The Effect of Armodafinil on Patient-Reported Functioning and Quality of Life in Patients With Excessive Sleepiness Associated With Shift Work Disorder: A Randomized, Double-Blind, Placebo-Controlled Trial

ABSTRACT
Objective: To examine whether treatment with armodafinil for 6 weeks affected patient-reported overall functioning and daily quality of life compared with placebo in patients with excessive sleepiness associated with shift work disorder.
Method: This 6-week, multicenter, randomized, double-blind, placebo-controlled, parallel-group study was conducted in 45 sleep centers across the United States between February and October 2010. Patients included in the study were 18 to 65 years of age and diagnosed with excessive sleepiness associated with shift work disorder on the basis of the International Classification of Sleep Disorders: Diagnostic and Coding Manual, Second Edition and DSM-IV-TR criteria. These patients also experienced late-in-shift sleepiness between 4 am and 8 am (Karolinska Sleepiness Scale score ≥ 6) and were functionally impaired (Global Assessment of Functioning score < 70). Patients were administered 150 mg of armodafinil or placebo on nights worked, and efficacy measures included changes in patient-reported overall functioning (modified Sheehan Disability Scale [SDS-M]) and daily quality of life (10-question Functional Outcomes of Sleep Questionnaire [FOSQ-10]).
Results: Patients treated with armodafinil had significantly greater improvement in SDS-M composite scores at final visit (last observation carried forward) (-6.8 vs -4.5, respectively, P = .0027) than those receiving placebo. Although the armodafinil group, compared to the placebo group, showed a greater improvement in total FOSQ-10 score from baseline to final visit (+3.4 vs +2.7, respectively, P = .0775), a statistically significant improvement was observed only at week 6 (+3.6 vs +2.7, respectively, P = .0351).
Conclusions: These findings are consistent with our previous report on clinician-rated measures of efficacy by demonstrating that armodafinil improves patient-rated functioning in patients with shift work disorder. Additionally, the current findings show for the first time that armodafinil may have benefits on quality of life after 6 weeks of treatment.
Trial Registration: ClinicalTrials.gov identifier: NCT01080807
Prim Care Companion CNS Disord 2012;14(4):doi:10.4088/PCC.12m01345
© Copyright 2012 Physicians Postgraduate Press, Inc.
Submitted: January 16, 2012; accepted May 11, 2012.
Published online: August 9, 2012.
Corresponding author: Milton K. Erman, MD, Pacific Sleep Medicine Services, Inc, 10052 Mesa Ridge Court #101, San Diego, CA ([email protected]).
Approximately 20% of employed adults in the United States work shifts outside the hours of 6 am and 6 pm.1 Due to their nighttime and sometimes rotating work schedules, such workers are at risk to develop shift work disorder (SWD). SWD is a circadian rhythm sleep disorder in which misalignment of the circadian rhythm results in excessive sleepiness during work hours and/or insomnia during sleep onset and sleep maintenance.2,3 The exact prevalence of workers with SWD is not known, although it is believed that up to 45% of night shift workers have excessive sleepiness with or without a formal diagnosis of SWD.4
As a result of their symptoms, shift workers (with or without a diagnosis of SWD) exhibit clinically significant impairment in their daily functioning and quality of life. Several aspects of a patient’s functioning (psychological, social, family, and work) are negatively affected by shift work. For example, the symptoms of SWD have been associated with psychological impairment such as increased rates of depression and anxiety, and SWD can exacerbate preexisting mood disorders.4-6 In addition, patients with SWD are more likely to neglect their social life and family obligations than workers without SWD.4 Shift workers are also more likely to be dissatisfied with their current job and are more likely to be involved in accidents while working or commuting home.7,8
To cope with the many symptoms of SWD, patients have a number of therapeutic options available. Currently, only 2 agents are indicated for the treatment of excessive sleepiness in patients with SWD: modafinil (containing R– and S-modafinil) and armodafinil (R-modafinil). Armodafinil is the longer-lasting stereoisomer of modafinil and has a half-life of approximately 15 hours (compared with the 3-hour half-life of S-modafinil).9 Czeisler et al10 demonstrated in a 12-week, multicenter, randomized, double-blind study that armodafinil significantly improved wakefulness, overall clinical condition, attention, and memory in patients with SWD compared with placebo. A recent report on a 6-week, multicenter, randomized, double-blind study showed that armodafinil was superior to placebo in improving clinician-rated clinical condition and wakefulness late in the night shift (between 4 am and 8 am) as well as overall daily functioning.11 The current report examines whether armodafinil also affected patient-reported outcomes related to functioning and quality of life.
METHOD
Patients
As previously described,11 men and women aged 18 to 65 years were included in the study if they were diagnosed with excessive sleepiness associated with SWD on the basis of the International Classification of Sleep Disorders: Diagnostic and Coding Manual, Second Edition3 and the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR)2 criteria. Patients were to have regularly worked five 6- to 12-hour night shifts (between 10 pm and 8 am) per month with 3 of these shifts worked consecutively. Patients were also required to have late-in-shift sleepiness defined as scores ≥ 4 on the Clinical Global Impressions-Severity of Illness scale (CGI-S) and ≥ 6 on the Karolinska Sleepiness Scale (KSS) between 4 am and 8 am.11 A score < 70 on the Global Assessment of Functioning (GAF) was also required for study participation. Major exclusion criteria included the presence of obstructive sleep apnea (apnea/hypopnea index > 5), any medical condition causing excessive sleepiness, or the use of agents that affected sleepiness or functioning in the past 7 days (eg, modafinil, caffeine ingestion of ≥ 600 mg per day, etc). Female participants of childbearing age were required to use a medically accepted method of birth control. Those female patients using oral contraceptives were required to also use a barrier method.11

- Patients with shift work disorder exhibit impairments in functioning and quality of life.
- Armodafinil is currently approved in the United States for the treatment of excessive sleepiness associated with shift work disorder.
- The results of this study suggest that armodafinil improves patient functioning and quality of life in patients with shift work disorder.
Study Procedure
Written informed consent was obtained from each patient before enrollment in the study. This 6-week, multicenter, randomized, double-blind, parallel-group, placebo-controlled study was conducted in accordance with the Guideline for Good Clinical Practice approved by the International Conference on Harmonisation and any applicable national and local laws and regulations. This study was conducted in 45 sleep centers across the United States between February and October 2010 and was registered in ClinicalTrials.gov (identifier: NCT01080807). Each of the 45 sites obtained institutional review board approval. Patients were randomized 1:1 in this trial to receive either armodafinil 150 mg/d or placebo 30-60 minutes before the start of their night shift, but no later than 11 pm, for 6 weeks. Patient diaries were used to assess dosing compliance. Armodafinil was administered orally only on nights worked in 50-mg tablets that were titrated up to 150 mg over 4 work days (1 tablet on first day worked, 2 tablets each on the second and third days worked, and 3 tablets on each subsequent day worked). Efficacy assessments were conducted during clinic visits at weeks 3 and 6.
Assessments
To assess overall functioning, patients were administered a modified version of the Sheehan Disability Scale (SDS-M).12 The scale was modified to be appropriate for patients with SWD. The SDS-M is a visual-analog scale that uses spatiovisual, numeric, and verbal anchors to assess whether the shift schedule disrupted their work, social life/leisure activities, and family life/home responsibilities. The effect of shift schedule on each domain was rated by the patient on a 10-point scale and was categorized as follows: 0 = not at all, 1-3 = mildly, 4-6 = moderately, 7-9 = markedly, and 10 = extremely. Patient-rated quality of life was assessed using the 10-question Functional Outcomes of Sleep Questionnaire (FOSQ-10).13 This version of the FOSQ is composed of 10 questions that measure the impact of sleepiness on daily living and quality of life with regard to physical, mental, and social functioning. Each question is rated on a 4-point scale, with a lower score indicating greater impairment. Total scores for the FOSQ-10 are based on 5 subscale scores (activity level, general productivity, vigilance, social outcome, and intimacy). Tolerability assessments for this study were reported earlier.11
Statistical Analysis
The full analysis included all patients who received at least 1 dose of study drug and had at least 1 postbaseline CGI-C assessment. The final visit results were calculated on an intention-to-treat basis. Results at specific time points are for observed patients only. The change in SDS-M and FOSQ-10 scores from baseline was analyzed using the analysis of variance, with treatment and baseline shift work duration as factors. All test comparisons were 2-tailed, with a significance level of .05. Data represented as final visit included study completers and last observation carried forward for noncompleters. Adverse events and other tolerability measures were analyzed using descriptive statistics.
RESULTS
Baseline Demographics and Patient Characteristics
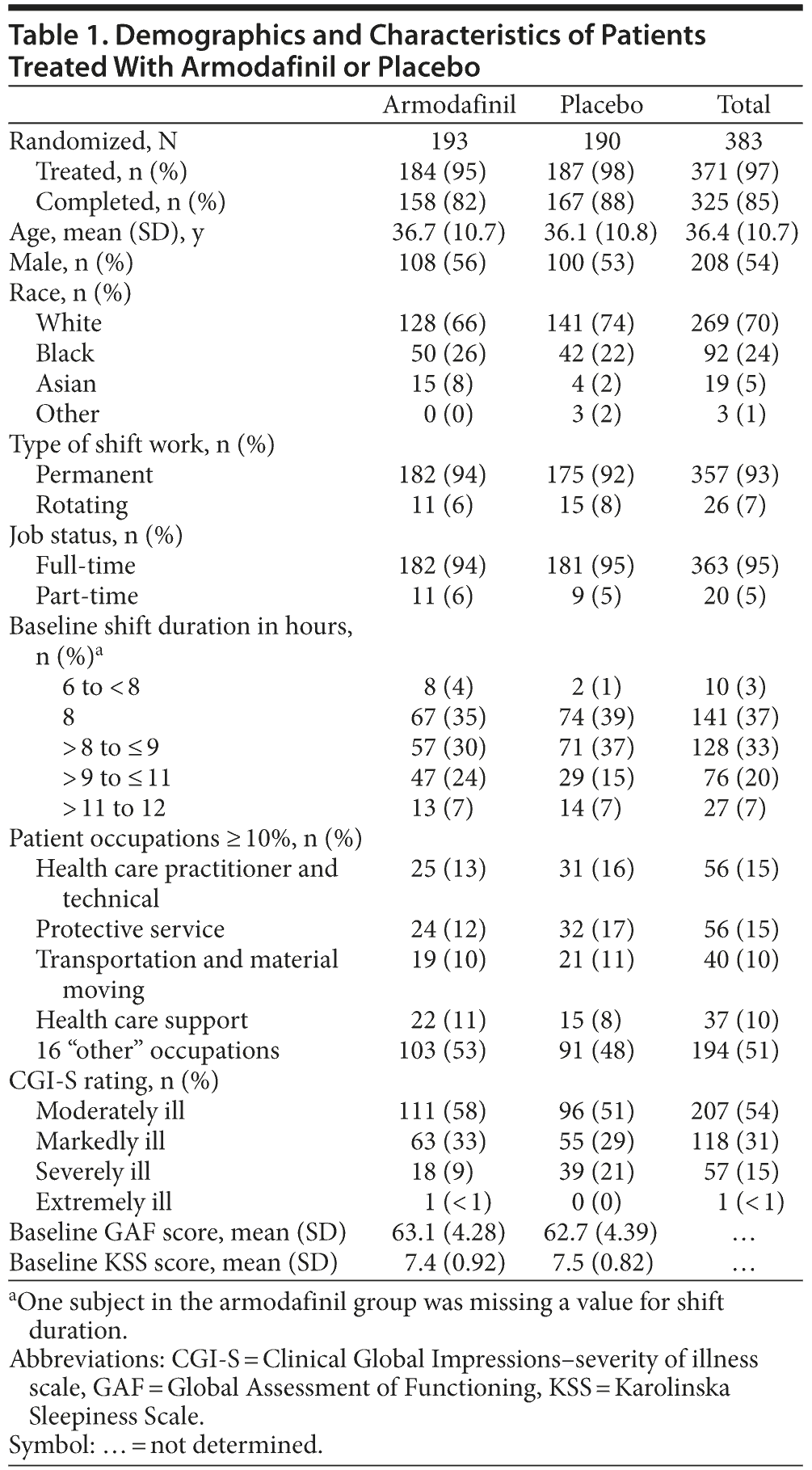
A total of 383 patients were randomly assigned to receive either armodafinil (n = 193) or placebo (n = 190). The proportion of completers was similar between groups (82% for armodafinil vs 88% for placebo). Discontinuation due to adverse events was greater in the armodafinil group (n = 9) compared with the placebo group (n = 1). Patient characteristics between the 2 groups were similar at baseline (Table 1). Most patients in the study worked permanent, full-time night shifts that were at least 8 hours in duration. As expected on the basis of inclusion criteria, patients in the study generally had moderate disease severity (CGI-S), functional impairment (GAF), and sleepiness (KSS) at baseline.
Patient Functioning
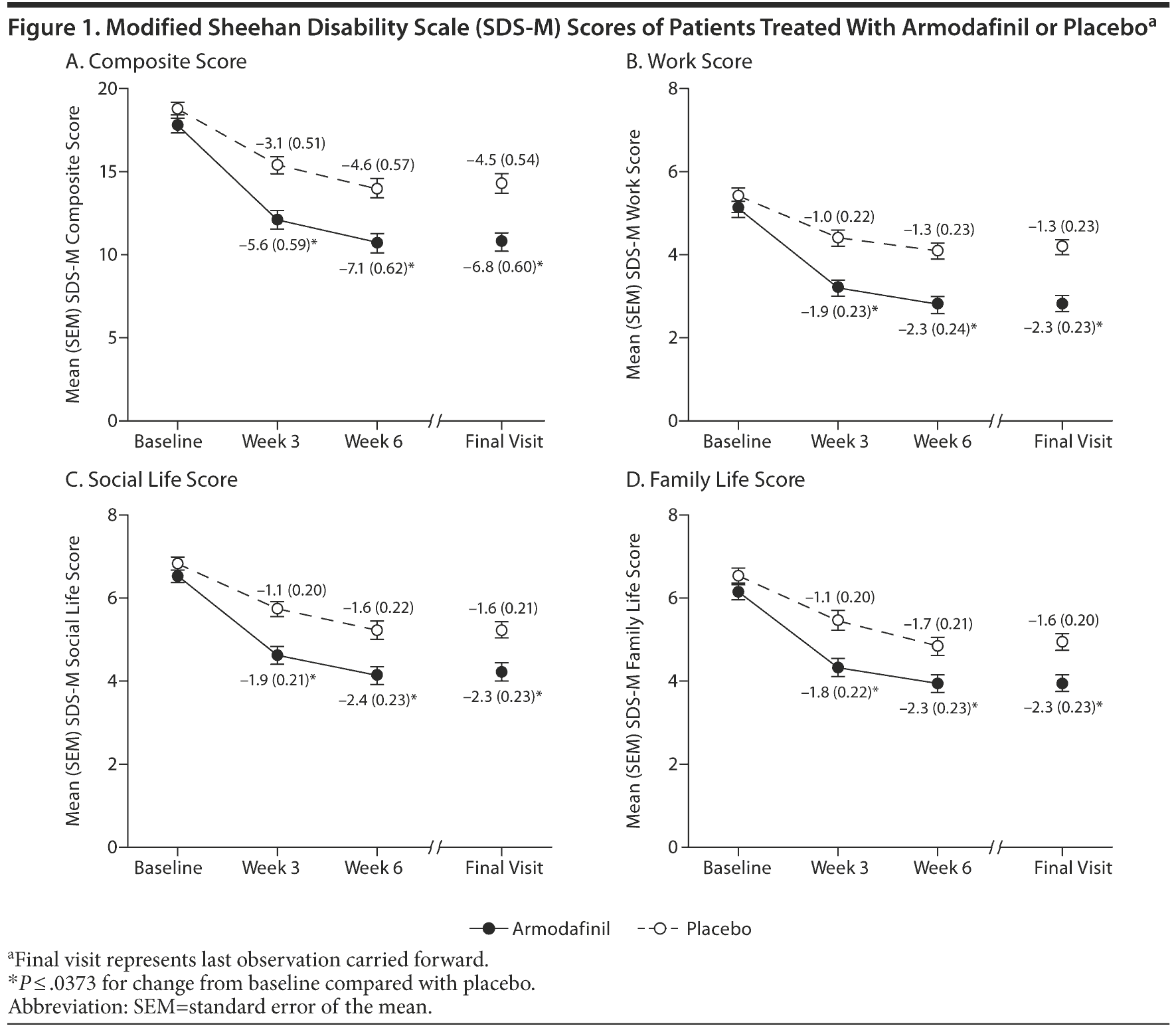
Patients treated with armodafinil had significantly greater improvements in SDS-M composite score from baseline to final visit compared with those receiving placebo (-6.8 for armodafinil vs -4.5 for placebo, P = .0027) (Figure 1A). At final visit, mean (SD) SDS-M composite scores were 10.8 (7.05) for the armodafinil group and 14.3 (7.08) for the placebo group, indicating superior functioning in patients treated with armodafinil at the end of the study. Greater improvements in SDS-M composite score were also observed in the armodafinil group at week 3 (P = .0014) and at week 6 (P = .0031). A similar statistically significant improvement at each time point following armodafinil administration was also observed for SDS-M work score (Figure 1B), social life score (Figure 1C), and family life score (Figure 1D) (P ≤ .0373 for all vs placebo).
Quality of Life
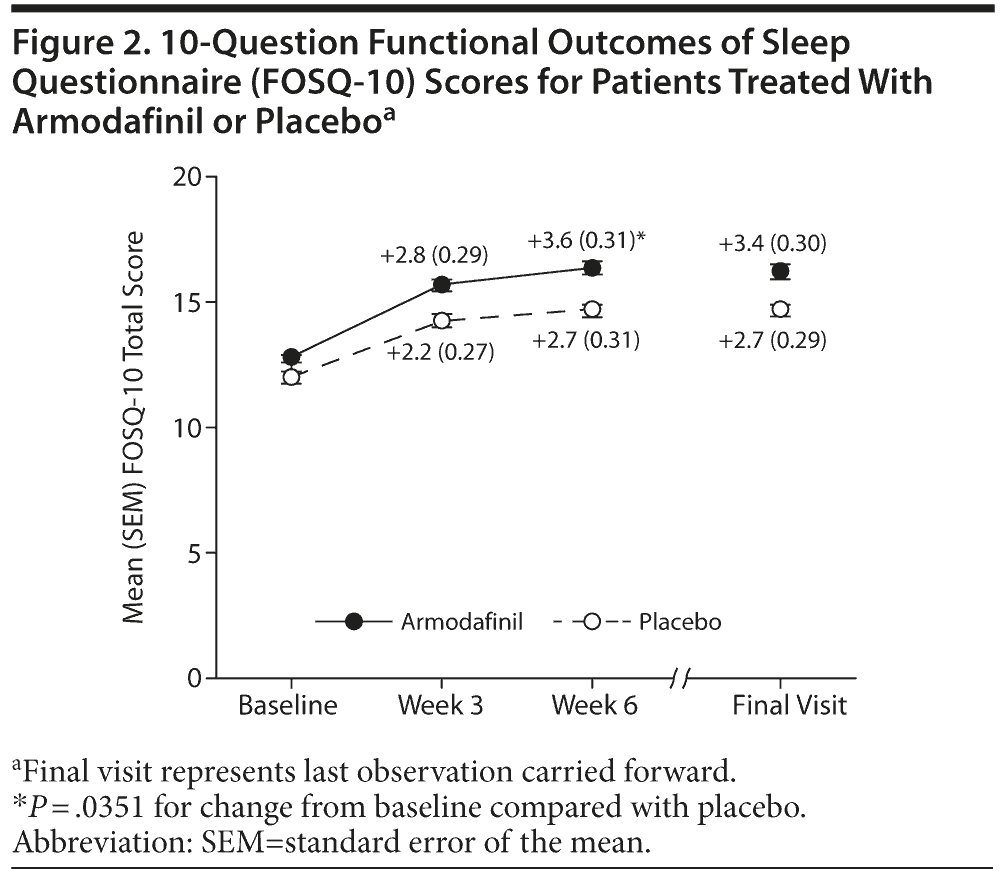
The change in total FOSQ-10 score from baseline was greater in patients treated with armodafinil compared with those receiving placebo, and this difference was statistically significant at week 6 (+3.6 vs +2.7, respectively, P = .0351) but not at final visit (+3.4 vs +2.7, respectively, P = .0775) (Figure 2). With regard to FOSQ-10 subscale scores (data not shown), changes from baseline in vigilance score were significantly greater in the armodafinil group compared with the placebo group at week 3 (+0.7 vs +0.5, respectively, P = .0323), week 6 (+0.8 vs +0.6, respectively, P = .0153), and final visit (+0.8 vs +0.6, respectively, P = .0337). Patients in the armodafinil group had significantly greater improvements in activity level (+0.8 for armodafinil vs +0.6 for placebo, P = .0273) and general productivity (+0.8 for armodafinil vs +0.6 for placebo, P = .0382) at week 6 only. Changes in social outcome score or intimacy score were not significantly different between the 2 groups at any of the time points examined.
DISCUSSION
Armodafinil has been previously shown to significantly improve wakefulness and clinician-rated measures of clinical condition and overall functioning in patients with SWD.10,11 The current report examined whether armodafinil affected patient-rated outcome measures of functioning and quality of life in patients with SWD. A significantly greater improvement in patient functioning was observed in patients treated with armodafinil at each time point compared with placebo. Patients in the armodafinil group also demonstrated a significantly greater improvement in quality of life after 6 weeks of treatment versus placebo. The results presented here are consistent with previous findings that patients with SWD benefit from armodafinil treatment in terms of patient functioning and show for the first time that 6 weeks of armodafinil treatment improved quality of life compared with placebo.
Shift workers are at a greater risk for psychological and social impairments than traditional daytime workers. In particular, patients with SWD demonstrate significant functional deficits that interfere with their daily lives. We have previously demonstrated that patients with SWD have considerable functional impairment as measured by the clinician-rated GAF and that armodafinil significantly improved functioning versus placebo.11 The GAF is Axis V of the DSM-IV-TR multiaxial assessment and is often used by psychiatrists to measure functional impairment.2 In the current report, patients were asked to rate their overall functioning using a modified version of the SDS (SDS-M), in which each domain was revised to assess the impact of shift schedule on the patient’s work, social, and family life. The SDS-M may be a useful and easy-to-administer assessment in identifying mental health-related functional impairment in primary care patients. The improvement in patient functioning with armodafinil observed in this report supports our earlier findings using the GAF.11 Thus, through the use of 2 different assessments for functioning, it would be reasonable to conclude that armodafinil treatment results in significantly greater improvements in overall functioning compared with placebo in patients with SWD.
The finding that improvements in each SDS-M subscale score (work, social life, and family life scores) were significantly greater in the armodafinil group was also notable. Baseline values for each of the mean SDS-M subscale scores were ≥ 4, indicating that patients enrolled in this study were experiencing at least “moderate” functional impairment at work, in their family life, and in their social life. At the end of the study, patients in the placebo group still showed “moderate” functional impairment (SDS-M score of 4 to 6) in each of the 3 domains. Conversely, armodafinil improved SDS-M work score to a value coinciding with “mild” functional impairment. Patients who received armodafinil were still moderately impaired with regard to social life and family life scores, although they demonstrated greater improvements in these domains than those patients administered placebo. Such residual impairment may be expected given the scheduling conflicts between night shift work and social and family activities.
In addition to functional impairment, patients with SWD often have a poor quality of life. Unlike other generic measures of quality of life, the FOSQ was developed to specifically address the impact of excessive sleepiness on 5 different aspects of a patient’s life.14 The results presented here show that armodafinil improved total FOSQ-10 score, although the difference was significant compared with placebo only in those patients who completed the 6-week study. Significant differences between treatment groups were also observed in FOSQ-10 subscores for activity level, general productivity, and vigilance, which were likely the result of improved wakefulness during the night shift following armodafinil treatment; however, changes in social outcome score and intimacy score were not significant. It should be noted that this study was specifically powered to detect treatment differences only in the Clinical Global Impressions of Change (CGI-C) score, the primary efficacy measure, and GAF score, the key secondary measure, rather than the secondary measures such as SDS-M and FOSQ-10 scores.11 To our knowledge, only 1 other study has examined quality of life with the FOSQ in patients with SWD. In that study, significant improvements in FOSQ (total, activity level, productivity, and vigilance only) were also observed in patients with SWD receiving 300 mg of modafinil for 12 weeks.15 However, caution should be used when comparing the FOSQ results of the current study with the aforementioned modafinil study, as there were differences in study duration.
A few limitations of the FOSQ-10 should be considered when interpreting the results of this study. First, the FOSQ-10 has not been validated for use in SWD. The FOSQ-10 asks questions related to the patient’s entire week and not only those nights worked. In the case of patients with SWD, impairment is likely most severe while the patient is at work, and the FOSQ-10 may not capture the true impact of excessive sleepiness in this population. Furthermore, unlike patients with other sleep disorders such as obstructive sleep apnea who take armodafinil daily, patients with SWD took armodafinil only on nights worked. Thus, the effects of armodafinil on FOSQ-10 in the current study may be underestimated due to the fact that the questions may not have been asked immediately after working a night shift. Another limitation of the FOSQ is that it was not designed to detect changes on the basis of treatment, although it has been used by patients with obstructive sleep apnea to rate treatment-induced changes in quality of life.16,17
The overall results of the current study support previous findings that armodafinil improves excessive sleepiness and associated symptoms of SWD. Our previous report using the same study population demonstrated that armodafinil improved late-in-shift (4 am to 8 am including the commute home) clinical condition (CGI-C), sleepiness (KSS), and overall functioning (GAF) in patients with SWD.11 In addition, Czeisler et al10 previously demonstrated in a 12-week study that armodafinil improved sleep latency (Multiple Sleep Latency Test), clinical condition (CGI-C), sleepiness (KSS), memory, and attention in patients with SWD. Taken together, the findings from the above-mentioned studies and the current study demonstrate a benefit of armodafinil in improving excessive sleepiness during either the last 4 hours of the night shift or throughout the entire night shift and the commute home.
It is important to note that armodafinil does not treat the full spectrum of symptoms associated with SWD. Instead, armodafinil is only used to treat excessive sleepiness on nights worked. SWD is a multifaceted disease, and while armodafinil-treated patients may not be as sleepy during their work hours or commute home as nontreated patients, the disruption of their circadian rhythm will lead to some sleepiness and/or insomnia during nonworking hours. As a result, even with successful armodafinil treatment, the symptoms of SWD can impact the patient’s social and family life. It is recommended that pharmacotherapy with agents such as armodafinil be part of a comprehensive treatment program to improve a patient’s overall symptom burden. This program should involve appropriate sleep hygiene, sleep education, planned napping, and appropriate diet and exercise. Employers can also assist with alleviating patient symptoms by providing sufficient time off between shifts, limiting shift duration, and appropriately timing light exposure to encourage circadian rhythm adjustment.
In summary, armodafinil significantly improved patient-rated functioning compared with placebo in patients with SWD. Patients who received armodafinil for 6 weeks also demonstrated improved quality of life compared with those receiving placebo. These findings extend earlier reports that armodafinil significantly improves excessive sleepiness in patients with SWD and support the use of armodafinil in a comprehensive treatment program that addresses the critical symptoms of SWD.
Drug names: armodafinil (Nuvigil), modafinil (Provigil).
Author affiliations: Pacific Sleep Medicine Services, San Diego, California (Dr Erman); Biometrics Department, Teva Pharmaceutical Industries Ltd, Frazer, Pennsylvania (Dr Yang); and Broward Research Group, Pembroke Pines, Florida (Dr Seiden).
Potential conflicts of interest: Dr Erman serves as a consultant to Cephalon (Teva Pharmaceutical Industries Ltd), Purdue, Somaxon, Sunovion, and Takeda; has received grant/research support from Arena, Aventis, Cephalon (Teva Pharmaceutical Industries Ltd), Eli Lilly, GlaxoSmithKline, Mallinckrodt, Merck, Neurocrine, Organon, Orphan, Pfizer, Pharmacia, ResMed, Sanofi-Synthelabo, Schwarz, Takeda, and Transcept; serves on the speakers or advisory boards of Cephalon (Teva Pharmaceutical Industries Ltd), Sunovion, and Takeda; and is a stock shareholder in Cephalon (Teva Pharmaceutical Industries Ltd), Forest, Merck, Pfizer, and Sanofi-Synthelabo. Dr Yang is an employee of Teva Pharmaceutical Industries Ltd. Dr Seiden is part owner and medical director of the Broward Research Group and received funding from Cephalon (Teva Pharmaceutical Industries Ltd) for this study.
Funding/support: This study was sponsored by Cephalon, now a wholly owned subsidiary of Teva Pharmaceutical Industries Ltd, Frazer, Pennsylvania.
Previous presentation: Oral presentation at the World Association of Sleep Medicine and Canadian Sleep Society Congress; September 10-14, 2011; Quebec City, Canada.
Acknowledgments: Editorial assistance was provided by Aji Nair, PhD (Global Scientific Communications, Teva Pharmaceutical Industries Ltd, Frazer, Pennsylvania). The authors thank Ryan Dammerman, MD, PhD, formerly of Cephalon (Teva Pharmaceutical Industries Ltd, Frazer, Pennsylvania), for contributions to trial design and data interpretation; Dr Dammerman is currently an employee of Shire, Chesterbrook, Pennsylvania. Drs Nair and Dammerman report no other conflicts of interest related to the subject of this article.
REFERENCES
1. McMenamin TM. A time to work: recent trends in shift work and flexible schedules. Mon Labor Rev. 2007;130:3-15.
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
3. American Academy of Sleep Medicine. The International Classification of Sleep Disorders: Diagnostic and Coding Manual, Second Edition. Westchester, IL: American Academy of Sleep Medicine; 2005.
4. Drake CL, Roehrs T, Richardson G, et al. Shift work sleep disorder: prevalence and consequences beyond that of symptomatic day workers. Sleep. 2004;27(8):1453-1462. PubMed
5. Cole RJ, Loving RT, Kripke DF. Psychiatric aspects of shiftwork. Occup Med. 1990;5(2):301-314. PubMed
6. Puca FM, Perrucci S, Prudenzano MP, et al. Quality of life in shift work syndrome. Funct Neurol. 1996;11(5):261-268. PubMed
7. Ohayon MM, Lemoine P, Arnaud-Briant V, et al. Prevalence and consequences of sleep disorders in a shift worker population. J Psychosom Res. 2002;53(1):577-583. PubMed doi:10.1016/S0022-3999(02)00438-5
8. Steele MT, Ma OJ, Watson WA, et al. The occupational risk of motor vehicle collisions for emergency medicine residents. Acad Emerg Med. 1999;6(10):1050-1053. PubMed doi:10.1111/j.1553-2712.1999.tb01191.x
9. Darwish M, Kirby M, Hellriegel ET, et al. Armodafinil and modafinil have substantially different pharmacokinetic profiles despite having the same terminal half-lives: analysis of data from three randomized, single-dose, pharmacokinetic studies. Clin Drug Investig. 2009;29(9):613-623. PubMed doi:10.2165/11315280-000000000-00000
10. Czeisler CA, Walsh JK, Wesnes KA, et al. Armodafinil for treatment of excessive sleepiness associated with shift work disorder: a randomized controlled study. Mayo Clin Proc. 2009;84(11):958-972. PubMed
11. Erman MK, Seiden DJ, Yang R, et al. Efficacy and tolerability of armodafinil: effect on clinical condition late in the shift and overall functioning of patients with excessive sleepiness associated with shift work disorder. J Occup Environ Med. 2011;53(12):1460-1465. PubMed doi:10.1097/JOM.0b013e318237a17e
12. Sheehan DV, Harnett-Sheehan K, Raj BA. The measurement of disability. Int Clin Psychopharmacol. 1996;11(suppl 3):89-95. PubMed doi:10.1097/00004850-199606003-00015
13. Chasens ER, Ratcliffe SJ, Weaver TE. Development of the FOSQ-10: a short version of the Functional Outcomes of Sleep Questionnaire. Sleep. 2009;32(7):915-919. PubMed
14. Weaver TE, Laizner AM, Evans LK, et al. An instrument to measure functional status outcomes for disorders of excessive sleepiness. Sleep. 1997;20(10):835-843. PubMed
15. Erman MK, Rosenberg R; Modafinil Shift Work Sleep Disorder Study Group. Modafinil for excessive sleepiness associated with chronic shift work sleep disorder: effects on patient functioning and health-related quality of life. Prim Care Companion J Clin Psychiatry. 2007;9(3):188-194. PubMed doi:10.4088/PCC.v09n0304
16. Dinges DF, Weaver TE. Effects of modafinil on sustained attention performance and quality of life in OSA patients with residual sleepiness while being treated with nCPAP. Sleep Med. 2003;4(5):393-402. PubMed doi:10.1016/S1389-9457(03)00108-4
17. Weaver TE, Chasens ER, Arora S. Modafinil improves functional outcomes in patients with residual excessive sleepiness associated with CPAP treatment. J Clin Sleep Med. 2009;5(6):499-505. PubMed