Abstract
Objectives: To evaluate the efficacy, safety, and durability of a single 25-mg dose of a proprietary, synthetic formulation of psilocybin with psychological support for reducing chronic suicidal ideation in a treatment-resistant population.
Methods: This was an open-label, single-arm study with a 12-week follow-up conducted between March 2022 and May 2025. Twenty adults with chronic suicidal ideation, major depressive disorder (DSM-5), and ≥2 prior antidepressant treatment failures received a single 25-mg dose of psilocybin administered within a structured preparatory and integration psychotherapy protocol. The primary outcome was change from baseline in the Modified Scale for Suicidal Ideation (MSSI) at week 3. Secondary outcomes included change in MSSI at weeks 1 and 12 and change in Montgomery-Asberg Depression Rating Scale (MADRS) scores at weeks 1, 3, and 12. Outcomes were analyzed using 1-way repeated-measures analysis of variance with Bonferroni-adjusted pairwise comparisons.
Results: Significant reductions in MSSI scores were observed from baseline to week 3 (primary end point: mean difference [MD] = 13.95; 95% CI, 8.63–19.27; P < .001; d = 1.73). Improvements were rapid and durable, with significant reductions at week 1 (MD = 15.10; P < .001; d = 2.11) and week 12 (MD = 13.00; P < .001; d = 1.46). By week 12, 70% (n=14) of participants achieved MSSI ≤2. MADRS scores showed similar significant reductions at all postbaseline time points (MD range: 17.55–20.50; all P < .001; d = 1.63–1.97). No serious adverse events occurred.
Conclusions: In this open-label single-arm study of adults with chronic suicidal ideation and prior treatment failures, a single administration of psilocybin with psychological support was associated with rapid, large-magnitude, and durable reductions in suicidal ideation and depressive symptoms through 12 weeks. These preliminary findings support further evaluation in larger randomized controlled trials.
Trial Registration: ClinicalTrials.gov Identifier: NCT05220410.
J Clin Psychiatry 2026;87(2):26m16338
Author affiliations are listed at the end of this article.
Suicide accounts for 1.4% of all deaths worldwide and is the 10th leading cause of death in the US.1 Chronic suicidal ideation (SI) presents a formidable clinical challenge often persisting despite intensive trials of standard pharmacotherapy or psychotherapy.2 While traditional interventions, such as antidepressant medications and cognitive-behavioral therapy, are effective in reducing suicidality for some patients, they typically require prolonged treatment courses and may not provide relief in the acute phase of suicidal distress.3–6 Accessibility and acceptability of electroconvulsive therapy limit its impact on suicidality.7–9
Novel interventions with rapid action, such as (es)ketamine, have shown promise in acute settings. However, the durability of their antisuicidal effect remains a limitation. One pivotal trial of esketamine demonstrated a significant reduction in SI at 4 hours postadministration, but this effect was not maintained at 24 hours or at 25-day follow-up, highlighting the need for interventions with more sustained effects.10
Psychedelics such as psilocybin have garnered considerable attention for their potential to induce rapid and enduring psychological change when administered with structured psychological support.11,12 Psilocybin has demonstrated robust antidepressant effects in open-label and randomized controlled trials for major depressive disorder (MDD) and treatment-resistant depression.12–14
Studies also suggest a therapeutic mechanism from psilocybin pharmacology to network-level changes. As a serotonin 5-HT2A receptor agonist, psilocybin acutely increases cortical excitability and desynchronizes activity particularly within the default mode network (DMN),15 a brain network implicated in self-referential thinking and rumination.16 This aligns with the Relaxed Beliefs Under Psychedelics (REBUS) model, wherein increased brain entropy facilitates the relaxation of higher-level “priors,” that is, fixed beliefs that may be rigid and maladaptive in MDD.17 Given that SI is characterized by ruminative rigidity particularly as it becomes chronic, psilocybin-induced DMN desynchronization may have particular relevance for this depressive symptom, potentially rapidly disrupting the “priors” of SI. These acute neurobiological effects are accompanied by lasting changes in synaptic plasticity, including increased dendritic spine density, which may underlie the durability of clinical response observed in trials of psilocybin for mood disorders.15,18
Despite these advances, there is a critical gap since the vast majority of clinical trials have excluded individuals with significant SI, viewing suicidality as a prohibitive risk factor rather than a primary therapeutic target.19 A recent review confirmed that in the trials of psychedelics where suicidality was assessed, it was evaluated as a secondary or safety end point, and studies were underpowered to detect therapeutic effects.20 This cautious approach has left a crucial question unanswered: Can psilocybin be safely and effectively administered to the individuals who may be in greatest need of a rapid and durable intervention? Alternatively, could psilocybin worsen, exacerbate, or induce suicidality? This study was designed to directly address this gap by investigating this historically excluded population defined by the presence of chronic SI.
We conducted an open-label naturalistic study of a single 25-mg dose of psilocybin combined with psychological support in a sample of individuals with chronic SI in the context of MDD. Participants had persistent suicidal thoughts despite at least 2 adequate trials of antidepressant medication.21 The primary objective was to evaluate the efficacy of this intervention in reducing SI, as measured by the Modified Scale for Suicidal Ideation (MSSI).22 Secondary objectives included assessing the durability of the effect over a 12-week period, evaluating changes in comorbid depressive symptoms using the Montgomery-Asberg Depression Rating Scale (MADRS), and carefully monitoring safety and tolerability in this high-risk population.
METHODS
Design and Participants
This 12-week, open-label, single-arm, naturalistic trial evaluated a single 25-mg dose of COMP360, a proprietary pharmaceutical-grade synthetic psilocybin formulation (COMPASS Pathways), in 20 adults with chronic SI. The study was approved by the Sheppard Pratt Institutional Review Board and conducted under a US Food and Drug Administration Investigational New Drug authorization. Enrollment began in March 2022, and the final participant completed follow-up in May 2025. All participants provided written informed consent prior to study procedures, and the trial was registered at ClinicalTrials.gov (NCT05220410).
Participants were recruited via web listings and word of mouth. Inclusion criteria were age 18–65 years, a DSM-5 diagnosis of major depressive disorder confirmed by the Mini-International Neuropsychiatric Interview (version 7.0.2),23 chronic SI (active thoughts on more days than not for ≥3 months, each lasting ≥1 hour), and treatment resistance, defined as insufficient response to at least 2 adequate antidepressant trials.21 Columbia-Suicide Severity Rating Scale (C-SSRS) scores of 3 or 4 within the past year were required at both screening and baseline. Individuals with a score of 5 (active plan and imminent intent) within the preceding 3 months were excluded to target sustained SI without acute risk. Additional exclusions mirrored prior psychedelic research and included a personal or first-degree family history of psychotic or bipolar I disorder, active substance use disorder within 12 months, or unstable medical conditions contraindicating psilocybin administration.
Intervention
Participants completed a supervised taper and washout of all psychotropic medications for a minimum of 2 weeks or 5 half-lives, whichever was longer, prior to psilocybin administration.
The therapeutic intervention followed a structured psychological support model encompassing preparation, administration, and integration phases. Each participant met with 2 trained therapists for 3 preparatory sessions (≥6 hours total) focused on rapport building, psychoeducation, and setting intentions. Psilocybin was administered orally in a dedicated room designed for comfort and psychological safety. Sessions lasted approximately 8 hours, during which participants reclined with eyeshades and listened to a curated music playlist while therapists remained present throughout.
Three postsession integration meetings occurred the day after and in subsequent weeks to facilitate reflection and incorporation of insights from the experience into daily functioning.
Outcome Measures
Efficacy and safety measures were evaluated at baseline and at weeks 1, 3, and 12 postdosing. The primary outcome was change in the clinician-rated MSSI total score from baseline to week 3. The MSSI is a 19-item, semistructured interview assessing the severity and features of SI. Secondary outcomes included MSSI change from baseline to weeks 1 and 12, change in MADRS total scores across all time points, rates of response (≥50% MSSI reduction) and remission (MSSI = 0) at week 12, and the correlation between MSSI and MADRS change from baseline to week 3. Safety was additionally assessed at each time point using the C-SSRS, administered by a study clinician to systematically evaluate SI severity and suicidal behavior and to guide risk monitoring and clinical escalation when indicated.
Safety and tolerability were also assessed through systematic monitoring and recording of all treatment-emergent adverse events (AEs) at each study visit. AEs were coded using the Medical Dictionary for Regulatory Activities (MedDRA) and evaluated by the study physician for severity (mild, moderate, or severe) and relationship to the study intervention. Relatedness determinations were based on consistency with the known pharmacologic effects of psilocybin. An independent Data Safety Monitoring Board met twice a year and provided oversight throughout the study, conducting periodic reviews of safety data and AEs.
Statistical Analysis
The primary analysis population included all participants who received psilocybin and completed the week 3 assessment. Changes in MSSI and MADRS scores across time (baseline and weeks 1, 3, and 12) were analyzed using repeated-measures analysis of variance. Bonferroni-corrected post hoc tests compared each postdosing time point (weeks 1, 3, and 12) with baseline. Effect sizes were calculated using Cohen d. Pearson correlation coefficients assessed the relationship between changes in MSSI and MADRS from baseline to week 3. All tests were 2-sided with α= .05. Analyses were conducted in SPSS (version 27; IBM Corp). Data visualizations were performed in R (version 4.4.1; ggplot2 package).
Exploratory analyses compared baseline and treatment-related variables between week 3 responders (≥50% reduction in MSSI) and nonresponders to identify potential predictors of response. Continuous and categorical variables were analyzed with t tests and Fisher exact tests, respectively, using R (version 4.4.1).
RESULTS
Participant Characteristics
A total of 20 participants were enrolled and received the psilocybin intervention. All participants completed the 12-week protocol, without dropout. Details of participant flow, including screening and enrollment, are provided in Supplementary Figure 1 (CONSORT diagram). The demographic and baseline clinical characteristics of the sample are presented in Table 1. The sample consisted of 12 men (60%) and 8 women (40%), with a mean (SD) age of 36.3 (11.1) years.
Participants presented with severe SI and moderate-to-severe depression. By clinical interview, the majority of participants had suffered from SI for years, or most of their adult lives.
Primary Efficacy Outcome: Change in Suicidal Ideation at Week 3
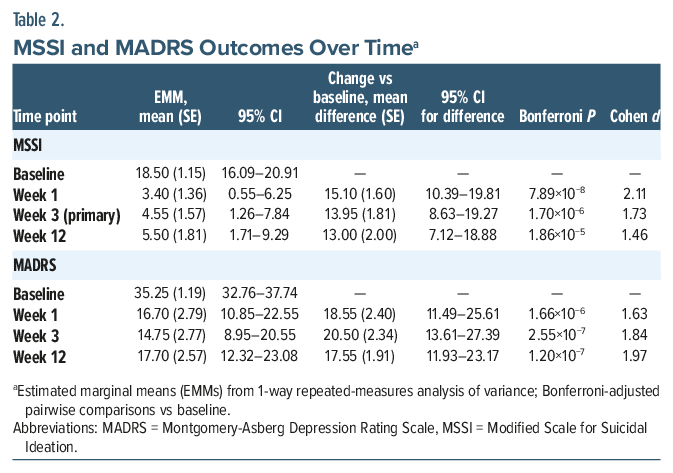
Psilocybin treatment was associated with a large and statistically significant reduction in SI at the week 3 primary end point (Table 2). The mean reduction from baseline was 13.95 points, with a large within-subject effect size (Cohen d = 1.73).
Secondary Efficacy Outcomes
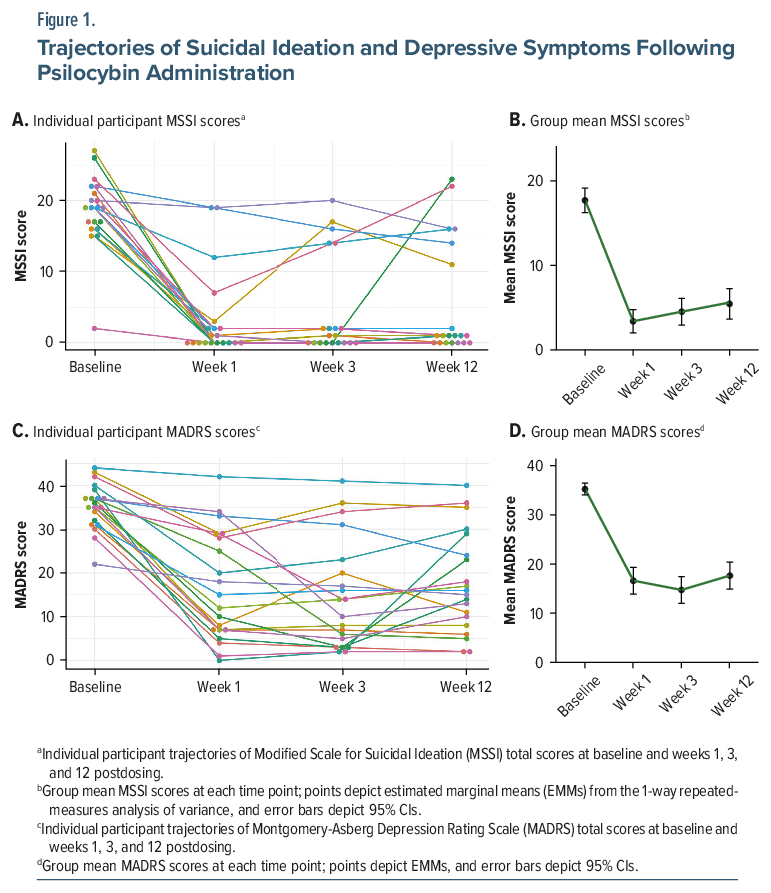
Temporal dynamics of MSSI and MADRS reductions. Reductions in SI were rapid and durable: Significant improvement was evident by week 1 and remained significant through week 12, with no significant differences among postbaseline assessments (Table 2; Figure 1A–B). Depressive symptoms showed a similarly robust and sustained improvement across follow-up (Table 2; Figure 1C–D).
Changes in SI and depressive symptom severity covaried: baseline-to-week 3 change scores on the MSSI and MADRS were strongly correlated (r = 0.70, P < .001).
Response and remission rates. At the week 3 primary end point, 15 of 20 participants (75%) met the criterion for an antisuicidal response (≥50% reduction from baseline MSSI). Nine participants (45%) achieved full remission of SI (MSSI = 0). For depressive symptoms, 12 of 20 participants (60%) met the criterion for MADRS response (≥50% reduction from baseline) at week 3, and 9 participants (45%) achieved remission, defined as a MADRS score of 10 or lower.
At the week 12 study end point, a substantial proportion of participants maintained clinical improvement. Seven of 20 participants (35%) had remission of SI (MSSI=0). An additional 7 participants (35%) had minimal residual ideation (MSSI score of 1 or 2). For depressive symptoms, 10 of 20 participants (50%) met the criteria for an antidepressant response (≥50% reduction in MADRS score) at week 12, and 5 of 20 participants (25%) were in remission, defined as a MADRS score of 10 or lower.
Medication Restarts
Across the 12-week follow-up, 12 of 20 participants (60%) restarted or initiated at least 1 psychotropic medication for MDD at or after week 3 (Supplementary Table 1). These patterns reflect the treatment-resistant nature of the cohort, though improvements observed through week 3 were largely maintained even as many participants resumed adjunctive pharmacotherapy. One participant with comorbid posttraumatic stress disorder (PTSD) required a rescue dose of lorazepam on the dosing day and as-needed clonazepam for 3 weeks thereafter to manage anxiety.
Safety and Tolerability
C-SSRS assessments provided supplementary safety measures, and results are given in Supplementary Tables 2 and 3. Psilocybin was generally well tolerated. No serious AEs were reported, and no participant discontinued the study due to an AE. The most frequent treatment-emergent AEs considered by the investigator to be related or possibly related to the intervention were transient and mild to moderate in severity. One patient required a rescue medication (lorazepam) on the day of dosing, as noted above. AEs are summarized in Table 3.
Two of 20 participants (10%) experienced an increase in MSSI score relative to their baseline value. For one of these participants, this increase was transient, with MSSI initially decreasing, rising at week 3, and then declining again to below baseline by week 12. For another patient, the increased SI was present at the week 12 end point, with their MSSI score rising from 17 at baseline to 23. There was no evidence of treatment-emergent psychosis or sustained manic/hypomanic symptoms in any participant.
Exploratory Analyses
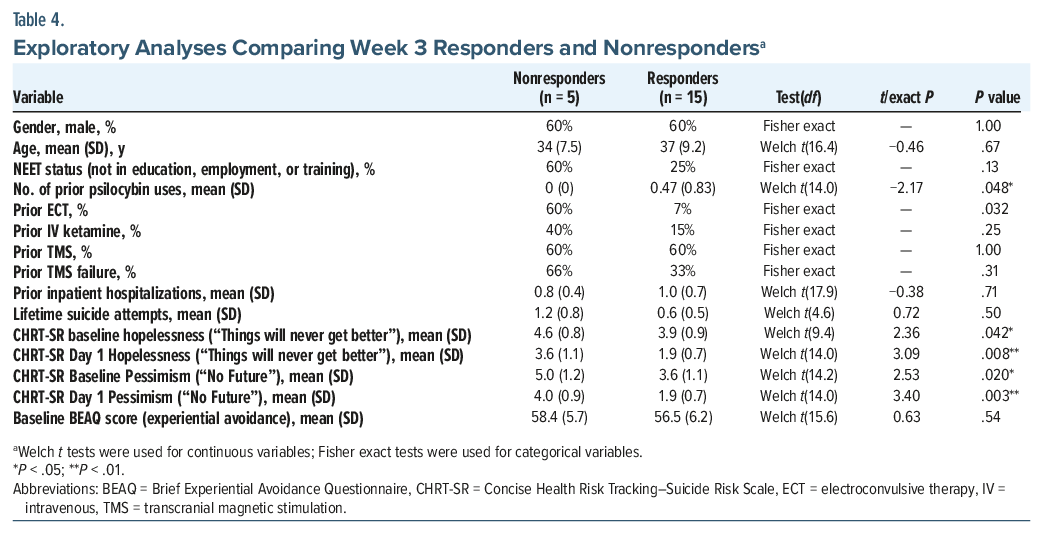
Exploratory comparisons of week 3 responders (n = 15) and nonresponders (n = 5) are summarized in Table 4. Variables examined included demographics, prior treatments (psilocybin, electroconvulsive therapy (ECT), intravenous ketamine, and transcranial magnetic stimulation [TMS]), baseline clinical burden (hospitalizations and suicide attempts), and psychological scales assessing hopelessness, pessimism, and experiential avoidance.
Responders had greater prior psilocybin exposure (mean [SD] number of prior uses: 0.47 [0.64] vs 0.00 [0.00]; P = .048) and were less likely to have a history of ECT (7% vs 60%; Fisher exact P = .032). Nonresponders endorsed greater hopelessness and pessimism on Concise Health Risk Tracking–Suicide Risk Scale (CHRT-SR) items, including higher ratings for “Things will never get better” at baseline (4.6 vs 3.9; P = .042) and day 1 (3.6 vs 1.9; P = .008) and “No future” at baseline (5.0 vs 3.6; P = .020) and day 1 (4.0 vs 1.9; P = .003). Entrenched negative expectancies appeared to attenuate early antisuicidal response to psilocybin therapy.
DISCUSSION
This open-label pilot study provides the first prospective data on the efficacy and safety of psilocybin-assisted therapy as an intervention targeting chronic, treatment-resistant suicidal ideation. The principal finding is that a single 25-mg dose of psilocybin, administered within a supportive therapeutic framework, was associated with rapid and substantial reductions in suicidal ideation that were sustained for 12 weeks (Figure 1). These antisuicidal effects were accompanied by significant and correlated antidepressant effects.
The magnitude of the observed effect on suicidal ideation is noteworthy. The within-group effect sizes (Cohen d ranging from 1.5 to 2.1) are among the largest reported for any psychiatric intervention for this condition, though these uncontrolled estimates must be interpreted with caution given the open-label design and the potential contribution of nonspecific factors including placebo response and expectancy effects. The rapidity of effect, with maximal reduction in suicidal ideation observed by the first posttreatment assessment at week 1, is clinically meaningful. Unlike conventional antidepressants, which often require several weeks for onset, psilocybin produced early improvement that may be critical in acutely suicidal populations. The durability of this effect through 12 weeks following a single administration also contrasts with the transient benefits often seen with other rapid-acting agents such as esketamine.10 This sustained response suggests that psilocybin may act not merely to suppress symptoms temporarily but also to catalyze more enduring psychological and behavioral change.
Might psilocybin exert some degree of primary antisuicidality effects independent of antidepressant effects? While the two outcomes were correlated (r=.70), the effect size for the reduction in suicidal ideation at week 1 (Cohen d=2.1) was numerically larger than that for depressive symptoms (Cohen d=1.7), and cause-effect relationship is indeterminate. Speculatively, psilocybin is known to occasion profound psychological experiences characterized by emotional catharsis, shifts in perspective, and an enhanced sense of meaning or connectedness.24,25 These processes could directly target core psychological drivers of suicidality, such as hopelessness, psychological pain, and social disconnection, in a manner partially distinct from the treatment of anhedonia or low mood, insomnia, appetite disturbance, or other symptoms of MDD. These psychological processes may be supported by psilocybin’s pharmacologic action as a serotonin 5-HT2A receptor agonist, which acutely disrupts DMN activity associated with rigid self-referential thinking and rumination. The resulting increase in neural flexibility, combined with evidence of enhanced synaptic plasticity following psilocybin administration, may provide a neurobiological substrate for the rapid and enduring shifts in perspective and connectedness reported by participants.
Exploratory analyses (Table 4) revealed the largest responder-nonresponder differences on CHRT-SR items endorsing “Things will never get better” and “No future.” These items were examined as potential surrogates for expectancy, or belief in the possibility of treatment benefit. Nonresponders consistently endorsed more severe negative expectancies, suggesting that entering treatment with stronger convictions that improvement was impossible may yield less benefit from psilocybin therapy. Prior psilocybin use was more common among responders, whereas prior ECT exposure showed suggested association with nonresponder status, indicating possible effects of treatment history on outcomes. Lifetime suicide attempts, experiential avoidance (Brief Experiential Avoidance Questionnaire), and prior TMS exposure did not differ between groups. These exploratory, hypothesis-generating findings should be interpreted cautiously given the small sample but may help identify expectancy-related moderators and guide stratification variables in future controlled trials.
The safety profile in this high-risk sample was generally favorable and consistent with previous psilocybin studies in nonsuicidal depressed populations, with most AEs being transient and mild. Notably, 1 participant with comorbid PTSD experienced a panic attack during the dosing session, requiring a rescue dose of lorazepam and subsequent short-term benzodiazepine use. While manageable, this underscores the need for vigilance in participants with high baseline anxiety or trauma-related disorders. Moreover, the findings that one participant experienced transient worsening in suicidal ideation and that another patient experienced a clinically significant worsening of suicidal ideation at the study end point are a critical safety signal that must be interpreted with caution. This finding provides a crucial bridge between the largely positive results from controlled clinical trials and the mixed results from observational studies and case reports, which have noted instances of psychedelics exacerbating psychiatric symptoms.26,27 It underscores that even within a highly controlled and supportive therapeutic context, a negative psychological outcome is possible for a subset of individuals. This highlights the heterogeneity of response and the absolute necessity of careful participant screening, preparation, and monitoring.
Another subtle but important observation is the temporal trajectory of the mean MSSI score. While the improvement from baseline was marked and sustained, the mean score was lowest at week 1 (3.4) and showed a slight upward drift at week 3 (4.6) and week 12 (5.5), suggesting that while the acute effect is profound, there may be a partial return of symptoms over time. This observation supports the notion that additional dosing or integration sessions could help consolidate and maintain therapeutic gains.
Limitations
The primary limitation is the open-label, single-arm design, which cannot control for nonspecific factors such as placebo effects. The small sample size (N=20) limits the generalizability and ability to draw conclusions about the incidence of rare adverse events. The study was conducted at a single, specialized academic center with highly trained therapists, which may not reflect real-world clinical settings. An additional limitation is that more than half of participants restarted or initiated adjunctive pharmacotherapy for MDD after week 3. Although improvements on primary outcomes were evident before these changes, the introduction of additional medications complicates the interpretation of long-term durability as psilocybin was not a stand-alone intervention across the entire follow-up. Finally, the sample size on MSSI nonresponders at week 3 was small (N=5), limiting the reliability of the exploratory comparisons with responders. Additionally, the open-label design raises the possibility that expectancy effects contributed to the large observed effect sizes. Participants who volunteer for a psilocybin trial may harbor strong positive expectations, and the subjective profundity of the psychedelic experience may amplify confirmation bias. In the broader antidepressant literature, nonspecific effects account for a substantial proportion of improvement in depression trials; there is no reason to assume that the contribution of such effects would be smaller in psychedelic-assisted therapy. Future controlled trials with an adequate comparator condition will be essential to disentangle specific pharmacologic effects from expectancy and other nonspecific factors.
CONCLUSION
Despite these limitations, this pilot study provides a strong preliminary signal that psilocybin-assisted therapy warrants further investigation as a treatment for chronic suicidal ideation. A single dose of psilocybin was associated with rapid, robust, and durable reductions in suicidal ideation and depression. Future studies should employ a suitable comparator group and include long-term follow-up to assess the full durability of treatment effects.
Article Information
Published Online: May 13, 2026. https://doi.org/10.4088/JCP.26m16338
© 2026 Physicians Postgraduate Press, Inc.
Submitted: January 22, 2026; accepted March 13, 2026.
To Cite: van der Vaart A, LaPratt J, Swartz K, et al. Efficacy and safety of a single dose of psilocybin for chronic suicidal ideation: an open-label trial.
J Clin Psychiatry. 2026;87(2):26m16338.
Author Affiliations: Institute for Advanced Diagnostics and Treatment, Sheppard Pratt Health System, Baltimore, Maryland (van der Vaart, LaPratt, Swartz, Shoultz, Lauterbach, Aaronson); Department of Psychiatry, University of Maryland, Baltimore, Maryland (van der Vaart, Lauterbach, Aaronson); VA Palo Alto Health Care System and Department of Psychiatry and Behavioral Sciences Stanford University School of Medicine, Palo Alto, California (Suppes); Department of Psychiatry, Medical University of South Carolina, Charleston, South Carolina (Sackeim).
Corresponding Author: Scott T. Aaronson, MD, Sheppard Pratt, 6501 N. Charles Street, Baltimore, MD 21204 ([email protected]).
Financial Disclosure: Dr Aaronson reported grants and nonfinancial support (supply of drug) from COMPASS Pathways during the conduct of the study and personal fees from LivaNova, Neuronetics, Genomind, and Sage Therapeutics outside the submitted work. Dr LaPratt reported grants from COMPASS Pathways during the conduct of the study. Dr Lauterbach reported other from Sheppard Pratt/COMPASS Pathways (supported study) during the conduct of the study and other from Brain Educators (one-third owner) outside the submitted work. Dr Suppes reported other from PsiloTec (stock options) outside the submitted work. Dr Sackeim reported personal fees from Cerebral Therapeutics, LivaNova, Neurolief, Neuronetics, Parow Enthiobiosciences, and SigmaStim outside the submitted work.
Funding/Support: This study was funded by Compass Pathways plc.
Role of the Funder: The funder had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Supplementary Material: Available at Psychiatrist.com.
Clinical Points
- Chronic suicidal ideation (SI) frequently persists despite multiple adequate antidepressant trials.
- Most psychedelic clinical trials exclude individuals with significant suicidality, leaving a critical gap in the evidence base for this high-risk population.
- In this open-label pilot study, a single dose of psilocybin with psychological support was associated with rapid, large-magnitude reductions in SI and depression sustained through 12 weeks.
- At week 12, 70% of participants had zero or minimal SI (Modified Scale for Suicidal Ideation ≤2), suggesting that psilocybin-assisted therapy merits further evaluation in controlled trials for patients with chronic suicidality.
Editor’s Note: We encourage authors to submit papers for consideration as a part of our Early Career Psychiatrists section. Please contact Joseph F. Goldberg, MD at Psychiatrist.com/contact/goldberg.
References (27)

- National Institute of Mental Health. Suicide. National Institute of Health; 2024. Accessed May 13, 2025. https://www.nimh.nih.gov/health/statistics/suicide
- Seemuller F, Kolter M, Musil R, et al. Chronic vs non-chronic depression in psychiatric inpatient care-data from a large naturalistic multicenter trial. J Affect Disord. 2022;299:73–84. PubMed CrossRef
- Gibbons RD, Brown CH, Hur K, et al Suicidal thoughts and behavior with antidepressant treatment: reanalysis of the randomized placebo-controlled studies of fluoxetine and venlafaxine. Arch Gen Psychiatry. 2012;69(6):580–587. PubMed CrossRef
- Cuijpers P, de Beurs DP, van Spijker BA, et al The effects of psychotherapy for adult depression on suicidality and hopelessness: a systematic review and meta-analysis. J Affect Disord. 2013;144(3):183–190. PubMed CrossRef
- Bergfeld IO, Mantione M, Figee M, et al. Treatment-resistant depression and suicidality. J Affect Disord. 2018;235:362–367. PubMed CrossRef
- Rhee TG, Shim S, Nasir M, et al. Longitudinal associations of electroconvulsive therapy with all-cause mortality and suicide deaths in depression and other psychiatric disorders: a systematic review and meta-analysis. Res Sq. 2025. doi: 10.21203/rs.3.rs-6180102/v1. PubMed CrossRef
- Kellner CH, Fink M, Knapp R, et al. Relief of expressed suicidal intent by ECT: a consortium for research in ECT study. Am J Psychiatry. 2005;162(5):977–982. PubMed CrossRef
- Sackeim HA. Modern electroconvulsive therapy: vastly improved yet greatly underused. JAMA Psychiatry. 2017;74(8):779–780. PubMed CrossRef
- Sienaert P, Brus O, Lambrichts S, et al. Suicidal ideation and ECT, ECT and suicidal ideation: a register study. Acta Psychiatr Scand. 2022;146(1):74–84. PubMed CrossRef
- Canuso CM, Singh JB, Fedgchin M, et al. Efficacy and safety of intranasal esketamine for the rapid reduction of symptoms of depression and suicidality in patients at imminent risk for suicide: results of a double-blind, randomized, placebo-controlled Study. Am J Psychiatry. 2018;175(7):620–630. PubMed CrossRef
- Aaronson ST, van der Vaart A, Miller T, et al. Single-Dose synthetic psilocybin with psychotherapy for treatment-resistant bipolar type II major depressive episodes: a nonrandomized open-label trial. JAMA Psychiatry. 2024;81(6):555–562. PubMed CrossRef
- Aaronson ST, van der Vaart A, Miller T, et al. Single-dose psilocybin for depression with severe treatment resistance: an open-label trial. Am J Psychiatry. 2025;182(1):104–113. PubMed CrossRef
- Goodwin GM, Aaronson ST, Alvarez O, et al. Single-dose psilocybin for a treatment-resistant episode of major depression. N Engl J Med. 2022;387(18):1637–1648. PubMed CrossRef
- Goodwin GM, Aaronson ST, Alvarez O, et al. Single-dose psilocybin for a treatment-resistant episode of major depression: impact on patient-reported depression severity, anxiety, function, and quality of life. J Affect Disord. 2023;327:120–127. PubMed CrossRef
- Siegel JS, Subramanian S, Perry D, et al. Psilocybin desynchronizes the human brain. Nature. 2024;632(8023):131–138. PubMed CrossRef
- Hamilton JP, Farmer M, Fogelman P, et al. Depressive rumination, the default-mode network, and the dark matter of clinical neuroscience. Biol Psychiatry. 2015;78(4):224–230. PubMed CrossRef
- Carhart-Harris RL, Friston KJ. REBUS and the anarchic brain: toward a unified model of the brain action of psychedelics. Pharmacol Rev. 2019;71(3):316–344. PubMed CrossRef
- Shao LX, Liao C, Gregg I, et al. Psilocybin induces rapid and persistent growth of dendritic spines in frontal cortex in vivo. Neuron. 2021;109(16):2535–2544 e4. PubMed CrossRef
- Iltis AS, McCall WV, Deria R. Suicidality, depression, and the FDA: health inequities and the ethical conduct of research. J Clin Psychiatry. 2020;81(2):19m13050. PubMed CrossRef
- Meshkat S, Malik T, Zeifman R, et al. Psychedelics and suicide-related outcomes: a systematic review. J Clin Med. 2025;14(5):1416. PubMed CrossRef
- Sackeim HA, Aaronson ST, Bunker MT, et al. Update on the assessment of resistance to antidepressant treatment: rationale for the Antidepressant Treatment History Form: Short Form-2 (ATHF-SF2). J Psychiatr Res. 2024;176:325–337. PubMed CrossRef
- Miller IW, Norman WH, Bishop SB, et al. The Modified Scale for Suicidal Ideation: reliability and validity. J Consult Clin Psychol. 1986;54(5):724–725. PubMed CrossRef
- Sheehan DV, Lecrubier Y, Sheehan KH, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59(Suppl 20:22-33):22–57. PubMed
- Griffiths RR, Richards WA, McCann U, et al. Psilocybin can occasion mystical-type experiences having substantial and sustained personal meaning and spiritual significance. Psychopharmacol Berl. 2006;187(3):268–283. PubMed CrossRef
- Roseman L, Nutt DJ, Carhart-Harris RL. Quality of acute psychedelic experience predicts therapeutic efficacy of psilocybin for treatment-resistant depression. Front Pharmacol. 2017;8:974. PubMed CrossRef
- Krebs TS, Johansen PO. Psychedelics and mental health: a population study. PLoS One. 2013;8(8):e63972. PubMed CrossRef
- Barber G, Nemeroff CB, Siegel S. A case of prolonged mania, psychosis, and severe depression after psilocybin use: implications of increased psychedelic drug availability. Am J Psychiatry. 2022;179(12):892–896. PubMed CrossRef
This PDF is free for all visitors!