Treatment sequences for the multisymptomatic child with bipolar disorder are not adequately described or based on a systematic clinical trial database, and systems for longitudinal tracking of symptoms are rarely utilized. We present a patient whose symptoms of depression, anxiety, attention-deficit/hyperactivity disorder, oppositional behavior, and mania are rated by a parent and plotted on a weekly basis in the Child Network under a Johns Hopkins Institutional Review Board-approved protocol. This 9-year-old girl remained inadequately responsive to lithium or risperidone. We describe a range of other treatment options and a possible sequence for their introduction. We encourage the use of systematic longitudinal ratings to help better visualize course of symptom fluctuations and response of the child to treatment. Given the highly fluctuating course of many symptoms in very young children as illustrated here, prospective monitoring appears essential. The current case also highlights the great unmet need for comparative effectiveness data in children less than 10 years of age to better guide clinical therapeutics.
A Multisymptomatic Child With Bipolar Disorder:
How to Track and Sequence Treatment
ABSTRACT
Treatment sequences for the multisymptomatic child with bipolar disorder are not adequately described or based on a systematic clinical trial database, and systems for longitudinal tracking of symptoms are rarely utilized. We present a patient whose symptoms of depression, anxiety, attention-deficit/hyperactivity disorder, oppositional behavior, and mania are rated by a parent and plotted on a weekly basis in the Child Network under a Johns Hopkins Institutional Review Board–approved protocol. This 9-year-old girl remained inadequately responsive to lithium or risperidone. We describe a range of other treatment options and a possible sequence for their introduction. We encourage the use of systematic longitudinal ratings to help better visualize course of symptom fluctuations and response of the child to treatment. Given the highly fluctuating course of many symptoms in very young children as illustrated here, prospective monitoring appears essential. The current case also highlights the great unmet need for comparative effectiveness data in children less than 10 years of age to better guide clinical therapeutics.
Prim Care Companion CNS Disord 2017;19(4):17r02122
https://doi.org/10.4088/PCC.17r02122
© Copyright 2017 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry and Behavioral Sciences, George Washington University School of Medicine, Washington, DC
bBipolar Collaborative Network, Bethesda, Maryland
cDivision of Child and Adolescent Psychiatry, Johns Hopkins Hospital/Bloomberg Children’s Center, Baltimore, Maryland
*Corresponding author: Robert M. Post, MD, Bipolar Collaborative Network, 5415 W Cedar Ln, Ste 201-B, Bethesda, MD 20814 ([email protected]).
Childhood-onset bipolar disorder is rarely characterized by distinct episodes of pure mania or depression, but more often is highly fluctuating, multifaceted, and accompanied by multiple comorbidities. Both inpatients and outpatients tend to run a difficult course on prospective follow-up, remaining symptomatic as much as two-thirds of the time of follow-up.1–6 How much of this adverse course is related to a more serious and difficult presentation of bipolar disorder in childhood than in adulthood7 as opposed to delayed8,9 or inadequate treatment6 is unknown.
Most longitudinal follow-up is accomplished with either repeated cross-sectional measures of psychopathology or periodic (usually 6-month to 1-year) retrospective ratings of the degree of symptomatology on a severity of dysfunction scale. We present the current case description in order to begin a dialogue about treatment alternatives and sequences for the difficult-to-treat child with a complex, multifaceted presentation of bipolar disorder and illustrate the unique advantages of more regular (weekly) prospective ratings of 5 symptom domains available for free in the Child Network (www.bipolarnews.org; click on Child Network).
The therapeutic approaches to childhood-onset bipolar illness have not been adequately studied or elucidated. Few controlled or industry-supported studies allow children under age 10 to enter, and most information is extrapolated from controlled studies in adults and is largely based on expert opinion10,11 aimed almost entirely toward children with a diagnosis of bipolar I disorder (BP-I). Yet, bipolar not otherwise specified is the most common presentation of bipolar spectrum illness in very young children,3 and these children are rarely systematically studied. One notable exception is the study by Geller et al12 assessing randomized lithium, valproate, or risperidone in a mixed group of bipolar spectrum patients wherein they found superiority of risperidone over both lithium and valproate both in the first monotherapy phase and in a second combination phase (ie, risperidone plus either other drug was superior to the lithium-valproate combination).
Complicating the picture are the findings that anxiety, attention-deficit/hyperactivity disorder (ADHD), and disruptive behavior disorders are common precursors to BP-I and BP-II diagnoses and frequent comorbidities once bipolar illness has emerged,2,13–15 particularly in children in the United States compared to European children.16,17 In addition, there is considerable debate and disagreement about the frequency and specificity of the diagnoses of severe mood dysregulation and its close relative recently added to the DSM-5, dysphoric mood dysregulation disorder.18 Although these latter syndromes are associated with considerable dysfunction, the therapeutic approaches to these disorders have also not been defined.
Given these deficits in the literature, we discuss treatment options for the multisymptomatic child with bipolar disorder with the awareness and caveat that these treatments remain speculative and perhaps controversial. We then speculate about what treatments, all of which are off-label, might be more helpful to this child. Given the dearth of systematic studies of this type of child in the literature, we hope this discussion will help engender a new dialogue among clinicians, as well as a much-needed new round of treatment studies.
Here, we present a case study of a 9-year-old girl with a complex presentation of multiple symptom domains (including anxiety, depression, ADHD, oppositional behavior, and mania) who had not adequately responded to lithium, risperidone, guanfacine, or melatonin. We illustrate the utility of the weekly ratings in describing the course of illness and response or inadequacy of response to treatment.
METHODS
The Child Network enlists parents of children aged 2–12 years to rate their child on a weekly basis on a secure website under the auspices of a protocol approved by the Johns Hopkins Institutional Review Board. After an informed consent, parents fill out a brief background/demographics form and a more detailed symptom checklist and then receive e-mails each Sunday for ratings of the severity of depression, anxiety, ADHD, oppositional behavior, and mania.19 Pharmacologic or psychosocial treatments are indicated, and side effects and tolerability are rated.
Parents could enter children into the Child Network if they were currently asymptomatic or already experiencing mood and behavioral difficulties. A key element of the Child Network is the ability of parents to print out the weekly longitudinal ratings and bring these to a treating clinician or physician to more easily evaluate symptom course and response to treatment.
In the current case, the patient’s father completed the informed consent for entry into this naturalistic study, a demographics questionnaire (which includes what diagnoses the child may have already received in the community), and a detailed symptom checklist, each of which take about 15–20 minutes to complete. Then, the weekly ratings of the 5 symptom domains, which take just a few minutes, were completed with a high degree of consistency for almost 1 year.
The longitudinal chart of the weekly ratings that the parent could print out is similar to that illustrated in Figure 1, but the format of the figure has been modified for clarity of presentation for the journal. Each of the weekly ratings of the symptoms of anxiety, depression, ADHD, oppositional behavior, and mania has been graphed as rated as 0 = not present, 1 = mild, 2 moderate, and 3 = severe based on the degree of dysfunction that was associated with the child’s usual family, social, or educational roles. Drug treatments and their doses are plotted above the symptoms, while the different psychotherapies that were employed were rated, but are not illustrated here, although they included intermittent family-focused therapy and special and home schooling.
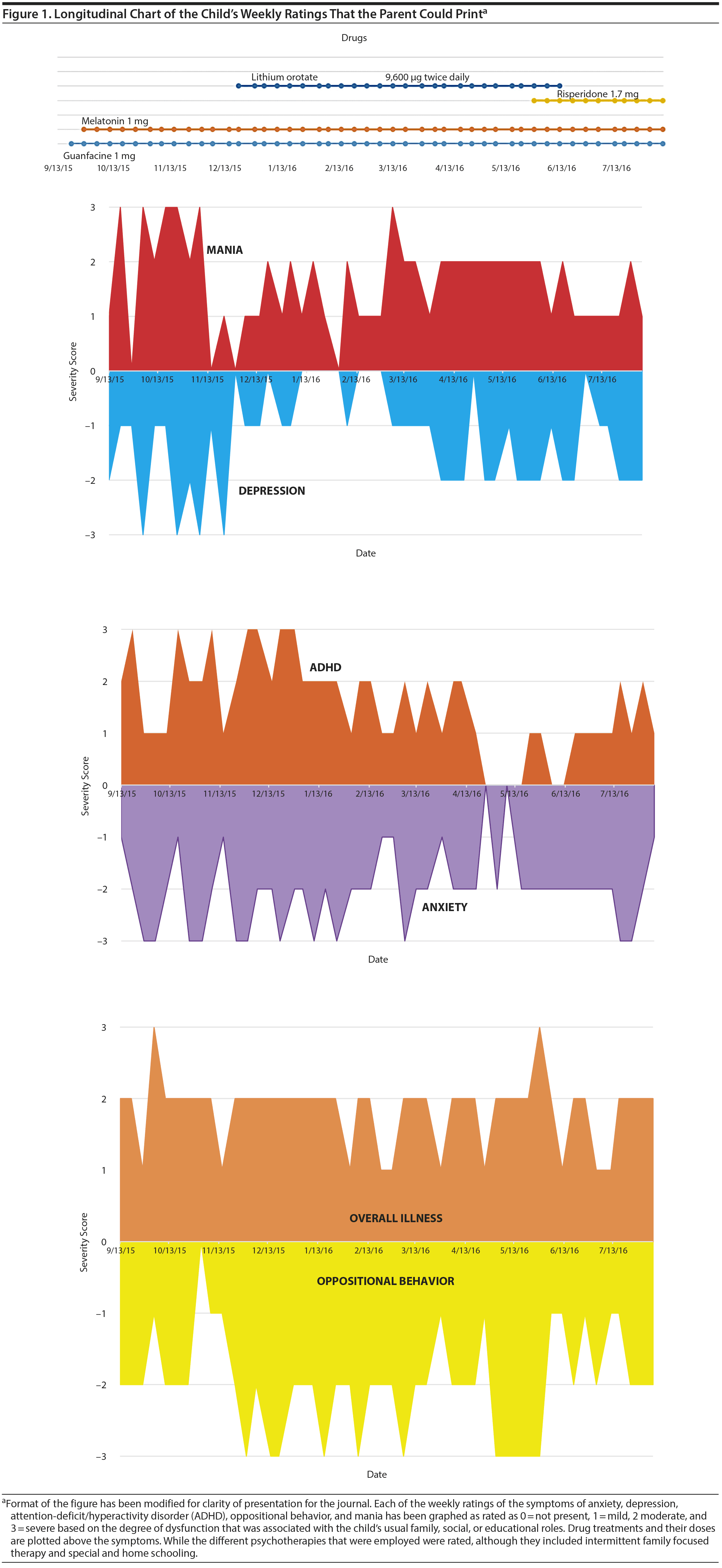
All of the demographic attributes of this girl and the case have been modified to maintain anonymity, and informed consent for publication of the weekly symptoms was obtained.
RESULTS
As illustrated in Figure 1, many weeks of severe mania, depression, and ADHD initially appeared to improve with the introduction of lithium orotate (4,800 ug in combination with the previously started guanfacine 1 mg/d and melatonin 1 mg/d). However, despite continued treatment with lithium (up to 9,800 ug twice a day), oppositional behavior worsened during the period from November 2015 to March 2016, and moderate depression re-emerged in April 2016. Anxiety was also generally less severe from December 2015 to July 2016, and weekly ratings of overall illness remained largely in the moderate severity range (not illustrated).
The switch from lithium to risperidone (maximum dose 1.7 mg/d) in June 2016 appeared to result in some improvement in mania (from mostly moderate to mostly mild), little change in moderate but highly fluctuating depression ratings, and an exacerbation in ADHD symptoms.
The patient’s diagnosis was assessed in 3 ways. In the community, the patient had been previously diagnosed as having BP-II and 3 anxiety disorder comorbidities, including school phobia, generalized anxiety disorder, and obsessive-compulsive disorder. These (1) retrospective diagnoses in the community obtained from the demographics questionnaire corresponded well with (2) the detailed parental checklist of behaviors and symptoms that the child had experienced in the 2 previous weeks. For example, mania items were scored 47 of a maximum score of 68, and a description of BP-II, ie, hypomania lasting at least 4 days, was positive. Depression was scored 15 of a maximum of 52, anxiety 24 of 32, ADHD 21 of 24, and disruptive behavioral disorder 19 of 40. (3) The prospective weekly ratings of anxiety, depression, ADHD, oppositional behavior, and mania illustrated in Figure 1 were then entirely consistent with (1) the community diagnoses and (2) the detailed symptom checklist.
Illness Course
Given the 6 weeks of moderate to severe mania (in October and November 2015), it would appear that this patient would now most likely meet criteria for a diagnosis of BP-I illness (DSM-5). She also experienced considerable other moderate to severe symptoms, including anxiety, depression (suggesting the presence of mixed mania), ADHD, and persisting moderate oppositional behavior (suggestive of a comorbid oppositional defiant disorder).
Lithium orotate appeared to initially help manic, ADHD, and anxious symptoms, but on lithium orotate, ADHD symptoms and depression worsened, and these were not further ameliorated with the switch to risperidone, although oppositional behavior did improve. Overall, the child had improved slightly during the 9 to 10 months of ratings, moving from generally severe ratings in multiple domains initially to generally moderate ratings, but she has remained persistently quite symptomatic.
DISCUSSION
Treatment Options for Targeting
Symptoms to Achieve Remission
Given the paucity of data in the literature to guide a systematic approach to therapeutics of very young children with multiple types of symptomatology, careful assessment of the responses to treatment of the individual child is of great importance. Having detailed and frequent feedback from parents about course of symptoms, degree of improvement observed, and tolerability of the treatments given that is systematized and available from parental ratings in the Child Network is likely to be of great assistance to the treating physician. The following is a discussion of possible treatment strategies and sequences that could be employed in complex multisymptomatic cases such as the one we have illustrated.
The general treatment goals would include mood stabilization prior to use of ADHD medications; choosing drugs with the greatest tolerability and safety; additionally targeting residual symptoms with appropriate medications as supplemented by nutraceuticals; recognizing that complex combination treatment may often be necessary; and combined use of medications and family education and therapy may be the most fruitful approach.
Mood Stabilizers and Atypical Antipsychotics
to Maximize Antimanic Effects
None of the options discussed here have US Food and Drug Administration (FDA) approval for use in children under 10 years of age; so, all of the suggestions are off-label. Furthermore, the authors list their suggestions with the full awareness that they may differ radically from what many other investigators in the field might suggest.20 In fact, it is this large array of diverse options (with a range of evidence supporting them from moderately strong to almost nonexistent) that is part of the rationale for this section. The algorithms for the inadequately responsive young child with bipolar disorder are poorly described in the literature and lack a systematic database.11,21 The traditional gold standard in evidence-based medicine of a positive randomized placebo-controlled clinical trial is of little use here because of the paucity of these studies in children with bipolar disorder.
Nonetheless, one potential approach to this patient’s difficulties that does have evidentiary support22 would be the add back of lithium to risperidone, as combinations were more effective than monotherapy in the study by Geller et al,12 especially when they involved an atypical antipsychotic such as risperidone. This might include the switch from lithium orotate to lithium carbonate for which there is a larger database.23 Combinations of lithium and valproate were also more effective than either monotherapy (with many patients also needing stimulants) in the studies by Findling et al.24 Most children also needed combinations of mood stabilizers (lithium, carbamazepine, valproate) in the study by Kowatch et al.25
Targeting ADHD
Increasing severity of ADHD despite improving mania speaks to the possible utility of adding a stimulant to the regimen that already includes the noradrenergic α2 agonist guanfacine. This would be supported by the data of Scheffer et al26 that stimulant augmentation for residual ADHD symptoms does not exacerbate mania and that the combination of a stimulant and guanfacine may have more favorable effects than stimulants alone.27 There is general consensus in the field that one should achieve adequate mood stabilization first prior to using low to moderate, but not high, doses of stimulants. Thus, in the face of an inadequate response to the lithium-risperidone combination in this child, stimulants could be deferred until better mood stabilization was achieved.
Other Approaches to
Mood Stabilization and Antianxiety Effects
The anticonvulsant mood stabilizers (carbamazepine, lamotrigine, and valproate) each have considerable mood-stabilizing and antianxiety effects at least in adults with bipolar disorder. With inadequate mood stabilization of this patient on lithium and risperidone, we would consider the further addition of lamotrigine. Lamotrigine appears particularly effective in adults with bipolar disorder who have a personal history and positive family history of anxiety (rather than mood disorders), and it has positive open data in adolescents with bipolar depression28 and in a controlled study29 of maintenance (but only in teenagers aged 13–17 and not in preteens aged 10–12). With better mood stabilization, anxiety symptoms usually diminish concomitantly, and we would pursue these strategies in preference to the use of antidepressants for depression and anxiety in young children with bipolar disorder.
Valproate is less desirable in females because of the development of menstrual irregularities, polycystic ovarian syndrome, and, in women of child-bearing age who become pregnant, the triple liabilities for the exposed fetus of spinal bifida, other major malformations, and major development delays with a loss of some 9 IQ points on average.30
Carbamazepine appears to be more effective in adults with bipolar disorder who have a negative family history of mood disorders19,31 (in contrast to lithium response predicted by a positive family history of mood disorder). While the overall results of oxcarbazepine in childhood mania were negative, they did exceed placebo in the youngest patients (aged 7–12) as opposed to the older adolescents (aged 13–18).32 There are long-acting preparations of both carbamazepine and oxcarbazepine (Equetro and Oxtellar) that would allow for all nighttime dosing to help with sleep and reduce daytime side effects and sedation.33,34 Although data for its antimanic and antidepressant effects in adults are stronger for carbamazepine than oxcarbazepine, given positive data with oxcarbazepine in younger children,32 the lower incidence of severe white count suppression, and its minimal effects in inducing hepatic enzymes cytochrome P450 3A4 compared to carbamazepine, oxcarbazepine might deserve careful consideration.
Other Alternative Atypical Antipsychotics: Aiming for Better Tolerability and Antidepressant Efficacy
In the study by Geller et al,12 risperidone had more side effects than lithium or valproate, including more weight gain and prolactin elevations. These findings along with the fact that risperidone is not FDA approved for unipolar or bipolar depression in adults suggest the possibility of switching this child to another atypical with better antidepressant and antianxiety effects. This might also open up the ability to go to higher doses for antimanic effects without having to worry as much about risperidone’s prolactin elevations, weight gain, and extrapyramidal side effects with the potential need for an anticholinergic.
Quetiapine and lurasidone are approved for bipolar depression in adults, but neither is approved for children, and while the weight gain of quetiapine is about equal to that of risperidone, lurasidone has little weight gain liability and no other metabolic side effects. Lurasidone has not been studied in mania, but DelBello et al35 presented data showing that compared to placebo, lurasidone (20–80 mg/d) exhibited highly significant antidepressant effects in children aged 13–17 years old with bipolar depression.
In children, weight gain on aripiprazole can be minimal to substantial, but aripiprazole decreases prolactin because of its partial dopamine agonist properties. While aripiprazole failed to show efficacy in bipolar depression in adults, it is indicated as an adjunct to antidepressants in unipolar depression. Ziprasidone has antimanic effects in adults and children36 and has the advantage of being relatively weight neutral, and recent data37 indicate its effectiveness as an adjunct in adults with mixed depression.
Olanzapine and clozapine are to be relatively avoided because of their propensity for weight gain greater than most of the other atypicals and the need for weekly white count monitoring with clozapine. Yet, clozapine has the highest rate of antimanic response among the atypicals in adults and has been used successfully in childhood-onset schizophrenia.
If the child developed considerable weight gain, one author (R.M.P.) would switch risperidone to lurasidone or ziprasidone, while another (R.L.F.) would consider the addition of aripiprazole instead. If anxiety and depression were compounded with continued insomnia (despite the use of melatonin), one might instead consider the switch to quetiapine even though it failed in a placebo-controlled trial38 of childhood bipolar depression. With an excellent mood and anxiety response, but substantial residual symptoms of ADHD and oppositional behavior remaining, a stimulant could then be added as noted previously.
From the uncertainties noted previously, the conduct of comparative studies of efficacy and tolerability of the atypical antipsychotics in children with bipolar disorder is sorely needed. Parenthetically, this could be accomplished with relative ease with practical clinical trials using randomized but open designs.39 This need is now further magnified by the FDA approval of 2 dopamine partial agonists in addition to aripiprazole. Brexpiprazole is approved for schizophrenia and as an adjunct to antidepressants in unipolar depression. Cariprazine is FDA approved for schizophrenia and mania and has positive placebo-controlled data in 1 of 2 studies40 each in bipolar depression and as an adjunct in unipolar depression.
Nutriceutical Approaches to Depression,
Anxiety, and Oppositionality
With some, but incomplete, degrees of improvement, such as that seen in Figure 1, we would add a series of adjuncts to the combination of an atypical, lithium, and another mood stabilizer. An excellent option for residual anxiety and depression would be the addition of N-acetylcysteine, which has positive placebo-controlled data in adults with bipolar depression, is helpful in potentiating the effects of selective serotonin reuptake inhibitors (SSRIs) in obsessive-compulsive disorder, and helps decrease a wide variety of pathological habits and addictions.19,41–43 Its tolerability and utility on irritability in children is documented by 3 placebo-controlled studies in children with autism either in monotherapy44 or in 2 others as an adjunct to risperidone.45,46 N-acetylcysteine is sold without a prescription in health food stores, and doses of 500 mg twice a day usually are increased slowly on a weekly basis to 2,000 to 2,700 mg/d.
Although the data are not unequivocal, there is some support for the use of omega-3 fatty acids to target depression47 and ADHD.48 Because of its safety, it may merit adjunctive use in the combined depression and ADHD symptoms seen in this patient.49
A very high percentage of children with serious psychiatric illness present with deficiencies in vitamin D3.50 Among other mixed results, a recent placebo-controlled study51 in unipolar depressed adults of vitamin D3 (1,500 IU/day in those who were not selected for a vitamin D3 deficiency) as augmentation of fluoxetine was positive. Thus, given the high incidence of outright deficiency of vitamin D3 and the positive open data52 in patients aged 6–17 years old with bipolar disorder, we would add supplemental vitamin D3.
In those who might have had gene testing, supplementation with l-methylfolate (rather than folate itself) for those with the methyl-tetrahydrofolate reductase deficiency could also be helpful with depression as it is in adults (as an augment to SSRIs).53
If this child persisted in having high levels of oppositional behavior despite multiple attempts at mood stabilization and stimulant augmentation, augmentation with a complex combination of vitamins and minerals such as EMPowerplus might be considered.47 Small open clinical trials54,55 have indicated the effectiveness and safety of EMPowerplus in the treatment of medication-nonresponsive children with bipolar disorder often complicated by extreme behavioral dyscontrol. However, there are potential adverse reactions when this preparation is used in combination with lithium such that the dose of lithium should be reduced or eliminated. A more systematic examination of this multifaceted vitamin and mineral compound in children with difficult-to-treat bipolar disorder is long past due.
Psychotherapeutic Approaches
The addition of family-focused therapy or a related family or group psychotherapeutic modality would be strongly recommended, particularly if it emphasized illness education, continued detailed mood and behavioral charting, improved family communication, cognitive-behavioral therapy and problem solving, and suggestions for not using highly punitive discipline measures.56–58 Such an expert psychotherapeutic practitioner or work in a group setting may be difficult to find in the community, but the ultimate result would most likely prove the effort worthwhile.47,59 Kessing et al60 demonstrated the superior outcome of patients with a first hospitalization for mania who were randomized to 2 years of specialty clinic compared to treatment as usual. Youngsters receiving the specialty care had fewer relapses over these 2 years, and the differences persisted and were enhanced over the next 4 years even when all patients were now receiving treatment as usual.
Mood Charting and Treatment Principles
We and many others believe that the mood charting in the Child Network or some other systematic longitudinal rating measures will be invaluable in helping to achieve an optimal outcome. We have found this to be the case for adults with bipolar disorder for whom the options and treatment sequences are somewhat better spelled out.31 In addition, charting symptoms in the Child Network will help identify prodromal symptoms and syndromes in those at high risk and thus help foster earlier and more effective intervention.61
However, in the realm of the pharmacotherapeutics of childhood bipolar disorders and related syndromes wherein the systematic treatment data are so sparse, the best guidance for optimal therapeutics is the response of the individual child herself or himself. Evidence gleaned from charting of an individual child of clear improvement or deterioration on a given treatment trumps all FDA approvals, academic or professional society treatment guidelines, or advocacy group recommendations. Having such a detailed record of symptom course and response to treatment will also facilitate the ability to get consultations and second opinions if the desired outcomes are not being achieved.
Submitted: February 27, 2017; accepted May 8, 2017.
Published online: August 31, 2017.
Potential conflicts of interest: In the past 12 months, Dr Findling receives or has received research support from and has acted as a consultant and/or served on a speaker’s bureau for Akili, Alcobra, American Academy of Child and Adolescent Psychiatry, American Psychiatric Press, Bracket, Epharma Solutions, Forest, Genentech, Ironshore, KemPharm, Lundbeck, Medgenics, National Institutes of Health, Neurim, PCORI, Pfizer, Physicians Postgraduate Press Inc, Roche, Sage, Shire, Sunovion, Supernus, Syneurx, Takeda, Teva, Tris, and Validus. Dr Post has been on the speakers bureau for AstraZeneca, Pamlabs, Sunovion, and Validus. Dr Rowe and Ms Kaplan report no conflicts of interest related to the subject of this article.
Funding/support: None.
REFERENCES
1. DelBello MP, Hanseman D, Adler CM, et al. Twelve-month outcome of adolescents with bipolar disorder following first hospitalization for a manic or mixed episode. Am J Psychiatry. 2007;164(4):582–590. PubMed doi:10.1176/ajp.2007.164.4.582
2. Axelson D, Goldstein B, Goldstein T, et al. Diagnostic precursors to bipolar disorder in offspring of parents with bipolar disorder: a longitudinal study. Am J Psychiatry. 2015;172(7):638–646. PubMed doi:10.1176/appi.ajp.2014.14010035
3. Birmaher B, Axelson D, Monk K, et al. Lifetime psychiatric disorders in school-aged offspring of parents with bipolar disorder: the Pittsburgh Bipolar Offspring Study. Arch Gen Psychiatry. 2009;66(3):287–296. PubMed doi:10.1001/archgenpsychiatry.2008.546
4. Wozniak J, Petty CR, Schreck M, et al. High level of persistence of pediatric bipolar-I disorder from childhood onto adolescent years: a four year prospective longitudinal follow-up study. J Psychiatr Res. 2011;45(10):1273–1282. PubMed doi:10.1016/j.jpsychires.2010.10.006
5. Geller B, Tillman R, Bolhofner K, et al. Child bipolar I disorder: prospective continuity with adult bipolar I disorder; characteristics of second and third episodes; predictors of 8-year outcome. Arch Gen Psychiatry. 2008;65(10):1125–1133. PubMed doi:10.1001/archpsyc.65.10.1125
6. Geller B, Tillman R, Bolhofner K, et al. Pharmacological and non-drug treatment of child bipolar I disorder during prospective eight-year follow-up. Bipolar Disord. 2010;12(2):164–171. PubMed doi:10.1111/j.1399-5618.2010.00791.x
7. Holtzman JN, Miller S, Hooshmand F, et al. Childhood-compared to adolescent-onset bipolar disorder has more statistically significant clinical correlates. J Affect Disord. 2015;179:114–120. PubMed doi:10.1016/j.jad.2015.03.019
8. Post RM, Leverich GS, Kupka RW, et al. Early-onset bipolar disorder and treatment delay are risk factors for poor outcome in adulthood. J Clin Psychiatry. 2010;71(7):864–872. PubMed doi:10.4088/JCP.08m04994yel
9. Suominen K, Mantere O, Valtonen H, et al. Early age at onset of bipolar disorder is associated with more severe clinical features but delayed treatment seeking. Bipolar Disord. 2007;9(7):698–705. PubMed doi:10.1111/j.1399-5618.2007.00388.x
10. Pavuluri MN, Birmaher B, Naylor MW. Pediatric bipolar disorder: a review of the past 10 years. J Am Acad Child Adolesc Psychiatry. 2005;44(9):846–871. PubMed doi:10.1097/01.chi.0000170554.23422.c1
11. Kowatch RA, Fristad M, Birmaher B, et al; Child Psychiatric Workgroup on Bipolar Disorder. Treatment Guidelines for Children and Adolescents With Bipolar Disorder. J Am Acad Child Adolesc Psychiatry. 2005;44(3):213–235. PubMed doi:10.1097/00004583-200503000-00006
12. Geller B, Luby JL, Joshi P, et al. A randomized controlled trial of risperidone, lithium, or divalproex sodium for initial treatment of bipolar I disorder, manic or mixed phase, in children and adolescents. Arch Gen Psychiatry. 2012;69(5):515–528. PubMed doi:10.1001/archgenpsychiatry.2011.1508
13. Duffy A, Alda M, Crawford L, et al. The early manifestations of bipolar disorder: a longitudinal prospective study of the offspring of bipolar parents. Bipolar Disord. 2007;9(8):828–838. PubMed doi:10.1111/j.1399-5618.2007.00421.x
14. Henin A, Biederman J, Mick E, et al. Psychopathology in the offspring of parents with bipolar disorder: a controlled study. Biol Psychiatry. 2005;58(7):554–561. PubMed doi:10.1016/j.biopsych.2005.06.010
15. Chang KD, Steiner H, Ketter TA. Psychiatric phenomenology of child and adolescent bipolar offspring. J Am Acad Child Adolesc Psychiatry. 2000;39(4):453–460. PubMed doi:10.1097/00004583-200004000-00014
16. Post RM, Altshuler L, Kupka R, et al. More pernicious course of bipolar disorder in the United States than in many European countries: implications for policy and treatment. J Affect Disord. 2014;160:27–33. PubMed doi:10.1016/j.jad.2014.02.006
17. Post RM, Altshuler LL, Kupka R, et al. More illness in offspring of bipolar patients from the US compared to Europe. J Affect Disord. 2016;191:180–186. PubMed doi:10.1016/j.jad.2015.11.038
18. Axelson D. Taking disruptive mood dysregulation disorder out for a test drive. Am J Psychiatry. 2013;170(2):136–139. PubMed doi:10.1176/appi.ajp.2012.12111434
19. Post RM. Treatment of bipolar depression: evolving recommendations. Psychiatr Clin North Am. 2016;39(1):11–33. PubMed doi:10.1016/j.psc.2015.09.001
20. Post RM, Wozniak J. Survey of expert treatment approaches for children with bipolar disorder-not otherwise specified and bipolar I presentations. Psych Annals. 2009;39(10):887–895. doi:10.3928/00485718-20090924-07
21. Leibenluft E. Flying almost blind. Am J Psychiatry. 2006;163(7):1129–1131. PubMed doi:10.1176/ajp.2006.163.7.1129
22. Findling RL, Kafantaris V, Pavuluri M, et al. Post-acute effectiveness of lithium in pediatric bipolar I disorder. J Child Adolesc Psychopharmacol. 2013;23(2):80–90. PubMed doi:10.1089/cap.2012.0063
23. Findling RL, Robb A, McNamara NK, et al. Lithium in the acute treatment of bipolar I disorder: a double-blind, placebo-controlled study. Pediatrics. 2015;136(5):885–894. PubMed doi:10.1542/peds.2015-0743
24. Findling RL, McNamara NK, Stansbrey R, et al. Combination lithium and divalproex sodium in pediatric bipolar symptom re-stabilization. J Am Acad Child Adolesc Psychiatry. 2006;45(2):142–148. PubMed doi:10.1097/01.chi.0000189135.05060.8a
25. Kowatch RA, Suppes T, Carmody TJ, et al. Effect size of lithium, divalproex sodium, and carbamazepine in children and adolescents with bipolar disorder. J Am Acad Child Adolesc Psychiatry. 2000;39(6):713–720. PubMed doi:10.1097/00004583-200006000-00009
26. Scheffer RE, Kowatch RA, Carmody T, et al. Randomized, placebo-controlled trial of mixed amphetamine salts for symptoms of comorbid ADHD in pediatric bipolar disorder after mood stabilization with divalproex sodium. Am J Psychiatry. 2005;162(1):58–64. PubMed doi:10.1176/appi.ajp.162.1.58
27. Spencer TJ, Greenbaum M, Ginsberg LD, et al. Safety and effectiveness of coadministration of guanfacine extended release and psychostimulants in children and adolescents with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2009;19(5):501–510. PubMed doi:10.1089/cap.2008.0152
28. Chang K, Saxena K, Howe M. An open-label study of lamotrigine adjunct or monotherapy for the treatment of adolescents with bipolar depression. J Am Acad Child Adolesc Psychiatry. 2006;45(3):298–304. PubMed doi:10.1097/01.chi.0000194566.86160.a3
29. Findling RL, Chang K, Robb A, et al. Adjunctive maintenance lamotrigine for pediatric bipolar I disorder: a placebo-controlled, randomized withdrawal study. J Am Acad Child Adolesc Psychiatry. 2015;54(12):1020–1031.e3. PubMed doi:10.1016/j.jaac.2015.09.017
30. Meador KJ, Baker GA, Browning N, et al; NEAD Study Group. Cognitive function at 3 years of age after fetal exposure to antiepileptic drugs. N Engl J Med. 2009;360(16):1597–1605. PubMed doi:10.1056/NEJMoa0803531
31. Post R, Leverich G. Treatment of Bipolar Illness: A Casebook for Clinicians and Patients. New York, NY: WW Norton and Company Inc; 2008.
32. Wagner KD, Kowatch RA, Emslie GJ, et al. A double-blind, randomized, placebo-controlled trial of oxcarbazepine in the treatment of bipolar disorder in children and adolescents. Am J Psychiatry. 2006;163(7):1179–1186. PubMed doi:10.1176/ajp.2006.163.7.1179
33. Joshi G, Wozniak J, Mick E, et al. A prospective open-label trial of extended-release carbamazepine monotherapy in children with bipolar disorder. J Child Adolesc Psychopharmacol. 2010;20(1):7–14. PubMed doi:10.1089/cap.2008.0162
34. Findling RL, Ginsberg LD. The safety and effectiveness of open-label extended-release carbamazepine in the treatment of children and adolescents with bipolar I disorder suffering from a manic or mixed episode. Neuropsychiatr Dis Treat. 2014;10:1589–1597. PubMed doi:10.2147/NDT.S68951
35. DelBello M, Goldman R, Deng L, et al. Efficacy and safety of lurasidone in children and adolescent patients with bipolar depression. Poster #14. Presented at the International Society for Bipolar Disorders; Arlington, Virginia; May 5, 2017.
36. Findling RL, Cavuş I, Pappadopulos E, et al. Efficacy, long-term safety, and tolerability of ziprasidone in children and adolescents with bipolar disorder. J Child Adolesc Psychopharmacol. 2013;23(8):545–557. PubMed doi:10.1089/cap.2012.0029
37. Patkar A, Gilmer W, Pae CU, et al. A 6 week randomized double-blind placebo-controlled trial of ziprasidone for the acute depressive mixed state. PLoS One. 2012;7(4):e34757. PubMed doi:10.1371/journal.pone.0034757
38. Findling RL, Pathak S, Earley WR, et al. Efficacy and safety of extended-release quetiapine fumarate in youth with bipolar depression: an 8 week, double-blind, placebo-controlled trial. J Child Adolesc Psychopharmacol. 2014;24(6):325–335. PubMed doi:10.1089/cap.2013.0105
39. Post RM, Kowatch RA. The health care crisis of childhood-onset bipolar illness: some recommendations for its amelioration. J Clin Psychiatry. 2006;67(1):115–125. PubMed doi:10.4088/JCP.v67n0118
40. Lao KS, He Y, Wong IC, et al. Tolerability and safety profile of cariprazine in treating psychotic disorders, bipolar disorder and major depressive disorder: a systematic review with meta-analysis of randomized controlled trials. CNS Drugs. 2016;30(11):1043–1054. PubMed doi:10.1007/s40263-016-0382-z
41. Post RM, Kalivas P. Bipolar disorder and substance misuse: pathological and therapeutic implications of their comorbidity and cross-sensitization. Br J Psychiatry. 2013;202(3):172–176. PubMed doi:10.1192/bjp.bp.112.116855
42. Kalivas BC, Kalivas PW. Corticostriatal circuitry in regulating diseases characterized by intrusive thinking. Dialogues Clin Neurosci. 2016;18(1):65–76. PubMed
43. Berk M, Malhi GS, Gray LJ, et al. The promise of N-acetylcysteine in neuropsychiatry. Trends Pharmacol Sci. 2013;34(3):167–177. PubMed doi:10.1016/j.tips.2013.01.001
44. Hardan AY, Fung LK, Libove RA, et al. A randomized controlled pilot trial of oral N-acetylcysteine in children with autism. Biol Psychiatry. 2012;71(11):956–961. PubMed doi:10.1016/j.biopsych.2012.01.014
45. Nikoo M, Radnia H, Farokhnia M, et al. N-acetylcysteine as an adjunctive therapy to risperidone for treatment of irritability in autism: a randomized, double-blind, placebo-controlled clinical trial of efficacy and safety. Clin Neuropharmacol. 2015;38(1):11–17. PubMed doi:10.1097/WNF.0000000000000063
46. Ghanizadeh A, Moghimi-Sarani E. A randomized double blind placebo controlled clinical trial of N-acetylcysteine added to risperidone for treating autistic disorders. BMC Psychiatry. 2013;13:196. PubMed doi:10.1186/1471-244X-13-196
47. Fristad MA. Evidence-based psychotherapies and nutritional interventions for children with bipolar spectrum disorders and their families. J Clin Psychiatry. 2016;77(suppl E1):e4. PubMed
48. Mischoulon D, Freeman MP. Omega-3 fatty acids in psychiatry. Psychiatr Clin North Am. 2013;36(1):15–23. PubMed doi:10.1016/j.psc.2012.12.002
49. Young AS, Arnold LE, Wolfson HL, et al. Psychoeducational psychotherapy and omega-3 supplementation improve co-occurring behavioral problems in youth with depression: results from a pilot RCT. J Abnorm Child Psychol. 2017;45(5):1025–1037. PubMed doi:10.1007/s10802-016-0203-3
50. Gracious BL, Finucane TL, Friedman-Campbell M, et al. Vitamin D deficiency and psychotic features in mentally ill adolescents: a cross-sectional study. BMC Psychiatry. 2012;12:38. PubMed doi:10.1186/1471-244X-12-38
51. Khoraminya N, Tehrani-Doost M, Jazayeri S, et al. Therapeutic effects of vitamin D as adjunctive therapy to fluoxetine in patients with major depressive disorder. Aust N Z J Psychiatry. 2013;47(3):271–275. PubMed doi:10.1177/0004867412465022
52. Sikoglu EM, Navarro AA, Starr D, et al. Vitamin D3 supplemental treatment for mania in youth with bipolar spectrum disorders. J Child Adolesc Psychopharmacol. 2015;25(5):415–424. PubMed doi:10.1089/cap.2014.0110
53. Papakostas GI, Shelton RC, Zajecka JM, et al. L-methylfolate as adjunctive therapy for SSRI-resistant major depression: results of two randomized, double-blind, parallel-sequential trials. Am J Psychiatry. 2012;169(12):1267–1274. PubMed doi:10.1176/appi.ajp.2012.11071114
54. Frazier EA, Gracious B, Arnold LE, et al. Nutritional and safety outcomes from an open-label micronutrient intervention for pediatric bipolar spectrum disorders. J Child Adolesc Psychopharmacol. 2013;23(8):558–567. PubMed
55. Simkin DR, Popper CW. Alternative and complementary therapies for psychiatric disorders. Part 2. Child Adolesc Psychiatr Clin N Am. 2014;23(3):ix–xii. PubMed doi:10.1016/j.chc.2014.04.002
56. Miklowitz DJ, Schneck CD, Singh MK, et al. Early intervention for symptomatic youth at risk for bipolar disorder: a randomized trial of family-focused therapy. J Am Acad Child Adolesc Psychiatry. 2013;52(2):121–131. PubMed doi:10.1016/j.jaac.2012.10.007
57. Fristad MA, Verducci JS, Walters K, et al. Impact of multifamily psychoeducational psychotherapy in treating children aged 8 to 12 years with mood disorders. Arch Gen Psychiatry. 2009;66(9):1013–1021. PubMed doi:10.1001/archgenpsychiatry.2009.112
58. Scott J, Colom F. Psychosocial treatments for bipolar disorders. Psychiatr Clin North Am. 2005;28(2):371–384. PubMed doi:10.1016/j.psc.2005.01.002
59. Miklowitz DJ. Evidence-based family interventions for adolescents and young adults with bipolar disorder. J Clin Psychiatry. 2016;77(suppl E1):e5. PubMed
60. Kessing LV, Hansen HV, Hvenegaard A, et al; Early Intervention Affective Disorders (EIA) Trial Group. Treatment in a specialized out-patient mood disorder clinic v. standard out-patient treatment in the early course of bipolar disorder: randomized clinical trial. Br J Psychiatry. 2013;202(3):212–219. PubMed doi:10.1192/bjp.bp.112.113548
61. Post RM, Chang K, Frye MA. Paradigm shift: preliminary clinical categorization of ultrahigh risk for childhood bipolar disorder to facilitate studies on prevention. J Clin Psychiatry. 2013;74(2):167–169. PubMed doi:10.4088/JCP.12com08136
This PDF is free for all visitors!


