Because this piece does not have an abstract, we have provided for your benefit the first 3 sentences of the full text.
To the Editor: There is a paucity of pharmacologic data on the prevention of postpartum depressive episodes in women with bipolar disorder. A single-blind, nonrandomized trial of valproate found the drug was ineffective in preventing mood episodes, particularly depression. Lithium is effective in the prevention of postpartum mania and psychosis; however, it is unclear whether lithium is also effective in preventing postpartum depressive episodes.
Lamotrigine in the Prevention of Bipolar II Postpartum Depression
To the Editor: There is a paucity of pharmacologic data on the prevention of postpartum depressive episodes in women with bipolar disorder.1 A single-blind, nonrandomized trial2 of valproate found the drug was ineffective in preventing mood episodes, particularly depression. Lithium is effective in the prevention of postpartum mania and psychosis3; however, it is unclear whether lithium is also effective in preventing postpartum depressive episodes. Lamotrigine alone or in combination with other mood stabilizers is recommended for acute and maintenance treatment of bipolar disorder.4 Due to its relatively safe profile during pregnancy, lamotrigine has been studied in the maintenance treatment of bipolar disorder in pregnant women.5 There are no studies on the effectiveness of lamotrigine in the prevention of postpartum mood episodes in spite of a recommendation for its use in the maintenance treatment of both bipolar I and bipolar II disorders.6 Compared to lithium, lamotrigine has a lower risk of major teratogenicity and is relatively safe during lactation.7
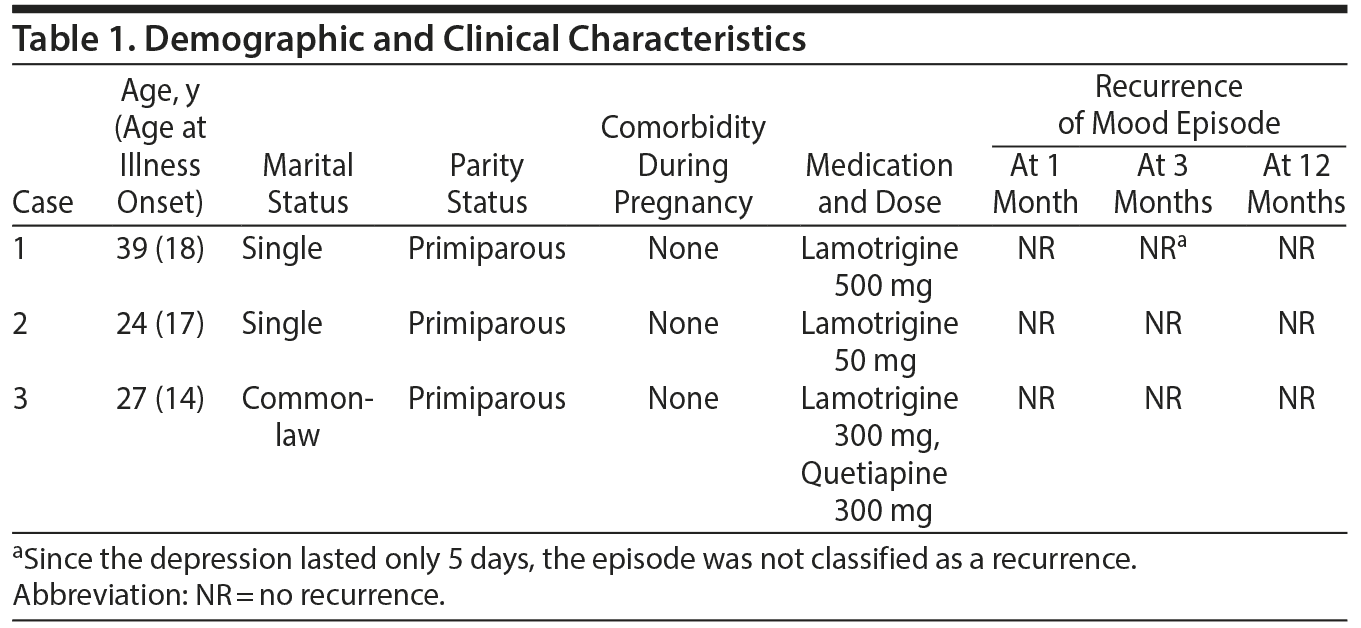
Case reports. We describe our clinical experience with lamotrigine in the prevention of postpartum depression in women with bipolar II disorder. Table 1 outlines demographic and clinical characteristics of patients who were diagnosed according to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). Written informed consent was obtained from women whose cases are described herein.
Case 1 had taken lamotrigine 500 mg for approximately 6 years and had been symptom-free for at least 4 years prior to the time of conception. She remained free of depression during and after pregnancy except a brief depressive period 2 weeks after childbirth that remitted spontaneously after 5 days. She was discharged to her family physician’s care after remaining free of depression for 1 year. Case 2 had several depressive episodes prior to pregnancy but was first treated during pregnancy. Within a month of taking lamotrigine 50 mg, she achieved remission and remained free of depression until she was discharged to her family physician’s care 1 year postpartum. Case 3 had required multiple psychiatric hospitalizations before experiencing mood stability on a combination of lamotrigine 300 mg and quetiapine 300 mg. Her mood had been stable for 2 months prior to her first pregnancy. She did not have recurrence of a mood episode during or after her 2 pregnancies and was discharged from our care when her youngest child was 1 year old.
These case histories suggest that lamotrigine alone or in combination with quetiapine may be effective in the prevention of postpartum depression in women with bipolar II disorder. Case 3 had failed to respond to quetiapine monotherapy prior to the addition of lamotrigine; therefore, it is difficult to ascertain whether lamotrigine alone or the combination provided protection against postpartum depression. Several factors, including early ages of illness onset, prior depressive episodes, and primiparity, meant these women were at risk of recurrence of depression after childbirth. Remarkably, there were no recurrences of depression during or after pregnancy once these women achieved remission.
Lamotrigine was well tolerated, and there were no apparent major adverse effects in 2 women who breastfed their children (Cases 2 and 3). Since there were no women with bipolar I disorder, caution needs to be exercised about generalizing our clinical observations to women with this disorder. Lamotrigine is not as effective in the prevention of mania as it is for depression and should not be used alone for postpartum prophylaxis in women with bipolar I disorder.1 Given these preliminary but promising results, randomized controlled trials are needed to assess the efficacy and tolerability of lamotrigine in the prevention of postpartum depressive episodes in women with bipolar II disorder.
References
1. Sharma V, Burt VK, Ritchie HL. Assessment and treatment of bipolar II postpartum depression: a review. J Affect Disord. 2010;125(1-3):18-26. PubMed doi:10.1016/j.jad.2009.09.014
2. Wisner KL, Hanusa BH, Peindl KS, et al. Prevention of postpartum episodes in women with bipolar disorder. Biol Psychiatry. 2004;56(8):592-596. PubMed doi:10.1016/j.biopsych.2004.07.022
3. Bergink V, Bouvy PF, Vervoort JS, et al. Prevention of postpartum psychosis and mania in women at high risk. Am J Psychiatry. 2012;169(6):609-615. PubMed doi:10.1176/appi.ajp.2012.11071047
4. Bowden CL, Calabrese JR, Sachs G, et al; Lamictal 606 Study Group. A placebo-controlled 18-month trial of lamotrigine and lithium maintenance treatment in recently manic or hypomanic patients with bipolar I disorder. Arch Gen Psychiatry. 2003;60(4):392-400. PubMed doi:10.1001/archpsyc.60.4.392
5. Yatham LN, Kennedy SH, Parikh SV, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) collaborative update of CANMAT guidelines for the management of patients with bipolar disorder: update 2013. Bipolar Disord. 2013;15(1):1-44. PubMed doi:10.1111/bdi.12025
6. Newport DJ, Stowe ZN, Viguera AC, et al. Lamotrigine in bipolar disorder: efficacy during pregnancy. Bipolar Disord. 2008;10(3):432-436. PubMed doi:10.1111/j.1399-5618.2007.00565.x
7. Clark CT, Klein AM, Perel JM, et al. Lamotrigine dosing for pregnant patients with bipolar disorder. Am J Psychiatry. 2013;170(11):1240-1247. PubMed doi:10.1176/appi.ajp.2013.13010006
aWestern University, Department of Psychiatry and Department of Obstetrics and Gynecology, London, Ontario, Canada
bParkwood Institute, Mental Health Care Building, London, Ontario, Canada
Potential conflicts of interest: Dr Sharma has received grant support from, participated on scientific advisory boards for, or served on the speakers’ bureaus of AstraZeneca, Bristol-Myers Squibb, Elan Pharmaceuticals, Eli Lilly, Janssen, Lundbeck, Neurocrine Biosciences, Pfizer, Stanley Medical Research Institute, Sunovion, and Servier. Ms Sommerdyk reports no conflict of interest.
Funding/support: None.
Published online: November 3, 2016.
Prim Care Companion CNS Disord 2016;18(6):doi:10.4088/PCC.16l01964
© Copyright 2016 Physicians Postgraduate Press, Inc.
Enjoy this premium PDF as part of your membership benefits!