or ziprasidone) within 48 hours of the first non-negative CAM-ICU-m score. Late antipsychotic administration was defined as administration of an antipsychotic medication beginning 48 to 240 hours after the first non-negative CAM-ICU-m score.
The primary outcome, delirium-coma–free hours (number of hours alive without delirium or coma), was selected to determine the effect of antipsychotics on “normal” brain function. This outcome was calculated on the basis of the date and time of the first non-negative CAM-ICU-m and last non-negative CAM-ICU-m score prior to delirium resolution. Delirium resolution was defined as a minimum of 48 hours of negative CAM-ICU-m scores. Delirium-coma–free hours (number of hours alive without delirium or coma) was calculated by subtracting the duration of delirium from 240 hours. If the patient died or was discharged in a delirious state during the observation period, they were considered to have zero delirium-coma–free hours. If a patient was discharged alive < 10 days after delirium onset but delirium resolved prior to discharge, the patient was presumed to have remained free of delirium for the remainder of the 240-hour observation period. Therefore, patients who had persistent delirium at day 10, died delirious, or were discharged delirious were considered to have zero delirium-coma–free hours. Ventilator-free hours alive were determined by subtracting the total time on mechanical ventilation from 240 hours. Total antipsychotic received over the follow-up period is reported in milligrams of chlorpromazine equivalents.13,14
Statistical Analysis
Descriptive statistics were used to compare the baseline demographic data and clinical variables among the early, late, and no antipsychotic groups. Nominal data were compared using χ2 methods, and continuous data were compared using analysis of variance (ANOVA) or nonparametric ANOVA.
We compared delirium-coma–free hours and ventilator-free hours among patients who received early antipsychotics, late antipsychotics, and no antipsychotics using a linear regression model. Delirium-coma–free hours and ventilator-free hours were transformed using a log-scale. The model included covariates for age, sex, race, APACHE II score, RASS category (0, positive, negative, or coma), discharge diagnosis (sepsis, respiratory, cardiovascular and diabetes, gastrointestinal, malignancy, or other), benzodiazepine exposure, and presence of an embedded psychiatrist. For analysis purposes, patients with zero delirium-coma–free hours or ventilator-free hours were set to 1 hour.
We further compared likelihood of delirium-coma resolution, likelihood of extubation, and likelihood of death among patients who received early antipsychotics, late antipsychotics, and no antipsychotics using Cox proportional hazards models to account for censoring due to death or discharge prior to the end of the 10-day surveillance period. Antipsychotic exposure was treated as a time-dependent variable. We ran this analysis with and without adjusting for the same covariates as in the primary regression model.
A P value < .05 was considered statistically significant. All statistical analyses were performed using SAS version 9.4.
RESULTS
We screened 889 patients for inclusion, of whom 625 patients had at least 1 positive CAM-ICU-m score and 322 met full inclusion criteria. A total of 303 patients were excluded due to missing CAM-ICU-m data (n = 198), receipt of antipsychotics prior to hospital admission (n = 56), alcohol or substance withdrawal (n = 47), or developmental delay (n = 2) (Figure 1).
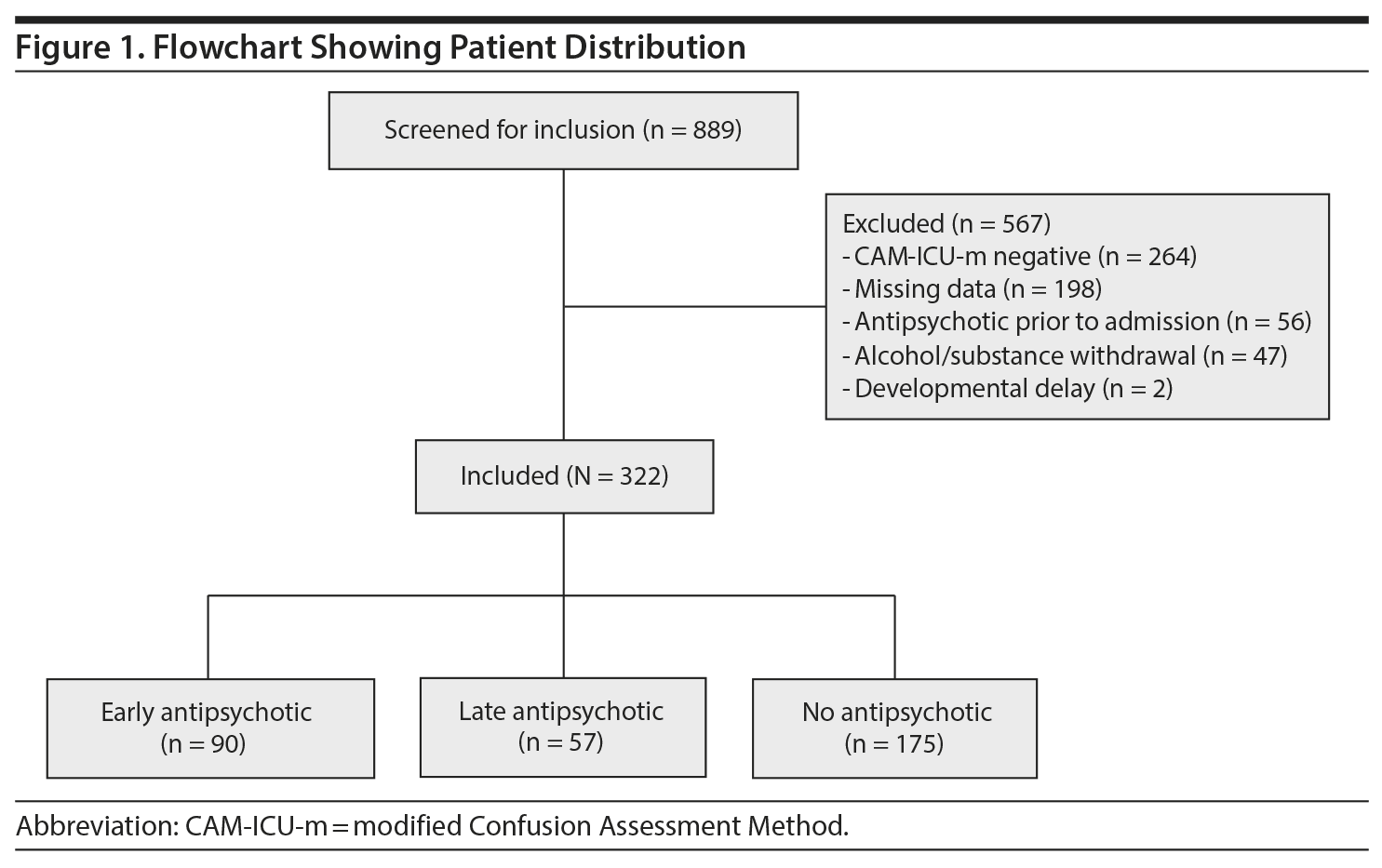
Baseline Demographics and Clinical Variables
Baseline characteristics for patients who were administered antipsychotics early (within 48 hours of first non-negative CAM-ICU-m score), late (after 48 hours of first non-negative CAM-ICU-M score), or never are summarized in Table 1. Age, sex, and APACHE II scores were not associated with antipsychotic administration. Patients of nonwhite race were less likely to receive antipsychotics. Comatose patients were less likely to receive antipsychotics overall, and if antipsychotics were administered, they were more likely to be administered late. Patients with RASS scores > 0 were more likely to receive antipsychotics early. Patients who received mechanical ventilation were more likely to receive antipsychotics than non–mechanically ventilated patients. The category of discharge diagnosis most highly associated with antipsychotic administration was a respiratory diagnosis. The presence of an embedded psychiatrist in one of the ICUs did not affect the overall percentage of patients to whom antipsychotics were administered or the frequency of early versus late antipsychotic administration.
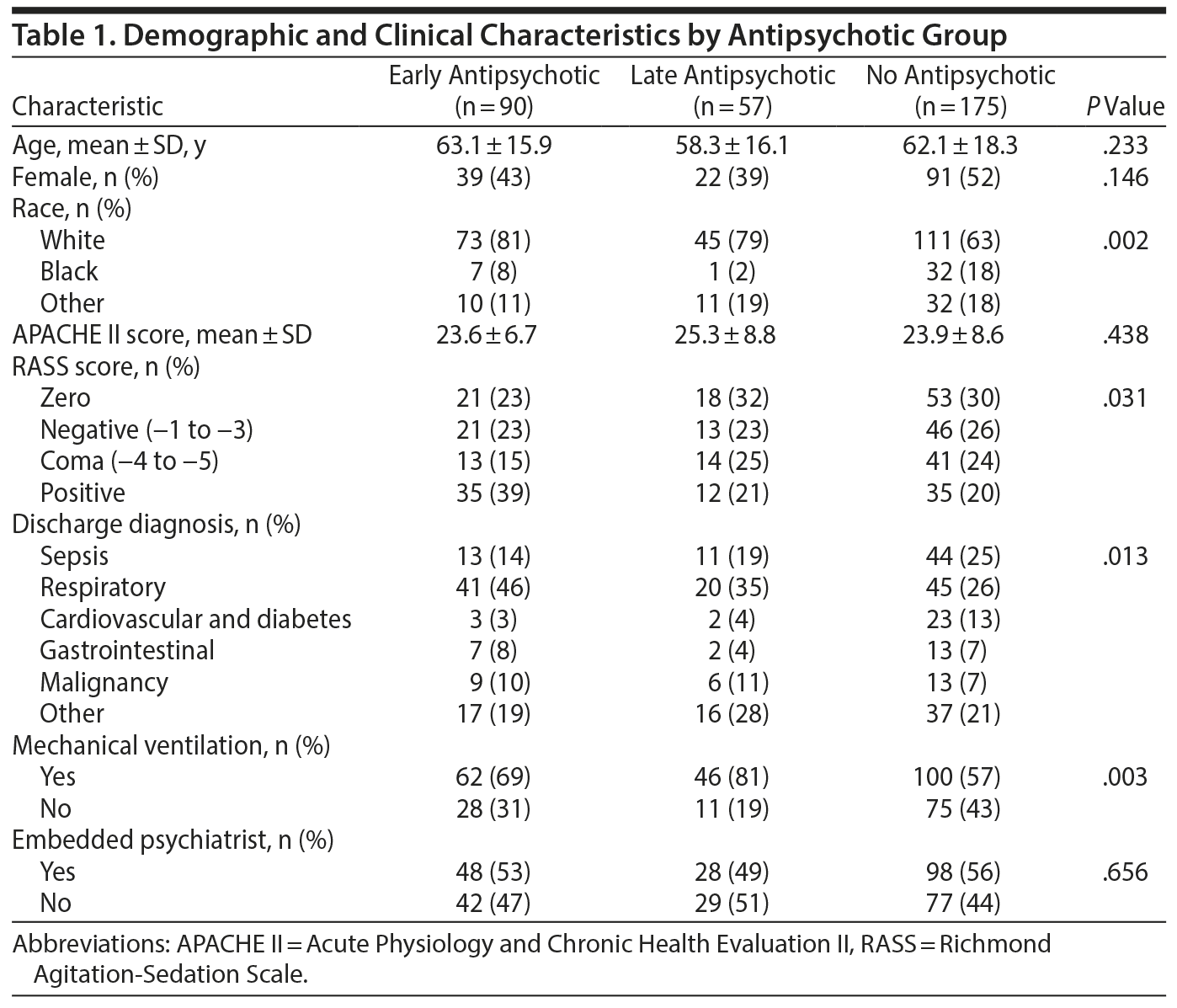
Primary Outcomes:
Delirium-Coma–Free Hours and Delirium Resolution
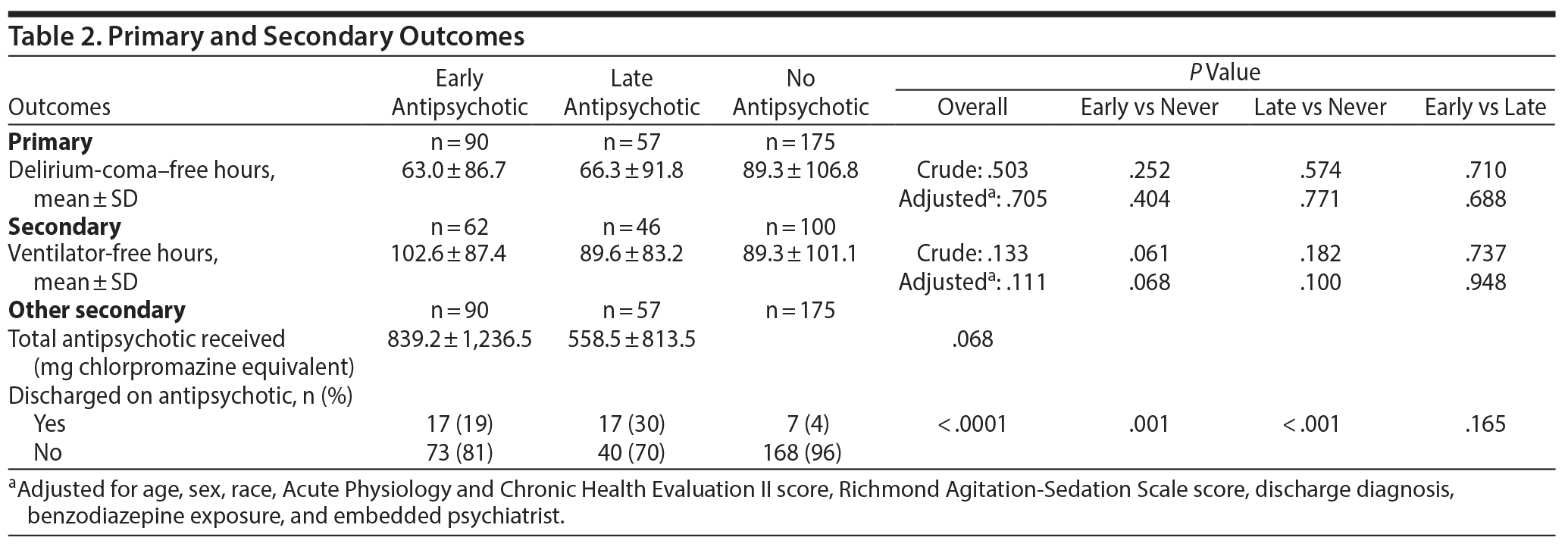
Neither early nor late antipsychotic exposure significantly increased delirium-coma–free hours (Table 2). The mean ± SD delirium-coma–free hours were 63.0 ± 86.7 hours (early) versus 66.3 ± 91.8 (late) versus 89.3 ± 106.8 (never), P = .50. After adjusting for age, sex, race, APACHE II, RASS category (positive, negative, zero, coma), discharge diagnosis, benzodiazepine exposure, and the presence of an embedded psychiatrist, the null effect of antipsychotic exposure on delirium-coma–free hours remained unchanged (P = .71).
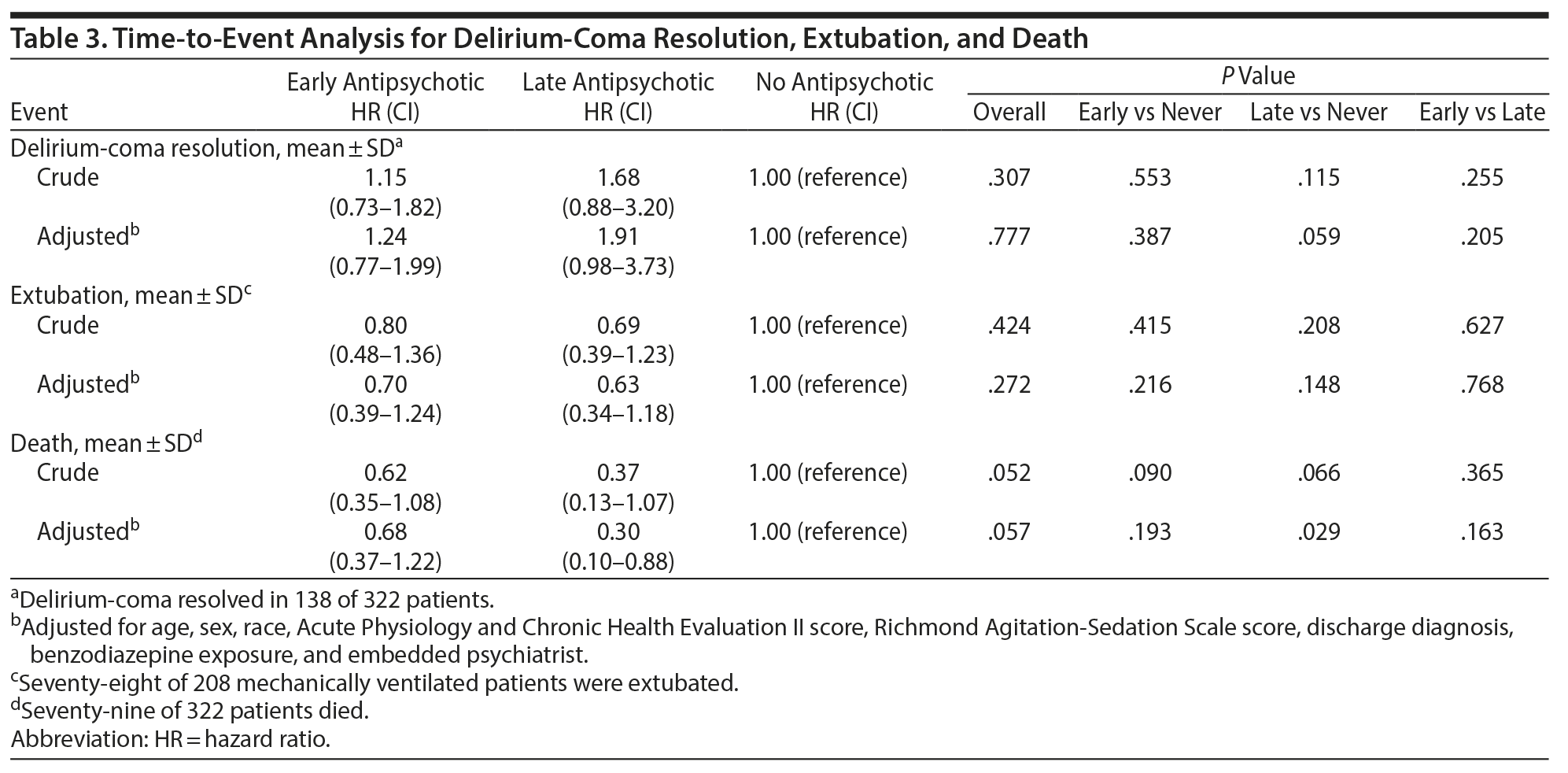
Likelihood of delirium-coma resolution within the 10-day follow-up period was determined using a survival analysis based on antipsychotic exposure as a time-dependent variable (Table 3). Antipsychotic exposure did not increase the likelihood of delirium-coma resolution. The hazard ratios (HRs) for delirium-coma resolution after adjusting for the same covariates as above were early HR = 1.24, 95% CI, 0.77–1.99; late HR = 1.91, 95% CI, 0.98–3.73; and never HR = 1 (reference); P = .78.
Secondary Outcomes:
Ventilator-Free Hours and Likelihood of Extubation
Antipsychotic exposure was not associated with an increase in ventilator-free hours (Table 2). The mean ± SD ventilator-free hours were 102.6 ± 87.4 hours (early) versus 89.6 ± 83.2 hours (late) versus 89.3 ± 101.1 hours (never) (P = .11, adjusted for age, sex, race, APACHE II, RASS category [positive, negative, zero, coma], discharge diagnosis, benzodiazepine exposure, and the presence of an embedded psychiatrist).
Likelihood of extubation was determined using a survival analysis based on antipsychotic exposure (Table 3). Antipsychotic exposure did not increase the likelihood of extubation. The HRs for likelihood of extubation after adjustment for the same covariates as above were early HR = 0.70, 95% CI, 0.39–1.24; late HR = 0.63, 95% CI, 0.34–1.18; and never HR = 1 (reference); P = .27.
Secondary Outcomes:
Antipsychotic Dosage, Discharge on an Antipsychotic, Antipsychotic-Associated Adverse Events, and Death
The timing of antipsychotic administration (early versus late) did not significantly affect the mean total dose of antipsychotic administered in the first 10 days following delirium onset, nor did early versus late administration of antipsychotics affect the likelihood of being discharged on an antipsychotic (Table 2).
In further exploratory analysis, we examined whether administration of antipsychotics was associated with harm including extrapyramidal symptoms, torsades de pointes, and mortality. Only 1 case of extrapyramidal symptoms was observed, which was in the late antipsychotic group. No cases of torsades de pointes or neuroleptic malignant syndrome were observed. Late antipsychotic administration (HR = 0.30, 95% CI, 0.10–0.88), but not early antipsychotic administration (HR = 0.68, 95% CI, 0.37–1.22), was associated with a statistically significant reduction in mortality compared with no antipsychotic administration (P = .03) after adjusting for age, sex, race, APACHE II, RASS category (positive, negative, zero, coma), discharge diagnosis, benzodiazepine exposure, and presence of an embedded psychiatrist (Table 3).
Subgroup Analyses:
Outcomes Excluding Comatose Patients
In post hoc exploratory analyses, we compared all outcomes among the noncomatose subgroup of patients (Tables 4 and 5). Within this subgroup, antipsychotic administration did not affect mean delirium-coma–free hours or ventilator-free hours. The mean ± SD delirium-coma–free hours in the early antipsychotic group was 65.4 ± 88.5 hours versus 76.8 ± 95.0 hours in the late antipsychotic group versus 104.9 ± 110.5 hours in the no antipsychotic group (P = .488 after adjusting for the same covariates as mentioned previously). The mean ventilator-free hours were 105.6 ± 89.8 (early) versus 101.7 ± 79.2 (late) versus 102.9 ± 104.8 (no antipsychotic) (adjusted P = .122).
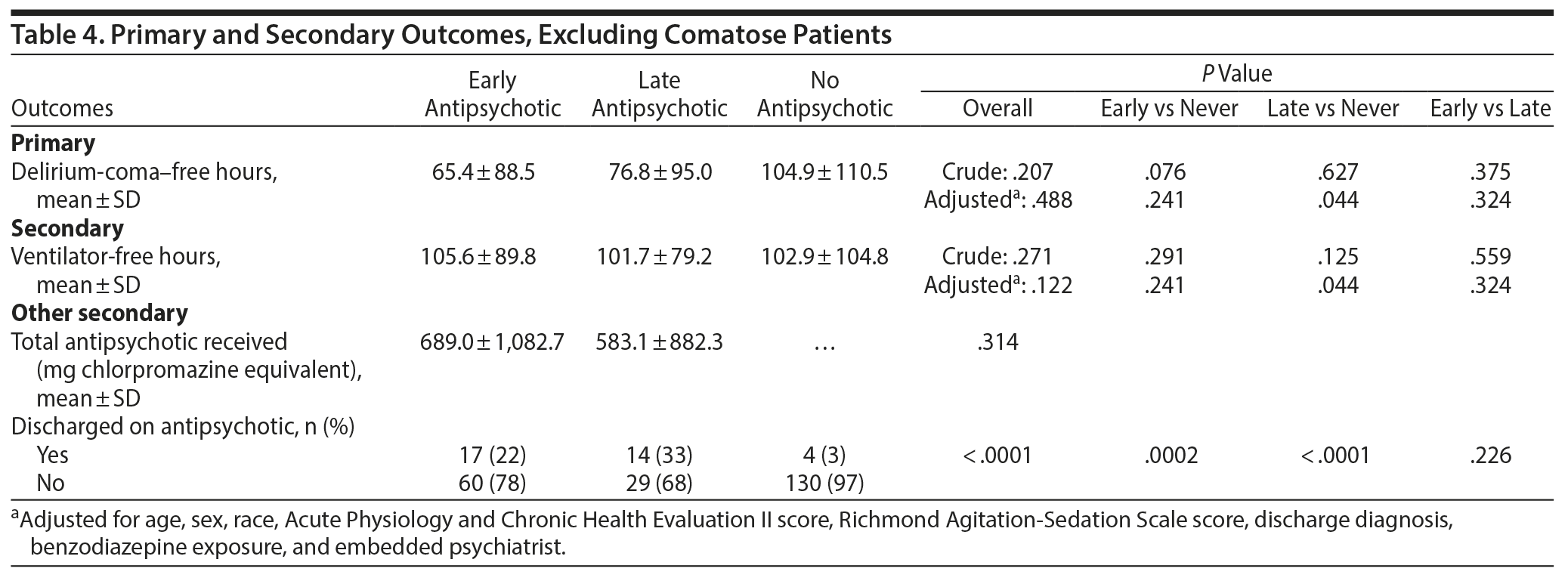
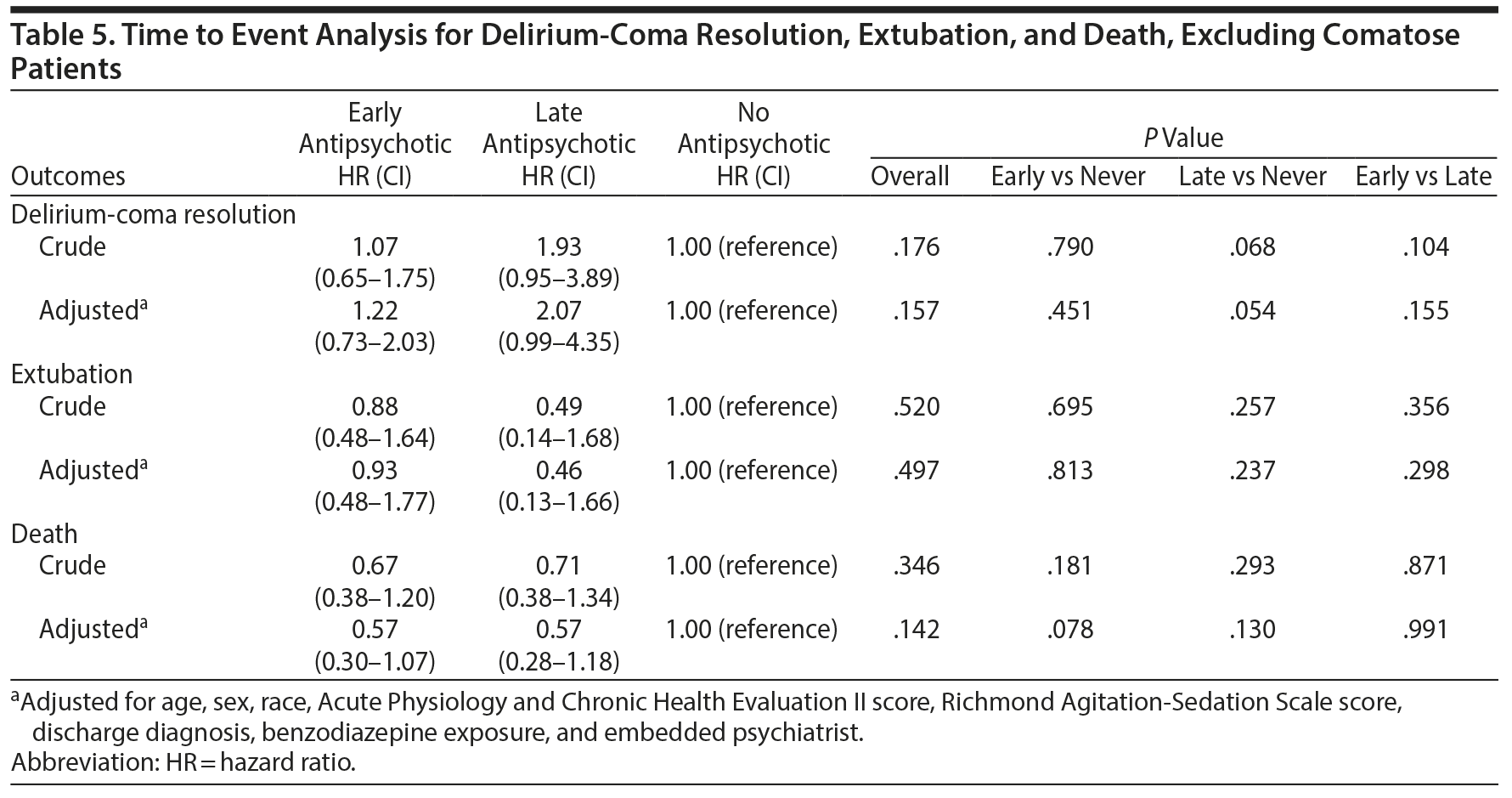
Antipsychotic administration was not observed to modify the likelihood of delirium resolution or extubation. The HRs for delirium-coma resolution after adjusting for the same covariates as mentioned previously were early HR = 1.22, 95% CI, 0.73–2.03; late HR = 2.07, 95% CI, 0.99–4.35; and never HR = 1 (reference); P = .157. The adjusted HRs for likelihood of extubation were early HR = 0.93, 95% CI, 0.48–1.77; late HR = 0.46, 95% CI, 0.13–1.66; and never HR = 1 (reference); P = .497.
Finally, neither early nor late antipsychotic administration among noncomatose patients was associated with a change in mortality. The adjusted HRs for likelihood of 10-day mortality were early HR = 0.57, 95% CI, 0.30–1.07; late HR = 0.57, 95% CI, 0.28–1.18; and never HR = 1 (reference); P = .142.
DISCUSSION
This retrospective analysis of antipsychotic management of critically ill patients with delirium found no association between antipsychotic use and duration of delirium or mechanical ventilation. Antipsychotic-related adverse events were minimal, with only 1 observed case of extrapyramidal symptoms and no episodes of torsades de pointes. Finally, we observed an association between late antipsychotic exposure and reduced 10-day mortality. When comatose patients were excluded, this effect was no longer observed, although power was limited.
Our findings that antipsychotic use did not increase delirium-coma–free hours, likelihood of delirium-coma resolution, ventilator-free hours, or extubation mirror many of the results of RCTs, including the MIND6 and HOPE-ICU7 studies. The MIND study6 was an RCT (N = 100) in which mechanically ventilated patients with altered mental status were randomized to haloperidol, ziprasidone, or placebo for the treatment of critical care delirium. Results showed that the 3 groups had similar delirium-coma–free days, ventilator-free days, and hospital length of stay.6 Similar results were reported in the HOPE study,7 which randomized 141 mechanically ventilated patients to either standing haloperidol or placebo irrespective of coma or CAM-ICU status. In contrast, a small study performed by Devlin and colleagues5 found that quetiapine was associated with reduced duration of delirium in critically ill patients. An explanation for this discrepancy may be that, unlike the other 2 RCTs6,7 and our study, Devlin’s study5 excluded comatose patients. The findings of Devlin et al5 are consistent with previous general hospital and surgical delirium studies,15,16 suggesting that antipsychotics may reduce delirium duration in less ill patients.
A recent retrospective study by Weaver and colleagues17 indicated that antipsychotic use in delirious critically ill patients was associated with a longer duration of delirium compared with no antipsychotic exposure. However, Weaver and colleagues17 included discharge from the ICU within their definition of delirium resolution. Given the retrospective nature of their study, it is possible that patients who were less ill, allowing for earlier discharge from the ICU, were less likely to receive antipsychotics, which represents a significant confounder. In our study, patients continued to be assessed with CAM-ICU-m scoring after ICU discharge, allowing us to more accurately determine when the episode of delirium resolved.
We found that neither early nor late exposure to antipsychotics impacted time to extubation. These findings contrast those of a retrospective study that showed early treatment of ICU delirium resulted in less time to extubation.18 In this prior study,18 however, delirium treatment was defined as the use of either antipsychotics or dexmedetomidine, making it difficult to draw conclusions regarding the efficacy and timing of antipsychotics in particular.
Our finding that antipsychotic use was associated with a reduced 10-day risk of death in the full cohort is intriguing and in contrast to 3 RCTs (MIND trial,6 HOPE-ICU trial,7 and REDUCE trial16), which demonstrated that randomization to an antipsychotic was not associated with a significant difference in mortality compared to placebo. However, after excluding comatose patients in a subgroup analysis, we no longer observed a protective effect of antipsychotics on mortality. A possible explanation for this discrepancy between the full cohort and the subgroup may be related to survivor bias, in that comatose patients who received late antipsychotics had to have survived the initial 48 hours after diagnosis of coma. A major limitation of our study is the retrospective, observational design, limiting our ability to discern which factors led to antipsychotic use. It is possible that antipsychotics were administered to less ill patients, which could account for the lower mortality rates observed in this group and was supported by the post hoc subgroup analysis.
While we observed that antipsychotics did not increase mortality, antipsychotic use in a medically vulnerable population is not without risk. A retrospective case-control study20 of outpatient veterans with dementia reported that haloperidol exposure was associated with an increased mortality risk of 3.8%, with a number needed to harm of 26. It is difficult, however, to compare the adverse effects seen in chronic outpatient antipsychotic use with the time-limited, carefully monitored use of antipsychotics in hospitalized delirious patients.21 This argument is supported by a systematic review by Hirota and Kishi22 that found no difference in the incidence of death, extrapyramidal symptoms, akathisia, or QT prolongation between medically hospitalized delirious patients treated with and without antipsychotics. In the current study, only 1 case of extrapyramidal symptoms was observed (akathisia), and no cases of torsades de pointes or neuroleptic malignant syndrome were observed. It should be noted that it is not our hospital’s practice to regularly monitor patients for extrapyramidal symptoms with a validated screening tool. This monitoring allows for the possibility of underdiagnosis and underdocumentation in the medical record. Studies have shown that many patients who are started on antipsychotics for hospital-acquired delirium remain on antipsychotics even after discharge. We found no association between timing of antipsychotic initiation in the ICU and likelihood of discharge on an antipsychotic.
This study has several important limitations. First, this was a retrospective observational analysis, and, thus, there may have been important reasons influencing the decision to administer and when to administer antipsychotics to different patients, including depth of sedation and overall clinical prognosis. This allows for the possibility of residual confounding between groups despite adjusting for multiple potential confounders. Second, we were unable to control for all anti- or prodeliriogenic medications that patients received. We controlled for benzodiazepines but did not control for opioids, other sedatives, or dexmedetomidine. Dexmedetomidine was not widely used for greater than 24 hours at our center during the study period. Third, our study did not distinguish between delirium and coma at the outset. It is possible that antipsychotics do not have a uniform effect on these 2 states. Finally, antipsychotic exposure was not protocolized. Therefore, our study does not account for the dosage, frequency, or kind of antipsychotics administered. While there are significant differences among antipsychotic agents in terms of side effects, the choice to group antipsychotics was made to gain a better understanding of whether there are important class effects of antipsychotics on the treatment of delirium. Finally, there may be significant heterogeneity within the treatment groups as defined as early versus late. Future research may attempt to further investigate whether very early (< 48 hour) administration of antipsychotics is of greater benefit.
CONCLUSIONS
In conclusion, we found no association between antipsychotic administration and the resolution of altered mental status or likelihood of extubation. Intriguingly, antipsychotic use among the full cohort was associated with reduced 10-day mortality; this effect was no longer observed when comatose patients were excluded.
Submitted: April 26, 2018; accepted August 28, 2018.
Published online: November 15, 2018.
Potential conflicts of interest: None.
Funding/support: This work was supported by an internal hospital innovation grant, the Brigham Care Redesign Incubator and Startup Program (BCRISP).
Role of the sponsor: The sponsor had no role in the design and conduct of the study; collection, management, analysis, and interpretation of data; or preparation, review, or approval of the manuscript.
Acknowledgments: The authors acknowledge David Wolfe, MD, Brigham and Women’s Hospital, Boston, Massachusetts, for reviewing this manuscript. Dr Wolfe has no conflicts of interest related to the subject of this article.
Previous presentation: This work was presented at the Academy of Psychosomatic Medicine Annual Meeting; November 8–11, 2017; Palm Springs, California.
REFERENCES
1. Gilchrist NA, Asoh I, Greenberg B. Atypical antipsychotics for the treatment of ICU delirium. J Intensive Care Med. 2012;27(6):354–361. PubMed CrossRef
2. American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders. 5th ed. Washington, DC: American Psychiatric Association; 2013.
3. Maldonado JR. Acute brain failure: pathophysiology, diagnosis, management, and sequelae of delirium. Crit Care Clin. 2017;33(3):461–519. PubMed CrossRef
4. Devlin JW, Skrobik Y, Gélinas C, et al. Clinical practice guidelines for the prevention and management of pain, agitation/sedation, delirium, immobility, and sleep disruption in adult patients in the ICU. Crit Care Med. 2018;46(9):e825–e873. PubMed CrossRef
5. CrossRef Devlin JW, Roberts RJ, Fong JJ, et al. Efficacy and safety of quetiapine in critically ill patients with delirium: a prospective, multicenter, randomized, double-blind, placebo-controlled pilot study. Crit Care Med. 2010;38(2):419–427. PubMed
6. Girard TD, Pandharipande PP, Carson SS, et al; MIND Trial Investigators. Feasibility, efficacy, and safety of antipsychotics for intensive care unit delirium: the MIND randomized, placebo-controlled trial. Crit Care Med. 2010;38(2):428–437. PubMed CrosssRef
7. Page VJ, Ely EW, Gates S, et al. Effect of intravenous haloperidol on the duration of delirium and coma in critically ill patients (Hope-ICU): a randomised, double-blind, placebo-controlled trial. Lancet Respir Med. 2013;1(7):515–523. PubMed CrossRef
8. van den Boogaard M, Schoonhoven L, van Achterberg T, et al. Haloperidol prophylaxis in critically ill patients with a high risk for delirium. Crit Care. 2013;17(1):R9. PubMed CrossRef
9. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the Confusion Assessment Method. A new method for detection of delirium. Ann Intern Med. 1990;113(12):941–948. PubMed CrossRef
10. Sessler CN, Gosnell MS, Grap MJ, et al. The Richmond Agitation-Sedation Scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166(10):1338–1344. PubMed CrossRef
11. Knaus WA, Draper EA, Wagner DP, et al. APACHE II: a severity of disease classification system. Crit Care Med. 1985;13(10):818–829. PubMed CrossRef
12. Skrobik Y, Dumont M, Ouimet S. Risk factors, coma, and consequences of intensive care unit delirium. Crit Care. 2005;9(suppl 1):138. CrossRef
13. Rose VL. APA practice guideline for the treatment of patients with schizophrenia. Am Fam Physician. 1997;56(4):1217–1220. PubMed
14. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003;64(6):663–667. PubMed CrossRef
15. Hu H, Deng W, Yang H. A prospective random control study comparison of olanzapine and haloperidol in senile delirium. Chong-qing Med J. 2004;8:1234–1237.
16. Kalisvaart KJ, de Jonghe JFM, Bogaards MJ, et al. Haloperidol prophylaxis for elderly hip-surgery patients at risk for delirium: a randomized placebo-controlled study. J Am Geriatr Soc. 2005;53(10):1658–1666. PubMed CrossRef
17. Weaver CB, Kane-Gill SL, Gunn SR, et al. A retrospective analysis of the effectiveness of antipsychotics in the treatment of ICU delirium. J Crit Care. 2017;41:234–239. PubMed CrossRef
18. Michaud CJ, Thomas WL, McAllen KJ. Early pharmacological treatment of delirium may reduce physical restraint use: a retrospective study. Ann Pharmacother. 2014;48(3):328–334. PubMed CrossRef
19. van den Boogaard M, Slooter AJC, Brüggemann RJM, et al; REDUCE Study Investigators. Effect of haloperidol on survival among critically ill adults with a high risk of delirium: the REDUCE randomized clinical trial. JAMA. 2018;319(7):680–690. PubMed CrossRef
20. Maust DT, Kim HM, Seyfried LS, et al. Antipsychotics, other psychotropics, and the risk of death in patients with dementia: number needed to harm. JAMA Psychiatry. 2015;72(5):438–445. PubMed CrossRef
21. Thom RP, Mock CK, Teslyar P. Delirium in hospitalized patients: risks and benefits of antipsychotics. Cleve Clin J Med. 2017;84(8):616–622. PubMed CrossRef
22. Hirota T, Kishi T. Prophylactic antipsychotic use for postoperative delirium: a systematic review and meta-analysis. J Clin Psychiatry. 2013;74(12):e1136–e1144. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!


