Objective: To investigate vilazodone, currently approved for major depressive disorder in adults, for generalized anxiety disorder (GAD).
Method: Three randomized, double-blind, placebo-controlled studies showing positive results for vilazodone (2,040 mg/d) in adult patients with GAD (DSM-IV-TR) were pooled for analyses; data were collected from June 2012 to March 2014. Post hoc outcomes in the pooled intent-to-treat population (n = 1,462) included mean change from baseline to week 8 in Hamilton Anxiety Rating Scale (HARS) total score, psychic and somatic anxiety subscale scores, and individual item scores; HARS response (≥ 50% total score improvement) and remission (total score ≤ 7) at week 8; and category shifts, defined as HARS item score ≥ 2 at baseline (moderate to very severe symptoms) and score of 0 at week 8 (no symptoms).
Results: The least squares mean difference was statistically significant for vilazodone versus placebo in change from baseline to week 8 in HARS total score (−1.83, P < .0001) and in psychic anxiety (−1.21, P < .0001) and somatic anxiety (−0.63, P < .01) subscale scores; differences from placebo were significant on 11 of 14 HARS items (P < .05). Response rates were higher with vilazodone than placebo (48% vs 39%, P < .001), as were remission rates (27% vs 21%, P < .01). The percentage of patients who shifted to no symptoms was significant for vilazodone on several items: anxious mood, tension, intellectual, depressed mood, somatic-muscular, somatic-sensory, cardiovascular, respiratory, and autonomic symptoms (P < .05).
Conclusions: Treatment with vilazodone versus placebo was effective in adult GAD patients, with significant differences between treatment groups found on both psychic and somatic HARS items.
Trial Registration: ClinicalTrials.gov identifiers: NCT01629966, NCT01766401, NCT01844115.
Post Hoc Analyses of Anxiety Measures in Adult Patients
With Generalized Anxiety Disorder Treated With Vilazodone
ABSTRACT
Objective: To investigate vilazodone, currently approved for major depressive disorder in adults, for generalized anxiety disorder (GAD).
Method: Three randomized, double-blind, placebo-controlled studies showing positive results for vilazodone (2,040 mg/d) in adult patients with GAD (DSM-IV-TR) were pooled for analyses; data were collected from June 2012 to March 2014. Post hoc outcomes in the pooled intent-to-treat population (n = 1,462) included mean change from baseline to week 8 in Hamilton Anxiety Rating Scale (HARS) total score, psychic and somatic anxiety subscale scores, and individual item scores; HARS response (≥ 50% total score improvement) and remission (total score ≤ 7) at week 8; and category shifts, defined as HARS item score ≥ 2 at baseline (moderate to very severe symptoms) and score of 0 at week 8 (no symptoms).
Results: The least squares mean difference was statistically significant for vilazodone versus placebo in change from baseline to week 8 in HARS total score (−1.83, P < .0001) and in psychic anxiety (−1.21, P < .0001) and somatic anxiety (−0.63, P < .01) subscale scores; differences from placebo were significant on 11 of 14 HARS items (P < .05). Response rates were higher with vilazodone than placebo (48% vs 39%, P < .001), as were remission rates (27% vs 21%, P < .01). The percentage of patients who shifted to no symptoms was significant for vilazodone on several items: anxious mood, tension, intellectual, depressed mood, somatic-muscular, somatic-sensory, cardiovascular, respiratory, and autonomic symptoms (P < .05).
Conclusions: Treatment with vilazodone versus placebo was effective in adult GAD patients, with significant differences between treatment groups found on both psychic and somatic HARS items.
Trial Registration: ClinicalTrials.gov identifiers: NCT01629966, NCT01766401, NCT01844115.
Prim Care Companion CNS Disord 2016;18(2):doi:10.4088/PCC.15m01904
© Copyright 2016 Physicians Postgraduate Press, Inc.
aNorthwest Clinical Research Center, Bellevue, Washington
bDuke University School of Medicine, Department of Psychiatry, Durham, North Carolina
cForest Research Institute, an Allergan affiliate, Jersey City, New Jersey
dPrescott Medical Communications Group, Chicago, Illinois
*Corresponding author: Arif Khan, MD, Northwest Clinical Research Center, 1900 116th Ave NE #112, Bellevue, WA 98004 ([email protected]).
Generalized anxiety disorder (GAD) is characterized by psychic and somatic symptoms including excessive worrying, fatigue, difficulty concentrating, irritability, muscle tension, and sleep disturbance.1 Such symptoms can be severe enough to cause clinically significant impairment in social and occupational activities1 and compromise patient quality of life.2 Although the goal of treatment is remission, often defined as an asymptomatic state and a return to premorbid levels of functioning,3 the prognosis for GAD recovery has generally been viewed pessimistically due to its chronic and recurrent nature.4
Although GAD is one of the most common psychiatric disorders seen in primary care settings, it is frequently unrecognized or misdiagnosed5; moreover, an estimated 58% of diagnosed patients are untreated.6 The diagnosis and treatment of GAD are often confounded by comorbidity with other physical and mental disorders, including major depressive disorder (MDD).7 The level of disability associated with GAD is comparable to the level of disability in MDD and greater than the disability associated with alcohol and drug use disorders, nicotine dependence, other anxiety disorders, and personality disorders.8 According to World Health Organization data,9 56.3% of individuals with GAD are severely disabled. GAD has also been associated with increased economic costs, including reduced workplace performance and overutilization of medical resources.10,11 Such costs greatly increase the economic burden of GAD, making this disorder an important mental health issue that affects patients, health care providers, payers, employers, and society at large.
Pharmacotherapy efficacy in the treatment of GAD has been demonstrated with agents from a variety of drug classes including selective serotonin reuptake inhibitors (SSRIs; escitalopram, paroxetine, sertraline), serotonin-norepinephrine reuptake inhibitors (SNRIs; duloxetine, venlafaxine), benzodiazepines (alprazolam, diazepam, lorazepam), and the 5-HT1A receptor partial agonist buspirone and the anticonvulsant pregabalin.12,13 Although some prior research14 suggests that SSRIs and SNRIs may have greater efficacy for psychic anxiety compared to somatic anxiety in GAD, evidence-based guidelines recommend these agents as the initial treatment option on the basis of proven overall efficacy and good tolerability.12
Vilazodone is an SSRI and 5-HT1A receptor partial agonist approved by the US Food and Drug Administration (FDA) for the treatment of MDD in adults; it is also approved for the treatment of MDD in Mexico and Canada. The efficacy and safety of vilazodone in MDD have been demonstrated in positive placebo-controlled phase 3 and 4 trials.15–18 The pharmacologic profile of vilazodone suggested a potential for treating symptoms of anxiety, which was supported by a post hoc analysis of data from patients with anxious depression in 2 of the MDD studies.19
Vilazodone was subsequently investigated for the treatment of GAD in 1 fixed-dose20 and 2 flexible-dose21,22 double-blind, randomized, placebo-controlled trials in adult patients. The primary efficacy parameter in each trial was mean change from baseline to week 8 in Hamilton Anxiety Rating Scale (HARS) total score. In the 2 flexible-dose trials,21,22 the difference in mean change in HARS total score for vilazodone 20–40 mg/d versus placebo was statistically significant (P < .05); in the fixed-dose trial of vilazodone 20 or 40 mg/d,20 a statistically significant difference in HARS total score mean change was seen in the 40-mg/d group (P < .05) but not in the 20-mg/d group.
Despite available pharmacotherapy options, GAD is still the least successfully treated anxiety disorder.23 Response to treatment in anxiety disorders is usually defined as a percent reduction in an anxiety rating scale score such as the HARS24; remission is often determined by meeting a predefined threshold score. However, while marked reductions in symptom severity on a rating scale can indicate significant treatment effect, many patients with rating scale improvements are still burdened by considerable anxiety symptoms.12
Controlled clinical trials provide important data about the ability of an agent to reduce anxiety symptoms, but a mean score change from baseline on a validated rating scale is not the only clinically relevant outcome. The extent of recovery in GAD may be more fully elucidated by additionally assessing changes in individual symptoms, subscales, and symptom categories to evaluate treatment effect. Diverse and comprehensive outcomes of interest may help inform clinicians and improve patient well-being and recovery in the treatment of GAD. As such, post hoc analyses were conducted on pooled data from the 3 vilazodone trials in GAD to more comprehensively evaluate the effect of treatment across the range of anxiety symptoms associated with GAD.
METHOD
Study Design and Patients
Data from the three 8-week, double-blind, randomized, placebo-controlled studies showing positive results for vilazodone in adult patients with GAD were pooled for post hoc analyses. These included a fixed-dose study of vilazodone 20 or 40 mg/d (NCT01629966)20 and 2 flexible-dose studies of vilazodone 20–40 mg/d (NCT01766401, NCT01844115)21,22; all vilazodone dose groups (20–40 mg/d) were pooled for the current post hoc analyses. Data were collected from June 2012 to March 2014. Detailed methods of the constituent studies have been previously published.20–22 The studies included male and female patients (18–70 years of age) who met DSM-IV-TR criteria25 for GAD. At baseline, patients were required to have a HARS total score ≥ 20, a score ≥ 2 on both HARS item 1 (anxious mood) and 2 (tension), a Clinical Global Impressions–Severity of Illness (CGI-S)26 score ≥ 4 (indicating at least moderate severity), and a 17-item Hamilton Depression Rating Scale (HDRS-17)27 total score ≤ 17.
Key exclusion criteria were an Axis I diagnosis other than GAD within 6 months (comorbid social anxiety disorder and specific phobias were allowed if secondary to GAD diagnosis), lifetime history of various psychiatric disorders (eg, bipolar disorder, psychotic disorder, depressive episode with psychotic or catatonic features), nonresponse to adequate treatment trials of ≥ 2 SSRIs or SNRIs for GAD (≥ 8 weeks at the recommended dose), and suicide risk (past year attempt, score ≥ 3 on item 3 of the HDRS-17, or investigator judgment). Medical conditions that could interfere with study conduct, confound the interpretation of study results, or endanger patient well-being were also exclusionary. Psychoactive drugs were prohibited, although eszopiclone, zopiclone, zaleplon, or zolpidem could be continued for insomnia.
Post Hoc Analyses
Post hoc analyses were based on the 14-item HARS in which items are scored on a scale ranging from 0 to 4, with higher scores indicating greater symptom severity (0 = not present, 1 = mild, 2 = moderate, 3 = severe, 4 = very severe). The psychic anxiety subscale consists of HARS items 1 to 6 (anxious mood, tension, fears, insomnia, intellectual, depressed mood) and item 14 (behavior at interview); the somatic anxiety subscale consists of HARS items 7 to 13 (somatic-muscular, somatic-sensory, cardiovascular symptoms, respiratory symptoms, gastrointestinal symptoms, genitourinary symptoms, autonomic symptoms).
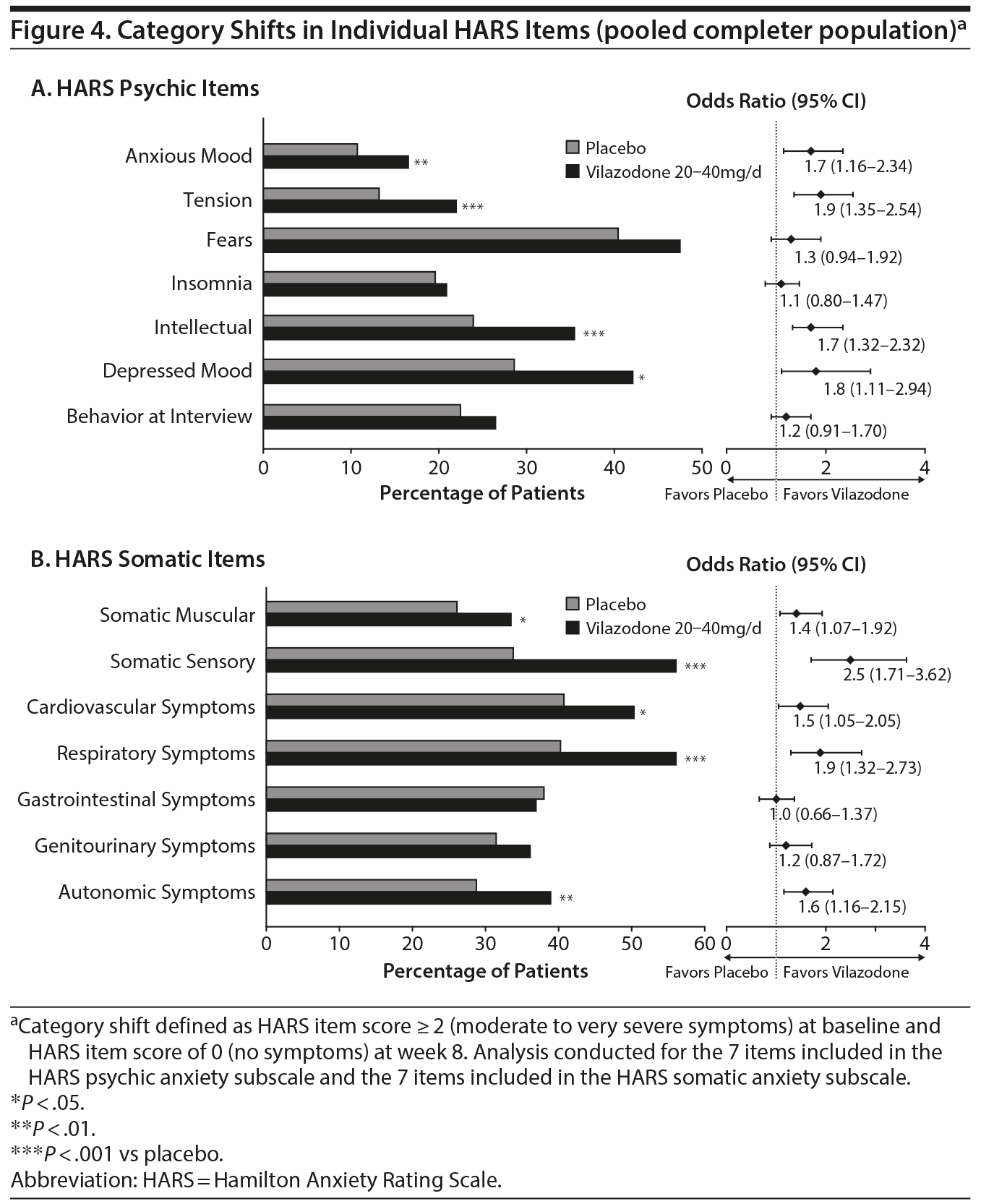
Efficacy was evaluated on the basis of mean changes from baseline to week 8 in HARS total score, HARS psychic and somatic anxiety subscale scores, and HARS individual item scores. HARS response (≥ 50% improvement from baseline) and remission (total score ≤ 7) at week 8 were also analyzed. In a novel category shift analysis, the percentage of patients who shifted from a more severe symptom category at baseline to no symptoms at the end of week 8 on each of the 14 individual HARS items was assessed. A category shift was defined as a HARS item score ≥ 2 (moderate to very severe) at baseline and score of 0 (no symptoms) at week 8.
Statistical Analyses
Most efficacy analyses were conducted in the pooled intent-to-treat (ITT) population, defined as all patients who received study medication and had ≥ 1 postbaseline HARS assessment. Category shift analyses were conducted in the pooled completer population, defined as patients in the pooled ITT population who had valid HARS total scores at both baseline and at week 8. Least squares (LS) mean change from baseline and least squares mean difference (LSMD) were calculated using a mixed-effects model for repeated measures with treatment group, pooled study center, visit, and treatment group by visit interaction as fixed effects and baseline value and baseline value by visit interaction as covariates; Cohen d effect sizes were estimated. Rates of HARS response and remission and the associated odds ratios (ORs) and 95% confidence intervals (CIs) were analyzed by logistic regression with treatment group as a factor and baseline value as a covariate. The ORs and P values for HARS category shifts were calculated using logistic regression with treatment group as a factor. The P values were not adjusted for multiple comparisons.
RESULTS
Baseline Demographics and Clinical Characteristics
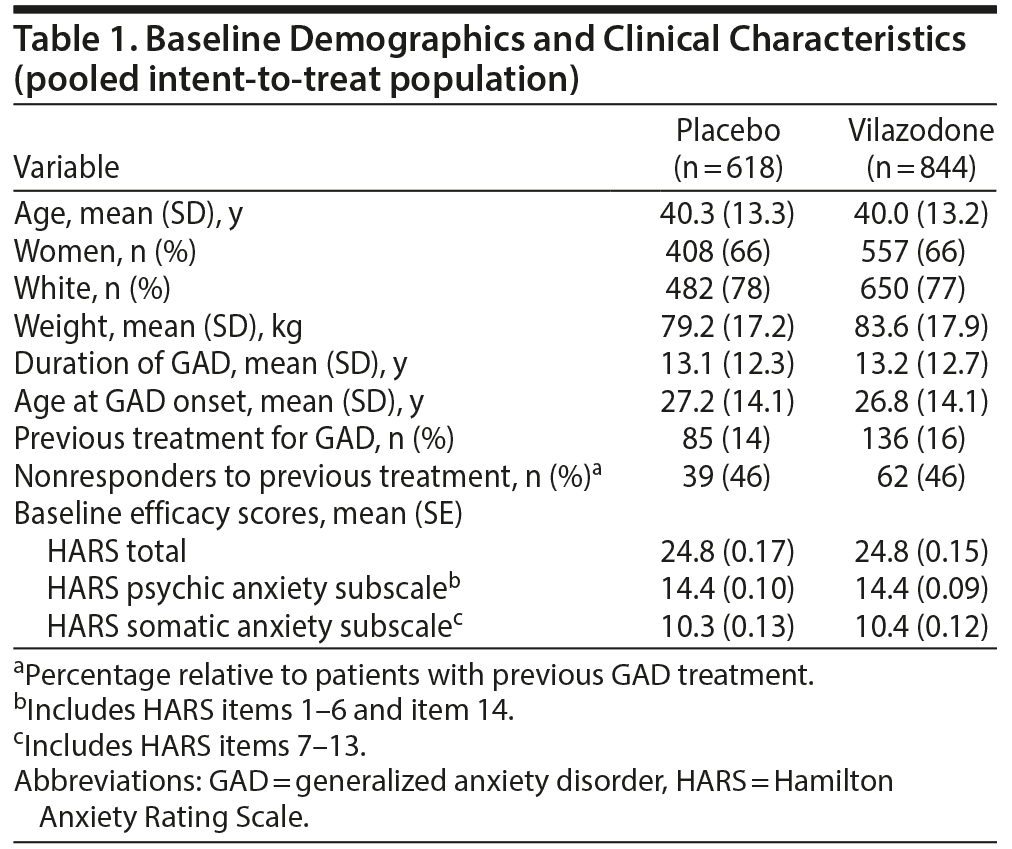
There were 1,462 patients in the pooled ITT population and 1,150 patients in the pooled completer population. Baseline demographics, clinical characteristics, and GAD duration were generally similar between the placebo and vilazodone groups (Table 1); characteristics were also similar between the pooled ITT and completer populations (data not shown). Baseline HARS psychic anxiety subscale scores were greater than baseline somatic anxiety subscale scores in the vilazodone and placebo treatment groups.
Post Hoc Efficacy Outcomes:
HARS Change From Baseline and Response/Remission
In the pooled ITT population, the LSMD (95% CI) for change from baseline to week 8 in HARS total score was statistically significant in favor of vilazodone over placebo (–1.83 [–2.62 to −1.04], P < .0001). Significant improvement for vilazodone versus placebo was also seen in change from baseline on the psychic anxiety subscale score (−1.21 [−1.69 to −0.73], P < .0001) and somatic anxiety subscale score (−0.63 [−1.01 to −0.25], P = .0012) (Figure 1).
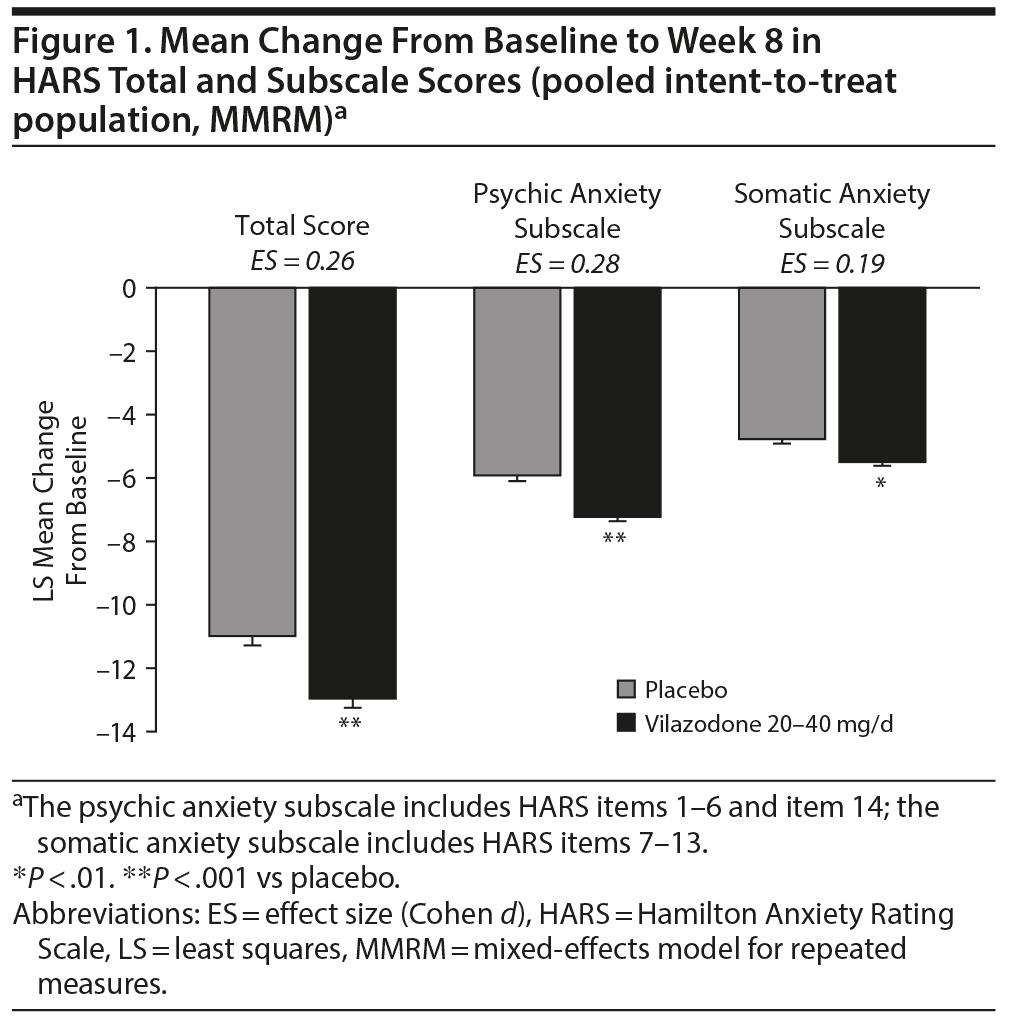
Rates of HARS response (total score reduction ≥ 50%) and remission (total score ≤ 7) at week 8 were significantly greater for vilazodone compared with placebo (Figure 2); the OR (95% CI) for response was 1.44 (1.16–1.77), P < .001 and for remission was 1.42 (1.10–1.82), P < .01.
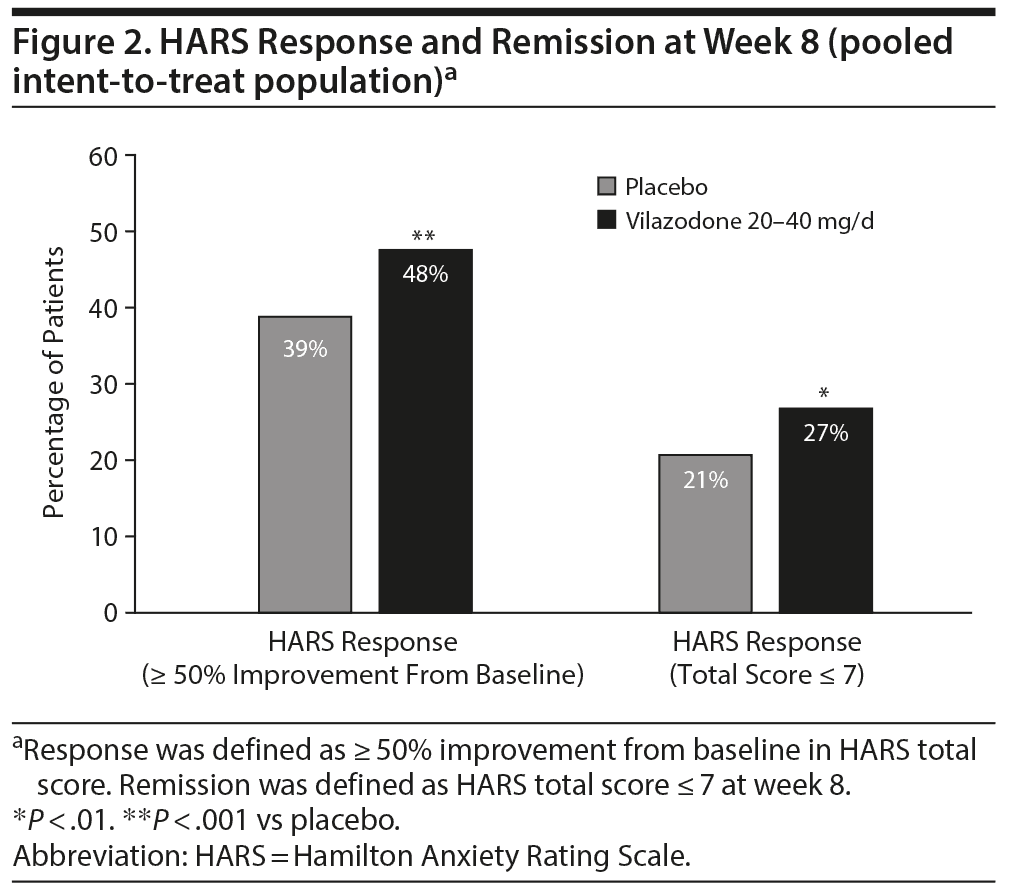
HARS Individual Items
On the individual HARS items that comprise the psychic anxiety subscale, the LS mean change from baseline to week 8 was significantly greater for vilazodone versus placebo on all items except insomnia (effect size range for all items: 0.11 to 0.32) (Figure 3A). On the items that comprise the somatic anxiety subscale, the LS mean change from baseline was significantly greater for vilazodone versus placebo on all items except for genitourinary symptoms and gastrointestinal symptoms (effect size range for all items: −0.07 to 0.20) (Figure 3B).
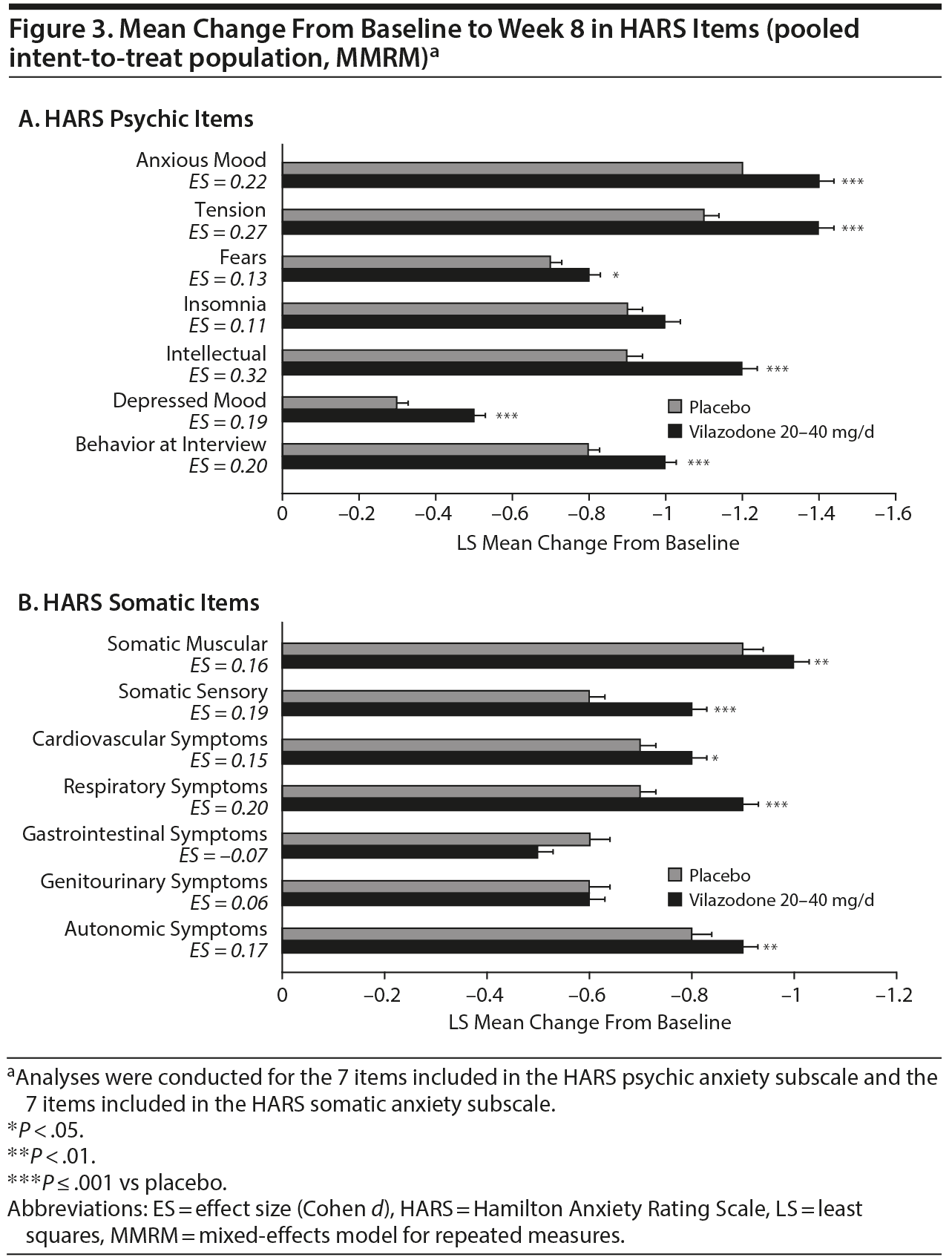
Category Shift Analyses
A significantly higher percentage of vilazodone- versus placebo-treated patients met the shift criteria (score ≥ 2 at baseline, score of 0 at week 8) on 4 items of the psychic anxiety subscale (anxious mood, tension, intellectual, depressed mood) and 5 items of the somatic anxiety subscale (somatic-muscular, somatic-sensory, cardiovascular symptoms, respiratory symptoms, and autonomic symptoms) (Figure 4).
DISCUSSION
The results of these post hoc analyses indicated greater mean improvements, along with higher odds of clinically relevant outcomes, for vilazodone 20–40 mg/d compared with placebo. The mean decrease from baseline to week 8 was statistically greater with vilazodone versus placebo in HARS total and subscale (psychic anxiety, somatic anxiety) scores and on 11 of 14 individual HARS item scores. At week 8, HARS response and remission rates were significantly greater for vilazodone than for placebo. In a unique category shift analysis, a significantly higher percentage of vilazodone-treated than placebo-treated patients shifted from the moderate to very severe symptom category at baseline to no symptoms at week 8 on 9 of 14 HARS items. This multifaceted approach to determining treatment effect suggests that adult patients with GAD who were treated with vilazodone achieved meaningful improvements across the range of psychic and somatic anxiety symptoms.
In the pooled ITT population, the HARS total score effect size for vilazodone was 0.26. In a meta-analysis of acute treatment trials in GAD,28 the overall effect size for placebo-controlled studies that reported change in HARS total score was 0.39, with some differences among drug classes or individual drugs (pregabalin, 0.50; hydroxyzine, 0.45; SNRIs, 0.42; benzodiazepines, 0.38; SSRIs, 0.36; buspirone, 0.17). While these values provide some context for discussing drug effects in GAD, interpretation of effect sizes in clinical trials is not straightforward due to methodological differences among studies that may influence the magnitude of effect. For the current analyses, the magnitude of effect sizes was affected by factors inherent to the study designs (eg, application of stringent inclusion and exclusion criteria) or the post hoc design (eg, constituent studies not prospectively designed or powered to evaluate single items). In addition, tolerability issues may have biased some of the efficacy results. Moreover, the symptomatic profile of individual GAD patients should be taken into account since different medications may have varying effects on specific symptoms, as suggested by the range of effect sizes that were found for individual HARS items in the current analysis.
Mean baseline scores were higher on the psychic anxiety subscale than on the somatic anxiety subscale, indicating a higher burden of psychic symptoms and the potential for more improvement in this symptom domain. As expected, the effect size for vilazodone was larger on the psychic anxiety subscale (0.28) than the somatic anxiety subscale (0.19). However, vilazodone appeared to improve both psychic and somatic anxiety symptoms, as shown by the significant improvement seen for vilazodone compared with placebo for 6 of 7 individual items on the HARS psychic anxiety subscale (all except insomnia) and for 5 of 7 items on the somatic anxiety subscale (all except genitourinary and gastrointestinal symptoms). Effect sizes for psychic anxiety subscale items ranged from 0.11 (insomnia) to 0.32 (intellectual); effect sizes for the somatic anxiety subscale items ranged from −0.07 (gastrointestinal symptoms) to 0.20 (respiratory symptoms).
Robust responses on certain HARS items may reflect particularly strong improvement in some of the core features of GAD. Compared with other HARS items, larger effect sizes were seen in anxious mood, tension (ie, restlessness, tension, fatigability), intellectual (ie, difficulty concentrating, poor memory), somatic-muscular, and autonomic symptoms, which closely correspond to the diagnostic symptoms for GAD as defined by the DSM-5.29 Improvement in respiratory symptoms may be related to improvement in respiratory conditions that are common in patients with GAD, such as asthma or chronic obstructive pulmonary disease; treating GAD may improve patient functioning even with the limitations of comorbid medical conditions.30 The lack of a statistically significant effect size in insomnia was unexpected and may be related to the permitted use of hypnotics for insomnia in the constituent studies.
Current pharmacotherapeutic options for the treatment of GAD may have differential effects on the psychic and somatic symptoms associated with GAD. For example, benzodiazepines, which facilitate γ-aminobutyric acid neurotransmission, are thought to have greater efficacy in treating somatic anxiety symptoms relative to psychic anxiety symptoms.31,32 Conversely, tricyclic antidepressants, azapirones (eg, buspirone), SSRIs, and SNRIs, all of which modulate monoamine neurotransmission, have been associated with greater reductions in psychic symptoms than somatic symptoms in several reports, although findings are not consistent throughout the literature.32–37 A meta-analysis investigating the efficacy of pregabalin,14 an antiepileptic drug licensed for treating GAD in Europe, found significant improvements relative to placebo across more anxiety symptoms with pregabalin 300–450 mg/d than with benzodiazepines (13 and 5 HARS items, respectively). Results from the present post hoc analyses indicate that mean decreases on most HARS item scores were significantly greater with vilazodone than placebo, suggesting efficacy in both psychic and somatic anxiety.
Perhaps of greater clinical relevance were the category shift analyses, which showed that among patients with moderate to very severe symptoms at baseline, the odds of achieving no symptoms after 8 weeks of treatment were significantly greater with vilazodone than with placebo on 4 psychic anxiety items (anxious mood, tension, intellectual, depressed mood) and 5 somatic anxiety items (somatic-muscular, somatic-sensory, cardiovascular symptoms, respiratory symptoms, and autonomic symptoms). These findings are noteworthy since excessive worry and anxiety are considered essential features of GAD, while somatic symptoms greatly contribute to disability in patients with GAD.29 In primary care settings, patients with GAD are more likely to present with a somatic symptom complaint than with anxiety,38 and many patients are unaware that physical symptoms can be indicative of a psychiatric disorder.39 Additionally, the level of somatic symptoms are similar in individuals with GAD regardless of the presence of a psychiatric comorbidity (eg, depression), suggesting that somatic symptoms are an inherent component of GAD itself.40 Therefore, choosing a treatment that is effective in both psychic and somatic anxiety symptom domains may be an important component of comprehensive treatment in GAD.
Despite its chronic nature and high level of recurrence, several follow-up studies41–44 have shown that full and partial recovery in GAD is possible in some patients and that chronicity is at least partly due to the high rate of symptom recurrence after remission. Results from 5-, 8- and 12year follow-up studies,41–43 all conducted in the same patient sample, have provided evidence pertaining to the chronicity of GAD in a psychiatric setting. After 5 years, 35% of men and 38% of women had full symptomatic remission, with 8-year rates of 46% and 56%, respectively.41 Over a 12-year period, 58% of patients in this sample had recovered, but the probability of recurrence in recovered patients was 45%.42 In a study of patients with GAD from a community sample, a 20-year follow-up showed that 16% of patients still had GAD, 29% had subthreshold syndromes or anxiety symptoms, and 52% were symptom-free, suggesting more improvement than progression within the anxiety spectrum.43 In patients with GAD in primary care, a 2-year follow-up44 found the probability of full and partial recovery were 0.39 and 0.54, respectively (Kaplan-Meier estimate); of the patients who fully recovered, 22% had partial recurrence of symptoms, 30% had full recurrence, and 48% experienced minimal symptom return, highlighting the persistence of GAD in many primary care patients.
In spite of its chronicity and the likelihood of recurrence, durable remission may be possible for some GAD patients, and measures that encourage treatment success should be implemented early in the disease course. Similar to the methods used in these post hoc analyses, deconstructing GAD into individual symptom components may be a clinically useful way to assess patient status at the end of acute treatment, potentially providing information that could enable a clinician to better manage subsyndromal or residual symptoms. Since GAD may remain at a subthreshold level even when it appears to be in remission,23 achieving meaningful reduction in symptom severity across psychic and somatic domains and symptom-free status after acute treatment may be an important step toward an extended recovery. It is important to note, however, that whereas residual symptoms have long been known to be an important factor in predicting recurrence in MDD,45,46 little is known about residual symptoms in GAD.47 Currently, predictors of response to treatment in GAD are largely based on clinical characteristics including severity of symptoms, shorter duration of illness, lack of comorbidities, and lower severity of psychosocial impairment.44,48,49
Limitations of these analyses included their pooled, post hoc nature, lack of an active comparator, and short treatment duration; results should be interpreted accordingly. In addition, the constituent studies were not powered to detect treatment differences in individual symptoms. Vilazodone treatment groups were pooled from 1 fixed-dose and 2 flexible-dose trials; therefore, no conclusions can be drawn regarding the effects of specific vilazodone doses on HARS total, subscale, or item scores. Conventional P values without adjustment for multiple comparisons were used, and although this is typical in post hoc analyses, random chance may have played a role in determining statistically significant differences. Patients with significant depressive symptoms were excluded from participation in the constituent studies, so the ability to generalize these post hoc findings to populations with comorbid depression and anxiety is limited.
CONCLUSIONS
In these post hoc analyses, acute treatment with vilazodone 20–40 mg/d demonstrated significant improvement across a wide range of symptoms associated with GAD. Statistically significant differences in favor of vilazodone versus placebo were noted on change in HARS total, subscale, and individual item scores. Rates of HARS response and remission were also significantly greater for vilazodone than for placebo. Moreover, a significantly higher percentage of vilazodone- than placebo-treated patients shifted from moderate or worse symptoms at baseline to no symptoms at week 8 on several HARS individual psychic and somatic anxiety items, an outcome that may be meaningful to clinicians and patients alike.
Submitted: November 4, 2015; accepted February 4, 2016.
Published online: April 28, 2016.
Drug names: alprazolam (Xanax, Niravam, and others), diazepam (Valium and others), duloxetine (Cymbalta), escitalopram (Lexapro and others), eszopiclone (Lunesta), hydroxyzene (Vistaril and others), lorazepam (Ativan and others), paroxetine (Paxil, Pexeva, and others), pregabalin (Lyrica), sertraline (Zoloft and others), vilazodone (Viibryd), zaleplon (Sonata and others), zolpidem (Ambien, Edluar, and others).
Potential conflicts of interest: Dr Khan is the medical director of the Northwest Clinical Research Center, which was an investigative site for the vilazodone phase 3 studies randomizing 241 patients; he has served as an uncompensated advisor to Forest Research Institute and is not compensated for his role as an author on scientific publications. Drs Durgam and Gommoll are employees of Forest Research Institute and stockholders in Allergan plc. Drs Tang and Mathews are former employees of Forest Research Institute and stockholders in Allergan plc. Dr Ruth is a former employee of Prescott Medical Communications, a contractor of Forest Research Institute.
Funding/support: Supported by funding from Forest Laboratories, LLC, an Allergan affiliate.
Role of the sponsor: Forest Laboratories was involved in the study design, data collection (via contracted clinical investigator sites), analysis and interpretation of data, and the decision to present these results.
Acknowledgment: Writing assistance and editorial support for the preparation of this manuscript were provided by Carol Brown, MS, of Prescott Medical Communications Group, Chicago, Illinois, with support from Forest Research Institute, an Allergan affiliate. Ms Brown reports no other conflicts of interest related to the subject of this article.
Previous presentations: The results reported in this article were previously presented at the Anxiety and Depression Association of America Annual Conference; April 9–12, 2015; Miami, Florida • College of Psychiatric and Neurologic Pharmacists Meeting; April 19–22, 2015; Tampa, Florida • American Psychiatric Association Annual Meeting; May 16–25, 2015; Chicago, Illinois • Annual Meeting of the American Society of Clinical Psychopharmacology; June 22–25, 2015; Miami, Florida • 28th Annual US Psychiatric and Mental Health Meeting; September 10–13, 2015; San Diego, California.
REFERENCES
1. Fricchione G. Clinical practice. generalized anxiety disorder. N Engl J Med. 2004;351(7):675–682. doi:10.1056/NEJMcp022342 PubMed
2. Mendlowicz MV, Stein MB. Quality of life in individuals with anxiety disorders. Am J Psychiatry. 2000;157(5):669–682. doi:10.1176/appi.ajp.157.5.669 PubMed
3. Bandelow B. Defining response and remission in anxiety disorders: toward an integrated approach. CNS Spectr. 2006;11(suppl 12):21–28. PubMed
4. Scholten WD, Batelaan NM, van Balkom AJ, et al. Recurrence of anxiety disorders and its predictors. J Affect Disord. 2013;147(1–3):180–185. doi:10.1016/j.jad.2012.10.031 PubMed
5. Davidson JR, Feltner DE, Dugar A. Management of generalized anxiety disorder in primary care: identifying the challenges and unmet needs. Prim Care Companion J Clin Psychiatry. 2010;12(2):PCC.09r00772. doi:10.4088/PCC.09r00772 PubMed
6. Wang PS, Lane M, Olfson M, et al. Twelve-month use of mental health services in the United States: results from the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):629–640. doi:10.1001/archpsyc.62.6.629 PubMed
7. Wittchen HU, Jacobi F. Size and burden of mental disorders in Europe—a critical review and appraisal of 27 studies. Eur Neuropsychopharmacol. 2005;15(4):357–376. doi:10.1016/j.euroneuro.2005.04.012 PubMed
8. Grant BF, Hasin DS, Stinson FS, et al. Co-occurrence of 12-month mood and anxiety disorders and personality disorders in the US: results from the national epidemiologic survey on alcohol and related conditions. J Psychiatr Res. 2005;39(1):1–9. doi:10.1016/j.jpsychires.2004.05.004 PubMed
9. Kessler RC, Aguilar-Gaxiola S, Alonso J, et al. The global burden of mental disorders: an update from the WHO World Mental Health (WMH) surveys. Epidemiol Psichiatr Soc. 2009;18(1):23–33. doi:10.1017/S1121189X00001421 PubMed
10. Hoffman DL, Dukes EM, Wittchen HU. Human and economic burden of generalized anxiety disorder. Depress Anxiety. 2008;25(1):72–90. doi:10.1002/da.20257 PubMed
11. Revicki DA, Travers K, Wyrwich KW, et al. Humanistic and economic burden of generalized anxiety disorder in North America and Europe. J Affect Disord. 2012;140(2):103–112. doi:10.1016/j.jad.2011.11.014 PubMed
12. Baldwin DS, Waldman S, Allgulander C. Evidence-based pharmacological treatment of generalized anxiety disorder. Int J Neuropsychopharmacol. 2011;14(5):697–710. doi:10.1017/S1461145710001434 PubMed
13. Bandelow B, Sher L, Bunevicius R, et al. WFSBP Task Force on Anxiety Disorders, OCD and PTSD. Guidelines for the pharmacological treatment of anxiety disorders, obsessive-compulsive disorder and posttraumatic stress disorder in primary care. Int J Psychiatry Clin Pract. 2012;16(2):77–84. doi:10.3109/13651501.2012.667114 PubMed
14. Lydiard RB, Rickels K, Herman B, et al. Comparative efficacy of pregabalin and benzodiazepines in treating the psychic and somatic symptoms of generalized anxiety disorder. Int J Neuropsychopharmacol. 2010;13(2):229–241. doi:10.1017/S1461145709990460 PubMed
15. Croft HA, Pomara N, Gommoll C, et al. Efficacy and safety of vilazodone in major depressive disorder: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2014;75(11):e1291–e1298. doi:10.4088/JCP.14m08992 PubMed
16. Khan A, Cutler AJ, Kajdasz DK, et al. A randomized, double-blind, placebo-controlled, 8-week study of vilazodone, a serotonergic agent for the treatment of major depressive disorder. J Clin Psychiatry. 2011;72(4):441–447. doi:10.4088/JCP.10m06596 PubMed
17. Rickels K, Athanasiou M, Robinson DS, et al. Evidence for efficacy and tolerability of vilazodone in the treatment of major depressive disorder: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2009;70(3):326–333. doi:10.4088/JCP.08m04637 PubMed
18. Mathews M, Gommoll C, Chen D, et al. Efficacy and safety of vilazodone 20 and 40 mg in major depressive disorder: a randomized, double-blind, placebo-controlled trial. Int Clin Psychopharmacol. 2015;30(2):67–74. doi:10.1097/YIC.0000000000000057 PubMed
19. Thase ME, Chen D, Edwards J, et al. Efficacy of vilazodone on anxiety symptoms in patients with major depressive disorder. Int Clin Psychopharmacol. 2014;29(6):351–356. doi:10.1097/YIC.0000000000000045 PubMed
20. Gommoll C, Durgam S, Mathews M, et al. A double-blind, randomized, placebo-controlled, fixed-dose phase III study of vilazodone in patients with generalized anxiety disorder. Depress Anxiety. 2015;32(6):451–459. doi:10.1002/da.22365 PubMed
21. Gommoll C, Forero G, Mathews M, et al. Vilazodone in patients with generalized anxiety disorder: a double-blind, randomized, placebo-controlled, flexible-dose study. Int Clin Psychopharmacol. 2015;30(6):297–306. doi:10.1097/YIC.0000000000000096 PubMed
22. Durgam S, Gommoll C, Forero G, et al. Efficacy and safety of vilazodone in patients with generalized anxiety disorder: a randomized, double-blind, placebo-controlled, flexible-dose trial. J Clin Psychiatry. In Press.
23. Newman MG, Llera SJ, Erickson TM, et al. Worry and generalized anxiety disorder: a review and theoretical synthesis of evidence on nature, etiology, mechanisms, and treatment. Annu Rev Clin Psychol. 2013;9(1):275–297. doi:10.1146/annurev-clinpsy-050212-185544 PubMed
24. Hamilton M. The assessment of anxiety states by rating. Br J Med Psychol. 1959;32(1):50–55. doi:10.1111/j.2044-8341.1959.tb00467.x PubMed
25. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
26. Guy W. The Clinician Global Severity and Impression scales. ECDEU Assessment Manual for Psychopharmacology. Rockville, MD: National Institute of Mental Health; 218–222. DHEW Publication No 76–338. 1976.
27. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56–62. doi:10.1136/jnnp.23.1.56 PubMed
28. Hidalgo RB, Tupler LA, Davidson JR. An effect-size analysis of pharmacologic treatments for generalized anxiety disorder. J Psychopharmacol. 2007;21(8):864–872. doi:10.1177/0269881107076996 PubMed
29. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Fifth Edition. Arlington, VA: American Psychiatric Association; 2013.
30. Culpepper L. Generalized anxiety disorder and medical illness. J Clin Psychiatry. 2009;70
(suppl 2):20–24. doi:10.4088/JCP.s.7002.04 PubMed
31. Rickels K, Weisman K, Norstad N, et al. Buspirone and diazepam in anxiety: a controlled study. J Clin Psychiatry.
1982;43(12 pt 2):81–86. PubMed
32. Rickels K, Zaninelli R, McCafferty J, et al. Paroxetine treatment of generalized anxiety disorder: a double-blind, placebo-controlled study. Am J Psychiatry. 2003;160(4):749–756. doi:10.1176/appi.ajp.160.4.749 PubMed
33. Feighner JP, Cohn JB. Analysis of individual symptoms in generalized anxiety—a pooled, multistudy, double-blind evaluation of buspirone. Neuropsychobiology. 1989;21(3):124–130. doi:10.1159/000118565 PubMed
34. Katz IR, Reynolds CF 3rd, Alexopoulos GS, et al. Venlafaxine ER as a treatment for generalized anxiety disorder in older adults: pooled analysis of five randomized placebo-controlled clinical trials. J Am Geriatr Soc. 2002;50(1):18–25. doi:10.1046/j.1532-5415.2002.50003.x PubMed
35. Meoni P, Hackett D, Lader M. Pooled analysis of venlafaxine XR efficacy on somatic and psychic symptoms of anxiety in patients with generalized anxiety disorder. Depress Anxiety. 2004;19(2):127–132. doi:10.1002/da.10141 PubMed
36. Pollack MH, Zaninelli R, Goddard A, et al. Paroxetine in the treatment of generalized anxiety disorder: results of a placebo-controlled, flexible-dosage trial. J Clin Psychiatry. 2001;62(5):350–357. doi:10.4088/JCP.v62n0508 PubMed
37. Rickels K, Pollack MH, Sheehan DV, et al. Efficacy of extended-release venlafaxine in nondepressed outpatients with generalized anxiety disorder. Am J Psychiatry. 2000;157(6):968–974. doi:10.1176/appi.ajp.157.6.968 PubMed
38. Wittchen HU, Kessler RC, Beesdo K, et al. Generalized anxiety and depression in primary care: prevalence, recognition, and management. J Clin Psychiatry.
2002;63(suppl 8):24–34. PubMed
39. Kessler RC, Keller MB, Wittchen HU. The epidemiology of generalized anxiety disorder. Psychiatr Clin North Am. 2001;24(1):19–39. doi:10.1016/S0193-953X(05)70204-5 PubMed
40. Brawman-Mintzer O, Lydiard RB, Crawford MM, et al. Somatic symptoms in generalized anxiety disorder with and without comorbid psychiatric disorders. Am J Psychiatry. 1994;151(6):930–932. doi:10.1176/ajp.151.6.930 PubMed
41. Yonkers KA, Bruce SE, Dyck IR, et al. Chronicity, relapse, and illness—course of panic disorder, social phobia, and generalized anxiety disorder: findings in men and women from 8 years of follow-up. Depress Anxiety. 2003;17(3):173–179. doi:10.1002/da.10106 PubMed
42. Bruce SE, Yonkers KA, Otto MW, et al. Influence of psychiatric comorbidity on recovery and recurrence in generalized anxiety disorder, social phobia, and panic disorder: a 12-year prospective study. Am J Psychiatry. 2005;162(6):1179–1187. doi:10.1176/appi.ajp.162.6.1179 PubMed
43. Angst J, Gamma A, Baldwin DS, et al. The generalized anxiety
spectrum: prevalence, onset, course and outcome. Eur Arch Psychiatry Clin Neurosci. 2009;259(1):37–45. doi:10.1007/s00406-008-0832-9 PubMed
44. Rodriguez BF, Weisberg RB, Pagano ME, et al. Characteristics
and predictors of full and partial recovery from generalized anxiety disorder in primary care patients. J Nerv Ment Dis. 2006;194(2):
91–97. doi:10.1097/01.nmd.0000198140.02154.32 PubMed
45. Fava GA. Subclinical symptoms in mood disorders: pathophysiological and therapeutic implications. Psychol Med. 1999;29(1):47–61. doi:10.1017/S0033291798007429 PubMed
46. Paykel ES. Remission and residual symptomatology in major depression. Psychopathology. 1998;31(1):5–14. doi:10.1159/000029018 PubMed
47. Lecrubier Y, Dolberg OT, Andersen HF, et al. Qualitative changes in symptomatology as an effect of treatment with escitalopram in generalized anxiety disorder and major depressive disorder. Eur Arch Psychiatry Clin Neurosci. 2008;258(3):171–178. doi:10.1007/s00406-007-0771-x PubMed
48. Pollack MH, Meoni P, Otto MW, et al. Predictors of outcome following venlafaxine extended-release treatment of DSM-IV generalized anxiety disorder: a pooled analysis of short- and long-term studies. J Clin Psychopharmacol. 2003;23(3):250–259. doi:10.1097/01.jcp.0000084025.22282.84 PubMed
49. Simon NM, Zalta AK, Worthington JJ 3rd, et al. Preliminary support for gender differences in response to fluoxetine for generalized anxiety disorder. Depress Anxiety. 2006;23(6):373–376. doi:10.1002/da.20184 PubMed
This PDF is free for all visitors!


