Acute Psychosis and Type 2 Diabetes Mellitus: Should Screening Guidelines Be Revised?
Although psychosis increases the risk for developing type 2 diabetes, the temporal relationship between the onset of psychosis and the onset of diabetes has not been studied. We present 6 cases of acute psychosis, which led to the new diagnosis of type 2 diabetes during inpatient psychiatric admission within days to weeks of the psychotic episode. The implications of these findings and the efficacy of current diabetes screening guidelines are discussed.
Primary Care Companion CNS Disord 2011;13(1):e1-e5
© Copyright 2011 Physicians Postgraduate Press, Inc.
Submitted: April 25, 2010; accepted September 1, 2010.
Published online: February 17, 2011 (doi:10.4088/PCC.10br01006).
Corresponding author: Leah K. Bauer, MD, Massachusetts General Hospital, 15 Parkman St, Wang 812, Boston, MA 02144 ([email protected]).
Prior research suggests that the prevalence of diabetes among individuals with schizophrenia and affective disorders is 1.5-2 times higher than that of the general population.1 Most studies have associated the increased risk for diabetes in this population with exposure to atypical antipsychotic agents.2-4 However, several studies have found a high rate of insulin resistance, diabetes, and metabolic syndrome in those with psychotic and affective disorders, independent of exposure to antipsychotic medication.5-7 Additionally, the rate of undiagnosed diabetes and impaired glucose tolerance among psychiatric patients in general is also high, over 10% in several samples.2,8
Clinical Points
- Clinicians should maintain a high level of suspicion for undiagnosed diabetes in patients admitted for acute psychotic episodes.
- Serial fasting glucose measurements may be a sensitive and cost-effective way to detect diabetes.
- Some cases of diabetes may not be detected if current screening guidelines are followed.
Several precipitating factors to acute psychosis have been identified, including medication noncompliance, amphetamine and excessive cannabis use, stressful life events, recent childbirth, steroid use, and general medical conditions such as systemic lupus erythematosus.9-13 Several studies have suggested the possibility that the stress of acute psychosis, life events, or metabolic insult may precipitate the development of glucose dysregulation and even diabetes.14-16 However, we found no research suggesting a close temporal relationship between the development of psychosis and the development of diabetes.
This case series raises questions about the possibility of a temporal relationship between psychosis and diabetes. It profiles 6 patients who were stable and off psychiatric medication at the time they developed acute psychosis without a clear precipitant. During inpatient hospitalization, all 6 patients received a new diagnosis of diabetes.
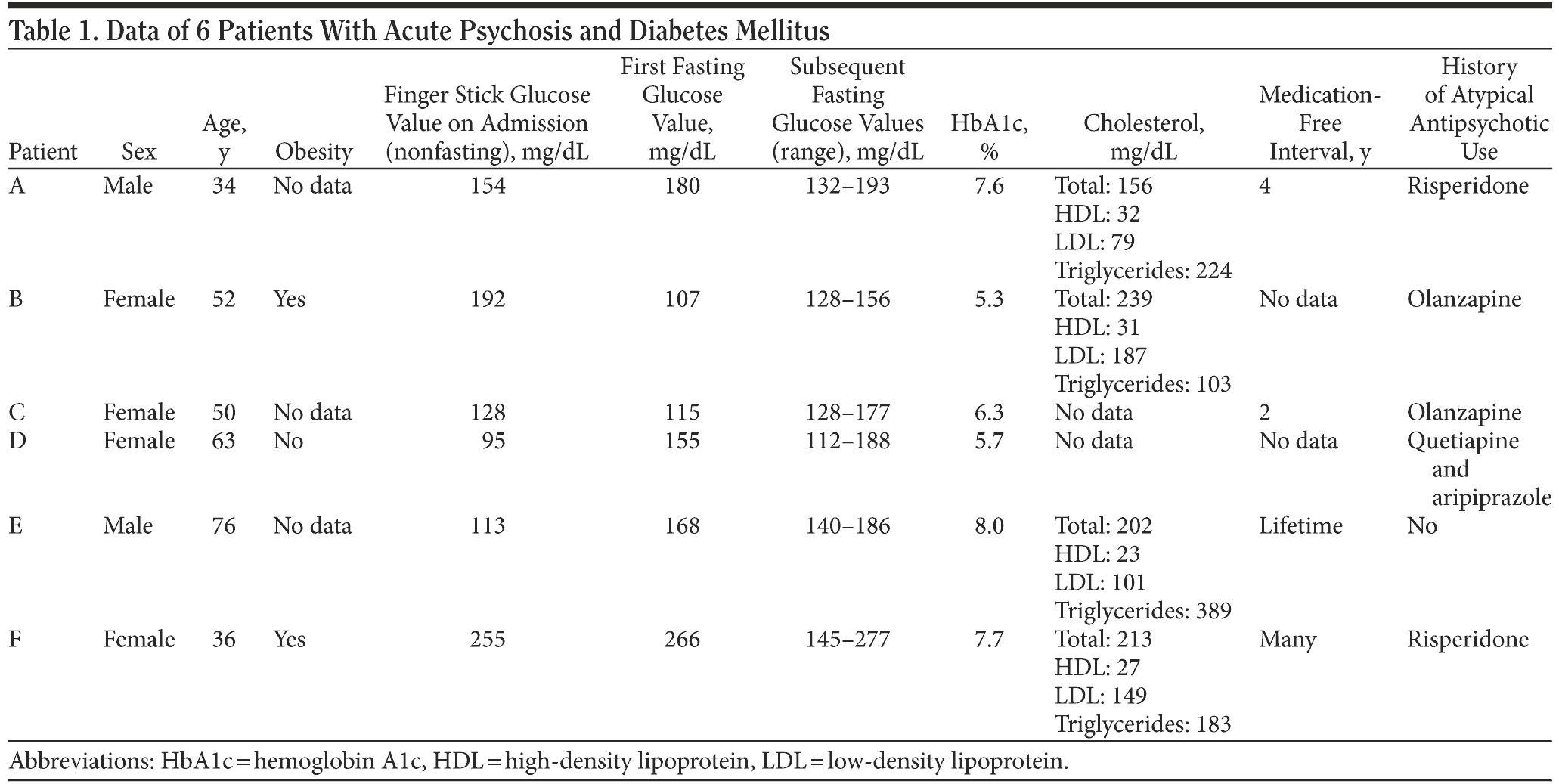
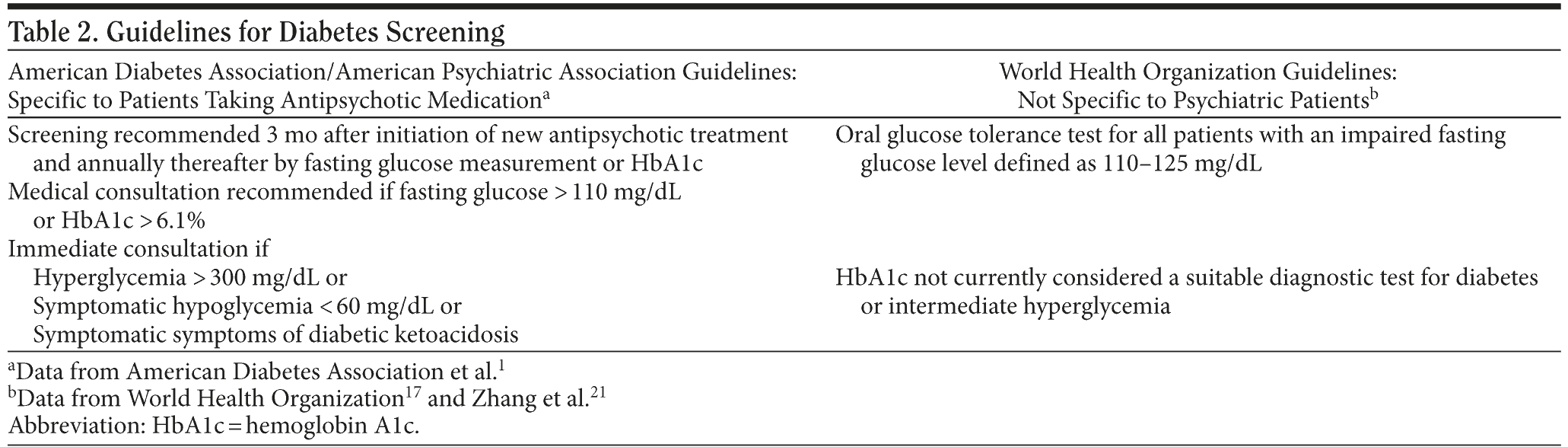
Current American Diabetes Association/American Psychiatric Association (ADA/APA) guidelines recommend a fasting glucose test or hemoglobin A1c (HbA1c) measurement annually for patients with schizophrenia and further investigation if fasting glucose is > 110 mg/dL or HbA1c is > 6.1%.1 The cases presented here call into question the efficacy and cost-effectiveness of current diabetes screening guidelines for psychiatric patients, suggesting the need for studies of the cost-effectiveness of including diabetes screening at the time of all new episodes of psychosis. Three of the 6 cases are presented in detail for illustrative purposes.
CASE 1
Patient A was a single 34-year-old white man with a history of schizophrenia and alcohol dependence. He was doing well and living in an apartment building where he also worked as the maintenance person until the police were called after he turned the power off in the building in response to psychotic symptoms. Patient A reported multiple persecutory and grandiose delusions; his affect was labile and his insight and judgment were impaired at the time that he was involuntarily admitted to the psychiatric unit.
Patient A had no known history of prior psychiatric admissions or significant past medical illnesses. He took no medications at the time of admission and reported no use of psychiatric medications in the 4 years prior to admission. In the past, he had taken risperidone, lamotrigine, bupropion, and citalopram. He smoked 1 pack of cigarettes per day and was drinking approximately 12 alcoholic drinks per day at the time of admission. He denied recent drug use, which was confirmed with a negative toxicology screen. His family history included schizophrenia in 2 cousins, alcohol dependence in his mother, depression in his father, and diabetes mellitus in both maternal grandparents.
At admission, Patient A’s laboratory values were within normal limits with the exception of a random blood glucose value of 154 mg/dL and small amounts of glucose and protein in his urine. Serial fasting finger stick blood sugar levels were checked throughout admission, which returned multiple values ranging from 132-193 mg/dL, thus establishing a diagnosis of diabetes mellitus per ADA guidelines. His HbA1c was elevated at 7.6%, which corresponds to a 3-month mean blood glucose value of 193 mg/dL.17 Patient A was diagnosed with schizophrenia, paranoid type (DSM-IV criteria) and was started on aripiprazole 10 mg/d during his hospitalization, to which he responded well. He was also started on metformin 500 mg for blood sugar control, which was tolerated well. Patient A was discharged 10 days after admission with adequate psychiatric and medical follow-up care arranged.
CASE 2
Patient B was a 52-year-old white woman with a self-reported history of bipolar I disorder. She was admitted to the hospital after being found by police walking along the side of the highway in a psychotic state. She had driven a long distance from her home several states away and then abandoned her vehicle and began walking for reasons unclear to her. She reported racing thoughts, irritability, and increased goal-directed activity on admission. She was noted to have pressured speech, religious preoccupation, magical thinking, disorganized thoughts, and multiple delusions.
Patient B had a history of several past psychiatric admissions, but documents from these admissions were unavailable. She was not taking any medications at the time of admission, but had taken haloperidol, lithium, and fluphenazine in the past. The patient was not able to attest to the duration of her medication-free interval. She had no recent substance abuse history, which was supported by a negative toxicology screen. She had a history of asthma and a family history of diabetes and alcohol dependence.
On admission, Patient B had a random blood glucose level of 192 mg/dL. Serial fasting glucose levels (mg/dL) were elevated in the 130s and 140s, establishing a diagnosis of diabetes per ADA guidelines. Her HbA1c was 5.3%, which is in the nondiabetic range, suggesting onset within recent weeks. She was diagnosed with bipolar affective disorder type 1, current episode manic with psychotic features (DSM-IV criteria). Patient B was started on several psychiatric medications that she did not respond to and was eventually placed on risperidone, oxcarbazepine, and metformin with good results. She was discharged to home in a stable condition after 36 days of inpatient stay, with adequate follow-up care arranged.
CASE 3
Patient C was a 50-year-old black woman with a history of schizoaffective disorder. She was brought to the emergency room by police after she was found flooding her apartment and trying to set it on fire. She denied all psychiatric symptoms at the time of admission, although she was noted to show signs of psychosis on mental status examination. She had not taken psychiatric medication for the past 2 years, but she had taken olanzapine in the past.
Patient C had 1 prior psychiatric admission 2 years prior to the current hospitalization. She had no past medical history. She denied a history of substance abuse and had a negative drug screen at the time of admission. Her family history was significant for a mother with schizophrenia. The patient was single with 2 grown children; she has had trouble holding a job and was on disability.
At admission, Patient C had a random blood glucose level of 128 mg/dL, which is in the normal range. Fasting blood glucose levels were checked, the first of which returned a value of 115 mg/dL, which suggests an impairment of blood sugar control, but is not diagnostic of diabetes. Follow-up fasting glucose levels ranged from 127 to 272 mg/dL, thus establishing a diagnosis of diabetes per ADA guidelines. Her HbA1c was 6.3%, which is abnormal and corresponds to a mean glucose level of 147 mg/dL.17 She was diagnosed with schizoaffective disorder, bipolar type (DSM-IV criteria). Her mental status improved after she was started on paliperidone. She was also started on metformin for glucose control. Patient C was discharged to home 10 days after her admission, with follow-up arranged for her mental health and her diabetes.
See Table 1 for data on patients D, E, and F.
DISCUSSION
The preceding cases raise several questions about the development of diabetes among individuals with psychosis. They also call the current recommended screening process for the detection of diabetes among this population into question. To our knowledge, the patients profiled in this case series had been stable and off psychotropic medications and illicit drugs, 4 of them for several years before the development of their acute psychotic episode. Since the common precipitants of medication noncompliance and illicit drug abuse were not contributing factors to the development of psychosis in these patients, it is reasonable to hypothesize that the glucose dysregulation documented in each of these patients could be associated with psychosis within a few weeks to months.
It is unclear, however, whether psychosis contributed to the development of glucose dysregulation, or inversely, whether glucose dysregulation and diabetes precipitated the psychosis. Although inconclusive, past research examines the former possibility, suggesting that the stress of acute psychosis can lead to glucose dysregulation.15,18 However, the cases presented here raise the possibility that the lack of glucose control could contribute to the development of psychosis, as these patients had no known history of diabetes prior to their psychotic symptoms and no other identifiable precipitant for psychosis. More prospective research is needed to delineate the temporal and causal relationship between psychosis and diabetes.
The present cases challenge the adequacy of current diabetes screening guidelines for patients with mental illness. Current ADA/APA guidelines recommend a fasting glucose test or HbA1c measurement annually for patients with schizophrenia and further investigation if fasting glucose is > 110 mg/dL or HbA1c is > 6.1%.1 If these measurements were applied to the above patients, we would have failed to detect diabetes in Patient B. Also, Patient D had a fasting glucose level above the cut-off value, but would not have met criteria for further investigation if HbA1c were measured alone (see Table 1). World Health Organization guidelines recommend performing an additional oral glucose tolerance test (OGTT) on all patients with an impaired fasting glucose (Table 2).17 Prior research comparing these 2 strategies in patients with schizophrenia and schizoaffective disorder revealed that the ADA/APA guidelines detected diabetes only 46.2% of the time, while the WHO strategy detected 96.2% of diabetes cases.19 Routine screening with a single annual fasting glucose test or HbA1c measurement may be inadequate to detect diabetes among the high-risk population of patients with new episodes of psychosis.
While authors of the van Winkel study19 recommended follow-up OGTT testing for patients with impaired fasting glucose, the cost-effectiveness and feasibility of this strategy warrant consideration. The estimated costs of a fasting plasma glucose test, HbA1c test, and OGTT in 1998 were $28, $38, and $106, respectively.20 These numbers suggest that a third strategy, such as serial fasting plasma glucose measurements, may be more cost-effective than OGTTs. However, the efficacy of this strategy in psychiatric inpatients is unknown.
Also, because of the careful timing and unpleasant liquid ingested during an OGTT, patients and health care staff may find serial fasting plasma glucose measurements easier to comply with than the OGTT. Zhang and associates21 agree that among all adults, fasting plasma glucose measurement is an effective detection strategy in those unwilling to comply with an OGTT. Additionally, in agreement with current WHO guidelines, they found that HbA1c testing was not a sensitive or cost-effective screening strategy when compared to other strategies.21
Of the 6 cases reviewed, all patients received a diagnosis of diabetes by the ADA criteria of fasting plasma glucose values above 126 mg/dL on 2 separate days. However, 3 of these 6 patients had random blood glucose values at admission under 130 mg/dL. We hope this finding encourages clinicians to maintain a high level of suspicion for diabetes among psychotic patients, even if a random blood glucose level on admission is not significantly elevated. Given the findings in this case series and the sporadic outpatient follow-up of many psychiatric patients, we believe that fasting plasma glucose screening is warranted for every psychiatric admission for psychosis, regardless of initial random glucose values, and that the impaired fasting glucose test should be followed by serial fasting measurements or an OGTT to adequately rule out a diagnosis of diabetes. We urge clinicians to keep a high index of suspicion for diabetes in patients with new psychotic episodes.
Limitations of this study include the small number of cases and the paucity of quantitative data. We were not able to determine the stability of blood glucose measurements in the time before admission, especially for patients who had few or no admissions in the years prior to the study interval. We also were not able to determine the true length of the reported medication-free intervals of these patients, as this information was sometimes not reported in the medical record, and patient history was thought to lack consistent reliability. It is unclear whether other precipitating factors for psychosis besides medication noncompliance and illicit drug abuse were present, such as increased life stress or other general medical conditions associated with psychosis. Additionally, alcohol abuse was a potential confounding factor in Patient A, which may have contributed to the development of psychosis. A great amount of case-to-case variability was present, with differences in diagnosis, medication-free interval, past medications taken, and family history. Factors such as these can possibly account for increased susceptibility to psychosis or diabetes and potentially could confound results.
Despite its limitations, this case series suggests several directions for future research. To further examine the temporal relationship between psychosis and diabetes, a prospective study of patients with schizophrenia that measures blood sugar levels at regular intervals over time may be helpful. It would also be of interest to examine the tenacity of a diabetes diagnosis in an individual with acute psychosis once the psychotic episode has resolved. Glucose dysregulation may be a transient phenomenon in these patients, and it is important to study whether elevated glucose during acute psychosis necessarily predicts diabetes later in life. If hyperglycemia was found to be often a transient phenomenon, there would be additional implications regarding use of glucose-regulating medication and outpatient follow-up. Additionally, a comparative study should examine the cost-effectiveness of diabetes screening strategies for all patients admitted for a new episode of psychosis.
Drug names: aripiprazole (Abilify), bupropion (Wellbutrin, Aplenzin, and others), citalopram (Celexa and others), haloperidol (Haldol and others), lamotrigine (Lamictal and others), lithium (Lithobid and others), metformin (Metaglip, Glucovance, and others), olanzapine (Zyprexa), oxcarbazepine (Trileptal and others), paliperidone (Invega), quetiapine (Seroquel), risperidone (Risperdal and others).
Author affiliations: Massachusetts General Hospital, Boston (Dr Bauer); and Department of Psychiatry, University of Cincinnati College of Medicine, Ohio (all authors).
Potential conflicts of interest: None reported.
Funding/support: None reported.
Acknowledgment: The authors thank Jeff Huffman, MD, Department of Psychiatry, Massachusetts General Hospital, Boston, for assistance with manuscript review. Dr Huffman has no financial or other affiliations relevant to the subject of this article.
REFERENCES
1. American Diabetes Association, American Psychiatric Association, American Association of Clinical Endocrinologists, et al. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;27(2):596-601. PubMed doi:10.2337/diacare.27.2.596
2. Taylor D, Young C, Mohamed R, et al. Undiagnosed impaired fasting glucose and diabetes mellitus amongst inpatients receiving antipsychotic drugs. J Psychopharmacol. 2005;19(2):182-186. PubMed doi:10.1177/0269881105049039
3. Reist C, Mintz J, Albers LJ, et al. Second-generation antipsychotic exposure and metabolic-related disorders in patients with schizophrenia: an observational pharmacoepidemiology study from 1988 to 2002. J Clin Psychopharmacol. 2007;27(1):46-51. PubMed doi:10.1097/JCP.0b013e31802e5126
4. Basu A, Meltzer HY. Differential trends in prevalence of diabetes and unrelated general medical illness for schizophrenia patients before and after the atypical antipsychotic era. Schizophr Res. 2006;86(1-3):99-109. PubMed doi:10.1016/j.schres.2006.04.014
5. Regenold WT, Thapar RK, Marano C, et al. Increased prevalence of type 2 diabetes mellitus among psychiatric inpatients with bipolar I affective and schizoaffective disorders independent of psychotropic drug use. J Affect Disord. 2002;70(1):19-26. PubMed doi:10.1016/S0165-0327(01)00456-6
6. Thakore JH. Metabolic disturbance in first-episode schizophrenia. Br J Psychiatry Suppl. 2004;184(47):S76-S79. PubMed doi:10.1192/bjp.184.47.s76
7. van Nimwegen LJ, Storosum JG, Blumer RM, et al. Hepatic insulin resistance in antipsychotic naive schizophrenic patients: stable isotope studies of glucose metabolism. J Clin Endocrinol Metab. 2008;93(2):572-577. PubMed doi:10.1210/jc.2007-1167
8. Arlotto E, Felicé MP, Favre C, et al. Intérêt du bilan biologique d’ entrée dans le dépistage de comorbidités chez les patients hospitalisés en psychiatrie. Encephale. 2008;34(1):61-65. PubMed doi:10.1016/j.encep.2007.01.003
9. Uçok A, Polat A, Cakir S, et al. One year outcome in first episode schizophrenia: predictors of relapse. Eur Arch Psychiatry Clin Neurosci. 2006;256(1):37-43. PubMed doi:10.1007/s00406-005-0598-2
10. Holtmann M, Becker K, Hartmann M, et al. Gibt es einen zeitlichen zusammenhang von substanzmissbrauch und psychose bei jugendlichen? Z Kinder Jugendpsychiatr Psychother. 2002;30(2):97-103. PubMed doi:10.1024//1422-4917.30.2.97
11. Cullberg J. Stressful life events preceding the first onset of psychosis: an explorative study. Nord J Psychiatry. 2003;57(3):209-214. PubMed doi:10.1080/08039480310001373
12. Kendell RE, Chalmers JC, Platz C. Epidemiology of puerperal psychoses. Br J Psychiatry. 1987;150(5):662-673. PubMed doi:10.1192/bjp.150.5.662
13. Appenzeller S, Cendes F, Costallat LT. Acute psychosis in systemic lupus erythematosus. Rheumatol Int. 2008;28(3):237-243. PubMed doi:10.1007/s00296-007-0410-x
14. Mooy JM, de Vries H, Grootenhuis PA, et al. Major stressful life events in relation to prevalence of undetected type 2 diabetes: the Hoorn Study. Diabetes Care. 2000;23(2):197-201. PubMed doi:10.2337/diacare.23.2.197
15. Shiloah E, Witz S, Abramovitch Y, et al. Effect of acute psychotic stress in nondiabetic subjects on beta-cell function and insulin sensitivity. Diabetes Care. 2003;26(5):1462-1467. PubMed doi:10.2337/diacare.26.5.1462
16. Marcelis M, Cavalier E, Gielen J, et al. Abnormal response to metabolic stress in schizophrenia: marker of vulnerability or acquired sensitization? Psychol Med. 2004;34(6):1103-1111. PubMed doi:10.1017/S0033291703001715
17. Definition and diagnosis of diabetes mellitus and intermediate hyperglycemia: report of a WHO/IDF consultation. World Health Organization. http://whqlibdoc.who.int/publications/2006/9241594934_eng.pdf. Accessed January 6, 2011.
18. Dinan TG. Stress and the genesis of diabetes mellitus in schizophrenia. Br J Psychiatry Suppl. 2004;184(47):S72-S75. PubMed doi:10.1192/bjp.184.47.s72
19. van Winkel R, De Hert M, Van Eyck D, et al. Screening for diabetes and other metabolic abnormalities in patients with schizophrenia and schizoaffective disorder: evaluation of incidence and screening methods. J Clin Psychiatry. 2006;67(10):1493-1500. PubMed doi:10.4088/JCP.v67n1002
20. The cost-effectiveness of screening for type 2 diabetes: CDC diabetes cost-effectiveness study group, centers for disease control and prevention. JAMA. 1998;280(20):1757-1763. PubMed
21. Zhang P, Engelgau MM, Valdez R, et al. Costs of screening for pre-diabetes among US adults: a comparison of different screening strategies. Diabetes Care. 2003;26(9):2536-2542. PubMed doi:10.2337/diacare.26.9.2536