The Association Between Alzheimer’s Disease Symptom Severity and Caregiver Outcomes: A Cross-Sectional Study
Objective: To determine the association between Alzheimer’s disease (AD) symptom severity and caregiver outcomes.
Method: This was a database analysis of the Alzheimer’s Disease Caregiver Study, a cross-sectional, caregiver-reported study conducted in 2007. Data were collected nationwide via the Internet and in 8 cities: Detroit, Michigan; Knoxville, Tennessee; Los Angeles, California; Miami, Florida; Philadelphia, Pennsylvania; Phoenix, Arizona; St Louis, Missouri; and Washington, DC. Participants were unpaid adult caregivers of AD patients (N = 1,077). Symptom severity was measured using the Revised Memory and Behavioral Problem Checklist (RMBPC). Caregiver outcomes included the Caregiver Burden Scale, diagnosis of anxiety and depression, use of the emergency room, hospitalization, number of physician visits, and missed workdays in the past 6 months. Linear and logistic regression models were developed to assess effects of AD symptom severity on outcomes. Covariates included caregiver and patient characteristics and interactions of AD symptom severity with covariates based on previous analyses.
Results: Of the 1,077 respondents, 1,034 had valid RMBPC overall symptom severity scores. AD symptom severity was a significant (P < .01) predictor of all caregiver outcomes except physician visits. Each unit increase in RMBPC severity score corresponded with an increase of 0.328 (95% CI, 0.101−0.554) units in caregiver burden. Each unit increase in severity resulted in increases in physician visits (b = 0.343; 95% CI, 0.052−0.635) and absenteeism (b = 1.722; 95% CI, 0.694−2.749). For each unit increase in RMBPC severity score, caregivers had greater likelihood of emergency room use (odds ratio = 1.506; 95% CI, 1.230−1.845), hospitalization (OR = 1.393; 95% CI, 1.091−1.777), anxiety (OR = 1.506; 95% CI, 1.257−1.805), and depression (OR = 1.811; 95% CI, 1.505−2.179).
Conclusions: AD symptom severity is significantly associated with poorer caregiver outcomes. Therefore, treatments that slow AD symptom progression may be beneficial to caregiver outcomes.
Prim Care Companion CNS Disord 2011;13(3):e1-e8
© Copyright 2011 Physicians Postgraduate Press, Inc.
Submitted: June 20, 2010; accepted January 5, 2011.
Published online: June 23, 2011 (doi:10.4088/PCC.10m01043).
Corresponding author: Charles D. Petrie, PhD, Outcomes Research, Clinical Development and Medical Affairs, Specialty Care Business Unit, Pfizer Global Development Headquarters, MS 6025-B2248, 50 Pequot Ave, New London, CT 06320 ([email protected]).
Dementia is a term that describes disorders that cause cognitive decline. The most common type of dementia is Alzheimer’s disease (AD).1 It is currently estimated that 5.3 million Americans have AD, and future projections estimate that, due to an increase in the aging population, there will be between 11 million and 16 million Americans with AD by 2050.1 Due in large part to the increasing size of the AD patient population, AD costs have risen in past decades and are expected to continue to rise.1 In 1991, the economic burden of caring for patients with AD per year was estimated to be $20.6 billion.2 In 2005, direct costs of AD to Medicare and Medicaid were $111 billion and indirect costs of caregiver lost productivity to employers were $36.5 billion. Additional costs were covered through the US Department of Veteran’s Affairs, private medical and long-term care insurers, and out-of-pocket payments by patients and their families.1
The economic and humanistic impact of AD on patients worsens with increasing disease severity. AD patients who were more dependent on others with respect to activities of daily living (ADLs) were found to have decreased quality of life.3 Patients with lower levels of physical and instrumental functioning also used the hospital and physicians more often than those with higher levels of physical and instrumental functioning.4 Across numerous studies, it has been consistently demonstrated that cognitive decline, behavioral disturbances, and depression associated with AD are strong predictors of nursing home admission.5
Clinical Points
- Alzheimer’s disease (AD) symptom severity is significantly associated with poorer caregiver outcomes.
- Poorer caregiver outcomes include greater degree of caregiver burden and greater likelihood of experiencing depression or anxiety, visiting the emergency room, hospitalization, and lost work productivity.
- Available treatments that slow AD symptom progression may positively impact caregiver outcomes and their associated costs.
The negative impact of AD is not limited to patients alone. Caregivers also experience negative consequences, especially as the condition progresses. The concept of caregiver burden among those who care for patients with AD in the United States has been well established for a number of years.1,6 The patient’s level of cognitive decline, extent of difficulty in performing ADLs, personality changes, and psychiatric symptoms have all been noted to increase caregiver burden.6,7 An association between decreased quality of life and increased caregiver burden has also been noted.8 More recent studies have found similar results among caregivers in other countries. A study of caregivers in Italy demonstrated a correlation between severity of behavioral disorders and caregiver quality of life, depression, and anxiety.9 A study of caregivers in Japan found that burden was associated with specific AD symptoms.10
The relationship of AD symptom severity with caregiver resource use and lost productivity has been less well established. A 2002 study by Toseland and colleagues11 showed that those who provide care for patients with lower levels of daily functioning (as measured by ADL scores) used more health services. A more recent study demonstrated greater rates of emergency room (ER) use and hospitalization among caregivers of patients with more severe behavioral and psychological symptoms.12 The effect on caregivers also has broader societal implications, with lost work productivity and early exit from the labor force due to caregiving.1 Only a single study, however, has demonstrated greater work loss among caregivers of more severe AD patients than among caregivers of less severe AD patients.13
In the past decade, the composition of the population of AD caregivers in the United States has changed. In the later 1990s, approximately 42%-48% of caregivers were adult children caring for a parent or parent-in-law with AD.14,15 A 2004 study reported that this proportion had risen to 57%.1 The relationship of AD symptom severity with caregiver burden, anxiety, depression, and work productivity loss has not been recently evaluated in the United States, and only a single study has recently evaluated the relationship of AD symptom severity with caregiver health care resource use.12 Due to the changing composition of American caregivers, it is important to reevaluate the effects of AD symptom severity on current caregivers.
The objective of this study is to understand the factors associated with self-reported outcomes of adults caring for a person with AD. We placed specific focus on quantifying the effects of AD symptom severity on caregiver outcomes, including caregiver burden, psychiatric conditions, health care resource use, and lost work productivity, so as to better understand the impact of AD progression on caregivers.
METHOD
Study Design
We obtained data from the Alzheimer’s Disease Caregiver Study (Kantar Health, formerly Consumer Health Sciences, Princeton, New Jersey) conducted in January and February 2007. The sample consisted of 1,077 unpaid adult (aged ≥ 18 years) caregivers of AD patients recruited to the study through 2 sources: a consumer Web-based panel and centralized locations in the following cities: Detroit, Michigan; Knoxville, Tennessee; Los Angeles, California; Miami, Florida; Philadelphia, Pennsylvania; Phoenix, Arizona; St Louis, Missouri; and Washington, DC. We collected 75 interviews via the centralized locations and 1,002 interviews via the Web-based panel.
The recruitment through centralized locations focused on recruiting older, spousal caregivers who may not have Internet access and may therefore be underrepresented in the Web-based arm of the study. Data were collected via self-administered questionnaires, and respondents were screened to ensure that they were currently caring for a spouse with AD and were not a paid caregiver. The recruitment via the Internet focused on respondents who completed the National Health and Wellness Survey, an Internet-based study fielded by Kantar Health annually, and indicated in the study that they were caregivers of adults with AD and also from the Lightspeed Research Panel of consumers who indicated that they were caregivers of adults in their household. All respondents recruited via the Internet were screened to ensure that they were at least 18 years old, currently caring for someone with AD, and not a paid caregiver.
The Alzheimer’s Disease Caregiver Study was not reviewed by an institutional review board, but all applicable principles regarding protection of human research subjects were adhered to. Most importantly, information about confidentiality and the voluntary nature of participation was included in the introduction to the questionnaire, survey responses were anonymized, and results were reported in aggregate form.
Operational Definitions
AD symptom severity. We assessed AD symptom severity for the past week using the overall severity score of the Revised Memory and Behavioral Problem Checklist (RMBPC).16 The RMBPC measures how frequently dementia-related problems occur (0 = never occurred to 4 = daily or more often) and the caregiver’s reaction to these problems. The 24 dementia-related problems are grouped into 3 broad categories: memory loss, depression, and behavioral disturbances. The overall severity score is calculated as the mean of the frequency ratings of the 24 problems and ranges from 0 to 4, with higher scores indicating greater AD symptom severity. For descriptive purposes only, we categorized RMBPC overall severity score as mild (0-1), moderate (> 1-2), moderate-severe (> 2-3), and severe (> 3-4). In all models, RMBPC overall severity score was entered as a continuous variable.
Patient and caregiver characteristics. We characterized patient living situation as living with the caregiver versus not. We did not include patient demographics, as these were highly correlated with caregiver demographics and would cause problems with multicollinearity in the models. Additionally, we assessed patient functional status using the Instrumental Activities of Daily Living (IADL) scale and the Physical Self-Maintenance Scale (PSMS).17 The 6-item PSMS asks observers to rate older persons’ competence in toileting, feeding, dressing, grooming, locomotion, and bathing behaviors. The IADL measures the competence of older people in instrumental activities: shopping, telephoning, food preparation, housekeeping, use of transportation, doing laundry, use of medicine, and money management. Possible ratings for all but toileting items are 1 = without any help, 2 = with some help, and 3 = unable to do on his/her own. Toileting includes 2 items: trouble getting to the bathroom on time (yes/no) and frequency of wetting or soiling self (never, less than once a week, once or twice a week, 3 times a week, or more). IADL scores range from 9 to 27, and PSMS scores range from 8 to 24. Higher scores on both scales indicate a higher level of functioning.
We included caregiver demographics and health conditions as covariates in our analyses. Caregiver demographics included gender, race, education, and type of caregiver (spouse, adult child, and other). Age and marital status were not included because they were highly correlated with type of caregiver and would cause problems with multicollinearity in the models. Caregiver health conditions were assessed as number of physical health conditions and presence of psychiatric condition.
Caregiver outcomes. We assessed caregiver burden for the past week using the overall burden score of the Caregiver Burden Scale,18,19 which measures the demand and difficulty of caring for a chronically ill person at home. Caregivers are asked to indicate how often they perform 15 caregiving tasks and how difficult they find these tasks. These tasks are broadly classified as instrumental care (eg, running errands, managing finances, or providing transportation), direct care (eg, providing personal care or administering medical treatments), and interpersonal care (eg, providing emotional support, monitoring the patient, or communicating with the patient). This instrument was originally validated among caregivers of cancer patients, but the individual items are not specific to any condition, and this scale has been used in previously published studies of caregivers of AD patients.13,20 Scores range from 0 to 5, with higher scores indicating a greater level of burden.
Caregivers were given a list of health conditions and asked if they had ever been diagnosed with any of them by a physician. Two psychiatric conditions were included: depression and anxiety. Caregivers were asked to report ER use, hospitalization, and visits to medical providers in the past 6 months for their own health conditions. We assessed caregiver health care resource use for the past 6 months with 3 metrics: use of ER, hospitalization, and number of visits to all medical providers. We assessed caregiver lost work productivity as the number of missed work days specifically due to caregiving in the past 6 months. This metric was assessed only for respondents who reported being employed full time.
Statistical Analyses
We compared caregiver outcomes, demographics, comorbidity, patient living situation, and functional status across the 4 descriptive categories of AD symptom severity. We used χ2 to test for significant differences in categorical variables across the 4 severity categories, and we used analysis of variance to test for significant differences in continuous variables across severity categories.
We developed separate linear regression models to assess the independent effects of AD symptom severity on the following caregiver outcomes: caregiver burden, caregiver lost work productivity, and caregiver provider visits. Linear regression was chosen after considering scatter plots of the relationship between RMBPC severity score and each of these outcomes, which illustrated a linear relationship between the variables. We developed separate logistic regression models to assess the independent effects of AD symptom severity on caregiver psychiatric conditions, ER use, and hospitalization. Logistic regression was chosen because presence of anxiety and presence of depression were dichotomous variables. ER use and hospitalization were considered dichotomized variables because they are relatively rare occurrences, and those who experienced any ER use or hospitalization generally reported single use. In all multivariable models, we entered symptom severity as RMBPC overall severity score. We included interactions of severity and other covariates in the models on the basis of results of previous analyses stratified by severity. Other covariates included caregiver demographics, caregiver comorbidity, patient living situation, and patient functional status. Throughout the analyses, significance was assessed at an α level of .01. All analyses were based on the full analytic sample (n = 1,034) and were conducted using SPSS 15.0 for Windows (IBM Corporation, Somers, New York).
RESULTS
Caregiver and Patient Characteristics
Of the 1,077 respondents, 1,034 had valid RMBPC overall symptom severity scores. The caregiver sample with valid RMBPC scores was 66.7% female, 20.2% nonwhite, and 59.7% adult child caregivers of a parent or parent-in-law. Nearly half (48%) reported experiencing either anxiety or depression, with 33% experiencing anxiety and 37% experiencing depression.
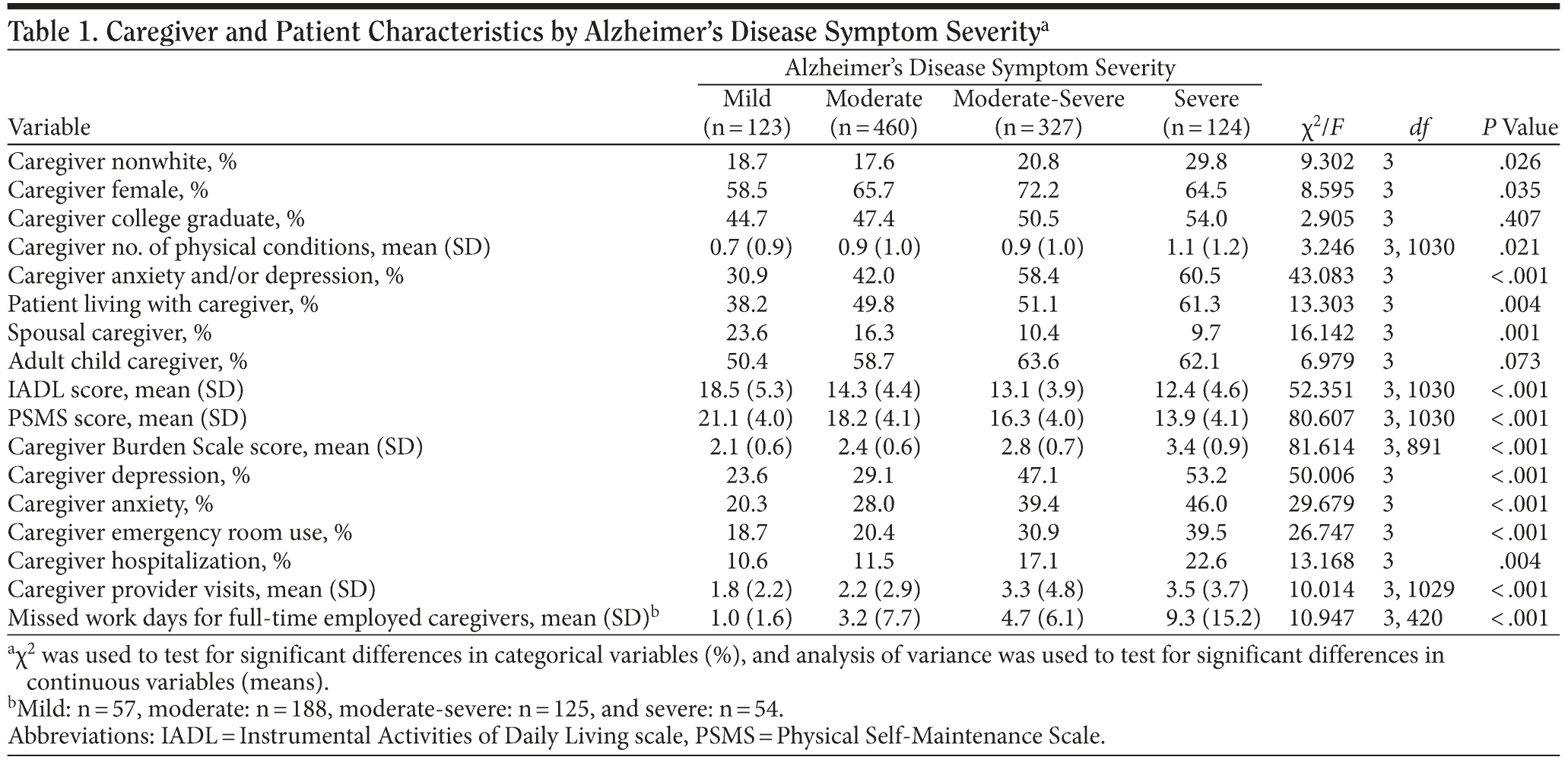
Table 1 provides caregiver and patient characteristics by AD symptom severity, which has been classified into 4 categories for descriptive purposes. Caregivers of patients with greater symptom severity were more likely to report a psychiatric condition. Caregivers of patients with more severe AD were less likely to be spousal caregivers, though the proportion of adult child caregivers did not significantly vary by severity, and they were more likely to live with the patient. Patients with greater symptom severity also experienced significantly poorer functional status, as measured by the IADL and PSMS.
Effects of AD Symptom Severity on Caregiver Burden
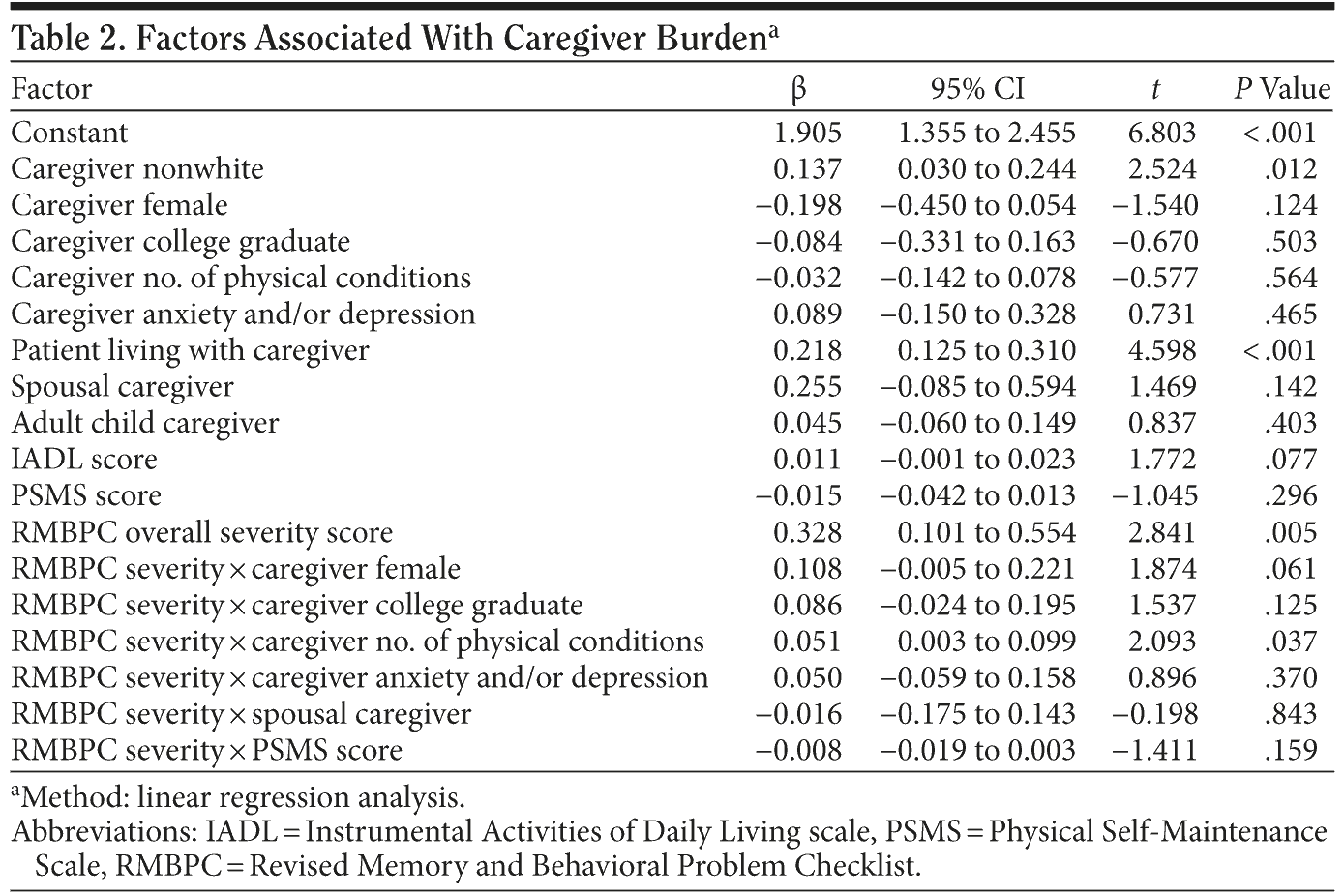
Prior to adjusting for covariates, caregiver burden varied significantly by AD symptom severity. Caregivers of patients with greater symptom severity experienced substantially more burden from caregiving tasks (Caregiver Burden Scale; mild: 2.1 to severe: 3.4; P < .001) (Table 1). Increase in RMBPC overall severity score corresponded to an increase in caregiver burden (Table 2). Among covariates, “patient living with the caregiver” was also a significant predictor of caregiver burden (Table 2).
Effects of AD Symptom Severity on Caregiver Psychiatric Conditions
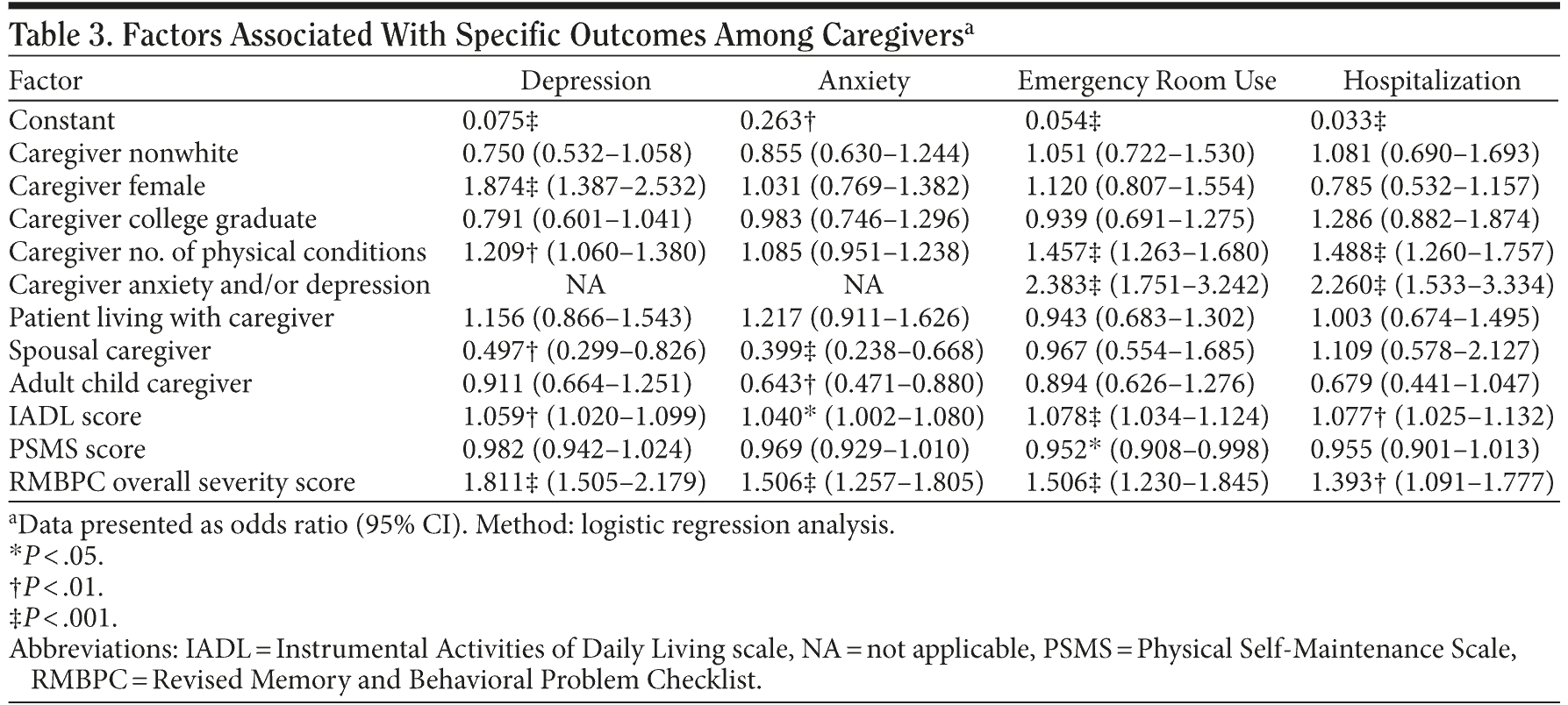
Caregiver psychiatric conditions were significantly associated with AD symptom severity. Caregivers of more severe patients were more likely to report anxiety than caregivers of more mild patients (Table 1). Adjusting for covariates, for each unit increase in RMBPC severity score, caregivers were about 1.5 times as likely to report a diagnosis of anxiety (Table 3) and almost twice as likely to report a diagnosis of depression (Table 3).
Other covariates were also significant predictors of caregiver psychiatric conditions. The caregiver being female, physical comorbidity, and higher patient functioning as measured by IADLs were also associated with a significantly greater likelihood of the caregiver experiencing depression. Being a spousal caregiver (compared with other caregivers) was associated with significantly lesser odds of experiencing depression. Being a spousal or adult child caregiver (compared with other caregivers) was associated with significantly lesser odds of experiencing anxiety (Table 3).
Effects of AD Symptom Severity on Caregiver Health Care Resource Use and Lost Productivity
Prior to adjusting for covariates, caregivers of more severe patients reported greater health care resource use. Specifically, in a 6-month period, caregivers of patients with more severe AD were more likely to use ERs (mild: 19% to severe: 40%) and to be hospitalized (mild: 11% to severe: 23%) (Table 1). They also had more visits to providers (mild: 1.8 visits to severe: 3.5 visits; P < .001) (Table 1). Adjusting for covariates, each unit increase in AD symptom severity corresponded to a greater likelihood of ER use (odds ratio [OR] = 1.506; 95% CI, 1.230-1.845) and hospitalization (OR = 1.393; 95% CI, 1.091-1.777) (Table 3). The association of AD symptom severity and number of provider visits was not significant (b = 0.343; 95% CI, 0.052-0.635; t = 2.309; P = .021).
Other covariates were also significant predictors of health care resource use. Caregiver physical and psychiatric comorbidity was associated with greater odds of ER use and hospitalization and a greater number of visits to health care providers. Additionally, higher patient functioning as measured by IADLs was associated with significantly greater odds of ER use and hospitalization (Table 3).
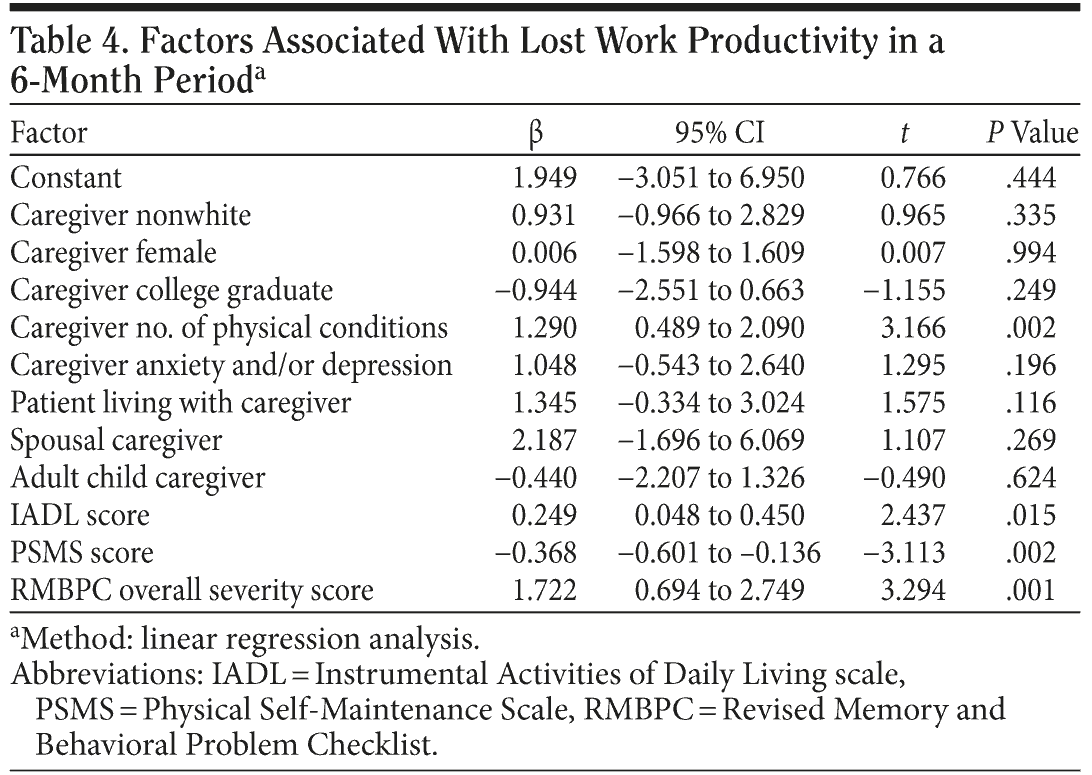
Among caregivers who worked full-time, those caring for patients with more severe AD missed significantly more days of work than those caring for patients with less severe disease (mild: 1.0 days to severe: 9.3 days) (Table 1). Adjusting for covariates, each unit increase in severity was associated with an increase of 1.722 (95% CI, 0.694-2.749) days of lost work due to caregiving in a 6-month period (Table 4). Physical comorbidity was also associated with a significantly greater number of lost workdays, and PSMS scores were associated with a significantly fewer number of lost workdays (Table 4).
CONCLUSIONS
With the increasing number of people with AD, the prominent role of informal caregivers in the daily care of patients, and the changing composition of informal caregivers, current understanding of the association of AD symptom severity with caregiver outcomes is more critical than ever. Previous research has found that patient cognitive decline and behavioral symptoms are associated with greater caregiver burden and poorer quality of life,6-10 as well as greater use of health care services11,12 and lost time from work.13 Results of our study were similar and update the current evidence on the economic and humanistic costs associated not only with caregiving, but also with caring for patients with more severe AD.
These results illustrate a consistent association of AD symptom severity with poorer caregiver outcomes and, ultimately, greater costs to individuals and society. Individual costs to caregivers associated with greater AD symptom severity include the greater levels of burden in performing caregiving tasks and the increased odds of experiencing anxiety and depression. Societal costs include both the direct and indirect economic impact of AD symptom severity. Caregivers of patients with more severe AD made greater use of health care resources, including greater likelihood of ER use and hospitalization. These results are consistent with research from 2008 that demonstrates a significant association between patient behavioral and psychological symptoms and caregiver use of ER and hospitalization, which correspond to a greater cost of care for caregivers of patients with more severe disease.12 In addition, employed caregivers of these patients face missing more days of work. This lost productivity negatively affects employers and may negatively affect the income of caregivers themselves.
In the past decade, caregivers of patients with AD have become increasingly more likely to be adult children caring for a parent or parent-in-law.1,14,15 Therefore, differential outcomes experienced by different types of caregivers are especially relevant. Spousal caregivers were less likely to experience depression and anxiety than other types of caregivers. In our initial analyses of the correlations between potential covariates, we found that spousal caregivers were older and less likely to be employed, which in itself is related to older age. These demographic differences likely affect depression and anxiety. For instance, being both employed and a caregiver may cause role strain, which may negatively affect mental well-being.
The natural course of AD is a continuing decline in cognitive functioning, often accompanied by an increase in behavioral symptoms.21,22 Cholinesterase inhibitors have been shown to slow the progression of symptoms.22 Results of this study illustrate the substantial effect of AD symptom severity on caregiver outcomes. Therefore, treatments that slow symptom progression may be expected to have a short-term positive effect on caregiver outcomes. Specifically, treatment of AD may reduce caregiver burden, presence of depression and anxiety, health care resource use, and lost work productivity in the short-term. By slowing disease progression and thereby reducing severity, reductions in caregiver burden, in depression and anxiety, in health care resource use, and in lost work productivity may be expected for the caregiver.
For example, in a 24-week, placebo-controlled trial, caregivers of patients with moderate to severe AD were assessed using the Caregiver Stress Scale.23 Caregivers of donepezil-treated patients reported lower levels of overload, relational deprivation, job-caregiving conflict, economic strains, role captivity, and loss of self compared with caregivers of placebo-treated patients.23 Caregivers of patients receiving donepezil also reported lower medication use-including antihypertensives, hypnotics/anxiolytics, and antidepressants-as well as 48% fewer visits to their primary care physician than caregivers of patients receiving placebo.24
Previous research has shown that treatment of patients also benefits caregivers. A study by Wimo et al25 indicated that treatment with donepezil had a positive impact with respect to decrease in caregiver time. An earlier study suggested that donepezil treatment reduced the difficulty associated with caregiving tasks.20 A meta-analysis of caregiver outcomes in clinical trials of cholinesterase inhibitors found a small beneficial effect of cholinesterase inhibitor use on caregiver burden and caregiver time.26 Further research should focus on associations of patient treatment with other caregiver outcomes such as presence of psychiatric conditions and costs associated with health care resource use and lost work productivity.
As current treatments slow but do not stop the progression of AD,22 long-term negative consequences for caregivers remain. In addition to prescription treatments, psychosocial interventions may further slow the progression of AD or act as a mediator between AD symptom severity and caregiver outcomes. Increasing caregiver knowledge of AD, awareness of available social support within the community, and resources to address legal and financial issues are believed to decrease caregiver stress.27 A study of AD caregivers demonstrated that psychoeducational training and respite vouchers, alone and in combination, were associated with less caregiver depression.28 Another study of AD caregivers demonstrated that psychosocial training of caregivers was associated with less caregiver distress, greater quality of life, and better perceived health.29 The Resources for Enhancing Alzheimer’s Caregivers Health study has demonstrated that intervention with home and telephone support group sessions is cost-effective in reducing caregiver time.30
The limitations of this study were primarily related to data collection methods. The study was a cross-sectional study and therefore causation could not be inferred. However, significant associations have been illustrated that add to the body of knowledge of caregiver outcomes. Also, a portion of the sample was Web-based and the non-Web-based sample was obtained from central city locations. Therefore, the sample may not be representative of people who do not have Internet access and/or who live in more rural areas. All data were self-reported by caregivers. Metrics such as AD symptom severity were not confirmed by clinicians or through patient records, though the RMBPC has been validated as a caregiver-reported metric of AD symptom severity. In addition, there may be other potential patient and caregiver characteristics that are associated with caregiver outcomes but were not considered in this analysis. These may include caregiver medication use, geographic region, and interactions between other covariates such as caregiver type and patient living situation. Future research should consider these and other potential covariates.
AD symptom severity is associated with poorer caregiver outcomes that have cost implications to the individual and society as a whole. Individual costs to caregivers of more severe patients include greater burden of direct, instrumental, and interpersonal care and greater likelihood of anxiety and depression. Greater health care resource use by caregivers of more severe patients translates to increased direct costs of care for caregivers, and greater lost work productivity increases indirect costs to employers. Implications of findings such as these are that available treatments that aid in the management of AD symptom progression may reasonably be expected to positively impact not only patient outcomes and their associated costs but also caregiver outcomes and their associated costs as well. Future research should focus on establishing possible causal effects among the associations found here.
Drug name: donepezil (Aricept and others).
Author affiliations: Consumer Health Sciences, Princeton, New Jersey (Dr Bolge and Ms Kannan); and Pfizer Inc, New York, New York (Drs Alvir and Petrie and Ms del Valle).
Potential conflicts of interest: Ms Kannan and Dr Bolge were employees of Consumer Health Sciences at the time of the study. Drs Alvir and Petrie are employees of and stock shareholders in Pfizer Inc. Ms del Valle was an employee of Pfizer Inc at the time of the study.
Funding/support: The Alzheimer’s Disease Caregiver Study is conducted by Consumer Health Sciences, Princeton, New Jersey. Pfizer Inc, New York, New York, licensed access to the study and funded the analysis presented in this article. Editorial assistance was provided by Hilary Bennett, MSc, from PPSI, a PAREXEL Company (Worthing West Sussex, United Kingdom), and Bill Kadish, MD, from PPSI, a PAREXEL Company (Stamford, Connecticut), and was funded by Pfizer Inc. Neither Ms Bennett or Dr Kadish report any conflicts of interest related to the subject of this article.
Previous presentation: Presented at The International Society for CNS Clinical Trials and Methodology, Autumn Meeting; October 6-7, 2008; Toronto, Ontario, Canada; and 24th Conference of Alzheimer’s Disease International; March 25-28, 2009; Singapore.
REFERENCES
1. Alzheimer’s Association. 2009 Alzheimer’s disease facts and figures. Alzheimers Dement. 2009;5(3):234-270. PubMed doi:10.1016/j.jalz.2009.03.001
2. Cummings JL, Cole G. Alzheimer disease. JAMA. 2002;287(18):2335-2338. PubMed doi:10.1001/jama.287.18.2335
3. Andersen CK, Wittrup-Jensen KU, Lolk A, et al. Ability to perform activities of daily living is the main factor affecting quality of life in patients with dementia. Health Qual Life Outcomes. 2004;2(1):52. PubMed doi:10.1186/1477-7525-2-52
4. Zhu CW, Leibman C, McLaughlin T, et al. The effects of patient function and dependence on costs of care in Alzheimer’s disease. J Am Geriatr Soc. 2008;56(8):1497-1503. PubMed doi:10.1111/j.1532-5415.2008.01798.x
5. Gaugler JE, Yu F, Krichbaum K, et al. Predictors of nursing home admission for persons with dementia. Med Care. 2009;47(2):191-198. PubMed doi:10.1097/MLR.0b013e31818457ce
6. Burns A, Rabins P. Carer burden dementia. Int J Geriatr Psychiatry. 2000;15(suppl 1):S9-S13. PubMed doi:10.1002/1099-1166(200007)15:1+<::AID-GPS160>3.0.CO;2-N
7. Black W, Almeida OP. A systematic review of the association between the behavioral and psychological symptoms of dementia and burden of care. Int Psychogeriatr. 2004;16(3):295-315. PubMed doi:10.1017/S1041610204000468
8. Karlawish JH, Casarett D, Klocinski J, et al. The relationship between caregivers’ global ratings of Alzheimer’s disease patients’ quality of life, disease severity, and the caregiving experience. J Am Geriatr Soc. 2001;49(8):1066-1070. PubMed doi:10.1046/j.1532-5415.2001.49210.x
9. Ferrara M, Langiano E, Di Brango T, et al. Prevalence of stress, anxiety and depression in with Alzheimer caregivers. Health Qual Life Outcomes. 2008;6(1):93. PubMed doi:10.1186/1477-7525-6-93
10. Matsumoto N, Ikeda M, Fukuhara R, et al. Caregiver burden associated with behavioral and psychological symptoms of dementia in elderly people in the local community. Dement Geriatr Cogn Disord. 2007;23(4):219-224. PubMed doi:10.1159/000099472
11. Toseland RW, McCallion P, Gerber T, et al. Predictors of health and human services use by persons with dementia and their family caregivers. Soc Sci Med. 2002;55(7):1255-1266. PubMed doi:10.1016/S0277-9536(01)00240-4
12. Schubert CC, Boustani M, Callahan CM, et al. Acute care utilization by dementia caregivers within urban primary care practices. J Gen Intern Med. 2008;23(11):1736-1740. PubMed doi:10.1007/s11606-008-0711-0
13. Small GW, McDonnell DD, Brooks RL, et al. The impact of symptom severity on the cost of Alzheimer’s disease. J Am Geriatr Soc. 2002;50(2):321-327. PubMed doi:10.1046/j.1532-5415.2002.50065.x
14. Yordi C, DuNah R, Bostrom A, et al. Caregiver supports: outcomes from the Medicare Alzheimer’s disease demonstration. Health Care Financ Rev. 1997;19(2):97-117. PubMed
15. Neumann PJ, Kuntz KM, Leon J, et al. Health utilities in Alzheimer’s disease: a cross-sectional study of patients and caregivers. Med Care. 1999;37(1):27-32. PubMed doi:10.1097/00005650-199901000-00005
16. Teri L, Truax P, Logsdon R, et al. Assessment of behavioral problems in dementia: the Revised Memory and Behavior Problems Checklist. Psychol Aging. 1992;7(4):622-631. PubMed doi:10.1037/0882-7974.7.4.622
17. Lawton MP. Scales to measure competence in everyday activities. Psychopharmacol Bull. 1988;24(4):609-614. PubMed
18. Carey PJ, Oberst MT, McCubbin MA, et al. Appraisal and caregiving burden in family members caring for patients receiving chemotherapy. Oncol Nurs Forum. 1991;18(8):1341-1348. PubMed
19. Oberst MT, Thomas SE, Gass KA, et al. Caregiving demands and appraisal of stress among family caregivers. Cancer Nurs. 1989;12(4):209-215. PubMed doi:10.1097/00002820-198908000-00003
20. Fillit HM, Gutterman EM, Brooks RL. Impact of donepezil on caregiving burden for patients with Alzheimer’s disease. Int Psychogeriatr. 2000;12(3):389-401. PubMed doi:10.1017/S1041610200006499
21. Bruen PD, McGeown WJ, Shanks MF, et al. Neuroanatomical correlates of neuropsychiatric symptoms in Alzheimer’s disease. Brain. 2008;131(Pt 9):2455-2463. PubMed doi:10.1093/brain/awn151
22. Shah S, Reichman WE. Treatment of Alzheimer’s disease across the spectrum of severity. Clin Interv Aging. 2006;1(2):131-142. PubMed doi:10.2147/ciia.2006.1.2.131
23. Feldman H, Gauthier S, Hecker J, et al; Donepezil MSAD Study Investigators Group. Efficacy of donepezil on maintenance of activities of daily living in patients with moderate to severe Alzheimer’s disease and the effect on caregiver burden. J Am Geriatr Soc. 2003;51(6):737-744. PubMed doi:10.1046/j.1365-2389.2003.51260.x
24. Winblad B, Wimo A, Mastey V, et al. Caregiver health benefits and associated reductions in healthcare costs as a consequence of treating patients with Aricept. Value Health. 2003;6:276.
25. Wimo A, Winblad B, Shah SN, et al. Impact of donepezil treatment for Alzheimer’s disease on caregiver time. Curr Med Res Opin. 2004;20(8):1221-1225. PubMed doi:10.1185/030079902125004349
26. Lingler JH, Martire LM, Schulz R. Caregiver-specific outcomes in antidementia clinical drug trials: a systematic review and meta-analysis. J Am Geriatr Soc. 2005;53(6):983-990. PubMed doi:10.1111/j.1532-5415.2005.53313.x
27. Skinner K. Nursing interventions to assist in decreasing stress in caregivers of Alzheimer’s patients. ABNF J. 2009;20(1):22-24. PubMed
28. Tompkins SA, Bell PA. Examination of a psychoeducational intervention and a respite grant in relieving psychosocial stressors associated with being an Alzheimer’s caregiver. J Gerontol Soc Work. 2009;52(2):89-104. PubMed doi:10.1080/01634370802561877
29. Mart×n-Carrasco M, Mart×n MF, Valero CP, et al. Effectiveness of a psychoeducational intervention program in the reduction of caregiver burden in Alzheimer’s disease patients’ caregivers. Int J Geriatr Psychiatry. 2009;24(5):489-499. PubMed doi:10.1002/gps.2142
30. Nichols LO, Chang C, Lummus A, et al; Resources for Enhancing Alzheimer’s Caregivers Health II Investigators. The cost-effectiveness of a behavior intervention with caregivers of patients with Alzheimer’s disease. J Am Geriatr Soc. 2008;56(3):413-420. PubMed doi:10.1111/j.1532-5415.2007.01569.x