Patients With Borderline Personality Disorder and Major Depressive Disorder Are Not Distinguishable by Their Neuropsychological Performance: A Case-Control Study
Objective: Patients with borderline personality disorder (BPD) and patients with major depressive disorder (MDD) exhibit a broad range of neuropsychological deficits. Studies in both groups of patients point to differences but also similarities. However, studies that compare both patient groups are missing from the literature. The present study aimed to compare neuropsychological functioning in BPD and MDD patients.
Method: Eighteen patients with BPD, 27 patients with MDD, 17 patients with BPD and MDD, and 76 healthy control subjects were included in the case-control study. Patients were treated for their disorders as inpatients of the Clinic of Psychiatry and Psychotherapy Bethel, Ev. Hospital Bielefeld (Bielefeld, Germany). All patients met DSM-IV diagnoses as assessed by trained psychotherapists within the first week of their admission. In addition to a comprehensive neuropsychological test battery, the inhibitory control of emotional stimuli was assessed. Data were collected between June 2004 and June 2007.
Results: Patients showed only a few impairments and no increased distractibility toward emotionally negative stimuli. Patients with BPD and patients with MDD were not distinguishable by the neuropsychological test results.
Conclusions: These data did not support the notion of specific neuropsychological profiles in BPD and MDD. Future research needs to clarify the overlap of symptoms between both disorders.
Prim Care Companion CNS Disord 2011;13(1):e1-e9
© Copyright 2011 Physicians Postgraduate Press, Inc.
Submitted: March 16, 2010; accepted June 24, 2010.
Published online: January 27, 2011(doi:10.4088/PCC.10m00982blu).
Corresponding author: Thomas Beblo, PhD, Division of Research, Evaluation and Documentation, Clinic of Psychiatry and Psychotherapy Bethel, Ev. Hospital Bielefeld, Remterweg 69-71, 33617 Bielefeld, Germany ([email protected]).
Cognitive deficits of patients with borderline personality disorder (BPD) have been systematically investigated for 2 decades. Patients have been shown to exhibit deficits in the domains of executive functions, memory, attention, and visuospatial abilities.1 Previously, we administered a comprehensive neuropsychological test battery and found that BPD patients exhibited impairments primarily in visual functions.2 In agreement with these findings, it was concluded that nonverbal functions might be more affected than verbal functions,1,3 especially in the domain of memory.4 Some experimental studies in BPD revealed impairments of inhibitory functions,3,5 especially the inhibition of stimuli with negative valence.6 Domes et al7 found BPD patients to show reduced inhibition of negative material in a directed forgetting task and in a negative priming task. However, the performance in the emotional stroop task was not affected. BPD patients in a study by Hurlemann et al8 displayed enhanced retrograde and anterograde amnesia in response to negative stimuli. The authors concluded that there is a negative emotional response bias with a reduced ability to inhibit negatively valenced stimuli.
Patients with MDD exhibit deficits in the domains of executive functions, memory, and attention as well.9 In an early meta-analysis, Veiel10 concluded that patients with major depressive disorder (MDD) show a dominant deficit in cognitive flexibility/fluency and diffuse impairments in other cognitive domains. Data from our previous studies supported this conclusion.11,12 Clark et al13 found impairments in cognitive flexibility but no further neuropsychological deficits in relatives of depressed patients. With regard to fluency, semantic fluency seems to be more impaired than phonological fluency.14 However, the profile and severity of impairment seem to depend on many factors such as comorbidity, subtype of the disorder, age, medication, and the experience of failure.15 Recent studies also underline the influence of rumination,16 motivation,17 and sleep.18 Further, experimental investigations indicate that cognitive changes in depression are more obvious with certain features of the neuropsychological tasks such as the consideration of affectively meaningful stimuli.19-21 As with BPD patients, inhibitory dysfunction in MDD patients is most likely valence specific. In the study of Lau et al,21 patients with MDD showed deficits in cognitive inhibition. These deficits were most pronounced for negatively valenced stimuli. An inhibition problem with negative distraction corresponds with an attentional bias toward negatively valenced information in MDD patients. This bias may lead to an enhanced memory for negatively valenced emotional material.22
Clinical Points
- On average, patients with major depressive disorder and patients with borderline personality disorder show only a few neuropsychological impairments as assessed by standardized neuropsychological tests. However, clinically significant deficits may occur in single cases, and, in particular, in more severely affected patients who were not included in the present study. In addition, clinically significant deficits in everyday life cannot be ruled out.
- Clinicians cannot differentiate patients with major depressive disorder from patients with borderline personality disorder by their neuropsychological performance.
These findings point to neuropsychological differences as well as similarities between BPD and MDD patients. Unfortunately, to our knowledge, there are no studies that compare neuropsychological profiles of both patient groups. Völker et al23 investigated executive functions in BPD patients as well as in subjects with a lifetime diagnosis of depression and healthy subjects. No differences between the 3 groups were found. However, conclusions are limited because other neuropsychological functions were not considered and acute MDD patients were not investigated. Fertuck et al24 administered a comprehensive neuropsychological battery in MDD patients with and without BPD as well as in healthy control subjects. Neuropsychological performance did not differ between the patient groups, thus indicating no additional negative effect of BPD in patients with MDD with regard to cognitive functioning. However, since a group with BPD only was missing, no clear conclusions about BPD can be drawn.24 Thus, the question of a possible neuropsychological overlap in BPD and MDD is still a matter of debate. This question implies important implications. Clinically, knowledge about neuropsychological profiles in BPD and MDD is helpful for diagnostic purposes. From a theoretical point of view, neuropsychological similarities between both disorders would bring up questions about common etiologic pathways.
The present study aimed at a comparison of neuropsychological functioning in BPD and MDD. We hypothesized (1) that patients with BPD would show specific deficits in visual functions, primarily visual memory; (2) that MDD patients predominantly would exhibit deficits in cognitive flexibility and semantic fluency; and (3) that BPD and MDD patients would show an increased distractibility toward emotionally negative stimuli (that is, reduced learning performance with the presentation of emotionally negative stimuli).
METHOD
Subjects
The case-control study included 27 patients with MDD but without any personality disorder, 18 patients with BPD but without acute MDD or any history of MDD, 17 patients with acute MDD and BPD, and 76 healthy control subjects. Patients were treated for their disorders as inpatients of the Clinic of Psychiatry and Psychotherapy Bethel, Ev. Hospital Bielefeld (Bielefeld, Germany). All patients met DSM-IV diagnoses as assessed by trained psychotherapists within the first week of their admission. Healthy subjects were recruited by advertisements in a local newspaper. Exclusion criteria for participation in the study were further comorbid Axis I disorders apart from anxiety disorders, somatization disorder, substance abuse of more than 6 months ago, and bulimia. In addition, patients with MDD with psychotic symptoms were not included. Healthy control subjects were free of any Axis I or II disorders. The subjects underwent careful clinical examination and were assessed to exclude the following medical conditions: pregnancy, endocrine system disorders, malignant diseases, liver cirrhosis, a history of neurologic disorders with central nervous system involvement, and mental retardation. After a complete explanation of the study, written informed consent was obtained from all subjects. The study was accepted by the institutional review board (University of Muenster Ethics Committee, Muenster, Germany). Data were collected between June 2004 and June 2007.
Instruments
Clinical examination. Psychiatric diagnoses were made using the Structured Clinical Interview for DSM-IV (SCID): SCID-I for Axis I disorders and SCID-II for personality disorders.25 These interviews were applied by trained psychotherapists. The clinical examination also included the assessment of depressive mood using the Beck Depression Inventory (BDI).26
Neuropsychological Assessment
Memory. Neuropsychological assessment was performed by a neuropsychologist (C.M.) and a master’s student. They were trained and supervised by a trained neuropsychologist (T.B.).
Visual learning with and without distraction. Subjects learned 3 lists (A, B, C) of 15 simple designs. The A and B sets of learning stimuli were drawn from the Rey Visual Design Learning Test.27 List C was developed by the authors themselves using variations of the stimuli from lists A and B. Pretests of the sets used showed comparable results for all item lists. In contrast to the standard procedure, each list was presented only 3 times.
All stimuli were presented on a video screen of a standard personal computer using the software Presentation 0.76.28 After each learning trial, the subjects were asked to draw the figures they remembered on a sheet of paper. The dependent variable was the sum of correctly drawn figures in trials 1-3. Three experimental conditions were presented to all subjects. (1) In order to assess baseline learning performance, subjects learned a design list without distraction. (2) In the first distraction condition, designs of a list were presented alternating with pictures from the International Affective Picture Series (IAPS) of neutral valence.29 For the 3 learning trials, 45 different IAPS pictures were used. (3) The second distraction condition corresponded to the first distraction condition, but IAPS pictures with negatively emotional valence were used. The ratings of emotional valence from the neutral and negative pictures differed as indicated by results of an analysis of variance (ANOVA) (F1,88 = 723.83, P < .0001). The assignment of the learning conditions (1-3) with the design lists (A-C) was randomized, and the lists were displayed in the same order (A-B-C).
In the distraction conditions, presentation started with a 350-ms interval presenting a black screen, followed by an interval of 1,000 ms in which a distractor was presented. Then, for a 350-ms interval, a black screen was shown, followed by a 1,000-ms presentation of a learning stimulus (design). The baseline condition was comparable, but instead of the distractor, a white screen was presented (1,000 ms). Thus, in all conditions, the interval between 2 learning stimuli was 2,700 ms, maintaining the duration of 1 learning trial by 40.5 seconds in total.
The Complex Figure Test (CFT)30 was applied for the additional assessment of visual memory. Subjects had to recall and draw a complex figure that they had previously been shown and had copied 30 minutes before.
Logical memory. In the subtest logical memory of the Wechsler Memory Scale-Revised,31,32 subjects had to recall 2 short stories as accurately as possible. Recall performance was assessed immediately after each story was heard (immediate recall) and after 20 minutes (delayed recall).
Working memory with and without interfering stimuli. Immediate visual memory spans were assessed by the Corsi Block Tapping Test.33 The examiner tapped a series of blocks and then asked the subjects to tap the blocks in the same order. These blocks were irregularly arranged on a board. Additionally, a modified version of the Corsi Block Tapping Test, the Block Suppression Test,34 was administered. Subjects were asked to reproduce only every second block beginning with the first block from a series of blocks tapped by the examiner.
Immediate verbal memory spans were assessed by the digit span forward subtest from the Wechsler Memory Scale-Revised. Subjects had to repeat a series of digits in a given order. The number of correctly recalled digit spans was assessed. In addition, the Digit Suppression Test 34 was administered. In the Digit Suppression Test, only every second digit of a series of orally presented digits had to be reproduced, beginning with the first digit. The number of correctly recalled digit spans was assessed.
Attention. Reaction time was assessed by means of the subtest “alertness” of the computerized Test-Battery of Attentional Performance (Testbatterie zur Aufmerksamkeitspruefung).35 Subjects had to press a button as fast as possible after a cross appeared on the screen. The subtest go/no-go assessed response selection and response inhibition. Two different crosses—1 target and 1 distractor—were presented in random order. The subjects had to respond to the target as quickly as possible. For the assessment of divided attention, in the divided attention subtest, subjects had to respond to visually and auditorily presented targets. Visuomotor tracking was assessed by means of the Trail Making Test, part A.36 In the Trail Making Test, part A, subjects had to connect 25 numbers as quickly as possible. The Frankfurt Attention Inventory (Frankfurter Aufmerksamkeits-Inventar37) was used to assess speed of visual scanning and selective attention. In this cancellation task, 2 critical stimuli consisting of a shape (square or circle) and dots (2 or 3) had to be crossed out among different distractors.
Executive functions. Lexical verbal fluency was assessed by requiring the subject to name as many words as possible with the initial letters F, A, and S. One minute is given for each letter. In the semantic verbal fluency task, subjects had to name as many animals as possible within 1 minute. Cognitive flexibility was assessed by means of the Trail Making Test, part B.36 This subtest required subjects to connect a series of numbers and letters in an alternating manner (1 to A, A to 2, 2 to B, etc). The test Logical Thinking of the Leistungspruefsystem38 was also administered. This test consists of 40 items with each item consisting of a series of digits and letters arranged with 1 exception according to a basic rule. The test required the subject to identify the wrong element and included a time limit of 8 minutes.
Visuospatial abilities. Construction was assessed by means of the Complex Figure Test.30 A complex figure had to be copied as accurately as possible.
Data Analysis
All statistical analyses were performed using the Statistical Package for the Social Sciences 14.0 (SPSS 14.0, IBM Corporation, Somers, New York). The level of significance was set at P < .05 and 2-tailed for all analyses. The effect of distraction on visual learning was investigated by a repeated measure 3 (learning condition: no distraction, neutral distraction, negative distraction) ×— 4 (group: healthy subjects, MDD, BPD, MDD/BPD) analysis of covariance (ANCOVA) with age as covariate. Post hoc tests were calculated if indicated.
For the analysis of the neuropsychological profile, all other group differences were compared for explorative purposes with ANCOVAs and age as covariate. We used Pearson correlations to investigate associations between depression, as assessed by the BDI, and neuropsychological performance. Given the large number of tests and a relatively small sample size, a strict α correction (eg, Bonferroni) would have resulted in a huge β error. We therefore decided to regard these outcomes strictly as exploratory. Post hoc tests were calculated if indicated. Demographic and clinical data were analyzed by ANOVAs or χ2 tests.
RESULTS
Sample Characteristics and Clinical Data
The mean age of the MDD patients was 38.0 years (SD = 13.9). All other patient groups were younger (F3,134 = 4.4, P = .005), and age did not differ between BPD patients (mean = 28.4, SD = 11.6), BPD/MDD patients (mean = 28.9, SD = 8.6), and healthy subjects (mean = 29.2, SD = 11.2). Most subjects had completed secondary school (Sekundarstufe II, 12 or 13 years of German basic school education). Groups did not differ with regard to their school education (healthy subjects: n = 6 [9 years, Hauptschule], n = 21 [10 years, Realschule], n = 49 [12/13 years, Abitur]; BPD: n = 1, n = 5, and n = 12, respectively; MDD: n = 7, n = 5, and n = 14; and BPD/MDD: n = 4, n = 7, and n = 6). Distribution by sex was comparable in all 4 groups, with 61% women (n = 11) in the BPD group, 59% women (n = 10) in the BPD/MDD group, 59% women (n = 16) in the MDD group, and 64% women (n = 49) in the healthy subject group.
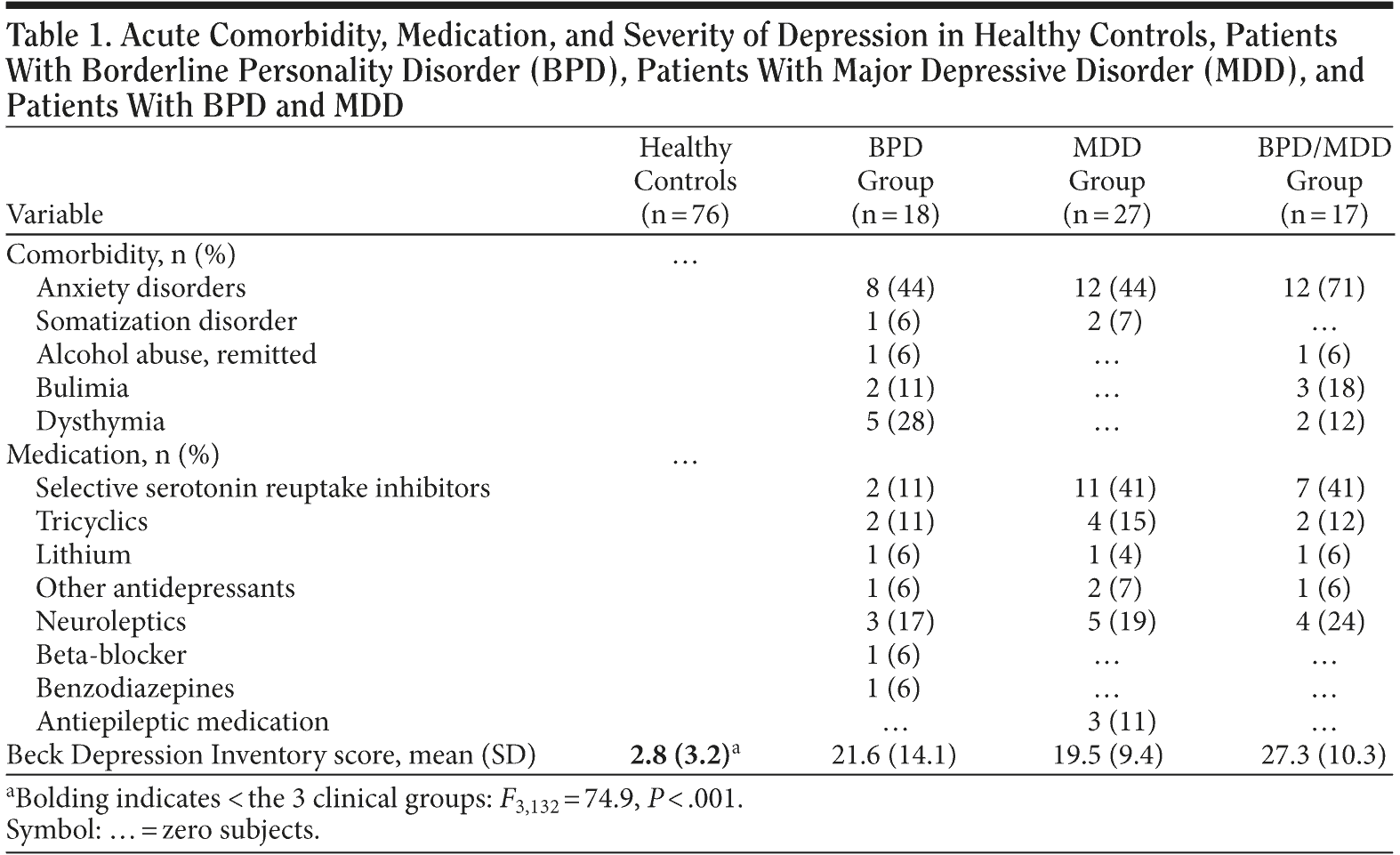
Severe comorbidity was excluded, but as expected, patients in all groups suffered from some comorbid Axis I disorders, primarily in the form of anxiety disorders (Table 1). Medication is also shown in Table 1.
With regard to the applied questionnaires, groups differed on the BDI (F3,132 = 74.9, P < .001) due to the low scores of the healthy controls (Table 1). Significant differences were not revealed between the 3 clinical groups for the BDI. For the 3 clinical groups, the results of the BDI indicate a relevant burden with symptoms of depression.
Visual Learning With and Without Distraction
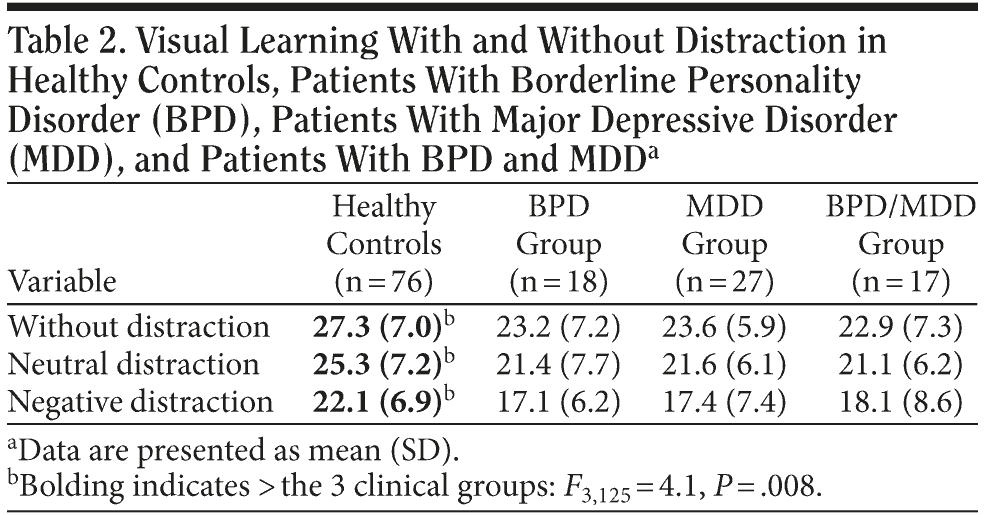
ANCOVA revealed that the 4 groups differed in their visual learning performance (F3,125 = 4.1, P = .008) due to a superior performance by the healthy subjects. However, post hoc analysis revealed no group differences between the 3 clinical groups. Compared to healthy subjects, BPD patients (F1,88 = 7.0, P < .010) and BPD/MDD patients (F1,87 = 5.9, P < .018) performed worse. MDD patients showed no deficits compared to healthy subjects. However, since the 3 patient groups did not differ, no specificity of deficits can be claimed for the BPD group; therefore, hypothesis 1 was not confirmed (Patients with BPD show specific deficits in visual functions.).
Subjects performed better when no distraction or a neutral distraction was presented; subjects performed worse when presented with negative distraction (main effect “condition”: F2,124 = 7.4, P < .001; without distraction vs neutral distraction: not significant; and neutral distraction vs negative distraction: F1,125 = 12.3, P < .001). However, hypothesis 3 (patients are disproportionally more distracted by emotionally relevant distractors) was not confirmed (no group ×— condition interaction). All means and standard deviations are given in Table 2.
Further Neuropsychological Test Results
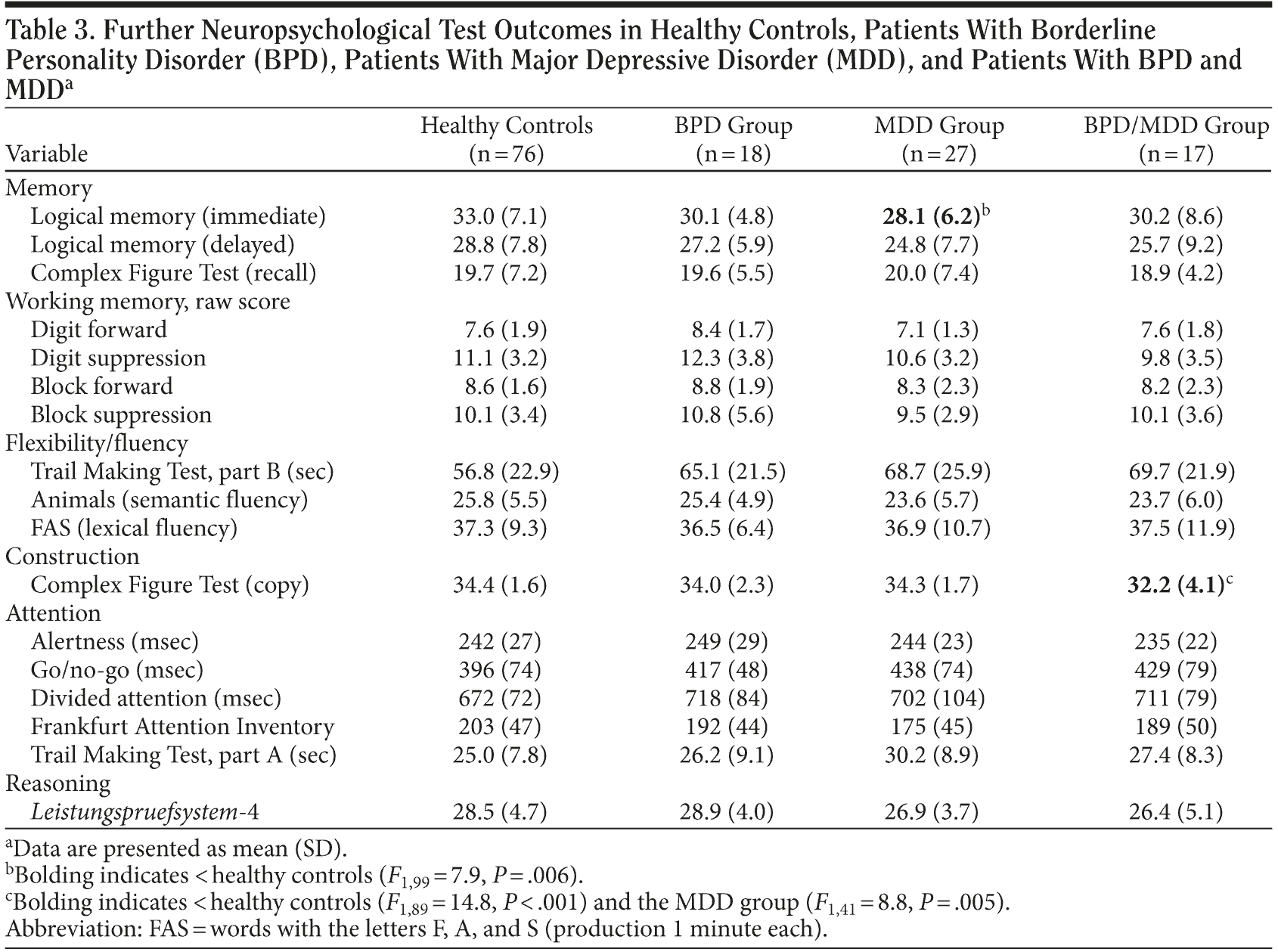
Comparison of the groups regarding their performance on other neuropsychological tests did not show group differences except in construction (copy of Rey`s Complex Figure: F3,132 = 6.0, P = .001) and verbal learning (logical memory, immediate recall: F3,131 = 3.0, P = .031). For construction, post hoc analysis showed an inferior performance of the BPD/MDD group compared to healthy subjects (F1,89 = 14.8, P < .001) and compared to the MDD patients (F1,41 = 8.8, P = .005) but not compared to the BPD patients (Table 3). BPD patients did not differ from the other groups; therefore, hypothesis 1 (BPD patients show specific deficits in visual functions) was not supported by the data. For verbal learning, post hoc analysis revealed that MDD patients performed worse than healthy subjects (F1,99 = 7.9, P = .006) but did not perform worse than the other patient groups; that is, poor performance on verbal learning was not specific to the MDD patient group. The BPD and BPD/MDD patient groups’ verbal learning performance was similar to the healthy subjects’ performance.
Associations Between Clinical Symptoms and Neuropsychological Test Results
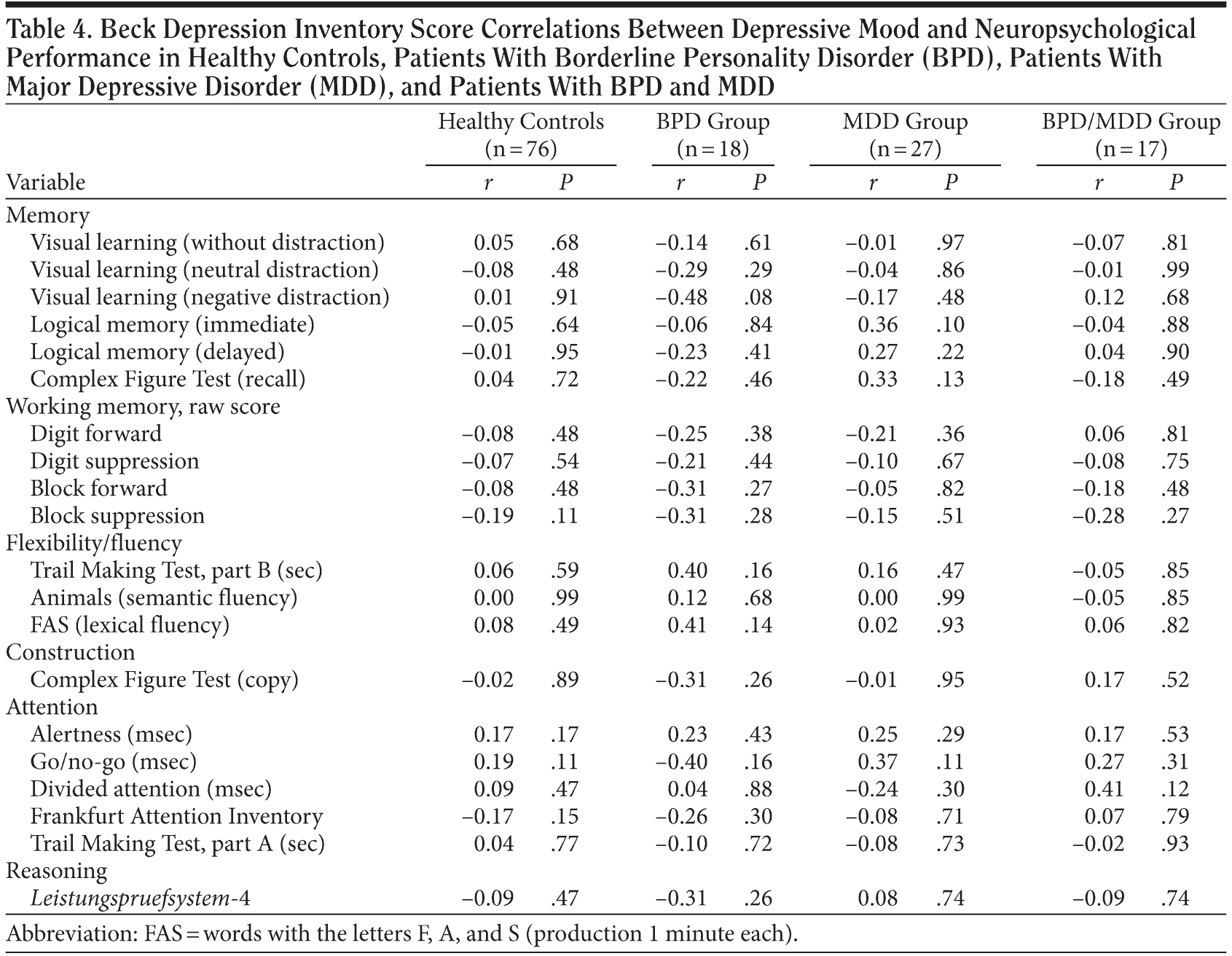
We did not find any associations between the severity of depression (as assessed by the BDI) and neuropsychological performance (Table 4).
Control of Comorbidity in the BPD and MDD Samples
In order to examine whether the reduced performances of the BPD and MDD group were due to comorbid disorders, comorbidity-defined subgroups (BPD patients with Axis I comorbidity vs BPD patients without, MDD patients with comorbidity vs MDD patients without) were compared with regard to the parameters with inferior performance of the patients (BPD: visual learning, MDD: verbal learning) via t tests. Subgroups did not differ in these exploratory analyses; that is, comorbid disorders were not a plausible explanation for reduced performances.
DISCUSSION
To our knowledge, this is the first study comparing the neuropsychological profiles of subjects with BPD, MDD, and BPD/MDD and healthy subjects. The main finding of the study is that the patient groups did not show a specific profile of neuropsychological deficits. In general, patient groups showed only a few impairments. Even the distractibility toward emotionally negative stimuli did not differ between the clinical groups and healthy subjects.
With regard to BPD, we found that visual memory was in fact impaired. This finding is in line with many other studies (eg, Swirsky-Sacchetti et al39) and with the conclusion of LeGris and van Reekum4 that visual memory is among the most reported deficits in BPD. A reduced volume of the hippocampus is a well-confirmed finding in BPD and may be associated with memory deficits.40,41 Interestingly, the study of Irle et al41 documented a visual memory deficit that was correlated to a volume reduction of the right hippocampus. Apart from visual memory, we did not find further visual deficits in BPD patients; construction, visual working memory, and other nonverbal neuropsychological functions were not impaired. These findings contrast with some other studies (eg, O’ Leary et al42). It might be speculated that the well-selected study patients without severe comorbidity do not present such deficits. In accordance with this interpretation, patients with BPD and comorbid MDD did show deficits in construction. In general, study samples without substantial comorbidity and medication (eg, Kunert et al43) rarely present neuropsychological deficits, whereas other studies (eg, Monarch et al44) that also included patients with substantial comorbidity and medication showed a broader range of impairments. In addition, the small sample size of our study might have prevented detection of subtle deficits. However, Table 3 indicates almost identical performances between healthy subjects and BPD subjects regarding construction and further visual functions.
The hypothesis that BPD patients would show an increased distractibility toward emotionally negative stimuli (hypothesis 3) was not supported by the data. In a previous study,45 we investigated the interference caused by emotional stimuli by using the emotional stroop paradigm. Only BPD patients with comorbid PTSD showed an increased interference to emotionally negative words. The majority of BPD patients in the present study did not suffer from PTSD. Korfine and Hooley6 and Domes et al7 demonstrated that BPD patients showed a decreased forgetting rate for negative words using the directed forgetting paradigm. These results might be interpreted as an impaired inhibition of emotionally negative stimuli. However, the paradigm of the present study requires the inhibition of distractors during task performance. In the directed forgetting paradigm, participants are initially asked to learn stimuli, and the instruction to forget them is placed later. Therefore, inhibition processes are relevant at a later stage of the process.
The MDD patients in the present study seem to show a reduced performance in cognitive flexibility and semantic fluency (Table 3). However, the results did not reach statistical significance, perhaps due to sample size and the associated lack of statistical power. However, as mentioned above, the profile of neuropsychological deficits in depression was proven to depend upon additional variables such as the patient’s type of affective disorder.15 Airaksinen et al46 found that problems in mental flexibility primarily characterize patients with dysthymia. MDD patients and patients with mixed anxiety-depressive disorders instead show impairments in verbal memory. In agreement with these findings, MDD patients in the present study showed a reduced performance in verbal memory. Some authors also discuss the patient’s age as an important confound for the presence of executive functions such as fluency. Porter et al stated that “‘ ¦ executive tasks were disproportionately impaired in the older depressed group.”47(p119) By contrast, Castaneda et al concluded that “Executive dysfunction seems to be a key factor of young adulthood MDD ‘ ¦ ,” whereas “Results about verbal memory and learning functions are inconsistent.”9(p17) However, as with executive dysfunction, memory impairment is consistent with neurobiological findings in depression, such as alterations of the prefrontal cortex or hippocampus.48
As with BPD patients, in MDD patients, we found no increased distractibility toward emotionally negative stimuli. This was surprising, as there is some evidence for an inhibitory dysfunction in depression for emotionally negative stimuli. In the study by Lau et al,21 MDD patients needed more time to read stories that were presented with emotionally negative distractor words (prose distraction task). Goeleven et al20 found that depressed patients showed a specific deficit when it came to inhibiting negative information during a priming task with pictures of sad and happy facial expressions. Investigations with the emotional stroop task revealed conflicting results, with some studies showing an increased reaction time specific to emotionally negative stimuli.49,50 Other studies did not show increased reaction times.51,52 We speculate that the extent of similarities between targets and distractors may account for differences between study results. In the present study, targets and distractors were very easy to distinguish, whereas different stimuli were very similar in the other studies. The combined distraction effect of similarity and emotion might be necessary to create a greater interference in MDD patients.
The results of the present study do not support the view of distinguishable neuropsychological deficits in BPD and MDD patients. In addition and in accordance with Fertuck et al,24 the results also fail to show that possible negative effects of BPD and MDD add to an inferior performance in the BPD/MDD group. BPD patients showed visual memory deficits, but their performance was not impaired when compared with MDD patients. Similarly, MDD patients showed verbal memory impairment, but the performance was not significantly worse than the performance of BPD patients. The BPD/MDD patients showed an inferior performance in construction when compared to healthy subjects and MDD patients but not when compared to BPD patients. In the majority of the tests applied, the 3 patient groups were not impaired. These results are in agreement with the outcomes of 2 studies23,24 that also failed to show neuropsychological differences between the patient groups. However, Keilp et al53 found that early visual information processing is impaired in MDD patients with BPD but not in MDD patients without comorbid BPD. Taken together, the results of the present study indicate that patients with BPD and MDD show a large overlap of neuropsychological performance, even though the study samples were well defined (BPD patients without MDD and MDD patients without BPD). Future research needs to clarify whether the overlap of symptoms point to common etiologic pathways in both disorders.
The main shortcoming of the present study is the small subsample sizes, especially in the BPD and BPD/MDD groups. Related to this shortcoming, we were not able to perform multivariate statistics or a strict α correction. Therefore, the reported impairments in visual and verbal memory have to be interpreted with caution. Furthermore, the small subsample sizes of each group may have prevented us from detecting small effect sizes. Therefore, we can only conclude that we did not find evidence for large differences between the patient groups. The BPD patients without present and past MDD are hard to find. Therefore, we decided to focus on a rather small but very well-selected sample. Furthermore, most of the patients took psychotropic medication with possible cognitive side effects. Since we did not find any impairment in attentional performance, it is unlikely that our results can be explained by cognitive side effects due to medication. By contrast, it cannot to be ruled out that some antidepressant drugs such as selective serotonin reuptake inhibitors might have a slightly stimulating effect on the neuropsychological performance.54 Although we controlled for some comorbidity, many patients showed comorbid disorders, primarily anxiety disorders. To control for this factor, we compared BPD patients with and without comorbid Axis I disorders as well as MDD patients with and without comorbid disorders. We found no differences in the performance of these patient subgroups. Therefore, it is unlikely that comorbid diagnoses explain the results of the present study.
In sum, the outcomes of the present study suggest only very few neuropsychological impairments in patients with BPD and MDD. Deficit profiles are not distinguishable between the clinical study samples. Our neuropsychological findings challenge the notion of disorder-specific neuropsychological findings in MDD and BPD patients. However, findings need to be replicated in larger samples in order to search for moderate group differences. The overlap of symptoms between BPD and MDD patients in the present study may point to common etiologic pathways in BPD and MDD. Since studies are missing from the literature that investigate patients with BPD and patients with MDD, the relation between the disorders remains unclear and requires further investigation.
Drug name: lithium (Lithobid and others).
Author affiliations: Division of Research, Evaluation and Documentation, Department of Psychiatry and Psychotherapy Bethel, Ev. Hospital Bielefeld, Bielefeld (Drs Beblo, Rullkoetter, and Driessen and Ms Schlosser); Department of Psychology, University of Bielefeld, Bielefeld (Drs Beblo and Driessen); Department of Psychosomatic Medicine and Psychotherapy, University Hospital Duisburg-Essen, Essen (Dr Mensebach); and Department of Psychosomatic Medicine and Psychotherapy, University Medical Center Hamburg-Eppendorf, Hamburg (Dr Wingenfeld), Germany.
Potential conflicts of interest: None reported.
Funding/support: This study was supported by the Deutsche Forschungsgemeinschaft, Bonn, Germany (DFG grant BE 2536/4-1).
REFERENCES
1. Ruocco AC. The neuropsychology of borderline personality disorder: a meta-analysis and review. Psychiatry Res. 2005;137(3):191-202. PubMed doi:10.1016/j.psychres.2005.07.004
2. Beblo T, Saavedra AS, Mensebach C, et al. Deficits in visual functions and neuropsychological inconsistency in borderline personality disorder. Psychiatry Res. 2006;145(2-3):127-135. PubMed doi:10.1016/j.psychres.2006.01.017
3. Fertuck EA, Lenzenweger MF, Clarkin JF, et al. Executive neurocognition, memory systems, and borderline personality disorder. Clin Psychol Rev. 2006;26(3):346-375. PubMed doi:10.1016/j.cpr.2005.05.008
4. LeGris J, van Reekum R. The neuropsychological correlates of borderline personality disorder and suicidal behaviour. Can J Psychiatry. 2006;51(3):131-142. PubMed
5. Rentrop M, Backenstrass M, Jaentsch B, et al. Response inhibition in borderline personality disorder: performance in a go/nogo task. Psychopathology. 2008;41(1):50-57. PubMed doi:10.1159/000110626
6. Korfine L, Hooley JM. Directed forgetting of emotional stimuli in borderline personality disorder. J Abnorm Psychol. 2000;109(2):214-221. PubMed doi:10.1037/0021-843X.109.2.214
7. Domes G, Winter B, Schnell K, et al. The influence of emotions on inhibitory functioning in borderline personality disorder. Psychol Med. 2006;36(8):1163-1172. PubMed doi:10.1017/S0033291706007756
8. Hurlemann R, Hawellek B, Maier W, et al. Enhanced emotion-induced amnesia in borderline personality disorder. Psychol Med. 2007;37(7):971-981. PubMed doi:10.1017/S0033291706009792
9. Castaneda AE, Tuulio-Henriksson A, Marttunen M, et al. A review on cognitive impairments in depressive and anxiety disorders with a focus on young adults. J Affect Disord. 2008;106(1-2):1-27. PubMed doi:10.1016/j.jad.2007.06.006
10. Veiel HOF. A preliminary profile of neuropsychological deficits associated with major depression. J Clin Exp Neuropsychol. 1997;19(4):587-603. PubMed doi:10.1080/01688639708403745
11. Beblo T, Baumann B, Bogerts B, et al. Neuropsychological correlates of major depression: a short term follow-up. Cogn Neuropsychiatry. 1999;4(4):333-341. doi:10.1080/135468099395864
12. Lahr D, Beblo T, Hartje W. Cognitive performance and subjective complaints before and after remission of major depression. Cogn Neuropsychiatry. 2007;12(1):25-45. PubMed doi:10.1080/13546800600714791
13. Clark L, Sarna A, Goodwin GM. Impairment of executive function but not memory in first-degree relatives of patients with bipolar I disorder and in euthymic patients with unipolar depression. Am J Psychiatry. 2005;162(10):1980-1982. PubMed doi:10.1176/appi.ajp.162.10.1980
14. Henry J, Crawford JR. A meta-analytic review of verbal fluency deficits in depression. J Clin Exp Neuropsychol. 2005;27(1):78-101. PubMed doi:10.1080/138033990513654
15. Beblo T, Lautenbacher S. Neuropsychologie der Depression. Göttingen, Germany: Hogrefe; 2006.
16. Donaldson C, Lam D. Rumination, mood and social problem-solving in major depression. Psychol Med. 2004;34(7):1309-1318. PubMed doi:10.1017/S0033291704001904
17. Scheurich A, Fellgiebel A, Schermuly I, et al. Experimental evidence for a motivational origin of cognitive impairment in major depression. Psychol Med. 2008;38(2):237-246. PubMed doi:10.1017/S0033291707002206
18. Goder R, Fritzer G, Hinze-Selch D, et al. Sleep in major depression: relation to memory performance and outcome after interpersonal psychotherapy. Neuropsychobiology. 2007;55(1):36-42. PubMed doi:10.1159/000103574
19. Erickson K, Drevets WC, Clark L, et al. Mood-congruent bias in affective go/no-go performance of unmedicated patients with major depressive disorder. Am J Psychiatry. 2005;162(11):2171-2173. PubMed doi:10.1176/appi.ajp.162.11.2171
20. Goeleven E, De Raedt R, Baert S, et al. Deficient inhibition of emotional information in depression. J Affect Disord. 2006;93(1-3):149-157. PubMed doi:10.1016/j.jad.2006.03.007
21. Lau MA, Christensen BK, Hawley LL, et al. Inhibitory deficits for negative information in persons with major depressive disorder. Psychol Med. 2007;37(9):1249-1259. PubMed doi:10.1017/S0033291707000530
22. Leppänen JM. Emotional information processing in mood disorders: a review of behavioral and neuroimaging findings. Curr Opin Psychiatry. 2006;19(1):34-39. PubMed doi:10.1097/01.yco.0000191500.46411.00
23. Völker KA, Spitzer C, Limberg A, et al. Executive dysfunctions in female patients with borderline personality disorder with regard to impulsiveness and depression. Psychother Psychosom Med Psychol. 2009;59(7):264-272. PubMed
24. Fertuck EA, Marsano-Jozefowicz S, Stanley B, et al. The impact of borderline personality disorder and anxiety on neuropsychological performance in major depression. J Pers Disord. 2006;20(1):55-70. PubMed doi:10.1521/pedi.2006.20.1.55
25. Wittchen H-U, Zaudig M, Fydrich T. Strukturiertes Klinisches Interview f×¼r DSM-IV (SKID). Göttingen, Germany: Hogrefe; 1997.
26. Beck AT, Steer RA. Beck-Depressions-Inventar (BDI): Testhandbuch. Bern, Switzerland: Huber; 1994.
27. Rey A. Épreuves Mnéstique et d’ Apprentissage. Neuchatel, Switzerland: Delachaux et Niestlé; 1968.
28. Presentation. 7.76 ed. Albany, CA: Neurobehavioral Systems, Inc; 2003.
29. Lang PJ, Bradley MM, Cutbert BN. The International Affective Picture System (IAPS). Bethesda, MD: NIMH Center for the Study of Emotion and Attention; 1987.
30. Rey A. L`examen de psychologique dans les cas d`encéphalopathie traumatique. Arch Psychol. 1941;28:286-340.
31. Haerting C, Markowitsch HJ, Neufeld H, et al. Wechsler Gedächtnistest: Revidierte Fassung. Bern, Switzerland: Huber; 2000.
32. Wechsler D. Wechsler Memory Scale-Revised Manual. San Antonio, TX: The Psychological Corporation; 1987.
33. Corsi PM. Human memory and the medial temporal region of the brain. Dissertation Abstracts International. 1972;34:819.
34. Beblo T, Macek C, Brinkers I, et al. A new approach in clinical neuropsychology to the assessment of spatial working memory: the block suppression test. J Clin Exp Neuropsychol. 2004;26(1):105-114. PubMed doi:10.1076/jcen.26.1.105.23938
35. Zimmermann P, Fimm B. Testbatterie zur Aufmerksamkeitspr×¼fung (TAP). Freiburg, Germany: Psytest; 1992.
36. Reitan RM. Trail Making Test. Tucson, AZ: Reitan Neuropsychology Laboratory; 1992.
37. Moosbrugger H, Oehlschlaegel J. Frankfurter Aufmerksamkeitsinventar (FAIR). Bern, Switzerland: Huber; 1996.
38. Horn W. Leistungspr×¼fsystem LPS. Göttingen, Germany: Hogrefe; 1983.
39. Swirsky-Sacchetti T, Gorton G, Samuel S, et al. Neuropsychological function in borderline personality disorder. J Clin Psychol. 1993;49(3):385-396. PubMed doi:10.1002/1097-4679(199305)49:3<385::AID-JCLP2270490313>3.0.CO;2-4
40. Driessen M, Herrmann J, Stahl K, et al. Magnetic resonance imaging volumes of the hippocampus and the amygdala in women with borderline personality disorder and early traumatization. Arch Gen Psychiatry. 2000;57(12):1115-1122. PubMed doi:10.1001/archpsyc.57.12.1115
41. Irle E, Lange C, Sachsse U. Reduced size and abnormal asymmetry of parietal cortex in women with borderline personality disorder. Biol Psychiatry. 2005;57(2):173-182. PubMed doi:10.1016/j.biopsych.2004.10.004
42. O’ Leary KM, Brouwers P, Gardner DL, et al. Neuropsychological testing of patients with borderline personality disorder. Am J Psychiatry. 1991;148(1):106-111. PubMed
43. Kunert HJ, Druecke HW, Sass H, et al. Frontal lobe dysfunctions in borderline personality disorder? neuropsychological findings. J Pers Disord. 2003;17(6):497-509. PubMed doi:10.1521/pedi.17.6.497.25354
44. Monarch ES, Saykin AJ, Flashman LA. Neuropsychological impairment in borderline personality disorder. Psychiatr Clin North Am. 2004;27(1):67-82, viii-ix [viii-ix.]. PubMed doi:10.1016/S0193-953X(03)00109-6
45. Wingenfeld K, Mensebach C, Rullkoetter N, et al. Attentional bias to personally relevant words in borderline personality disorder is strongly related to comorbid posttraumatic stress disorder. J Pers Disord. 2009;23(2):141-155. PubMed doi:10.1521/pedi.2009.23.2.141
46. Airaksinen E, Larsson M, Lundberg I, et al. Cognitive functions in depressive disorders: evidence from a population-based study. Psychol Med. 2004;34(1):83-91. PubMed doi:10.1017/S0033291703008559
47. Porter RJ, Bourke C, Gallagher P. Neuropsychological impairment in major depression: its nature, origin and clinical significance. Aust N Z J Psychiatry. 2007;41(2):115-128. PubMed doi:10.1080/00048670601109881
48. Drevets WC, Price JL, Furey ML. Brain structural and functional abnormalities in mood disorders: implications for neurocircuitry models of depression. Brain Struct Funct. 2008;213(1-2):93-118. PubMed doi:10.1007/s00429-008-0189-x
49. Dudley R, O’ Brien J, Barnett N, et al. Distinguishing depression from dementia in later life: a pilot study employing the emotional stroop task. Int J Geriatr Psychiatry. 2002;17(1):48-53. PubMed doi:10.1002/gps.514
50. Mitterschiffthaler MT, Williams SC, Walsh ND, et al. Neural basis of the emotional stroop interference effect in major depression. Psychol Med. 2008;38(2):247-256. PubMed doi:10.1017/S0033291707001523
51. Kerr N, Scott J, Phillips ML. Patterns of attentional deficits and emotional bias in bipolar and major depressive disorder. Br J Clin Psychol. 2005;44(pt 3):343-356. PubMed doi:10.1348/014466505X57755
52. McNeely HE, Lau MA, Christensen BK, et al. Neurophysiological evidence of cognitive inhibition anomalies in persons with major depressive disorder. Clin Neurophysiol. 2008;119(7):1578-1589. PubMed doi:10.1016/j.clinph.2008.03.031
53. Keilp JG, Klain HM, Brodsky B, et al. Early visual information processing deficit in depression with and without borderline personality disorder. Psychiatry Res. 2007;149(1-3):139-145. PubMed
54. Dumont GJ, de Visser SJ, Cohen AF, et al; Biomarker Working Group of the German Association for Applied Human Pharmacology. Biomarkers for the effects of selective serotonin reuptake inhibitors (SSRIs) in healthy subjects. Br J Clin Pharmacol. 2005;59(5):495-510. PubMed doi:10.1111/j.1365-2125.2005.02342.x