Article Abstract
Because this piece does not have an abstract, we have provided for your benefit the first 3 sentences of the full text.
Ms A is a 70-year-old right-handed white woman who presented with her family to the memory clinic at Banner Alzheimer’s Institute for evaluation and treatment of cognitive impairment. Her family initially noted insidious onset of speech changes, approximately 4 years prior to the evaluation, with gradual progression. Word-finding difficulty, mild stuttering, mispronounced words, and made-up words became increasingly evident.

CASE CONFERENCE
The Banner Alzheimer’s Institute Case Conference is a weekly event in which physicians and staff discuss challenging and/or teaching cases of patients seen at the Institute’s Memory Disorders Clinic. These conferences are attended by a multidisciplinary group that includes Banner Alzheimer’s Institute dementia specialists, community physicians (internal medicine, family medicine, and radiology), physician assistants, social workers, nurses, medical students, residents, and fellows.
BANNER ALZHEIMER’ S INSTITUTE
The Banner Alzheimer’s Institute located in Phoenix, Arizona, has an unusually ambitious mission: to end Alzheimer’s disease without losing a generation, set a new standard of care for patients and families, and forge a model of collaboration in biomedical research. The Institute provides high-level care and treatment for patients affected by Alzheimer’s disease, dementia, and related disorders. In addition, the Institute offers extensive support services for families and many unique and rewarding research opportunities.
Prim Care Companion CNS Disord 2016;18(4):doi:10.4088/PCC.16alz01994
© Copyright 2016 Physicians Postgraduate Press, Inc.
Submitted: May 25, 2016; accepted June 8, 2016.
Published online: August 18, 2016.
AUTHORS
David A. Weidman, MD, is a neurologist and dementia specialist at Banner Alzheimer’s Institute and a clinical assistant professor of neurology at the University of Arizona College of Medicine, Phoenix.
Anna D. Burke, MD, is a geriatric psychiatrist and dementia specialist at the Stead Family Memory Clinic of Banner Alzheimer’s Institute and a clinical assistant professor of psychiatry at the University of Arizona College of Medicine, Phoenix.
Garrett H. Riggs, MD, is a behavioral neurologist and dementia specialist at the Stead Family Memory Clinic of Banner Alzheimer’s Institute.
Helle Brand, PA, is a physician assistant at the Stead Family Memory Clinic of Banner Alzheimer’s Institute.
Jacquelynn Copeland, PhD, is a neuropsychologist at Banner Alzheimer’s Institute.
William J. Burke, MD, is a geriatric psychiatrist and the director of the Stead Family Memory Clinic of Banner Alzheimer’s Institute and a research professor of psychiatry at the University of Arizona College of Medicine, Phoenix.
Corresponding author: David A. Weidman, MD, Banner Alzheimer’s Institute, 901 E. Willetta St, Phoenix, AZ 85006 ([email protected]).
CME Background
Articles are selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME articles on a variety of topics throughout each volume. This special series of case reports about dementia was deemed valuable for educational purposes by the Publisher, Editor in Chief, and CME Institute Staff. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the article, correctly answer the questions in the Posttest, and complete the Evaluation.
CME Objective
After studying this article, you should be able to:
- Evaluate and treat geriatric patients with cognitive impairment
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1.0 hour of Category I credit for completing this program.
Date of Original Release/Review
This educational activity is eligible for AMA PRA Category 1 Creditâ„¢ through August 31, 2018. The latest review of this material was August 2016.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Larry Culpepper, MD, MPH, Editor in Chief, has been a consultant for Forest, Lundbeck, Merck, Shire, Takeda, and Elsevier Press; has been a stock shareholder of M3 My Mood Monitor; and has received royalties from UpToDate and Oxford University Press. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears on the next page.
HISTORY OF PRESENTING ILLNESS
Ms A is a 70-year-old right-handed white woman who presented with her family to the memory clinic at Banner Alzheimer’s Institute for evaluation and treatment of cognitive impairment. Her family initially noted insidious onset of speech changes, approximately 4 years prior to the evaluation, with gradual progression. Word-finding difficulty, mild stuttering, mispronounced words, and made-up words became increasingly evident. About 3 years prior to presentation, she sustained 2 minor head injuries: the first from a fall to the ground related to a mechanical trip and the second when she was struck as a pedestrian in a parking lot by a car backing out of a parking space, prompting emergency department evaluation, which included unremarkable brain computed tomography imaging. Neither trauma resulted in significant postconcussive symptoms. Functional impairments emerged 1 year before the evaluation, including errors calculating a restaurant tip, making change, and navigating when driving in an unfamiliar setting. Her husband needed to give occasional reminders about upcoming events and appointments. She was not repetitive. She was aware that she was struggling and could become visibly frustrated but with no verbal outbursts. She eventually sought out assistance from her family to help accomplish a number of complex daily tasks. In particular, her husband began managing household finances after she made a few mistakes. The cognitive symptoms appeared to worsen with excessive activity or stimuli and to improve after rest periods. She denied other mood changes, but apathy was occasionally evident to her family, with loss of interest in some of her usual activities. There were no aggressive or inappropriate behaviors, personality changes, delusions, or hallucinations. Ms A slept well, albeit while chronically taking eszopiclone at bedtime. Her appetite was fine, and she remained independent in all aspects of personal hygiene and grooming.
PAST MEDICAL HISTORY
Ms A has a history of hypertension, hyperlipidemia, premature atrial and ventricular contractions, vasovagal syncope, osteoarthritis, and mild anxiety. Bouts of stabbing left headaches and a flare-up of benign positional vertigo resolved several months prior to presentation. Her surgical history includes a hysterectomy.
ALLERGIES
Ms A reported an allergy to sulfa-containing antibiotics and shellfish.
MEDICATIONS
Current medications included eszopiclone 3 mg when necessary, metoprolol extended release 50 mg/d, lisinopril 20 mg/d, nabumetone 500 mg/d, pitavastatin 2 mg/d, esomeprazole 40 mg/d, donepezil 5 mg/d, verapamil 40 mg/d, a multivitamin, fish oil, coenzyme Q10, a probiotic, and a calcium/zinc/magnesium/vitamin D3 combination supplement.
SOCIAL HISTORY
Ms A has 17 years of formal education and worked for more than 30 years as a registered nurse. She currently lives at home with her husband and has 2 adult children, both of whom are supportive.
SUBSTANCE USE HISTORY
Ms A drank 4 glasses of wine per night until 2 years ago, and she currently drinks 2 glasses per night. Ms A smoked cigarettes for 10 years, a half pack per day, and quit in 1967.
FAMILY HISTORY
Ms A’s paternal great aunt and maternal grandmother had dementia, not formally diagnosed as Alzheimer’s disease. Her father and paternal uncles had heart disease. Her mother and maternal grandmother suffered from strokes.
On the basis of the information so far, do you think a major neurocognitive disorder is present?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Yes | 100% |
| B. No | 0% |
| C. Not enough information | 0% |
All of the participants felt that the symptoms and functional deficits provided in the clinical history were sufficient to meet the definition of a major neurocognitive disorder.
Evidence of significant cognitive decline from a previous level of performance in 1 or more areas of cognitive domains (complex attention, executive function, learning and memory, language, perceptual-motor or social cognition) based on:The DSM-5 (American Psychiatric Association, 2013) defines a major neurocognitive disorder as follows:
-
- Concern of the individual, a knowledgeable informant, or the clinician that there has been a significant decline in cognitive function and
- Substantial impairment in cognitive performance, preferably documented by standardized neuropsychological testing or, in its absence, another quantified clinical assessment.
- The cognitive deficits interfere with independence in everyday activities.
- The cognitive deficits do not occur exclusively in the context of a delirium.
- The cognitive deficits are not better explained by another mental disorder
On the basis of the information so far, what would you expect to see on the neurologic examination?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Normal | 13% |
| B. Objective nonfocal neurologic findings (including frontal release signs) | 63% |
| C. Focal neurologic findings | 0% |
| C. Insufficient information provided | 24% |
The majority of the participants present expected objective nonfocal neurologic findings, such as word-finding difficulty, paraphasic errors, and neologisms. Of the participants, 13% believed the examination would be normal; 24% thought that insufficient information was provided, specifically Ms A’s ability to read or write was not mentioned in the history.
Paraphasic error is a type of language output error commonly associated with aphasia characterized by the production of unintended syllables, words, or phrases during the effort to speak.
Neologism refers to a newly coined word or expression.
NEUROLOGIC EXAMINATION
No abnormalities were seen on gait, sensory, motor, coordination, deep tendon reflex, or cranial nerve testing. Mental status examination revealed mostly fluent speech but with occasional hesitations, “broken speech,” light stuttering, paraphasic errors, and neologisms. Ms A was able to repeat simple phrases, but complex phrases were more difficult. Comprehension, object naming, reading, and writing were intact.
PHYSICAL EXAMINATION
The physical examination was unremarkable. Ms A’s blood pressure was 120/70 mm Hg, pulse was 68 bpm, and weight was 178 lb.
LABORATORY AND RADIOLOGY RESULTS
Laboratory studies including complete blood count with differential, a comprehensive metabolic panel, vitamin B12 level, thyroid-stimulating hormone level, and sedimentation revealed no clinically significant abnormalities. Magnetic resonance images of the brain revealed mild to moderate periventricular white matter disease and mild global atrophy.
On the basis of the information so far, what would you expect the Mini-Mental State Examination (MMSE) score to be?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 26-30 | 0% |
| B. 21-25 | 44% |
| C. 16-20 | 56% |
| D. 11-15 | 0% |
| E. < 11 | 0% |
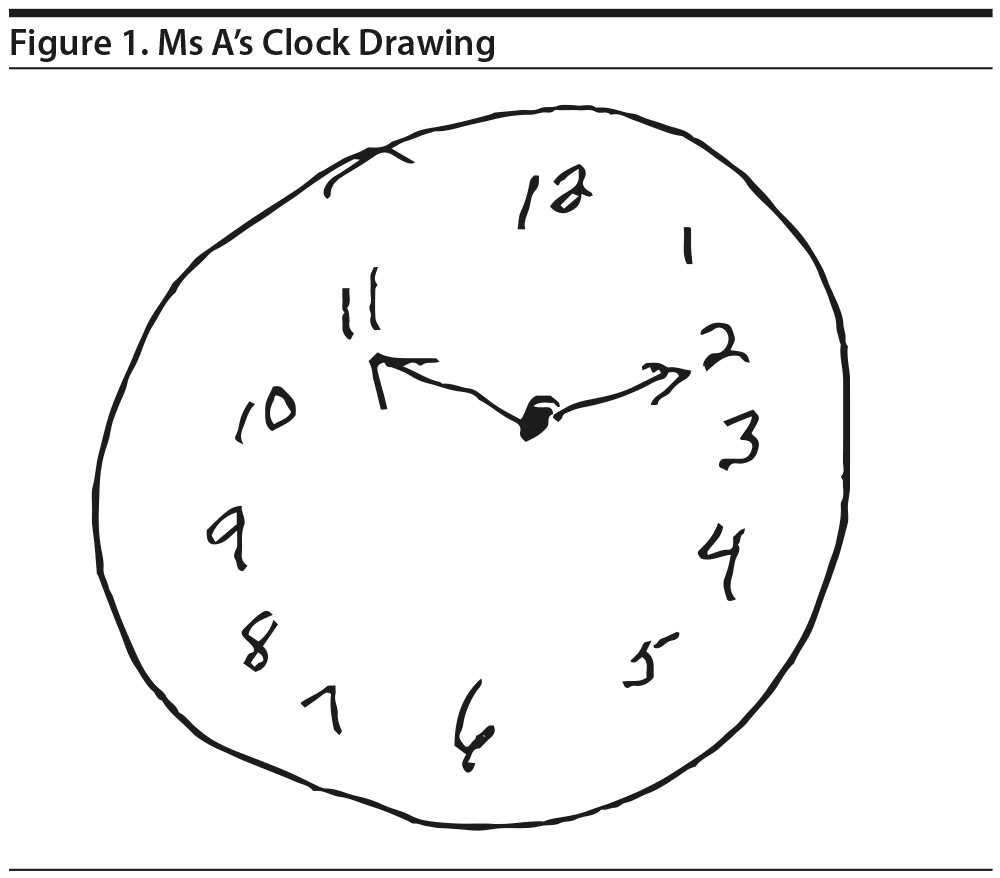
Ms A scored better than the participants expected, 27/30 on the MMSE (Folstein et al, 1975), with impairments in orientation (2 points lost) and registration (1 point lost). Other cognitive screening tests included clock drawing, which was essentially normal, but 1 point was taken off for equal lengths of the hour and minute hands (Figure 1). An auditory verbal learning test, which involves learning a list of 15 unrelated nouns, was within the normal range. Performance on initial learning trials was within normal limits, with increased recall across 5 trials, and recall of 12 of 13 initially learned words following a 20-minute delay.
On the basis of the information so far, what would you expect the Montreal Cognitive Assessment (MoCA) score to be?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 26-30 | 10% |
| B. 21-25 | 80% |
| C. 16-20 | 10% |
| D. 11-15 | 0% |
| E. < 11 | 0% |
The majority of the participants believed that the score would fall within the range of 21 to 25 points. Ms A’s MoCA test revealed a score of 21/30, with impairments in visuospatial/executive function, attention, orientation, and language. Delayed recall was intact (Figure 2).
The Montreal Cognitive Assessment (MoCA) has been shown to have a better sensitivity and specificity in detecting more subtle cognitive impairments, such as mild cognitive impairment (MCI), when compared to the MMSE. Nasreddine et al (2005) found that the MMSE had a sensitivity of 18% to detect MCI, whereas the MoCA detected 90% of MCI subjects. In the mild Alzheimer’s disease group, the MMSE had a sensitivity of 78%, whereas the MoCA detected 100%. Specificity was excellent for both the MMSE and MoCA (100% and 87%, respectively).
On the basis of the information so far, what underlying etiologic subtype of major neurocognitive disorder is present?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Alzheimer’s disease | 0% |
| B. Frontotemporal lobar dementia | 70% |
| C. Lewy body disease | 0% |
| D. Vascular disease | 0% |
| E. Traumatic brain injury | 0% |
| F. Parkinson’s disease | 0% |
| G. Due to another medical condition | 0% |
| H. Due to multiple etiologies | 30% |
The majority of participants felt that Ms A’s clinical presentation of a predominant and gradually worsening language impairment was most likely due to a frontotemporal lobar dementia. Other participants thought more than 1 etiology was probable, including the possibility of Alzheimer’s disease in conjunction with vascular features, given the insidious onset and gradual progression of symptoms, with mild to moderate vascular changes present in cerebral white matter and stroke risk factors of hypertension and hyperlipidemia.
THE TREATING PHYSICIAN’ S IMPRESSION
On the basis of the history and clinical presentation as well as the results of the cognitive and physical examinations, the treating physician felt that Ms A had a major neurocognitive disorder of mild severity (per DSM-5 criteria). The pattern of symptoms and signs was not typical for Alzheimer’s disease given that language abnormalities were the most prominent feature and verbal as well as visual short-term memory functions were intact on cognitive screening tests.
Which of the following evaluations would you schedule next?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Neuropsychological evaluation | 100% |
| B. FDG-PET | 0% |
| C. Amyloid PET | 0% |
| D. Cerebrospinal fluid analysis | 0% |
| E. APOE genotype testing | 0% |
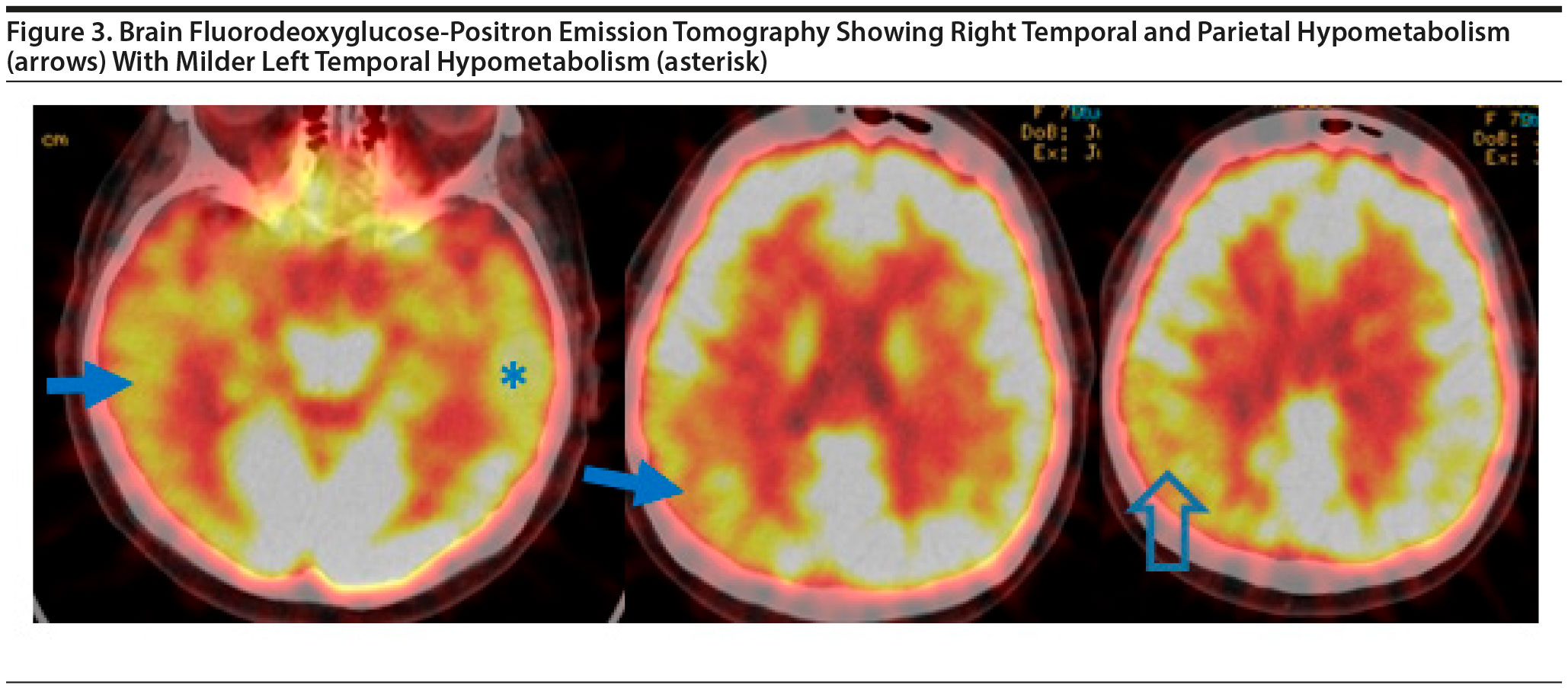
The participants felt that a neuropsychological evaluation should be carried out. In comparison to the MoCA, MMSE, and other screening tests, a comprehensive neuropsychological evaluation would better define the language dysfunction and more accurately include or exclude short-term memory impairment, and the pattern of cognitive strengths and weaknesses could help clarify the etiology of symptoms. Neuropsychological testing was not scheduled. Subsequent to the initial evaluation, the treating physician was able to review a FDG-PET scan, which had been done prior to the evaluation. The FDG-PET scan revealed predominantly temporal hypometabolism, right parietal hypometabolism, and milder left temporal hypometabolism, most consistent with Alzheimer’s disease (Figure 3).
The treating physician felt a progressive nonfluent aphasia, a type of frontotemporal lobar degeneration (FTLD), remained a possible etiology of the neurocognitive disorder, while recognizing that the patient met criteria for possible Alzheimer’s disease. Encountering diagnostic uncertainty, the treating physician had a detailed discussion with the patient and family about additional testing. An amyloid PET scan was ordered to help clarify the diagnosis.
A diagnosis of possible Alzheimer’s disease dementia can be made in 1 of 2 circumstances: (1) an atypical course, meaning the pattern of cognitive deficits meets the criteria for probable Alzheimer’s disease, but either a sudden onset of cognitive impairment is present or there is insufficient historical detail or objective documentation of progressive decline or (2) clinical criteria are met for probable Alzheimer’s disease, but there is evidence of concomitant strokes or significant cerebrovascular disease, or features of dementia with Lewy bodies other than the cognitive symptoms themselves, or evidence for another neurological disease, medical comorbidity, or medication use that could substantially impair cognition (McKhann et al, 2011).
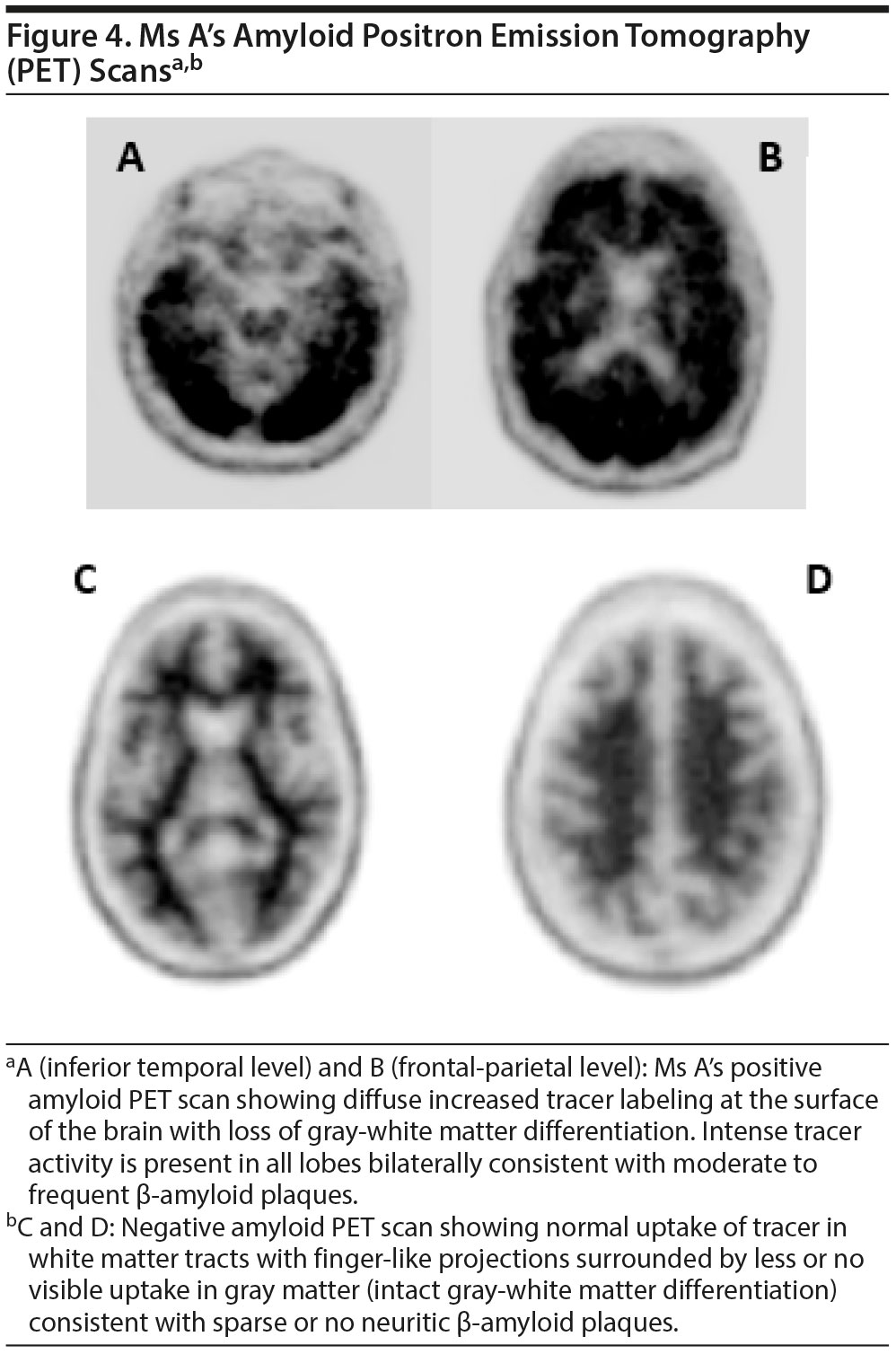
The amyloid PET scan was positive, revealing diffuse cortical uptake of the tracer (Figure 4), consistent with moderate to elevated amyloid plaque present in the cerebral cortex. A diagnosis of probable Alzheimer’s disease was made—compatible with a nonamnestic presentation, a “language variant”—with the most prominent deficits in word-finding and calculating and milder deficits in cognitive domains of executive function and attention.
Probable Alzheimer’s disease is characterized by insidious or gradual onset over months to years, not sudden over hours or days, with clear-cut history of worsening reported or observed by others. The initial and most prominent cognitive deficits most commonly reflect short-term memory dysfunction, with impairment in learning and recall of recently learned information. There should also be evidence of cognitive dysfunction in at least 1 other cognitive domain, such as reasoning and handling of complex tasks, visuospatial abilities, language functions, or changes in behavior or comportment. Less commonly, nonamnestic presentations occur when the most prominent deficits are in language, visuospatial skills, or executive function (McKhann et al, 2011).
Ms A had been taking donepezil 5 mg for several months prior to presentation. What treatment would you recommend?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Increase donepezil dosage | 100% |
| B. Switch to a different cholinesterase inhibitor | 0% |
| C. NMDA antagonist | 0% |
| D. Combination memantine/donepezil capsule | 0% |
| E. Methylphenidate | 0% |
All of the participants stated that they would increase the donepezil dosage. Cholinesterase inhibitors are first-line agents for Alzheimer’s disease. Initial US Food and Drug Administration approval of donepezil was in 1996, with efficacy demonstrated at mild to moderate stages of Alzheimer’s disease. Expansion of donepezil’s indication to use in treatment of the severe stage was approved in 2006.
Ms A experienced intermittent diarrhea, nausea, and diffuse myalgias shortly after donepezil was increased to 10 mg daily. Ms A also reported that she had felt “foggy” and fatigued ever since donepezil was initially started. What other medication indicated for Alzheimer’s disease would you recommend next?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Oral rivastigmine | 0% |
| B. Rivastigmine transdermal | 100% |
| C. Galantamine | 0% |
| D. NMDA antagonist (memantine) | 0% |
All of the participants believed that transdermal rivastigmine should be recommended next (Winblad et al, 2007). The incidence of gastrointestinal side effects with oral rivastigmine is comparable to, if not greater than, donepezil. There are no proven clinically meaningful differences among the agents in terms of efficacy that would justify changing to galantamine (Raina et al, 2008). Memantine is not indicated for the mild stage of Alzheimer’s disease. Ms A tolerated transdermal rivastigmine well, and after receiving a more specific diagnosis, she expressed interest in participating in an interventional clinical research trial for mild Alzheimer’s disease.
DISCUSSION
Alzheimer’s disease occasionally presents as a progressive aphasia characterized by logopenia, with prominent impairment in language abilities, along with problems in calculating, as Ms A’s case illustrates. She was not significantly forgetful at the initial evaluation. Logopenic aphasia due to Alzheimer’s disease is 1 of 3 well-described and not infrequently encountered variant Alzheimer’s syndromes, which may not cause significant short-term memory dysfunction at early stages, yet meet core clinical criteria of probable Alzheimer’s disease on the basis of clear-cut functional decline. Relatively isolated and disabling problems in language, visuospatial skills, or executive function can occur, classified as (1) progressive aphasia with logopenia, (2) posterior cortical atrophy (“visual variant”), and (3) a behavioral (dysexecutive) variant, respectively. A take-home lesson is to follow an epidemiologic rule of thumb: that atypical presentations of common disorders (such as Alzheimer’s disease) are more frequently encountered than typical presentations of much less common disorders (such as FTLD), and in these circumstances, early referral to a dementia specialist should strongly be considered.
As defined by postmortem neuropathologic confirmation of senile amyloid plaques and neurofibrillary tangles, Alzheimer’s disease is the most common cause of a progressive aphasia with logopenia, but a FTLD, defined by FTLD-tau neuropathology, can present similarly. In-depth assessment for a loss of grammar (agrammatism)—in speech, writing, or both—may help differentiate the 2 pathophysiologic processes. Case series have shown that FTLD is a more common etiology than Alzheimer’s disease in patients who show significantly diminished grammatical ability early in the course of a neurocognitive disorder, while Alzheimer’s disease is the more common cause when grammatical ability is preserved (Mesulam et al, 2014). Nonetheless, an overlap exists, imposing a limit on the ability of neurolinguistic testing to accurately predict underlying neuropathology (Vandenberghe, 2016). In Ms A’s case, an in vivo biomarker of underlying pathology—amyloid PET imaging—helped increase confidence in a diagnosis of Alzheimer’s disease (Wolk, 2013). Amyloid PET scanning is a powerful and recently available imaging test that can help confirm or exclude the diagnosis of Alzheimer’s disease, including its variants. Brain amyloid imaging should be considered only as an added diagnostic tool, within a comprehensive evaluation of objectively cognitively impaired patients, in diagnostically uncertain cases (Witte et al, 2015).
Logopenic aphasia is characterized by intermittent word-retrieval deficits, both in free speech and with naming. Patients may speak with fluent utterances, interspersed with frequent word-finding pauses, and on cognitive testing often show difficulty with sentence repetition, yet no signs of a disorder of motor speech, grammar, or comprehension.
The core feature of agrammatism is disrupted sentence production, with additional effort and time to retrieve most words, at virtually all times, with loss of syntax (word order and phrase structure), labored pronunciation, loss of verbs more than nouns, and misuse of parts of words that modify tense or number
FUNDING/SUPPORT
None.
DRUG NAMES
Donepezil (Aricept and others), donepezil-memantine (Namzaric), esomeprazole (Nexium and others), eszopiclone (Lunesta), galantamine (Razadyne and others), memantine (Namenda), methylphenidate (Ritalin and others), metoprolol extended release (Toprol-XL and others), pitavastatin (Livalo), rivastigmine (Exelon and others), verapamil (Calan and others).
DISCLOSURE OF OFF-LABEL USAGE
The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Financial Disclosure
Dr Anna D. Burke is a consultant for Eli Lilly and Axovant. Drs Weidman, Riggs, Copeland, and W. J. Burke and Ms Brand have no personal affiliations or financial relationships with any commercial interest to disclose relevant to the activity.
Case Conference
The Banner Alzheimer’s Institute Case Conference is a weekly event in which physicians and staff discuss challenging or teaching cases of patients seen at the Institute’s Stead Family Memory Clinic. These conferences are attended by a multidisciplinary group that includes Banner Alzheimer’s Institute dementia specialists, community physicians (internal medicine, family medicine, and radiology), physician assistants, social workers, nurses, medical students, residents, and fellows.
Disclaimer
The opinions expressed are those of the authors, not of Banner Health or Physicians Postgraduate Press, Inc.
Clinical Points
- Alzheimer’s disease occasionally will present atypically, with relative sparing and sometimes absence of impairment in short-term memory, learning, or recall. The primary care physician may encounter a dementia patient with functional decline due to predominant impairment in language, visual-spatial skills, or executive functions. In that situation, the physician should consider an Alzheimer’s disease variant and referral to a dementia specialist for further assessment.
- Amyloid positron emission tomography scanning is a powerful and recently available imaging test that can help confirm or exclude the diagnosis of Alzheimer’s disease, including its variants. Brain amyloid imaging should be considered only as an added diagnostic tool, within a comprehensive evaluation of objectively cognitively impaired patients, in diagnostically uncertain cases.
This CME activity is expired. For more CME activities, visit cme.psychiatrist.com.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Association; 2013.
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198. PubMed doi:10.1016/0022-3956(75)90026-6
McKhann GM, Knopman DS, Chertkow H, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):263-269. PubMed doi:10.1016/j.jalz.2011.03.005
Mesulam MM, Rogalski EJ, Wieneke C, et al. Primary progressive aphasia and the evolving neurology of the language network. Nat Rev Neurol. 2014;10(10):554-569. PubMed doi:10.1038/nrneurol.2014.159
Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699. PubMed doi:10.1111/j.1532-5415.2005.53221.x
Raina P, Santaguida P, Ismaila A, et al. Effectiveness of cholinesterase inhibitors and memantine for treating dementia: evidence review for a clinical practice guideline. Ann Intern Med. 2008;148(5):379-397. PubMed doi:10.7326/0003-4819-148-5-200803040-00009
Vandenberghe R. Classification of the primary progressive aphasias: principles and review of progress since 2011. Alzheimers Res Ther. 2016;8(1):16. doi:10.1186/s1
Winblad B, Cummings J, Andreasen N, et al. A six-month double-blind, randomized, placebo-controlled study of a transdermal patch in Alzheimer’s disease—rivastigmine patch versus capsule. Int J Geriatr Psychiatry. 2007;22(5):456-467. PubMed doi:10.1002/gps.1788
Witte MM, Foster NL, Fleisher AS, et al; MM. Clinical use of amyloid-positron emission tomography neuroimaging: Practical and bioethical considerations. Alzheimers Dement (Amst). 2015;1(3):358-367. PubMed
Wolk DA. Amyloid imaging in atypical presentations of Alzheimer’s disease. Curr Neurol Neurosci Rep. 2013;13(12):412. PubMed doi:10.1007/s11910-013-0412-x3195-016-0185-
Enjoy this premium PDF as part of your membership benefits!