Dementia With Lewy Bodies: Diagnosis and Management for Primary Care Providers
Objective: The purpose of this review is to aid primary care providers in distinguishing dementia with Lewy bodies (DLB) from Alzheimer’s disease and from Parkinson’s disease with dementia. Differentiating these entities has important treatment implications.
Data Sources: A PubMed search was undertaken using the keywords Lewy body dementia, dementia with Lewy bodies, and Lewy body disease. There were no date restrictions. Only articles in the English language were reviewed. References of selected articles were reviewed for additional sources.
Data Selection and Extraction: Initially, 2,967 articles were retrieved. All 3 authors participated in data selection and extraction. Articles were further selected for content specific to epidemiology, clinical presentation, diagnostic studies, treatment, and prognosis. For articles with repetitive information, the most current article was used. This resulted in a total of 62 articles included in the review.
Data Synthesis: Dementia with Lewy bodies is the second leading cause of dementia after Alzheimer’s disease. The core symptoms of DLB, including cognitive fluctuations, visual hallucinations, and parkinsonism, may not always be present as a triad, and clinicians may be unaware of associated symptoms. Thus, this diagnosis is frequently missed by primary care providers. Often, DLB is misdiagnosed as Alzheimer’s disease, Parkinson’s disease, or a primary psychiatric illness. Treatments for DLB include cholinesterase inhibitors and N-methyl-d-aspartate antagonists. Antipsychotics should be avoided or used with caution.
Conclusions: Dementia with Lewy bodies is an often missed diagnosis. Symptoms are often attributed to other disorders. A high clinical suspicion is helpful in accurate diagnosis, and presence of any of the core symptoms should initiate clinical suspicion of DLB. Distinguishing DLB from other disorders has important treatment implications.
Prim Care Companion CNS Disord 2011;13(5):doi:10.4088/PCC.11r01190
© Copyright 2011 Physicians Postgraduate Press, Inc.
Submitted: March 31, 2011; accepted June 2, 2011.
Published online: October 27, 2011.
Corresponding author: Melanie Zupancic, MD, Department of Internal Medicine, Division Medicine/Psychiatry, Southern Illinois University School of Medicine, PO Box 19363, Springfield, IL 62794-9636 ([email protected]).
Dementia with Lewy bodies (DLB) accounts for 15% to 35% of dementia cases.1,2 This disorder presents several difficulties regarding both diagnosis and management. Many patients initially present to their primary care physician. Thus, the tasks of diagnosing DLB, differentiating it from other types of dementia, and initiating evidence-based treatment often fall on primary care providers. The purpose of this review is to summarize the diagnostic criteria and clinical features, radiographic features, pathology, and treatment of this disorder to aid primary care physicians in caring for this population.
METHOD
A PubMed search was undertaken using the keywords Lewy body dementia, dementia with Lewy bodies, and Lewy body disease. References of selected articles were reviewed for additional sources. Initially, 2,967 articles were retrieved. There were no date restrictions. Only articles in English were reviewed. All 3 authors participated in data selection and extraction. Articles were further selected for relevance regarding epidemiology, clinical presentation, diagnostic studies, treatment, and prognosis. For articles with repetitive information, the most current article was used. This resulted in a total of 62 articles included in the review.
Clinical Points
- Dementia with Lewy bodies is an underrecognized diagnosis.
- Clinical suspicion is key to accurate diagnosis, and core symptoms should be assessed in all patients presenting with cognitive complaints.
- Cholinesterase inhibitors are the first-line treatment for dementia with Lewy bodies.
DIAGNOSTIC FEATURES
DLB is the second most common cause of dementia after Alzheimer’s disease.1,2 Thus, accurate recognition is crucial. Diagnostic criteria (Table 1) were initially established in 1996 and subsequently revised to improve sensitivity.3,4 However, despite the revised diagnostic criteria, the recognition of DLB remains poor, with a reported rate of less than 50% in one study.5 One reason for the low recognition of DLB is the difficulty associated with recognizing the diagnostic criteria. A high index of clinical suspicion is needed along with collateral information from family and caretakers.
Dementia
The diagnosis of dementia requires a gradual decline of cognitive function that impedes daily functioning and is accompanied by aphasia, apraxia, agnosia, or impaired executive function.6 The pattern of cognitive deficits in DLB differs from that observed in Alzheimer’s disease. Memory impairments may not be present until the latter stages of DLB. Standard bedside tests such as the Mini-Mental State Examination (MMSE) cannot reliably discriminate between DLB and other forms of dementia.3 Neuropsychological testing reveals several specific deficits. When matched for age, education, and MMSE score, patients with DLB tend to demonstrate more deficits in visuoperceptive tasks, visuoconstructive tasks, nonverbal reasoning, attention, and executive function compared to patients with Alzheimer’s disease.7-13 Additionally, DLB patients may show greater impairments in functional abilities earlier in the disease than Alzheimer’s disease patients.14 Deficits in neuropsychological testing demonstrated in DLB patients are similar to those seen in patients with Parkinson’s disease with dementia.3,15 Thus, neuropsychological testing is not a reliable tool to differentiate Parkinson’s disease with dementia from DLB. The differentiation of these two disorders is discussed in detail later in this review.
Fluctuating Cognition
The prevalence of fluctuating cognition in DLB has varied considerably across different studies, ranging from 30% to 89%.16,17 Fluctuations vary in intensity, and their duration can range from hours to days. The variable temporal profile makes these features difficult to detect by physicians who do not see the patient for prolonged periods of time. Distinguishing the “good days” and “bad days” often reported by caregivers of Alzheimer’s disease patients from the fluctuations indicative of DLB can be challenging to an inexperienced physician. It may be helpful to use validated scales such as the Clinician Assessment of Fluctation and the One Day Fluctuation Assessment Scale.18,19 The polarity of fluctuations can be dramatic. For example, on good days, patients may appear to be close to their previous baseline, attending to activities of daily living and participating in meaningful conversation. Hours later, they may be unable to find their bedroom, hallucinating, and having obvious motor deficits. History from family members and caregivers is essential in determining the presence of fluctuating mental status. Clinicians are encouraged to ask about lethargy, daytime somnolence, sustained periods of staring into space, periods of decreased attention, periods of improved memory, and episodes of disorganized speech, in addition to more dramatic fluctuations as described above.4 Caregivers of individuals with DLB often report observing episodes of blank staring with complete disengagement from the environment, whereas Alzheimer’s disease caregivers more frequently report episodes of heightened memory impairment and repetitiveness in conversation.18,19 Fluctuations in cognition in DLB tend not to be related to demands of memory function as they are with Alzheimer’s disease patients. Additionally, periods of confabulation and delusional thinking are more often reported in DLB patients.18,19
Recurrent Visual Hallucinations
The presence of recurrent complex visual hallucinations is a critical feature in differentiating between Alzheimer’s disease and DLB. Hallucinations in other sensory modalities also occur, but with decreased frequency. Visual hallucinations are present earlier in the course of disease in DLB while memory may be relatively preserved. Although the presence of visual hallucinations is highly specific, the sensitivity has ranged from 39% to 77%.14,16,17 Typical images include people and animals. Although the patient is aware that the images are not real, emotional responses are common.4
One caveat is that antiparkinsonian drugs commonly administered to this population because of the presence of extrapyramidal symptoms (EPS) may exacerbate or cause hallucinations. Thus, the presence of visual hallucinations with concurrent medication administration may not be as significant as hallucinations in drug-naive patients, but this has not been systematically studied. Hallucinations that persist after antiparkinsonian medications have been withdrawn in a patient with EPS should prompt the clinician to consider DLB as the primary diagnosis.
Spontaneous Parkinsonism
The prevalence of parkinsonism in DLB has ranged from 60% to 92%.16,17 The severity of EPS is similar to that observed in age-matched patients with Parkinson’s disease.16,17 Given the high rate of dementia in Parkinson’s disease patients, differentiating DLB from Parkinson’s disease with dementia can be difficult. It is suggested that if the onset of dementia occurs more than 12 months after the onset of motor symptoms, then Parkinson’s disease with dementia is the appropriate label. If cognitive deficits appear within 12 months of motor symptoms, then DLB is more likely.3 However, this 12-month rule is a generalization. The main point is that cognitive and motor symptoms in DLB appear close together in time, while in Parkinson’s disease with dementia, cognitive deficits appear years to decades after the onset of motor symptoms. Other key differences between DLB and Parkinson’s disease include a lower prevalence of resting tremor and a higher prevalence of postural instability, stooped posture, hypophonic speech, ataxia, and facial immobility in DLB compared to Parkinson’s disease.3,4 Additionally, responsiveness to l-dopa has been reported to be less in DLB patients compared to Parkinson’s disease patients.3,4 Although parkinsonism occurs in Alzheimer’s disease, it tends to appear later in the course of illness than in DLB or Parkinson’s disease with dementia.
REM Sleep Behavior Disorder
REM sleep behavior disorder (RBD) is a parasomnia characterized by the presence of vivid dreams and complex motor movement during REM sleep without muscle atonia. It predominately occurs in males and has an onset after the age of 50. Patients will act out their dreams through speech and movement, often shouting or talking with accompanying limb flailing and/or falling out of bed. Violent behavior can also occur. Iranzo et al20 reported on a series of 17 patients with RBD and DLB. Self-awareness of behaviors was present in 29% of patients, and the recollection of unpleasant dreams, in 82%. Unpleasant dreams most frequently involved being attacked by someone, arguing with someone, and being chased by someone. The most frequently reported behaviors in descending order were talking, shouting, falling out of bed, punching, and laughing. Forty percent of patients reported sustaining injuries, and 6% injured their bed partner.20 Often the history is obtained from a bed partner, and symptoms can be present for decades before the onset of dementia.21,22
Idiopathic RBD can be a harbinger of a subsequent neurodegenerative disease, namely the α-synucleinopathies (Parkinson’s disease, multiple system atrophy, and DLB). The prevalence of polysomnogram-confirmed RBD in patients with α-synucleinopathies is 39% versus 0% in patients with nonsynucleinopathies (Alzheimer’s disease, frontotemporal dementia, corticobasal degeneration, progressive supranuclear palsy, mild cognitive impairment, primary progressive aphasia, and posterior cortical atrophy).23 The frequency of probable RBD in Parkinson’s disease and DLB patients compared to those with Alzheimer’s disease and mild cognitive impairment was 56% versus 2%.23 In 10 autopsied patients with clinically diagnosed RBD, 9 had Lewy body disease.23 The estimated 10-year risk of developing a neurodegenerative disease has been reported to be 40% in patients with RBD.24 The reported risk of development of DLB has ranged from 7% to 14% in patients presenting with idiopathic RBD.22,24 The exact prevalence of RBD in patients with DLB is unknown. However, studies have suggested rates of 50% to 83%.17
Clinicians with patients diagnosed with idiopathic RBD should monitor for DLB as the patient ages, and in patients suspected of having dementia, the presence of RBD should prompt consideration of DLB as the primary diagnosis.
Neuroleptic Sensitivity
Primary care providers understandably may consider the use of antipsychotics in this population. However, the decision to utilize these medications is not without significant risk. In a series of 20 patients with DLB, McKeith et al25 reported that 80% received neuroleptics. Of those, 46% developed EPS for the first time after neuroleptic administration. Additionally, 81% of patients receiving antipsychotics developed a neuroleptic sensitivity syndrome ranging from mild (54%) to severe (46%). A mild syndrome was characterized by increased tone, rigidity, or bradykinesia. Severe reactions were characterized by sudden onset of sedation, immobility, fever, and confusion at times resembling neuroleptic malignant syndrome. Patients suffering severe reactions did not differ in age, sex, presence of psychosis, EPS at presentation, or syncopal episodes from those not suffering severe reactions. Severe reactions were also associated with a 2-fold increase in mortality. In a follow-up study, Ballard et al26 found that 29% of DLB patients receiving neuroleptics had severe reactions, half of whom died within 2 months.
Supportive Features
As highlighted in Table 1, there are several features that support a diagnosis of DLB, but are not specific enough to allow definitive diagnosis. Autonomic instability is also present in other α-synucleopathies and, in addition to parkinsonism, may predispose patients to falls or syncope. In turn, patients may come to clinical attention because of repeated falls or syncopal episodes. McKeith et al25 found that 35% of DLB patients reported repeated unexplained falls, while 25% endorsed syncopal episodes at presentation. In patients presenting with frequent falls or loss of consciousness, cognitive deficits may be elicited only through careful questioning. Psychiatric symptoms including depression, anxiety, and psychosis are common. In a series of 92 DLB patients, Borroni et al27 found that 67% reported anxiety, 62% endorsed depression, 58% complained of apathy, and half the sample had psychotic symptoms. Psychotic symptoms including delusions and hallucinations, and anxiety worsened with disease progression, although depression remained stable. No relationship between neuropsychiatric symptoms and motor symptoms was observed. McKeith et al25 found that 45% of DLB patients endorsed auditory hallucinations, while 80% had delusions at some point during their illness.
Radiographic Features
Clinically, differentiating DLB and other dementia subtypes such as Parkinson’s disease with dementia and Alzheimer’s disease can be difficult. Considering poor sensitivity of the diagnostic criteria, it is important to look at additional distinguishing investigations to improve diagnostic precision.5 The American Academy of Neurology28 recommends structural neuroimaging with either noncontrast head computed tomography (CT) or magnetic resonance imaging (MRI) in the routine initial evaluation of all patients with dementia to rule out malignancy, normal pressure hydrocephalus, and subdural hematomas as possible causes.
An MRI is more sensitive than a CT scan for studying regional changes in tissue volume in the brain. Alzheimer’s disease is characteristically defined by diffuse cerebral atrophy with a disproportionate amount in the medial temporal lobes with relative sparing of subcortical structures. Ventricular enlargement also is present as disease progresses.29 Conversely, DLB and Parkinson’s disease with dementia show similar changes on MRI consisting of atrophy of subcortical structures including the basal ganglia with relative preservation of medial temporal lobe structures.3 Additionally, DLB patients show slower rates of cerebral atrophy and ventricular enlargement compared to Alzheimer’s disease patients.29
Of increasing interest in differentiating Alzheimer’s disease from DLB are functional neuroimaging studies that examine cerebral perfusion and cerebral metabolism utilizing single-photon emission computed tomography (SPECT) or positron emission tomography (PET), respectively. This stems from the hypothesis that even those with DLB without parkinsonism will show degeneration in the nigrostriatal system, whereas those with Alzheimer’s disease will not. Using molecules that bind to the dopamine reuptake and transporter molecule found in presynaptic membranes of dopamine-producing neurons in the caudate and putamen, SPECT can be used to measure the density of dopaminergic neurons in the nigrostriatal system. Thus, reduced dopamine transporter binding has been proposed as a means to differentiate between DLB and Alzheimer’s disease.30 In a series of 23 patients with dementia, Walker and Walker30 found that the sensitivity and specificity for diagnosing DLB via SPECT was 100% and 92%, respectively. Other studies31-33 have reported abnormal dopamine transporter scans to have a sensitivity of over 75% and a specificity of over 90% for DLB. PET scans have shown hypometabolism in the posterior cortical region as well, which is thought to explain the increased incidence of visual hallucinations in this population versus Alzheimer’s disease. Both SPECT and PET scanning appeared useful for the diagnosis of DLB, but SPECT provided more robust results than PET.34 However, lack of availability limits the use of all functional neuroimaging.
Although an electroencephalogram (EEG) is not routinely done in evaluation of dementia, several investigators have examined its diagnostic utility. Bonnani et al35 identified slow activities (pre-α) in posterior derivations of all DLB patients that notably differentiated these patients from those with Alzheimer’s disease. Intermittent delta and sharp transients also occur more in patients with DLB than those with Alzheimer’s disease, although their frequency may be too small to aid in diagnosis.3,35 In short, EEG is not a useful tool in identifying DLB.
The role of biomarkers, including tau and amyloid β 42 concentrations in cerebrospinal fluid, in diagnosing dementia has garnered increased clinical attention recently and may have some diagnostic relevance. However, routine use of biomarkers in diagnosis is not recommended at this time by the American Academy of Neurology.28,36
PATHOLOGY
Pathological assessment of DLB reveals aggregates of α-synuclein, referred to as Lewy bodies, found in neurons and glia. They are hypothesized to stem from abnormal protein processing giving rise to cytoplasmic inclusions identifiable by immunohistochemistry.37 Their presence is required for the DLB diagnosis at autopsy. Brainstem involvement is almost universal, and variable amounts of Lewy body burden are found in the limbic system and neocortex.3,38 In addition to central nervous system involvement, Lewy bodies are also found in the peripheral nervous system and autonomic nervous system and thought to be related to orthostasis.38 There is some sense that the location of Lewy bodies relates to neural dysfunction. Brainstem lesions are thought to account for EPS, while cognitive and neuropsychiatric symptoms are attributed to neocortical and limbic involvement. However, despite the intuitive nature of this hypothesis, the burden of Lewy bodies in specific brain regions has not been conclusively demonstrated to correlate with clinical symptoms. Whether Lewy bodies are involved in neurodegeneration directly, a cellular defense mechanism, or by-products of an unidentified degenerative process has yet to be determined.38
Additionally, the mere presence of Lewy bodies is not in itself enough to cause clinical symptoms. In fact, an estimated 24% to 55% of neuropsychiatrically healthy elderly adults are found to have significant Lewy bodies.38 Confounding the pathological picture is the high incidence of comorbid Alzheimer’s pathology, which is present in 35% to 90% of cases.39
Similar to Alzheimer’s disease, DLB is also associated with deficiencies in cholinergic transmission. Additionally, relative to patients with Alzheimer’s disease, those with DLB have lower choline acetyltransferase activity.40-43 It has been hypothesized that the increased prevalence of psychotic symptoms and visuospatial dysfunction in DLB patients may be secondary to the greater deficits in cholinergic transmission.40-43 Reduced activity of choline acetyltransferase in the temporal and parietal neocortex has been observed in DLB patients and correlated with the presence of visual hallucinations, while up-regulation of muscarinic M1 postsynaptic receptors in the temporal lobe has been correlated with delusional thinking.43,44 Relative to Alzheimer’s disease, DLB is also associated with reductions in dopamine in the basal ganglia and norepinephrine in the putamen.41 Abnormalities in glutamate receptors and second messenger signaling have also been demonstrated.45
TREATMENT
Cognitive Symptoms
The marked decrease in cholinergic functioning observed in DLB patients relative to Alzheimer’s disease patients led to speculation that cholinesterase inhibitors (ChEIs) may have a greater potential for improvement in DLB than in Alzheimer’s disease. Additionally, the observation of less neuronal loss in DLB than in Alzheimer’s disease suggests that cortical neurons in DLB are more viable than those in Alzheimer’s disease and could be more responsive to cholinergic stimulation.29,41 Studies have not confirmed this hypothesis, though.
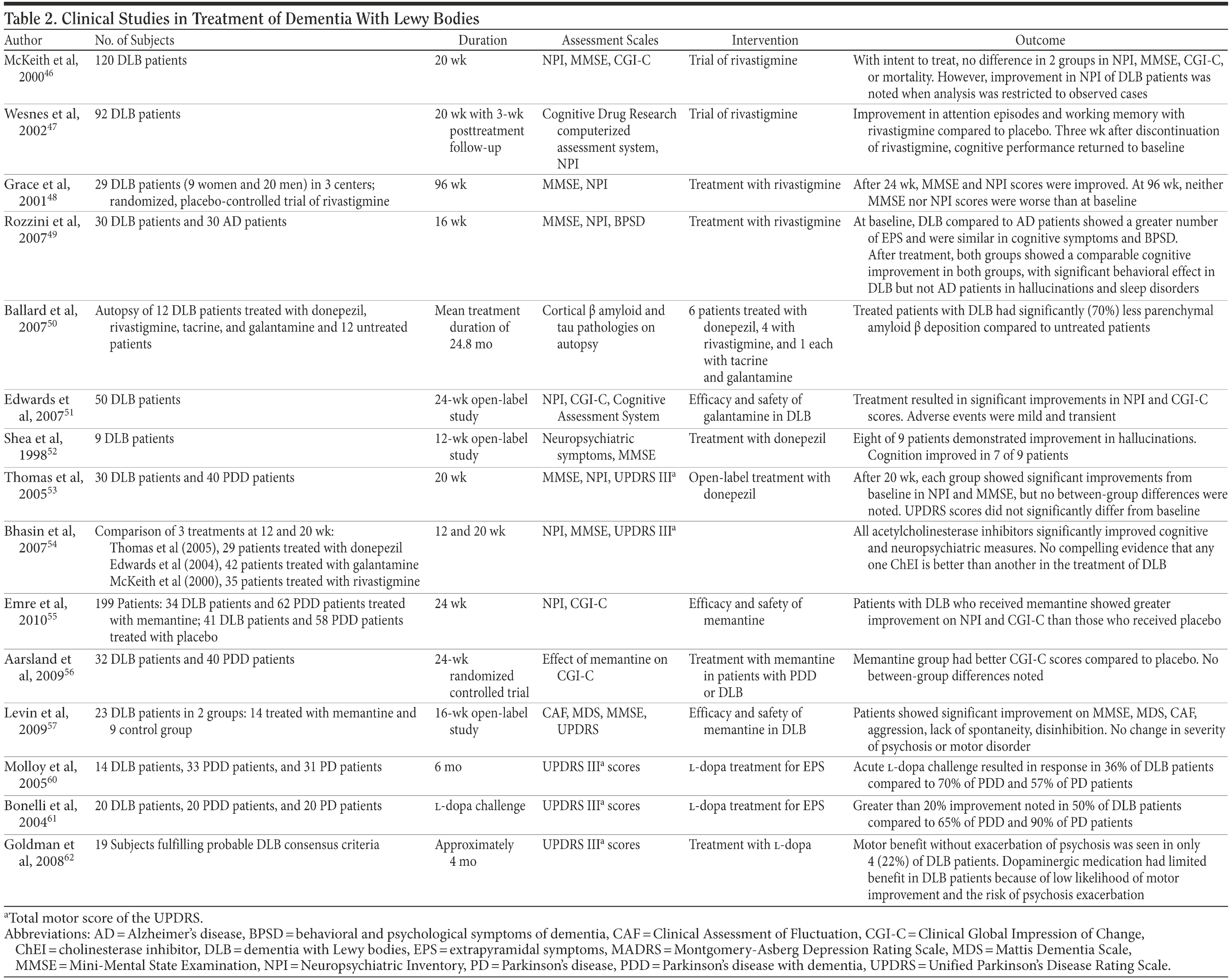
Patients with DLB who are treated with ChEIs have shown improvement in various neuropsychiatric measures. In a placebo-controlled, double-blind study conducted in 120 patients with DLB, McKeith et al46 found significant improvements in measures of attention as well as reductions in psychotic symptoms. There was no observed mortality benefit. In a prospective, randomized, double-blind, placebo-controlled trial involving 92 DLB patients treated with rivastigmine, Wesnes et al47 observed improvements in measures of attention, working memory, and episodic memory. Grace et al48 observed improvements in MMSE scores and neuropsychiatric symptoms over 24 weeks, a finding that has been observed by others.49 Grace and colleagues’ study was extended over 96 weeks, and neither MMSE nor Neuropsychiatric Inventory scores significantly differed from baseline, suggesting that ChEIs may attenuate degeneration.48 This hypothesis is further supported by findings by Ballard et al,50 who observed a 70% reduction in amyloid β pathology in autopsies of 12 patients with DLB receiving ChEI therapy versus patients not receiving such therapy. Groups were matched for age as well as duration and severity of dementia.
Galantamine has also been studied. It not only is a ChEI but also allosterically modulates neuronal nicotinic receptors. In patients with DLB, there is a selective loss of the alpha7 nicotinic receptors in the hippocampus, suggesting possible added benefits of galantamine for cognition and behavior due to its selective nicotinic receptor modulation. One study51 showed substantial benefits in visual hallucinations and fluctuating cognitive deficits. Finally, donepezil has also shown positive results in measures of cognition and neuropsychiatric symptoms.52,53 Differences between ChEIs have not been found,54 although head-to-head trials are lacking.
Thus, it appears that all ChEIs can be effective in this population. However, results should be viewed cautiously, as the number of trials is small, with few being placebo controlled, and the number of patients is limited. Additionally, although results suggest statistically significant improvement in symptoms, it is uncertain whether these correspond to clinically significant differences. To date, there has been no mortality benefit with any pharmacologic treatment of DLB, and the clinical course tends to be one of progressive deterioration.
Given the observed aberrations observed in glutaminergic transmission, it seems intuitive that the N-methyl-d-aspartate antagonist memantine may have a role in treatment. Memantine has been shown to decrease neuropsychiatric symptoms including aggression and disinhibition, as well as Clinical Global Impression of Change and MMSE scores.55-57
Neuropsychiatric Symptoms
DLB patients suffer from a variety of neuropsychiatric symptoms, as discussed previously, including anxiety, depression, and psychosis. Comorbid anxiety and depression is best addressed with selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs).3 There are no studies showing differences between SSRIs or SNRIs. However, it makes intuitive sense that those with stronger anticholinergic side effects such as paroxetine may not be the best first-line agents. Tricyclic antidepressants and other antidepressants with anticholinergic properties are best avoided, as they can worsen cognition and visual hallucinations. Efforts should also be made to avoid benzodiazepines, as they can worsen cognition and predispose this vulnerable population to falls. However, at times they may be necessary, particularly for RBD. In these cases, very low doses of clonazepam can be administered prior to bedtime. Other agents showing effectiveness in RBD include melatonin and very small doses of quetiapine.3,58,59 The latter should be used with caution, however, because of enhanced neuroleptic sensitivity.3 Apathy and aggression may respond to ChEIs and memantine, as mentioned above. Psychotic symptoms may improve with ChEIs.46,51 However, if the symptoms are refractory to this treatment and cause significant impairment and burden to caregivers, second-generation antipsychotics such as quetiapine and clozapine can be used in lower doses with caution and attention to neuroleptic sensitivity. This course of action involves a careful discussion with family members about the risk of neuroleptic sensitivity as well as the US Food and Drug Administration black box warning regarding use of these medications in patients with dementia. Unfortunately, there are no clear-cut guidelines to aid clinicians. If the question of antipsychotic use arises, referral to a specialist is appropriate.
Motor Symptoms
Motor symptoms can be particularly disabling for patients. Dopaminergic agents are typically administered for parkinsonism, but their use must be balanced against the risk of exacerbation of psychotic symptoms. Additionally, patients with DLB show a lower response than those with Parkinson’s disease, and it has been suggested that this be a distinguishing diagnostic feature.3 In a series of 14 patients with DLB, Molloy et al60 found that after an acute single-dose challenge with l-dopa, only 36% demonstrated a response, defined as a 20% or greater improvement in 2 of 3 tests of motor function on the Unified Parkinson’s Disease Rating Scale (UPDRS). Seventy percent of Parkinson’s disease with dementia patients and 57% of Parkinson’s disease patients were classified as responders. Medication dosage did not differ significantly between groups.60 Bonelli et al61 found a greater than 20% improvement in UPDRS scores in 50% of DLB patients compared to 65% of Parkinson’s disease with dementia patients and 90% of Parkinson’s disease patients. Adverse events were not reported. Goldman et al62 reported improvement in motor symptoms in 6 of 19 DLB patients. Of the responders, 2 developed worsening hallucinations. Thus, only 22% of the cohort was able to achieve improvement in motor symptoms without exacerbation of psychosis.
Further research is required to determine the pathophysiology underlying these results, but they suggest a fundamental difference in disease process. Hypotheses put forward to explain these differences include intrinsic α-synuclein pathology in the striatal system, loss of dopamine receptors in the caudate and putamen, and loss of dopamine receptors on corticostriatal projections.60 Other antiparkinsonian medications besides l-dopa, including selegiline, amantadine, catechol-O-methyl transferase inhibitors, anticholinergics, and dopamine agonists, should be used with caution in view of concerns about inducing confusion and psychosis.62
Thus, the decision to use dopaminergic agents must be undertaken with caution. Clinicians are advised to start with low doses and titrate slowly, with constant monitoring for worsening cognition and psychosis.
Management of orthostatic hypotension consists of elimination of offending medications, increased water intake, and use of ankle pumps and compression stockings. If these measures are ineffective, fludrocortisone and midodrine may be tried.63,64
A summary of clinical studies involving treatment is presented in Table 2.
Nonpharmacologic Treatment
Given the limited efficacy of medications for this disease and the substantial burden caregivers encounter, it is important to encourage families to use nonpharmacologic treatment strategies. These include using walkers and canes to minimize the risk of falls as well as using ramps instead of stairs and installing grab bars.63 Physical therapy and occupational therapy are appropriate referrals to aid in maintaining mobility and preventing falls.63 Keeping patients away from heavy machinery to prevent injury and removing driving privileges are important components of care. Finally, caregivers should be given education and monitored for caregiver burnout, as DLB patients often require around-the-clock care as the disease progresses. Providers should initiate conversations of nursing home and extended care facility placement with families. Additionally, code status and conversations regarding end-of-life issues need to take place early in the disease process while the patient can still participate.
PROGNOSIS
The prognosis of DLB is poor. Jellinger et al65 found a median time of survival of 5 years from symptom onset in a series of 243 autopsy-confirmed cases of DLB and Parkinson’s disease. Williams et al66 found a median survival of 7.3 years after onset of dementia. Relative to Alzheimer’s disease, DLB is associated with increased overall mortality and reduced survival after onset of dementia in some but not all studies. The time to institutionalization and rate of cognitive decline is similar between the 2 illnesses. These effects are independent of baseline cognitive status, comorbidities, and medications. Male gender has been reported to be a risk factor for increased mortality in some but not all studies.65-68
CONCLUSION
Dementia with Lewy bodies is a common cause of dementia in the elderly. It is a diagnosis that is often missed because of the difficulty in obtaining core symptoms. Often, patients are not forthcoming with psychiatric symptoms and may only endorse cognitive impairment or motor symptoms, leading to a misdiagnosis of Alzheimer’s disease or Parkinson’s disease. Additionally, patients presenting with psychotic symptoms may be misdiagnosed with a primary psychotic disorder. Accurate diagnosis is critical in order to avoid using neuroleptics and antiparkinsonian agents in this population. Patients presenting with motor symptoms or repeated falls should be carefully questioned about cognitive and psychiatric symptoms. Similarly, older patients presenting with new-onset psychotic symptoms should have a thorough evaluation, including neuropsychiatric testing, before their complaints are attributed to a late-onset primary psychotic disorder. Additional history should be obtained from family members or caregivers, as patients may not have insight into their symptoms. Education should be provided to family members about the overall prognosis and nature of psychotic symptoms, as these can be particularly distressing to caregivers.
Drug names: amantadine (Symmetrel and others), clonazepam (Klonopin and others), clozapine (Clozaril, FazaClo, and others), donepezil (Aricept and others), galantamine (Razadyne), memantine (Namenda), midodrine (Proamatine and others), paroxetine (Paxil, Pexeva, and others), quetiapine (Seroquel), rivastigmine (Exelon and others), selegiline (Eldepryl and others), tacrine (Cognex).
Author affiliations: Department of Internal Medicine, Division Medicine/Psychiatry (Dr Zupancic) and Department of Psychiatry (Drs Mahajan and Handa), Southern Illinois University School of Medicine, Springfield.
Potential conflicts of interest: None reported.
Funding/support: None reported.
REFERENCES
1. Aarsland D, Rongve A, Nore SP, et al. Frequency and case identification of dementia with Lewy bodies using the revised consensus criteria. Dement Geriatr Cogn Disord. 2008;26(5):445-452. PubMed doi:10.1159/000165917
2. Rahkonen T, Eloniemi-Sulkava U, Rissanen S, et al. Dementia with Lewy bodies according to the consensus criteria in a general population aged 75 years or older. J Neurol Neurosurg Psychiatry. 2003;74(6):720-724. PubMed doi:10.1136/jnnp.74.6.720
3. McKeith IG, Dickson DW, Lowe J, et al; Consortium on DLB. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology. 2005;65(12):1863-1872. PubMed doi:10.1212/01.wnl.0000187889.17253.b1
4. McKeith IG, Galasko D, Kosaka K, et al. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the consortium on DLB international workshop. Neurology. 1996;47(5):1113-1124. PubMed
5. Nelson PT, Jicha GA, Kryscio RJ, et al. Low sensitivity in clinical diagnoses of dementia with Lewy bodies. J Neurol. 2010;257(3):359-366. PubMed doi:10.1007/s00415-009-5324-y
6. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
7. Mori E, Shimomura T, Fujimori M, et al. Visuoperceptual impairment in dementia with Lewy bodies. Arch Neurol. 2000;57(4):489-493. PubMed doi:10.1001/archneur.57.4.489
8. Nervi A, Reitz C, Tang MX, et al. Comparison of clinical manifestations in Alzheimer disease and dementia with Lewy bodies. Arch Neurol. 2008;65(12):1634-1639. PubMed doi:10.1001/archneur.65.12.1634
9. Oda H, Yamamoto Y, Maeda K. The neuropsychological profile in dementia with Lewy bodies and Alzheimer’s disease. Int J Geriatr Psychiatry. 2009;24(2):125-131. PubMed doi:10.1002/gps.2078
10. Metzler-Baddeley C. A review of cognitive impairments in dementia with Lewy bodies relative to Alzheimer’s disease and Parkinson’s disease with dementia. Cortex. 2007;43(5):583-600. PubMed doi:10.1016/S0010-9452(08)70489-1
11. Calderon J, Perry RJ, Erzinclioglu SW, et al. Perception, attention, and working memory are disproportionately impaired in dementia with Lewy bodies compared with Alzheimer’s disease. J Neurol Neurosurg Psychiatry. 2001;70(2):157-164. PubMed doi:10.1136/jnnp.70.2.157
12. Collerton D, Burn D, McKeith I, et al. Systematic review and meta-analysis show that dementia with Lewy bodies is a visual-perceptual and attentional-executive dementia. Dement Geriatr Cogn Disord. 2003;16(4):229-237. PubMed doi:10.1159/000072807
13. Crowell TA, Luis CA, Cox DE, et al. Neuropsychological comparison of Alzheimer’s disease and dementia with Lewy bodies. Dement Geriatr Cogn Disord. 2007;23(2):120-125. PubMed doi:10.1159/000097791
14. Stavitsky K, Brickman AM, Scarmeas N, et al. The progression of cognition, psychiatric symptoms, and functional abilities in dementia with Lewy bodies and Alzheimer disease. Arch Neurol. 2006;63(10):1450-1456. PubMed doi:10.1001/archneur.63.10.1450
15. Mondon K, Gochard A, Marqué A, et al. Visual recognition memory differentiates dementia with Lewy bodies and Parkinson’s disease dementia. J Neurol Neurosurg Psychiatry. 2007;78(7):738-741. PubMed doi:10.1136/jnnp.2006.104257
16. Serby M, Samuels SC. Diagnostic criteria for dementia with Lewy bodies reconsidered. Am J Geriatr Psychiatry. 2001;9(3):212-216. PubMed
17. Del Ser T, McKeith I, Anand R, et al. Dementia with Lewy bodies: findings from an international multicentre study. Int J Geriatr Psychiatry. 2000;15(11):1034-1045. PubMed doi:10.1002/1099-1166(200011)15:11<1034::AID-GPS231>3.0.CO;2-5
18. Bradshaw J, Saling M, Hopwood M, et al. Fluctuating cognition in dementia with Lewy bodies and Alzheimer’s disease is qualitatively distinct. J Neurol Neurosurg Psychiatry. 2004;75(3):382-387. PubMed doi:10.1136/jnnp.2002.002576
19. Walker MP, Ayre GA, Cummings JL, et al. Quantifying fluctuation in dementia with Lewy bodies, Alzheimer’s disease, and vascular dementia. Neurology. 2000;54(8):1616-1625. PubMed
20. Iranzo A, Santamaria J, Tolosa E. The clinical and pathophysiological relevance of REM sleep behavior disorder in neurodegenerative diseases. Sleep Med Rev. 2009;13(6):385-401. PubMed doi:10.1016/j.smrv.2008.11.003
21. Claassen DO, Josephs KA, Ahlskog JE, et al. REM sleep behavior disorder preceding other aspects of synucleinopathies by up to half a century. Neurology. 2010;75(6):494-499. PubMed doi:10.1212/WNL.0b013e3181ec7fac
22. Iranzo A, Molinuevo JL, Santamar×a J, et al. Rapid-eye-movement sleep behaviour disorder as an early marker for a neurodegenerative disorder: a descriptive study. Lancet Neurol. 2006;5(7):572-577. PubMed doi:10.1016/S1474-4422(06)70476-8
23. Boeve BF, Silber MH, Ferman TJ, et al. Association of REM sleep behavior disorder and neurodegenerative disease may reflect an underlying synucleinopathy. Mov Disord. 2001;16(4):622-630. PubMed doi:10.1002/mds.1120
24. Postuma RB, Gagnon JF, Vendette M, et al. Quantifying the risk of neurodegenerative disease in idiopathic REM sleep behavior disorder. Neurology. 2009;72(15):1296-1300. PubMed
25. McKeith I, Fairbairn A, Perry R, et al. Neuroleptic sensitivity in patients with senile dementia of Lewy body type. BMJ. 1992;305(6855):673-678. PubMed doi:10.1136/bmj.305.6855.673
26. Ballard C, O’ Brien J, Gray A, et al. Attention and fluctuating attention in patients with dementia with Lewy bodies and Alzheimer disease. Arch Neurol. 2001;58(6):977-982. PubMed doi:10.1001/archneur.58.6.977
27. Borroni B, Agosti C, Padovani A. Behavioral and psychological symptoms in dementia with Lewy-bodies (DLB): frequency and relationship with disease severity and motor impairment. Arch Gerontol Geriatr. 2008;46(1):101-106. PubMed doi:10.1016/j.archger.2007.03.003
28. Knopman DS, DeKosky ST, Cummings JL, et al; Report of the Quality Standards Subcommittee of the American Academy of Neurology. Practice parameter: diagnosis of dementia (an evidence-based review). Neurology. 2001;56(9):1143-1153. PubMed
29. Watson R, Blamire AM, O’ Brien JT. Magnetic resonance imaging in Lewy body dementias. Dement Geriatr Cogn Disord. 2009;28(6):493-506. PubMed doi:10.1159/000264614
30. Walker RW, Walker Z. Dopamine transporter single photon emission computerized tomography in the diagnosis of dementia with Lewy bodies. Mov Disord. 2009;24(suppl 2):S754-S759. PubMed doi:10.1002/mds.22591
31. McKeith I, O’ Brien J, Walker Z, et al; DLB Study Group. Sensitivity and specificity of dopamine transporter imaging with 123I-FP-CIT SPECT in dementia with Lewy bodies: a phase III, multicentre study. Lancet Neurol. 2007;6(4):305-313. PubMed doi:10.1016/S1474-4422(07)70057-1
32. Walker Z, Jaros E, Walker RW, et al. Dementia with Lewy bodies: a comparison of clinical diagnosis, FP-CIT single photon emission computed tomography imaging and autopsy. J Neurol Neurosurg Psychiatry. 2007;78(11):1176-1181. PubMed doi:10.1136/jnnp.2006.110122
33. O’ Brien JT, Colloby S, Fenwick J, et al. Dopamine transporter loss visualized with FP-CIT SPECT in the differential diagnosis of dementia with Lewy bodies. Arch Neurol. 2004;61(6):919-925. PubMed doi:10.1001/archneur.61.6.919
34. Lim SM, Katsifis A, Villemagne VL, et al. The 18F-FDG PET cingulate island sign and comparison to 123I-beta-CIT SPECT for diagnosis of dementia with Lewy bodies. J Nucl Med. 2009;50(10):1638-1645. PubMed doi:10.2967/jnumed.109.065870
35. Bonanni L, Thomas A, Tiraboschi P, et al. EEG comparisons in early Alzheimer’s disease, dementia with Lewy bodies and Parkinson’s disease with dementia patients with a 2-year follow-up. Brain. 2008;131(Pt 3):690-705. PubMed doi:10.1093/brain/awm322
36. Bibl M, Esselmann H, Lewczuk P, et al. Combined analysis of CSF tau, Aβ42, Aβ1-42% and Aβ1-40% in Alzheimer’s disease: dementia with Lewy bodies and Parkinson’s disease dementia. Int J Alzheimers Dis. 2010; pii: 761571. doi:10.4061/2010/761571 PubMed
37. Weisman D, McKeith I. Dementia with Lewy bodies. Semin Neurol. 2007;27(1):42-47. PubMed doi:10.1055/s-2006-956754
38. Jellinger KA. Formation and development of Lewy pathology: a critical update. J Neurol. 2009;256(suppl 3):270-279. PubMed doi:10.1007/s00415-009-5243-y
39. Hansen LA, Samuel W. Criteria for Alzheimer’s disease and the nosology of dementia with Lewy bodies. Neurology. 1997;48(1):126-132. PubMed
40. Langlais PJ, Thal L, Hansen L, et al. Neurotransmitters in basal ganglia and cortex of Alzheimer’s disease with and without Lewy bodies. Neurology. 1993;43(10):1927-1934. PubMed
41. Perry EK, Haroutunian V, Davis KL, et al. Neocortical cholinergic activities differentiate Lewy body dementia from classical Alzheimer’s disease. Neuroreport. 1994;5(7):747-749. PubMed doi:10.1097/00001756-199403000-00002
42. Ballard C, Piggott M, Johnson M, et al. Delusions associated with elevated muscarinic binding in dementia with Lewy bodies. Ann Neurol. 2000;48(6):868-876. PubMed doi:10.1002/1531-8249(200012)48:6<868::AID-ANA7>3.0.CO;2-0
43. Perry EK, McKeith I, Thompson P, et al. Topography, extent, and clinical relevance of neurochemical deficits in dementia of Lewy body type, Parkinson’s disease, and Alzheimer’s disease. Ann N Y Acad Sci. 1991;640:197-202. PubMed
44. Dalfó E, Albasanz JL, Martin M, et al. Abnormal metabotropic glutamate receptor expression and signaling in the cerebral cortex in diffuse Lewy body disease is associated with irregular alpha-synuclein/phospholipase C (PLCbeta1) interactions. Brain Pathol. 2004;14(4):388-398. PubMed doi:10.1111/j.1750-3639.2004.tb00082.x
45. Lippa CF, Johnson R, Smith TW. The medial temporal lobe in dementia with Lewy bodies: a comparative study with Alzheimer’s disease. Ann Neurol. 1998;43(1):102-106. PubMed doi:10.1002/ana.410430117
46. McKeith IG, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet. 2000;356(9247):2031-2036. PubMed doi:10.1016/S0140-6736(00)03399-7
47. Wesnes KA, McKeith IG, Ferrara R, et al. Effects of rivastigmine on cognitive function in dementia with Lewy bodies: a randomised placebo-controlled international study using the Cognitive Drug Research computerised assessment system. Dement Geriatr Cogn Disord. 2002;13(3):183-192. PubMed doi:10.1159/000048651
48. Grace J, Daniel S, Stevens T, et al. Long-term use of rivastigmine in patients with dementia with Lewy bodies: an open-label trial. Int Psychogeriatr. 2001;13(2):199-205. PubMed doi:10.1017/S104161020100758X
49. Rozzini L, Chilovi BV, Bertoletti E, et al. Cognitive and psychopathologic response to rivastigmine in dementia with Lewy bodies compared to Alzheimer’s disease: a case control study. Am J Alzheimers Dis Other Demen. 2007;22(1):42-47. doi:10.1177/1533317506297517 PubMed
50. Ballard CG, Chalmers KA, Todd C, et al. Cholinesterase inhibitors reduce cortical Aβ in dementia with Lewy bodies. Neurology. 2007;68(20):1726-1729. PubMed doi:10.1212/01.wnl.0000261920.03297.64
51. Edwards K, Royall D, Hershey L, et al. Efficacy and safety of galantamine in patients with dementia with Lewy bodies: a 24-week open-label study. Dement Geriatr Cogn Disord. 2007;23(6):401-405. PubMed doi:10.1159/000101512
52. Shea C, MacKnight C, Rockwood K. Donepezil for treatment of dementia with Lewy bodies: a case series of nine patients. Int Psychogeriatr. 1998;10(3):229-238. PubMed doi:10.1017/S1041610298005341
53. Thomas AJ, Burn DJ, Rowan EN, et al. A comparison of the efficacy of donepezil in Parkinson’s disease with dementia and dementia with Lewy bodies. Int J Geriatr Psychiatry. 2005;20(10):938-944. PubMed doi:10.1002/gps.1381
54. Bhasin M, Rowan E, Edwards K, et al. Cholinesterase inhibitors in dementia with Lewy bodies: a comparative analysis. Int J Geriatr Psychiatry. 2007;22(9):890-895. PubMed doi:10.1002/gps.1759
55. Emre M, Tsolaki M, Bonuccelli U, et al; 11018 Study Investigators. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9(10):969-977. PubMed doi:10.1016/S1474-4422(10)70194-0
56. Aarsland D, Ballard C, Walker Z, et al. Memantine in patients with Parkinson’s disease dementia or dementia with Lewy bodies: a double-blind, placebo-controlled, multicentre trial. Lancet Neurol. 2009;8(7):613-618. PubMed doi:10.1016/S1474-4422(09)70146-2
57. Levin OS, Batukaeva LA, Smolentseva IG, et al. Efficacy and safety of memantine in Lewy body dementia. Neurosci Behav Physiol. 2009;39(6):597-604. PubMed doi:10.1007/s11055-009-9167-x
58. Boeve BF, Silber MH, Ferman TJ. Melatonin for treatment of REM sleep behavior disorder in neurologic disorders: results in 14 patients. Sleep Med. 2003;4(4):281-284. PubMed doi:10.1016/S1389-9457(03)00072-8
59. Kunz D, Mahlberg R. A two-part, double-blind, placebo-controlled trial of exogenous melatonin in REM sleep behaviour disorder. J Sleep Res. 2010;19(4):591-596. PubMed doi:10.1111/j.1365-2869.2010.00848.x
60. Molloy S, McKeith IG, O’ Brien JT, et al. The role of levodopa in the management of dementia with Lewy bodies. J Neurol Neurosurg Psychiatry. 2005;76(9):1200-1203. PubMed doi:10.1136/jnnp.2004.052332
61. Bonelli SB, Ransmayr G, Steffelbauer M, et al. l-dopa responsiveness in dementia with Lewy bodies, Parkinson disease with and without dementia. Neurology. 2004;63(2):376-378. PubMed
62. Goldman JG, Goetz CG, Brandabur M, et al. Effects of dopaminergic medications on psychosis and motor function in dementia with Lewy bodies. Mov Disord. 2008;23(15):2248-2250. PubMed doi:10.1002/mds.22322
63. Tinetti ME, Baker DI, McAvay G, et al. A multifactorial intervention to reduce the risk of falling among elderly people living in the community. N Engl J Med. 1994;331(13):821-827. PubMed doi:10.1056/NEJM199409293311301
64. Kearney F, Moore A. Pharmacological options in the management of orthostatic hypotension in older adults. Expert Rev Cardiovasc Ther. 2009;7(11):1395-1400. PubMed doi:10.1586/erc.09.130
65. Jellinger KA, Wenning GK, Seppi K. Predictors of survival in dementia with Lewy bodies and Parkinson dementia. Neurodegener Dis. 2007;4(6):428-430. PubMed doi:10.1159/000107703
66. Williams MM, Xiong C, Morris JC, et al. Survival and mortality differences between dementia with Lewy bodies vs Alzheimer disease. Neurology. 2006;67(11):1935-1941. PubMed doi:10.1212/01.wnl.0000247041.63081.98
67. Hanyu H, Sato T, Hirao K, et al. Differences in clinical course between dementia with Lewy bodies and Alzheimer’s disease. Eur J Neurol. 2009;16(2):212-217. PubMed doi:10.1111/j.1468-1331.2008.02388.x
68. Walker Z, Allen RL, Shergill S, et al. Three years survival in patients with a clinical diagnosis of dementia with Lewy bodies. Int J Geriatr Psychiatry. 2000;15(3):267-273. PubMed doi:10.1002/(SICI)1099-1166(200003)15:3<267::AID-GPS107>3.0.CO;2-7