Because this piece does not have an abstract, we have provided for your benefit the first 3 sentences of the full text.
Bipolar disorder is both common and serious, frequently leading to functional disability, comorbid medical conditions, and suicide. The disorder is seen in 1 in 20 primary care patients, with an estimated population prevalence of 1 in 25 people. The lifetime prevalence of bipolar disorder is approximately 4%.
Obstructive Sleep Apnea as a Complication
of Bipolar Disorder and Its Treatment:
A Review and Approach to Management
Bipolar disorder is both common and serious, frequently leading to functional disability, comorbid medical conditions, and suicide.1 The disorder is seen in 1 in 20 primary care patients, with an estimated population prevalence of 1 in 25 people.2 The lifetime prevalence of bipolar disorder is approximately 4%.2
Despite the severity of this psychiatric condition, most patients with bipolar disorder in the United States are treated by primary care physicians and not by psychiatrists.3 Therefore, primary care physicians should be mindful of medication side effects and their complications. First-line treatments for bipolar disorder include lithium and valproic acid supplemented by use of atypical antipsychotics, all of which are associated with myriad multisystem side effects, especially weight gain, that predispose patients to obstructive sleep apnea (OSA) among other potential complications.4 The following case vignette and discussion should prove useful with regard to screening for OSA in patients with bipolar disorder, choosing medications for bipolar disorder with consideration of metabolic factors, and initiating treatments that mitigate weight gain.
CASE VIGNETTE
Ms A, a 56-year-old woman with bipolar disorder, alcohol use disorder (in remission), type 2 diabetes (diagnosed 3 years earlier), and hypothyroidism, was brought from home by ambulance to the emergency department for shortness of breath, cough, and confusion. Ms A was found to be in acute hypoxic respiratory failure; she was placed on bilevel positive airway pressure (BiPAP) at admission and did not require intubation. After evaluation, it was determined that this presentation was not the result of a suicide attempt. Ms A’s baseline mental health was well controlled with current pharmacotherapy; however, her respiratory health was not fully optimized given a lack of follow-up for previously diagnosed pulmonary hypertension and inconsistent use of BiPAP.
Four years prior to this admission, Ms A had a respiratory arrest at home in the context of cocaine intoxication and was hospitalized with intubation for 3 days. On the basis of her overall clinical picture and echocardiographic findings, she was diagnosed with pulmonary arterial hypertension and OSA. She was unaware of any snoring and had no partner to corroborate a snoring history.
At admission, her Mallampati classification was I (soft palate, uvula, and pillars were visible). Her neck circumference was not obtained, and no previous measurements were found in her records. Of note, Ms A’s bipolar disorder diagnosis was long established by this time. She had taken lithium and quetiapine for several years before the diagnoses of OSA and pulmonary hypertension. For the past year, she had been treated with aripiprazole.
Ms A had a history of multiple prior psychiatric hospitalizations and had repeatedly improved with treatment that included lithium, aripiprazole, and quetiapine. At admission, her serum lithium level was 0.9 mEq/L (in the therapeutic range). Her urine toxicology screen was negative for alcohol, benzodiazepines, and opiates. Unfortunately, her weight had continued to rise over time in association with use of lithium and second-generation antipsychotics, which most likely contributed to the onset and progression of OSA and pulmonary hypertension. Ms A’s current weight was 215 lb, her height was 64 in, and her body mass index (BMI) was 36.9 kg/m2. Per hospital records, she had gained approximately 12 lb over the 8 months prior to hospitalization.
During the admission, Ms A’s lithium dose was decreased from 450 mg twice/d to 300 mg once in the morning and 450 mg once in the evening, aripiprazole was reduced from 10 mg/d to 5 mg/d, and quetiapine was discontinued. Quetiapine was discontinued because it has been associated with acute respiratory dysfunction in patients with OSA.5 Lithium was decreased because of sedating side effects; furthermore, her serum lithium level was supratherapeutic for the prophylaxis against mania. Aripiprazole was also decreased because of known respiratory side effects, primarily dyspnea. She continued using BiPAP.
BIPOLAR DISORDER
The DSM-56 divides bipolar disorder into several categories (bipolar I disorder, bipolar II disorder, cyclothymic disorder, and other specified bipolar and related disorders, with or without mixed features). A person must have had at least 1 manic episode during his or her lifetime to meet criteria for bipolar I disorder. This diagnosis can be made in those who have met criteria for major depressive disorder (MDD) in the past, but a depressive episode is not required for the diagnosis of bipolar I disorder.7
A manic episode is characterized by a persistently elevated, expansive, or irritable mood with increased goal-directed activity/energy that lasts most of the day nearly every day for ≥ 1 week (or any duration that requires hospitalization of the patient).7 Symptoms must also cause marked social or occupational impairment, require hospitalization, or include psychotic symptoms. Of the following symptoms, ≥ 3 must be present (4 if the mood is only irritable): grandiosity, decreased sleep, increased talking/pressured speech, flight of ideas, distractibility, increased goal-directed activity/psychomotor agitation, and participation in dangerous activities.7 As with many other DSM-5–defined disorders, substance use and medical conditions cannot contribute to the manic episode (with the exception of antidepressant use).
Bipolar II disorder is characterized by a history of hypomanic episodes alternating with major depressive episodes.7 A depressive episode is required to diagnose bipolar II disorder but is not required to diagnose bipolar I disorder.7 Hypomania requires the same number of mania-defining symptoms described previously; however, the episode must last no more than 4 days and must not be severe enough to cause marked social or occupational impairment. Importantly, the presence of psychotic features rules out hypomania and suggests that the patient is suffering from a manic episode.7
“Mixed features” is a specifier applied to manic, hypomanic, and depressive episodes in the bipolar spectrum (also a specifier for MDD). It involves ≥ 3 hypomanic/manic symptoms in a major depressive episode or ≥ 3 depressive symptoms in a hypomanic/manic episode.8 The presence of mixed features alone places a patient at high risk (elevated risk of completed suicide) given that it predicts more severe illness and treatment resistance.8
What Causes Bipolar Disorder?
The etiology of bipolar disorder is still not fully understood, although it is believed to have both genetic and environmental factors. The circadian dysregulation hypothesis is currently at the center of environmental factors and is linked with genes responsible for the molecular clock (located in the suprachiasmatic nucleus of the hypothalamus).8 Bipolar disorder is characterized by disrupted sleep-wake schedules, daily activities, and appetite, all of which are integrally related to the circadian system. Mood exacerbations may also be provoked by seasonal patterns and alterations in light intensity, which may explain associated irregular control of sleep, hormone levels, and body temperature (that are all under circadian control).8
Neurobiologic studies9 of euthymic bipolar disorder patients have shown reduced responsiveness in brain areas associated with cognition (eg, anterior cingulate cortex and dorsolateral/medial prefrontal cortex) and increased responsiveness of limbic/paralimbic regions responsible for emotions (eg, amygdala, ventrolateral prefrontal cortex, and ventral anterior cingulate cortex). Other studies10 suggest irregularities in important genes, such as CACNA1C and ANK3 and their implicated pathways involved with calcium signaling, in patients with bipolar disorder. Mitochondrial and endoplasmic reticulum dysfunction, autophagy, and glutamatergic neurotransmission are additional molecular pathways implicated in bipolar disorder.11 White matter abnormalities suggesting axonal disorganization have also been described as an early marker for bipolar disorder and as part of its pathogenesis.12
What Elements of the Differential
Diagnosis Should Be Considered?
It is critical to consider bipolar disorder when evaluating a patient with symptoms of MDD, because the treatment for MDD can precipitate a hypomanic/manic episode in some individuals with bipolar disorder. “Red flags” the primary care physician should be aware of when treating patients with depressive episodes include poor functioning in social/occupation contexts, risky behaviors, history of legal disputes, family history of bipolar disorder, associated symptoms of psychosis, early age at onset, antidepressant resistance, and hypomanic/manic symptoms after starting antidepressants.3,13 An initial depressive episode, while appropriately diagnosed by the primary care physician as MDD, may in hindsight be the first depressive episode experienced by a patient who will develop hypomania/mania. This progression requires conversion of the diagnosis of MDD to bipolar disorder. Thus, patients with the aforementioned warning signs and recurrent episodes of depression should be carefully monitored for manic features. Substance- or medication-induced mania must be monitored in all patients and managed by dose adjustment or discontinuation of the offending agent.13 Moreover, individuals with attention-deficit/hyperactivity disorder, prominent irritability, or borderline personality disorder have symptoms that overlap with those of bipolar disorder.14 Identification of the timeline of manic symptoms (episodic versus baseline behavior) is critical to avoid misdiagnosis.
Which Psychiatric Comorbidities
Should Be Considered?
Anxiety disorders are often comorbid with bipolar disorder, occurring in 75% of patients with bipolar disorder.7 Given that anxiety disorders are frequently treated with selective serotonin reuptake inhibitors, all anxious patients should be screened for bipolar disorder to avoid precipitation of a manic episode with pharmacologic treatment of anxiety.
Bipolar disorder is also strongly associated with substance use disorders, with alcohol use disorder occurring in nearly 50% of patients with bipolar disorder.7 Given that comorbid alcohol use disorder increases the risk of suicide attempts and completed suicide, it is crucial to screen patients with these comorbid conditions to guide treatment. For example, a double-blind, randomized, placebo-controlled study15 with 59 patients with bipolar disorder and alcohol dependence found that treatment with valproate could decrease heavy drinking.
How Is Bipolar Disorder Treated?
First-line treatment of bipolar disorder includes lithium carbonate or valproic acid often in conjunction with a second-generation antipsychotic and supportive therapy. Lithium treats acute mania, prevents future manic episodes, and is the only drug known to decrease suicidality in patients with bipolar disorder. Patients typically require plasma levels between 0.8 and 1.2 mmol/L to treat acute mania, with concentrations measured 12 hours after the last dose.16 Serum levels between 0.6 and 0.75 mmol/L are often recommended for effective mania prophylaxis.17
Antiepileptic drugs, including valproic acid, carbamazepine, and lamotrigine, have mood-stabilization properties that make them effective second-line treatments for bipolar disorder. Atypical antipsychotics (such as olanzapine, aripiprazole, quetiapine, risperidone, and ziprasidone) are approved for the treatment of acute mania.18 Only olanzapine and aripiprazole are approved by the US Food and Drug Administration for long-term maintenance treatment in bipolar disorder (olanzapine is preferred in underweight patients, and aripiprazole is preferred in overweight patients).18 Atypical antipsychotic medications differ vastly in their metabolic side effect profiles. For purposes of clarity in this article, atypical antipsychotics with high metabolic risk profiles (olanzapine, quetiapine) will be referred to as high-risk atypical antipsychotics and those with low metabolic risk profiles (aripiprazole, ziprasidone) will be referred to as low-risk atypical antipsychotics. Psychotherapy/education is an effective long-term treatment; it is vital to emphasize medication adherence, awareness of prodromal symptoms, and circadian rhythm management. There has only been anecdotal use of electroconvulsive therapy in the treatment of drug-resistant mania, and it has not been described as long-term management.18
Unfortunately, several of the pharmacologic treatments are associated with weight gain. There have been case reports19 documenting massive weight gain on carbamazepine; however, a 12-month double-blind trial20 of carbamazepine versus lithium associated carbamazepine with weight loss (carbamazepine: n = 15, lithium: n = 16). Lamotrigine is also generally considered more weight neutral.19,21 There is little evidence that either carbamazepine or lamotrigine have been independently associated with OSA. Thus, we will focus on use of lithium, valproic acid, and the atypical antipsychotics.
What Is the Mechanism of Action
of Mood-Stabilizing Agents
There have been several hypotheses on the mechanism of action of lithium. Lithium depletes inositol in the central nervous system, inhibits overactive protein kinase C intracellular signaling, and preserves white matter, especially with long-term administration.11,22 The mechanism of weight gain associated with lithium is not well understood but is thought to result from increased stimulation of hexokinase and pyruvate kinase, leading to increased cellular uptake of glucose and inhibition of muscle protein kinase.23
The mechanism of valproic acid as a mood stabilizer also includes interference with inositol metabolism. Valproic acid enhances γ-aminobutyric acid (GABA)–mediated neurotransmission and alters gene expression through direct inhibition of histone deacetylases.24 Valproic acid also provides neuroprotection by increasing expression of antiapoptotic proteins, suppressing local and systemic inflammatory responses and up-regulating neurotrophic factors. These mechanisms have not been directly correlated to mood stabilization in bipolar disorder but should be further explored given the correlation between white matter disease and manic symptoms.25 Some studies26 have hypothesized that the mechanism of valproic acid–associated weight gain is associated with increases in appetite and energy storage via up-regulation of pancreatic insulin secretion.
Atypical antipsychotics treat the manic symptoms associated with bipolar disorder by blocking dopamine D2 receptors; they also block 5-HT2A receptors, which are thought to decrease the incidence of extrapyramidal side effects.27 The mechanism for atypical antipsychotic–associated weight gain includes perturbation of multiple receptors such as 5-HT2c, H1, D2, M3, and α-adrenergic receptors.28 High- and low-risk atypical antipsychotics do not affect these receptors in the same way, which is reflected in their different side effect profiles.29 For example, high-risk atypical antipsychotics such as olanzapine were found to have high affinity for H1 receptors (consistent with associated weight gain), whereas low-risk atypical antipsychotics such as aripiprazole and ziprasidone were associated with low affinity for H1 receptors.29
Who Is More Likely to Sustain Weight Gain?
Patients with an elevated baseline body weight are more likely to gain weight while taking lithium.30,31 Thus, patients with elevated BMIs should be counseled on the risks of increased weight gain while on lithium. In contrast, patients with a low BMI who were treated with high-risk atypical antipsychotics gained more weight than patients with a high BMI.32 Moreover, 45% of underweight and normal-weight individuals increased their baseline body weight ≥ 7% with high-risk atypical antipsychotic treatment (olanzapine specifically) compared to only 20% of obese individuals.33 With both high-risk atypical antipsychotic and valproic acid use, women are more likely to gain weight than men.34 However, with valproic acid use, no difference exists in the treatment of adults versus children and teenagers. Regarding insulin resistance, patients with a family history of diabetes are at increased risk for developing glucose intolerance after starting high-risk atypical antipsychotics such as olanzapine and quetiapine.35
When Do Patients Start to Develop Side Effects
and How Much Weight Do They Gain?
Studies30 examining patients treated with lithium have shown that weight gain is maximal in the first 1 to 2 years. An open-label, nonrandomized chart review36 of patients receiving lithium (N = 70, treatment duration mean = 89 days, SD = 41 days) demonstrated a cumulative average weight gain of 6.3 kg (14 lb) with 77% of patients experiencing weight gain, 2.9% having no change, and 20% losing weight. Multiple studies37 showed that up to 20% of patients would gain ≥ 10 kg (22 lb). A double-blind study38 observed a mean weight gain of 4.2 kg (9 lb) on lithium versus 1.2 kg (3 lb) on placebo at 76 weeks (results nonsignificant, lithium n = 121, placebo: n = 121). Patients taking valproic acid can start to gain weight within the first 3 months, with a peak at 6 months.39 One study26 showed an increase of 0.49 kg (1 lb) in 21 days (N = 52, P = .03) in patients treated with valproic acid versus placebo.
Many of the atypical antipsychotics (both high and low risk) used as adjuvant therapy for patients with bipolar disorder are implicated in weight gain. Olanzapine is the most strongly associated, with an average weight gain of 2.3 kg (5 lb)/month. Others include quetiapine (1.8 kg/month), risperidone (1.0 kg (4 lb)/month), and ziprasidone (0.8 kg/month (2 lb)).40 All of the atypical antipsychotics, have the potential to cause sharp rises in body weight within the first few months. One case study41 described 2 months of aripiprazole treatment (commonly considered weight neutral) resulting in an 11-kg (24-lb) weight gain.
How Can Side Effects of Pharmacologic
Treatments for Bipolar Disorder Be Mitigated?
Given that the mechanism of lithium-associated weight gain is mostly unclear, the best treatment is prevention. Primary care physicians should be aware of this side effect before starting lithium and make routine exercise and dietary plans available for the patient. Given that strong thirst is a common side effect of lithium, patients should be advised to satisfy thirst with non- or low-calorie beverages. One case report42 documented the use of lithium in a patient whose weight increased from 68 kg (150 lb) to 80 kg (176 lb) after 9 months and to 93 kg (205 lb) after 2 years and 9 months of therapy. Initiation of metformin treatment in that patient led to an 8-kg (18-lb) weight loss after 1 year.42
Patients who gain more than 4 lb on valproic acid within the first month are advised to switch to a different mood-stabilizing medication. The amount of weight gained is independent of valproic acid dosages and levels; thus, similar guidelines on side effect management should apply to the treatment of bipolar disorder as well as epilepsy,39 specifically monitoring of body weight before and after initial administration of valproic acid therapy and considering change in medication if body weight increases by at least 4 lb after 1 month of treatment. Pharmacologic interventions with weight loss medications (eg, orlistat, metformin) and behavioral modifications have not proved effective in preventing weight gain in patients started on high-risk atypical antipsychotics.43 Some trials44 have shown that topiramate can reduce or prevent atypical antipsychotic–associated weight gain. The proposed mechanism of action is related to activation of γ-aminobutyric acid receptors and reduction of calorie deposition with reduced insulin levels.23 Thus, topiramate may be a useful treatment for primary care physicians who must manage a patient’s weight gain while on high-risk atypical antipsychotics. It is important to note that topiramate has a documented drug-drug interaction with metformin, requiring close monitoring of patients’ blood glucose levels.23
Metformin effectively improves insulin sensitivity that is often compromised by high-risk atypical antipsychotics such as olanzapine and sometimes quetiapine. Primary care physicians are therefore advised to start their patients on metformin concurrently with these antipsychotics to prevent diabetes mellitus; however, they may also consider whether the patient must remain on olanzapine or could be switched to low-risk atypical antipsychotics such as aripiprazole or ziprasidone (medications that would not require the patient to be on metformin).35
OBSTRUCTIVE SLEEP APNEA
There are 3 known types of sleep apnea: central, obstructive, and mixed. OSA is a disorder of sleep defined by the presence of interruptions or reductions in respiration (> 10 seconds in duration) during sleep associated with either oxygen desaturation or transient arousal from sleep. Clinically, the patient may describe awakenings from sleep with gasping or choking, daytime somnolence, witnessed breathing interruptions during sleep, or loud snoring.45 Of importance, patients may also be unaware of those symptoms and actually deny them. Thus, information from bed partners becomes invaluable. Additional symptoms to be aware of include morning headaches and daytime somnolence.46 OSA is classified as mild, moderate, or severe based on the number of obstructive respiratory events per hour, patients’ oxygen levels during those events, and severity of daytime somnolence.47
The obstructive type should be distinguished from central sleep apnea (eg, idiopathic, high altitude–induced, narcotic-induced, obesity hypoventilation syndrome, and Cheyne-Stokes breathing [commonly seen in patients with heart failure]).48 The mechanism behind central sleep apnea is different from OSA in that it is related to abnormalities in respiratory drive centers or carbon dioxide levels.49 Mixed sleep apnea is a combination of the etiologic factors present in obstructive and central sleep apnea.
Who Has Obstructive Sleep Apnea?
One complication of weight gain is OSA, a condition that affects at least 2%–4% of adults. Risk factors for OSA include obesity, large neck circumference, and being a middle-aged male or a woman in menopause.50 Patients who smoke and drink are also at increased risk of OSA.51 There also is mounting evidence that OSA is highly comorbid in patients with insulin resistance and cardiovascular morbidities, including hypertension, coronary artery disease, and atrial fibrillation.52 The Adult Obstructive Sleep Apnea Task Force of the Academy of Sleep Medicine (AASM) recommends diagnostic confirmation and severity qualification by objective testing for OSA in all patients with congestive heart failure, atrial fibrillation, type 2 diabetes, stroke, or a BMI ≥ 35.45 The absence of these characteristics, however, should not preclude attempts to diagnose OSA. For example, neck size and BMI may not always be adequate objective markers in the elderly population; those characteristics have a smaller influence on the Apnea-Hypopnea Index (AHI) in this population.53
What Predisposes to Obstructive Sleep Apnea?
Alterations in the anatomy of the upper airway, comprised by muscles and soft tissues, are the driving force in the development of OSA.54 Fiberoptic nasopharyngoscopy studies55 have found that the AHI correlates with the degree or shape of pharyngeal narrowing. Sleep endoscopy studies55 showed obstruction of the velopharynx in 89% of patients, with 72% of patients having multiple sites of obstruction from anatomic variations. Increased volumes of adipose tissue in the soft palate, parapharyngeal, and retroglossal regions are all significant predicators of OSA.56
It is also important to consider injury to muscles and neuronal fibers from repetitive opening and closing of the upper airway; this can lead to sensorimotor dysfunction with concurrent pharyngeal myopathy.57 This sensorineural adaptation can result in decreased responsiveness of the genioglossus negative pressure reflex, which is responsible for increasing upper airway dilator muscle tone to prevent airway collapse during sleep.58
Why Do We Screen for and
Treat Obstructive Sleep Apnea?
OSA has important effects on a patient’s cognitive well-being, work performance, and quality of life.51 Furthermore, this condition can result in pulmonary hypertension, which can be further complicated by acute hypoxemic respiratory failure, cor pulmonale, and death. Untreated OSA is estimated to result in a 2-fold increase in medical expenses due to related cardiovascular disease.59 Patients can also develop morning headaches and depressive symptoms with increased daytime somnolence and neurocognitive slowing that is associated with a higher risk of motor vehicle accidents.46 Treatment of OSA with continuous positive airway pressure (CPAP) (≥ 4 hours/night) resulted in a reduction of motor vehicle accidents (7.6 to 2.5 accidents/1,000 drivers/year).60 In the workplace, patients with OSA had significantly more difficulty concentrating on new tasks, learning new tasks, and performing monotonous tasks compared to non-OSA patients (OR = 7.5, 9.1, and 20, respectively).61 The risk of lost workdays was 1.6 and 1.8 times greater in men and women, respectively, with OSA compared to patients without OSA.61
How Do We Screen for Obstructive Sleep Apnea?
Common screening tools utilized by primary care physicians include the Epworth Sleepiness Scale,62 the STOP (Snoring, Tiredness, Observed apnea, high blood Pressure) Questionnaire,63 the STOP-Bang (BMI, Age, Neck circumference and Gender) questionnaire,63 and the Berlin Questionnaire.64,65 The Mallampati score, used to assess ease of endotracheal intubation, is also suggested as part of the physical examination before evaluating patients with polysomnography (PSG) (N = 137, 1-point increases in Mallampati score increased odds of having OSA by 2.5-fold).66 Acute dystonic reactions occur in 3%–10% of patients exposed to antipsychotics.67 Acute laryngeal-pharyngeal dystonia has been reported since the 1970s68–70 as a potential cause of asphyxia after haloperidol administration. Christodoulou and Kalaitzi71 reported 26 cases of acute laryngeal-pharyngeal dystonia associated with oral or parenteral first-generation antipsychotics. Raja and Azzoni72 reported significantly lower incidence of acute dystonic reactions in general (about 3%) with newer antipsychotics. Overall, the likelihood of this reaction occurring with olanzapine of any form appears low.
What Tests Do We Use to
Diagnose Obstructive Sleep Apnea?
Primary care physicians should recommend that their patients have either home sleep studies with portable monitoring or laboratory-based sleep studies. In the laboratory, a patient has the option of attending a split-night or a full-night PSG.73 The Portable Monitoring Task Force of the AASM recommends that those patients with a high pretest probability for moderate-severe OSA should undergo portable monitoring.74 Negative or inconclusive portable monitoring should be followed by PSG in these populations as portable monitoring has a high rate of false negatives.74 Of note, portable monitoring should only be performed in conjunction with a comprehensive sleep evaluation.
The sleep laboratory is generally responsible for choosing specific machinery. There are 4 types of sleep study monitors, characterized by the number of respiratory channels, used to diagnosis OSA.74 Type I is a facility-based PSG that also measures AHI; it is the gold standard for diagnosis, but it must only be used when clinical suspicion is high, as PSGs are costly and inconvenient for patients.74 Advantages to using home portable monitoring tests (types II–IV) are that they are cheaper alternatives and allow patients access from home; the disadvantage to using types III–IV is that they do not measure AHI.74
What Treatment Options Are Available
for Patients With Obstructive Sleep Apnea?
Lifestyle interventions—including diet and exercise with the goal of weight loss—with CPAP continue to be the gold standard for treating OSA. There is low-quality evidence supporting the use of a mandibular advancement device.75 Positional therapy, oropharyngeal exercises, palatal implants, surgical interventions, pharmacologic therapy, and atrial overdrive pacing were described by the American College of Physicians as having insufficient evidence to recommend as alternative treatments to CPAP.75
Why Do We Monitor Patients With
Bipolar Disorder for Obstructive Sleep Apnea?
Early diagnosis and monitoring of the side effects of medications used to treat bipolar disorder are critical. In a study76 of more than 4 million patients from the Veterans Health Administration data bank, 118,105 were diagnosed with OSA. Of those patients with OSA, bipolar disorder was identified in 3.3%.76 A chart review77 of 482 patients diagnosed with bipolar disorder and screened for OSA found a positive screen in 214 patients, and only 114 patients followed up with a sleep study. Of those 114 patients, 101 were diagnosed with OSA, suggesting the group prevalence is between 21% and 47.5%.77 Thus, given the high prevalence of OSA in patients with bipolar disorder, it would be wise to monitor for this condition.
How Do We Screen for the Development
of Obstructive Sleep Apnea in Patients
With Bipolar Disorder and Why?
In patients with bipolar disorder, self-report screening tools, like the Berlin Questionnaire,64 are often inaccurate, showing both a poor positive and negative predictive value.78 Therefore, use of a patient-operative portable monitoring device should be considered in patients with high pretest probabilities (based on previously discussed risk factors, including snoring, daytime somnolence, and obesity). This can provide an objective measure of AHI and is a more accurate tool for referring patients to additional in-laboratory PSG.78
We screen for OSA in patients with bipolar disorder for the same health-risk implications of OSA previously described. However, there are currently no guidelines for how often to screen for OSA in patients being treated for bipolar disorder. Plante and Winkelman79 recommended that all patients with bipolar disorder with any sleep complaints be screened. Routine observations based on appearance (obesity and large neck circumference) and questions about snoring or daytime somnolence should also prompt health care providers to screen for OSA. Given how common OSA is in patients with bipolar disorder and its health implications, it would be prudent for primary care physicians to consider screening routinely for this condition, especially after starting a medication that is associated with weight gain. However, there is no evidence to date on how many patients started on treatment for bipolar disorder should have a PSG or an in-home monitoring device. More research is required to determine if it would be cost efficient to have primary care physicians screen bipolar disorder patients with portable monitoring devices before any weight gain occurs. However, primary care physicians are encouraged to utilize the predictable patterns of certain medications’ side effects to monitor for the development of OSA, especially in the first year of a medication trial. If a patient shows weight stability after use of long-term medication treatment (> 1 year), screening by routine questioning and observations alone may suffice.
Which Medications Are Implicated
In the Development or Worsening
of Obstructive Sleep Apnea?
Lithium and valproic acid increase the risk of OSA through previously described mechanisms of weight gain; an independent mechanism for OSA on these medications has not been described. With regard to all atypical antipsychotics, several alternative mechanisms for the development of OSA have been suggested. As previously discussed, a common pharmacologic feature of the atypical antipsychotics is blockade of D2 and 5-HT2A receptors.80 Previous studies81 have shown serotonergic agonists to positively regulate central respiratory drive and increase airway diameter. Therefore, inhibition of this serotonin pathway is suggested to worsen OSA by increasing upper airway resistance; this, however, is only a theoretical risk.
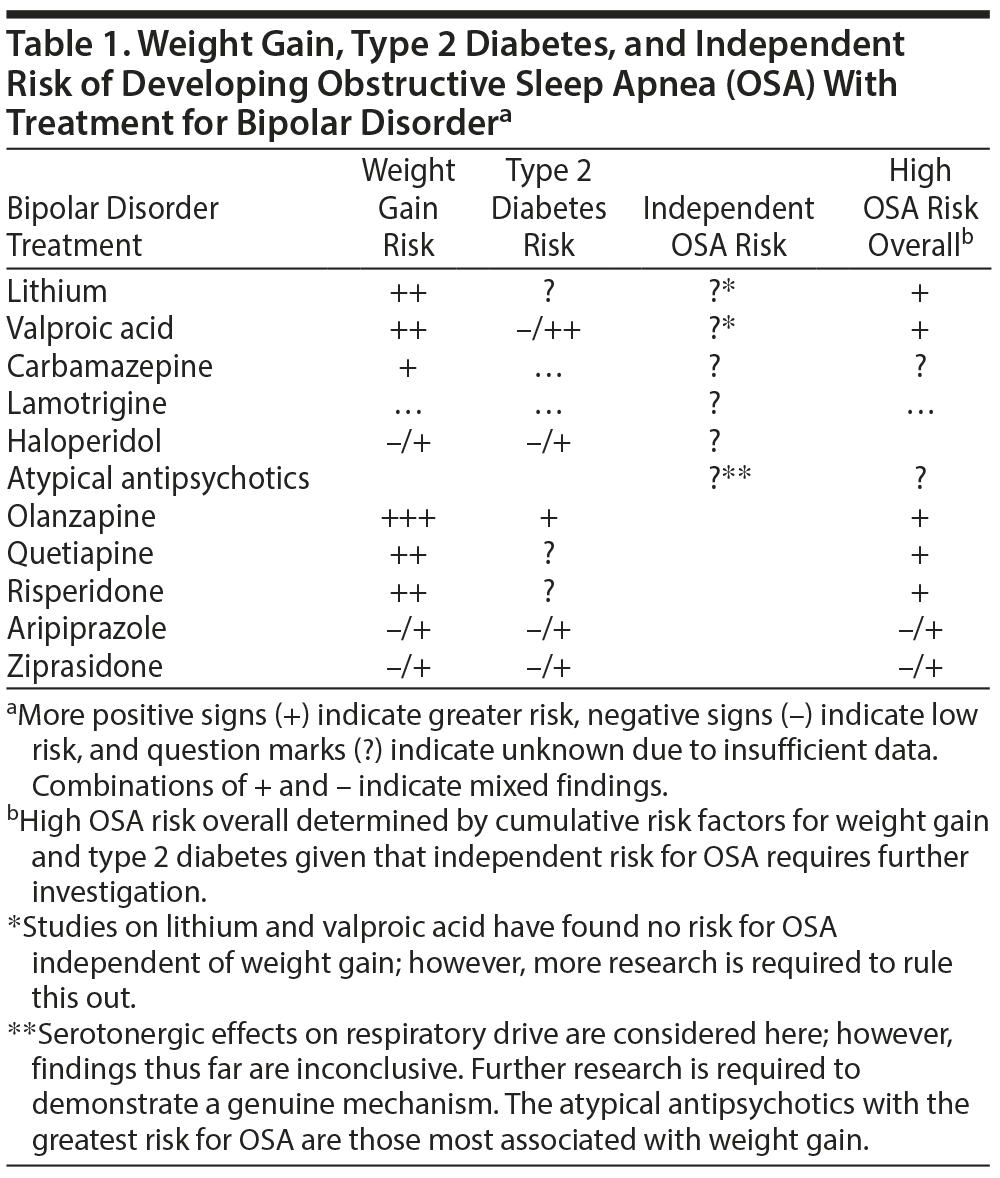
Furthermore, the ventilatory depression that can be induced by certain atypical antipsychotics (specifically quetiapine and olanzapine) can further exacerbate sleep-disordered breathing.77 As previously discussed, decreased reflex activation of the genioglossus through inhibition of laryngeal mechanoreceptors can result in airway collapse during sleep. The tranquilizing properties of high-risk atypical antipsychotics have been postulated to decrease laryngeal mechanoreceptor stimulation, thus decreasing upper airway tone and worsening symptoms of OSA.82 Thus, it becomes especially important to monitor patients treated with atypical antipsychotics for an exacerbation of OSA symptoms in the emergency setting. Quetiapine has been associated with acute respiratory dysfunction in OSA patients without comorbid bipolar disorder.5 A case report83 described an obese male diagnosed with a sleep-related disorder (likely OSA) who received a large dose of sedating medications and quetiapine that led to respiratory failure and death. Another case report84 described the administration of olanzapine that led to carbon dioxide narcosis with respiratory failure in an elderly patient with preexisting lung disease. These reports support careful early observation of patients with OSA or its risk factors who are started on high-risk atypical antipsychotics. A summary of these risks factors by medication is described in Table 1.32,85
Postinjection delirium/sedation syndrome is an uncommon adverse event occurring after the injection of olanzapine pamoate. It is reported to occur in approximately 0.05%–0.07% of injections and 0.75%–1.4% of patients86–88 and does not seem to occur with other long-acting injections.89 It is thought to be related to accidental intravascular entry of the drug secondary to vessel injury during the injection, given that the salt form is more soluble in blood than in muscle.90 There are limited cases91 of delirium from short-acting olanzapine injections. Delirium from oral olanzapine appears to also be rare and has been reported primarily in elderly people, presumably as a result of anticholinergic activity.92 In summary, it appears that postinjection delirium/sedation syndrome, while unique to the long-acting injectable olanzapine, is uncommon, suggesting safety for those who receive the medication.
How Do We Manage Patients With
Bipolar Disorder and Obstructive Sleep Apnea?
As previously discussed, lithium/valproic acid/high-risk atypical antipsychotic–related weight gain may not be the only mechanism for development of OSA. Nevertheless, lifestyle changes including diet and exercise continue to remain invaluable. For patients taking lithium, there may be additional value in starting medications such as metformin.42 Some benefit may be provided by switching patients to more low-risk atypical antipsychotics; however, primary care physicians should continue to think about OSA (not necessarily screen) with these medications given possible serotonergic antagonist mechanisms. Regardless of the psychiatric medication used for the treatment of bipolar disorder, CPAP continues to be a first-line treatment for OSA.93
Furthermore, primary care physicians are advised to regularly check the HbA1c in bipolar disorder patients diagnosed with OSA. OSA has been independently linked as a risk factor for the development of diabetes.94 High-risk atypical antipsychotics used to treat bipolar disorder can also lead to impaired glucose metabolism, resulting in type 2 diabetes. The mechanism of development of diabetes with atypical antipsychotic use includes antagonism of 5-HT2A receptors and increased free fatty acids; this results in decreased skeletal muscle and adipose tissue uptake of glucose, leading to insulin resistance, hyperglycemia, and diabetes mellitus.95
What Should We Be Aware of After Starting Continuous Positive Airway Pressure?
There is scant literature on the relationship between bipolar disorder and CPAP–induced mania. A possible explanation for this phenomenon is related to CPAP’s improvement of clock gene (hPer1) dysfunction caused by OSA.93 It may be useful for primary care physicians to observe patients for worsening manic symptoms in the setting of new CPAP treatment.96 In a study97 aimed at patients with bipolar disorder without OSA, actigraphy, which is useful in recording circadian rhythmicity and disruption, was suggested as a tool to estimate sleep and wakefulness in patients with clinical insomnia despite taking sedating medications (including mood stabilizers and antipsychotics). Given the ability of actigraphy to study circadian rhythms, it may prove useful to utilize this technology in patients with bipolar disorder who start CPAP for OSA to see if any specific changes in rhythmicity correlate to clinical features of mania.
CONCLUSION
Motor vehicle accidents, cognitive impairment, work disability/impaired function, and decreased quality of life are all documented adverse clinical outcomes of untreated OSA by the Adult OSA Task Force of the AASM.45 As previously mentioned, this task force recommends objective testing to diagnosis OSA in all patients with congestive heart failure, atrial fibrillation, type 2 diabetes, stroke, or a BMI ≥ 35. Here, we examined bipolar disorder management and how medication side effects may be implicated in the development of OSA. On the basis of this literature review, patients treated for bipolar disorder with lithium, valproic acid, or a high-risk atypical antipsychotic should be considered for type I or type II sleep study monitors to diagnose OSA. Our patient discussed previously would have greatly benefited from early intervention with CPAP after starting treatment for bipolar disorder. Our patient, like many others, developed OSA complicated by pulmonary hypertension. As primary care physicians treat and manage patients with bipolar disorder, we strongly recommend the aforementioned screening approaches for OSA and treatments that mitigate metabolic side effects.
REFERENCES
1. Forty L, Ulanova A, Jones L, et al. Comorbid medical illness in bipolar disorder. Br J Psychiatry. 2014;205(6):465–472. PubMed CrossRef
2. Jann MW. Diagnosis and treatment of bipolar disorders in adults: a review of the evidence on pharmacologic treatments. Am Health Drug Benefits. 2014;7:489–499. PubMed
3. Culpepper L. The role of primary care clinicians in diagnosing and treating bipolar disorder. Prim Care Companion J Clin Psychiatry. 2010;12:4–9. PubMed CrossRef
4. Chengappa KN, Suppes T, Berk M. Treatment of bipolar mania with atypical antipsychotics. Expert Rev Neurother. 2004;4(sup2):S17–S25. PubMed CrossRef
5. Freudenmann RW, Sussmuth SD, Wolf RC, et al. Respiratory dysfunction in sleep apnea associated with quetiapine. Pharmacopsychiatry. 2008;41(3):119–121. PubMed CrossRef
6. Severus E, Bauer M. Diagnosing bipolar disorders in DSM-5. Int J Bipolar Disord. 2013;1(1):14. PubMed CrossRef
7. American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders. Fifth Edition. Washington, DC: American Psychiatric Association; 2013.
8. Muneer A. Mixed states in bipolar disorder: etiology, pathogenesis and treatment. Chonnam Med J. 2017;53(1):1–13. PubMed CrossRef
9. Maletic V, Raison C. Integrated neurobiology of bipolar disorder. Front Psychiatry. 2014;5:98. PubMed CrossRef
10. Harrison PJ. Molecular neurobiological clues to the pathogenesis of bipolar disorder. Curr Opin Neurobiol. 2016;36:1–6. PubMed CrossRef
11. Kim Y, Santos R, Gage FH, et al. Molecular mechanisms of Bipolar Disorder: progress made and future challenges. Front Cell Neurosci. 2017;11:30. PubMed CrossRef
12. Adler CM, Adams J, DelBello MP, et al. Evidence of white matter pathology in bipolar disorder adolescents experiencing their first episode of mania: a diffusion tensor imaging study. Am J Psychiatry. 2006;163(2):322–324. PubMed CrossRef
13. Culpepper L. Misdiagnosis of bipolar depression in primary care practices. J Clin Psychiatry. 2014;75(03):e05. PubMed CrossRef
14. Kernberg OF, Yeomans FE. Borderline personality disorder, bipolar disorder, depression, attention deficit/hyperactivity disorder, and narcissistic personality disorder: Practical differential diagnosis. Bull Menninger Clin. 2013;77(1):1–22. PubMed CrossRef
15. Salloum IM, Cornelius JR, Daley DC, et al. Efficacy of valproate maintenance in patients with bipolar disorder and alcoholism: a double-blind placebo-controlled study. Arch Gen Psychiatry. 2005;62(1):37–45. PubMed CrossRef
16. Drugs for psychiatric disorders. Treat Guidel Med Lett. 2006;4:35–46. PubMed
17. Severus WE, Kleindienst N, Seemuller F, et al. What is the optimal serum lithium level in the long-term treatment of bipolar disorder–a review? Bipolar Disord. 2008;10(2):231–237. PubMed CrossRef
18. Vieta E, Sanchez-Moreno J. Acute and long-term treatment of mania. Dialogues Clin Neurosci. 2008;10:165–179. PubMed
19. Lampl Y, Eshel Y, Rapaport A, et al. Weight gain, increased appetite, and excessive food intake induced by carbamazepine. Clin Neuropharmacol. 1991;14(3):251–255. PubMed CrossRef
20. Coxhead N, Silverstone T, Cookson J. Carbamazepine versus lithium in the prophylaxis of bipolar affective disorder. Acta Psychiatr Scand. 1992;85(2):114–118. PubMed CrossRef
21. Biton V, Mirza W, Montouris G, et al. Weight change associated with valproate and lamotrigine monotherapy in patients with epilepsy. Neurology. 2001;56(2):172–177. PubMed CrossRef
22. Gildengers AG, Butters MA, Aizenstein HJ, et al. Longer lithium exposure is associated with better white matter integrity in older adults with bipolar disorder. Bipolar Disord. 2015;17(3):248–256. PubMed CrossRef
23. Gracious BL, Meyer AE. Psychotropic-induced weight gain and potential pharmacologic treatment strategies. Psychiatry. 2005;2:36–42 [Edgmont]. PubMed
24. Rosenberg G. The mechanisms of action of valproate in neuropsychiatric disorders: can we see the forest for the trees? Cell Mol Life Sci. 2007;64(16):2090–2103. PubMed CrossRef
25. Chen S, Wu H, Klebe D, et al. Valproic acid: a new candidate of therapeutic application for the acute central nervous system injuries. Neurochem Res. 2014;39(9):1621–1633. PubMed CrossRef
26. Martin CK, Han H, Anton SD, et al. Effect of valproic acid on body weight, food intake, physical activity and hormones: results of a randomized controlled trial. J Psychopharmacol. 2009;23(7):814–825. PubMed CrossRef
27. Reynolds GP. Receptor mechanisms of antipsychotic drug action in bipolar disorder—focus on asenapine. Ther Adv Psychopharmacol. 2011;1(6):197–204. PubMed CrossRef
28. Murray R, Correll CU, Reynolds GP, et al. Atypical antipsychotics: recent research findings and applications to clinical practice: proceedings of a symposium presented at the 29th Annual European College of Neuropsychopharmacology Congress, 19 September 2016, Vienna, Austria. Ther Adv Psychopharmacol. 2017;7(1 suppl):1–14. PubMed CrossRef
29. Miron IC, Baroana VC, Popescu F, et al. Pharmacological mechanisms underlying the association of antipsychotics with metabolic disorders. Curr Health Sci J. 2014;40:12–17. PubMed
30. Vestergaard P, Poulstrup I, Schou M. Prospective studies on a lithium cohort, 3: tremor, weight gain, diarrhea, psychological complaints. Acta Psychiatr Scand. 1988;78(4):434–441. PubMed CrossRef
31. Vendsborg PB, Bech P, Rafaelsen OJ. Lithium treatment and weight gain. Acta Psychiatr Scand. 1976;53(2):139–147. PubMed CrossRef
32. Bak M, Fransen A, Janssen J, et al. Almost all antipsychotics result in weight gain: a meta-analysis. PLoS One. 2014;9(4):e94112. PubMed CrossRef
33. Bushe CJ, Slooff CJ, Haddad PM, et al. Weight change by baseline BMI from three-year observational data: findings from the Worldwide Schizophrenia Outpatient Health Outcomes Database. J Psychopharmacol. 2013;27(4):358–365. PubMed CrossRef
34. Seeman MV. Secondary effects of antipsychotics: women at greater risk than men. Schizophr Bull. 2009;35(5):937–948. PubMed CrossRef
35. Schumann SA, Ewigman B. Can metformin undo weight gain induced by antipsychotics? J Fam Pract. 2008;57:526–530. PubMed
36. Chengappa KN, Chalasani L, Brar JS, et al. Changes in body weight and body mass index among psychiatric patients receiving lithium, valproate, or topiramate: an open-label, nonrandomized chart review. Clin Ther. 2002;24(10):1576–1584. PubMed CrossRef
37. Gitlin M. Lithium side effects and toxicity: prevalence and management strategies. Int J Bipolar Disord. 2016;4(1):27. PubMed CrossRef
38. Calabrese JR, Bowden CL, Sachs G, et al. A placebo-controlled 18-month trial of lamotrigine and lithium maintenance treatment in recently depressed patients with bipolar I disorder. J Clin Psychiatry. 2003;64(9):1013–1024. PubMed CrossRef
39. Verrotti A, D’Egidio C, Mohn A, et al. Weight gain following treatment with valproic acid: pathogenetic mechanisms and clinical implications. Obes Rev. 2011;12(5):e32–e43. PubMed CrossRef
40. Shrivastava A, Johnston ME. Weight-gain in psychiatric treatment: risks, implications, and strategies for prevention and management. Mens Sana Monogr. 2010;8(1):53–68. PubMed CrossRef
41. Singh T. Aripiprazole-induced weight gain. Psychiatry (Edgemont). 2005;2:19. PubMed
42. Praharaj SK. Metformin for lithium-induced weight gain: a case report. Clin Psychopharmacol Neurosci. 2016;14(1):101–103. PubMed CrossRef
43. Faulkner G, Cohn T, Remington G. Interventions to reduce weight gain in schizophrenia. Cochrane Database Syst Rev. 2007;CD005148. PubMed
44. Mahmood S, Booker I, Huang J, et al. Effect of topiramate on weight gain in patients receiving atypical antipsychotic agents. J Clin Psychopharmacol. 2013;33(1):90–94. PubMed CrossRef
45. Epstein LJ, Kristo D, Strollo PJ Jr, et al. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5:263–276. PubMed
46. Harding SM. Complications and consequences of obstructive sleep apnea. Curr Opin Pulm Med. 2000;6(6):485–489. PubMed CrossRef
47. Treating Sleep Apnea. A Review of the Research for Adults. Rockville, MD: Comparative Effectiveness Review Summary Guides for Consumers; 2005.
48. Eckert DJ, Jordan AS, Merchia P, et al. Central sleep apnea: pathophysiology and treatment. Chest. 2007;131(2):595–607. PubMed CrossRef
49. Costanzo MR, Khayat R, Ponikowski P, et al. Mechanisms and clinical consequences of untreated central sleep apnea in heart failure. J Am Coll Cardiol. 2015;65(1):72–84. PubMed CrossRef
50. Spicuzza L, Caruso D, Di Maria G. Obstructive sleep apnoea syndrome and its management. Ther Adv Chronic Dis. 2015;6(5):273–285. PubMed CrossRef
51. Punjabi NM. The epidemiology of adult obstructive sleep apnea. Proc Am Thorac Soc. 2008;5(2):136–143. PubMed CrossRef
52. Kohli P, Balachandran JS, Malhotra A. Obstructive sleep apnea and the risk for cardiovascular disease. Curr Atheroscler Rep. 2011;13(2):138–146. PubMed CrossRef
53. Lee W, Nagubadi S, Kryger MH, et al. Epidemiology of obstructive sleep apnea: a population-based perspective. Expert Rev Respir Med. 2008;2(3):349–364. PubMed CrossRef
54. Eckert DJ, Malhotra A. Pathophysiology of adult obstructive sleep apnea. Proc Am Thorac Soc. 2008;5(2):144–153. PubMed CrossRef
55. Owens RL, Eckert DJ, Yeh SY, et al. Upper airway function in the pathogenesis of obstructive sleep apnea: a review of the current literature. Curr Opin Pulm Med. 2008;14(6):519–524. PubMed CrossRef
56. Li Y, Lin N, Ye J, et al. Upper airway fat tissue distribution in subjects with obstructive sleep apnea and its effect on retropalatal mechanical loads. Respir Care. 2012;57(7):1098–1105. PubMed CrossRef
57. Patil SP, Schneider H, Schwartz AR, et al. Adult obstructive sleep apnea: pathophysiology and diagnosis. Chest. 2007;132(1):325–337. PubMed CrossRef
58. Horner RL, Innes JA, Morrell MJ, et al. The effect of sleep on reflex genioglossus muscle activation by stimuli of negative airway pressure in humans. J Physiol. 1994;476:141–151. PubMed
59. Tarasiuk A, Reuveni H. The economic impact of obstructive sleep apnea. Curr Opin Pulm Med. 2013;19(6):639–644. PubMed CrossRef
60. Karimi M, Hedner J, Habel H, et al. Sleep apnea-related risk of motor vehicle accidents is reduced by continuous positive airway pressure: Swedish Traffic Accident Registry data. Sleep. 2015;38(3):341–349. PubMed CrossRef
61. Hirsch Allen AJM, Bansback N, Ayas NT. The effect of OSA on work disability and work-related injuries. Chest. 2015;147(5):1422–1428. PubMed CrossRef
62. Wu S, Wang R, Ma X, et al. Excessive daytime sleepiness assessed by the Epworth Sleepiness Scale and its association with health related quality of life: a population-based study in China. BMC Public Health. 2012;12(1):849. PubMed CrossRef >
63. Chung F, Yang Y, Brown R, et al. Alternative scoring models of STOP-bang questionnaire improve specificity to detect undiagnosed obstructive sleep apnea. J Clin Sleep Med. 2014;10(9):951–958. PubMed CrossRef >
64. Thurtell MJ, Bruce BB, Rye DB, et al. The Berlin questionnaire screens for obstructive sleep apnea in idiopathic intracranial hypertension. J Neuroophthalmol. 2011;31(4):316–319. PubMed CrossRef
65. Jonas DE, Amick HR, Feltner C, et al. Screening for Obstructive Sleep Apnea in Adults: An Evidence Review for the US Preventive Services Task Force. Rockville, MD: Agency for Healthcare Research and Quality; 2017.
66. Nuckton TJ, Glidden DV, Browner WS, et al. Physical examination: Mallampati score as an independent predictor of obstructive sleep apnea. Sleep. 2006;29(7):903–908. PubMed CrossRef
67. Goga JK, Seidel L, Walters JK, et al. Acute laryngeal dystonia associated with aripiprazole. J Clin Psychopharmacol. 2012;32(6):837–839. PubMed CrossRef
68. Flaherty JA, Lahmeyer HW. Laryngeal-pharyngeal dystonia as a possible cause of asphyxia with haloperidol treatment. Am J Psychiatry. 1978;135(11):1414–1415. PubMed CrossRef
69. Menuck M. Laryngeal-pharyngeal dystonia and haloperidol. Am J Psychiatry. 1981;138(3):394–395. PubMed CrossRef
70. Ilchef R. Neuroleptic-induced laryngeal dystonia can mimic anaphylaxis. Aust N Z J Psychiatry. 1997;31(6):877–879. PubMed CrossRef
71. Christodoulou C, Kalaitzi C. Antipsychotic drug-induced acute laryngeal dystonia: two case reports and a mini review. J Psychopharmacol. 2005;19(3):307–311. PubMed CrossRef
72. Raja M, Azzoni A. Novel antipsychotics and acute dystonic reactions. Int J Neuropsychopharmacol. 2001;4(04):393–397. PubMed CrossRef
73. McArdle N, Grove A, Devereux G, et al. Split-night versus full-night studies for sleep apnoea/hypopnoea syndrome. Eur Respir J. 2000;15(4):670–675. PubMed CrossRef
74. Collop NA, Anderson WM, Boehlecke B, et al. Clinical guidelines for the use of unattended portable monitors in the diagnosis of obstructive sleep apnea in adult patients: Portable Monitoring Task Force of the American Academy of Sleep Medicine. J Clin Sleep Med. 2007;3:737–747. PubMed
75. Qaseem A, Holty JE, Owens DK, et al. Management of obstructive sleep apnea in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2013;159:471–483. PubMed
76. Sharafkhaneh A, Giray N, Richardson P, et al. Association of psychiatric disorders and sleep apnea in a large cohort. Sleep. 2005;28(11):1405–1411. PubMed CrossRef
77. Kelly T, Douglas L, Denmark L, et al. The high prevalence of obstructive sleep apnea among patients with bipolar disorders. J Affect Disord. 2013;151(1):54–58. PubMed CrossRef
78. Soreca I, Buttenfield JA, Hall MH, et al. Screening for obstructive sleep apnea in patients with bipolar I disorder: comparison between subjective and objective measures. Bipolar Disord. 2015;17(3):345–348. PubMed CrossRef
79. Plante DT, Winkelman JW. Sleep disturbance in bipolar disorder: therapeutic implications. Am J Psychiatry. 2008;165(7):830–843. PubMed CrossRef
80. Kuroki T, Nagao N, Nakahara T. Neuropharmacology of second-generation antipsychotic drugs: a validity of the serotonin-dopamine hypothesis. Prog Brain Res. 2008;172:199–212. PubMed CrossRef
81. Shirani A, Paradiso S, Dyken ME. The impact of atypical antipsychotic use on obstructive sleep apnea: a pilot study and literature review. Sleep Med. 2011;12(6):591–597. PubMed CrossRef
82. Rishi MA, Shetty M, Wolff A, et al. Atypical antipsychotic medications are independently associated with severe obstructive sleep apnea. Clin Neuropharmacol. 2010;33(3):109–113. PubMed CrossRef
83. Fleischman JK, Ananthamoorthy R, Greenberg H, et al. An unexplained death in the psychiatric emergency room: a case of undiagnosed obstructive sleep apnea? Gen Hosp Psychiatry. 2008;30(1):83–86. PubMed CrossRef
84. Mouallem M, Wolf I. Olanzapine-induced respiratory failure. Am J Geriatr Psychiatry. 2001;9(3):304–305. PubMed CrossRef
85. Correll CU, Detraux J, De Lepeleire J, et al. Effects of antipsychotics, antidepressants and mood stabilizers on risk for physical diseases in people with schizophrenia, depression and bipolar disorder. World Psychiatry. 2015;14(2):119–136. PubMed CrossRef
86. Detke HC, McDonnell DP, Brunner E, et al. Post-injection delirium/sedation syndrome in patients with schizophrenia treated with olanzapine long-acting injection, I: analysis of cases. BMC Psychiatry. 2010;10(1):43. PubMed CrossRef
87. Jones ME, Andrews JS, Faries DE, et al. Baseline characteristics and hospitalizations in patients with schizophrenia receiving olanzapine long-acting injection: an interim analysis from a non-interventional, prospective observational safety study. BMC Psychiatry. 2015;15(1):278. PubMed CrossRef
88. Eli Lilly Company Limited. Summary of Product Characteristics (SPC)—ZYPADHERA 210 mg, 300 mg, and 405 mg, powder and solvent for prolonged release suspension for injection. eMC website. https://www.medicines.org.uk/emc/medicine/21361. 2016.
89. Alphs L, Gopal S, Karcher K, et al. Are the long-acting intramuscular formulations of risperidone or paliperidone palmitate associated with post-injection delirium/sedation syndrome? an assessment of safety databases. Curr Drug Saf. 2011;6(1):43–45. PubMed CrossRef
90. McDonnell DP, Detke HC, Bergstrom RF, et al. Post-injection delirium/sedation syndrome in patients with schizophrenia treated with olanzapine long-acting injection, II: investigations of mechanism. BMC Psychiatry. 2010;10(1):45. PubMed CrossRef
91. Marder SR, Sorsaburu S, Dunayevich E, et al. Case reports of postmarketing adverse event experiences with olanzapine intramuscular treatment in patients with agitation. J Clin Psychiatry. 2010;71(04):433–441. PubMed CrossRef
92. Aggarwal A, Sharma RC. Delirium associated with olanzapine therapy in an elderly man with bipolar affective disorder. J Neuropsychiatry Clin Neurosci. 2011;23(2):E54–E55. PubMed CrossRef
93. Heck T, Zolezzi M. Obstructive sleep apnea: management considerations in psychiatric patients. Neuropsychiatr Dis Treat. 2015;11:2691–2698. PubMed
94. Pamidi S, Tasali E. Obstructive sleep apnea and type 2 diabetes: is there a link? Front Neurol. 2012;3:126. PubMed CrossRef
95. Buchholz S, Morrow AF, Coleman PL. Atypical antipsychotic-induced diabetes mellitus: an update on epidemiology and postulated mechanisms. Intern Med J. 2008;38(7):602–606. PubMed CrossRef
96. Aggarwal R, Baweja R, Saunders EF, et al. CPAP-induced mania in bipolar disorder: a case report. Bipolar Disord. 2013;15(7):803–807. PubMed CrossRef
97. Kaplan KA, Talbot LS, Gruber J, et al. Evaluating sleep in bipolar disorder: comparison between actigraphy, polysomnography, and sleep diary. Bipolar Disord. 2012;14(8):870–879. PubMed CrossRef
LESSONS LEARNED AT THE INTERFACE
OF MEDICINE AND PSYCHIATRY
The Psychiatric Consultation Service at Massachusetts General Hospital (MGH) sees medical and surgical inpatients with comorbid psychiatric symptoms and conditions. During their twice-weekly rounds, Dr Stern and other members of the Consultation Service discuss diagnosis and management of hospitalized patients with complex medical or surgical problems who also demonstrate psychiatric symptoms or conditions. These discussions have given rise to rounds reports that will prove useful for clinicians practicing at the interface of medicine and psychiatry.
Mr Moonga is a fourth-year medical student at Stony Brook School of Medicine, Stony Brook, New York. Dr Mischoulon is the director of the Depression Clinical and Research Program, Department of Psychiatry, Massachusetts General Hospital and professor of psychiatry at Harvard Medical School, Boston, Massachusetts. Dr Winkelman is an associate professor of psychiatry at Harvard Medical School and medical director of the Sleep Health Center, Brigham and Women’s Hospital, Boston, Massachusetts. Dr Stern is chief emeritus of the Avery D. Weisman Psychiatry Consultation Service, director of the Thomas P. Hackett Center for Scholarship in Psychosomatic Medicine, director of the Office of Clinical Careers at Massachusetts General Hospital, and the Ned H. Cassem professor of psychiatry in the field of psychosomatic medicine/consultation at Harvard Medical School, Boston, Massachusetts, and is the editor in chief of Psychosomatics.
Prim Care Companion CNS Disord 2017;19(6):17f02194
https://doi.org/10.4088/PCC.17f02194
© Copyright 2017 Physicians Postgraduate Press, Inc.
Submitted: July 12, 2017; accepted August 28, 2017.
Published online: November 30, 2017.
Potential conflicts of interest: Dr. Stern is an employee of the Academy of Psychosomatic Medicine and has received royalties from Elsevier and the Massachusetts General Hospital Psychiatry Academy. Drs Mischoulon and Winkelman and Mr Moonga report no conflicts of interest related to the subject of this article.
Funding/support: None.
Corresponding author: Theodore A. Stern, MD, Department of Psychiatry, Massachusetts General Hospital, Fruit St, WRN 605, Boston, MA 02114 ([email protected]).
Enjoy this premium PDF as part of your membership benefits!

