Objective: To evaluate the efficacy and tolerability of a once-monthly intramuscular (IM) depot formulation of the dopamine partial agonist aripiprazole as maintenance treatment in adults meeting DSM-IV-TR schizophrenia criteria.
Method: The study was conducted from July 2008 until February 2011. Subjects requiring chronic treatment with an antipsychotic entered a 4- to 12-week oral stabilization phase and received oral aripiprazole (10-30 mg/d). Subjects meeting stability criteria for 4 weeks entered an IM-depot stabilization phase in which they received 400-mg aripiprazole-IM-depot injections every 4 weeks (single decrease to 300 mg permitted) with coadministration of oral aripiprazole tablets in the first 2 weeks. Subjects meeting stability criteria for 12 consecutive weeks were randomly assigned (2:1) to aripiprazole-IM-depot or placebo during a 52-week, double-blind maintenance phase. The primary outcome measure was time to exacerbation of psychotic symptoms/impending relapse (event). Safety and tolerability were also assessed.
Results: 710 patients entered oral stabilization, 576 progressed to IM-depot stabilization, and 403 were randomly assigned to double-blind treatment. The study was terminated early because efficacy was demonstrated by the preplanned interim analysis (conducted after 64 events). Time to impending relapse was significantly delayed with aripiprazole-IM-depot treatment compared with placebo in both the interim analysis and the final analysis (P < .0001, log-rank test). The hazard ratio (placebo/aripiprazole-IM-depot) at final analysis was 5.03 (95% CI, 3.15-8.02). The rate of impending relapse was significantly lower with aripiprazole-IM-depot than placebo at endpoint (final analysis, 10.0% [n = 27/269] vs 39.6% [n = 53/134]). Improvements in Clinical Global Impressions-Severity of Illness scale and Positive and Negative Syndrome Scale total scores were maintained with aripiprazole-IM-depot treatment but showed significant worsening with placebo (change from double-blind baseline, P < .0001 for aripiprazole-IM-depot vs placebo). The most common treatment-emergent adverse events (occurring in ≥ 5% of aripiprazole-IM-depot subjects and greater than placebo) were insomnia, tremor, and headache.
Conclusions: Aripiprazole-IM-depot significantly delayed time to impending relapse compared with placebo and appears to be a well-tolerated maintenance treatment option for schizophrenia.
Trial Registration: ClinicalTrials.gov identifier: NCT00705783
J Clin Psychiatry 2012;73(5):617-624
© Copyright 2012 Physicians Postgraduate Press, Inc.
Submitted: November 10, 2011; accepted February 14, 2012(doi:10.4088/JCP.11m07530).
Corresponding author: John M. Kane, MD, The Zucker Hillside Hospital, 75-59 263rd St, Kaufmann Bldg, Ste 103, Glen Oaks, NY 11004-1100 ([email protected]).
Aripiprazole Intramuscular Depot as Maintenance Treatment in Patients With Schizophrenia: A 52-Week, Multicenter, Randomized, Double-Blind, Placebo-Controlled Study
ABSTRACT
Objective: To evaluate the efficacy and tolerability of a once-monthly intramuscular (IM) depot formulation of the dopamine partial agonist aripiprazole as maintenance treatment in adults meeting DSM-IV-TR schizophrenia criteria.
Method: The study was conducted from July 2008 until February 2011. Subjects requiring chronic treatment with an antipsychotic entered a 4- to 12-week oral stabilization phase and received oral aripiprazole (10–30 mg/d). Subjects meeting stability criteria for 4 weeks entered an IM-depot stabilization phase in which they received 400-mg aripiprazole-IM-depot injections every 4 weeks (single decrease to 300 mg permitted) with coadministration of oral aripiprazole tablets in the first 2 weeks. Subjects meeting stability criteria for 12 consecutive weeks were randomly assigned (2:1) to aripiprazole-IM-depot or placebo during a 52-week, double-blind maintenance phase. The primary outcome measure was time to exacerbation of psychotic symptoms/impending relapse (event). Safety and tolerability were also assessed.
Results: 710 patients entered oral stabilization, 576 progressed to IM-depot stabilization, and 403 were randomly assigned to double-blind treatment. The study was terminated early because efficacy was demonstrated by the preplanned interim analysis (conducted after 64 events). Time to impending relapse was significantly delayed with aripiprazole-IM-depot treatment compared with placebo in both the interim analysis and the final analysis (P < .0001, log-rank test). The hazard ratio (placebo/aripiprazole-IM-depot) at final analysis was 5.03 (95% CI, 3.15–8.02). The rate of impending relapse was significantly lower with aripiprazole-IM-depot than placebo at endpoint (final analysis, 10.0% [n = 27/269] vs 39.6% [n = 53/134]). Improvements in Clinical Global Impressions-Severity of Illness scale and Positive and Negative Syndrome Scale total scores were maintained with aripiprazole-IM-depot treatment but showed significant worsening with placebo (change from double-blind baseline, P < .0001 for aripiprazole-IM-depot vs placebo). The most common treatment-emergent adverse events (occurring
in ≥ 5% of aripiprazole-IM-depot subjects and greater than placebo) were insomnia, tremor, and headache.
Conclusions: Aripiprazole-IM-depot significantly delayed time to impending relapse compared with placebo and appears to be a well-tolerated maintenance treatment option for schizophrenia.
Trial Registration: ClinicalTrials.gov identifier: NCT00705783
J Clin Psychiatry 2012;73(5):617–624
© Copyright 2012 Physicians Postgraduate Press, Inc.
Submitted: November 10, 2011; accepted February 14, 2012
(doi:10.4088/JCP.11m07530).
Corresponding author: John M. Kane, MD, The Zucker Hillside Hospital, 75-59 263rd St, Kaufmann Bldg, Ste 103, Glen Oaks, NY 11004-1100 ([email protected]).
Schizophrenia is a chronic condition, and maintenance treatment with antipsychotics is a key feature of long-term management. Medication adherence, a factor substantially overestimated by both patients and physicians, is an important element in treatment success.1,2 Rates of nonadherence to antipsychotic medication are high and are associated with a significantly increased risk of relapse and hospitalization,3 resulting in high health care costs—in part, due to the increased costs associated with hospitalization.4 Nonadherence also represents a barrier to patient recovery.5,6 In addition, discontinuation of medication is the largest predictor of relapse risk in schizophrenia.7,8 Long-acting injectable antipsychotics relieve patients from the daily need to take medication and subsequently have the potential to improve adherence.8 Furthermore, failure to attend an injection visit serves as a signal for treatment nonadherence.9
Aripiprazole has a unique pharmacology with partial agonist activity at dopamine D2/D3 receptors, associated with a low risk of hyperprolactinemia, and partial agonist activity at 5-HT1A receptors10,11 and antagonist activity at 5-HT2A receptors.12,13 Oral aripiprazole has demonstrated a low potential for metabolic disturbances,14,15 an important consideration given the high rates of medical morbidity and mortality in schizophrenia.16 The long-term efficacy and safety profile of oral aripiprazole thus suggests that a long-acting injectable formulation of aripiprazole would be a suitable long-term treatment option in schizophrenia. Aripiprazole depot is a lyophilized powder of aripiprazole; the aripiprazole molecule is unmodified. Following reconstitution with water, the depot formulation is administered as a suspension into the gluteal muscle. Aripiprazole is then absorbed gradually from the muscle over time, resulting in plasma levels that are consistent with those of the oral product.17 The safety and tolerability of aripiprazole-IM-depot when transitioning from oral aripiprazole have been previously demonstrated.18 Furthermore, intramuscular injections resulted in sustained concentrations of aripiprazole for more than 1 month, with the achieved steady-state concentrations being comparable to those of the once-daily oral aripiprazole formulation.18
Here, we report the efficacy and safety of maintenance therapy with an IM-depot formulation of aripiprazole from a 52-week, randomized, double-blind, placebo-controlled study in patients with schizophrenia. The primary outcome measure was time to impending relapse (clinical worsening, psychiatric hospitalization, increased risk of suicide, or violent behavior) in subjects who were stabilized on treatment with aripiprazole-IM-depot for at least 12 weeks and then randomly assigned to either aripiprazole-IM-depot
or placebo.
METHOD
Patients
Patients were individuals 18–60 years of age requiring chronic antipsychotic treatment who could understand protocol requirements and provide informed written consent. Enrolled subjects had a diagnosis of schizophrenia, as defined by the Diagnostic and Statistical Manual of Mental Health Disorders, Fourth Edition, Text Revision (DSM-IV-TR) criteria, for at least 3 years (to exclude first-episode patients) and a history of symptom exacerbation or relapse when not receiving antipsychotic treatment.
Key exclusion criteria included a DSM-IV-TR diagnosis other than schizophrenia, any clinically significant medical or neurologic disorder, and any medically significant abnormal laboratory test or electrocardiogram (ECG) results at screening. Subjects considered refractory to antipsychotic treatment by history or responsive to clozapine treatment were excluded. In accordance with the Declaration of Helsinki, the ethics committee at each site approved the protocol. The study is registered with ClinicalTrials.gov (identifier: NCT00705783).
Study Design
This multicenter study (108 centers in the United States, Mexico, Argentina, Bulgaria, Romania, Serbia, Slovakia, Russia, India, Taiwan, Malaysia, and the Philippines) was conducted from July 2008 until February 2011. The study consisted of a screening phase and 4 treatment phases. Eligibility was determined during the screening phase (2–42 days). In the oral conversion phase (phase 1, 4–6 weeks), subjects not already on aripiprazole monotherapy were cross-titrated during weekly visits from other antipsychotic(s) to oral aripiprazole monotherapy. Aripiprazole was initiated at 5 mg/d for the first week, then increased to 10 mg/d in the second week, while the same dose of the previous antipsychotic was maintained; in weeks 3 and 4, the dose of the previous antipsychotic was tapered down, and the antipsychotic was discontinued at the end of week 4, 5, or 6. In the oral stabilization phase (phase 2, 4–12 weeks), subjects were assessed biweekly and stabilized on treatment with oral aripiprazole (10–30 mg/d). Stability was defined as meeting all of the following criteria for 4 consecutive weeks (2 consecutive visits 2 weeks apart):
- Outpatient status
- Positive and Negative Syndrome Scale (PANSS)19 total score ≤ 80
- Lack of specific psychotic symptoms on the PANSS, as measured by a score of ≤ 4 on each of the following items: conceptual disorganization, suspiciousness, hallucinatory behavior, unusual thought content
- Clinical Global Impressions-Severity of Illness
(CGI-S)20 score ≤ 4 (moderately ill) - Clinical Global Impression for Severity of Suicidality (CGI-SS)21 score ≤ 2 (mildly suicidal) on part 1 and ≤ 5 (minimally worsened) on part 2
Patients meeting all of the stability criteria in the oral stabilization phase were assigned to single-blind aripiprazole-IM-depot (400 mg) in the IM-depot stabilization phase (phase 3, 12–36 weeks). Oral aripiprazole (10–20 mg/d) was continued for the first 2 weeks following the initial IM-depot dose to maintain therapeutic plasma concentrations of antipsychotic medication and to ensure a smooth transition between the oral and the extended-release formulation. During IM-depot stabilization, patients could have a single decrease to aripiprazole 300 mg based on tolerability and could have their dose increased back to 400 mg for symptomatic control, if needed. Subjects who met stability criteria (as defined above) on single-blind aripiprazole-IM-depot for 12 consecutive weeks were randomly assigned 2:1 to maintenance treatment (phase 4, up to 52 weeks) with aripiprazole-IM-depot at the dose they were receiving (400 or 300 mg) or placebo-IM-depot. Aripiprazole-IM-depot and placebo-IM-depot were administered every 4 weeks and injected into the gluteal muscle in an alternating fashion.
Antidepressants and mood stabilizers were prohibited throughout the study and were washed out during screening, as appropriate. Antipsychotics other than aripiprazole were prohibited throughout the study; subjects receiving other antipsychotics were tapered off during the conversion phase. Benzodiazepines (maximum 6 mg/d) and anticholinergics (≤ 4 mg/d benztropine or equivalent) were permitted, although not within 8 hours or 12 hours, respectively, of rating scale assessments.
Assessments
The primary outcome measure was time to exacerbation of psychotic symptoms/impending relapse, defined as
meeting any or all of the following 4 criteria at any time during phase 4.
- Clinical worsening as defined by CGI-Improvement score of ≥ 5 and an increase on any of 4 individual PANSS items (conceptual disorganization, hallucinatory behavior, suspiciousness, or unusual thought content) to a score > 4 with an absolute increase of ≥ 2 on that specific item since randomization or an increase > 4 on these PANSS items and an absolute increase of ≥ 4 on the combined score
of these items since randomization - Hospitalization due to worsening of psychotic symptoms
- Risk of suicide as defined by CGI-SS score of 4 (severely suicidal) or 5 (attempted suicide) on part 1 or a score of 6 (much worse) or 7 (very much worse) on part 2
- Violent behavior resulting in clinically significant self-injury, injury to another person, or property damage
The key secondary endpoint was the proportion of subjects meeting impending relapse criteria in phase 4. Other secondary efficacy assessments included mean change from double-blind baseline to endpoint in PANSS total score and mean change from baseline to endpoint in CGI-S score.
Safety was assessed by adverse event (AE) reporting, clinical laboratory tests (hematology and fasting clinical chemistry), urinalysis, 12-lead ECG, vital signs, injection-site evaluation, and physical examination. In addition, body weight and serum prolactin concentrations were monitored. The following scales were used to assess extrapyramidal symptoms (EPS): Abnormal Involuntary Movement
Scale (AIMS),22 Simpson-Angus Scale (SAS),23 and Barnes
Akathisia Rating Scale.24 Intensity of injection pain was assessed using a visual analog scale (VAS; 0 = no pain to 100 = unbearably painful).
Interim Analysis and Conditions for Early Termination
The study included 2 prespecified interim analyses conducted by an Independent Data Monitoring Committee (IDMC) to assess efficacy, performed at approximately 50% and 75% of impending relapses for all randomized subjects. On the basis of advice from regulatory authorities, results needed to demonstrate statistically significant effects in a series of 4 sensitivity analyses. The IDMC could discontinue the study for ethical reasons, most notably continued exposure to placebo, if efficacy was established (at a significance level of .001) at the preplanned first interim analysis
(64 events).
Statistical Analyses
Time to impending relapse from randomization date was plotted on Kaplan-Meier curves and compared using a log-rank test. The hazard ratio (HR) (placebo/aripiprazole-IM-depot) and its 95% confidence intervals (CIs) were derived from the Cox proportional hazard model, with treatment as factor in the model.
Changes in PANSS total score, CGI-S score, and EPS rating scale scores were analyzed using an analysis of covariance (ANCOVA) model, with terms for treatment and baseline value as a covariate for both last-observation-carried-forward (LOCF) and observed-case (OC) datasets. Time to discontinuation due to all reasons other than termination of the study by the sponsor was also plotted on Kaplan-Meier curves and compared using the log-rank test. Changes in weight were analyzed using ANCOVA, with terms for treatment and baseline value as a covariate. Incidences of weight gain ≥ 7% were compared using the χ2 test. Other safety endpoints were summarized using descriptive statistics.
RESULTS
Patient Disposition and Characteristics
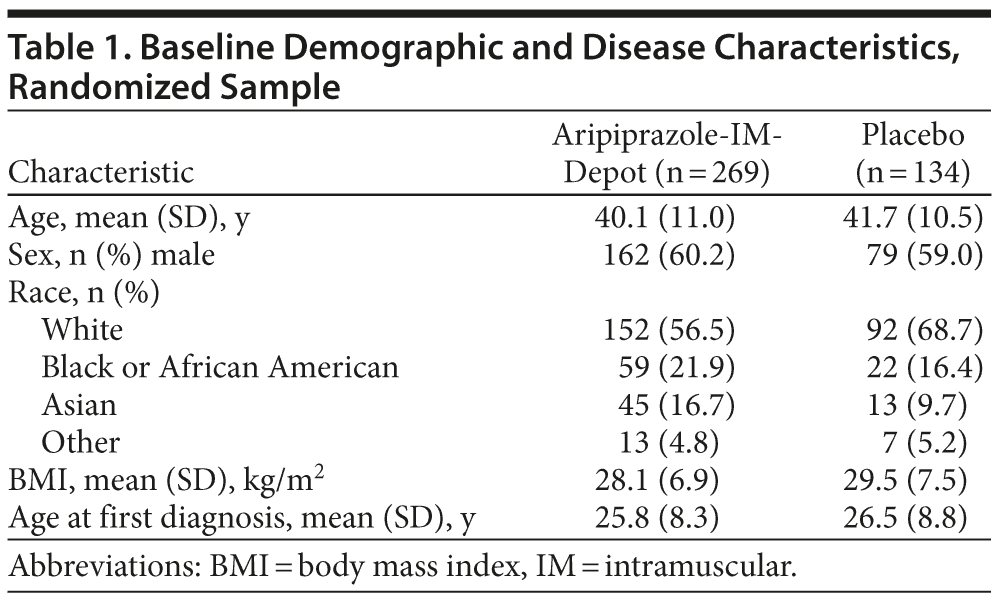
Of 1,025 screened patients, 843 were enrolled in the study, and 403 patients were randomly assigned to double-blind treatment in phase 4 (aripiprazole-IM-depot, n = 269; placebo, n = 134; Figure 1). On the basis of the results of the preplanned interim analysis conducted after 64 relapse events, the IDMC determined that the primary endpoint had been achieved and that there were no safety issues of particular concern. Thus, the study was terminated early to avoid continued exposure to placebo, and all patients were brought in for a final visit. As such, the main reason for discontinuation from all study phases was early study termination: 237 patients (58.8%) discontinued during double-blind treatment due to this early termination. Baseline demographic and disease characteristics of randomized subjects were similar between treatment groups (Table 1).
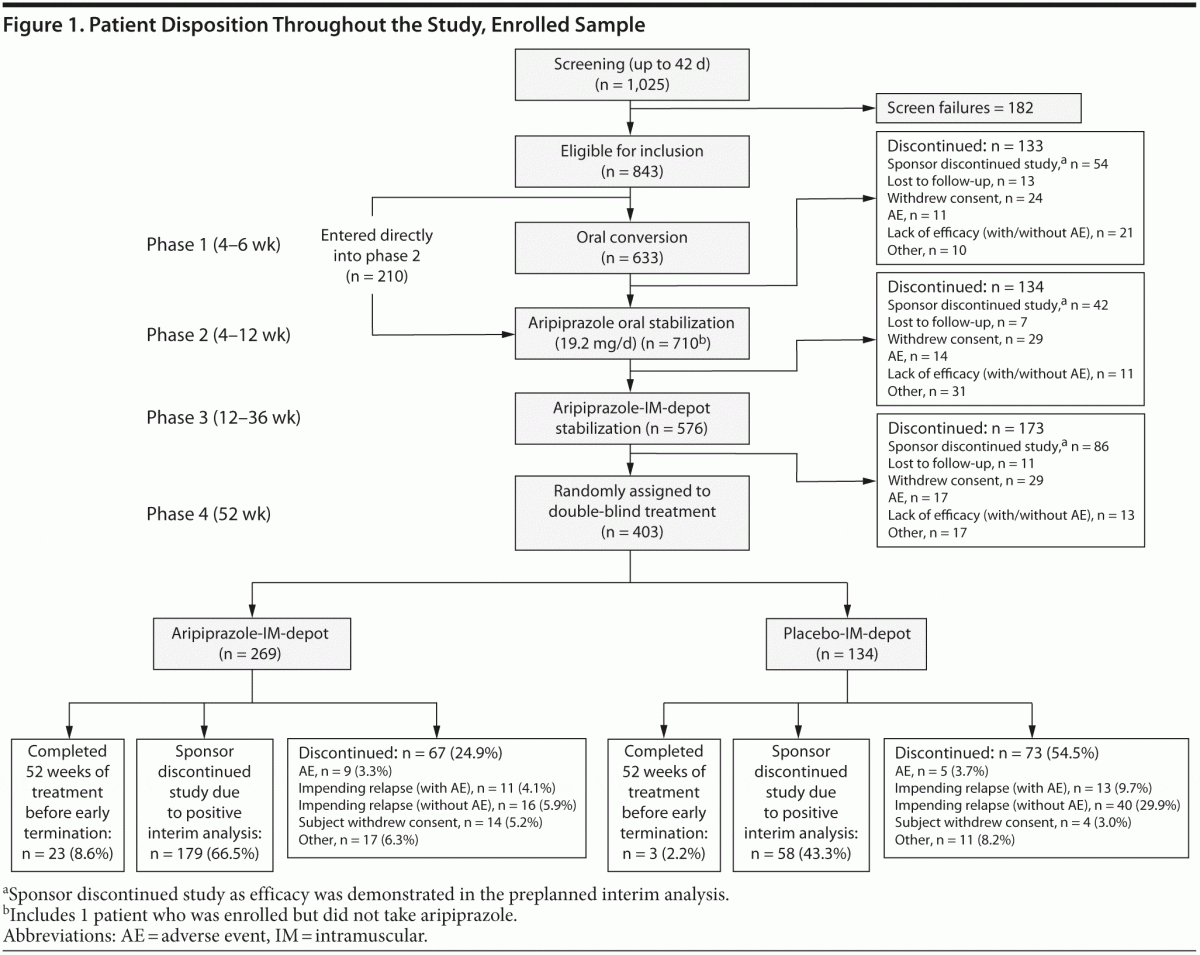
Treatment Exposure
Of the patients who received aripiprazole-IM-depot (n = 576), 33.7% (n = 194) received at least 7 injections (6 months) and 8.9% (n = 51) received at least 13 injections (12 months) of aripiprazole-IM-depot during phases 3 and 4. The mean dose of oral aripiprazole during the oral stabilization phase was 19.2 mg/d in enrolled (n = 709) and randomized patients (n = 403). During the aripiprazole-IM-depot stabilization phase, 88.6% of randomized patients (ie, those who would receive aripiprazole-IM-depot in the double-blind phase) received IM-depot 400 mg. Of those randomized to aripiprazole-IM-depot, 96.3% started on and continued to receive 400 mg with no change in dose.
Efficacy
Time to impending relapse was significantly delayed with aripiprazole-IM-depot compared with placebo in the final analysis (Figure 2A). Relapse rates were also significantly lower with aripiprazole-IM-depot than placebo at the final analysis time point (80 events; 10.0% [n = 27/269] vs 39.6% [n = 53/134]; HR = 5.03; 95% CI, 3.15–8.02). Similar results were observed at the interim analysis, with a significantly longer time to impending relapse with aripiprazole-IM-depot compared with placebo (log-rank test, P < .0001) and significantly lower relapse rates with aripiprazole-IM-depot (9.6%; n = 22/230) than placebo (36.8%; n = 42/114; HR = 4.72, 95% CI, 2.81–7.94). In the final analysis, reasons for relapse were as follows (aripiprazole-IM-depot vs placebo): clinical worsening according to CGI/PANSS, 74.1% (n = 20/27) vs 86.8% (n = 46/53); hospitalization, 25.9% (n = 7/27) vs 9.4% (n = 5/53); suicide risk, 3.7% (n = 1/27) vs 1.9% (n = 1/53); and violent behavior, 3.7% (n = 1/27) vs 7.5% (n = 4/53).
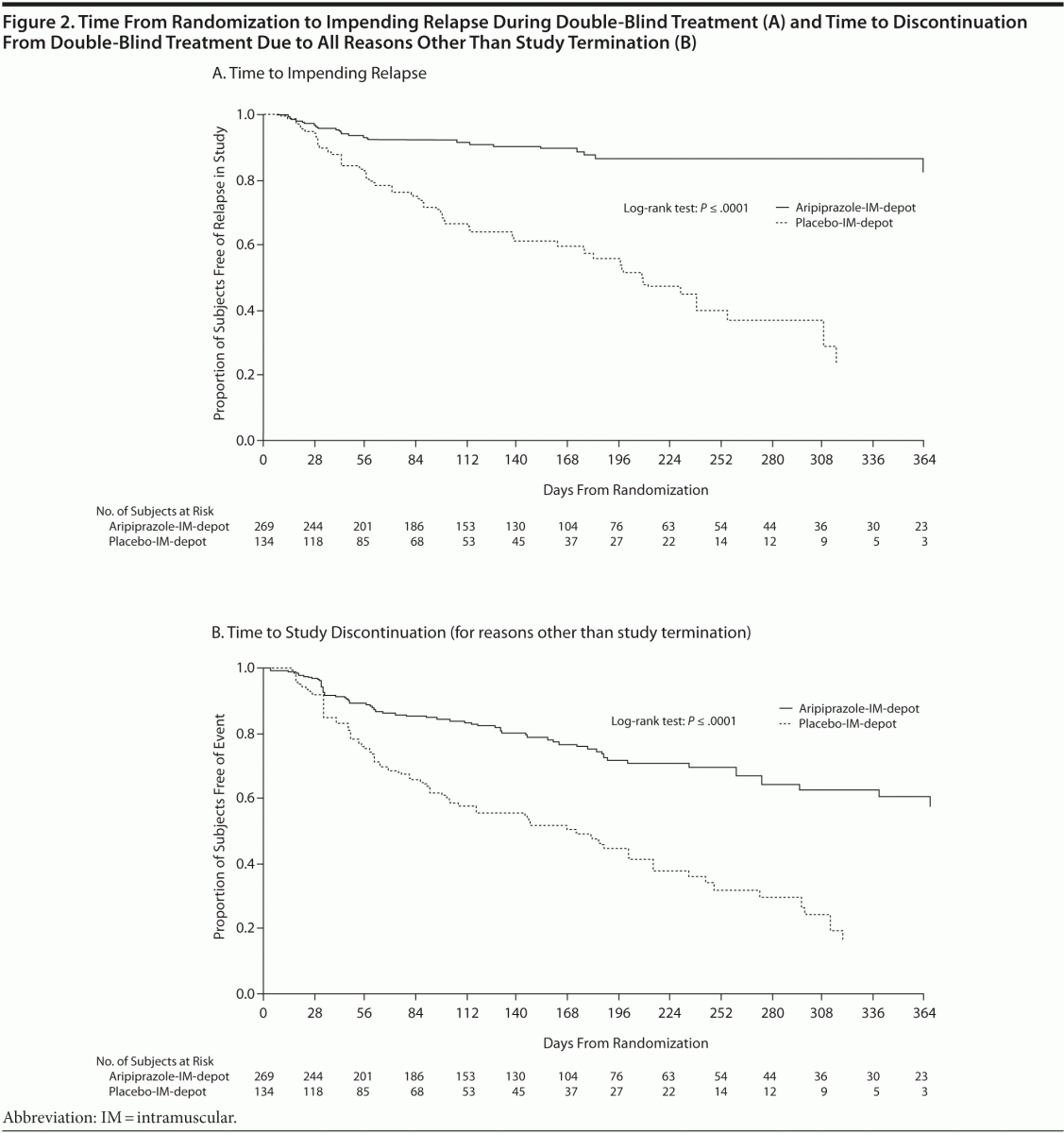
Overall time to study discontinuation from double-blind treatment due to all reasons other than termination of the study by the sponsor was significantly delayed with aripiprazole-IM-depot compared with placebo (Figure 2B), with discontinuation rates of 24.9% vs 54.5%, respectively.
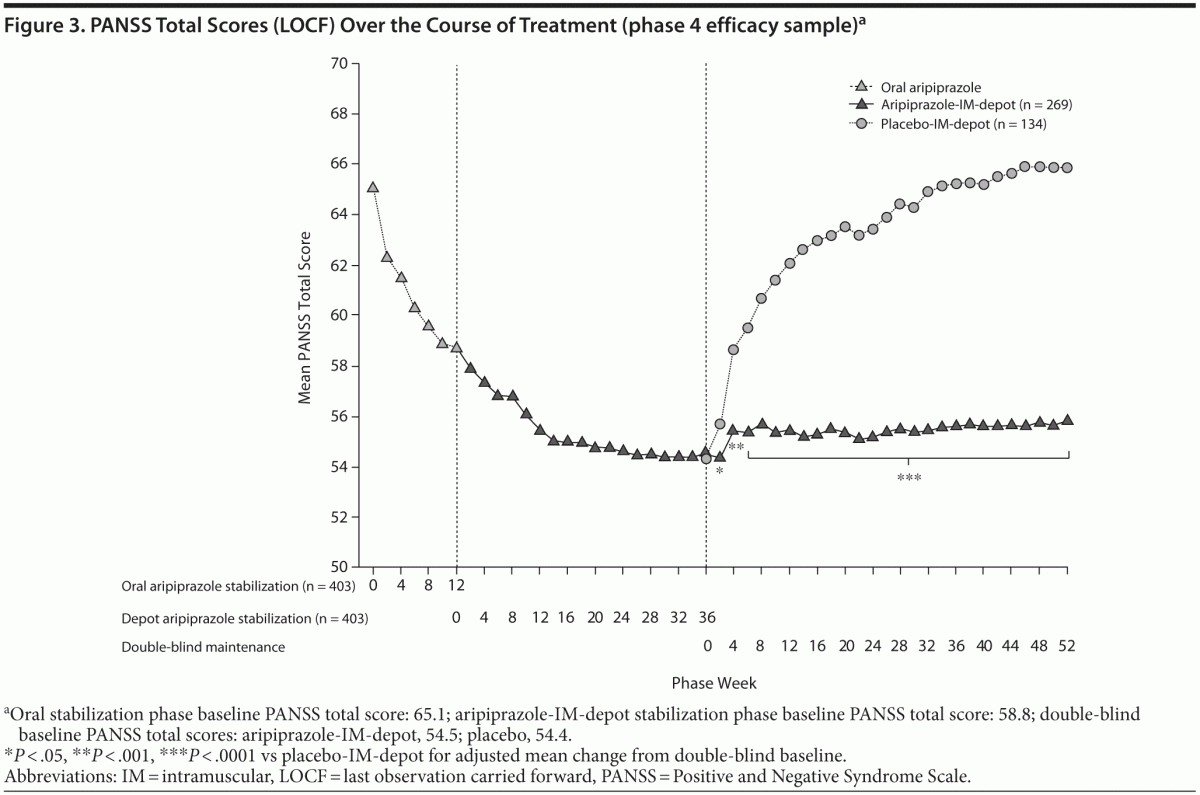
Mean PANSS total scores improved during the oral and aripiprazole-IM-depot stabilization phases. There were significant mean increases from double-blind baseline for those randomly assigned to placebo (+11.6) vs those patients randomly assigned to aripiprazole-IM-depot (+1.4; P < .0001; Figure 3). Significant differences in mean PANSS total score between aripiprazole-IM-depot and placebo were observed as early as week 2 and were consistently observed at all subsequent time points. Similar results were observed for CGI-S scores. In the randomized patient sample, CGI-S scores decreased from the oral stabilization phase baseline (3.4) through to the end of oral stabilization (3.1) and showed further improvement in the IM-depot stabilization phase (2.9). The mean change in CGI-S (LOCF) score during double-blind treatment was statistically significantly in favor of aripiprazole at week 52 (0.1 vs 0.7; P < .0001) and at every assessment from week 4 onward.
Safety and Tolerability
Treatment-emergent AEs during phases 3 and 4 are shown in Table 2. During double-blind treatment, the most common treatment-emergent AEs (occurring in ≥ 5% of aripiprazole-IM-depot subjects and greater than placebo) were insomnia, headache, and tremor.
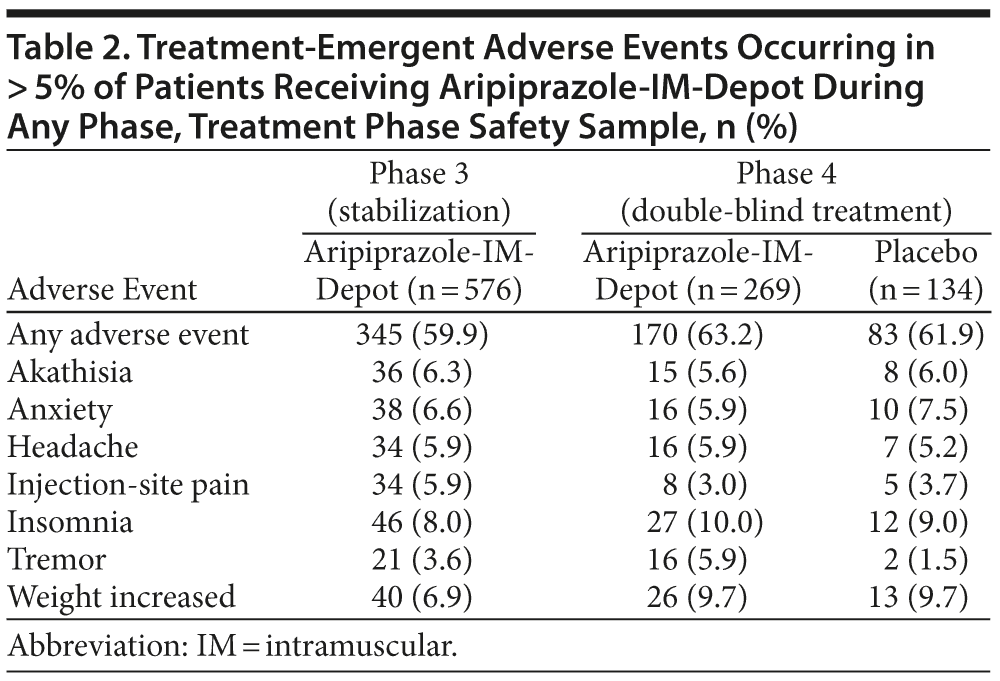
Serious treatment-emergent AEs occurred in 1.4% and 4.3% of patients during oral and IM-depot stabilization, respectively. In the double-blind treatment phase, serious AEs were reported by 4.1% of aripiprazole-IM-depot patients and 6.7% of placebo patients. The only serious AEs reported by > 1% of patients in either group were psychotic disorder (1.5% in aripiprazole-IM-depot vs 3.0% placebo patients) and schizophrenia (0.7% aripiprazole-IM-depot patients vs 1.5% placebo). Other serious AEs reported in the randomized phase were generally resolved without medical consequence.
Overall discontinuations due to treatment-emergent AEs were low, occurring in 3.0% (n = 21/709) and 4.9% (n = 28/576) of patients (safety sample) during oral and IM-depot stabilization, respectively. During double-blind treatment, 7.1% (n = 19/269) of aripiprazole-IM-depot and 13.4% (n = 18/134) of placebo patients discontinued due to treatment-emergent AEs. Two deaths were reported during the study: 1 during IM-depot stabilization (coronary artery insufficiency) and 1 patient receiving aripiprazole-IM-depot (pancreatic carcinoma) during double-blind treatment. Both events were considered to be unrelated to study treatment by the respective investigators. Four suicide-related AEs were reported during double-blind treatment. Three (1.1%) aripiprazole-IM-depot patients experienced suicidal ideation, and 1 (0.4%) aripiprazole-IM-depot patient attempted suicide.
Injections of aripiprazole-IM-depot were generally well tolerated (see Table 2, injection-site pain). Mean intensity of injection pain, measured with a 100-point VAS, was minimal during depot stabilization; reductions were observed from the first (6.1) to the last (4.9) injection. VAS scores also showed minimal pain during the double-blind treatment phase, and reductions were reported from first to last injection by both aripiprazole-IM-depot (5.1/4.0) and placebo (5.1/4.9) subjects. Following the first injection in the IM-depot stabilization phase, investigators’ evaluations rated redness, swelling, induration, and pain as absent in the majority of patients (73.8%–95.8%; n = 568 evaluable subjects following first injection). Investigator ratings during double-blind treatment were similar between aripiprazole-IM-depot and placebo. Injection-site induration was reported more frequently in aripiprazole-IM-depot patients (1.9%, n = 5/269) than in placebo patients (0%).
The incidence of potentially clinically relevant prolactin elevation (> upper limit of normal) during double-blind treatment was lower with aripiprazole-IM-depot than placebo (1.9% vs 7.1%). The incidence of potentially clinically relevant changes in vital signs, orthostatic hypotension, and ECG parameters was similar between treatment groups during double-blind treatment, as was the mean change in QTc intervals.
During double-blind treatment, 14.9% of aripiprazole-IM-depot and 9.7% of placebo patients experienced treatment-emergent EPS AEs (akathisia [5.6% vs 6.0%], dyskinetic [0.7% vs 1.5%], dystonic [1.9% vs 1.5%], parkinsonism [8.2% vs 3.0%], and residual [0.4% vs 0.0%]). There was 1 report of tardive dyskinesia during double-blind treatment in a patient receiving placebo. The mean changes in AIMS movement score (−0.02 vs −0.02; P = .957), SAS total score (−0.02 vs −0.06; P = .689), and Barnes Akathisia Rating Scale global score (0.02 vs −0.02; P = .303) during double-blind treatment were not significantly different between aripiprazole-IM-depot and placebo at week 52 (LOCF). A total of 16.7% of patients in the aripiprazole-IM-depot group received anticholinergic agents for the alleviation of EPS during double-blind treatment, compared with 10.4% in the placebo group.
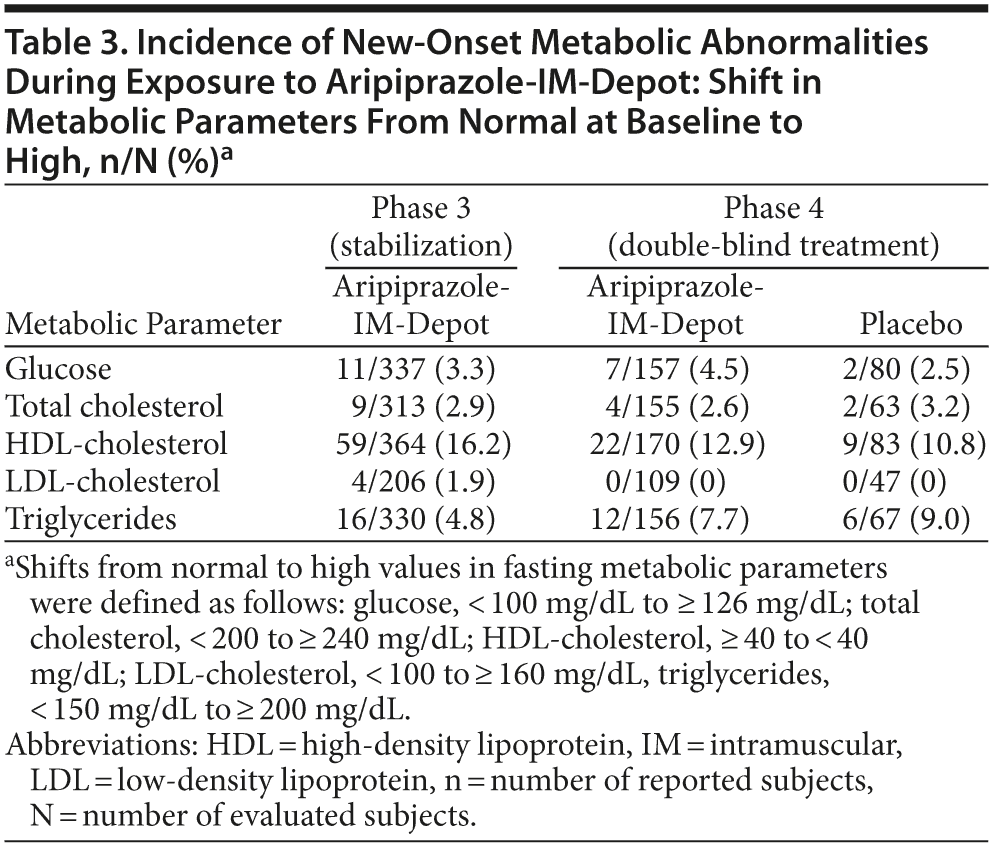
Mean weight changes during the oral and IM-depot stabilization phases were 0.1 kg and −0.2 kg, respectively. During double-blind treatment, mean change in body weight from double-blind baseline to last visit was −0.2 kg (n = 267) for aripiprazole-IM-depot and −0.4 kg (n = 134) for placebo (P = .812, LOCF analysis). The incidence of clinically significant weight gain (≥ 7% increase from baseline) was 0.9% during oral stabilization, 5.4% during IM stabilization, and 6.4% and 5.2% for aripiprazole-IM-depot and placebo, respectively, during double-blind treatment (LOCF). The incidence of potentially clinically significant new-onset metabolic abnormalities was similar between aripiprazole-IM-depot and placebo (Table 3).
DISCUSSION
Results from this randomized, double-blind, placebo-controlled study showed that aripiprazole-IM-depot is an effective maintenance treatment option in schizophrenia. Time to impending relapse and all-cause discontinuation (other than early termination of the study) for aripiprazole-IM-depot was significantly longer than with placebo, whereas relapse rates were significantly lower for patients receiving aripiprazole-IM-depot than for those receiving placebo. Discontinuations due to AEs were low and similar to placebo. Thus, aripiprazole-IM-depot offers a new long-acting antipsychotic treatment option.
When evaluating the risk-benefit profile of long-acting injectable antipsychotics, consideration should be given to potential AEs such as EPS, especially tardive dyskinesia, and metabolic disturbances, as these can have lasting long-term health consequences. Reported rates of akathisia were low across treatment phases and lower than with placebo during double-blind treatment, and there were no significant differences from placebo on movement disorder scales. Weight changes and the onset of new metabolic abnormalities were low with aripiprazole IM injections and similar to those observed with placebo. Furthermore, the metabolic profile of aripiprazole-IM-depot was similar to that of oral aripiprazole.25,26
Although there are inherent limitations in comparing results between trials using different methodologies and of differing durations, the impending relapse rates with aripiprazole-IM-depot compared with placebo reported here (10% vs 40%) were similar to those observed with paliperidone palmitate (18% vs 48% for placebo) in a double-blind, placebo-controlled study of similar design to evaluate the benefits of an atypical long-acting injectable antipsychotic in delaying time to relapse in schizophrenia.27 Prospective comparative analyses are needed to better understand the potential differences in overall risk-benefit profile of the
2 agents.
The risk-benefit profile of long-acting antipsychotic agents compared to oral agents should also be considered when making treatment decisions. A systematic meta-analysis of long-term studies of both typical and atypical depot antipsychotics concluded that depot medication was associated with clinically meaningful reductions in relapse rates compared with oral antipsychotic treatment in outpatients with schizophrenia28; similarly, an epidemiologic study conducted in Finland (n = 2,588)29 strongly favored the benefits of long-acting agents versus oral agents. In contrast, a recent study conducted in the US Veterans Affairs system (n = 369) showed that the rate of hospitalization with the long-acting injectable risperidone (39%) was not superior to the rate with oral antipsychotics (45%).30 The benefits of preventing relapse in the context of reducing disease progression are important to consider in terms of potential benefits of long-acting agents.
Some methodological differences may account for the lack of consistent results when comparing the efficacy of depot and oral antipsychotics. Alternatively, patient selection may be one factor, as patient participation in a clinical trial may lead to biased selection of relatively adherent patients, thereby diminishing the likelihood of finding differences between antipsychotic formulations. An additional factor may be a study’s length; recent evidence shows that differences in efficacy between oral and depot antipsychotics emerge over time.31,32 Moreover, participation in a clinical trial program involving regular symptom assessment might also reduce rates of nonadherence by providing for more careful overall management and monitoring. Ultimately, the decision to use a long-acting agent should be made on the basis of evidence, clinical judgment, and the individual needs of each patient.
The limitations of this study include the requirement that all patients be stabilized on aripiprazole-IM-depot for at least 12 weeks prior to randomization may have resulted in a patient population different from that of clinical practice. Interpretation of study findings should consider the entry criteria used, such as duration of illness of at least
3 years and no requirement for a maximum time from last exacerbation—factors that are particularly relevant when making treatment decisions. In addition, selection bias for a double-blind patient population that responded to oral and depot aripiprazole may have affected study outcome. However, the incidence and severity of treatment-emergent adverse events (including discontinuations) between phases of treatment were similar, suggesting a fair representation of patients in the double-blind phase. Finally, early termination of the study limited the number of patients with exposure to aripiprazole-IM-depot for the planned 12 months.
Aripiprazole-IM-depot significantly delayed time to impending relapse compared with placebo and is a relatively well-tolerated maintenance treatment option in schizophrenia that has a different risk-benefit profile than currently available options.
Drug names: aripiprazole (Abilify), benztropine (Cogentin and others), clozapine (Clozaril, FazaClo, and others), paliperidone (Invega), risperidone (Risperdal and others).
Author affiliations: The Zucker Hillside Hospital and The Hofstra North Shore-LIJ School of Medicine, Glen Oaks, New York (Dr Kane); Otsuka Pharmaceutical Development and Commercialization, Inc, Princeton, New Jersey (Drs Sanchez, Forbes, McQuade, and Carson; Mss Perry and Jin; and Mr Johnson); and Department of Psychiatry and Psychotherapy, Medical University Innsbruck, Innsbruck, Austria (Dr Fleischhacker).
Potential conflicts of interest: Dr Kane has received honoraria for lectures and/or consulting from Alkermes, Amgen, Bristol-Myers Squibb, Cephalon, Eisai, Boehringer Ingelheim, Eli Lilly, Intracellular Therapeutics, Janssen, Johnson & Johnson, Lundbeck, Merck, Novartis, Otsuka, Pfizer, Pierre Fabre, Proteus, Roche, Sunovion, and Targacept
and is a shareholder of MedAvante. Dr Fleischhacker has received research grants from Otsuka, Pfizer, Janssen, Alkermes, and Eli Lilly; consulting honoraria from Lundbeck, Roche, Bristol-Myers Squibb, Otsuka, Janssen, Pfizer, United BioSource, MedAvante, Sunovion,
and Merck; and speaker honoraria from Lundbeck, Sunovion, Janssen,
Eli Lilly, Otsuka, and AstraZeneca and holds stock in MedAvante.
Drs Sanchez, Forbes, McQuade, and Carson; Mss Perry and Jin; and Mr Johnson are employees of Otsuka Pharmaceutical Development and Commercialization, Inc.
Funding/support: This study was supported by Otsuka Pharmaceutical Commercialization, Inc (Tokyo, Japan).
Acknowledgments: Editorial support for the preparation of this manuscript was provided by Ogilvy Healthworld Medical Education; funding for this support was provided by Otsuka Pharmaceutical Commercialization, Inc.
REFERENCES
1. Keith S. Advances in psychotropic formulations. Prog Neuropsychopharmacol Biol Psychiatry. 2006;30(6):996–1008. doi:10.1016/j.pnpbp.2006.03.031 PubMed
2. Byerly MJ, Thompson A, Carmody T, et al. Validity of electronically monitored medication adherence and conventional adherence measures in schizophrenia. Psychiatr Serv. 2007;58(6):844–847. doi:10.1176/appi.ps.58.6.844 PubMed
3. Valenstein M, Ganoczy D, McCarthy JF, et al. Antipsychotic adherence over time among patients receiving treatment for schizophrenia:
a retrospective review. J Clin Psychiatry. 2006;67(10):1542–1550. doi:10.4088/JCP.v67n1008 PubMed
4. Gilmer TP, Dolder CR, Lacro JP, et al. Adherence to treatment with antipsychotic medication and health care costs among Medicaid beneficiaries with schizophrenia. Am J Psychiatry. 2004;161(4):692–699. doi:10.1176/appi.ajp.161.4.692 PubMed
5. Ascher-Svanum H, Faries DE, Zhu B, et al. Medication adherence and long-term functional outcomes in the treatment of schizophrenia in usual care. J Clin Psychiatry. 2006;67(3):453–460. doi:10.4088/JCP.v67n0317 PubMed
6. Herings RM, Erkens JA. Increased suicide attempt rate among patients interrupting use of atypical antipsychotics. Pharmacoepidemiol Drug Saf. 2003;12(5):423–424. doi:10.1002/pds.837 PubMed
7. Nasrallah HA. The case for long-acting antipsychotic agents in
the post-CATIE era. Acta Psychiatr Scand. 2007;115(4):260–267. doi:10.1111/j.1600-0447.2006.00982.x PubMed
8. Kane JM. Review of treatments that can ameliorate nonadherence in patients with schizophrenia. J Clin Psychiatry. 2006;67(suppl 5):9–14. PubMed
9. Lindenmayer JP. Long-acting injectable antipsychotics: focus on olanzapine pamoate. Neuropsychiatr Dis Treat. 2010;6(6):261–267. doi:10.2147/NDT.S3072 PubMed
10. Burris KD, Molski TF, Xu C, et al. Aripiprazole, a novel antipsychotic,
is a high-affinity partial agonist at human dopamine D2 receptors. J Pharmacol Exp Ther. 2002;302(1):381–389. doi:10.1124/jpet.102.033175 PubMed
11. Shapiro DA, Renock S, Arrington E, et al. Aripiprazole, a novel
atypical antipsychotic drug with a unique and robust pharmacology. Neuropsychopharmacology. 2003;28(8):1400–1411. doi:10.1038/sj.npp.1300203 PubMed
12. Stark AD, Jordan S, Allers KA, et al. Interaction of the novel antipsychotic aripiprazole with 5-HT1A and 5-HT 2A receptors: functional receptor-binding and in vivo electrophysiological studies. Psychopharmacology (Berl). 2007;190(3):373–382. doi:10.1007/s00213-006-0621-y PubMed
13. Jordan S, Koprivica V, Chen R, et al. The antipsychotic aripiprazole is a potent, partial agonist at the human 5-HT1A receptor. Eur J Pharmacol. 2002;441(3):137–140. doi:10.1016/S0014-2999(02)01532-7 PubMed
14. Pigott TA, Carson WH, Saha AR, et al; Aripiprazole Study Group. Aripiprazole for the prevention of relapse in stabilized patients
with chronic schizophrenia: a placebo-controlled 26-week study.
J Clin Psychiatry. 2003;64(9):1048–1056. doi:10.4088/JCP.v64n0910 PubMed
15. Fleischhacker WW, McQuade RD, Marcus RN, et al. A double-blind, randomized comparative study of aripiprazole and olanzapine in patients with schizophrenia. Biol Psychiatry. 2009;65(6):510–517. doi:10.1016/j.biopsych.2008.07.033 PubMed
16. Goff DC, Cather C, Evins AE, et al. Medical morbidity and mortality
in schizophrenia: guidelines for psychiatrists. J Clin Psychiatry 2005;
66(2):183–194; quiz 147, 273–184. PubMed
17. Mallikaarjun S, Salazar DE, Bramer SL. Pharmacokinetics, tolerability, and safety of aripiprazole following multiple oral dosing in normal healthy volunteers. J Clin Pharmacol. 2004;44(2):179–187. doi:10.1177/0091270003261901 PubMed
18. Fleischhacker WW, Kane JM, Sanchez R, et al. A pharmacokinetic study of once-monthly aripiprazole extended-release injectable suspension (ARI-ERIS) in adult patients with schizophrenia. Presented at the 164th Annual Meeting of the American Psychiatric Association; May 14–16, 2011; Honolulu, Hawaii.
19. Kay SR, Fiszbein A, Opler LA. The Positive and Negative Syndrome Scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13(2):261–276. PubMed
20. Guy W. ECDEU Assessment Manual for Psychopharmacology-Revised. Rockville, MD: US Department of Health Services; 1976.
21. Lindenmayer JP, Czobor P, Alphs LD, et al. InterSePT Study Group. The InterSePT Scale for Suicidal Thinking: reliability and validity. Schizophr Res. 2003;63(1–2):161–170. doi:10.1016/S0920-9964(02)00335-3 PubMed
22. Guy W. Abnormal Involuntary Movement Scale (AIMS). US Department of Health Education and Welfare ECDEU Assessment Manual for Psychopharmacology. Rockville, MD: National Institute
of Mental Health; 1976:534–537.
23. Simpson GM, Angus JW. A rating scale for extrapyramidal side effects. Acta Psychiatr Scand suppl. 1970;45(s212):11–19. doi:10.1111/j.1600-0447.1970.tb02066.x PubMed
24. Barnes TR. A rating scale for drug-induced akathisia. Br J Psychiatry. 1989;154(5):672–676. doi:10.1192/bjp.154.5.672 PubMed
25. Newcomer JW. Metabolic considerations in the use of antipsychotic medications: a review of recent evidence. J Clin Psychiatry. 2007;
68(suppl 1):20–27. PubMed
26. Stroup T, McEvoy J, Ring K, et al. A randomized trial examining the effectiveness of switching from olanzapine, quetiapine, or risperidone to aripiprazole to reduce metabolic risk: Comparison of Antipsychotics for Metabolic Problems (CAMP). Am J Psychiatry. 2011; 168(9):
947–956. PubMed
27. Hough D, Gopal S, Vijapurkar U, et al. Paliperidone palmitate maintenance treatment in delaying the time-to-relapse in patients with schizophrenia: a randomized, double-blind, placebo-controlled study. Schizophr Res. 2010;116(2–3):107–117. doi:10.1016/j.schres.2009.10.026 PubMed
28. Leucht C, Heres S, Kane JM, et al. Oral versus depot antipsychotic drugs for schizophrenia—a critical systematic review and meta-analysis of randomised long-term trials. Schizophr Res. 2011;127(1–3):83–92. doi:10.1016/j.schres.2010.11.020 PubMed
29. Tiihonen J, Haukka J, Taylor M, et al. A nationwide cohort study of oral and depot antipsychotics after first hospitalization for schizophrenia. Am J Psychiatry. 2011;168(6):603–609. doi:10.1176/appi.ajp.2011.10081224 PubMed
30. Rosenheck RA, Krystal JH, Lew R, et al; CSP555 Research Group. Long-acting risperidone and oral antipsychotics in unstable schizophrenia. N Engl J Med. 2011;364(9):842–851. doi:10.1056/NEJMoa1005987 PubMed
31. Gaebel W, Schreiner A, Bergmans P, et al.. Relapse prevention in schizophrenia and schizoaffective disorder with risperidone long-acting injectable vs quetiapine: results of a long-term, open-label, randomized clinical trial. Neuropsychopharmacology. 2010;35(12):2367–2377. PubMed
32. Keks NA, Ingham M, Khan A, et al. Long-acting injectable risperidone v olanzapine tablets for schizophrenia or schizoaffective disorder: randomised, controlled, open-label study. Br J Psychiatry. 2007;
191(2):131–139. doi:10.1192/bjp.bp.105.017020 PubMed
This PDF is free for all visitors!




