Objective: The current study investigates whether milnacipran is effective in reducing pain and improving function in patients with persistent pain ≥ 1 year after total knee arthroplasty.
Method: This was a 12-week open-label study of flexibly dosed milnacipran in patients (N = 5) experiencing chronic persistent knee pain ≥ 1 year following total knee arthroplasty in the absence of new injury, infection, or implant failure. Subjects were identified from October 2010 to August 2011 through the Duke University Medical Center orthopedic clinic (Durham, North Carolina), typically during 1-year postoperative follow-up visits, and were referred by their orthopedic surgeon.
Results: Milnacipran treatment was associated with reduction in pain according to the primary outcome measure of the visual analog scale (VAS) score for pain (effect size of 1.15) and secondary outcome measures of Knee Society Score (KSS) evaluation subscale score (effect size of 1.37) and Medical Outcomes Study 36-item Short-Form Health Survey (SF-36) bodily pain subscale (effect size of 1.16) at week 12. Secondary outcome measures of functional change were mixed in such that, at week 12, the SF-36 physical functioning subscale showed improvement (effect size of 1.16), but the KSS function subscale score was just below the threshold for meaningful effect size (0.98).
Conclusions: Open-label milnacipran demonstrated reduced pain and some evidence of functional improvement in this small sample of patients with chronic persistent pain 1 year or more after total knee arthroplasty such that well-powered studies are warranted.
Trial Registration: ClinicalTrials.gov identifier: NCT01780389
Prim Care Companion CNS Disord 2013;15(4): doi:10.4088/PCC.12m01496
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: November 23, 2012; accepted February 22, 2013.
Published online: July 11, 2013.
Corresponding author: David M. Marks, MD, Department of Psychiatry, Duke University Medical Center, Box 3333, Durham NC 27705 ([email protected]).
Open-Label Milnacipran for Patients With Persistent Knee Pain
1 Year or Longer After Total Knee Arthroplasty: A Pilot Study
ABSTRACT
Objective: The current study investigates whether milnacipran is effective in reducing pain and improving function in patients with persistent pain ≥ 1 year after total knee arthroplasty.
Method: This was a 12-week open-label study of flexibly dosed milnacipran in patients (N = 5) experiencing chronic persistent knee pain ≥ 1 year following total knee arthroplasty in the absence of new injury, infection, or implant failure. Subjects were identified from October 2010 to August 2011 through the Duke University Medical Center orthopedic clinic (Durham, North Carolina), typically during 1-year postoperative follow-up visits, and were referred by their orthopedic surgeon.
Results: Milnacipran treatment was associated with reduction in pain according to the primary outcome measure of the visual analog scale (VAS) score for pain (effect size of 1.15) and secondary outcome measures of Knee Society Score (KSS) evaluation subscale score (effect size of 1.37) and Medical Outcomes Study 36-item Short-Form Health Survey (SF-36) bodily pain subscale (effect size of 1.16) at week 12. Secondary outcome measures of functional change were mixed in such that, at week 12, the SF-36 physical functioning subscale showed improvement (effect size of 1.16), but the KSS function subscale score was just below the threshold for meaningful effect size (0.98).
Conclusions: Open-label milnacipran demonstrated reduced pain and some evidence of functional improvement in this small sample of patients with chronic persistent pain 1 year or more after total knee arthroplasty such that well-powered studies are warranted.
Trial Registration: ClinicalTrials.gov identifier: NCT01780389
Prim Care Companion CNS Disord 2013;15(4): doi:10.4088/PCC.12m01496
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: November 23, 2012; accepted February 22, 2013.
Published online: July 11, 2013.
Corresponding author: David M. Marks, MD, Department of Psychiatry, Duke University Medical Center, Box 3333, Durham NC 27705 ([email protected]).
Total knee arthroplasty is an effective and widely used surgical treatment for osteoarthritis. Approximately 300,000 total knee arthroplasties are performed annually in the United States,1 and this number is expected to rise as the population ages, with projections of up to 1.5 million annual operations by 2020.2 Despite total knee arthroplasty being regarded as effective, a significant number of postoperative patients continue to experience pain and functional impairment. A large Canadian survey study3 revealed that only 70% of patients felt their expectations had been met 1 year after undergoing total knee arthroplasty, and post–total knee arthroplasty respondents reported continued pain, stiffness, and physical dysfunction. Brander et al4 demonstrated that approximately 1 in 8 patients is dissatisfied with the results of total knee arthroplasty due to chronic persistent pain as much as 1 year postoperatively in the face of radiographic evidence of successful surgery without postsurgical complications; these patients utilized more health care resources including physician visits, physical therapy, and manipulations. Brander et al4 reported that persistent pain and joint dysfunction 1 year after total knee arthroplasty measured by the visual analog scale (VAS) and Knee Society Score (KSS) were associated with higher scores on measures of depression and anxiety including the Beck Depression Inventory (BDI) and State-Trait Anxiety Inventory (STAI). Similarly, Anderson et al5 reported an approximately 90% satisfaction rate (reported 12–67 months following total knee arthroplasty) in patients > 75 years of age, and dissatisfaction correlated with poorer mental health scores, as well as decreased physical function and increased bodily pain scores. Although it appears that persistent pain after total knee arthroplasty is potentially linked to mood and anxiety levels, its pathophysiology remains mysterious, and treatment interventions for persistent pain after total knee arthroplasty have not been systematically studied.
Milnacipran is a serotonin-norepinephrine reuptake inhibitor (SNRI) uniquely shown to inhibit the reuptake of serotonin and norepinephrine (in vitro and in vivo) with approximately equal potency.6,7 Milnacipran has demonstrated efficacy in pain syndromes such as fibromyalgia8,9 and orofacial pain10 and has shown benefit in case reports for phantom limb pain, trigeminal neuralgia, and chronic pain of various causes.11 Other SNRIs have yielded positive results in studies of multiple pain syndromes, including, but not limited to, fibromyalgia,12 “functional” chest pain and dyspepsia,13,14 diabetic neuropathy,15–17 headache,18 and, perhaps most relevant to the current study, osteoarthritis of the knee.19,20 SNRIs, including milnacipran, are believed to inhibit pain by activating descending serotonergic and noradrenergic pathways from brainstem nuclei, and this process may be most effective when ascending pathways are sensitized.21 With regard to persistent pain after total knee arthroplasty, the relative contributions of peripheral nociception, as well as peripheral and central sensitization, are unclear.
Milnacipran (similar to other SNRIs) has demonstrated efficacy in depressive disorders and in anxiety.22 The efficacy of milnacipran in the treatment of major depressive disorder (MDD) has been established in a number of double-blind, placebo-controlled clinical trials,23,24 as well as in a series of randomized, double-blind, comparator studies using tricyclic antidepressants (amitriptyline,25 imipramine,26–28 clomipramine29,30) or selective serotonin reuptake inhibitors (SSRIs) (fluoxetine,31–33 fluvoxamine,34,35 paroxetine,36 sertraline37). Although randomized controlled trials of milnacipran in anxiety disorders have not been published to date, milnacipran has shown efficacy in an open-label study of panic disorder,38 and milnacipran has been demonstrated to reduce anxiety-specific items in depression rating scales in studies of MDD.23,24 Research with antidepressants, including milnacipran, on pain syndromes has typically shown that the analgesic effects of these drugs are independent of any changes in mood or anxiety symptoms.39–41 As such, the current study posits that milnacipran may have analgesic effects in patients with persistent pain after total knee arthroplasty, presumably due to the enhancement of descending inhibitory pain pathways.
METHOD
Study Design
The study was designed as a 12-week open-label study of flexibly dosed milnacipran on pain and functional outcomes in patients who experience chronic persistent knee pain 1 year or longer following total knee arthroplasty. The protocol was conducted at Duke University Medical Center, Durham, North Carolina, and approved by the local Internal Review Board (Clinicaltrials.gov Identifier: NCT01780389). All subjects provided written informed consent after the study was explained to them.
Subjects
Subjects were identified from October 2010 to August 2011 through the Duke University Medical Center orthopedic clinic, typically during 1-year postoperative follow-up visits, and were referred by their orthopedic surgeon. Key inclusion criteria included (1) chronic persistent pain ≥ 1 year after total knee arthroplasty without history of new injury, infection, or implant failure and (2) VAS score ≥ 40 mm (out of 100 mm) at screen and baseline visits. Key exclusion criteria included (1) history of bipolar disorder or psychosis as confirmed by the Mini-International Neuropsychiatric Interview (MINI),42 (2) current or recent (within the last 6 months) drug dependence or substance abuse disorder according to DSM-IV-TR criteria (excluding nicotine),43 and (3) treatment with antidepressant medication within 4 weeks of screening visit (6 weeks for fluoxetine).
Interventions
After a brief screening period to determine subject eligibility, appropriate subjects received oral milnacipran according to a fixed schedule for 2 weeks followed by flexible dosing (50–100 mg twice daily) based on efficacy and tolerability (day 1: 12.5 mg in the morning; days 2–3: 12.5 mg twice daily; days 4–7: 25 mg twice daily; days 8–14: 50 mg twice daily, if tolerated; day 15–84: 25 mg, 50 mg, 75 mg, or 100 mg twice daily). At the termination of the study, the protocol specified a taper of milnacipran unless subjects elected to continue on milnacipran following the study.
Outcome Measures
The primary outcome measure for the study was change in mean VAS pain score (0–100 mm) between baseline and week 12. Secondary outcome measures included both subscales of the KSS44 (evaluation score measuring subjective pain and function score measuring range of motion), self-report Global Rating of Change (GROC),45 Medical Outcomes Study 36-item Short-Form Health Survey (SF-36) as a quality of life measure,46 and the 20-item Multidimensional Fatigue Inventory (MFI-20).47 To evaluate whether any observed changes in pain/function outcome measures were confounded by changes in anxiety or depression, the self-report BDI,48 rater-administered Montgomery-Asberg Depression Rating Scale (MADRS),49 and self-report STAI50 were included as additional secondary measures.
Statistical Analysis
Analysis was performed at multiple time points: baseline and 1, 2, 4, 6, 8, 10, and 12 weeks. As this was a pilot study with a small sample size, a standardized effect size statistic was used to interpret the results. A baseline comparison was performed at week 4 and week 12 to assess short-term and long-term effects. A conservative cutoff score of 1.0 was used to indicate a meaningful clinical effect. Missing data were carried forward from the last available visit. For the KSS, ≥ 90 points was considered excellent, 80–89 was considered good, 70–79 was considered fair, and < 70 was considered poor.51,52
RESULTS
Individual subject demographic information and baseline scores on all outcome measures are shown in Table 1. Subjects uniformly scored low on depression rating scales, and no subjects met diagnostic criteria for MDD on the MINI. One subject scored moderately high on the STAI, but similar to other subjects, did not exhibit diagnostic criteria for an anxiety disorder on the MINI.
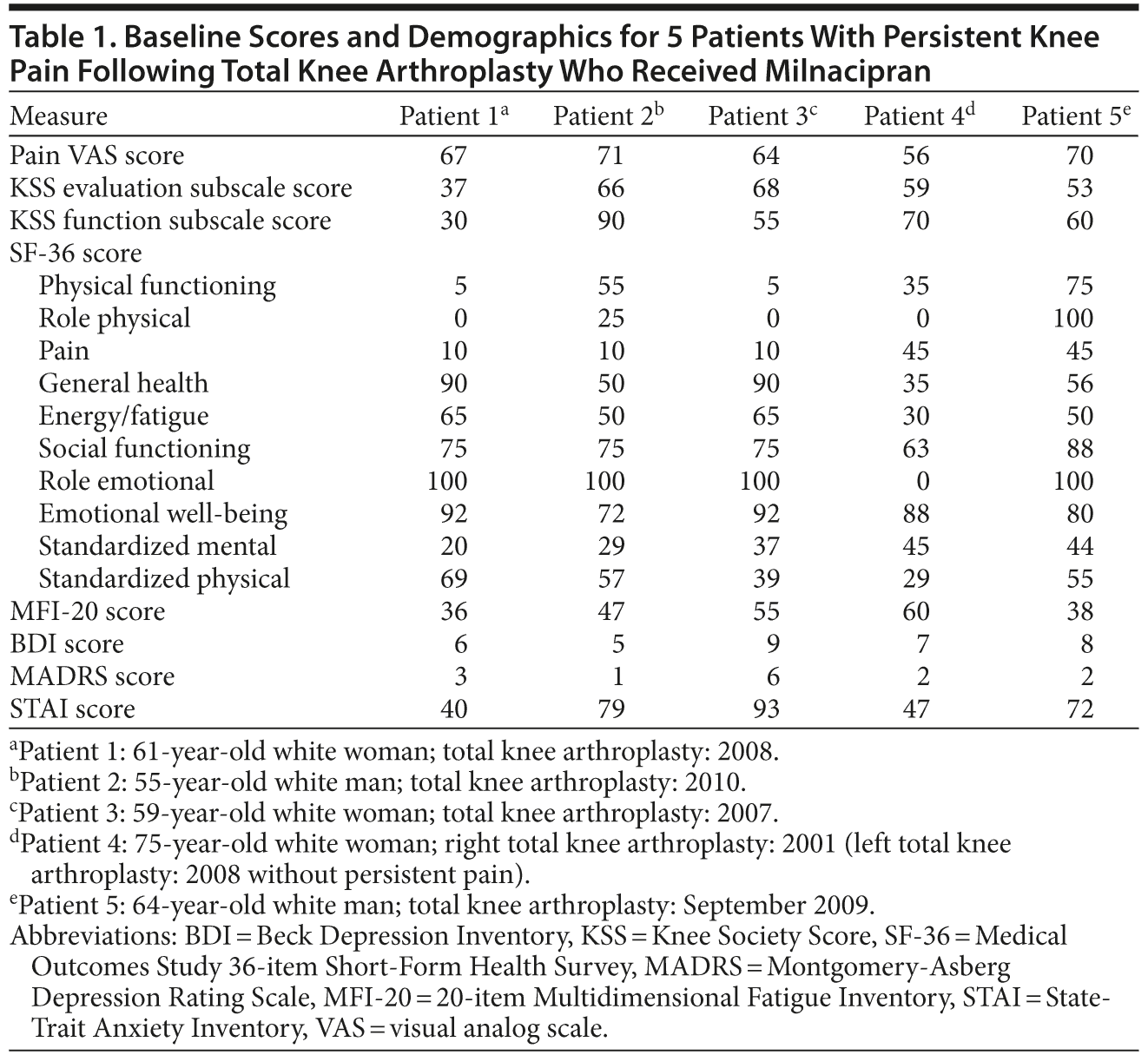
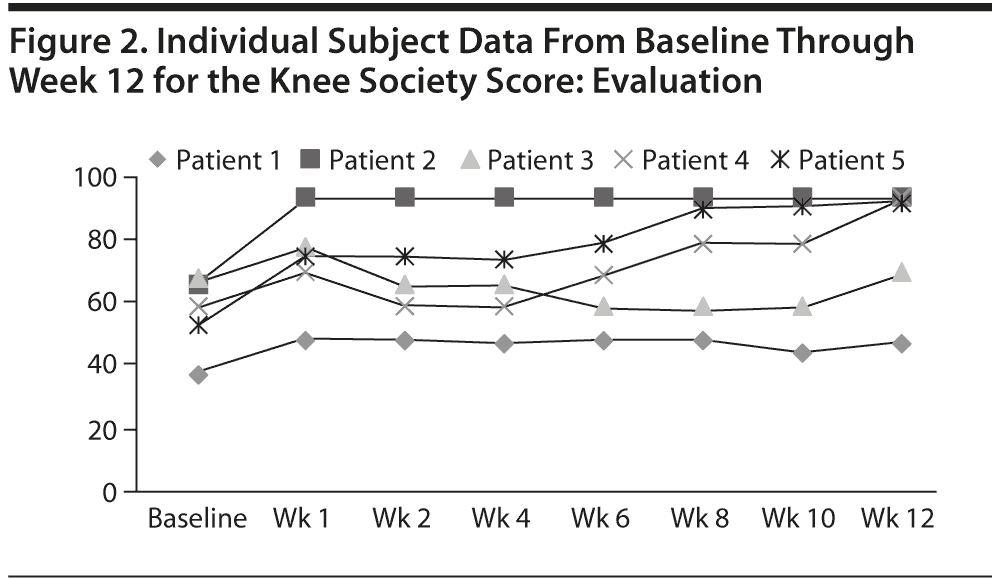
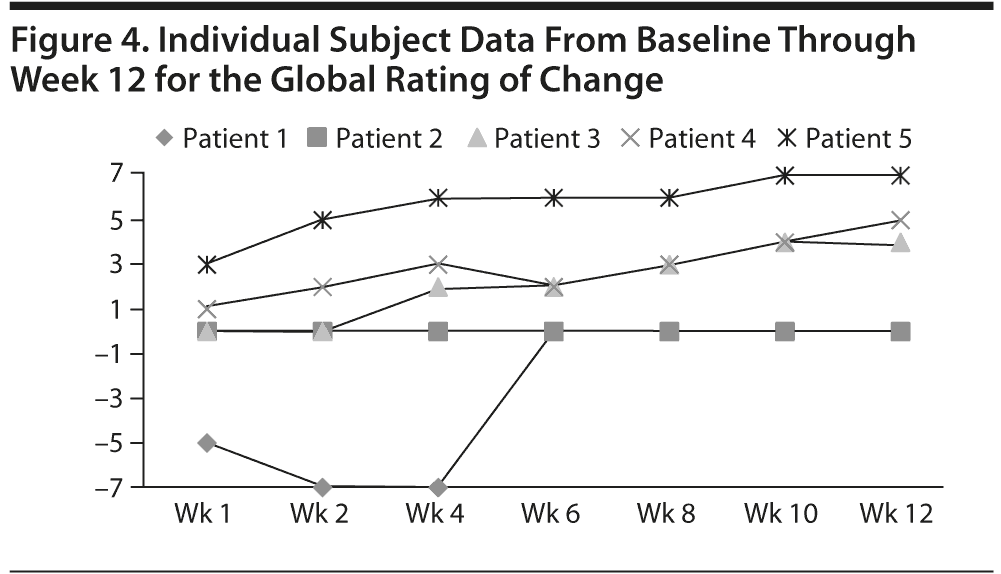
Individual subject data throughout the study for the VAS (Figure 1), KSS evaluation (Figure 2), KSS function (Figure 3), and GROC (Figure 4) are presented. One subject experienced an increase in VAS score during the trial for unclear reasons, while 4 subjects experienced a decrease. Effect sizes for all outcome measures at week 4 and week 12 are shown in Table 2.
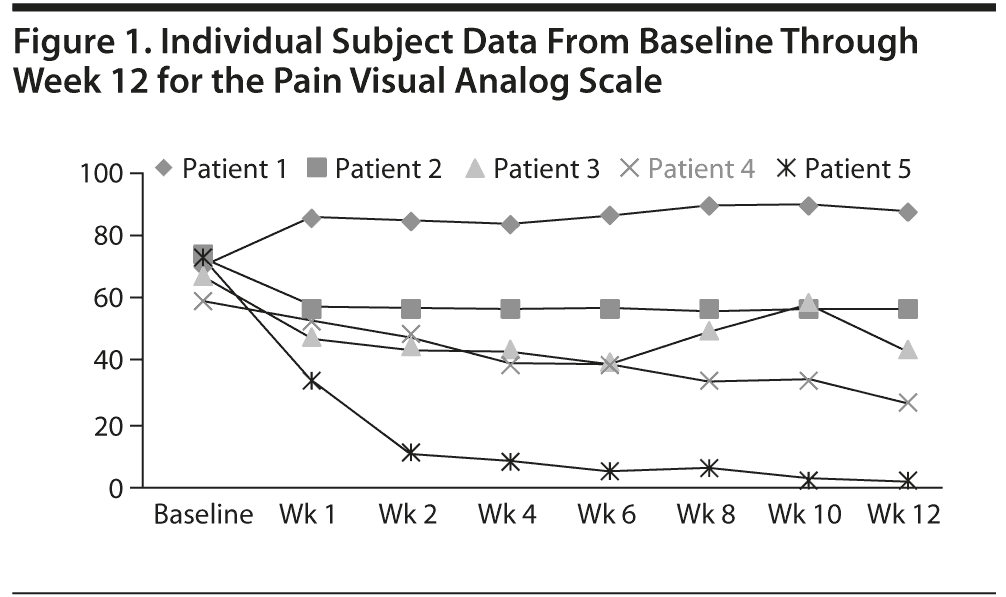
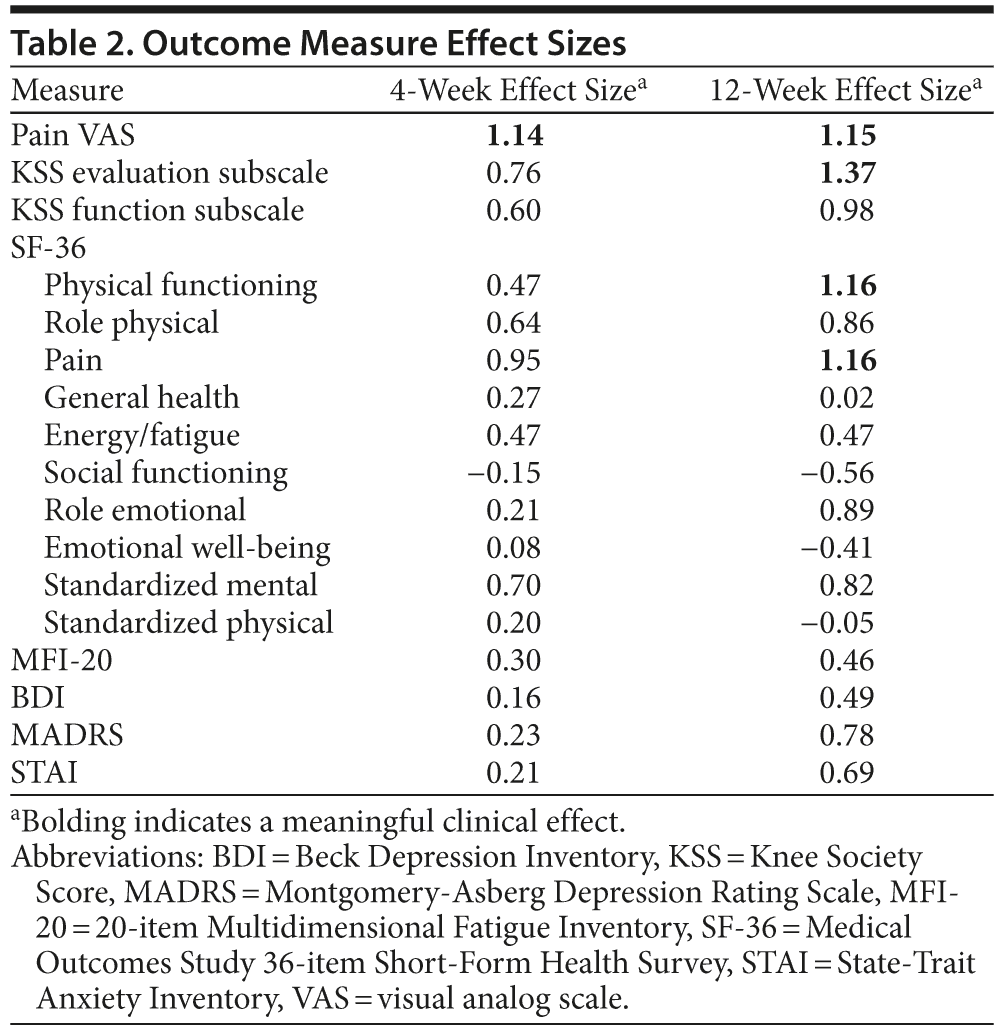
Pain measures consistently demonstrated a meaningful clinical effect at endpoint week 12, including the primary outcome measure of VAS pain score (1.14 at week 4, 1.15 at week 12) and the secondary outcome measure of KSS evaluation subscale score (0.76 at week 4, 1.37 at week 12). At baseline, all subjects were classified as having a poor result on the KSS evaluation subscale, whereas at week 12, 3 were classified as excellent, 1 was classified as fair, and 1 remained classified as poor. Additionally, a meaningful clinical effect was observed on the SF-36 bodily pain subscale (0.95 at week 4, 1.16 at week 12). As noted, the primary outcome measure of VAS pain score demonstrated meaningful clinical effect on pain at week 4, although the secondary measures did not.
Functional measures showed mixed results (Table 2) in that the SF-36 physical functioning subscale demonstrated a meaningful clinical effect at endpoint (0.47 at week 4, 1.16 at week 12), while the KSS function subscale narrowly missed our defined criterion for meaningful improvement (0.60 at week 4, 0.98 at week 12). The SF-36 subscales of bodily pain and physical functioning were the only subscales to show meaningful change.
Three of 5 subjects achieved a meaningful clinical change on the GROC (≥ 4 = moderately better, Figure 4). There were no significant changes per effect size in fatigue on the MFI-20 (0.30 at week 4, 0.46 at week 12) or on any psychological measure.
Spontaneously reported adverse events that were considered to be potentially related to milnacipran included nausea (1 subject), headache, (1 subject), constipation (1 subject), and painful ejaculation (1 subject who discontinued due to this adverse event). No serious adverse events occurred.
At the end of study, patient 3 (titrated to 75 mg twice daily) and patient 5 (titrated to 50 mg twice daily) opted to continue on milnacipran. Patient 4 (titrated to 100 mg twice daily) achieved improvement in knee pain but declined to continue on milnacipran after the study due to the perception that it did not alleviate other osteoarthritic joint pain (wrist, lumbar). Patient 2 (titrated to 50 mg twice daily) terminated the study early due to an adverse event. Patient 1 (titrated to a dose of 75 mg twice daily) completed the study but did not elect to continue milnacipran due to lack of efficacy.
DISCUSSION
The current pilot study has the chief limitation of a small sample size, which is insufficient for statistical hypothesis testing. Nonetheless, the observed effect sizes are compelling in that, overall, a meaningful clinical improvement was demonstrated in all measures of pain and in some measures of function. Additionally, the majority of subjects reported meaningful improvement via the GROC. A strength of this study is the inclusion of subjects with long-standing knee pain occurring persistently for at least 1 year after total knee arthroplasty such that it is unlikely that observed improvement represents spontaneous remission or tissue healing. However, since no placebo group was included in the study, placebo effects cannot be excluded. An additional strength of the study was the inclusion of mood and anxiety rating scales; consistent with previous research on SNRIs, in the current study, it does not appear that improvements in pain are due to changes in depressive or anxiety symptoms.
Although this study was limited in its analysis due to a small sample size, the notion of exploring the efficacy and tolerability of milnacipran in persistent pain after total knee arthroplasty is relevant for multiple reasons. First, milnacipran and other SNRIs have shown benefit in a variety of pain syndromes as described, and milnacipran may be particularly well suited as an analgesic in light of its equipotent serotonin and norepinephrine reuptake inhibition. In preclinical animal models, milnacipran has shown superior effects of ameliorating hyperalgesia and allodynia compared to some other antidepressant drugs.21 Persistent pain results from changes in sensitivity within both ascending and descending pain pathways in the brain and the spinal cord, and both serotonin and norepinephrine are implicated in modulating descending inhibitory pain pathways in the central nervous system.
Furthermore, persistent pain after total knee arthroplasty is linked to depression and anxiety epidemiologically (as described) if not pathophysiologically. As such, it seems that it would be useful to identify treatments that are effective for mood and anxiety symptoms/disorders and persistent pain after total knee arthroplasty due to high frequency of comorbidity.
As has been proposed in fibromyalgia,53 it is possible that depression/anxiety and persistent pain after total knee arthroplasty share pathophysiologic mechanisms, including genetic predisposition and neuroendocrine abnormalities that predispose toward psychiatric disorder and disorders of central pain sensitization. As described, the relative contribution of central sensitization, peripheral sensitization, and other physiologic processes is mysterious with respect to persistent pain after total knee arthroplasty. Despite the link between depression and anxiety and post–total knee arthroplasty pain, it should be noted that the current study demonstrates improvement in post–total knee arthroplasty pain in the absence of psychiatric diagnosis (after comprehensive psychiatric interview and MINI assessment) or meaningful change in scores of depression and anxiety rating scales. This result is consistent with multiple studies showing that the analgesic effects of antidepressants (including SNRIs and tricyclic antidepressants) are not due simply to improvements in depressive or anxiety symptoms.54 Clinicians should be aware that SNRIs such as milnacipran have the potential to interact with other serotonergic agents to cause serotonin syndrome; as such, milnacipran should not be prescribed with monoamine oxidase inhibitors (infrequently used antidepressants) and should be prescribed with some caution with triptans (used for migraine abortive treatment). Risk of serotonin syndrome should be considered but is less significant with concomitant use of other agents that increase serotonin by the same mechanism as milnacipran (reuptake inhibition) such as trazodone, tramadol, and SSRIs.
In summary, the current open-label study of milnacipran in 5 patients with persistent pain after total knee arthroplasty demonstrates clinically meaningful improvement in pain and some evidence of functional improvement as well. In light of the degree of improvement observed, the well-established efficacy of milnacipran and other SNRIs in pain syndromes, and the frequent comorbidities of depressive and anxiety disorders, well-powered placebo-controlled studies of milnacipran in persistent pain after total knee arthroplasty are warranted.
Drug names: clomipramine (Anafranil and others), fluoxetine (Prozac and others), fluvoxamine (Luvox and others), imipramine (Tofranil and others), milnacipran (Savella), paroxetine (Paxil, Pexeva, and others), sertraline (Zoloft and others), tramadol (Ultram, Ryzolt, and others), trazodone (Oleptro and others).
Author affiliations: Departments of Psychiatry (Dr Marks) and Orthopaedic Surgery (Dr Bolognesi), Duke University Medical Center, Durham, North Carolina.
Potential conflicts of interest: Dr Bolognesi has received grant/research support from Forest. Dr Marks reports no conflicts of interest related to the subject of this article.
Funding/support: This study was sponsored by an investigator-initiated grant (SAV-MD-08) from Forest Laboratories.
Role of sponsor: Forest Laboratories had no role in the design and conduct of the study or the collection, management, analysis, and interpretation of the data. Forest Laboratories reviewed and approved the manuscript for submission.
REFERENCES
1. Parvizi J, Jain N, Schmidt AH. Periprosthetic knee fractures. J Orthop Trauma. 2008;22(9):663–671. doi:10.1097/BOT.0b013e31816ed989 PubMed
2. Kurtz S, Ong K, Lau E, et al. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780–785. doi:10.2106/JBJS.F.00222 PubMed
3. Bourne RB, Chesworth B, Davis A, et al. Comparing patient outcomes after THA and TKA: is there a difference? Clin Orthop Relat Res. 2010;468(2):542–546. PubMed
4. Brander VA, Stulberg SD, Adams AD, et al. Predicting total knee replacement pain: a prospective, observational study. Clin Orthop Relat Res. 2003;416(416):27–36. doi:10.1097/01.blo.0000092983.12414.e9 PubMed
5. Anderson JG, Wixson RL, Tsai D, et al. Functional outcome and patient satisfaction in total knee patients over the age of 75. J Arthroplasty. 1996;11(7):831–840. PubMed
6. Moret C, Charveron M, Finberg JP, et al. Biochemical profile of midalcipran (F 2207), 1-phenyl-1-diethyl-aminocarbonyl-2-aminomethyl-cyclopropane (Z) hydrochloride, a potential fourth generation antidepressant drug. Neuropharmacology. 1985;24(12):1211–1219. doi:10.1016/0028-3908(85)90157-1 PubMed
7. Marks DM, Pae CU, Patkar AA. Triple reuptake inhibitors: the next generation of antidepressants. Curr Neuropharmacol. 2008;6(4):338–343. doi:10.2174/157015908787386078 PubMed
8. Clauw DJ, Mease P, Palmer RH, et al. Milnacipran for the treatment of fibromyalgia in adults: a 15-week, multicenter, randomized, double-blind, placebo-controlled, multiple-dose clinical trial. Clin Ther. 2008;30(11):1988–2004. doi:10.1016/j.clinthera.2008.11.009 PubMed
9. Mease PJ, Clauw DJ, Gendreau RM, et al. The efficacy and safety of milnacipran for treatment of fibromyalgia. a randomized, double-blind, placebo-controlled trial. J Rheumatol. 2009;36(2):398–409. doi:10.3899/jrheum.080734 PubMed
10. Ito M, Kimura H, Yoshida K, et al. Effectiveness of milnacipran for the treatment of chronic pain in the orofacial region. Clin Neuropharmacol. 2010;33(2):79–83. doi:10.1097/WNF.0b013e3181cb5793 PubMed
11. Kamata M, Takahashi H, Naito S, et al. Effectiveness of milnacipran for the treatment of chronic pain: a case series. Clin Neuropharmacol. 2004;27(5):208–210. doi:10.1097/01.wnf.0000145507.07261.a6 PubMed
12. Arnold LM, Clauw DJ, Wohlreich MM, et al. Efficacy of duloxetine in patients with fibromyalgia: pooled analysis of 4 placebo-controlled clinical trials. Prim Care Companion J Clin Psychiatry. 2009;11(5):237–244. doi:10.4088/PCC.08m00680 PubMed
13. van Kerkhoven LA, Laheij RJ, Aparicio N, et al. Effect of the antidepressant venlafaxine in functional dyspepsia: a randomized, double-blind, placebo-controlled trial. Clin Gastroenterol Hepatol. 2008;6(7):746–752. doi:10.1016/j.cgh.2008.02.051 PubMed
14. Lee H, Kim JH, Min BH, et al. Efficacy of venlafaxine for symptomatic relief in young adult patients with functional chest pain: a randomized, double-blind, placebo-controlled, crossover trial. Am J Gastroenterol. 2010;105(7):1504–1512. doi:10.1038/ajg.2010.82 PubMed
15. Rowbotham MC, Goli V, Kunz NR, et al. Venlafaxine extended release in the treatment of painful diabetic neuropathy: a double-blind, placebo-controlled study. Pain. 2004;110(3):697–706. doi:10.1016/j.pain.2004.05.010 PubMed
16. Pae CU, Park MH, Marks DM, et al. Desvenlafaxine, a serotonin-norepinephrine uptake inhibitor for major depressive disorder, neuropathic pain and the vasomotor symptoms associated with menopause. Curr Opin Investig Drugs. 2009;10(1):75–90. PubMed
17. Wernicke JF, Pritchett YL, D’Souza DN, et al. A randomized controlled trial of duloxetine in diabetic peripheral neuropathic pain. Neurology. 2006;67(8):1411–1420. doi:10.1212/01.wnl.0000240225.04000.1a PubMed
18. Ozyalcin SN, Talu GK, Kiziltan E, et al. The efficacy and safety of venlafaxine in the prophylaxis of migraine. Headache. 2005;45(2):144–152. doi:10.1111/j.1526-4610.2005.05029.x PubMed
19. Chappell AS, Desaiah D, Liu-Seifert H, et al. A double-blind, randomized, placebo-controlled study of the efficacy and safety of duloxetine for the treatment of chronic pain due to osteoarthritis of the knee. Pain Pract. 2011;11(1):33–41. doi:10.1111/j.1533-2500.2010.00401.x PubMed
20. Frakes EP, Risser RC, Ball TD, et al. Duloxetine added to oral nonsteroidal anti-inflammatory drugs for treatment of knee pain due to osteoarthritis: results of a randomized, double-blind, placebo-controlled trial. Curr Med Res Opin. 2011;27(12):2361–2372. doi:10.1185/03007995.2011.633502 PubMed
21. Marks DM, Shah MJ, Patkar AA, et al. Serotonin-norepinephrine reuptake inhibitors for pain control: premise and promise. Curr Neuropharmacol. 2009;7(4):331–336. doi:10.2174/157015909790031201 PubMed
22. Pae CU, Marks DM, Shah M, et al. Milnacipran: beyond a role of antidepressant. Clin Neuropharmacol. 2009;32(6):355–363. doi:10.1097/WNF.0b013e3181ac155b PubMed
23. Macher JP, Sichel JP, Serre C, et al. Double-blind placebo-controlled study of milnacipran in hospitalized patients with major depressive disorders. Neuropsychobiology. 1989;22(2):77–82. doi:10.1159/000118596 PubMed
24. Lecrubier Y, Pletan Y, Solles A, et al. Clinical efficacy of milnacipran: placebo-controlled trials. Int Clin Psychopharmacol. 1996;11(suppl 4):29–33. doi:10.1097/00004850-199609004-00004 PubMed
25. Ansseau M, von Frenckell R, Mertens C, et al. Controlled comparison of two doses of milnacipran (F 2207) and amitriptyline in major depressive inpatients. Psychopharmacology (Berl). 1989;98(2):163–168. doi:10.1007/BF00444686 PubMed
26. Van Amerongen AP, Ferrey G, Tournoux A. A randomized, double-blind comparison of milnacipran and imipramine in the treatment of depression. J Affect Disord. 2002;72(1):21–31. doi:10.1016/S0165-0327(01)00422-0 PubMed
27. Tignol J, Pujol-Domenech J, Chartres JP, et al. Double-blind study of the efficacy and safety of milnacipran and imipramine in elderly patients with major depressive episode. Acta Psychiatr Scand. 1998;97(2):157–165. doi:10.1111/j.1600-0447.1998.tb09980.x PubMed
28. Lopez-Ibor JJ, Conesa A; Spanish Milnacipran/Imipramine Study. A comparative study of milnacipran and imipramine in the treatment of major depressive disorder. Curr Med Res Opin. 2004;20(6):855–860. doi:10.1185/030079904125003719 PubMed
29. Leinonen E, Lepola U, Koponen H, et al. Long-term efficacy and safety of milnacipran compared to clomipramine in patients with major depression. Acta Psychiatr Scand. 1997;96(6):497–504. doi:10.1111/j.1600-0447.1997.tb09953.x PubMed
30. Steen A, Den Boer JA. A double-blind six months comparative study of milnacipran and clomipramine in major depressive disorder. Int Clin Psychopharmacol. 1997;12(5):269–281. doi:10.1097/00004850-199709000-00004 PubMed
31. Ansseau M, Papart P, Troisfontaines B, et al. Controlled comparison of milnacipran and fluoxetine in major depression. Psychopharmacology (Berl). 1994;114(1):131–137. doi:10.1007/BF02245454 PubMed
32. Guelfi JD, Ansseau M, Corruble E, et al. A double-blind comparison of the efficacy and safety of milnacipran and fluoxetine in depressed inpatients. Int Clin Psychopharmacol. 1998;13(3):121–128. doi:10.1097/00004850-199805000-00005 PubMed
33. Lee MS, Ham BJ, Kee BS, et al. Comparison of efficacy and safety of milnacipran and fluoxetine in Korean patients with major depression. Curr Med Res Opin. 2005;21(9):1369–1375. doi:10.1185/030079905X59166 PubMed
34. Ansseau M, von Frenckell R, Gérard MA, et al. Interest of a loading dose of milnacipran in endogenous depressive inpatients: comparison with the standard regimen and with fluvoxamine. Eur Neuropsychopharmacol. 1991;1(2):113–121. doi:10.1016/0924-977X(91)90712-4 PubMed
35. Clerc G; Milnacipran/Fluvoxamine Study Group. Antidepressant efficacy and tolerability of milnacipran, a dual serotonin and noradrenaline reuptake inhibitor: a comparison with fluvoxamine. Int Clin Psychopharmacol. 2001;16(3):145–151. doi:10.1097/00004850-200105000-00003 PubMed
36. Sechter D, Vandel P, Weiller E, et al; Study Co-coordinators. A comparative study of milnacipran and paroxetine in outpatients with major depression. J Affect Disord. 2004;83(2–3):233–236. doi:10.1016/j.jad.2004.07.002 PubMed
37. Yang JCK, Yu SWBH. Milnacipran versus sertraline in major depressive disorder: a double-blind randomized comparative study on the treatment effect and beta-adrenergic receptor responsiveness. Korean J Psychopharmacol. 2003;14:(4)387–396.
38. Blaya C, Seganfredo AC, Dornelles M, et al. The efficacy of milnacipran in panic disorder: an open trial. Int Clin Psychopharmacol. 2007;22(3):153–158. doi:10.1097/YIC.0b013e32802c7bf5 PubMed
39. Arnold LM, Palmer RH, Gendreau RM, et al. Relationships among pain, depressed mood, and global status in fibromyalgia patients: post hoc analyses of a randomized, placebo-controlled trial of milnacipran. Psychosomatics. 2012;53(4):371–379. doi:10.1016/j.psym.2012.02.005 PubMed
40. Marangell LB, Clauw DJ, Choy E, et al. Comparative pain and mood effects in patients with comorbid fibromyalgia and major depressive disorder: secondary analyses of four pooled randomized controlled trials of duloxetine. Pain. 2011;152(1):31–37. doi:10.1016/j.pain.2010.05.029 PubMed
41. Raskin J, Pritchett YL, Wang F, et al. A double-blind, randomized multicenter trial comparing duloxetine with placebo in the management of diabetic peripheral neuropathic pain. Pain Med. 2005;6(5):346–356. doi:10.1111/j.1526-4637.2005.00061.x PubMed
42. Sheehan DV, Lecrubier Y, Sheehan KH, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59(suppl 20):22–33. PubMed
43. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
44. Insall JN, Dorr LD, Scott RD, et al. Rationale of the Knee Society clinical rating system. Clin Orthop Relat Res. 1989;248(248):13–14. PubMed
45. Jaeschke R, Singer J, Guyatt GH. Measurement of health status: ascertaining the minimal clinically important difference. Control Clin Trials. 1989;10(4):407–415. doi:10.1016/0197-2456(89)90005-6 PubMed
46. Ware JE Jr, Sherbourne CD. The MOS 36-Item Short-Form Health Survey (SF-36), 1: conceptual framework and item selection. Med Care. 1992;30(6):473–483. doi:10.1097/00005650-199206000-00002 PubMed
47. Smets EM, Garssen B, Bonke B, et al. The Multidimensional Fatigue Inventory (MFI) psychometric qualities of an instrument to assess fatigue. J Psychosom Res. 1995;39(3):315–325. doi:10.1016/0022-3999(94)00125-O PubMed
48. Beck AT, Ward CH, Mendelson M, et al. An inventory for measuring depression. Arch Gen Psychiatry. 1961;4(6):561–571. doi:10.1001/archpsyc.1961.01710120031004 PubMed
49. Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134(4):382–389. doi:10.1192/bjp.134.4.382 PubMed
50. Spielberger CD, Gorsuch RL, Lushene PR, et al. Manual for the State-Trait Anxiety Inventory (Form Y). Palo Alto, California: Consulting Psychologists Press, Inc; 1983.
51. Singh J, Sloan JA, Johanson NA. Challenges with health-related quality of life assessment in arthroplasty patients: problems and solutions. J Am Acad Orthop Surg. 2010;18(2):72–82. PubMed
52. Medical Advisory Secretariat. Total knee replacement: an evidence-based analysis. Ont Health Technol Assess Ser. 2005;5(9):1–51. PubMed
53. Pae CU, Luyten P, Marks DM, et al. The relationship between fibromyalgia and major depressive disorder: a comprehensive review. Curr Med Res Opin. 2008;24(8):2359–2371. doi:10.1185/03007990802288338 PubMed
54. Dharmshaktu P, Tayal V, Kalra BS. Efficacy of antidepressants as analgesics: a review. J Clin Pharmacol. 2012;52(1):6–17. doi:10.1177/0091270010394852
Enjoy this premium PDF as part of your membership benefits!


