Objective: To evaluate the efficacy and tolerability of pharmacotherapy in pediatric anxiety disorders using network meta-analysis.
Data Sources: PubMed, Cochrane Database, Web of Science, PsycNET, and ClinicalTrials.gov were searched for double-blind, controlled pharmacotherapy trials in youth with anxiety disorders from 1966 to September 2017.
Data Selection: All double-blind, placebo-controlled trials of pharmacotherapy in the treatment of pediatric patients with generalized, social, and/or separation anxiety disorders were included.
Data Extraction: We extracted demographic, symptom severity, global improvement, discontinuation, and suicidality data. Risk of bias was assessed with the Cochrane risk-of-bias tool, and a network meta-analysis comparing the efficacy and tolerability of medications and medication classes was performed using the gemtc package (R).
Results: We identified 20 citations (22 RCTs, 24 treatment arms) with 2,623 patients. Selective serotonin reuptake inhibitors (SSRIs) were the only class that was superior in reducing anxiety (standardized mean difference: 5.2; credible interval [CrI]: [2.8 to 8.8]) and in likelihood of treatment response compared to placebo (odds ratio [OR]: 4.6; CrI: [3.1 to 7.5]). Serotonin-norepinephrine reuptake inhibitor (SNRI) and α2 agonist treatment were associated with more frequent treatment response compared to placebo. The likelihood of treatment response was greater for SSRIs compared to SNRIs (OR: 1.9; CrI: [1.1 to 3.5]). All-cause discontinuation and treatment-emergent suicidality significantly differed among medications but not medication class.
Conclusions: Although multiple medications reduce anxiety in children and adolescents, treatment response, tolerability, and treatment-emergent suicidality differ among these medications and medication classes. Determining whether efficacy and tolerability differences represent true differences (or reflect differences in trial design) requires additional head-to-head medication trials and—to exclude the impact of missing treatment interventions—requires trials of medications that successfully treat anxiety in adults but that have not been evaluated in youth.

CME Background
Articles are selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME articles on a variety of topics throughout each volume. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the article, correctly answer the questions in the Posttest, and complete the Evaluation. A $10 processing fee will apply.
CME Objective
After studying this article, you should be able to:
‘ ¢ Consider data on differences in efficacy, tolerability, and treatment-emergent suicidality when selecting pharmacotherapy for anxiety disorders in pediatric patients
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1 hour of Category I credit for completing this program.
Release, Expiration, and Review Dates
This educational activity was published in January 2019 and is eligible for AMA PRA Category 1 Creditâ„¢ through February 28, 2021. The latest review of this material was December 2018.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Marlene P. Freeman, MD, Editor in Chief, has received research funding from JayMac and Sage; has been a member of the advisory boards for Otsuka, Alkermes, and Sunovion; has been a member of the Independent Data Safety and Monitoring Committee for Janssen; and, as a Massachusetts General Hospital (MGH) employee, works with the MGH National Pregnancy Registry, which is sponsored by Teva, Alkermes, Otsuka, Actavis, and Sunovion, and works with the MGH Clinical Trials Network and Institute, which receives research funding from multiple pharmaceutical companies and the National Institute of Mental Health. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears at the end of the article.
ABSTRACT
Objective: To evaluate the efficacy and tolerability of pharmacotherapy in pediatric anxiety disorders using network meta-analysis.
Data Sources: PubMed, Cochrane Database, Web of Science, PsycNET, and ClinicalTrials.gov were searched for double-blind, controlled pharmacotherapy trials in youth with anxiety disorders from 1966 to September 2017.
Data Selection: All double-blind, placebo-controlled trials of pharmacotherapy in the treatment of pediatric patients with generalized, social, and/or separation anxiety disorders were included.
Data Extraction: We extracted demographic, symptom severity, global improvement, discontinuation, and suicidality data. Risk of bias was assessed with the Cochrane risk-of-bias tool, and a network meta-analysis comparing the efficacy and tolerability of medications and medication classes was performed using the gemtc package (R).
Results: We identified 20 citations (22 RCTs, 24 treatment arms) with 2,623 patients. Selective serotonin reuptake inhibitors (SSRIs) were the only class that was superior in reducing anxiety (standardized mean difference: 5.2; credible interval [CrI]: [2.8 to 8.8]) and in likelihood of treatment response compared to placebo (odds ratio [OR]: 4.6; CrI: [3.1 to 7.5]). Serotonin-norepinephrine reuptake inhibitor (SNRI) and α2 agonist treatment were associated with more frequent treatment response compared to placebo. The likelihood of treatment response was greater for SSRIs compared to SNRIs (OR: 1.9; CrI: [1.1 to 3.5]). All-cause discontinuation and treatment-emergent suicidality significantly differed among medications but not medication class.
Conclusions: Although multiple medications reduce anxiety in children and adolescents, treatment response, tolerability, and treatment-emergent suicidality differ among these medications and medication classes. Determining whether efficacy and tolerability differences represent true differences (or reflect differences in trial design) requires additional head-to-head medication trials and—to exclude the impact of missing treatment interventions—requires trials of medications that successfully treat anxiety in adults but that have not been evaluated in youth.
J Clin Psychiatry 2019;80(1):17r12064
To cite: Dobson ET, Bloch MH, Strawn JR. Efficacy and tolerability of pharmacotherapy for pediatric anxiety disorders: a network meta-analysis. J Clin Psychiatry. 2019;80(1):17r12064.
To share: https://doi.org/10.4088/JCP.17r12064
© Copyright 2019 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry and Behavioral Neuroscience, University of Cincinnati, College of Medicine, Cincinnati, Ohio
bDepartment of Psychiatry and Behavioral Sciences, Medical University of South Carolina, Charleston, South Carolina
cYale Child Study Center, New Haven, Connecticut
dDivision of Child and Adolescent Psychiatry, Department of Pediatrics, Cincinnati Children’s Hospital Medical Center, Cincinnati, Ohio
*Corresponding author: Jeffrey R. Strawn, MD, University of Cincinnati, Department of Psychiatry, Box 670559, Cincinnati, OH 45267-0559 ([email protected]).
Anxiety disorders are prevalent and impairing in children and adolescents1 but frequently improve with psychopharmacologic treatment.2 Nearly two dozen randomized controlled trials (RCTs) have evaluated the efficacy of antidepressants, benzodiazepines, α2 agonists, and other classes of medication for the treatment of pediatric anxiety disorders, and 2 studies directly compared more than 1 psychopharmacologic treatment.3,4 Guidelines from the American Academy of Child and Adolescent Psychiatry recommend selective serotonin reuptake inhibitors (SSRIs) and selective serotonin-norepinephrine reuptake inhibitors (SNRIs)5 but note that other classes of medication may effectively reduce anxiety. However, clinicians treating anxious youth have limited comparative efficacy and tolerability data to guide treatment. In fact, pediatricians—who are frequently the first providers initiating pharmacotherapy for anxious youth—report that their personal preference and experience with specific medications drive medication choice,6 and qualitative studies suggest that these first-line providers feel that there are insufficient data regarding the differential tolerability of medications in anxious youth.6
Meta-analyses provide a platform for examining the efficacy and tolerability of pharmacotherapy. In fact, 4 meta-analyses suggest that antidepressants are effective in treating pediatric patients with anxiety disorders.7,8,10,11 However, these meta-analyses provide limited data as to which medications are most effective and which medications are most tolerable and, with 1 exception,10 only examine antidepressants. A recent meta-analysis comparing antidepressant efficacy and tolerability in pediatric patients with depressive and anxiety disorders, as well as obsessive-compulsive and posttraumatic stress disorders,11 reports that antidepressants are particularly effective in anxiety disorders compared to the other 3 disorders but does not compare specific medications with one another. In a second meta-analysis, Wang et al10 explore the efficacy and safety of pharmacotherapy and cognitive behavioral therapy in childhood anxiety disorders, although differences in medication tolerability and treatment-emergent suicidality were not included. Additionally, data from 3 trials, involving more than 600 patients (one quarter of available RCT data in pediatric anxiety disorders) and 2 previously unstudied classes of medication (serotonin-1A [5-hydroxytryptamine, 5-HT1A] agonists and α2 agonists),12,13 were not available at the time these prior meta-analyses of pharmacotherapy in anxious youth were completed.10,12,13
With these considerations in mind, we sought to answer several important clinical questions using network meta-analysis: (1) Are there differences in efficacy (as measured by treatment response and anxiety symptom improvement) between medication classes and medications commonly prescribed for pediatric anxiety disorders? (2) Are there differences in tolerability (as measured by all-cause discontinuation and discontinuation due to adverse event) between medication classes and medications commonly prescribed for pediatric anxiety disorders? and (3) Are there differences in treatment-emergent suicidality between medication classes and medications commonly prescribed for pediatric anxiety disorders? Based on the magnitude of the efficacy signals detected in primary studies, we hypothesized that SSRIs and SNRIs would be more effective compared to 5-HT1A agonists and benzodiazepines and that benzodiazepines would be associated with poorer tolerability compared to antidepressants. Finally, we hypothesized, based on prior analyses in patients with depressive disorders,14,15 that antidepressants and specifically paroxetine and venlafaxine would be associated with greater treatment-emergent suicidality compared to other classes and medications, respectively.

- In anxious youth, treatment response was more likely with selective serotonin reuptake inhibitors (SSRIs) compared to serotonin-norepinephrine reuptake inhibitors (SNRIs); however, SSRIs were associated with a greater likelihood of discontinuation secondary to adverse events compared to SNRIs.
- In terms of all-cause discontinuation, SSRIs were the most tolerable class of medications, while tricyclic antidepressants were the least tolerable.
- Treatment-emergent suicidality did not differ significantly among classes of medications but did differ by specific medication.
METHODS
Identification and Selection of Studies
A literature review of the National Library of Medicine (PubMed), the Cochrane Database, Web of Science, PsycINFO, and Embase, as well as the federal clinical trials registry (ClinicalTrials.gov), from 1966 to October 2017 was completed using the following search terms: “(pediatric OR child or children or adolescent OR youth) AND (anxiety or “generalized anxiety disorder” or “overanxious disorder” or “separation anxiety disorder” or “social phobia” or “social anxiety disorder” or “school avoidance” or SAD or GAD) AND (SSRI or SNRI or TCA or benzodiazepine or NRI or “serotonin norepinephrine reuptake inhibitor” or “selective serotonin reuptake inhibitor” or anxiolytic or pregabalin or gabapentin or antidepressant or fluoxetine or venlafaxine or desvenlafaxine or duloxetine or vortioxetine or vilazodone or buspirone or sertraline or citalopram or escitalopram or fluvoxamine or levomilnacipran or chlordiazepoxide or diazepam or lorazepam or oxazepam or temazepam or clonazepam or pregabalin or alprazolam or triazolam or midazolam or atomoxetine or guanfacine or clomipramine or desipramine or doxepin or nortriptyline or amoxapine or mirtazapine or paroxetine or tricyclic or imipramine).” The references of all eligible trials and review articles were searched for additional clinical trials. Importantly, from a missing treatment intervention standpoint, we did not include some treatments that are used clinically but for which no randomized clinical trials exist, including hydroxyzine, bupropion, and second-generation antipsychotics.16
All prospective, randomized, placebo-controlled clinical trials that evaluated a specific pharmacotherapy intervention in the treatment of anxiety disorders in patients < 18 years of age and used a validated rating scale to measure anxiety symptom severity were selected for further analysis. Trials involving concurrent psychotherapy were excluded, as were those that were unavailable in English.
Data Extraction
Study data and characteristics (eg, year of trial publication, duration of trial, treatment response and anxiety symptom improvement, treatment-emergent adverse events, dropout rates, and rates of treatment-emergent suicidality) were extracted from primary articles and/or clinical study reports into a database (Microsoft Excel). Data were extracted from week 8 or the time point closest to week 8 favoring the study’s primary end point. When full data (response rate in the case of treatment response outcomes or mean change from baseline and standard deviation in the case of anxiety symptom improvement) were not available at week 8 or could not be reasonably calculated, the value closest to the original end point was used; data were digitally extracted using WebPlotDigitizer when presented graphically.17
For treatment response, the following measures were specified a priori (in order of preference): (1) Clinical Global Impression-Improvement (CGI-I) scores ≤ 2 (indicating much improved or very much improved), (2) 50% improvement on Pediatric Anxiety Rating Scale (PARS) severity, and (3) global improvement or global assessment of functioning. When CGI-I summary statistics were reported, a large pseudosample was generated based on a normal distribution with results binned into discrete CGI-I values (R, version 3.4.2, function: rnorm). For anxiety symptom severity, the following were utilized (in order of preference): (1) PARS,18 (2) Hamilton Anxiety Rating Scale (HARS),19 (3) Multidimensional Anxiety Rating Scale for Children,20 (4) Columbia Kiddie Schedule for Affective Disorders and Schizophrenia—Generalized Anxiety Disorder scale, (5) Liebowitz Social Anxiety Scale for Children and Adolescents, (6) Social Phobia and Anxiety Inventory for Children, and (7) Revised Children’s Manifest Anxiety Scale. When only baseline and endpoint data were available, standard deviation of the change from baseline was imputed using a correlation coefficient.21 Our hierarchy of anxiety symptom severity rating scales was based on (1) comparability and psychometric properties of rating scales, (2) appropriateness of use in a pediatric population, (3) consistency of use across trials, and (4) inclusion of somatic symptoms that may be obfuscated by the emergence of treatment side effects.
Network Meta-Analysis
Using a Bayesian approach, we performed a random-effects network meta-analysis (R, version 3.4.2, package gemtc, version 0.8-2)22 to compare treatment response and anxiety symptom improvement, all-cause discontinuation, discontinuation due to adverse event, and treatment-emergent suicidality for medication and medication class. Pairwise comparisons from each model were made using relative effect tables with treatment response expressed as log of odds ratio (logOR) and anxiety symptom improvement expressed as mean difference with 95% credible intervals (CrIs). A node-splitting approach in which differences between actual trial data (direct) and inferred data (with head-to-head trials excluded) was employed to evaluate the consistency of direct and indirect comparisons.23
To assess the likelihood that a given treatment is the best, second best, and so on within a network, rank probabilities were determined and converted to cumulative rank probabilities from which surface under the cumulative ranking (SUCRA) curves were generated. Then, each treatment model was described hierarchically using SUCRA values.24
Publication bias was assessed using funnel plots for each efficacy and tolerability measure and Egger test.25 Study quality was rated using the Cochrane Risk of Bias Assessment Tool.21 Heterogeneity was assessed in dichotomous direct comparisons with Cochran Q and I2 when 2 or more direct comparisons between classes were available. We did not test for heterogeneity in our analysis of anxiety symptom improvement given the large heterogeneity in symptom severity reporting methods. Finally, we checked the sensitivity of our model by rerunning our treatment response analysis without the treatment with the highest scoring SUCRA value and then again without the lowest scoring SUCRA value.
RESULTS
Included Studies
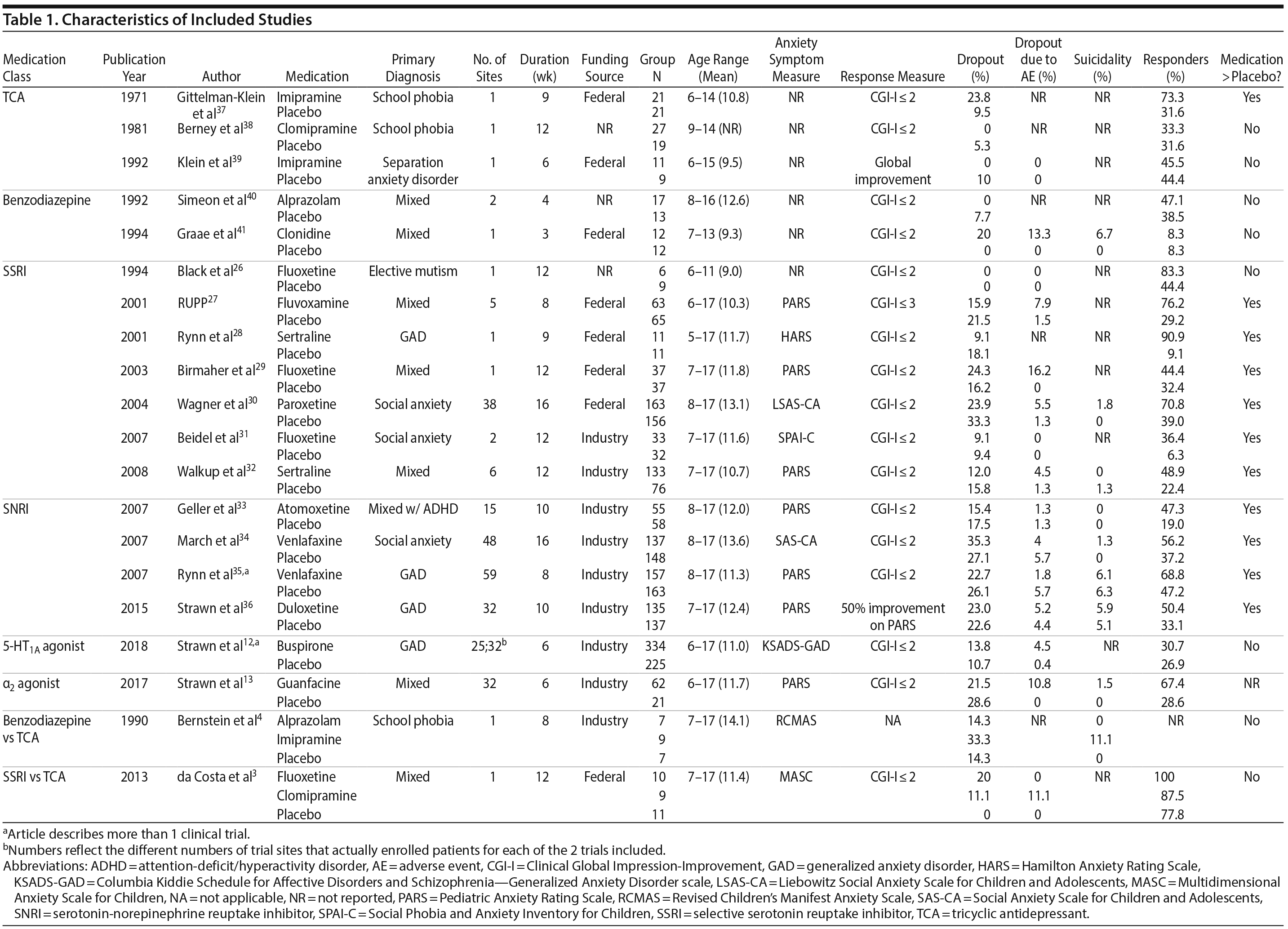
Our search identified 20 published articles including data from 22 randomized, double-blind, placebo-controlled trials with 2,623 patients.3,4,12,13,26-41 Among these were 24 active treatment arms with 8 SSRI arms, 5 SNRI arms, 5 tricyclic antidepressant (TCA) arms, 3 benzodiazepine arms, 2 5-HT1A agonist arms, and 1 α2 agonist arm. Two studies involved more than 2 treatment arms. Primary diagnoses were children with mixed anxiety disorders in 7 RCTs, generalized anxiety disorder (GAD) in 6, social anxiety disorder in 3, school phobia in 3, separation anxiety disorder in 1, elective mutism in 1, and mixed anxiety disorders with comorbid attention-deficit/hyperactivity disorder (ADHD) in 1. In studies involving school phobia and elective mutism, the majority of patients were diagnosed with an anxiety disorder (GAD, social anxiety disorder, or separation anxiety disorder); in fact, in the one trial of youth with elective mutism, 100% of participants met DSM-III criteria for social phobia.26 Eleven RCTs were funded by industry, 8 were federally funded, and 3 did not disclose funding source. Trials ranged in duration from 3 to 16 weeks, with an average of 9 weeks. The average age was 11.6 years (range, 5-17 years), and additional details are shown in Table 1. A network plot of qualifying studies has been included in Figure 1.
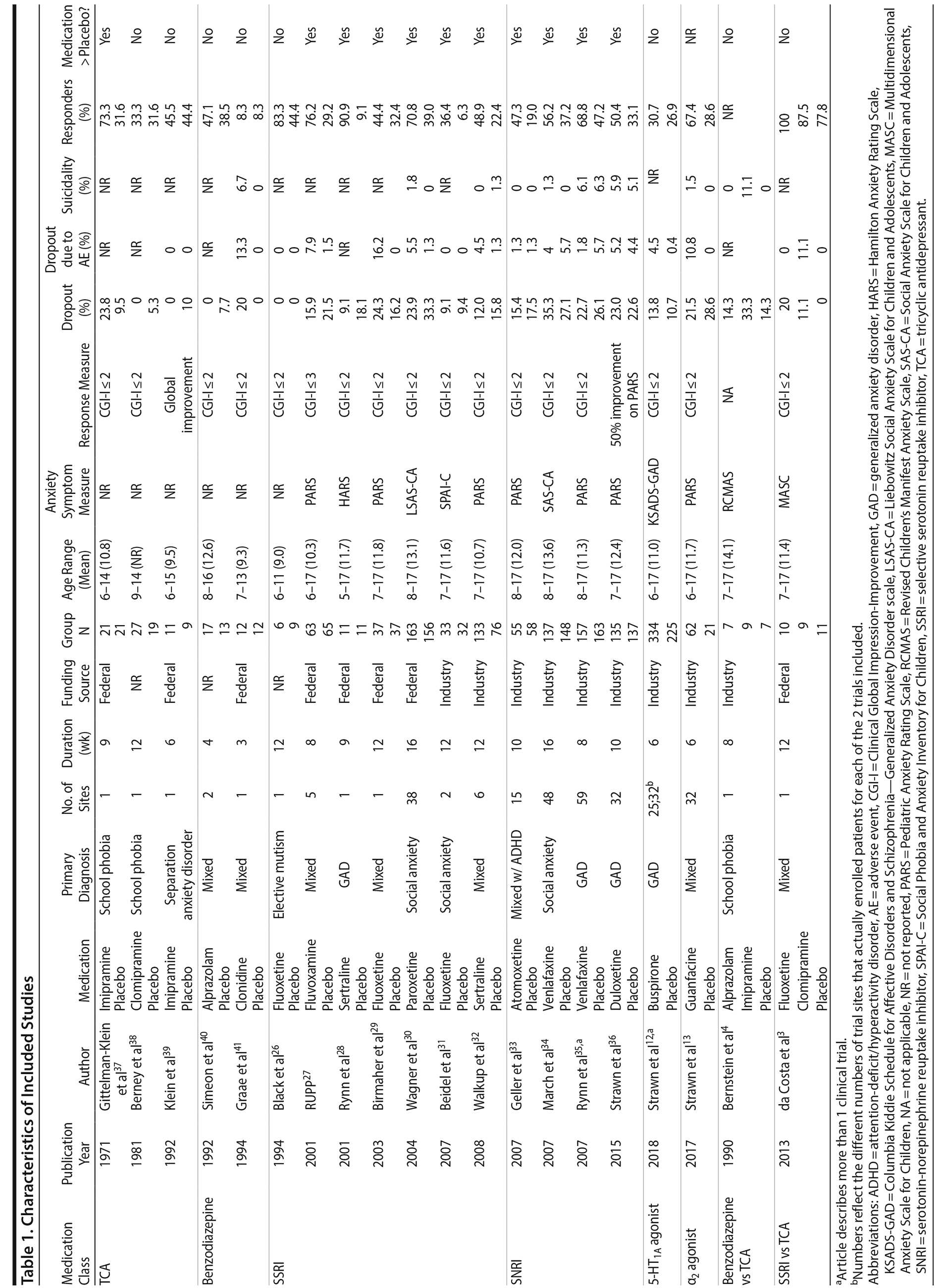
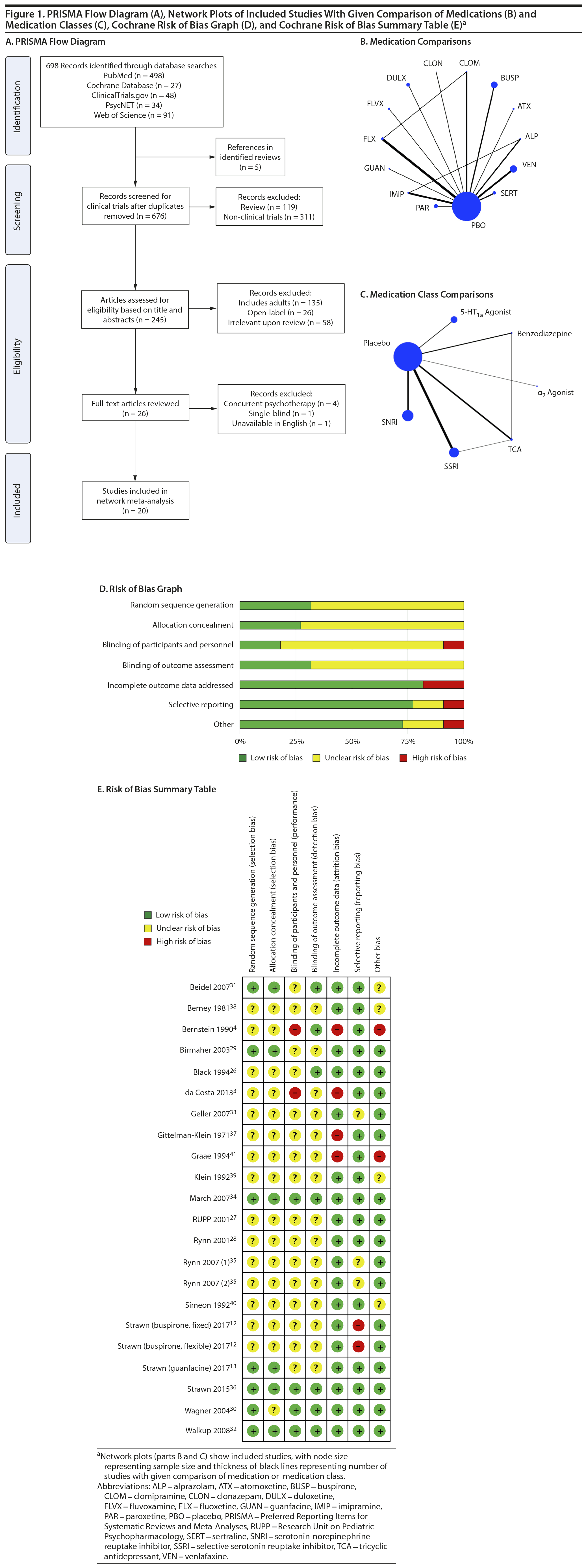
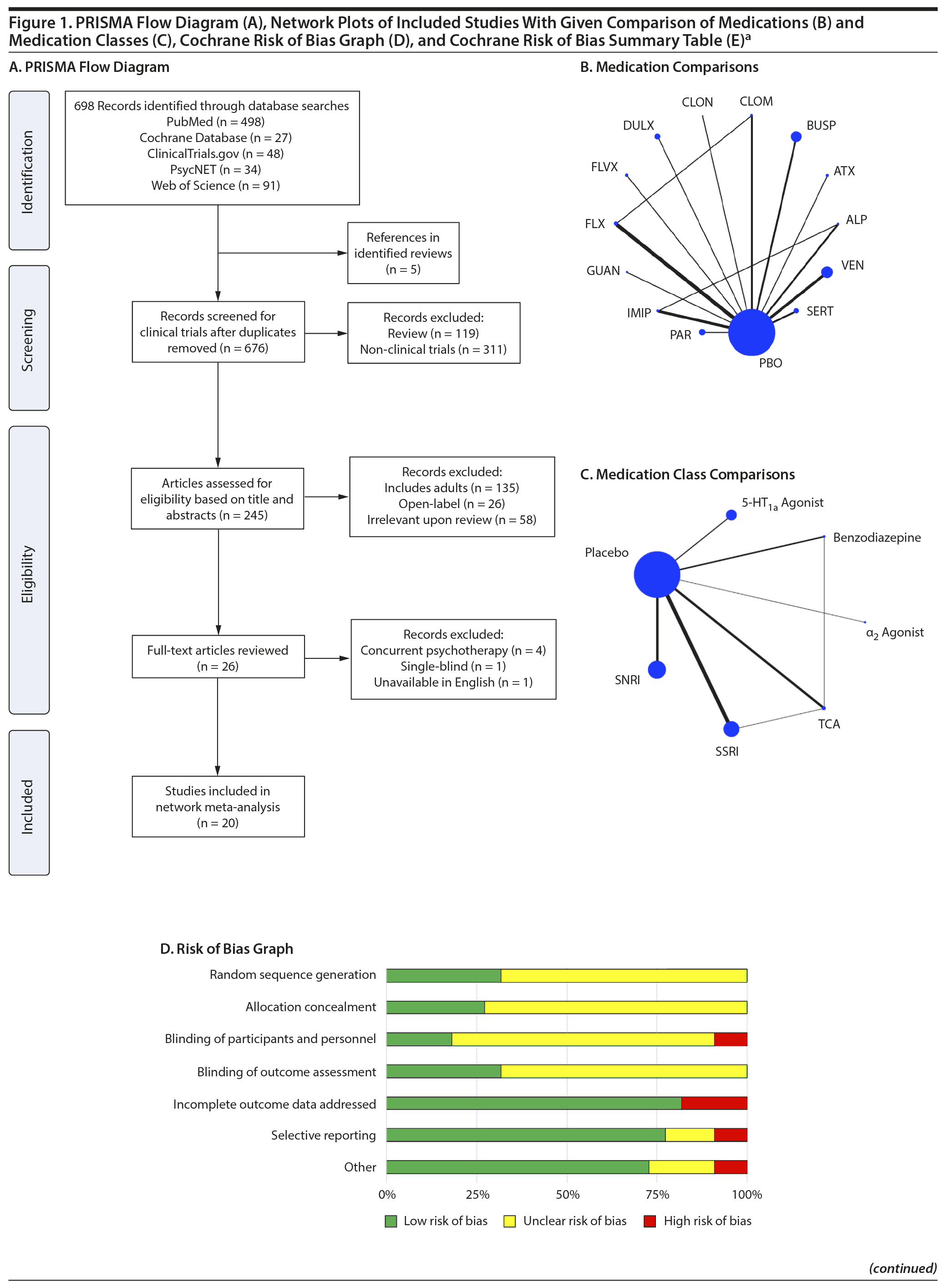

Risk of bias was generally low or unclear with few exceptions. Components of studies at high risk of bias included a study with crossover design in which dropout in the active arm precluded treatment in the placebo arm,41 studies in which patients had limited interaction with personnel not blinded to treatment (ie, a non-assessing nurse monitoring for side effects),3,4 large difference between groups in dropout rate or reasons for dropout (ie, lack of efficacy or adverse events),3,4,37,41 inconsistency in data reporting (eg, a study identified percent responders as primary outcome but only reported mean CGI-I scores),12 and differences in baseline anxiety severity or presence of comorbidities.4 With the exception of the study with inconsistent data reporting,12 all instances of high bias risk occurred in small studies (N < 40). The most common reasons for unclear risk of bias in studies were statements regarding the study being “double-blind” or “randomized” without specifying how subjects were randomized or who specifically was blinded, representing unclear risk of bias in random sequence generation, allocation concealment, blinding of participants and personnel, and blinding of outcome assessment.
Efficacy
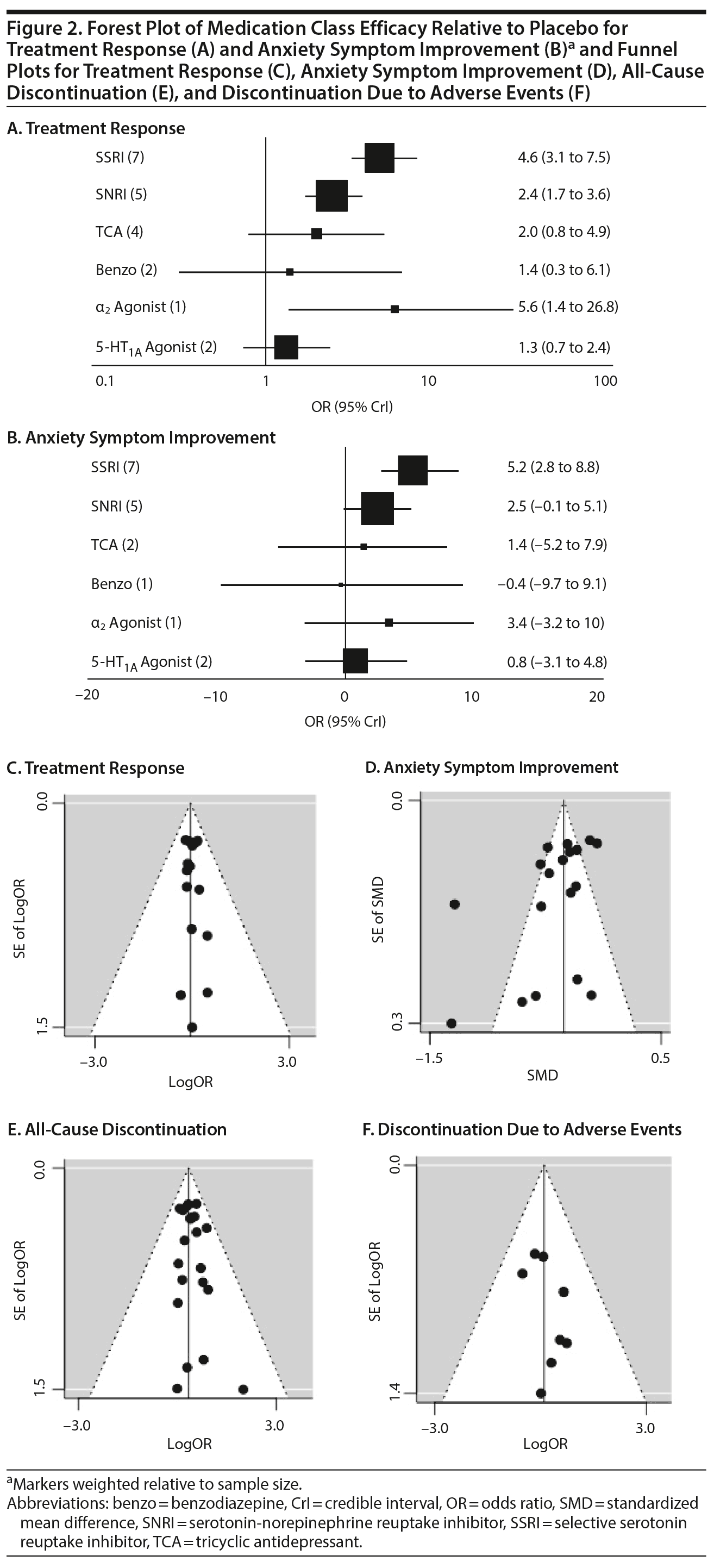
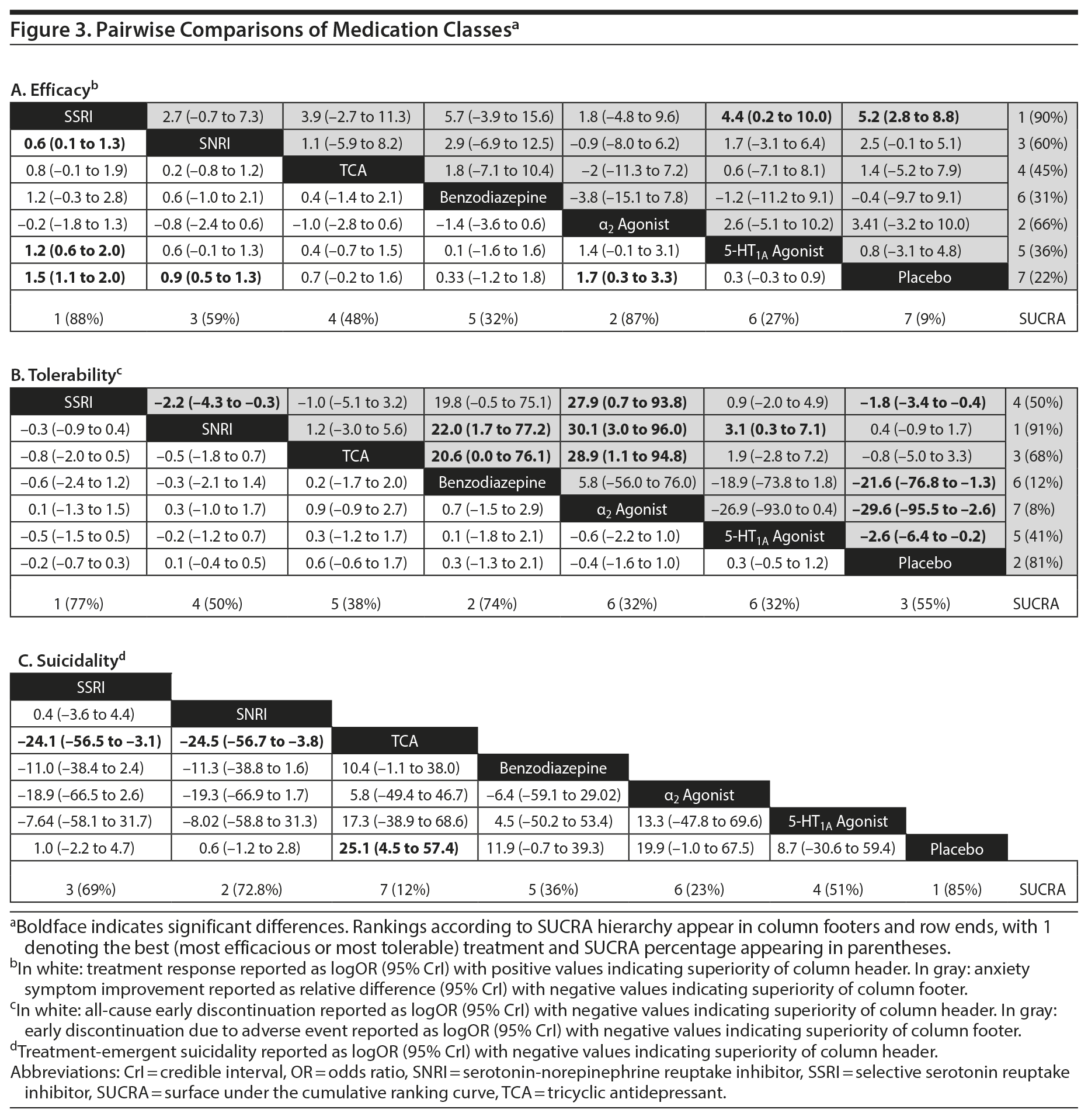
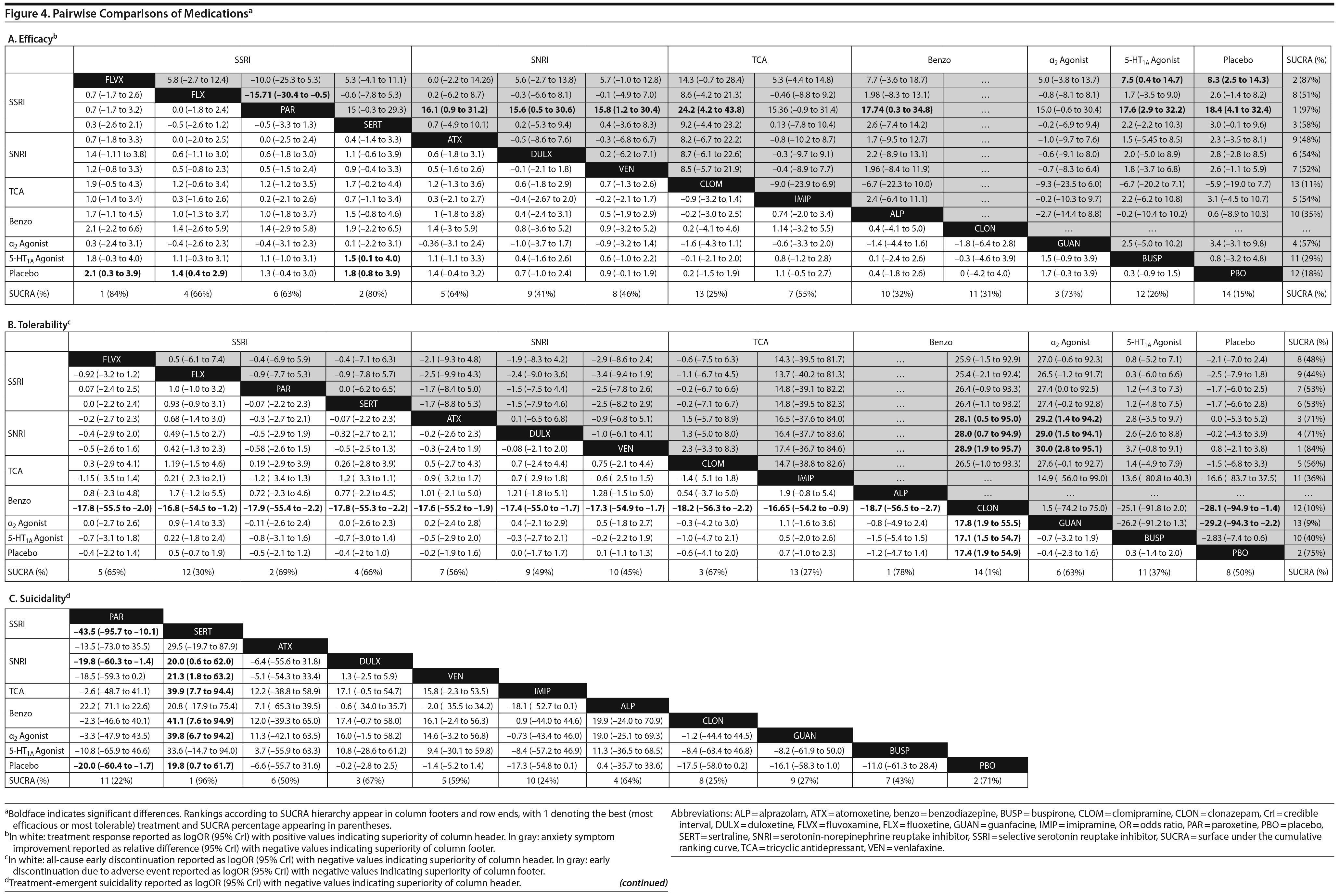
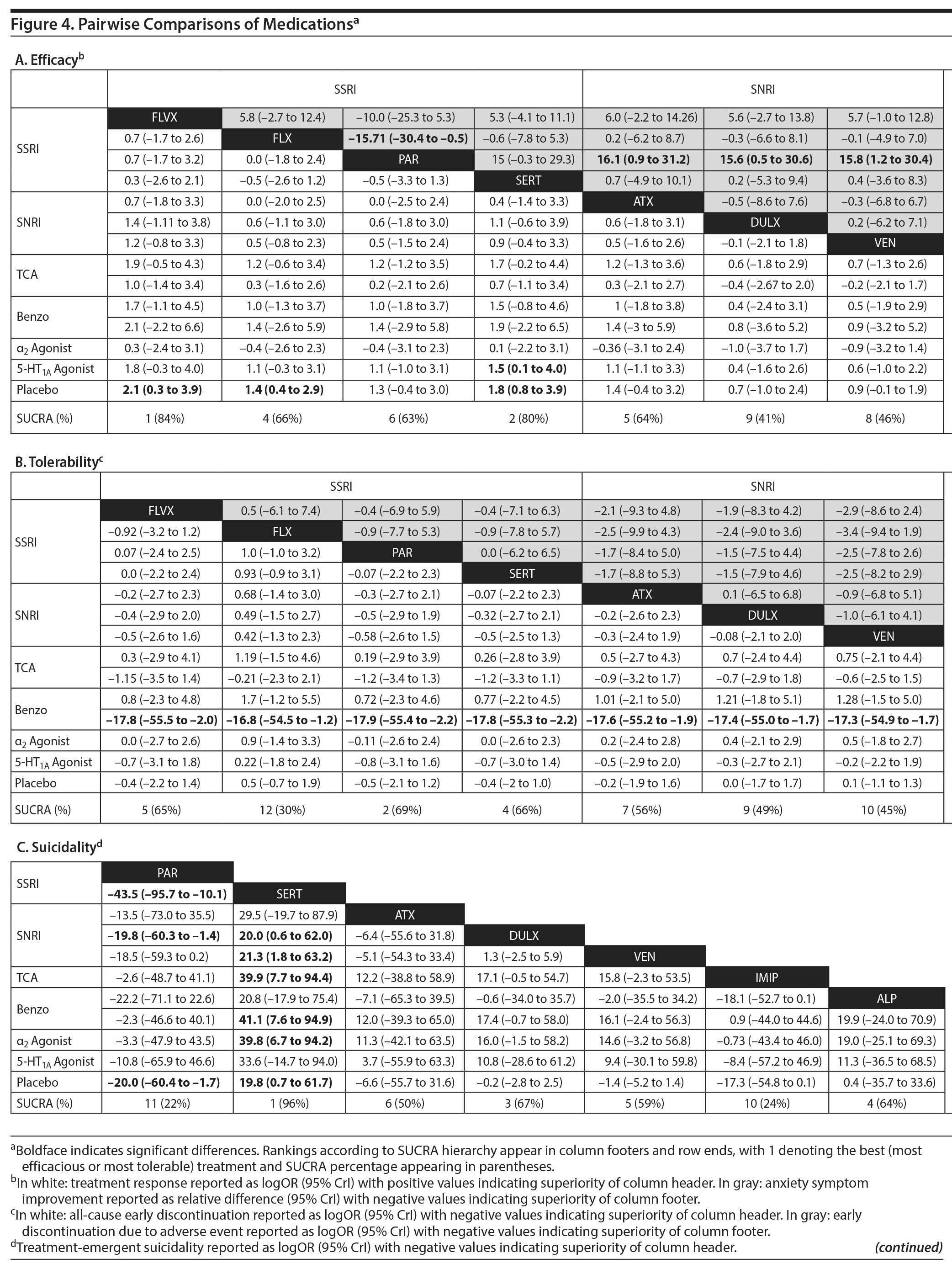
Treatment response. In pairwise comparisons by medication class (Figure 2A, Figure 3A), 3 were superior to placebo: α2 agonists (logOR: 1.7; 95% CrI: [0.3 to 3.3]), SSRIs (logOR: 1.5; 95% CrI: [1.1 to 2.0]), and SNRIs (logOR: 0.9; 95% CrI: [0.5 to 1.3]). SSRIs were superior to 5-HT1A agonists (logOR: 1.2; 95% CrI: [0.6 to 2.0]) and SNRIs (logOR: 0.6; 95% CrI: [0.1 to 1.3]). In terms of treatment response, SSRIs were the most effective class and 5-HT1A agonists were the least.
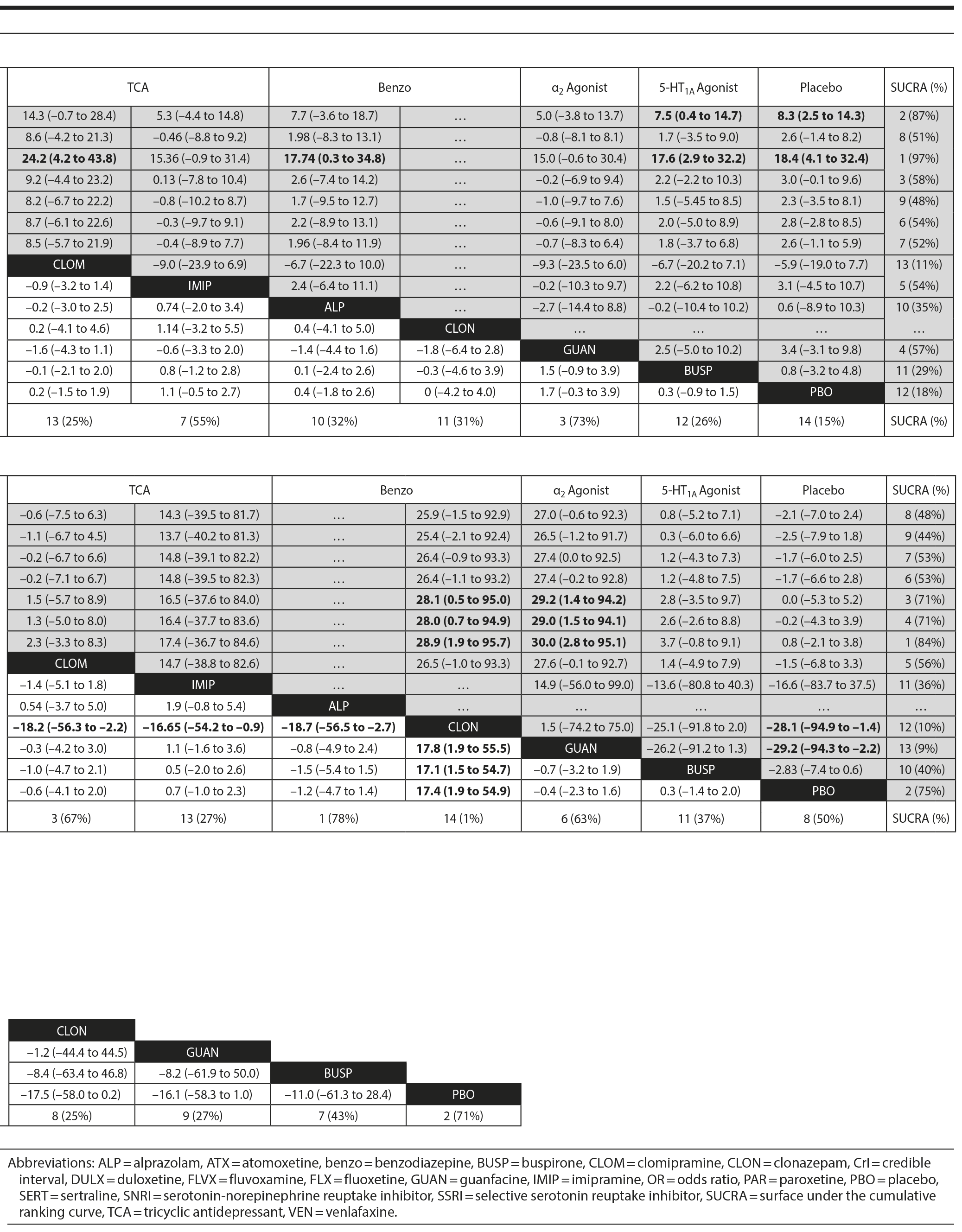
Pairwise comparisons by medication (Figure 4A) revealed sertraline to be superior to buspirone (logOR: 1.5; 95% CrI: [0.1 to 4.0]). Three treatments were superior to placebo: fluvoxamine (logOR: 2.1; 95% CrI: [0.31 to 3.89]), sertraline (logOR: 1.8, 95% CrI: [0.8 to 3.9]), and fluoxetine (logOR: 1.4, 95% CrI: [0.4 to 2.9]). In terms of treatment response, the most effective active treatment was fluvoxamine and the least effective was clomipramine.
A funnel plot of these studies appeared symmetric, and the Egger test did not indicate publication bias (P = .49) (Figure 2C). Heterogeneity for comparisons with placebo was as follows: SSRI: Q7 = 12.06, I2 = 38.6%; SNRI: Q4 = 1.77, I2 = 0%; TCA: Q3 = 3.45, I2 = 24.6%; benzodiazepine: Q1 = 0.05, I2 = 0%; 5-HT1A agonist: Q1 = 0.36, I2 = 0%. Our sensitivity analysis revealed this to be a stable network. When the medication with the highest SUCRA value (fluvoxamine) was removed, there was no change in the relative ranking of the remaining medications. When the medication with the lowest SUCRA value (clomipramine) was removed, only alprazolam and clonazepam shifted in ranking from 10th to 11th and from 11th to 10th, respectively. Notably, in our initial analysis alprazolam and clonazepam had very similar SUCRA values: 32% and 31%, respectively.
Anxiety symptom improvement. In pairwise comparisons of medication classes, SSRIs were superior to 5-HT1A agonists (mean difference: 4.4; CrI: [0.2 to 10.0]) and placebo (mean difference: 5.2; CrI: [2.8 to 8.8]), but there were no further significant findings (Figure 2B, Figure 3A). In terms of improvement in anxiety symptoms, the most effective class was the SSRIs, and benzodiazepines were the least effective.
Pairwise comparisons supported the superiority of 2 medications relative to placebo (Figure 4A): paroxetine (mean difference: 18.4; CrI: [4.1 to 32.4]) and fluvoxamine (mean difference: 8.3; CrI: [2.5 to 14.3]). Paroxetine was superior to 7 active treatments: clomipramine (mean difference: 24.2; CrI: [4.2 to 43.8]), alprazolam (mean difference: 17.7; CrI: [0.3 to 34.8]), buspirone (mean difference: 17.6; CrI: [2.9 to 32.2]), atomoxetine (mean difference: 16.1; CrI: [0.9 to 31.2]), venlafaxine (mean difference: 15.8, CrI: [1.2 to 30.4]), fluoxetine (mean difference: 15.7, CrI: [0.5 to 30.4], and duloxetine (mean difference: 15.6, CrI: [0.5 to 31.2]). In terms of symptom improvement, the most effective active treatment was paroxetine, while clomipramine was the least.
A funnel plot of these studies was not symmetric, and the Egger test suggested possible publication bias (P = .04) (Figure 2D).
Safety and Tolerability
All-cause discontinuation. No significant differences in all-cause discontinuation were detected among medication classes (Figure 3B). In terms of all-cause discontinuation, SSRI was the most tolerable class while TCA was the least.
Pairwise comparison of medications revealed early discontinuation was more likely in patients treated with clonazepam than all other active medications and placebo (Figure 4B). In terms of all-cause discontinuation, the most tolerable active treatment was alprazolam and the least effective was clonazepam.
A funnel plot of these studies appeared symmetric, and the Egger test did not indicate publication bias (P = .88) (Figure 2E). Heterogeneity for comparisons with placebo was as follows: SSRI: Q7 = 4.57, I2 = 0%; SNRI: Q3 = 2.75, I2 = 9.0%; TCA: Q4 = 3.49, I2 = 0%.
Discontinuation due to adverse events. Pairwise comparisons revealed significant differences between medication classes (Figure 3B). SNRIs were superior to 4 classes including α2 agonists (logOR: 30.1, 95% CrI: [3.0 to 95.9]), benzodiazepines (logOR: 22.0, 95% CrI: [1.7 to 77.2]), 5-HT1A agonists (logOR: 3.1, CrI: [0.3 to 7.1]), and SSRIs (logOR: 2.2, CrI: [0.3 to 4.3]). TCAs were superior to 2 classes including α2 agonists (logOR: 28.9, 95% CrI: [1.1 to 94.8]) and benzodiazepines (logOR: 20.6, 95% CrI: [0.03 to 76.0]). SSRIs were superior to α2 agonists (logOR: 27.9, 95% CrI: [0.7 to 93.8]). Placebo was associated with a lower likelihood of discontinuation compared to 4 medication classes: α2 agonists (logOR: 29.6, 95% CrI: [2.6, 95.5]), benzodiazepines (logOR: 21.6 [1.3 to 76.8]), 5-HT1A agonists (logOR: 2.6, 95% CrI: [0.2 to 6.4]), and SSRIs (logOR: 1.8, 95% CrI: [0.4 to 3.4]). In terms of discontinuation due to adverse events, SNRIs were the most tolerable and α2 agonists were the least.
In terms of specific medications, 4 were superior to clonazepam: duloxetine (logOR: 28.0, 95% CrI: [0.7 to 94.9]), venlafaxine (logOR: 28.9, 95% CrI: [1.9 to 95.7]), atomoxetine (logOR: 28.1, 95% CrI: [0.5 to 28.1]), and placebo (logOR: 28.1, 95% CrI: [1.4 to 94.9]) (Figure 4B). Four medications were also superior to guanfacine ER: duloxetine (logOR: 29.0, 95% CrI: [1.5 to 94.1]), venlafaxine (logOR: 30.0, 95% CrI: [2.8 to 95.1]), atomoxetine (logOR: 29.2, 95% CrI: [1.4 to 94.2]), and placebo (logOR: 29.2, 95% CrI: [2.2 to 94.3]. In terms of discontinuation due to adverse events, the most tolerable active treatment was venlafaxine and the least effective was guanfacine.
A funnel plot of these studies appeared symmetric, and the Egger test did not indicate publication bias (P = .46) (Figure 2F). Heterogeneity for comparisons with placebo was as follows: SSRI: Q6 = 2.08, I2 = 0%; SNRI: Q3 = 2.42, I2 = 0%; TCA: Q2 = 1.49, I2 = 0%; 5-HT1A agonist: Q1 = 1.10, I2 = 9.2%.
Treatment-emergent suicidality. Treatment-emergent suicidality did not differ across medication classes (Figure 3C). In terms of treatment-emergent suicidality, SNRIs were the most tolerable while TCAs were the least.
Pairwise comparisons between specific medications revealed significant differences in treatment-emergent suicidality (Figure 4C). Treatment-emergent suicidality was significantly greater in paroxetine-treated patients compared to those receiving sertraline (logOR: 43.5, 95% CrI: [10.1 to 96.0]), placebo (logOR: 19.5, 95% CrI: [1.7 to 60.4]), and duloxetine (logOR: 20.3, 95% CrI: [1.5 to 67.7]). Five other treatments were also associated with a higher rate of treatment-emergent suicidality than sertraline, including guanfacine (logOR: 42.7, 95% CrI: [7.2 to 102.4]), clonazepam (logOR: 41.0, 95% CrI: [7.0 to 97.8]), duloxetine (logOR: 20.0, 95% CrI: [0.5 to 65.1]), placebo (logOR: 19.79, 95% CrI: [0.5 to 64.8]), and venlafaxine (logOR: 21.5, 95% CrI: [1.6 to 66.3]). In terms of treatment-emergent suicidality, sertraline was the most tolerable active treatment while paroxetine was the least.
A funnel plot could not be generated as multiple studies lacked treatment-emergent suicidality events in 1 or more arms, which precluded calculating odds ratios. Heterogeneity for comparisons with placebo was as follows: SNRI: Q3 = 0.79, I2 = 0%; SSRI: Q1 = 2.58, I2 = 61.3%; benzodiazepine: Q1 = 1.49, I2 = 0%.
DISCUSSION
This comprehensive evaluation of comparative efficacy and tolerability of pharmacotherapy in pediatric anxiety disorders—which includes more trials than any prior meta-analysis and uses a probabilistic approach—suggests that SSRIs are superior to SNRIs and 5-HT1A agonists. Moreover, this report reveals clinically relevant class- and medication-specific tolerability differences in anxious youth. These findings confirm recommendations from the American Academy of Child and Adolescent Psychiatry and others regarding SSRIs as first-line pharmacotherapy for pediatric anxiety disorders.2,5 Moreover, given the heterogeneity of pharmacologic treatment approaches for pediatric anxiety disorders,6 these findings also guide clinicians’ medication selection within classes.
Our finding that treatment response is greater for SSRIs compared to SNRIs is consistent with prior analyses,8,11 including a recent report suggesting that the rate and magnitude of response to SSRIs are greater compared to SNRIs.42 This medication class-related difference in efficacy may relate to multiple clinical and pharmacologic factors. First, the serotonergic system matures earlier than the noradrenergic system, and this developmental lag in the noradrenergic system could subtend differences in the effectiveness of antidepressants targeting norepinephrine (eg, SNRIs and tricyclic antidepressants) versus serotonin (ie, SSRIs) between youth and adults.43 Second, the pathophysiology of anxiety may involve more serotonergic dysfunction relative to noradrenergic dysfunction,44 which could underlie the difference in efficacy between SSRIs and SNRIs. Third, the degree of serotonin blockade at a given dose may impact treatment response in pediatric patients with anxiety disorders; however, the degree to which an SNRI blocks serotonin reuptake at a given dose in the pediatric population is unknown.
That discontinuation due to adverse events is more likely with SSRIs (compared to SNRIs) is of interest for several reasons, including relative differences in side effect profiles and anxiety-specific experiencing of side effects (eg, differences in interoception). Among common side effects in children and adolescents is activation, a constellation of treatment-emergent symptoms including irritability, impulsivity, hyperactivity, and disinhibition.8 This adverse event cluster may be pathoetiologically linked to serotonergic neurotransmission. In this regard, in lower animals, administration of a 5-HT1A antagonist reverses SSRI-related activation-like effects, suggesting that activation may be due to an acutely hyposerotonergic state triggered by 5-HT1A agonism.45 However, specific side effects leading to discontinuation are infrequently reported, and we did not investigate differences in types of adverse events leading to discontinuation. Nevertheless, activation or similar serotonergically driven effects may explain observed differences in tolerability. Additionally, SSRI-related increases in serotonin release from the enterochromaffin cells of the gut and subsequent increased gastrointestinal motility may underlie some of the SSRI-related gastrointestinal adverse events, and these may contribute to increased discontinuation. Moreover, these symptoms may be particularly problematic in anxious youth, of whom up to 30% have abdominal symptoms prior to treatment.46 Finally, a recent study suggests that the presence of an anxiety disorder increases the reporting of side effects. In this study, adults with major depressive disorder (MDD) and co-occurring panic disorder were more likely to develop gastrointestinal, cardiac, neurologic, and genitourinary side effects during SSRI treatment compared to those without co-occurring panic disorder, a finding that may be potentially related to “heightened interoceptive awareness of changes in their body.”47
Treatment-emergent suicidality was associated with benzodiazepine treatment compared to placebo; however, 1 small RCT significantly influenced this result.41 Drug-to-drug differences in suicidality are of high interest. Sertraline-treated youth had lower rates of treatment-emergent suicidality compared to those receiving placebo and compared to 6 other active treatments (including 2 SNRIs and 1 SSRI [paroxetine]). Our finding that SSRIs may be differentially associated with both higher and lower rates of suicidality than placebo is of interest especially given classwise comparisons that did not reveal differences in treatment-emergent suicidality. This finding raises additional concerns regarding the broad black box warning concerning suicidal thinking and behavior in children, adolescents, and young adults that was added to the labeling of all antidepressant medications for all indications in 2004.14 Ostensibly, some antidepressants may be associated with higher risks of treatment-emergent suicidality (eg, paroxetine), while others (eg, sertraline) have a lower incidence of treatment-emergent suicidality compared to placebo in pediatric patients with anxiety disorders. Further, our analyses indicate that, as a class, the only medications with significantly more treatment-emergent suicidality relative to placebo in pediatric anxiety disorders are benzodiazepines, a class of medication currently without a black box warning for suicidal thinking or behavior in youth. Importantly, several recent studies in children and adolescents suggest that benzodiazepines may be associated with significant tolerability concerns. For example, in the Treatment of SSRI-Resistant Depression in Adolescents (TORDIA) study, suicidal adverse events occurred in 60% of adolescents treated with benzodiazepines versus 13% in patients not receiving benzodiazepines.15 However, patients with anxiety disorders in these studies may have had more severe psychopathology. This is an important consideration given the established increased risk of suicidality in patients with anxiety plus MDD.48 Moreover, prior meta-analyses of short-term treatment with benzodiazepines failed to reveal an increased risk of suicidality.49 Finally, the meta-analysis of antidepressant medications that—in part—gave birth to the black box warning for suicidal thinking and behavior in youth primarily relied on spontaneously reported suicidal thoughts and behaviors, and many of the early antidepressant studies lacked structured categorization of suicidality. It is possible that different methods for assessing side effects differentially affected the reporting of suicidality related adverse events between medications and placebo. These differences in side effect reporting differed across agents (as they had different industry sponsors) and may subtend the differences in suicidality risk across agents rather than actual differences between the agents themselves. Since most antidepressant trials systemically assessed suicidal thoughts and behaviors using structured, clinician-administered instruments (eg, Columbia-Suicide Severity Rating Scale),50 this will likely become a lesser issue in future trials.
Although our network meta-analysis provides clinically important findings, several treatments that are superior to placebo in individual RCTs failed to achieve significant efficacy. Placebo response in pediatric anxiety disorders varies significantly across studies (6%-78%) and is associated with trial-specific factors that varied considerably across the included studies.51 In this regard, we have previously reported that primary diagnosis, funding source, study setting, and the number of sites predict placebo response.51 For this reason, pooling placebo response across studies may attenuate the ability to detect drug-placebo differences. Of note, our primary outcome (treatment response, most commonly CGI-I) was more sensitive to class-placebo and drug-placebo differences, with 3 identified each, than anxiety symptom severity measures (most commonly PARS), which identified only 1 class-placebo difference (SSRIs) and 2 drug-placebo differences.
Limitations
While this is the largest network meta-analysis of psychopharmacologic treatments for pediatric anxiety disorders, there are several important limitations. First, multiarm studies, which enhance our ability to compare efficacy and tolerability, were infrequent in this sample,3,4 which further restricted our node-splitting consistency analysis. Second, trial design varied across trials in terms of the primary disorder under study (ie, social phobia/social anxiety disorder, GAD, separation anxiety disorder, or a mixed population), comorbidity (eg, inclusion or exclusion of comorbid MDD or ADHD),33 functional impairment (eg, school refusal),4 titration schedule (eg, fixed vs flexible dosing), and duration. This heterogeneity could obscure our ability to detect some medication- or medication class-specific differences in efficacy and tolerability. Third, the specificity of our primary preferred outcome measure, the CGI-I, to improvements in anxiety may have been confounded by comorbidities such as MDD and ADHD in some studies, so treatment may have improved these conditions without necessarily improving anxiety-related symptoms leading to global improvement overall. Additionally, the use of multiple disorder-specific rating scales and associated differences in the variance of symptom severity and response may have further degraded our ability to detect medication or class-specific differences in efficacy or tolerability. Similarly, the variability in measures of symptom severity may confound measures of anxiety by attributing some somatic symptoms to anxiety rather than to treatment (eg, gastrointestinal or neurologic symptoms on the HARS that overlap with SSRI-related side effects). Fourth, treatment-emergent suicidality was uncommon and not systematically evaluated in earlier studies that may have reduced the precision of comparative evaluations of treatment-emergent suicidality. As such, treatment-emergent suicidality may have been underestimated in this meta-analysis. Fifth, wide variability in placebo response and pooling placebo response decreased our ability to detect medication-placebo differences.51 Sixth, one of the included studies13 was presented as an exploratory analysis not intended to demonstrate efficacy and stated no primary outcome. Seventh, the inclusion of “zero event” trials with our Bayesian approach in some cases led to inordinately large logORs in some pairwise comparisons. The inclusion of zero event trials is a contentious issue in meta-analytic literature, with most discussion focusing on a frequentist rather than a Bayesian approach.52-54 We chose to include zero event arms, as excluding them would have essentially precluded a discussion of suicidal events in anxiety disorder clinical trials given the rarity of such events in both placebo and medication arms.
Finally, transitivity represents an endemic concern in meta-analyses and especially network meta-analyses. In this regard, differences between studies may impact the magnitude of treatment effect and may not necessarily reflect medication-related differences in anxiety symptom reduction. Such factors include the primary diagnosis, titration schedule, symptom severity, and comorbidity. Controlling for these factors is problematic, although the anxiety disorders studied herein are commonly evaluated together, respond similarly to antidepressant treatment, and share risk factors and neurobiology, and there is strong precedent for studying them together. Additionally, most of these studies have required at least “moderate” anxiety symptom severity as an inclusion criterion, and comorbidity has been generally similar across trials, with nearly all excluding patients with co-occurring major depressive disorder, bipolar disorder, eating disorders, and psychotic spectrum disorders. Regarding the potential impact of medication titration, all antidepressant trials have included flexible dosing with the exception of 2, and standardizing titration across medication classes with different mechanisms of action represents an intractable design problem.
Regarding the network geometry for efficacy comparisons (Figure 1), the networks include 2 closed loops (ie, direct comparison controlled trials).55 Thus, while these networks could be classified as complex, some might classify them as predominantly star-shaped56; predominantly star-shaped networks increase the dependence on indirect comparisons.57 Trials where placebo comparisons dominate the clinical trial landscape, as is the case for many pediatric trials, “suggest treatment preference bias.”56 However, the preference for placebo-controlled trials may also relate to regulatory mandates and the regulatory environment in which the trials were conducted.12,56 The networks examined herein tend to have moderate diversity (ie, number of treatments and treatment classes evaluated) but have low co-occurrence (ie, the balance of comparison frequency between specific treatments) and low homophily (the degree to which comparisons are made between treatments from within or between a class [SSRI vs TCA]).
CONCLUSIONS
SSRIs are superior to other classes of medications in reducing anxiety in pediatric patients with anxiety disorders, but are associated with a greater likelihood of discontinuation as a result of adverse events. For clinicians, these results suggest that among SSRIs, sertraline might be considered prior to other SSRIs based on its tolerability and efficacy and that some SSRIs should be considered as “second line” interventions (eg, paroxetine) based on their comparative tolerability profiles. The “most probable” ranking of efficacy and tolerability lays the groundwork for developing an evidence-based treatment hierarchy. Moving forward, large, multiple comparator RCTs are essential to inform further network meta-analyses and ultimately develop specific evidence-based guidelines to supplement or supplant heuristic judgments. Finally, our findings with regard to suicidality across medications and medication classes call for a reassessment of the specificity of the antidepressant black box warning, in particular with regard to youth with primary anxiety disorders.
Submitted: December 5, 2017; accepted August 16, 2018.
Published online: January 29, 2019.
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, none of the drugs discussed in the article are approved by the US Food and Drug Administration for the treatment of pediatric anxiety disorders, except that duloxetine is approved for the treatment of generalized anxiety disorder in children aged 7 to 17 years and fluvoxamine is approved for the treatment of obsessive-compulsive disorder in children aged 8 to 17 years.
Financial disclosure: Dr Dobson has received support from the American Academy of Child and Adolescent Psychiatry (Campaign for America’s Kids). Dr Bloch is on the Scientific Advisory Board of Therapix Biosciences and receives research support from Biohaven Pharmaceuticals, Therapix Biosciences, National Institutes of Health, Tourette Association of America, the Brain & Behavior Research Foundation (formerly NARSAD), and the Patterson Foundation. Dr Strawn has received research support from Edgemont, Allergan, Lundbeck, Shire, Neuronetics, and the National Institute of Mental Health (NIMH and NIEHS); receives royalties from Springer Publishing and UpToDate; and has received material support from and consulted to GeneSight Assurex.
Funding/support: This work was supported by the National Institute of Mental Health (grant K23MH10637, to Dr Strawn).
Role of sponsor: The National Institute of Mental Health had no role in the conduct or publication of this study.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1. Merikangas KR, He J-P, Burstein M, et al. Lifetime prevalence of mental disorders in US adolescents: results from the National Comorbidity Survey Replication: Adolescent Supplement (NCS-A). J Am Acad Child Adolesc Psychiatry. 2010;49(10):980-989. PubMed CrossRef
2. Wehry AM, Beesdo-Baum K, Hennelly MM, et al. Assessment and treatment of anxiety disorders in children and adolescents. Curr Psychiatry Rep. 2015;17(7):52. PubMed CrossRef
3. da Costa CZ, de Morais RM, Zanetta DM, et al. Comparison among clomipramine, fluoxetine, and placebo for the treatment of anxiety disorders in children and adolescents. J Child Adolesc Psychopharmacol. 2013;23(10):687-692. PubMed CrossRef
4. Bernstein GA, Garfinkel BD, Borchardt CM. Comparative studies of pharmacotherapy for school refusal. J Am Acad Child Adolesc Psychiatry. 1990;29(5):773-781. PubMed CrossRef
5. Connolly SD, Bernstein GA; Work Group on Quality Issues. Practice parameter for the assessment and treatment of children and adolescents with anxiety disorders. J Am Acad Child Adolesc Psychiatry. 2007;46(2):267-283. PubMed CrossRef
6. Tulisiak AK, Klein JA, Harris E, et al. Antidepressant prescribing by pediatricians: a mixed-methods analysis. Curr Probl Pediatr Adolesc Health Care. 2017;47(1):15-24. PubMed CrossRef
7. Bridge JA, Iyengar S, Salary CB, et al. Clinical response and risk for reported suicidal ideation and suicide attempts in pediatric antidepressant treatment: a meta-analysis of randomized controlled trials. JAMA. 2007;297(15):1683-1696. PubMed CrossRef
8. Strawn JR, Welge JA, Wehry AM, et al. Efficacy and tolerability of antidepressants in pediatric anxiety disorders: a systematic review and meta-analysis. Depress Anxiety. 2015;32(3):149-157. PubMed CrossRef
9. Locher C, Koechlin H, Zion SR, et al. Efficacy and safety of selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors, and placebo for common psychiatric disorders among children and adolescents: a systematic review and meta-analysis. JAMA Psychiatry. 2017;74(10):1011-1020. PubMed CrossRef
10. Wang Z, Whiteside SPH, Sim L, et al. Comparative effectiveness and safety of cognitive behavioral therapy and pharmacotherapy for childhood anxiety disorders: a systematic review and meta-analysis. JAMA Pediatr. 2017;171(11):1049-1056. PubMed CrossRef
11. Locher C, Koechlin H, Zion SR, et al. Efficacy and safety of selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors, and placebo for common psychiatric disorders among children and adolescents: a systematic review and meta-analysis. JAMA Psychiatry. 2017;74(10):1011-1020. PubMed CrossRef
12. Strawn JR, Mills JA, Cornwall GJ, et al. Buspirone in children and adolescents with anxiety: a review and Bayesian analysis of abandoned randomized controlled trials. J Child Adolesc Psychopharmacol. 2018;28(1):2-9. PubMed CrossRef
13. Strawn JR, Compton SN, Robertson B, et al. Extended release guanfacine in pediatric anxiety disorders: a pilot, randomized, placebo-controlled trial. J Child Adolesc Psychopharmacol. 2017;27(1):29-37. PubMed CrossRef
14. US Food and Drug Administration. Relationship between psychotropic drugs and pediatric suicidality: review and evaluation of clinical data. FDA.gov website. http://www.fda.gov/ohrms/dockets/ac/04/briefing/2004-4065b1-10-TAB08-Hammads-Review.pdf.
15. Brent DA, Emslie GJ, Clarke GN, et al. Predictors of spontaneous and systematically assessed suicidal adverse events in the Treatment of SSRI-Resistant Depression in Adolescents (TORDIA) study. Am J Psychiatry. 2009;166(4):418-426. PubMed CrossRef
16. Strawn JR, Geracioti L, Rajdev N, et al. Pharmacotherapy for generalized anxiety disorder in adult and pediatric patients: an evidence-based treatment review. Expert Opin Pharmacother. 2018;19(10):1057-1070. PubMed CrossRef
17. Rohatgi A. WebPlotDigitizer. 2017. https://automeris.io/WebPlotDigitizer/.
18. The Pediatric Anxiety Rating Scale (PARS): development and psychometric properties. J Am Acad Child Adolesc Psychiatry. 2002;41(9):1061-1069. PubMed CrossRef
19. Hamilton M. The assessment of anxiety states by rating. Br J Med Psychol. 1959;32(1):50-55. PubMed CrossRef
20. March JS, Parker JD, Sullivan K, et al. The Multidimensional Anxiety Scale for Children (MASC): factor structure, reliability, and validity. J Am Acad Child Adolesc Psychiatry. 1997;36(4):554-565. PubMed CrossRef
21. Higgins J, Green S. Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0. London, UK: The Cochrane Collaboration; 2011.
22. Dias S, Welton NJ, Caldwell DM, et al. Checking consistency in mixed treatment comparison meta-analysis. Stat Med. 2010;29(7-8):932-944. PubMed CrossRef
23. Cipriani A, Zhou X, Del Giovane C, et al. Comparative efficacy and tolerability of antidepressants for major depressive disorder in children and adolescents: a network meta-analysis. Lancet. 2016;388(10047):881-890. PubMed
24. Salanti G, Ades AE, Ioannidis JPA. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol. 2011;64(2):163-171. PubMed CrossRef
25. Duval S, Tweedie R. Trim and fill: a simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000;56(2):455-463. PubMed CrossRef
26. Black B, Uhde TW. Treatment of elective mutism with fluoxetine: a double-blind, placebo-controlled study. J Am Acad Child Adolesc Psychiatry. 1994;33(7):1000-1006. PubMed CrossRef
27. The Research Unit on Pediatric Psychopharmacology Anxiety Study Group. Fluvoxamine for the treatment of anxiety disorders in children and adolescents. N Engl J Med. 2001;344(17):1279-1285. PubMed CrossRef
28. Rynn MA, Siqueland L, Rickels K. Placebo-controlled trial of sertraline in the treatment of children with generalized anxiety disorder. Am J Psychiatry. 2001;158(12):2008-2014. PubMed CrossRef
29. Birmaher B, Axelson DA, Monk K, et al. Fluoxetine for the treatment of childhood anxiety disorders. J Am Acad Child Adolesc Psychiatry. 2003;42(4):415-423. PubMed CrossRef
30. Wagner KD, Berard R, Stein MB, et al. A multicenter, randomized, double-blind, placebo-controlled trial of paroxetine in children and adolescents with social anxiety disorder. Arch Gen Psychiatry. 2004;61(11):1153-1162. PubMed CrossRef
31. Beidel DC, Turner SM, Sallee FR, et al. SET-C versus fluoxetine in the treatment of childhood social phobia. J Am Acad Child Adolesc Psychiatry. 2007;46(12):1622-1632. PubMed CrossRef
32. Walkup JT, Albano AM, Piacentini J, et al. Cognitive behavioral therapy, sertraline, or a combination in childhood anxiety. N Engl J Med. 2008;359(26):2753-2766. PubMed CrossRef
33. Geller D, Donnelly C, Lopez F, et al. Atomoxetine treatment for pediatric patients with attention-deficit/hyperactivity disorder with comorbid anxiety disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(9):1119-1127. PubMed CrossRef
34. March JS, Entsuah AR, Rynn M, et al. A randomized controlled trial of venlafaxine ER versus placebo in pediatric social anxiety disorder. Biol Psychiatry. 2007;62(10):1149-1154. PubMed CrossRef
35. Rynn MA, Riddle MA, Yeung PP, et al. Efficacy and safety of extended-release venlafaxine in the treatment of generalized anxiety disorder in children and adolescents: two placebo-controlled trials. Am J Psychiatry. 2007;164(2):290-300. PubMed CrossRef
36. Strawn JR, Prakash A, Zhang Q, et al. A randomized, placebo-controlled study of duloxetine for the treatment of children and adolescents with generalized anxiety disorder. J Am Acad Child Adolesc Psychiatry. 2015;54(4):283-293. PubMed CrossRef
37. Gittelman-Klein R, Klein DF. School phobia: controlled imipramine treatment. Calif Med. 1971;115(3):42. PubMed
38. Berney T, Kolvin I, Bhate SR, et al. School phobia: a therapeutic trial with clomipramine and short-term outcome. Br J Psychiatry. 1981;138(2):110-118. PubMed CrossRef
39. Klein RG, Koplewicz HS, Kanner A. Imipramine treatment of children with separation anxiety disorder. J Am Acad Child Adolesc Psychiatry. 1992;31(1):21-28. PubMed CrossRef
40. Simeon JG, Ferguson HB, Knott V, et al. Clinical, cognitive, and neurophysiological effects of alprazolam in children and adolescents with overanxious and avoidant disorders. J Am Acad Child Adolesc Psychiatry. 1992;31(1):29-33. PubMed CrossRef
41. Graae F, Milner J, Rizzotto L, et al. Clonazepam in childhood anxiety disorders. J Am Acad Child Adolesc Psychiatry. 1994;33(3):372-376. PubMed CrossRef
42. Strawn JR, Mills JA, Sauley BA, et al. The impact of antidepressant dose and class on treatment response in pediatric anxiety disorders: a meta-analysis. J Am Acad Child Adolesc Psychiatry. 2018;57(4):235-244.e2. PubMed CrossRef
43. Murrin LC, Sanders JD, Bylund DB. Comparison of the maturation of the adrenergic and serotonergic neurotransmitter systems in the brain: implications for differential drug effects on juveniles and adults. Biochem Pharmacol. 2007;73(8):1225-1236. PubMed CrossRef
44. Ressler KJ, Nemeroff CB. Role of serotonergic and noradrenergic systems in the pathophysiology of depression and anxiety disorders. Depress Anxiety. 2000;12(suppl 1):2-19. PubMed CrossRef
45. Rahn KA, Cao YJ, Hendrix CW, et al. The role of 5-HT1A receptors in mediating acute negative effects of antidepressants: implications in pediatric depression. Transl Psychiatry. 2015;5(5):e563. PubMed CrossRef
46. Crawley SA, Caporino NE, Birmaher B, et al. Somatic complaints in anxious youth. Child Psychiatry Hum Dev. 2014;45(4):398-407. PubMed CrossRef
47. Shankman SA, Gorka SM, Katz AC, et al. Side effects to antidepressant treatment in patients with depression and comorbid panic disorder. J Clin Psychiatry. 2017;78(4):433-440. PubMed CrossRef
48. Foley DL, Goldston DB, Costello EJ, et al. Proximal psychiatric risk factors for suicidality in youth: the Great Smoky Mountains Study. Arch Gen Psychiatry. 2006;63(9):1017-1024. PubMed CrossRef
49. Kuang H, Johnson JA, Mulqueen JM, et al. The efficacy of benzodiazepines as acute anxiolytics in children: a meta-analysis. Depress Anxiety. 2017;34(10):888-896. PubMed CrossRef
50. Posner K, Brown GK, Stanley B, et al. The Columbia-Suicide Severity Rating Scale: initial validity and internal consistency findings from three multisite studies with adolescents and adults. Am J Psychiatry. 2011;168(12):1266-1277. PubMed CrossRef
51. Dobson ET, Strawn JR. Placebo response in pediatric anxiety disorders: implications for clinical trial design and interpretation. J Child Adolesc Psychopharmacol. 2016;26(8):686-693. PubMed CrossRef
52. Ma Y, Chu H, Mazumdar M. Meta-analysis of proportions of rare events-a comparison of exact likelihood methods with robust variance estimation. Commun Stat Simul Comput. 2016;45(8):3036-3052. PubMed CrossRef
53. Bhaumik DK, Amatya A, Normand S-L, et al. Meta-analysis of rare binary adverse event data. J Am Stat Assoc. 2012;107(498):555-567. PubMed CrossRef
54. Friedrich JO, Adhikari NKJ, Beyene J. Inclusion of zero total event trials in meta-analyses maintains analytic consistency and incorporates all available data. BMC Med Res Methodol. 2007;7(1):5. PubMed CrossRef
55. Mills EJ, Ioannidis JPA, Thorlund K, et al. How to use an article reporting a multiple treatment comparison meta-analysis. JAMA. 2012;308(12):1246-1253. PubMed CrossRef
56. Salanti G, Kavvoura FK, Ioannidis JPA. Exploring the geometry of treatment networks. Ann Intern Med. 2008;148(7):544-553. PubMed CrossRef
57. Hutton B, Salanti G, Caldwell DM, et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med. 2015;162(11):777-784. PubMed CrossRef
This PDF is free for all visitors!