Objective: The primary objective was to evaluate the safety and tolerability of lurasidone, a new atypical antipsychotic agent, in the longer-term treatment of schizophrenia (DSM-IV). Persistence of symptom improvement was assessed as a secondary outcome.
Method: Patients who completed a 6-week, double-blind, placebo-controlled study evaluating the efficacy of fixed doses of once-daily lurasidone (40 or 120 mg) or olanzapine 15 mg (to confirm assay sensitivity) were eligible to receive flexibly dosed lurasidone 40 to 120 mg/d in this 6-month, open-label extension study (conducted from March 2008 to December 2009). Assessments of safety and tolerability were conducted at open-label baseline, at day 10, and monthly thereafter.
Results: Of 254 enrolled patients, 113 (44.5%) completed 6 months of open-label treatment. During the open-label study (month 6 observed cases), small decreases were observed in mean weight (−0.1 kg) and median lipid levels (total cholesterol, −6.5 mg/dL; low-density lipoprotein, 0.0 mg/dL; high-density lipoprotein, 0.0 mg/dL; triglycerides, −8.5 mg/dL). Patients previously treated with olanzapine (n = 69) experienced decrease in weight and improvement in lipid levels, whereas patients previously treated with lurasidone (n = 115) or placebo (n = 62) experienced minimal changes. No clinically meaningful changes were observed in median prolactin levels. The 2 most commonly reported adverse events were akathisia (13.0%) and insomnia (11.0%). Persistent antipsychotic efficacy of lurasidone was shown for patients who had previously received lurasidone, olanzapine, or placebo; further reductions from open-label baseline to final visit were observed in mean PANSS total score (−8.7) for all patients.
Conclusions: Open-label treatment with flexibly dosed lurasidone (40-120 mg/d) was generally safe, well tolerated, and effective over a 6-month period in patients who had completed a preceding 6-week, double-blind study.
Trial Registration: ClinicalTrials.gov identifier: NCT00615433
J Clin Psychiatry
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: August 7, 2012; accepted February 15, 2013.
Online ahead of print: March 13, 2013 (doi:10.4088/JCP.12m08084).
Corresponding author: Stephen M. Stahl, MD, PhD, 1930 Palomar Point Way, Ste 103, Carlsbad, CA 92008 ([email protected]).
Effectiveness of Lurasidone for Patients With Schizophrenia Following 6 Weeks of Acute Treatment With Lurasidone, Olanzapine, or Placebo: A 6-Month, Open-Label, Extension Study
ABSTRACT
Objective: The primary objective was to evaluate
the safety and tolerability of lurasidone, a new atypical antipsychotic agent, in the longer-term treatment of schizophrenia (DSM-IV). Persistence of symptom improvement was assessed as a secondary outcome.
Method: Patients who completed a 6-week, double-blind, placebo-controlled study evaluating the efficacy of fixed doses of once-daily lurasidone (40 or 120 mg) or olanzapine 15 mg (to confirm assay sensitivity) were eligible to receive flexibly dosed lurasidone 40 to 120 mg/d in this 6-month, open-label extension study (conducted from March 2008 to December 2009). Assessments of safety and tolerability were conducted at open-label baseline, at day 10, and monthly thereafter.
Results: Of 254 enrolled patients, 113 (44.5%) completed 6 months of open-label treatment. During the open-label study (month 6 observed cases), small decreases were observed in mean weight (−0.1 kg) and median lipid levels (total cholesterol, −6.5 mg/dL; low-density lipoprotein, 0.0 mg/dL; high-density lipoprotein, 0.0 mg/dL; triglycerides, −8.5 mg/dL). Patients previously treated with olanzapine (n = 69) experienced decrease in weight and improvement in lipid levels, whereas patients previously treated with lurasidone (n = 115) or placebo (n = 62) experienced minimal changes. No clinically meaningful changes were observed in median prolactin levels. The 2 most commonly reported adverse events were akathisia (13.0%) and insomnia (11.0%). Persistent antipsychotic efficacy of lurasidone was shown for patients who had previously received lurasidone, olanzapine, or placebo; further reductions from open-label baseline to final visit were observed
in mean PANSS total score (−8.7) for all patients.
Conclusions: Open-label treatment with flexibly dosed lurasidone (40–120 mg/d) was generally safe, well tolerated, and effective over a 6-month period in patients who had completed a preceding 6-week, double-blind study.
Trial Registration: ClinicalTrials.gov identifier: NCT00615433
J Clin Psychiatry
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: August 7, 2012; accepted February 15, 2013.
Online ahead of print: March 13, 2013 (doi:10.4088/JCP.12m08084).
Corresponding author: Stephen M. Stahl, MD, PhD, 1930 Palomar Point Way, Ste 103, Carlsbad, CA 92008 ([email protected]).
Lurasidone is an atypical antipsychotic agent that acts as an antagonist with high affinity for the 5-hydroxytryptamine (5-HT)2A, dopamine D2, and 5-HT7 receptors.1 Lurasidone has shown efficacy in the treatment of acute exacerbation of schizophrenia in a series of short-term, placebo-controlled studies.2–6 Although treatment with some atypical antipsychotics has been associated with weight gain, elevated blood glucose, and dyslipidemia,7–9 treatment with lurasidone has not been associated with significant disturbances in lipids or glycemic control and had minimal effect on weight, based on combined data from 5 short-term (6-week), placebo-controlled studies6,10 and data from a 12-month active-controlled study.11 Similarly, 3 weeks to 12 months of treatment with lurasidone was not associated with clinically meaningful changes in the QTc interval.2–4,11
This 6-month, open-label extension study was preceded by a 6-week, double-blind study in which the short-term safety and efficacy of once-daily, fixed doses of lurasidone (40 mg or 120 mg) or olanzapine 15 mg for the treatment of schizophrenia were compared with placebo.4 In the preceding double-blind study, both doses of lurasidone and olanzapine demonstrated comparable efficacy versus placebo, based on change in Positive and Negative Syndrome Scale (PANSS) and Clinical Global Impressions-Severity of Illness (CGI-S) scores. Lurasidone was generally well tolerated in this 6-week trial, although it was associated with higher rates of akathisia, parkinsonism, and dystonia than was olanzapine. Because of the chronicity of schizophrenia and the need for prolonged therapy, it is important to assess the long-term effects of new antipsychotic medications. This 6-month, open-label extension study evaluated the safety and tolerability of once-daily, flexibly dosed lurasidone (40–120 mg/d) for patients who completed the preceding double-blind, placebo-controlled registration study.4
METHOD
Study Design
Detailed methods and results for the preceding 6-week, double-blind, placebo-controlled core study have been published previously.4 Patients who completed the double-blind study were given the option to continue into this 6-month (28-week) open-label extension study, conducted from March 2008 to December 2009, at sites in the United States (n = 24), India (n = 12), Colombia (n = 5), Lithuania (n = 4), and the Philippines (n = 4). Patients who elected to participate in the open-label extension study received a single-blind placebo washout for 3 days. Most patients remained as outpatients during the 3-day washout period; however, inpatient hospitalization was permitted at the discretion of the investigator. All patients then received lurasidone 80 mg/d for 7 days. Following these 7 days and at any subsequent monthly study visits, the dose could be increased or decreased by 40 mg/d based on the judgment of the investigator, up to 4 times. Medication was administered once daily in the morning with food.
Written informed consent was provided by all patients prior to enrollment into this study. The study protocol was approved by an Independent Ethics Committee associated with each study center. The study was conducted in accordance with Good Clinical Practice.12 The study is registered with ClinicalTrials.gov (identifier: NCT00615433).
Assessments
The primary objective of this 6-month, open-label extension study was to assess the long-term safety and tolerability of lurasidone in patients with schizophrenia; in addition, the longer-term efficacy of lurasidone was evaluated.
Assessment visits occurred at baseline of the open-label study, day 10 (following 3-day placebo washout and 7 days of lurasidone 80 mg/d), and monthly thereafter for 6 months. Adverse events were recorded at each visit; assessment of medication tolerability was based, in part, on treatment discontinuation due to adverse events. Extrapyramidal symptoms (EPS) were assessed at each visit with the Simpson-Angus Scale,13 Barnes Akathisia Rating Scale,14 and Abnormal Involuntary Movement Scale.15 Safety evaluation also included laboratory tests (chemistry and hematology panels, lipid panel, glycosylated hemoglobin [HbA1c], and prolactin), electrocardiograms (ECGs), and vital sign measurements. Efficacy was assessed using the PANSS total and subscale scores16 and CGI-S.15
Treatment compliance was evaluated at each monthly visit using tablet counts. Patients were instructed to return with the blister packs dispensed at the previous study visit, and the remaining tablets of study medication were counted. Treatment compliance was calculated as (number of tablets missing)/(number of tablets prescribed) × 100. Patients
were considered compliant if the number of doses missing was within 75%–125% of the prescribed doses. Mean estimated treatment compliance was the average of individual compliance values for patients with available compliance data across all visits. Such tablet counts, as obtained in clinical trials, may not fully reflect actual adherence to treatment.17
Statistical Methods
The primary analysis population comprised all patients who continued into the extension study and received at least 1 lurasidone dose (safety population). For each safety and efficacy measure, change scores were calculated from double-blind baseline to open-label study endpoint and from open-label baseline to open-label study endpoint (month 6). Additional prespecified analyses were conducted for 3 patient cohorts based on randomized treatment assignment in the preceding double-blind study (to lurasidone, olanzapine, or placebo). For patients who did not complete the study, the last postbaseline observation was carried forward to open-label study endpoint for the LOCF analyses as appropriate.
RESULTS
Patient Disposition and Study Treatment
Of the 298 patients who completed the preceding double-blind study, 254 (85.2%) continued into the open-label extension study. Patients entered the open-label extension study from double-blind treatment with lurasidone 40 mg/d or 120 mg/d (n = 118), olanzapine 15 mg/d (n = 71), or placebo (n = 65). Of the 254 patients, 113 (44.5%) completed the open-label extension study (Figure 1). The safety population (N = 246) excluded 8 patients who did not receive any dose of study medication after entering the extension study. The overall discontinuation rate for patients who had initially received placebo was 58.5%, compared with 56.3% for patients who had initially received olanzapine and 53.4% for patients who continued treatment with lurasidone. Adverse events resulted in early discontinuation for 12.6% of patients (14.4% of patients who had received lurasidone, 12.3% of patients who had received placebo, and 9.9% of patients who had received olanzapine). “Insufficient clinical response” was the reason for discontinuation for 6.7% of patients (7.6% of patients who had received lurasidone, 7.0% of patients who had received olanzapine, and 4.6% of patients who had received placebo).
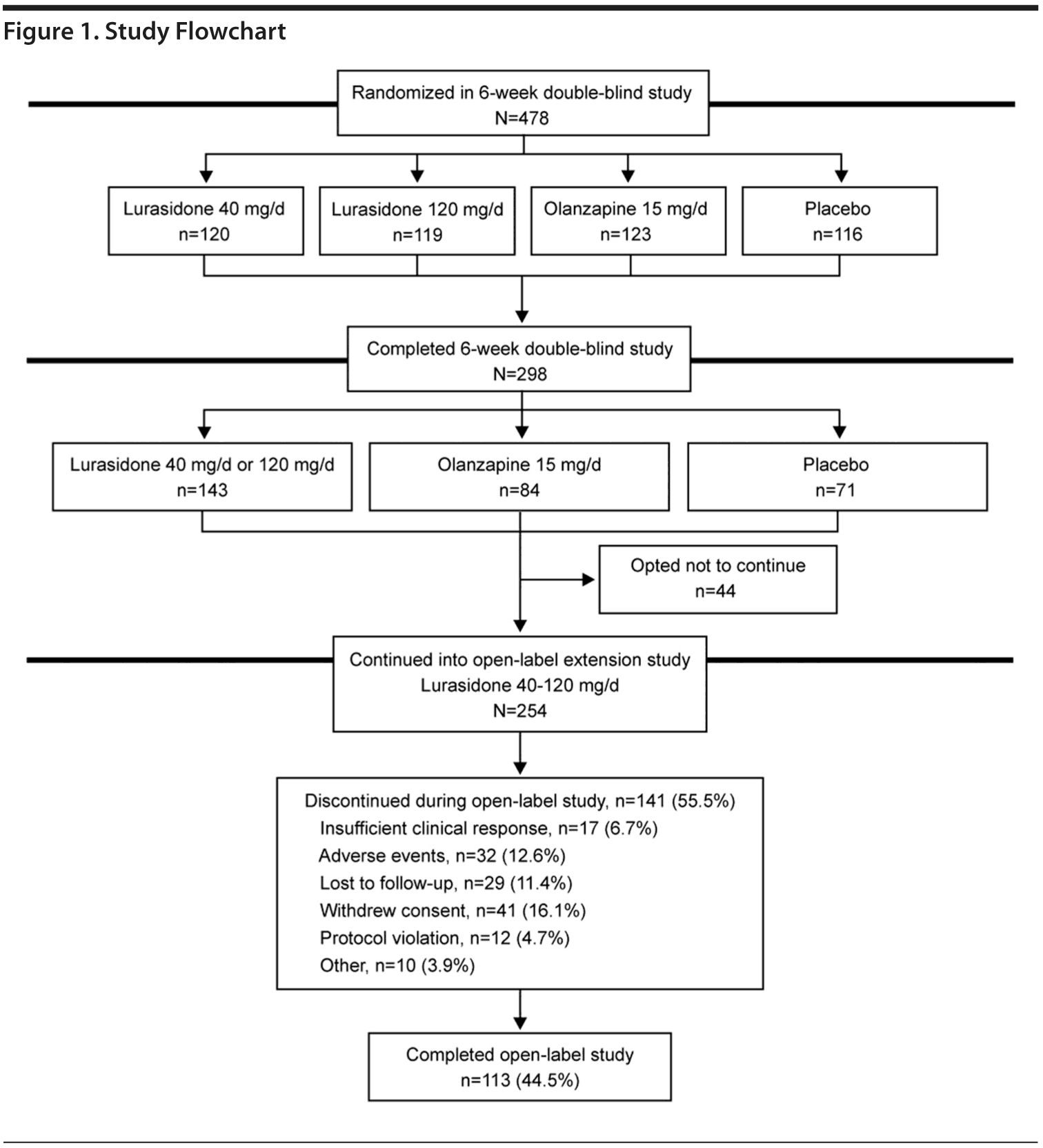
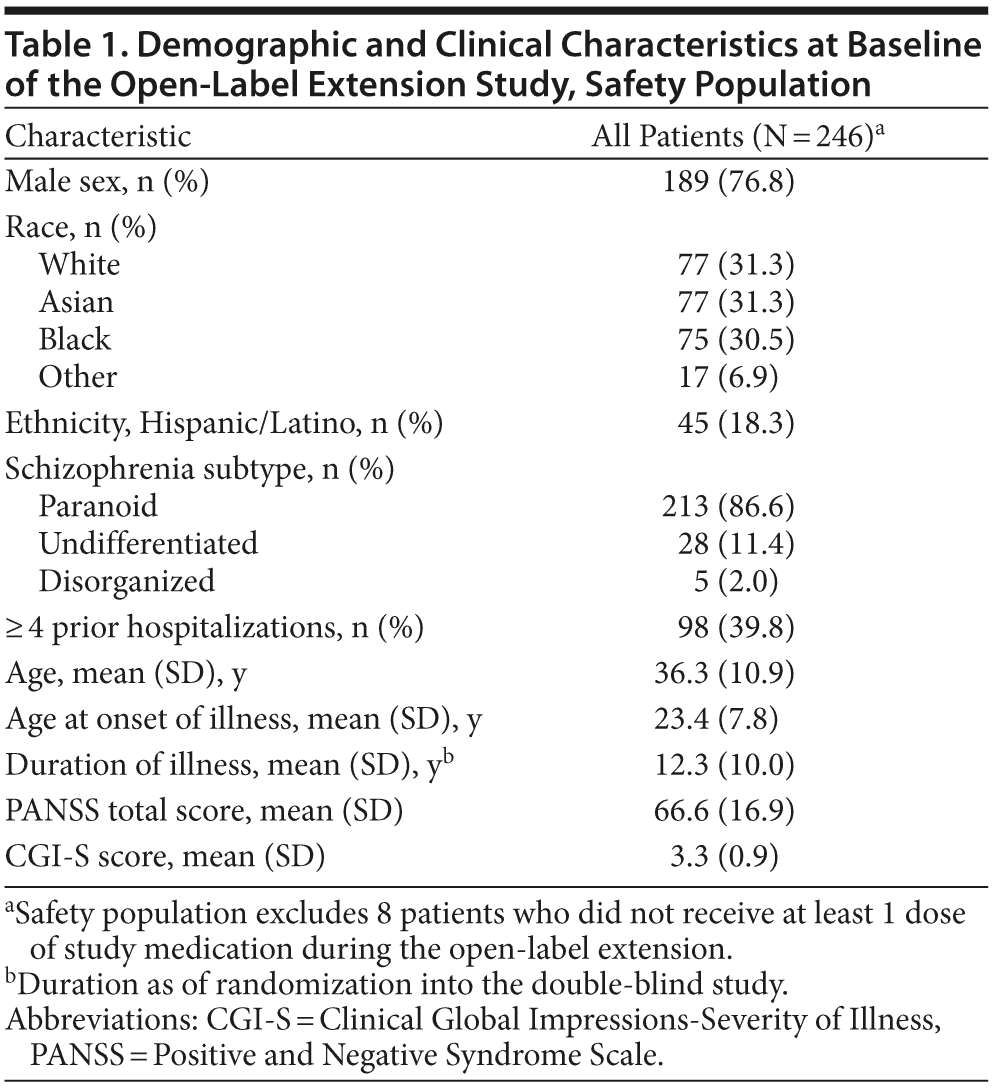
Patient demographic and clinical characteristics at baseline are shown in Table 1 (safety population). The mean daily dose of lurasidone during the open-label extension study was 86.3 mg; the modal dose was 80 mg/d for 63.0% of patients, 40 mg/d for 9.9% of patients, and 120 mg/d for 27.2% of patients. Median duration of lurasidone exposure during the open-label extension study was 152.0 days; study completers received 193 days of open-label treatment. The proportion of patients continuing at 6, 12, 16, and 24 weeks of the extension study was 72.0%, 58.8%, 53.1%, and 46.5%, respectively.
Information about treatment compliance was available for 197 patients (80.1%). Mean treatment compliance for these patients was 99.4%. Seventy-four percent of patients (n = 182) received concomitant medication(s). The most commonly used medications were anxiolytics (35% of patients), anticholinergics (32% of patients), and hypnotics and sedatives (26% of patients).
Safety
Adverse events. During this study, 162 patients (65.9%) reported at least 1 adverse event. The most commonly reported adverse events were akathisia (13.0%), insomnia (11.0%), nausea (9.8%), somnolence (9.8%), and parkinsonism (9.3%) (Table 2). The majority of adverse events were classified as mild or moderate. Thirty of 246 patients in the safety population (12.2%) discontinued because of an adverse event. The most common adverse events resulting in discontinuation were psychiatric disorders (n = 16 [6.5%]) and nervous system disorders (n = 8 [3.3%]).
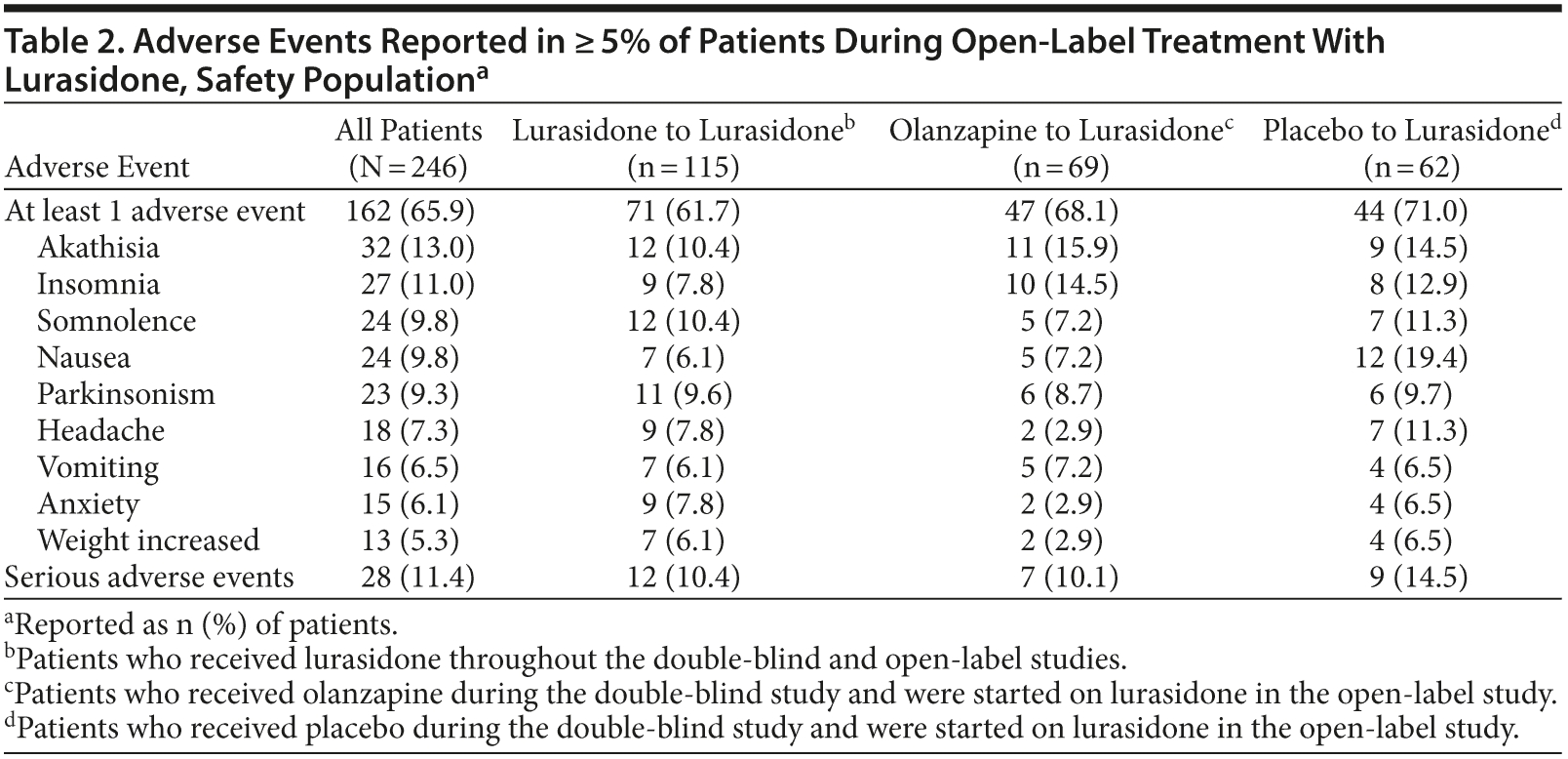
Twenty-eight patients (11.4%) experienced serious adverse events (14.5% of patients who had received placebo, 10.4% of patients who had received lurasidone, and 10.1%
of patients who had received olanzapine); no deaths occurred. Worsening of schizophrenia (n = 9 [3.7%]) and psychotic disorder or acute psychosis (n = 7 [2.8%]) were the 2 most common serious adverse events. In addition, there was 1 case (0.4%) of suicidal ideation and 1 (0.4%) suicide attempt.
In general, the frequency of adverse events appeared not to be related to treatment assignment during the preceding double-blind study, with the exception of nausea, which was reported in 19.4% of patients who had received placebo during the double-blind study and were started on lurasidone (Table 2). Half of these reports of nausea occurred during the first 3 weeks of treatment with lurasidone in the open-label extension study.
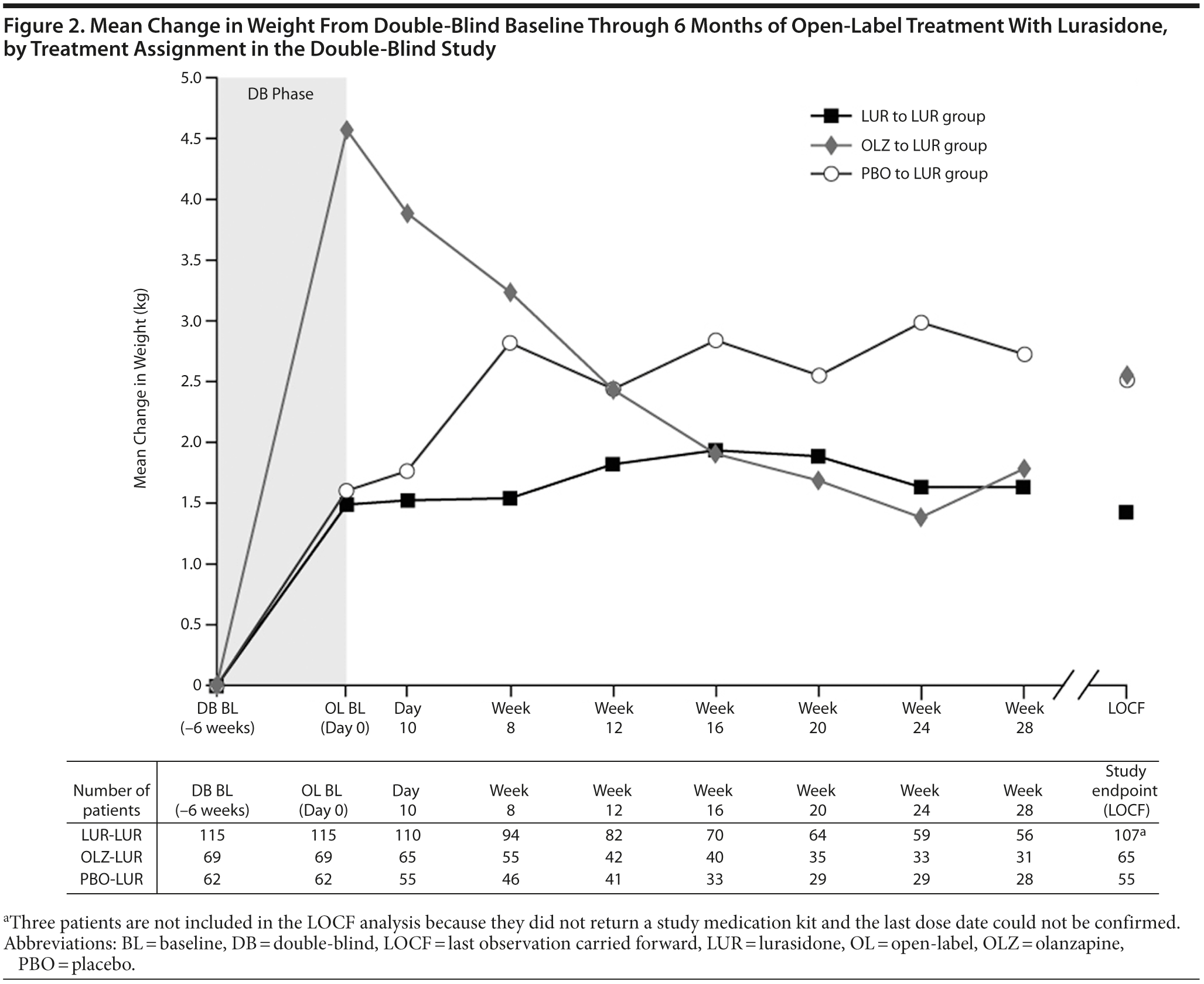
Body weight, body mass index, and waist circumference. For patients who continued into the open-label extension phase, mean weight gain (observed cases [OC]) during the preceding 6-week, double-blind study, which included at least 3 weeks of inpatient hospitalization, was similar for patients receiving lurasidone (1.5 kg, n = 115) or placebo (1.6 kg, n = 62), but higher for patients receiving olanzapine (4.6 kg, n = 69) (Figure 2). Patients who received lurasidone during both the double-blind and open-label studies experienced mean weight gain of 1.6 kg during 8 months of lurasidone exposure (OC, n = 56). For all patients in the open-label extension study, there was a slight mean decrease in weight (−0.1 kg) from open-label baseline to study endpoint (OC). During the open-label extension study, mean weight (OC) was generally stable for patients continuing on lurasidone (+0.4 kg, n = 56) and increased slightly (+0.9 kg, n = 28) for patients who had received placebo during the preceding double-blind study (Table 3; Figure 2). In contrast, patients who had received olanzapine during the preceding double-blind study experienced a reduction in mean weight (−1.9 kg, n = 31) during open-label treatment with lurasidone. A similar pattern of results was observed for body mass index. Overall, 8.7% of patients experienced ≥ 7% weight gain and 13.9% of patients experienced ≥ 7% weight loss during the open-label extension study (OC; Table 3); a ≥ 7% weight increase was most common in patients who had received placebo during the preceding double-blind study (17.9%), and a ≥ 7% weight reduction was most common in patients who had received olanzapine (29.0%).
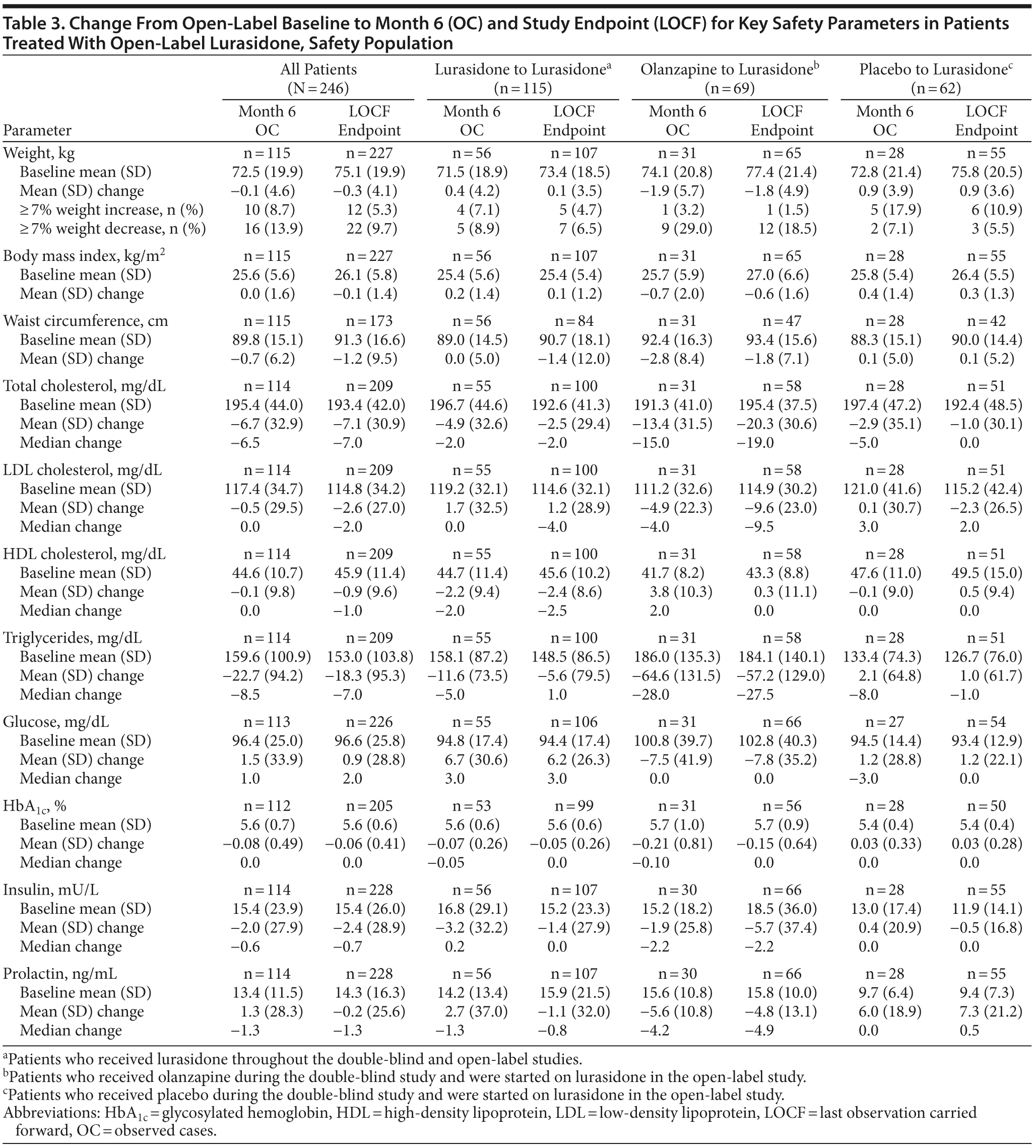
Metabolic parameters. Lipids. For patients who continued into the open-label extension phase, median changes in lipid levels during the preceding 6-week, double-blind study (OC) were generally small and similar for patients who received lurasidone (−3.0 mg/dL for total cholesterol, −3.0 mg/dL
for low-density lipoprotein [LDL] cholesterol, 0.0 mg/dL for high-density lipoprotein [HDL] cholesterol, +1.0 mg/dL for triglycerides) or placebo (−1.5 mg/dL for total cholesterol, 0.0 mg/dL for LDL cholesterol, −0.5 mg/dL for HDL cholesterol, +1.5 mg/dL for triglycerides), whereas patients who received olanzapine had increases in median total cholesterol (+15.0 mg/dL), LDL cholesterol (+7.0 mg/dL), and triglycerides (+27.0 mg/dL) and a decrease in median HDL cholesterol (−2.0 mg/dL) during the double-blind study.
For patients who received lurasidone during both the double-blind and open-label studies, changes in median lipid levels after 8 months of lurasidone exposure (OC) were −12.0 mg/dL for total cholesterol, −2.0 mg/dL for LDL cholesterol, −2.0 mg/dL for HDL cholesterol, and +4.0 mg/dL for triglycerides. During open-label treatment with lurasidone, patients who had received olanzapine in the double-blind study experienced larger changes in median lipid levels (−15.0 mg/dL in total cholesterol, −4.0 mg/dL in LDL cholesterol, and −28.0 mg/dL in triglycerides; OC) compared with patients who had received lurasidone during the double-blind study (−2.0 mg/dL for total cholesterol, 0.0 mg/dL for LDL cholesterol, and −5.0 mg/dL for triglycerides; OC) (Table 3). Median HDL cholesterol increased (+2.0) in the olanzapine-to-lurasidone subgroup and decreased (−2.0 mg/dL) for patients continuing on lurasidone (OC). The placebo-to-lurasidone subgroup showed a decrease in median total cholesterol (−5.0 mg/dL) and triglycerides (−8.0 mg/dL) during the open-label extension study, a slight increase (+3.0 mg/dL) in LDL cholesterol, and no change in HDL cholesterol (OC).
For patients who had received placebo during the double-blind study, a shift from normal values at open-label baseline to high values at LOCF endpoint was observed in 9.8% of patients for total cholesterol (high defined as > 200 mg/dL), 7.8% of patients for LDL cholesterol (high defined as > 129 mg/dL), and 2.0% of patients for triglycerides (high defined as > 203 mg/dL); 5.9% of patients who had received placebo shifted from normal to low values of HDL cholesterol (low defined as < 35 mg/dL) in the open-label study. For patients who had received olanzapine, shifts were noted in 6.9%, 5.2%, 8.6%, and 3.4% of patients for total cholesterol, HDL cholesterol, LDL cholesterol, and triglycerides, respectively; for patients continuing on lurasidone, shifts were experienced by 12.0%, 19.0%, 14.0%, and 8.0% of patients for the respective lipid parameters. Smaller shifts in lipid parameters for the cohort previously treated with olanzapine (compared with those previously treated with lurasidone) may have been due to the effects of olanzapine treatment on these parameters during the preceding 6-week, double-blind study.
Glucose, insulin, and glycosylated hemoglobin. For patients who continued into the open-label extension phase, median changes in glucose, insulin, and HbA1c levels during the preceding double-blind study were small in every treatment group (OC). Median changes during the open-label extension study were similarly small (Table 3); the proportion of patients who experienced a shift from normal value to high value (LOCF) was 16.8% for glucose (high defined as > 99 mg/dL), 7.0% for insulin (high defined as > 28.0 mU/L), and 1.9% for HbA1c (high defined as > 6.0%).
Extrapyramidal symptoms. The proportion of patients who reported any EPS-related treatment-emergent adverse event, including akathisia, during the extension study was 26.4%. The most commonly reported EPS-related adverse events were akathisia (13.0%) and parkinsonism (9.3%) (Table 2). There was a low incidence of other EPS-related symptoms such as tremor (2.4%), restlessness (3.3%), and dystonia (3.7%). The incidence of any EPS-related adverse event was similar irrespective of double-blind treatment assignment. Five patients (2.0%) discontinued the study because of an EPS-related adverse event: 3 patients for akathisia (2 who had received olanzapine and 1 who had received lurasidone in the preceding double-blind study), 1 patient (who had initially received lurasidone) for dystonia (torticollis), and 1 patient (who had initially received lurasidone) for tremor. Mean change from open-label baseline to study endpoint (LOCF) was 0.0 on the Simpson-Angus Scale mean score, 0.1 on the Barnes Akathisia Rating Scale global clinical assessment of akathisia, and 0.0 on the Abnormal Involuntary Movement Scale total score.
Prolactin. For patients who continued into the open-label extension phase, median change in prolactin level during the preceding double-blind study was +0.7 ng/mL for patients receiving lurasidone, −0.75 ng/mL for patients receiving placebo, and +3.9 ng/mL for patients receiving olanzapine (OC). For the overall patient population, there was a small decrease in median prolactin level from open-label baseline to study endpoint (−1.3 ng/mL, OC); the median change was −1.3 ng/mL for patients who continued on lurasidone, −4.2 ng/mL for patients who had been treated with olanzapine, and 0.0 ng/mL for patients who had received placebo. A change from normal prolactin levels at open-label baseline to high prolactin levels at LOCF endpoint occurred in 21 male patients (12.1%; criterion for high value > 17.7 ng/mL) and 6 female patients (10.9%; criterion for high value > 29.2 ng/mL).
Electrocardiographic parameters. There were no clinically meaningful changes in mean ECG parameters during the open-label period. No patients had a QTcF interval of > 500 msec. One patient (0.4%) had a ≥ 60-msec increase in QTcF interval from open-label baseline at the month 6 assessment.
Physical examination and vital signs. There were no clinically meaningful changes in physical examination measures or vital signs (heart rate, systolic and diastolic blood pressure, body temperature) during open-label treatment with lurasidone.
Efficacy
Patients who continued into the open-label extension phase had shown substantial improvement in PANSS total score during the preceding double-blind study (−33.0 points for patients receiving lurasidone, −31.6 for patients receiving olanzapine, and −23.1 for patients receiving placebo). In the open-label extension study, patients demonstrated continued improvement in PANSS total score: overall mean change of –8.7 from open-label baseline to month 6 (OC). Mean changes in PANSS total score during the extension study were −12.0 for the placebo-to-lurasidone subgroup, −8.6 for patients who continued on lurasidone, and −5.8 for patients switched from olanzapine to lurasidone. A similar pattern of change was observed for the PANSS positive, negative, and general psychopathology subscales and the CGI-S.
DISCUSSION
This open-label extension study followed a 6-week, double-blind, placebo-controlled study of lurasidone at fixed daily doses of 40 mg or 120 mg, or olanzapine 15 mg/d.4 During the double-blind study, treatment with lurasidone and treatment with olanzapine both produced significant improvement in symptoms in patients experiencing an acute exacerbation of schizophrenia.4 Post hoc analyses showed no significant differences between lurasidone and olanzapine on improvement in PANSS total or CGI-S scores.4 In this 6-month extension study, open-label treatment with lurasidone, flexibly dosed between 40 mg/d and 120 mg/d, was associated with sustained improvement in symptoms
of schizophrenia, with further improvement noted in PANSS total and CGI-S scores at study endpoint compared with open-label baseline. Symptom control was maintained irrespective of treatment group assignment in the preceding double-blind study; as expected, the greatest numeric improvement from open-label baseline in PANSS total score was seen for patients initially treated with placebo in the acute study phase. The modal 80-mg/d dose of lurasidone, used by almost two-thirds of patients in this extension study, is consistent with that found in previous lurasidone flexible-dose studies,11,18 suggesting this may be an optimal dose for many outpatients with schizophrenia.
The safety and tolerability profile of lurasidone in this extension study was consistent with findings from previously reported short- and longer-term lurasidone studies in patients with schizophrenia.2,4,10,11,19 Notably, open-label treatment with lurasidone over 6 months in this study did not produce further weight gain beyond that observed during the initial 6 weeks of lurasidone treatment in the preceding acute study and was associated with improvement in lipid parameters, with no adverse changes in measures of glycemic control.
The incidence of parkinsonism (9.3%) and akathisia (13.0%) was also consistent with previous studies. In the preceding 6-week, double-blind study (which included olanzapine as an active control), a greater incidence of akathisia, parkinsonism, and dystonia was observed with lurasidone than with olanzapine.4 However, evening administration of lurasidone in a recent 6-week study20 was associated with lower rates of akathisia (7.4%–8.0%), parkinsonism (5.6%–6.6%), and dystonia (1.7%–2.4%) than were observed in other clinical trials in which lurasidone was dosed in the morning.6 It is possible that evening dosing may be better for lurasidone than the morning dosing employed in this study. In general, based on this and other studies, the incidence of akathisia with lurasidone appears comparable to that of other atypical antipsychotic agents, with the exception
of somewhat higher rates than observed with olanzapine or quetiapine.2,4,18,21
This open-label extension study was not intended as a formal switch study. However, because the preceding double-blind study included olanzapine, the study design permitted assessment of the effects of switching from treatment with olanzapine to lurasidone (implemented after a brief washout and without cross-taper). Switch studies in patients with schizophrenia generally include patients who have experienced suboptimal efficacy or safety/tolerability with their current antipsychotic treatment.22 In contrast, all patients who had completed double-blind treatment with olanzapine were eligible to participate in this extension study, independent of their acute phase treatment outcome. In this context, it is notable that patients treated with lurasidone after conversion from olanzapine showed sustained improvement in efficacy, with a decrease in weight and lipid parameters to levels comparable to those of patients who were not initially treated with olanzapine. The magnitude of the reduction in weight and lipid parameters found after switching from olanzapine to lurasidone in this extension study appeared clinically relevant and suggests that lurasidone may be a useful treatment for patients with antipsychotic-induced weight gain or dyslipidemia. These improvements in metabolic parameters were consistent with those reported in other studies that have examined the effects of switching from atypical antipsychotic medications with higher metabolic liability to those with lower metabolic liability,23–28 as well as a recent meta-analysis of short- and longer-term placebo-controlled and head-to-head trials of several atypical antipsychotics in patients with schizophrenia and bipolar disorder, which reported that lurasidone had the lowest potential for weight gain among the atypical antipsychotics studied.29
Improvements in psychopathology and reductions in weight and dyslipidemia were also observed in a recently completed formal switch study in which stable outpatients with schizophrenia were switched to lurasidone from other antipsychotic agents.18 However, switching from olanzapine to lurasidone in our extension study resulted in a modest increase in the incidence of akathisia, consistent with data reported from the formal lurasidone switch study.18
Overall study discontinuation rates were comparable between patients switching from olanzapine and those continuing on lurasidone in this extension study. A comparable proportion of patients who switched from olanzapine or continued on lurasidone discontinued open-label study participation because of insufficient clinical response. The rate of discontinuation because of an adverse event was slightly lower for patients switched from olanzapine compared with patients continued on lurasidone. There was an increased incidence of insomnia in patients who switched from olanzapine, but no increased incidence of anxiety, agitation, or other activation-like events that may be associated with a switch to a less sedating agent.
All first-generation antipsychotic agents and some second-generation agents (notably, amisulpride and risperidone) have the potential to increase serum prolactin levels, which can lead to hyperprolactinemia-associated complications such as infertility and sexual dysfunction.30–32 After a modest increase during the preceding 6-week double-blind study in patients treated with either lurasidone or olanzapine, prolactin levels decreased during this 6-month open-label extension study, and median values at study endpoint were similar to those observed at double-blind baseline.
Less than half of the patients (44.5%) who entered the open-label extension completed the study; discontinuation was attributed to insufficient clinical response for 6.7% of patients. A similar discontinuation rate (53.2%) was shown in a pooled analysis of atypical antipsychotic studies with comparable duration of treatment (24–28 weeks).33 Much greater 26-week discontinuation rates (78%–83%) were observed in a health care claims database analysis of atypical antipsychotic medication use.34
Limitations of this extension study include the open-label design (which may bias in favor of the investigational treatment) and the lack of an active control arm (eg, patients who continued on olanzapine). In addition, although any open-label extension study has potential for selection bias, 254 of the 298 patients (85.2%) who completed the preceding double-blind study elected to continue in the open-label extension. The demographic characteristics of patients at the acute phase baseline4 were comparable with those reported at extension baseline, limiting the potential for a nonrepresentative patient sample in the current study.
The results of this open-label, flexible-dose, 6-month extension study indicate that lurasidone in flexible doses ranging from 40 mg/d to 120 mg/d was generally safe and well tolerated and associated with sustained efficacy in the longer-term treatment of patients with schizophrenia. Lurasidone showed a low propensity for weight gain and minimal effects on metabolic parameters or prolactin and was associated with modest rates of EPS. These findings support the clinical utility of longer-term treatment with lurasidone in patients with schizophrenia.
Drug names: lurasidone (Latuda), olanzapine (Zyprexa),
quetiapine (Seroquel), risperidone (Risperdal and others).
Author affiliations: Department of Psychiatry, University of California–San Diego School of Medicine, San Diego (Dr Stahl); and Sunovion Pharmaceuticals, Inc., Marlborough, Massachusetts, and Fort Lee, New
Jersey (Drs Cucchiaro, Simonelli, Hsu, Pikalov, and Loebel).
Potential conflicts of interest: Dr Stahl is the founder/owner of the Neuroscience Education Institute and has served as a consultant to Advent, Allergan, Alkermes, Arena, AstraZeneca, BioMarin, BioVail (now Valeant), Boehringer Ingelheim, Bristol-Myers Squibb, CeNeRx BioPharma, Covance, Cypress Bioscience, Dainippon Sumitomo, Eisai, Eli Lilly, Forest, GenOmind, GlaxoSmithKline, Janssen, Jazz, Labopharm, Lundbeck, Marinus, Meda, Meiji, Merck, Neuronetics, Novartis, Ono, Orexigen Therapeutics, Otsuka, Pamlab, Pfizer, Pfizer Canada, Pierre Fabre, PGxHealth (now Trovis), Prexa, Propagate, Rexahn, Roche, Royalty, Sanofi, Schering-Plough (now Merck), Sepracor (now Sunovion), Servier, Shire, SK Corporation, Soffinova, Solvay, Sunovion (formerly Sepracor), Valeant, Vanda, Vivus, and Wyeth (now Pfizer); has served on speakers bureaus for Dainippon Sumitomo, Eli Lilly, Merck, Pamlab, Pfizer, Schering-Plough (now Merck), Shire, Sepracor (now Sunovion), Servier, Sunovion (formerly Sepracor), and Wyeth (now Pfizer); and has received research and/or grant support from AstraZeneca, BioMarin, Boehringer Ingelheim, Bristol-Myers Squibb, Cephalon, Dainippon Sumitomo, Eli Lilly, Forest, GenOMind, Lundbeck, Merck, Novartis, Pamlab, Pfizer, Pfizer Canada, PGxHealth (now Trovis), Pharmasquire, Sanofi-Aventis, Schering-Plough (now Merck), Sepracor (now Sunovion), Servier, Shire, Sunovion (formerly Sepracor), Torrent, Trovis, and Wyeth (now Pfizer). Drs Cucchiaro, Simonelli, Hsu, Pikalov, and Loebel are
full-time employees of Sunovion.
Funding/support: This study was sponsored by Sunovion Pharmaceuticals, Inc., Marlborough, Massachusetts, and Fort Lee, New Jersey.
Previous presentations: Initial results of this study were presented, in part, at the Annual Meeting of the American College of Neuropsychopharmacology; December 5–9, 2010; Miami Beach, Florida; and the International Congress on Schizophrenia Research; April 2–6, 2011; Colorado Springs, Colorado.
Acknowledgments: Technical editorial and medical writing assistance was provided by Lynne Kolton Schneider, PhD, and Nancy Holland, PhD, for Synchrony Medical Communications, LLC, West Chester, Pennsylvania. Funding for this support was provided by Sunovion Pharmaceuticals Inc., Marlborough, Massachusetts, and Fort Lee, New Jersey. The authors thank the patients who participated in this study, as well as the study investigators.
REFERENCES
1. Ishibashi T, Horisawa T, Tokuda K, et al. Pharmacological profile of lurasidone, a novel antipsychotic agent with potent 5-hydroxytryptamine 7 (5-HT7) and 5-HT1A receptor activity. J Pharmacol Exp Ther. 2010;334(1):
171–181. doi:10.1124/jpet.110.167346 PubMed
2. Nakamura M, Ogasa M, Guarino J, et al. Lurasidone in the treatment of acute schizophrenia: a double-blind, placebo-controlled trial. J Clin Psychiatry. 2009;70(6):829–836. doi:10.4088/JCP.08m04905 PubMed
3. Potkin SG, Ogasa M, Cucchiaro J, et al. Double-blind comparison of the safety and efficacy of lurasidone and ziprasidone in clinically stable outpatients with schizophrenia or schizoaffective disorder. Schizophr Res. 2011;132(2–3):101–107. doi:10.1016/j.schres.2011.04.008 PubMed
4. Meltzer HY, Cucchiaro J, Silva R, et al. Lurasidone in the treatment of schizophrenia: a randomized, double-blind, placebo- and olanzapine-controlled study. Am J Psychiatry. 2011;168(9):957–967. doi:10.1176/appi.ajp.2011.10060907 PubMed
5. Loebel A, Cucchiaro J, Silva R, et al. Efficacy of lurasidone in schizophrenia: results of a pooled analysis based on a 5-factor model of schizophrenia. Schizophr Res. 2010;117(2–3):267. doi:10.1016/j.schres.2010.02.425
6. Citrome L. Lurasidone for schizophrenia: a review of the efficacy and safety profile for this newly approved second-generation antipsychotic. Int J Clin Pract. 2011;65(2):189–210. doi:10.1111/j.1742-1241.2010.02587.x PubMed
7. Reynolds GP, Kirk SL. Metabolic side effects of antipsychotic drug treatment—pharmacological mechanisms. Pharmacol Ther. 2010;125(1):
169–179. doi:10.1016/j.pharmthera.2009.10.010 PubMed
8. Stahl SM, Mignon L, Meyer JM. Which comes first: atypical antipsychotic treatment or cardiometabolic risk? Acta Psychiatr Scand. 2009;119(3):
171–179. doi:10.1111/j.1600-0447.2008.01334.x PubMed
9. Newcomer JW. Second-generation (atypical) antipsychotics and metabolic effects: a comprehensive literature review. CNS Drugs. 2005;19(suppl 1):
1–93. doi:10.2165/00023210-200519001-00001 PubMed
10. Pikalov A, Cucchiaro J, Ogasa M, et al. Effect of lurasidone on weight and metabolic parameters: results from pooled short-term placebo-controlled and long-term trials in schizophrenia [abstract no. NR06-49]. Presented at: 164th Annual Meeting of the American Psychiatric Association; May 14–18, 2011; Honolulu, HI.
11. Citrome L, Cucchiaro J, Sarma K, et al. Long-term safety and tolerability of lurasidone in schizophrenia: a 12-month, double-blind, active-controlled study. Int Clin Psychopharmacol. 2012;27(3):165–176. doi:10.1097/YIC.0b013e32835281ef PubMed
12. International Conference on Harmonisation (ICH). ICH Harmonised Tripartite Guideline: Guideline for Good Clinical Practice. http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Efficacy/E6_R1/Step4/E6_R1__Guideline.pdf. Updated June 1996. Accessed January 17, 2012.
13. Simpson GM, Angus JWS. A rating scale for extrapyramidal side effects. Acta Psychiatr Scand suppl. 1970;212(S212):11–19. doi:10.1111/j.1600-0447.1970.tb02066.x PubMed
14. Barnes TR. A rating scale for drug-induced akathisia. Br J Psychiatry. 1989;
154(5):672–676. doi:10.1192/bjp.154.5.672 PubMed
15. Guy W, ed. ECDEU Assessment Manual for Psychopharmacology. US Department of Health, Education and Welfare publication (ADM) 76-338. Rockville, MD: National Institute of Mental Health; 1976.
16. Kay SR, Fiszbein A, Opler LA. The Positive and Negative Syndrome Scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13(2):261–276. doi:10.1093/schbul/13.2.261 PubMed
17. Sajatovic M, Velligan DI, Weiden PJ, et al. Measurement of psychiatric treatment adherence. J Psychosom Res. 2010;69(6):591–599. doi:10.1016/j.jpsychores.2009.05.007 PubMed
18. McEvoy JP, Citrome L, Hernandez D, et al. Effectiveness of lurasidone in patients with schizophrenia or schizoaffective disorder switched from other antipsychotics: a randomized, 6-week, open-label study. J Clin Psychiatry. 2013;74(2):170–179. 10.4088/JCP.12m07992.
19. Ogasa M, Kimura T, Nakamura M, et al. Lurasidone in the treatment of schizophrenia: a 6-week, placebo-controlled study. Psychopharmacology (Berl). 2013;225(3):519–530. doi:10.1007/s00213-012-2838-2 PubMed
20. Loebel A, Cucchiaro J, Sarma K, et al. Efficacy and safety of lurasidone 80mg/day and 160mg/day in the treatment of schizophrenia: a randomized, double-blind, placebo- and active-controlled trial [published online ahead of print February 13, 2013]. Schizophr Res. doi:10.1016/j.schres.2013.01.009 PubMed
21. Kane JM, Fleischhacker WW, Hansen L, et al. Akathisia: an updated review focusing on second-generation antipsychotics. J Clin Psychiatry. 2009;
70(5):627–643. doi:10.4088/JCP.08r04210 PubMed
22. Masand PS. A review of pharmacologic strategies for switching to atypical antipsychotics. Prim Care Companion J Clin Psychiatry. 2005;7(3):121–129. doi:10.4088/PCC.v07n0309 PubMed
23. Casey DE, Carson WH, Saha AR, et al; Aripiprazole Study Group. Switching patients to aripiprazole from other antipsychotic agents: a multicenter randomized study. Psychopharmacology (Berl). 2003;166(4):391–399. doi:10.1007/s00213-002-1344-3 PubMed
24. Newcomer JW, Campos JA, Marcus RN, et al. A multicenter, randomized, double-blind study of the effects of aripiprazole in overweight subjects with schizophrenia or schizoaffective disorder switched from olanzapine. J Clin Psychiatry. 2008;69(7):1046–1056. doi:10.4088/JCP.v69n0702 PubMed
25. Stroup TS, McEvoy JP, Ring KD, et al; Schizophrenia Trials Network. A randomized trial examining the effectiveness of switching from olanzapine, quetiapine, or risperidone to aripiprazole to reduce metabolic risk: Comparison of Antipsychotics for Metabolic Problems (CAMP).
Am J Psychiatry. 2011;168(9):947–956. doi:10.1176/appi.ajp.2011.10111609 PubMed
26. Weiden PJ, Daniel DG, Simpson G, et al. Improvement in indices of health status in outpatients with schizophrenia switched to ziprasidone. J Clin Psychopharmacol. 2003;23(6):595–600. doi:10.1097/01.jcp.0000095347.32154.08 PubMed
27. Weiden PJ, Newcomer JW, Loebel AD, et al. Long-term changes in weight and plasma lipids during maintenance treatment with ziprasidone. Neuropsychopharmacology. 2008;33(5):985–994. doi:10.1038/sj.npp.1301482 PubMed
28. Weiden PJ, Simpson GM, Potkin SG, et al. Effectiveness of switching to ziprasidone for stable but symptomatic outpatients with schizophrenia.
J Clin Psychiatry. 2003;64(5):580–588. doi:10.4088/JCP.v64n0514 PubMed
29. De Hert M, Yu W, Detraux J, et al. Body weight and metabolic adverse effects of asenapine, iloperidone, lurasidone and paliperidone in the treatment of schizophrenia and bipolar disorder: a systematic review and exploratory meta-analysis. CNS Drugs. 2012;26(9):733–759. doi:10.2165/11634500-000000000-00000 PubMed
30. Rettenbacher MA, Hofer A, Ebenbichler C, et al. Prolactin levels and sexual adverse effects in patients with schizophrenia during antipsychotic treatment. J Clin Psychopharmacol. 2010;30(6):711–715. doi:10.1097/JCP.0b013e3181faf0e3 PubMed
31. Lu ML, Shen WW, Chen CH. Time course of the changes in antipsychotic-induced hyperprolactinemia following the switch to aripiprazole. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(8):1978–1981. doi:10.1016/j.pnpbp.2008.09.016 PubMed
32. Melkersson K, Berinder K, Hulting AL. Effect of antipsychotic-induced hyperprolactinemia on anthropometric measures, insulin sensitivity and lipid profile in patients with schizophrenia or related psychoses. Neuroendocrinol Lett. 2011;32(4):428–436. PubMed
33. Liu-Seifert H, Adams DH, Kinon BJ. Discontinuation of treatment of schizophrenic patients is driven by poor symptom response: a pooled post-hoc analysis of four atypical antipsychotic drugs. BMC Med. 2005;3(1):21. doi:10.1186/1741-7015-3-21 PubMed
34. Mullins CD, Obeidat NA, Cuffel BJ, et al. Risk of discontinuation of atypical antipsychotic agents in the treatment of schizophrenia. Schizophr Res. 2008;
98(1–3):8–15. doi:10.1016/j.schres.2007.04.035 PubMed
This PDF is free for all visitors!





