Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
CASE CONFERENCE
The Banner Alzheimer’s Institute Case Conference is a weekly event in which physicians and staff discuss challenging and/or teaching cases of patients seen at the Institute’s Memory Disorders Clinic. These conferences are attended by a multidisciplinary group that includes Banner Alzheimer’s Institute dementia specialists, community physicians (internal medicine, family medicine, and radiology), physician assistants, social workers, nurses, medical students, residents, and fellows.
BANNER ALZHEIMER’ S INSTITUTE
The Banner Alzheimer’s Institute located in Phoenix, Arizona, has an unusually ambitious mission: to end Alzheimer’s disease without losing a generation, set a new standard of care for patients and families, and forge a model of collaboration in biomedical research. The Institute provides high-level care and treatment for patients affected by Alzheimer’s disease, dementia, and related disorders. In addition, the Institute offers extensive support services for families and many unique and rewarding research opportunities.
Prim Care Companion CNS Disord 2012;14(3):doi:10.4088/PCC.12alz01398
© Copyright 2012 Physicians Postgraduate Press, Inc.
Received: March 29, 2012; accepted March 29, 2012.
Published online: June 28, 2012.
AUTHORS
Anna D. Burke, MD, is a geriatric psychiatrist and dementia specialist at the Memory Disorders Clinic of Banner Alzheimer’s Institute.
Roy Yaari, MD, MAS, a neurologist, is associate director of the Memory Disorders Clinic of Banner Alzheimer’s Institute and a clinical professor of neurology at the College of Medicine, University of Arizona, Tucson.
Pierre N. Tariot, MD, a geriatric psychiatrist, is director of Banner Alzheimer’s Institute and a research professor of psychiatry at the College of Medicine, University of Arizona, Tucson.
Jan Dougherty, RN, MS, is director of Family and Community Services at Banner Alzheimer’s Institute.
Adam S. Fleisher, MD, MAS, is associate director of Brain Imaging at the Banner Alzheimer’s Institute, a neurologist at the Institute’s Memory Disorders Clinic, and an associate professor in the Department of Neurosciences at the University of California, San Diego.
Helle Brand, PA, is a physician assistant at the Memory Disorders Clinic of Banner Alzheimer’s Institute.
Corresponding author: Anna D. Burke, MD, Banner Alzheimer’s Institute, 901 E. Willetta St, Phoenix, AZ 85006 ([email protected]).
CME Background
Original material is selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME activities on a variety of topics from volume to volume. This special series of case reports about dementia was deemed valuable for educational purposes by the Publisher, Editor in Chief, and CME Institute Staff. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the material and go to PrimaryCareCompanion.com to complete the Posttest and Evaluation online.
CME Objective
After studying this case, you should be able to:
- Evaluate and treat an elderly outpatient with recent delirium, parkinsonian symptoms, cognitive fluctuation, and visual hallucinations
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1.0 hour of Category I credit for completing this program.
Date of Original Release/Review
This educational activity is eligible for AMA PRA Category 1 Creditâ„¢ through June 30, 2015. The latest review of this material was June 2012.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Larry Culpepper, MD, MPH, Editor in Chief, has been a consultant for AstraZeneca, Pfizer, Takeda, and Trovis; and has been a member of the speakers/advisory boards for Forest and Merck. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears at the end of the article.
HISTORY OF PRESENT ILLNESS
Dr A, an 81-year-old married right-handed man, presented with his wife to the Banner Alzheimer’s Institute in 2010 due to a 2-year history of memory loss and fluctuating periods of confusion. According to his wife, symptoms initially began in 2005 after Dr A underwent cataract surgery. She noted that Dr A reported difficulty reading. He described words as “falling off the page.” Dr A also had difficulty comprehending what he was reading at the time, which resulted in significant frustration and irritability. His wife also noted increased difficulty with decision making, and Dr A deferred more decisions to her.
After the cataract surgery, Dr A continued to work as a physician. Concerned about her husband’s well-being and his reputation, his wife delicately asked his colleagues to watch for callbacks at work. She reported that no callbacks or problems with his work were noted by colleagues. Dr A decided to retire in 2008 due to his “vision problems” and increasing fatigue.
In the fall of 2008, the couple went on a cruise, during which Dr A’s wife noticed increasing confusion that fluctuated over the course of the day. Worsening confusion was noted in the evenings as well as upon awakening from sleep. Dr A had difficulty with simple tasks such as wiping his nose when instructed to do so. He displayed geographic disorientation and had problems finding his way back to his cabin. His wife also noted that Dr A was “slow.” He had more difficulty answering questions in a timely manner and at times appeared to be more tangential and verbose. His speech was softer and “mumbling.” In addition to cognitive slowing, Dr A began to shuffle when he walked and appeared more unsteady on his feet. No falls or injuries occurred.
In mid-2009, Dr A was hospitalized due to pneumonia and developed delirium. He could not remember how to perform simple tasks such as placing his hearing aids in his ears. Visual hallucinations of “shadow people” in his room were also present. Dr A spent 1 week in the hospital and then 2 weeks in rehabilitation, during which time, the delirium slowly resolved.
Over the course of the past year, Dr A’s wife began to notice gradually worsening short-term memory deficits including increasing repetition during conversations and questioning, misplacing items around the house, forgetting recent events such as family gatherings, and more difficulty with managing the calendar and appointments. As a result of his memory impairment, Dr A began to “fill in” gaps in stories with imaginary events that had never occurred. Dr A also became more quiet and withdrawn. He was content with spending much of the day napping or sitting in a chair. He had difficulty initiating activities and appeared less interested in hobbies that he had previously enjoyed.
Dr A had more difficulty with instrumental and basic activities of daily living. He was no longer able to appropriately use the telephone, TV remote, and microwave. He became confused about how to take his medications, and his wife took over this task. He began to take much longer to pay bills and write out checks. His wife noticed that Dr A was voiding more checks and that his writing was becoming smaller and more illegible. A bilateral resting tremor developed. Dr A also became more lax in his personal hygiene and grooming. His spouse attributed this behavior to his worsening physical symptoms including prominent psychomotor slowing and upper extremity tremor. Dr A’s wife also began choosing clothing for him, as he would wear the same dirty clothing for several days.
Dr A continued to experience fluctuating periods of confusion, during which he displayed bizarre behaviors such as looking for his sweater in a recycling bin or accidentally going to the bathroom in a closet. He was more suspicious of other’s intents. On occasion, he would describe seeing “shadow people” in the room. No auditory hallucinations were present, and the visual hallucinations did not appear to result in significant distress. No behavioral disturbances were noted by his wife.
Dr A’s sleep was impaired due to restlessness and thrashing during sleep. Dr A would frequently act out his dreams during the night and would become confused upon awakening, believing that his dreams were real.
Approximately 1 week prior to evaluation at Banner Alzheimer’s Institute, Dr A was briefly hospitalized after an episode of atrial fibrillation. Over the course of the next week, he became increasingly sedated, confused, and quiet. While in the hospital, Dr A was diagnosed with Alzheimer’s disease by a consulting physician and prescribed memantine, which he had not yet started.
PAST MEDICAL HISTORY
Dr A’s past medical history included treatment for tuberculosis in 1950, atrial fibrillation since 1986, right bundle branch block, hypothyroidism, hiatal hernia, glaucoma, and gastroesophageal reflux disease.
ALLERGIES
Dr A was allergic to tetracycline and quinaglute. He also reported an episode of atrial fibrillation after administration of fluticasone propionate. Transient hyponatremia resulted from use of escitalopram, which had been prescribed to treat symptoms of apathy.
MEDICATIONS
Dr A’s current medications included chlorhexidine oral mouth wash, flecainide acetate, levothyroxine, omeprazole, warfarin, latanoprost eye drops, azithromycin and amoxicillin/clavulanate extended release for a recently diagnosed upper respiratory tract infection, super B complex, vitamin C, folic acid, and extra strength acetaminophen. As noted previously, Dr A was recently started on memantine but had not received any doses of the medication as of yet.
SOCIAL HISTORY
Dr A had a 24-year education history. He was a physician with a 47-year work history and was retired since 2008. He resided in his own home with his spouse.
SUBSTANCE ABUSE HISTORY
Dr A denied any history of illicit drug use. He smoked 3 to 4 packs of cigarettes per day for approximately 30 years prior to quitting in 1975. He drank approximately 20 drinks per week before stopping in the fall of 2004.
FAMILY HISTORY
There was a family history of dementia in Dr A’s maternal uncle; age at onset was unclear. His mother suffered a stroke and developed parkinsonian symptoms thereafter. There was no history of mental illness in the family.
Review of Systems
A complete review of systems was performed and revealed no history of head injuries or toxic exposures. Apathy, shuffling gait, difficulty with posture, orthostatic hypotension, constipation, and tremor were present on review.
Based on the information so far, do you think a dementia is present?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Yes | 100% |
| B. No | 0% |
| C. Not enough information | 0% |
All of the clinicians present felt that criteria for a dementia were met since Dr A displayed a gradually progressive cognitive decline that was significantly impairing his functional abilities. The argument was made that Dr A’s cognitive status during the time of the evaluation could have been adversely affected by his recent medical issues, and a delirium may have been present. The presence of a delirium would be suggestive of an underlying cognitive impairment. Research indicates that individuals suffering from an underlying neurodegenerative disorder are more prone to developing delirium when they encounter additional physical stress such as infection, illness, or surgery (Fick et al, 2002).
The DSM-IV defines dementia as multiple cognitive deficits that include memory impairment and at least 1 of the following cognitive disturbances: aphasia, apraxia, agnosia, or a disturbance in executive functioning. The cognitive deficits must be sufficiently severe to cause impairment in social or occupational functioning and must represent a decline from a previously higher level of functioning. A diagnosis of dementia should not be made if the cognitive deficits occur exclusively during the course of a delirium (American Psychiatric Association, 2000).
Delirium is an acute change in mental status, developing over hours to days, presenting with the following symptoms:
- Clouding of state of consciousness;
- Disorganized thinking;
- Disorientation to time/place;
- Inattention or distraction;
- Drowsiness, disturbed sleep;
- Increase or decrease in psychomotor activity;
- Fluctuation of signs and symptoms; and
- Improvement/normalization of mental status after treatment of the underlying condition.
Based on the information so far, what would you expect to see on the neurologic examination?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Normal | 0% |
| B. Objective nonfocal neurologic findings | 90% |
| (including frontal release signs) | |
| C. Focal neurologic findings | 10% |
Most clinicians answered B and felt that Dr A would display no evidence of any focal neurologic findings. However, they did expect that frontal release signs and parkinsonian symptoms would be present. Due to Dr A’s cardiac history and increased risk of stroke, a few clinicians who answered C felt that focal neurologic findings such as mild hemiparesis or unilateral brisk deep tendon reflexes indicating an infarct might be present on examination.
Different dementias may be associated with various physical examination findings. However, most often the physical examination is normal in the early stages. Some subtle general findings can include frontal release signs such as a positive snout, glabellar, or palmomental reflex (Links et al, 2010).
NEUROLOGIC EXAMINATION
Cranial nerves II-XII were intact except for broken smooth pursuits and bilateral decreased hearing to finger rub. Dr A displayed speech and motor apraxia. Deep tendon reflexes were symmetrically hypoactive throughout and absent in the ankles. His gait was shuffling, slow, and festinating with en bloc turning. His arm swing was decreased bilaterally. Increased tone but no cogwheeling was present on examination of the upper extremities. A bilateral upper extremity resting tremor was also noted; it was more pronounced in the right upper extremity. Otherwise, the neurologic examination was unremarkable.
Apraxia is characterized by loss of the ability to execute or carry out learned purposeful movements, despite having the desire and the physical ability to perform the movements. Apraxia is a disorder of motor planning, which may be acquired or developmental, but may not be caused by incoordination, sensory loss, or failure to comprehend simple commands.
PHYSICAL EXAMINATION
The physical examination was remarkable for kyphosis, masked facies, hypophonic speech, and psychomotor retardation. The cardiac examination revealed an irregular rate and rhythm. Otherwise, the physical examination was largely unremarkable.
MENTAL STATUS EXAMINATION
Dr A was a well-groomed elderly white man appearing to be in no acute distress. He displayed significant psychomotor retardation as well as masked facies. Decreased blink was present. Dr A was pleasant and cooperative during the examination. His affect was blunted. Dr A’s thought process was confused, and he had difficulty answering simple questions. There was no evidence of any suicidal or homicidal ideations. No auditory hallucinations were present at the time of the visit, but Dr A did report occasionally seeing shadow people. Mild paranoid ideations were noted on history. His judgment and insight appeared impaired. Dr A was oriented to person but not to place or time. His recent memory was impaired, as were attention and concentration. Fund of knowledge was decreased for Dr A’s age and education level.
Based on the information so far, what would you expect the Mini-Mental State Examination (MMSE) score to be?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 26-30 | 90% |
| B. 21-25 | 10% |
| C. 16-20 | 0% |
| D. 11-15 | 0% |
| E. ≤ 10 | 0% |
The majority of clinicians answered A and felt that Dr A’s high education status and high cognitive reserve would impact his MMSE (Folstein et al, 1975) scores positively. Some clinicians who answered B indicated that, despite Dr A’s high cognitive reserve, his current clinical impairments were prominent and would translate to lower MMSE scores. A MMSE performed at the time of the visit revealed a total score of 18 out of 30, with impairments in orientation, delayed recall, drawing, comprehension, and attention.
Cognitive reserve is the concept that has been developed to explain how it is that some elderly people with extensive neuropathology associated with dementia show little in the way of cognitive decline and is intimately related to cortical plasticity. Cognitive reserve appears from epidemiologic studies to be positively influenced by extended years of education, physical exercise, and social and intellectual pursuits in middle and late life (Stern et al, 1999).
Based on the information so far, what would you expect the Montreal Cognitive Assessment (MoCA) score to be?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 26-30 | 0% |
| B. 21-25 | 0% |
| C. 16-20 | 50% |
| D. 11-15 | 50% |
| E. ≤ 10 | 0% |
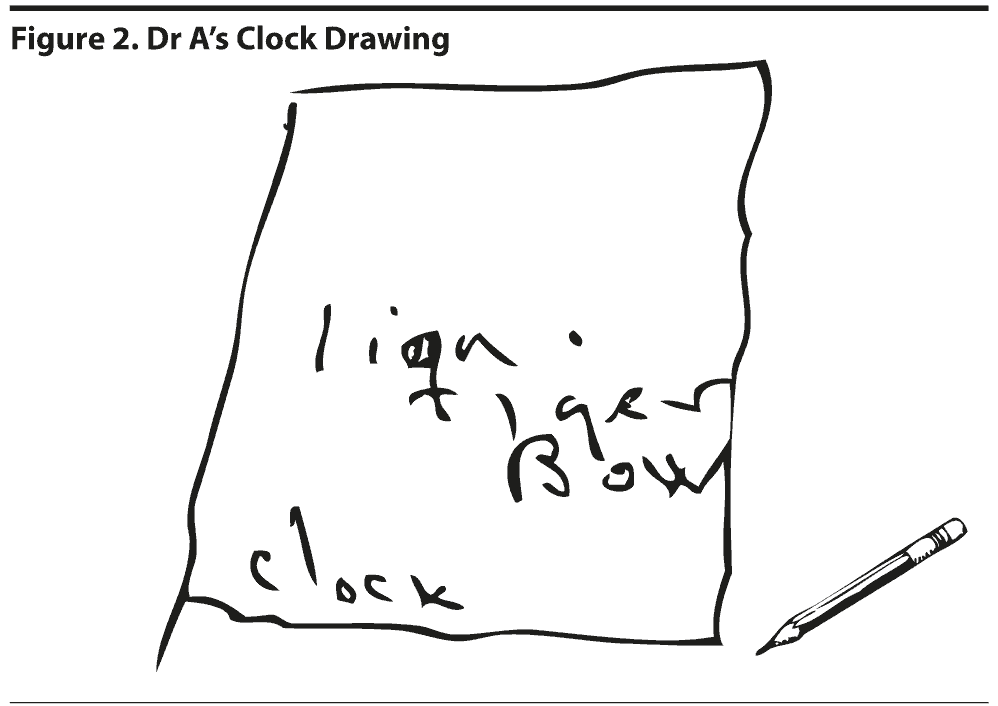
All of the clinicians present believed that the MoCA would be better able to pick up on more subtle cognitive deficits, particularly in areas of visuospatial abilities and executive function, regardless of the patient’s cognitive reserve. The MoCA has been shown to have a better sensitivity and specificity in detecting more subtle cognitive impairments, such as mild cognitive impairment, when compared to the MMSE. The MoCA is less affected by education and intellectual level than the MMSE. Nasreddine et al (2005) showed that the MMSE had a sensitivity of 18% to detect mild cognitive impairment, whereas the MoCA detected 90% of subjects with mild cognitive impairment. In the mild Alzheimer’s disease group, the MMSE had a sensitivity of 78%, whereas the MoCA detected 100% of cases. Specificity was excellent for both the MMSE and MoCA (100% and 87%, respectively; Nasreddine et al, 2005). A MoCA test revealed a total score of 12 out of 30, with Dr A displaying impairments in the areas of executive function, visuospatial abilities, attention, language abilities, delayed recall, and orientation (Figure 1). The clock drawing was significantly impaired, with Dr A being unable to draw even the outline of the clock and instead drawing a box (Figure 2).
The clock drawing is frequently impaired in various types of dementia. Patients with dementia with Lewy bodies were found to be more likely to make conceptual errors than patients with Alzheimer’s disease and Parkinson’s disease. Patients with Parkinson’s disease and dementia with Lewy bodies displayed more planning errors than patients with Alzheimer’s disease (Cahn-Weiner et al, 2003).
Based on the information so far, do you think this is dementia?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Yes | 100% |
| B. No | 0% |
| C. Can’ t determine at this time | 0% |
All of the clinicians felt that the results of cognitive screening tests along with the clinical history were compelling evidence of a dementia.
IMPRESSION
Dr A presented for evaluation of cognitive impairment of approximately 2 plus years’ duration. The patient’s clinical course of insidious onset of short-term memory deficits and gradual decline was complicated by numerous medical conditions causing acute decompensations as a result of delirium. The most recent episode occurred approximately 1 week prior to his evaluation at Banner Alzheimer’s Institute when Dr A suffered atrial fibrillation and required hospitalization. He was also suffering from an upper respiratory infection at the time of the evaluation. The clinician felt that it was unclear whether Dr A’s current deficits on cognitive screening tests were impacted by this recent medical illness and possible underlying delirium. The physician felt that testing should be repeated at a later date once Dr A had recovered to clarify the etiology of the current clinical symptoms; however, an underlying neurodegenerative process appeared to be present.
Many symptoms were consistent with a Lewy body-type pathology including parkinsonian symptoms, visual hallucination, fluctuating course, and rapid eye movement (REM) sleep disturbances. However, Alzheimer’s -type pathology could not fully be ruled out. Further workup was indicated.
Rapid eye movement sleep behavior disorder is a sleep disorder (parasomnia) that involves abnormal behavior during the sleep phase with REM. The disorder is characterized by the dreamer acting out his or her dreams as a result of loss of muscle paralysis (atonia) during REM sleep. These dreams often involve kicking, screaming, punching, grabbing, and even jumping out of bed.
What should the next step be?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Neuropsychological testing | 0% |
| B. Laboratories (CBC, CMP, TSH, | 0% |
| vitamin B12 level) | |
| C. Structural brain scan | 0% |
| D. Repeat cognitive screening test at next visit | 0% |
| E. B and C | 0% |
| F. A, B, C | 0% |
| G. B, C, D | 90% |
| H. All of the above | 10% |
The majority of clinicians felt that laboratory studies to rule out metabolic and infectious causes along with structural neuroimaging to rule out intracranial pathology would be of greatest benefit. A few clinicians felt that neuropsychological testing might be beneficial in further understanding Dr A’s areas of cognitive strength and weakness and in utilizing these strengths and weaknesses to ascertain a diagnosis and treatment plan. However, the majority of clinicians would have first completed a repeat brief cognitive screening battery at the next clinic visit. The results of a brief battery would further elucidate the cognitive impact of Dr A’s recent medical problems and clarify his true cognitive baseline, provided that he remained medically stable with no recurrence of delirium.
What would you do next?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Watch and wait | 0% |
| B. Start a cholinesterase inhibitor (donepezil, | 40% |
| rivastigmine, or galantamine) | |
| C. Start the NMDA antagonist memantine | 60% |
| D. Start an antipsychotic | 0% |
| E. Stop flecainide | 0% |
Although there are currently no US Food and Drug Administration (FDA)-approved treatments for dementia with Lewy bodies, evidence from studies indicates that traditional pharmacologic options used in Alzheimer’s disease, such as acetylcholinesterase inhibitors and memantine, may be effective in treating cognitive deficits and even behavioral symptoms associated with the disease (Wild et al, 2003). Anecdotal evidence of more robust response by patients with dementia with Lewy bodies to acetylcholinesterase inhibitors when compared to patients with Alzheimer’s disease has been noted. Recent studies indicate that memantine may exhibit similar beneficial effects on global cognitive function and behavior (Emre et al, 2010).
The treating physician recommended the following:
- Obtain prior medical records including neuroimaging and laboratory studies from the patient’s primary care physician.
- Advise the patient to start memantine. The choice of memantine as opposed to another cognitive enhancer was guided by the fact that Dr A had already purchased the medication, and recent studies indicate that memantine may be of benefit on cognitive function and behavior in patients with dementia with Lewy bodies (Emre et al, 2010).
- At the next visit, repeat cognitive testing to assess for level of cognitive impairment and possible improvement of acute mental status changes.
FOLLOW-UP
Dr A presented to the Memory Disorders Clinic 5 weeks later for follow-up. His wife reported that since the last visit, his memory had slowly but gradually improved, though Dr A never returned to his baseline level. He was notably less confused during the follow-up evaluation, although he continued to display some difficulty following conversations. As per family report, continued periods of fluctuating cognition were present with no specific temporal pattern noted. Dr A remained quite forgetful, struggling with remembering names and tasks, and occasionally had difficulty completing his thoughts. Word-finding deficits were apparent.
Dr A also improved functionally, although he did not return to his baseline level. He continued to make the bed every morning, but had difficulty with other household chores such as the laundry, meal preparation, and yard work. Occasional problems using the cell phone were noted. For example, Dr A had attempted to use his cell phone as a TV remote control. Some periods of confusion regarding his ability to tend to personal hygiene such as changing his sanitary pad and undergarments or getting dressed were also noted. He appeared to tolerate memantine well, and no medication side effects were noted.
Dr A denied any previous head injuries but did report frequent falls related to impaired mobility. However, a magnetic resonance image of the brain revealed that there was focal subtle encephalomalacia involving the mid-inferior left temporal lobe, most consistent with a sequela of a prior traumatic contusion. There was also mild diffuse cerebral volume loss, most pronounced in the bitemporal and biparietal distributions. Increased ventricular sizes were seen, most likely secondary to central white matter volume loss or involution. Laboratory studies including a CBC and CMP as well as TSH and vitamin B12 levels were unremarkable.
Since the last visit, Dr A also underwent a sleep study, which revealed obstructive sleep apnea. He was started on continuous positive airway pressure.
Based on the information so far, what would you expect the follow-up MMSE score to be?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 26-30 | 5% |
| B. 21-25 | 90% |
| C. 16-20 | 5% |
| D. 11-15 | 0% |
| E. ≤ 10 | 0% |
Dr A’s repeat MMSE confirmed the pronounced improvement subjectively observed by family. At the time of the follow-up visit, he scored 24 out of 30.
Based on the information so far, what would you expect the MoCA score to be?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 26-30 | 0% |
| B. 21-25 | 0% |
| C. 16-20 | 100% |
| D. 11-15 | 0% |
| E. ≤ 10 | 0% |
The results of the MoCA test also revealed a modest improvement. Dr A scored 15 out of 30 (Figure 3).
Based on the information so far, what is the most likely diagnosis?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Dementia with Lewy bodies | 100% |
| B. Parkinson’s dementia | 0% |
| C. Frontotemporal dementia syndrome | 0% |
| D. Alzheimer’s dementia | 0% |
| E. Mild cognitive impairment | 0% |
| F. Dementia not otherwise specified | 0% |
The clinicians believed that the pattern of cognitive impairment noted on screening tests and clinical history were most consistent with a dementia with Lewy bodies. On cognitive testing, Dr A displayed prominent impairment in visuospatial abilities and executive dysfunction.
The treating physician felt that Dr A’s current pattern of cognitive deficits, including more prominent visuospatial and executive functional deficits, prominent early onset of parkinsonian symptoms, REM sleep behavior disorder, fluctuating nature of cognitive impairment, and sensitivity to medications, was most consistent with a diagnosis of dementia with Lewy bodies of moderate severity. Dr A was thought to be clearing from a delirium and appeared to be displaying an improvement in cognitive and functional abilities, although a return to baseline was unlikely.
The rivastigmine patch was started at a dose of 4.6 mg daily and was increased to 9.5 mg daily after a month. The off-label use of this medication was discussed with Dr A and his spouse. He appeared to tolerate the medication well.
Over the next several months, visual hallucinations of “old buddies” intensified. Dr A felt that these hallucinations were real and that they were his guardian angels. A thorough medical workup revealed no evidence of infections or other medical illness accounting for this increase in symptoms. Dr A suffered several minor falls due to worsening gait instability and orthostasis. No head injuries or loss of consciousness took place. His mobility was becoming increasingly impaired, and hypophonia was more notable. The hallucinations were not disturbing to Dr A. No agitation or aggression were noted. Apathy became more prominent, and Dr A began to express passive suicidal ideations with no plan or intent present.
Based on the information so far, what pharmacotherapy would you start?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Antidepressant | 70% |
| B. Atypical antipsychotic | 0% |
| C. Conventional antipsychotic | 0% |
| D. Mood stabilizer | 0% |
| E. Carbidopa/levodopa | 0% |
| F. Stimulant | 0% |
| G. None | 30% |
Symptoms of apathy and depression are quite commonly seen in dementia with Lewy bodies. Most of the clinicians present felt that due to worsening depressive symptoms and the emergence of suicidal ideations, a trial of an antidepressant was warranted. Although Dr A reported visual hallucinations, he was not distressed by them. In fact, he saw these hallucinations as comforting symbols. None of the clinicians felt that treatment with an antipsychotic was indicated. Patients with dementia with Lewy bodies tend to be more sensitive to the extrapyramidal symptoms of antipsychotics.
In 2005, an FDA warning highlighted a significant increase in mortality risk for patients with Alzheimer’s disease treated with atypical antipsychotic drugs compared with individuals receiving placebo in 17 randomized, controlled trials (US Food and Drug Administration, 2005). The warning stated, “Elderly patients with dementia-related psychosis treated with atypical antipsychotic drugs are at an increased risk of death compared to placebo. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks) in these patients revealed a risk of death in the drug-treated patients of between 1.6 to 1.7 times that seen in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (eg, heart failure, sudden death) or infectious (eg, pneumonia) in nature” (US Food and Drug Administration, 2005). Subsequent studies, mainly involving large public databases, have found similar or even higher death rates among elders receiving conventional antipsychotics, which resulted in a second broader black box warning for all antipsychotics in 2008 (US Food and Drug Administration, 2008). Despite the FDA warning, atypical antipsychotics are commonly used off-label to treat behavioral and psychotic symptoms in dementia with good effect. Currently, there are no FDA-approved treatments for behavioral and psychotic symptoms in dementia.
Based on the information so far, what nonpharmacologic treatments would you start?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Physical therapy | 0% |
| B. Speech therapy | 0% |
| C. Psychodynamic psychotherapy | 0% |
| D. Cognitive-behavioral therapy | 0% |
| E. A and B | 100% |
| F. No therapy is necessary | 0% |
The clinicians felt that due to his cognitive limitations, Dr A would be unable to participate in psychodynamic psychotherapy or cognitive-behavioral therapy in a meaningful manner. Both of these forms of psychiatric intervention are useful tools in treatment of depression and anxiety in cognitively intact individuals. However, both therapies require the ability to remember the sessions and work through problems in the time between sessions. Dr A displays impairments in short-term memory and executive dysfunction and periods of confusion, which would interfere with appropriate participation.
Physical, speech, and occupational programs are available for individuals suffering from parkinsonian syndromes. These programs, such as Lee Silverman Voice Treatment (LSVT Global, 2012), focus on “speaking loud” and “moving big.” The increased amplitude of limb and body movement (bigness) in people with parkinsonian syndromes like dementia with Lewy bodies has shown documented improvements in amplitude (trunk rotation/gait) that translate to improved speed in the upper and lower limbs, balance, and quality of life. Speech therapy stimulates the motor speech system and incorporates sensory awareness training to help individuals with Parkinson’s disease recognize that their voice is too soft, convincing them that the louder voice is within normal limits, and making them comfortable with their new louder voice.
After checking electrolyte levels due to a previous history of hyponatremia, Dr A was started on venlafaxine extended release 75 mg daily. He was also referred to the Lee Silverman Voice Treatment “Big and Loud” classes to help improve mobility, gait, balance, and speech.
DISCUSSION
In contrast to Alzheimer’s disease, which is characterized histopathologically by the presence of neurofibrillary tangles and amyloid plaques, dementia with Lewy bodies is associated with α-synuclein intracellular inclusions (Lewy bodies) in neurons in the limbic, neocortical, and subcortical regions. Diagnostic criteria for dementia with Lewy bodies include progressive cognitive decline plus at least 2 of the following symptoms: cognitive fluctuations, visual hallucinations (typically of mute animate objects appearing early in the course of the disease), or parkinsonism. Some studies also suggest the presence of prominent visuospatial deficits and executive dysfunction on cognitive testing early on in the disease. Individuals with dementia with Lewy bodies may also display more drug sensitivities, dysautonomia, and REM sleep behavior disorders than their Alzheimer’s disease counterparts.
Dementia with Lewy bodies is estimated to comprise approximately 10% of all dementias. “Pure” dementia with Lewy bodies is rarely present, and autopsy frequently reveals the co-occurrence of Alzheimer’s pathology. There are currently no FDA-approved treatments for dementia with Lewy bodies and no evidence base to direct treatment. Anecdotal evidence and limited studies indicate that cholinesterase inhibitors and memantine may be effective treatment options for individuals suffering from this disorder. Additional treatment of frequently associated affective and psychotic symptoms may be necessary but requires caution, as individuals with dementia with Lewy bodies are more sensitive to the extrapyramidal effects of medications, such as antipsychotics. Nonpharmacologic approaches, including physical, occupational, and voice therapy, are also valuable options for comprehensive treatment.
Disclosure of off-label usage
The authors have determined that, to the best of their knowledge, memantine is not approved by the US Food and Drug Administration for the treatment of cognitive impairment in dementia with Lewy bodies.
FINANCIAL DISCLOSURE
Dr Yaari is a consultant for Amedisys Home Health. Dr Tariot has served as a consultant for Acadia, AC Immune, Allergan, Eisai, Epix, Forest, Genentech, MedAvante, Memory Pharmaceuticals, Myriad, Novartis, Sanofi-Aventis, Schering-Plough, and Worldwide Clinical Trials; has received consulting fees and grant/research support from Abbott, AstraZeneca, Avid, Baxter, Bristol-Myers Squibb, GlaxoSmithKline, Elan, Eli Lilly, Medivation, Merck, Pfizer, Toyama, and Wyeth; has received educational fees from Alzheimer’s Foundation of America; has received other research support from Alzheimer’s Association, Arizona Department of Health Services, GE, Institute for Mental Health Research, Janssen, National Institute of Mental Health, and National Institute on Aging; has received honoraria from AstraZeneca, Eisai, Eli Lilly, and Pfizer; is a stock shareholder in Adamas and MedAvante; and holds a patent for “Biomarkers of Alzheimer’s Disease.” Drs Burke and Fleisher and Mss Dougherty and Brand have no personal affiliations or financial relationships with any commercial interest to disclose relative to the activity.
FUNDING/SUPPORT
None reported.
DISCLAIMER
The opinions expressed are those of the authors, not of Banner Health or Physicians Postgraduate Press.
Clinical Points
- Dementia with Lewy bodies (DLB) is a neurodegenerative disorder characterized by progressive cognitive decline plus at least 2 of the following symptoms: cognitive fluctuations, visual hallucinations (typically of mute animate objects appearing early in the course of the disease), or parkinsonism.
- There are currently no US Food and Drug Administration-approved treatments for DLB and no evidence base to direct treatment. Anecdotal evidence and limited studies indicate that cholinesterase inhibitors and memantine may be effective treatment options for individuals suffering from this disorder.
- Nonpharmacologic approaches, including physical, occupational, and voice therapy, are also valuable options for comprehensive treatment of DLB
This CME activity is expired. For more CME activities, visit cme.psychiatrist.com.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
References
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Fourth Edition. Arlington, VA: American Psychiatric Association; 2000.
Cahn-Weiner DA, Williams K, Grace J, et al. Discrimination of dementia with Lewy bodies from Alzheimer disease and Parkinson disease using the Clock Drawing Test. Cogn Behav Neurol. 2003;16(2):85-92. PubMed doi:10.1097/00146965-200306000-00001
Emre M, Tsolaki M, Bonuccelli U, et al. 11018 Study Investigators. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomized, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9(10):969-977. PubMed doi:10.1016/S1474-4422(10)70194-0
Fick DM, Agostini JV, Inouye SK. Delirium superimposed on dementia: a systematic review. J Am Geriatr Soc. 2002;50(10):1723-1732. PubMed
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198. doi:10.1016/0022-3956(75)90026-6 PubMed
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198. doi:10.1016/0022-3956(75)90026-6 PubMed
Links KA, Merims D, Binns MA, et al. Prevalence of primitive reflexes and parkinsonian signs in dementia. Can J Neurol Sci. 2010;37(5):601-607. PubMed
LSVT Global. Rehabilitative Therapy for People With Parkinson’s . http://www.lsvtglobal.com/index.php?action=home. Accessed May 23, 2012
MMSE (Folstein et al, 1975) score generally correlates with disease severity. A score ≤ 9 points can indicate severe dementia, a score between 10-20 points can indicate moderate dementia, and a score > 20 can indicate mild dementia (Mungas, 1991).
Mungas D. In-office mental status testing: a practical guide. Geriatrics. 1991;46(7):54-58, 63, 66. PubMed
Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699. PubMed doi:10.1111/j.1532-5415.2005.53221.x
Stern Y, Albert S, Tang MX, et al. Rate of memory decline in AD is related to education and occupation: cognitive reserve? Neurology. 1999;53(9):1942-1947. PubMed
US Food and Drug Administration. Deaths With Antipsychotics in Elderly Patients With Behavioral Disturbances. April 11, 2005. http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsand
Providers/DrugSafetyInformationforHeathcareProfessionals/PublicHealthAdvisories/
ucm053171.htm?utm_campaign=Google2&utm_source=fdaSearch&utm_medium=website&utm_term=Deaths With Antipsychotics in Elderly Patients With Behavioral Disturbances 2008&utm_content=1. Accessed May 23, 2012
US Food and Drug Administration. FDA Requests Boxed Warnings on Older Class of Antipsychotic Drugs. June 16, 2008. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2008/ucm116912.htm?utm_campaign=Google2&utm_source=fdaSearch&utm_medium=website&utm_term=
antipsychotic drug warnings&utm_content=1. Accessed May 23, 2012
Wild R, Pettit T, Burns A. Cholinesterase inhibitors for dementia with Lewy bodies. Cochrane Database Syst Rev. 2003;(3):CD003672. PubMed
Enjoy this premium PDF as part of your membership benefits!