This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.
Objective: Cognitive impairment frequently accompanies major depressive disorder (MDD) and can persist during remission. This review examined pharmacotherapy effects on cognitive function in MDD.
Data Sources: PubMed and EMBASE searches were conducted on July 30, 2013, for English language reports of cognitive assessments following pharmacologic monotherapy or augmentation therapy in MDD.
Study Selection: A total of 43 research reports were identified (31 monotherapy [8 placebo-controlled, 11 active-comparator, 12 open-label], 12 augmentation therapy [7 placebo-controlled, 5 open-label]).
Data Extraction: Results reported in each publication were examined for open-label and placebo- or active comparator-controlled studies.
Results: Studies varied widely in terms of size (median, 50 participants; interquartile range, 21-143 participants), populations examined, duration (median, 8 weeks; interquartile range, 6-12 weeks), and neurocognitive assessments used. Most individual studies reported some benefit to cognition with pharmacotherapy, but there was no pattern of response in specific domains and only 12% of individually analyzed changes favored active treatment over placebo or untreated healthy controls. Sample weighted mean effect sizes revealed that verbal memory improved with monotherapy, while the largest treatment effect with augmentation therapy was for visual memory.
Conclusions: Pharmacotherapy may have benefit in reducing cognitive impairment in MDD, with augmentation therapy being a potential approach for addressing cognitive deficits that persist after monotherapy has brought about clinical response or remission. However, given the wide variability in study design and treatment duration across studies, these findings should be interpreted cautiously. More definitive research is required before firm conclusions can be reached.
Cognitive Effects of Pharmacotherapy for Major Depressive Disorder: A Systematic Review

ABSTRACT
Objective: Cognitive impairment frequently accompanies major depressive disorder (MDD) and can persist during remission. This review examined pharmacotherapy effects on cognitive function in MDD.
Data Sources: PubMed and EMBASE searches were conducted on July 30, 2013, for English language reports of cognitive assessments following pharmacologic monotherapy or augmentation therapy in MDD.
Study Selection: A total of 43 research reports were identified (31 monotherapy [8 placebo-controlled, 11 active-comparator, 12 open-label], 12 augmentation therapy [7 placebo-controlled, 5 open-label]).
Data Extraction: Results reported in each publication were examined for open-label and placebo- or active comparator-controlled studies.
Results: Studies varied widely in terms of size (median, 50 participants; interquartile range, 21-143 participants), populations examined, duration (median, 8 weeks; interquartile range, 6-12 weeks), and neurocognitive assessments used. Most individual studies reported some benefit to cognition with pharmacotherapy, but there was no pattern of response in specific domains and only 12% of individually analyzed changes favored active treatment over placebo or untreated healthy controls. Sample weighted mean effect sizes revealed that verbal memory improved with monotherapy, while the largest treatment effect with augmentation therapy was for visual memory.
Conclusions: Pharmacotherapy may have benefit in reducing cognitive impairment in MDD, with augmentation therapy being a potential approach for addressing cognitive deficits that persist after monotherapy has brought about clinical response or remission. However, given the wide variability in study design and treatment duration across studies, these findings should be interpreted cautiously. More definitive research is required before firm conclusions can be reached.
J Clin Psychiatry 2014;75(8):864-876
© Copyright 2014 Physicians Postgraduate Press, Inc.
Submitted: May 31, 2013; accepted January 17, 2014.
Online ahead of print: July 8, 2014 (doi:10.4088/JCP.13r08609).
Corresponding author: Richard S. E. Keefe, PhD, Department of Psychiatry and Behavioral Sciences, Room 3425 Purple Zone, 200 Trent Drive, Box #3270 DUHS, Duke University Medical Center, Durham, NC 27710 ([email protected]).
It is now recognized that individuals with major depressive disorder (MDD) may exhibit deficits in cognition, including cognitive inefficiency.1,2 However, of the 9 diagnostic criteria for MDD in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5),3 “diminished ability to think or concentrate, or indecisiveness,” is the only disturbance that is clearly cognitive in nature. Other symptoms, such as diminished energy and sleep disturbances, can also adversely affect cognitive function.
Cognition may be impaired in several domains, including processing speed, psychomotor skills, attention, memory, and executive functions. The domains of memory and executive function tend to show the most severe impairment, especially in elderly people with MDD.1
For most people with MDD, it is unclear whether cognitive impairment varies with the severity of depressive symptoms or persists during times of euthymia. The degree of correlation between the severity of cognitive impairment and the severity of depressive symptoms has been reported to vary widely.4-6 However, it is known that in some individuals, cognitive impairment can persist during periods of remission from depressive symptoms.7 Moreover, prior research suggests that persistent cognitive deficits (including cognitive inefficiency) can have detrimental effects on coping ability8 and execution of basic and instrumental activities of daily living.9
The US Food and Drug Administration has not approved any pharmaceuticals for the treatment of cognitive impairment in MDD, nor is there an expert clinical and scientific consensus on this issue. Nevertheless, numerous antidepressant therapy studies have included endpoints to detect treatment-associated cognitive improvement as a therapeutic benefit or treatment-associated cognitive impairment as an adverse event.
Determination of the magnitude of effect of antidepressant treatment on cognitive function is made difficult by the wide variety of study designs and assessment methodologies that have been implemented. A broad range of neuropsychological instruments has been used across studies; findings have been reported in different formats even when similar instruments are used; and there is considerable inconsistency in the manner by which these instruments have been categorized in terms of assessing specific domains of cognitive function.
In addition, because antidepressant monotherapy is effective in only some individuals with MDD, and only a minority achieve complete remission with initial monotherapy,10 many patients have residual depressive symptoms, including decreased concentration,11 which can interfere with higher-order cognitive function. Therefore, MDD treatment often requires augmentation of antidepressant monotherapy. However, relatively little is known about the effects of augmentation therapy on cognitive function in individuals with MDD.
This systematic review reports on the cognitive effects of antidepressant monotherapy and augmentation pharmacotherapy on cognitive function in individuals with MDD. The goals of the review were (1) to summarize the published studies and conference presentations that have assessed the effects of antidepressant pharmacotherapy for MDD on cognitive impairment, with attention to identifying the domains of cognitive function most likely to be affected by treatment; and (2) to describe the impact of augmentation pharmacotherapy on cognitive function in people with MDD whose depressive symptoms are stable or in remission after monotherapy.

- Individuals with major depressive disorder (MDD) may exhibit deficits across a range of cognitive function domains, which can persist during depressive symptom remission.
- No current pharmacotherapy is approved for treating cognitive dysfunction in MDD, but pharmacotherapy may have beneficial effects in this area.
- Based on the biomedical literature review, antidepressant monotherapy or augmentation of antidepressant monotherapy can help improve cognitive dysfunction in MDD; however, cautious interpretation is warranted because of intrinsic study limitations and variability in study designs.
METHOD
Literature Searches
We performed a series of literature searches on the effects of therapy on cognitive function in MDD on July 30, 2013. The first PubMed search, focused on monotherapy, was conducted using the following search string: (major depressive disorder OR unipolar depression) AND (executive function OR cognitive function OR cognition OR cognitive impairment OR cognitive dysfunction OR executive dysfunction). The search was limited to humans, clinical trials, meta-analyses, randomized controlled trials, and English language. The second search, focused specifically on augmentation therapy, was conducted using the following search string: (major depressive disorder OR MDD OR unipolar depression) AND (augmentation OR augment OR adjunct OR adjunctive OR combination therapy OR add-on) AND (therapy OR treatment). The same search parameter limits described above were applied. To confirm and coordinate these searches, a final search was conducted using the following string: (major depressive disorder OR unipolar depression) AND (executive function OR cognitive function OR cognition OR cognitive impairment OR cognitive dysfunction OR executive dysfunction) AND (augmentation OR augment OR adjunct OR adjunctive OR combination therapy OR add-on) AND (therapy OR treatment). Again, the same search parameter limits were applied.
We obtained full-text versions of all potentially pertinent references from these 3 independent searches. These articles were reviewed to confirm their relevance to the topic. During this review, we obtained publications cited in the selected papers that appeared to be relevant and had not already been identified in the independent literature searches. Duplicates (reports that came up on more than one search) were eliminated. Also, reports on nonpharmacologic treatment modalities (such as transcranial magnetic stimulation) were excluded so that this review could focus on the effects of pharmacotherapy.
Finally, a search of EMBASE was conducted to identify recent conference presentations of relevance to this topic that were not yet published as peer-reviewed manuscripts. This search, conducted on July 30, 2013, used the terms major depressive disorder AND (cognition OR cognitive) AND (drug OR treatment OR pharmacotherapy OR pharmacotherapeutic) NOT (magnetic OR electromagnetic OR transcranial OR electroconvulsive); the limits were English language, human subjects, years 2010-2013; publication type: conference presentation/abstract/review.
Statistical Analyses
Effect sizes for the treatment differences in the change from baseline (end-of-study values – baseline values) for all cognitive endpoints from trials that included a placebo control, healthy control, or active comparator and that adequately reported the data were calculated using Cohen d ([active treatment – control]/pooled population standard deviation at end of study). The pooled standard deviation at end of study was chosen for this analysis because it may better approximate the standard deviation for the individual changes from baseline for each study. In addition, sample weighted mean effect sizes with 95% CIs were calculated for each cognitive function domain across studies assessing monotherapy versus placebo or augmentation therapy versus placebo augmentation. As clinical improvement is associated with increased values for some measures and decreased values for others, all analyses were conducted so that positive effect sizes favor active treatment. The 95% CI of the effect size was based on the normal distribution of the estimators of effect size. Due to the lack of a rigorous peer-review process, published abstracts were not included in the assessment of effect size.
Literature Review Results
The 4 searches on the cognitive effects of antidepressant monotherapy and augmentation therapy in MDD yielded a total of 1,032 hits (PubMed, n = 759; EMBASE, n = 273). hits. After exclusion of duplicates, items deemed not relevant and items not reporting cognition data, and the selection of additional articles identified from the reference lists of included publications, 43 research reports were identified. These reports included 31 monotherapy studies12-42 (26 published articles, 5 abstracts for one of which40 additional study design information was obtained from a reference43 cited in the published abstract) and 12 augmentation studies44-55 (10 published articles, 2 abstracts). Three reports present pooled data,17,19,36 2 represent a primary study and its extension,22,33 and 2 are separate assessments of a single study population.25,30
RESULTS
Descriptive Analysis
Supplementary eTable 1 summarizes the main characteristics of the 43 reports included in this review. In terms of study design, there are 15 reports on placebo-controlled studies (8 monotherapy, 7 augmentation therapy), 11 reports on active-comparator studies (all monotherapy), and 17 reports on open-label studies (single-arm studies or studies of treated patients vs either healthy controls or untreated patients, or time-course comparisons of treated patients without a control arm; 12 monotherapy, 5 augmentation therapy).
Study participants were individuals with mild to severe depression; entry criteria in all studies included a minimum score on a depression symptom rating instrument to verify the level of severity of depression. Among all the studies included in this review, only 144 assessed a population defined by complete or partial remission of MDD (based on Montgomery-Asberg Depression Rating Scale score ≤ 18). In 15 of the 43 reports, the populations were described as elderly (including 3 studies in which age ≥ 50 years was an entry criterion). An additional 3 studies assessed poststroke patients (mean ages, 58, 65, and 67 years). Most of the reports described cognitive effects of pharmacotherapy in participants with MDD. However, some dealt with depressive symptoms in the setting of comorbidities such as heart failure,12 alcohol dependence,15 stroke,27-29 or other conditions30; 1 report dealt with psychotic depression.50
Across all 43 studies, 4,828 participants were evaluated: monotherapy versus placebo, n = 2,149; active versus placebo augmentation, n = 384; monotherapy versus active control, n = 1,410; open-label monotherapy, n = 745; open-label augmentation, n = 140. Population size ranged from 12 to 776 participants per monotherapy study (mean ± SD, 139 ± 169; median, 63) and from 11 to 143 participants per augmentation therapy study (mean ± SD, 44 ± 39; median, 30). The mean ± SD number of participants across all studies was 112 ± 151 but the median was only 50 (interquartile range, 21-143), indicating that the distribution was skewed toward smaller populations.
Most of the studies were of relatively short duration. Excluding single-observation studies13,21,40 and a long-term extension,33 study duration ranged from 4 to 36 weeks in monotherapy studies (mean ± SD, 11 ± 7 weeks; median, 8 weeks) and from 3 to 104 weeks in augmentation therapy studies (mean ± SD = 16 ± 28 weeks; median, 6 weeks). Across all studies, mean ± SD study duration was 13 ± 16 weeks, but the distribution was skewed toward shorter lengths; the median study duration was 8 weeks (interquartile range, 6-12 weeks). The most frequent study durations were 8 weeks (10 studies) and 12 weeks (11 studies).
In most studies, antidepressant pharmacotherapy consisted of commonly used antidepressant agents (selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors, or tricyclic antidepressants). In some studies, other psychotropic agents were tested: apomorphine,13 lithium,48 estrogen,49 the serotonin-reuptake enhancer tianeptine,24 the antipsychotics aripiprazole54 and amisulpride,50 the mineralocorticoid receptor modulators fludrocortisone and spironolactone,53 the cognition-enhancing drugs galantamine46 and donepezil,51 the dissociative anesthetic/N-methyl-d-aspartate antagonist ketamine,40 and the d-amphetamine prodrug lisdexamfetamine dimesylate.44
Assessment of cognitive function was described as a primary/coprimary assessment in 32 reports (24 monotherapy12,13,15-19,22,24-35,37,38,41,42; 8 augmentation44,46,47,49,51-54) and as a secondary assessment in 9 reports (6 monotherapy14,20,23,36,39,40; 3 augmentation45,50,55). In contrast, it was a safety assessment in only 2 reports (1 monotherapy21; 1 augmentation48).
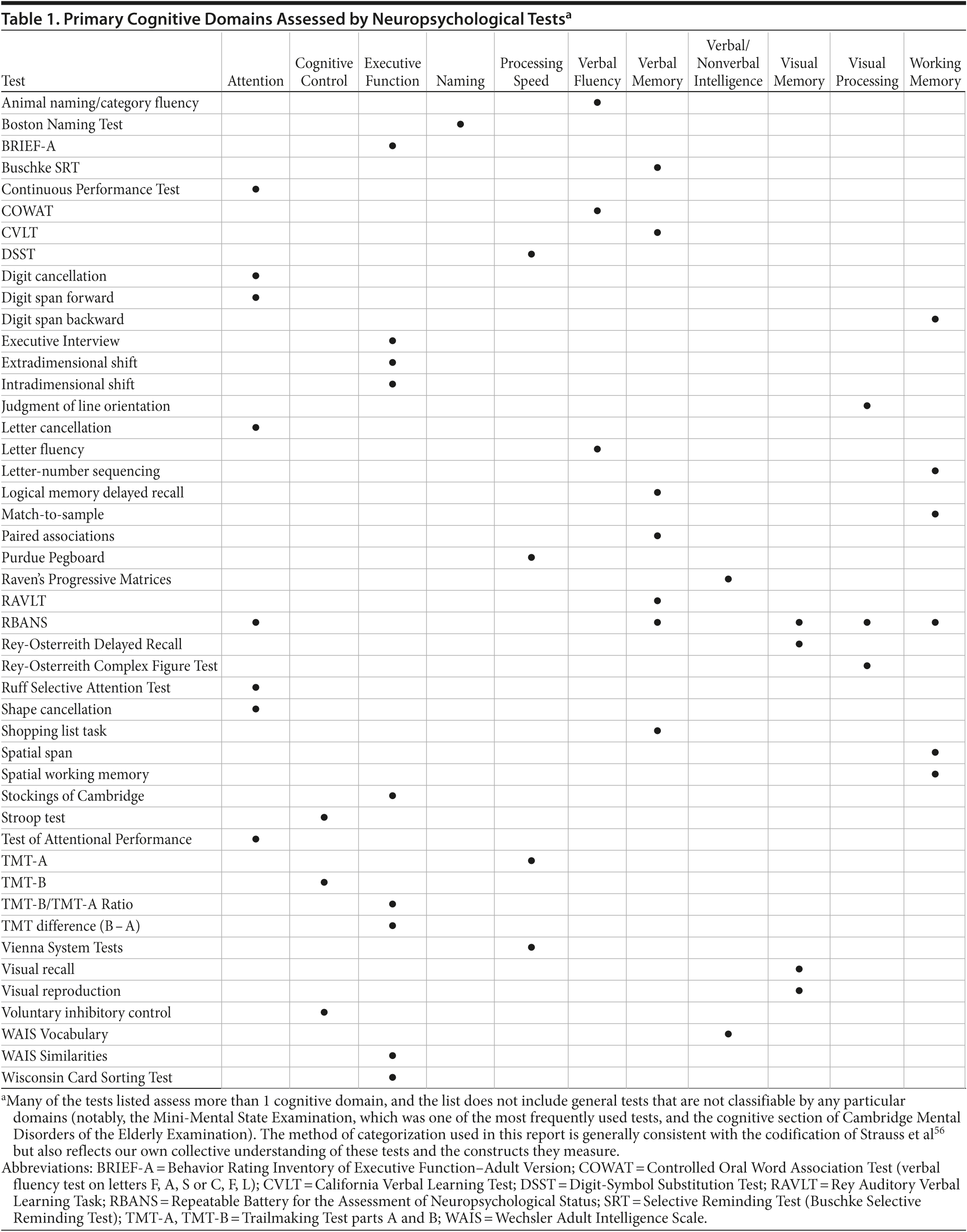
Numerous cognitive assessment tools were employed (see Supplementary eTable 1 at PSYCHIATRIST.COM). The most frequently used instrument was the Digit-Symbol Substitution Test (DSST, 13 studies); versions of this test also appear as part of the revised Wechsler Adult Intelligence Scale (WAIS-R, where it is called Digit Symbol), the third-edition WAIS (WAIS-III, where it is called Digit Symbol Coding), and the Brief Assessment of Cognition for Affective Disorders (BAC-A, where it is called Symbol Coding). Other frequently used instruments included the Mini-Mental State Examination (MMSE, 10 studies), Trail Making Test (9 studies), and the Stroop test (6 studies).
The cognitive domains assessed included processing speed, psychomotor function, attention, verbal learning and memory, verbal fluency, visuospatial awareness, and executive function. In some of the reports, the assessment instruments used are identified as measures of one or more specific domains of cognitive function. However, such identification was inconsistent. For example, in different studies, the Stroop test was described as a measure of executive function,31,33,35,42,45,51 information processing speed,22,31 or attention31; similarly, DSST was described as a measure of attention,13,25,30,32 psychomotor function,17,20,21,42 information processing speed,20,39 or visuospatial awareness.17,51
Table 1 lists the neurocognitive measures used in the studies that are included in this review, with the present authors’ own assignment of the primary cognitive domain assessed by each test. It is recognized that many tests assess multiple domains and that there may be disagreement with these assignments. In some cases, our assignment of primary cognitive domain was based on the codification by Strauss et al (2006),56 and as such may differ from that of the authors of the reviewed publication. For example, we labeled the Stroop test as a measure of cognitive control, a domain not named in the publications included in this review.
Data on the cognitive effects reported in the 43 studies of pharmacotherapy for depression included in this review are summarized in Supplementary eTable 2. In 6 studies, cognitive effects were specifically reported as a function of treatment response (defined by prespecified improvement in depressive symptoms) versus nonresponse; among these 6 studies, there was no reported correlation with treatment response in 1 open-label study16 and 1 active-comparator study35 but treatment responders tended to perform better than nonresponders in 2 active-comparator studies,17,24 1 placebo-controlled study,42 and 1 open-label study.32
In the majority of reports, there was statistically significant cognitive benefit with monotherapy12,14,17,19,20,22-39,41,42 or augmentation therapy.44,45,47,51-55 As might be expected, significant benefit was reported frequently with active treatment versus placebo (7 of 8 monotherapy studies19,25,30,34,36,39,42) and with active augmentation versus placebo augmentation (5 of 7 studies44,47,51,52,55). Of the 3 placebo-controlled studies that did not report significant benefit, all had relatively small treatment groups.13,46,49 Significant improvement from baseline appeared to be less consistent in open-label studies (7 of 12 monotherapy,12,27-29,31,32,41 3 of 5 augmentation45,53,54). In 10 of 11 active-comparator trials (all monotherapy),14,17,20,22-24,26,33,35,38 significant between-treatment differences were reported, with sertraline consistently having superior effects to nortriptyline in 3 studies14,17,35 and to fluoxetine in 2 studies.20,23
In many reports, benefit was seen on certain cognitive measures but not others, or in certain subpopulations (eg, treatment responders vs nonresponders) but not others, or during the course of the study but not at study end. In a minority of reports, no statistically significant cognitive benefit was seen with monotherapy13,15,16,18,21 or augmentation therapy.46,48-50 Across all studies, clear descriptions of the use of statistical procedures to adjust for multiple comparisons or time course analyses was found in only 7 reports.12,18,23,29,33,45,48
Based on the present authors’ assignment of the primary cognitive domain assessed by each test (Table 1), the cognitive tests most frequently used in the reports included in this review assessed the domains of verbal memory, working memory, and processing speed. Antidepressant pharmacotherapy was more likely than not to help improve performance in those domains, but the reported estimates of improvement were relatively small compared with the magnitude of the deficits. Keeping in mind that many studies included multiple tests for a given domain and that relatively few studies clearly stated that correction for multiple comparisons was conducted, verbal memory improved in 12 studies,14,17,22,23,25,26,31,32,35,39,41,52 working memory improved in 7 studies,19,22,31,33,51,53,54 and processing speed improved in 8 studies.17,19,20,23,32,39,41,53 Executive function, sometimes considered a key clinical and functional indicator, showed statistically significant improvement in 2 monotherapy studies33,37 and 3 augmentation therapy studies.44,51,54
Effect Size Analysis
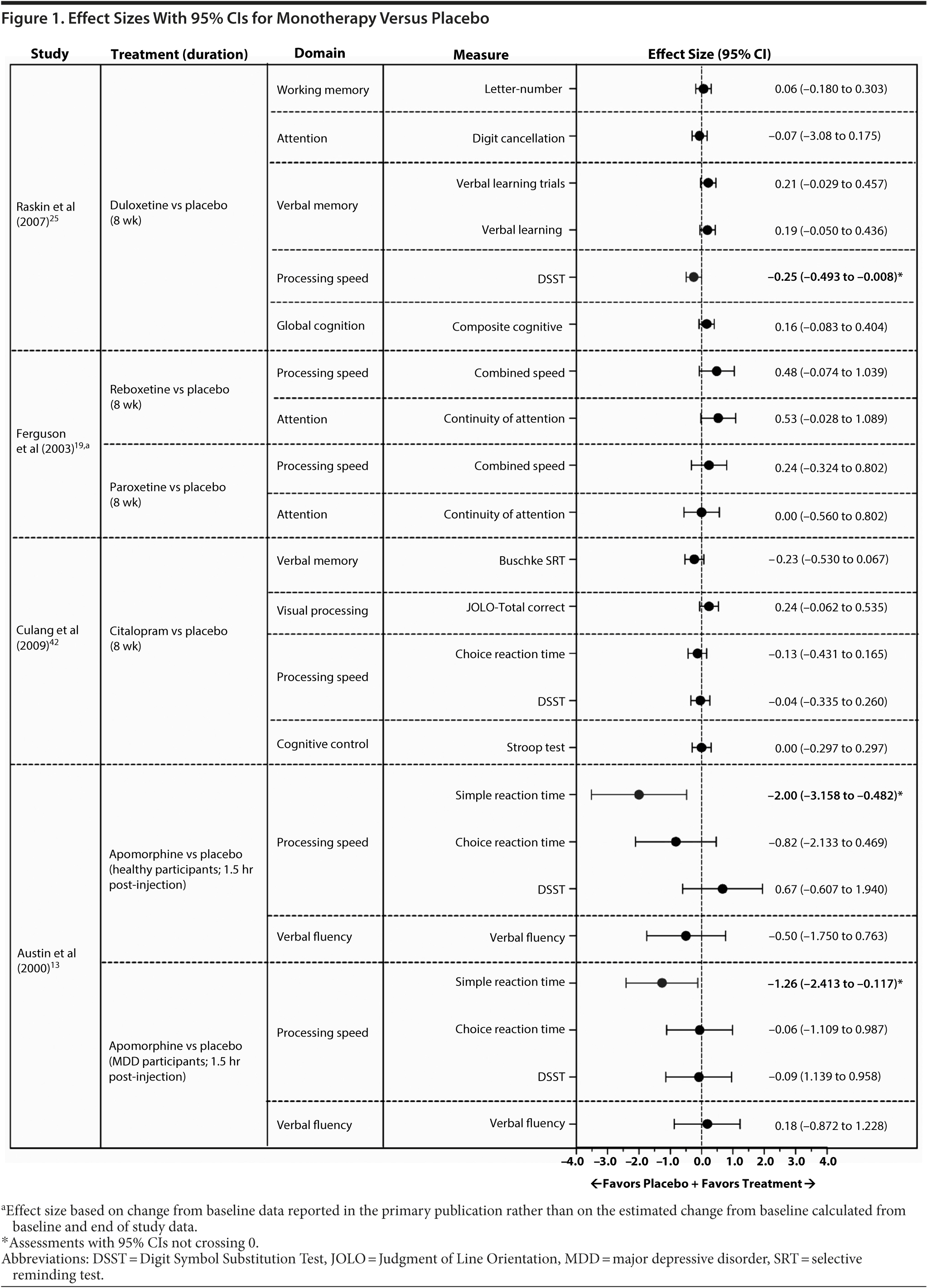
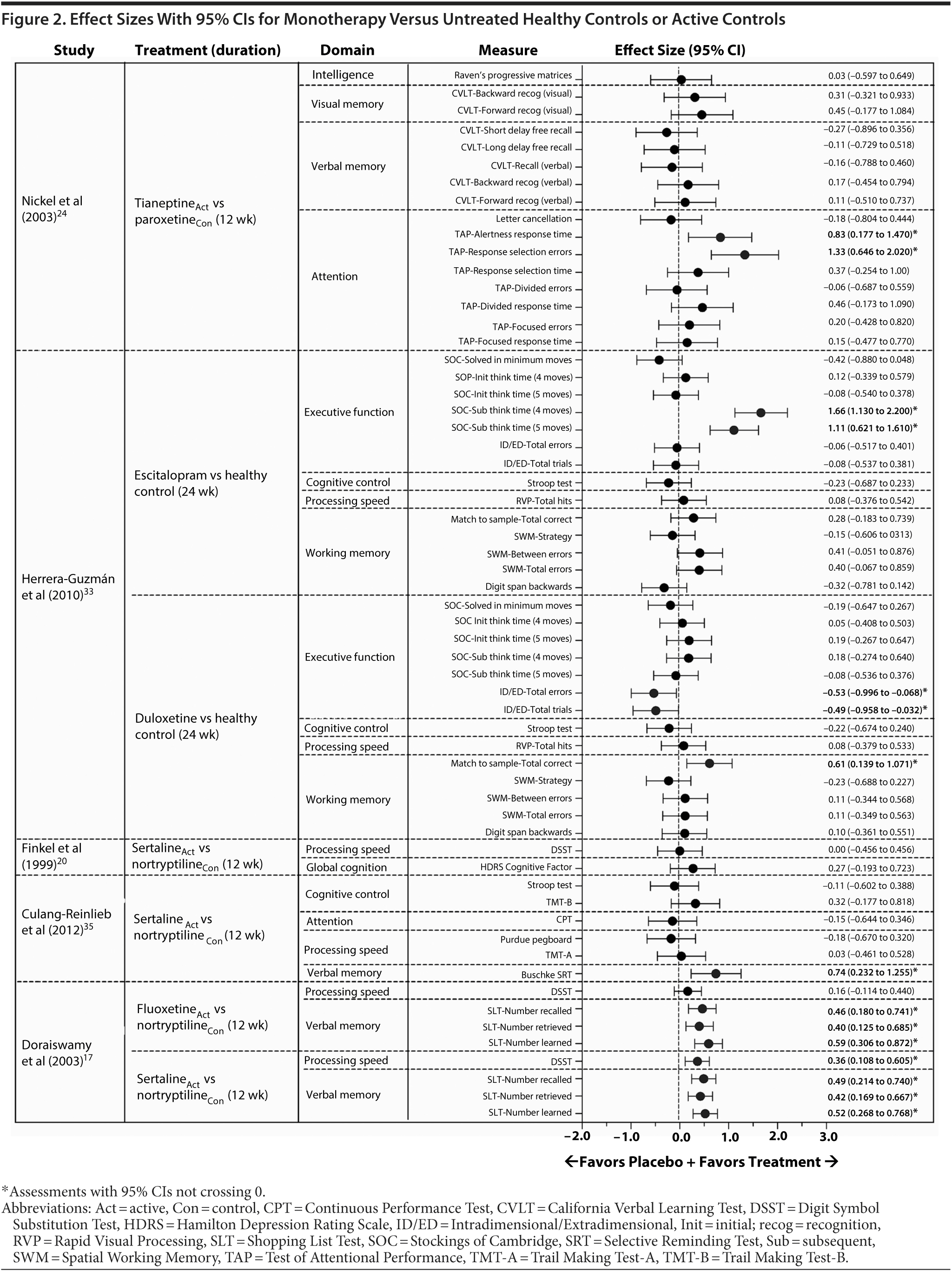
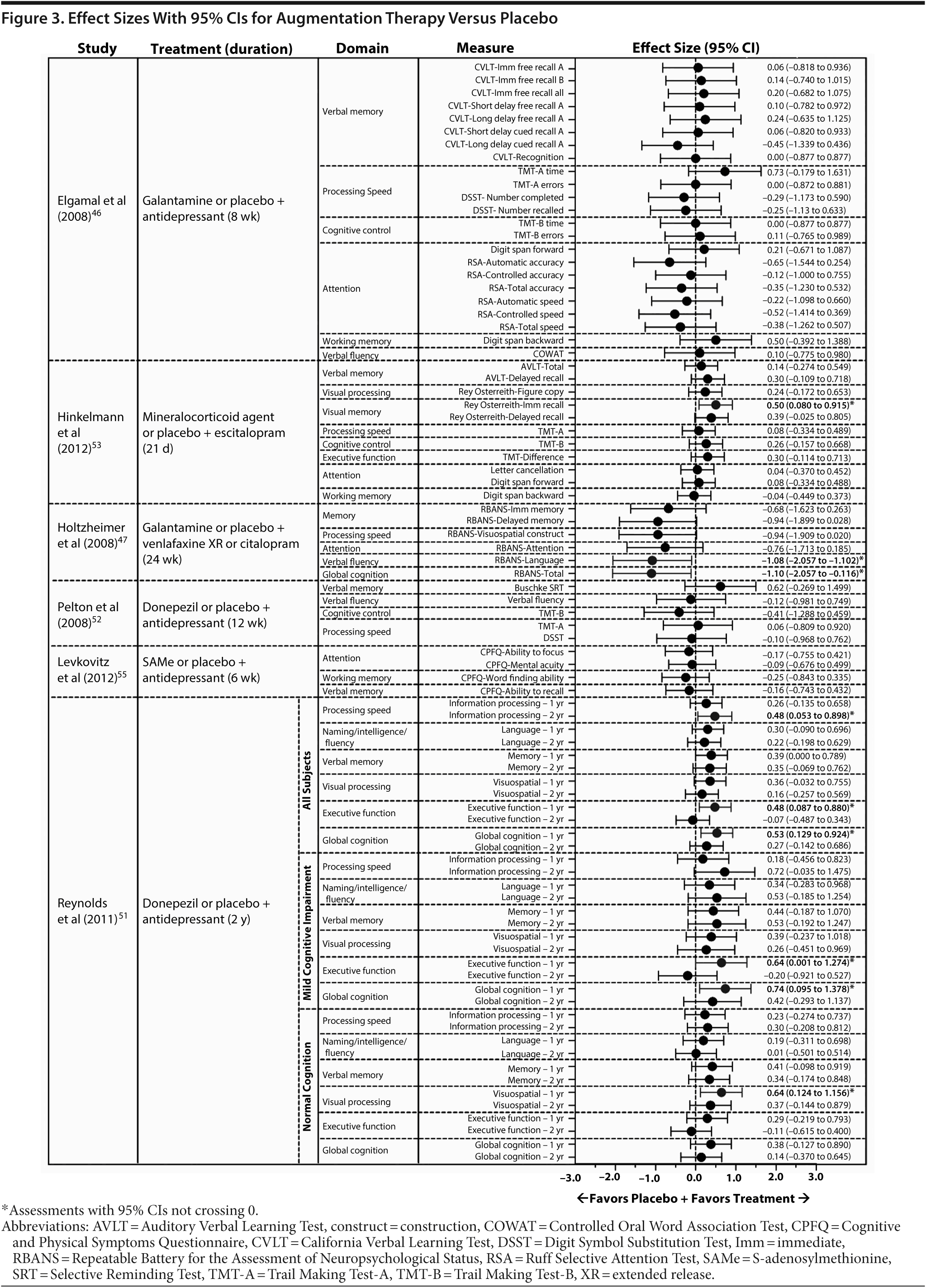
Based on data from 15 of the 43 identified publications, it was feasible to calculate an effect size for 168 cognitive measures. Assessments were based on data from 8 monotherapy publications13,17,19,20,24,33,35,42 and 4 augmentation publications47,52,53,55 and on data requested from the authors of 1 monotherapy publication25 and 2 augmentation publications.46,51 The 95% CI of the effect size did not cross 0 in 27 of these analyses (16%), indicating an effect favoring one of the treatment arms (Figures 1, 2, and 3; see values with asterisks), but did cross 0 in the remaining 141 analyses (84%). Of those analyses with 95% CIs that did not cross 0, 20 favored an active treatment (12% of all analyses [20/168]) over placebo or untreated healthy controls; the remaining analyses favored placebo or untreated healthy controls over active treatment (4% of all analyses [7/168]).
In monotherapy studies that included a placebo control, the 95% CIs did not cross 0 for 3 effect sizes from 2 studies.13,25 Small to large effect sizes indicated that improvements from baseline in the processing speed domain were greater with placebo than with apomorphine or duloxetine (Figure 1). The remaining effect sizes (n = 20) in monotherapy studies with a placebo control had 95% CIs that crossed 0; these effect sizes generally favored active treatment and tended to be of small to moderate magnitude (Figure 1).
In 1 monotherapy study that included healthy controls,33 10 effect sizes had 95% CIs that did not cross 0 (Figure 2). The large effect sizes indicated that improvement from baseline in individuals with MDD treated with duloxetine was greater than in untreated healthy controls in the working memory domain and that improvement in individuals with MDD treated with escitalopram was greater than in untreated healthy controls in the executive function domain. Additionally, large effect sizes indicated that improvement from baseline in untreated healthy controls in the executive function domain was greater than in individuals with MDD treated with duloxetine. The remaining effect sizes (n = 23) in monotherapy studies with healthy controls had 95% CIs that crossed 0; these effect sizes generally favored active treatment and tended to be of small to moderate magnitude (Figure 2).
In 3 monotherapy trials that included active comparators,17,24,35 5 effect sizes had 95% CIs that did not cross 0 (Figure 2). In 1 study,35 a large effect size indicated that improvement from baseline in the verbal memory domain was greater with sertraline than with nortryptiline. In a second study,17 moderate to large effect sizes indicated that improvement in the verbal memory domain was greater with fluoxetine than with nortryptiline and that improvement in the verbal memory and processing speed domains were greater with sertraline than with nortriptyline. In a third study,24 large effect sizes indicated that improvement from baseline in the attention domain was greater with tianeptine than with paroxetine. The remaining effect sizes (n = 22) in monotherapy studies with active treatment comparators had 95% CIs that crossed 0; across these assessments, effect sizes tended to be of small to moderate magnitude (Figure 2).
In augmentation therapy studies that included a placebo control, 9 effect sizes from 3 studies47,51,53 had 95% CIs that did not cross 0 (Figure 3). In 1 study,47 large effect sizes indicated that improvements from baseline in the verbal fluency domain and for global cognition were greater with placebo than with galantamine augmentation. In a second study,53 a large effect size indicated that improvement in the visual memory domain was greater with augmentation with a mineralocorticoid agonist or antagonist than with placebo. In the third study,51 moderate to large effect sizes for the executive function domain, visual spatial domain, information processing domain, and for global cognition indicated that improvements in performance were greater with donezepil augmentation than with placebo. The remaining effect sizes (n = 76) in augmentation therapy studies with a placebo control had 95% CIs that crossed 0; these effect sizes generally favored active treatment and tended to be of small to moderate magnitude (Figure 3).
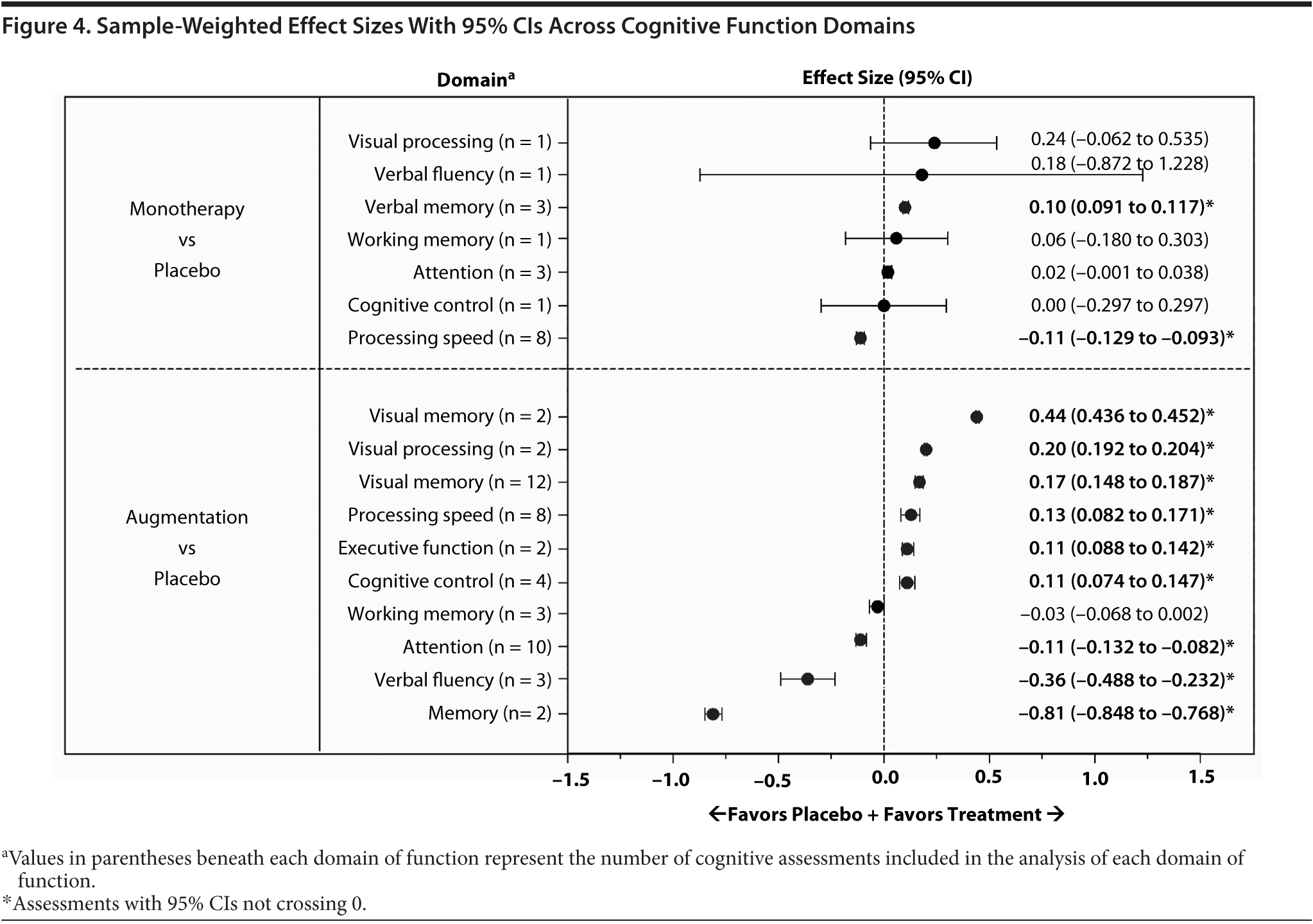
An examination of the cognitive domains across studies indicated that the 95% CIs for the sample weighted mean effect sizes did not cross 0 for 2 domains in monotherapy studies and for 9 domains in augmentation therapy studies (Figure 4, see values with asterisks). In monotherapy studies, a small effect size favoring active treatment was observed for verbal memory and a small effect size favoring placebo was observed for processing speed (Figure 4, top). In augmentation therapy trials, a moderate effect size favoring active treatment was observed for visual memory and small effect sizes favoring active treatment were observed for visual processing, verbal memory, processing speed, executive function, and cognitive control; a large effect size favoring placebo was reported for memory and small to moderate effect sizes favoring placebo were reported for attention and verbal fluency (Figure 4, bottom).
DISCUSSION
Although it is known that cognitive dysfunction is associated with MDD, the data regarding the cognitive effects of antidepressant pharmacotherapy are relatively limited. Overall, the reports included in this review suggest that antidepressant monotherapy or augmentation therapy can have beneficial effects on cognitive function. This interpretation is partially supported by the effect size analyses included in this review. Although the effect size analyses based on individual measures within studies indicated that there was a tendency for monotherapy and augmentation therapy to be favored over placebo across a range of cognitive domains, improvement with pharmacotherapy versus placebo or untreated healthy controls was meaningful (95% CI of the effect size not crossing 0) for only a minority (approximately 12%) of the analyzed cognitive assessments. Examination of sample weighted mean effect sizes for cognitive domains across studies indicated that only verbal memory was improved with monotherapy versus placebo; multiple domains of function improved with augmentation therapy versus placebo, with the largest effect size observed for visual memory. In regard to the effect size analyses reported in this review, it is important to note that the magnitude of the effects should be interpreted with caution because they represent estimates based on the pooled standard deviation at the end of each study. These effect sizes may overestimate or underestimate the “true” effect size based on the pooled standard deviation of the individual changes from baseline for each study. Furthermore, the number of measures included for each sample weighted mean effect size was ≤ 3 for all but 1 domain in monotherapy trials and was ≤ 4 in 7 of 9 domains. Another reason for caution is the high degree of variability in study design and data presentation among the studies included in this review.
Because many of the studies were relatively small or assessed selected subgroups of the depressed populations (eg, elderly patients or those with specific comorbidities), it remains unclear whether the findings can be generalized to a larger and more heterogeneous population of depressed individuals. Furthermore, the studies were generally of short duration, so long-term effects of treatment remain uncertain. Finally, it should be noted that in the trials comparing multiple active treatments, cognitive benefit with treatment versus lack of treatment was suggested but not confirmed.
With the exception of the report by Madhoo et al,44 which used a population of individuals in partial remission (MADRS total scores of 10-18) or full remission (MADRS total scores < 10), the studies included in this review assessed cognitive function in the presence of mild to severe depressive symptoms, which makes it difficult to establish whether cognitive improvement was an independent outcome or a consequence of clinical response and remission. Studies in elderly individuals generally did not draw clear distinctions between cognitive decline due to aging versus cognitive inefficiency and impairment due to depression. Similarly, except for the report by Wise et al,30 the potential impact of comorbid medical conditions on cognitive function were generally not addressed.
Statistical methodology was inconsistent, and some of the included reports were incomplete or unclear in this area. Examples include outcomes reported as interaction statistics without data on within-group changes from baseline and between-group differences at study end24,27,51 and drug effects described in words but not substantiated with specific data.46,48-50 Moreover, few of the reports specifically stated whether any type of correction for multiple comparisons was made; 7 reports12,18,23,29,33,45,48 stated that such corrections were made and 6 reports24,25,30,31,41,55 stated or described the analyses in such a manner as to indicate that corrections were not made. In addition, some neurocognitive measures were used more frequently than others, potentially increasing the likelihood that a positive outcome would be observed in these domains by chance.
A major issue in the interpretation of our findings in this review is the specific instruments used for assessment of cognitive function in the included studies. Several of the studies relied solely on the MMSE for assessment of cognitive function, but this scale is a poor choice for this purpose because it broadly measures global cognitive function, has no alternate form, and has extreme ceiling effects. In addition, many instruments assess multiple cognitive domains, and categorization was inconsistent across studies. For example, the Controlled Oral Word Association Test (COWAT) has been classified as a test of phonemic verbal fluency, which falls within the cognitive domain of language function,31 but it has also been deemed a measure of processing speed or executive function.57 The method of categorization used in this report is generally consistent with the codification of Strauss et al56 but also reflects our own collective understanding of these tests and the constructs they measure. Furthermore, different versions of the same assessment tool could have been used in different studies, which could influence the consistency of findings across studies. Lastly, many of the assessment tools used were developed to assess cognitive function in healthy individuals or in those with specific neurologic deficits. As such, their use in individuals with MDD may imply that there is a more complex array of deficits in depression when, in fact, there is a simpler factor structure associated with depression.
One method to ensure consistency across studies would be to use a cognitive battery specific and sensitive to MDD. This approach is reflected in the Measurement and Treatment Research to Improve Cognition in Schizophrenia58 (MATRICS) initiative directed by the National Institutes of Mental Health (NIMH), which recognized that lack of consensus on the optimal tools for assessing cognitive function was a barrier to the development of effective treatments for cognitive impairment in schizophrenia. Thus, a standardized test battery, developed in accordance with criteria suggested by the MATRICS participants, could facilitate interpretation across separate studies; however, a limitation of this approach is that researchers may feel obligated to use the standard battery even in situations for which other tests might be more appropriate.
The complexities of research in this area are illustrated by assessing the similarities and differences between the present review and that of a recently published meta-analysis (with a psychological rather than pharmacotherapeutic focus) of executive function impairment in MDD.59 Both publications suggest that MDD is associated with deficits across a wide range of cognitive domains, that cognitive impairment may correlate with depressive symptom severity on some measures, and that impairments may persist despite remission of depressive symptoms. However, the focus on the role of pharmacotherapy in MDD differed between publications. The present review examined the impact of pharmacotherapy on cognitive dysfunction and found some evidence for a positive effect. In contrast, the meta-analysis of Snyder focused on identifying the nature of the cognitive deficits in MDD and their possible mediators, which included detrimental effects of pharmacotherapy on cognitive function (although the author acknowledged that this finding may reflect the profile of patients using antidepressant drugs rather than adverse effects of the drugs). In addition, the categorization of neurocognitive tests offered in the meta-analysis sometimes differed from our own, again indicating the need for greater understanding of and consensus on the various domains of cognition, their interrelationships, and the optimal means by which they may be measured.
It should be noted that this literature review was restricted to searches of PubMed and EMBASE, which are databases that include publications from the most methodologically rigorous journals and congresses. However, utilization of other databases might have provided additional information. In addition, the inclusion of abstracts in the review can be viewed as a limitation because the peer-review process is less rigorous for abstracts than it is for published manuscripts. As such, data from these abstracts were not included in the assessment of effect sizes. However, as presentations at biomedical congresses represent the most recent data that is available, a description of these studies and their findings is relevant to this topic. Lastly, although several negative findings were reported in the studies we included in our review, it should be noted that publication bias could influence these findings as negative findings are less likely to be published.
The common occurrence of cognitive impairment in individuals with MDD may be related to the shared neurobiologic systems and processes that can modulate cognitive function and mood. Although a detailed review of these neurobiologic mechanisms is beyond the scope of the current review, an improved understanding of how common neurobiologic substrates and processes, including those related to cortical and subcortical structure and function and neurogenesis,60,61 mediate cognition and mood will be critical to furthering the field’s development of treatments for cognitive impairment in depression. Because the methods for studying cognition in MDD has varied substantially been studied differently, the construct of cognition has been deemed important enough for the National Institutes of Health to consider it a key component of the Research Domain Criteria initiative.
Considering all these concerns, it is difficult to draw any firm conclusions regarding differential effects of antidepressant pharmacotherapy on various cognitive domains. Our own tally of results from the individual studies included in this review yielded no consistent patterns. However, a review of the sample weighted mean effect size data for cognitive domains across studies suggested that verbal memory was slightly improved with monotherapy and that visual and verbal memory were improved by augmentation therapy. Additionally, there was evidence for improved executive function and cognitive control with augmentation therapy. Overall, the results summarized in this review may be interpreted cautiously as suggesting that antidepressant pharmacotherapy may have a beneficial effect on cognitive impairment associated with MDD.
CONCLUSIONS
There is a need for more systematic examination of the cognitive effects of pharmacotherapy in MDD, similar to the examination already underway in schizophrenia. Larger-scale, longer-term, placebo-controlled studies are warranted to assess drug effects on cognitive function in populations representative of the general MDD population. Furthermore, study designs and statistical methods that maximize test validity and minimize the influence of confounding factors should be used. Finally, more rigorous research into treatment effects on specific domains of cognitive function would be valuable.
Based on a literature review and effect size analysis, it appears that some antidepressant pharmacotherapies can help improve cognitive deficits associated with MDD, including augmentation therapies when the response to antidepressant monotherapy is inadequate, with certain types of memory showing the most consistent improvement with pharmacotherapy. As in other clinical conditions in which an appropriate regimen of monotherapy has resulted in only partial response, augmentation therapy, especially with an agent that employs a different mechanism of action, may be a more sensible strategy than dose escalation (which could increase the incidence and/or severity of adverse effects) or drug switching (which could yield results no better than those obtained with the initial drug and which could incur adverse events not seen with the initial drug). Further research should seek to overcome substantive methodological limitations of prior investigations and to confirm whether pharmacotherapy would be beneficial for the treatment of cognitive deficits associated with MDD.
Drug names: apomorphine (Apokyn), aripiprazole (Abilify), donepezil (Aricept and others), duloxetine (Cymbalta), escitalopram (Lexapro and others), fluoxetine (Prozac and others), galantamine (Razadyne), ketamine (Ketalar and others), lisdexamfetamine (Vyvanse), lithium (Lithobid and others), nortriptyline (Pamelor, Aventyl, and others), paroxetine (Paxil, Pexeva, and others), sertraline (Zoloft and others), spironolactone (Aldactone and others).
Author affiliations: Department of Psychiatry and Behavioral Sciences (Drs Keefe, McClintock, and Doraiswamy), Department of Medicine (Dr Doraiswamy), and the Duke Institute for Brain Sciences (Dr Doraiswamy), Duke University Medical Center, Durham; Department of Psychiatry, UT Southwestern Medical Center, Dallas (Dr McClintock); Department of Psychiatry, Geisel School of Medicine at Dartmouth, Lebanon, New Hampshire (Dr Roth); Complete Healthcare Communications, Inc (CHC), Chadds Ford, Pennsylvania (Mr Tiger); and Global Medical Affairs, Shire Development LLC, Wayne, Pennsylvania (Dr Madhoo). Mr Tiger is now retired.
Author contributions: The concept for this manuscript was developed by the authors in conjunction with Shire Development LLC (Wayne, Pennsylvania). The authors exercised full control over the content of the manuscript and in the final decision to submit to The Journal of Clinical Psychiatry.
Potential conflicts of interest: Dr Keefe currently receives or in the past 3 years has received investigator-initiated research funding support from the Department of Veterans Affairs, Feinstein Institute for Medical Research, GlaxoSmithKline, National Institute of Mental Health, Novartis, Psychogenics, Research Foundation for Mental Hygiene, Inc, and the Singapore National Medical Research Council. He currently receives or in the past 3 years has received honoraria, served as a consultant, or advisory board member for Abbott, Abbvie, Akebia, Amgen, Astellas, Asubio, AviNeuro/ChemRar, BiolineRx, Biomarin, Boehringer-Ingelheim, Bristol-Myers Squibb, Eli Lilly, EnVivo, Helicon, Lundbeck, Merck, Mitsubishi, Novartis, Otsuka, Pfizer, Roche, Shire, Sunovion, Takeda, Targacept. Dr Keefe receives royalties from the BACS testing battery and the MATRICS Battery (BACS Symbol Coding). He is also a shareholder in NeuroCog Trials, Inc. Dr McClintock has received research support from the National Institutes of Health, National Center for Research Resources, and the Brain and Behavior Foundation; he has also served as a consultant for Shire. Dr Roth is an author of the Behavior Rating Inventory of Executive Function-Adult Version (BRIEF-A) and receives a royalty from the publisher and has served as a research consultant to Shire. Dr Doraiswamy has received research grants (through Duke University) from Elan, Avid, Lilly, Novartis, Neuronetrix, Medivation, Wyeth, Janssen, Pfizer, and National Institutes of Health. He has received advisory or speaking fees from Accera, Avid, AstraZeneca, Abbvie, Baxter, Cognoptix, Lundbeck, Takeda, Piramal, Genomind, Genentech, Sonexa, Shire, Targacept, Grifols, Neuronetrix, TauRx, Medivation, Danone, Neurocog Trials, Alzheimer’s Association, Alzheimer’s Foundation, University of California, National University of Singapore, and University of Copenhagen. He owns shares in Maxwell Health, Sonexa, Clarimedix, and Adverse Events Inc, whose products are not discussed here. Mr Tiger was a full-time employee of CHC at the time this manuscript was developed. CHC was funded by Shire Development LLC for the development of this manuscript. Dr Madhoo is an employee of Shire Development LLC and holds stock and/or stock options in Shire.
Funding/support: The concept for this manuscript was developed by Shire Development LLC (Wayne, Pennsylvania) in conjunction with the authors. Under the direction of the authors, Craig Slawecki, PhD, an employee of CHC, provided proofreading, copyediting, fact checking, and formatting assistance for this manuscript. CHC was funded by Shire Development LLC for the development of this manuscript. Editorial assistance was funded by Shire Development LLC. Statistical support for this manuscript was provided by Ben Adeyi, MS, from Shire Development LLC. Brian Scheckner, PharmD, an employee of Shire Development LLC (Wayne, Pennsylvania), also reviewed and edited the manuscript for scientific accuracy.
Role of the sponsor: The authors exercised full control over the content of the manuscript and in the final decision to submit to The Journal of Clinical Psychiatry.
Supplementary material: Available at PSYCHIATRIST.COM.
REFERENCES
1. Marazziti D, Consoli G, Picchetti M, et al. Cognitive impairment in major depression. Eur J Pharmacol. 2010;626(1):83-86. PubMed doi:10.1016/j.ejphar.2009.08.046
2. Sierksma AS, van den Hove DL, Steinbusch HW, et al. Major depression, cognitive dysfunction and Alzheimer’s disease: is there a link? Eur J Pharmacol. 2010;626(1):72-82. PubMed doi:10.1016/j.ejphar.2009.10.021
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Washington, DC: American Psychiatric Association; 2013.
4. Maalouf FT, Brent D, Clark L, et al. Neurocognitive impairment in adolescent major depressive disorder: state vs trait illness markers. J Affect Disord. 2011;133(3):625-632. PubMed doi:10.1016/j.jad.2011.04.041
5. McClintock SM, Cullum M, Husain MM, et al. Evaluation of the effects of severe depression on global cognitive function and memory. CNS Spectr. 2010;15(5):304-313. PubMed
6. Gorlyn M, Keilp J, Burke A, et al. Neurocognitive impairment in MDD is an independent component of symptom severity. Biol Psychiatry. 2012;71(suppl 1):76S.
7. Fava M. Symptoms of fatigue and cognitive/executive dysfunction in major depressive disorder before and after antidepressant treatment. J Clin Psychiatry. 2003;64(suppl 14):30-34. PubMed
8. Gualtieri CT, Johnson LG, Benedict KB. Neurocognition in depression: patients on and off medication versus healthy comparison subjects. J Neuropsychiatry Clin Neurosci. 2006;18(2):217-225. PubMed doi:10.1176/appi.neuropsych.18.2.217
9. Reppermund S, Ising M, Lucae S, et al. Cognitive impairment in unipolar depression is persistent and non-specific: further evidence for the final common pathway disorder hypothesis. Psychol Med. 2009;39(4):603-614. PubMed doi:10.1017/S003329170800411X
10. Trivedi MH, Rush AJ, Wisniewski SR, et al; STAR*D Study Team. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. 2006;163(1):28-40. PubMed doi:10.1176/appi.ajp.163.1.28
11. McClintock SM, Husain MM, Wisniewski SR, et al. Residual symptoms in depressed outpatients who respond by 50% but do not remit to antidepressant medication. J Clin Psychopharmacol. 2011;31(2):180-186. PubMed doi:10.1097/JCP.0b013e31820ebd2c
12. Alves TC, Rays J, Telles RM, et al. Effects of antidepressant treatment on cognitive performance in elderly subjects with heart failure and comorbid major depression: an exploratory study. Psychosomatics. 2007;48(1):22-30. PubMed doi:10.1176/appi.psy.48.1.22
13. Austin MP, Mitchell P, Hadzi-Pavlovic D, et al. Effect of apomorphine on motor and cognitive function in melancholic patients: a preliminary report. Psychiatry Res. 2000;97(2-3):207-215. PubMed doi:10.1016/S0165-1781(00)00222-5
14. Bondareff W, Alpert M, Friedhoff AJ, et al. Comparison of sertraline and nortriptyline in the treatment of major depressive disorder in late life. Am J Psychiatry. 2000;157(5):729-736. PubMed doi:10.1176/appi.ajp.157.5.729
15. Brown ES, Bobadilla L, Nejtek VA, et al. Open-label nefazodone in patients with a major depressive episode and alcohol dependence. Prog Neuropsychopharmacol Biol Psychiatry. 2003;27(4):681-685. PubMed doi:10.1016/S0278-5846(03)00079-4
16. Deuschle M, Kniest A, Niemann H, et al. Impaired declarative memory in depressed patients is slow to recover: clinical experience. Pharmacopsychiatry. 2004;37(4):147-151. PubMed doi:10.1055/s-2004-827168
17. Doraiswamy PM, Krishnan KR, Oxman T, et al. Does antidepressant therapy improve cognition in elderly depressed patients? J Gerontol A Biol Sci Med Sci. 2003;58(12):M1137-M1144. PubMed doi:10.1093/gerona/58.12.M1137
18. Farabaugh AH, Sonawalla SB, Fava M, et al. Differences in cognitive factors between “true drug” versus “placebo pattern” response to fluoxetine as defined by pattern analysis. Hum Psychopharmacol. 2006;21(4):221-225. PubMed doi:10.1002/hup.765
19. Ferguson JM, Wesnes KA, Schwartz GE. Reboxetine versus paroxetine versus placebo: effects on cognitive functioning in depressed patients. Int Clin Psychopharmacol. 2003;18(1):9-14. PubMed doi:10.1097/00004850-200301000-00002
20. Finkel SI, Richter EM, Clary CM, et al. Comparative efficacy of sertraline vs fluoxetine in patients age 70 or over with major depression. Am J Geriatr Psychiatry. 1999;7(3):221-227. PubMed doi:10.1097/00019442-199908000-00006
21. Gorenstein C, de Carvalho SC, Artes R, et al. Cognitive performance in depressed patients after chronic use of antidepressants. Psychopharmacology (Berl). 2006;185(1):84-92. PubMed doi:10.1007/s00213-005-0274-2
22. Herrera-Guzmán I, Gudayol-Ferré E, Herrera-Guzmán D, et al. Effects of selective serotonin reuptake and dual serotonergic-noradrenergic reuptake treatments on memory and mental processing speed in patients with major depressive disorder. J Psychiatr Res. 2009;43(9):855-863. PubMed doi:10.1016/j.jpsychires.2008.10.015
23. Newhouse PA, Krishnan KR, Doraiswamy PM, et al. A double-blind comparison of sertraline and fluoxetine in depressed elderly outpatients. J Clin Psychiatry. 2000;61(8):559-568. PubMed doi:10.4088/JCP.v61n0804
24. Nickel T, Sonntag A, Schill J, et al. Clinical and neurobiological effects of tianeptine and paroxetine in major depression. J Clin Psychopharmacol. 2003;23(2):155-168. PubMed doi:10.1097/00004714-200304000-00008
25. Raskin J, Wiltse CG, Siegal A, et al. Efficacy of duloxetine on cognition, depression, and pain in elderly patients with major depressive disorder: an 8-week, double-blind, placebo-controlled trial. Am J Psychiatry. 2007;164(6):900-909. PubMed doi:10.1176/appi.ajp.164.6.900
26. Richardson JS, Keegan DL, Bowen RC, et al. Verbal learning by major depressive disorder patients during treatment with fluoxetine or amitriptyline. Int Clin Psychopharmacol. 1994;9(1):35-40. PubMed doi:10.1097/00004850-199400910-00006
27. Sato S, Yamakawa Y, Terashima Y, et al. Efficacy of milnacipran on cognitive dysfunction with post-stroke depression: preliminary open-label study. Psychiatry Clin Neurosci. 2006;60(5):584-589. PubMed doi:10.1111/j.1440-1819.2006.01562.x
28. Spalletta G, Caltagirone C. Sertraline treatment of post-stroke major depression: an open study in patients with moderate to severe symptoms. Funct Neurol. 2003;18(4):227-232. PubMed
29. Spalletta G, Ripa A, Bria P, et al. Response of emotional unawareness after stroke to antidepressant treatment. Am J Geriatr Psychiatry. 2006;14(3):220-227. PubMed doi:10.1097/01.JGP.0000194647.72654.a1
30. Wise TN, Wiltse CG, Iosifescu DV, et al. The safety and tolerability of duloxetine in depressed elderly patients with and without medical comorbidity. Int J Clin Pract. 2007;61(8):1283-1293. PubMed doi:10.1111/j.1742-1241.2007.01476.x
31. Wroolie TE, Williams KE, Keller J, et al. Mood and neuropsychological changes in women with midlife depression treated with escitalopram. J Clin Psychopharmacol. 2006;26(4):361-366. PubMed doi:10.1097/01.jcp.0000227699.26375.f8
32. Devanand DP, Pelton GH, Marston K, et al. Sertraline treatment of elderly patients with depression and cognitive impairment. Int J Geriatr Psychiatry. 2003;18(2):123-130. PubMed doi:10.1002/gps.802
33. Herrera-Guzmán I, Herrera-Abarca JE, Gudayol-Ferré E, et al. Effects of selective serotonin reuptake and dual serotonergic-noradrenergic reuptake treatments on attention and executive functions in patients with major depressive disorder. Psychiatry Res. 2010;177(3):323-329. PubMed doi:10.1016/j.psychres.2010.03.006
34. Reilly JL, Lencer R, Thase ME, et al. Effects of sertraline on neurocognition in outpatients with major depressive disorder. Neuropsychopharmacology. 2011;36(suppl 1):S117-S118.
35. Culang-Reinlieb ME, Sneed JR, Keilp JG, et al. Change in cognitive functioning in depressed older adults following treatment with sertraline or nortriptyline. Int J Geriatr Psychiatry. 2012;27(8):777-784. PubMed doi:10.1002/gps.2783
36. Alev L, Altin M, Ozbek K, et al. Effect of duloxetine on functional outcomes in patients with major depressive disorder. Klinik Psikofarmakoloji Bulteni. 2011;21:S115.
37. Chang HH, Lee IH, Gean PW, et al. Treatment response and cognitive impairment in major depression: association with C-reactive protein. Brain Behav Immun. 2012;26(1):90-95. PubMed doi:10.1016/j.bbi.2011.07.239
38. Hashemian F, Tabatabayi MS, Sharifi A, et al. Comparison of the effects of bupropion and fluoxetine on reaction time in adults with major depressive disorder in a 4-week, single-blind study. Klinik Psikofarmakoloji Bulteni. 2011;21:S113.
39. Katona C, Hansen T, Olsen CK. A randomised, double-blind, placebo-controlled, active-referenced study of the multimodal antidepressant Lu AA21004 in the treatment of elderly depressed patients. Eur Neuropsychopharmacol. 2012;22:S258-S259. doi:10.1016/S0924-977X(12)70388-7
40. Murrough JW, Wan LB, Glicksberg B, et al. Ketamine and neurocognition in depression: the modulating effects of lamotrigine. Neuropsychopharmacology. 2012;38:S375.
41. Boeker H, Schulze J, Richter A, et al. Sustained cognitive impairments after clinical recovery of severe depression. J Nerv Ment Dis. 2012;200(9):773-776. PubMed doi:10.1097/NMD.0b013e318266ba14
42. Culang ME, Sneed JR, Keilp JG, et al. Change in cognitive functioning following acute antidepressant treatment in late-life depression. Am J Geriatr Psychiatry. 2009;17(10):881-888. PubMed doi:10.1097/JGP.0b013e3181b4bf4a
43. Mathew SJ, Murrough JW, aan het Rot M, et al. Riluzole for relapse prevention following intravenous ketamine in treatment-resistant depression: a pilot randomized, placebo-controlled continuation trial. Int J Neuropsychopharmacol. 2010;13(1):71-82. PubMed doi:10.1017/S1461145709000169
44. Madhoo M, Keefe RSE, Roth RM, et al. Efficacy and safety of lidexamfetamine dimesylate in adults with executive dysfunction and partial or full remission of major depressive disorder. Int J Neuropsychopharmacol. 2012;15(suppl):189.
45. DeBattista C, Lembke A, Solvason HB, et al. A prospective trial of modafinil as an adjunctive treatment of major depression. J Clin Psychopharmacol. 2004;24(1):87-90. PubMed doi:10.1097/01.jcp.0000104910.75206.b9
46. Elgamal S, MacQueen G. Galantamine as an adjunctive treatment in major depression. J Clin Psychopharmacol. 2008;28(3):357-359. PubMed doi:10.1097/JCP.0b013e318172756c
47. Holtzheimer PE 3rd, Meeks TW, Kelley ME, et al. A double blind, placebo-controlled pilot study of galantamine augmentation of antidepressant treatment in older adults with major depression. Int J Geriatr Psychiatry. 2008;23(6):625-631. PubMed doi:10.1002/gps.1951
48. Kok RM, Vink D, Heeren TJ, et al. Lithium augmentation compared with phenelzine in treatment-resistant depression in the elderly: an open, randomized, controlled trial. J Clin Psychiatry. 2007;68(8):1177-1185. PubMed doi:10.4088/JCP.v68n0803
49. Morgan ML, Cook IA, Rapkin AJ, et al. Estrogen augmentation of antidepressants in perimenopausal depression: a pilot study. J Clin Psychiatry. 2005;66(6):774-780. PubMed doi:10.4088/JCP.v66n0617
50. Politis AM, Papadimitriou GN, Theleritis CG, et al. Combination therapy with amisulpride and antidepressants: clinical observations in case series of elderly patients with psychotic depression. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(5):1227-1230. PubMed doi:10.1016/j.pnpbp.2008.03.011
51. Reynolds CF 3rd, Butters MA, Lopez O, et al. Maintenance treatment of depression in old age: a randomized, double-blind, placebo-controlled evaluation of the efficacy and safety of donepezil combined with antidepressant pharmacotherapy. Arch Gen Psychiatry. 2011;68(1):51-60. PubMed doi:10.1001/archgenpsychiatry.2010.184
52. Pelton GH, Harper OL, Tabert MH, et al. Randomized double-blind placebo-controlled donepezil augmentation in antidepressant-treated elderly patients with depression and cognitive impairment: a pilot study. Int J Geriatr Psychiatry. 2008;23(7):670-676. PubMed doi:10.1002/gps.1958
53. Hinkelmann K, Moritz S, Botzenhardt J, et al. Changes in cortisol secretion during antidepressive treatment and cognitive improvement in patients with major depression: a longitudinal study. Psychoneuroendocrinology. 2012;37(5):685-692. PubMed doi:10.1016/j.psyneuen.2011.08.012
54. Greer TL, Sunderajan P, Grannemann BD, et al. Aripiprazole augmentation improves aspects of executive function in major depressive disorder: a pilot study. Neuropsychopharmacology. 2011;36(suppl 1):S350-S351.
55. Levkovitz Y, Alpert JE, Brintz CE, et al. Effects of S-adenosylmethionine augmentation of serotonin-reuptake inhibitor antidepressants on cognitive symptoms of major depressive disorder. J Affect Disord. 2012;136(3):1174-1178. PubMed doi:10.1016/j.jad.2011.04.059
56. Strauss E, Sherman EMS, Spreen O. A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary. 3rd ed. New York, NY: Oxford University Press; 2006.
57. Barry D, Bates ME, Labouvie E. FAS and CFL forms of verbal fluency differ in difficulty: a meta-analytic study. Appl Neuropsychol. 2008;15(2):97-106. PubMed doi:10.1080/09084280802083863
58. Green MF, Nuechterlein KH, Gold JM, et al. Approaching a consensus cognitive battery for clinical trials in schizophrenia: the NIMH-MATRICS conference to select cognitive domains and test criteria. Biol Psychiatry. 2004;56(5):301-307. PubMed doi:10.1016/j.biopsych.2004.06.023
59. Snyder HR. Major depressive disorder is associated with broad impairments on neuropsychological measures of executive function: a meta-analysis and review. Psychol Bull. 2013;139(1):81-132. PubMed doi:10.1037/a0028727
60. Femen×a T, Gómez-Galán M, Lindskog M, et al. Dysfunctional hippocampal activity affects emotion and cognition in mood disorders. Brain Res. 2012;1476:58-70. PubMed doi:10.1016/j.brainres.2012.03.053
61. Duman RS, Aghajanian GK. Synaptic dysfunction in depression: potential therapeutic targets. Science. 2012;338(6103):68-72. PubMed doi:10.1126/science.1222939
This PDF is free for all visitors!