Abstract
Objective: To compare rehospitalization outcomes and medication practice patterns between long-acting injectable antipsychotics (LAIs) and oral antipsychotics (OAs) in US patients with bipolar I disorder (BD-I).
Methods: This retrospective cohort study using the Premier Hospital database (October 2020–September 2023) grouped adults (aged ≥18 years) hospitalized with BD-I by discharge medication: OA, LAI, or second-generation LAI (SG LAI). LAI and SG LAI patients were propensity score matched (1:4) to OA patients. Rehospitalization rates and risk (BD-I–related and all-cause) within 30, 60, and 90 days were assessed. Medication continuation vs switching at rehospitalization was also analyzed.
Results: Among 98,088 eligible patients, 78.1% were in the OA and 2.4% in the LAI group. BD-I–related rehospitalization rates were lower for LAI users at 30 (3.9% vs 5.0%, P=.033) and 60 days (5.9% vs 7.2%, P=.030) vs OAs. SG LAI users showed reductions in rehospitalizations at 30 (3.6% vs 5.4%; P=.010), 60 (5.2% vs 7.5%; P=.008), and 90 (6.8% vs 9.1%; P=.015) days. Risk of first BD-I rehospitalization within 30–90 days was reduced for LAI (hazard ratio [HR]: 0.784–0.856) and SG LAI (HR: 0.653–0.742) groups vs OAs.
Conclusions: LAIs, especially SG formulations, are associated with reduced rehospitalization in BD-I compared to OAs, supporting their broader use to improve adherence and reduce readmissions.
J Clin Psychiatry 2026;87(2):25m16154
Author affiliations are listed at the end of this article.
Bipolar I disorder (BD-I) is a chronic psychiatric illness characterized by episodes of mania, hypomania, and depression, or mixtures of manic and depressive features.1,2 In 2019, 2.12 million people in the United States (US) had bipolar disorder (BD) (age-standardized prevalence of 601.3 per 100,000),3 and BD-I had a lifetime prevalence of 1.0%.4 Individuals with BD-I are at risk for severe psychosocial dysfunction, psychiatric hospitalization, and relapse (ie, regression to symptoms of hypomania, mania, or depression).2,5 Those with higher relapse rates experience more frequent hospitalizations, higher rates of disability and suicide, impaired cognitive functioning, more coexisting medical conditions, poorer interpersonal relationships, and overall impaired quality of life.6 Repeated relapses can escalate to increasingly severe interepisode residual symptoms of mania and depression, and other adverse symptoms.6 With effective treatment and medication adherence, patients are at lower risk of relapse.7 Pharmacologic treatments for BD-I include mood stabilizers (eg, lithium and valproate), second-generation antipsychotics, and antidepressants and are usually supplemented with psychosocial interventions such as cognitive-behavioral therapy.8
Nonadherence to oral treatments is an obstacle to the long-term well-being of patients with BD.7 Long-acting injectable antipsychotics (LAIs) are associated with greater medication adherence9–12 and subsequent lower rates of relapse or hospitalization9,12–15 than with oral antipsychotics (OAs) in studies of patients with schizophrenia. A retrospective analysis also found reductions in rehospitalization for patients with BD, schizophrenia, or schizoaffective disorder taking LAIs compared with patients taking OAs.16 To date, LAI formulations approved by the US Food and Drug Administration for use as a monotherapy or adjunctive therapy in the treatment of patients with BD-I include aripiprazole monohydrate and risperidone extended-release microspheres.17,18
LAIs are infrequently used to treat patients with BD-I in clinical practice, even though they could offer benefits.19,20 This could be due to lack of clinician and patient awareness, overestimation of patient adherence, underestimation of patient LAI acceptance, and lack of clinical guidance regarding the use of LAIs in BD.21–23 Analyses of a US electronic health record database estimated that only 18% of patients diagnosed with schizophrenia or schizoaffective disorder were prescribed LAIs.12,24 In outpatient settings, the proportion was even lower, ranging from just 3%–10%.25,26 A recent single-center study reported that patients with schizophrenia (41.5%) and schizoaffective disorder (36.0%) accounted for the majority of LAI users, while those with BD represented a smaller share (13.1%).27 In addition, LAI prescription rates among patients hospitalized with BD remain low at approximately 14%.28
Considering that BD has been associated with increasing healthcare costs and high inpatient hospitalization,29 additional data on rehospitalization following OA or LAI use, specifically in patients with BD-I, are required. In addition, real-world data on practice patterns for switching from OA to LAIs are needed. The aim of this study was to examine rehospitalization rates and the risk of rehospitalization of hospitalized patients with BD-I who use LAIs vs OAs. Medication switching patterns at rehospitalization were also investigated.
METHODS
Study Design
This retrospective cohort study was conducted using the Premier Hospital database between October 2020 and September 2023. The 2020–2023 study period was selected to include most recent prescribing patterns, including the use of newer antipsychotics. This large (>231 million patients), US hospital-based, service-level, all-payer database contains information on inpatient discharges primarily collected from nonprofit, nongovernmental community and teaching hospitals and health systems in rural and urban areas.
Population
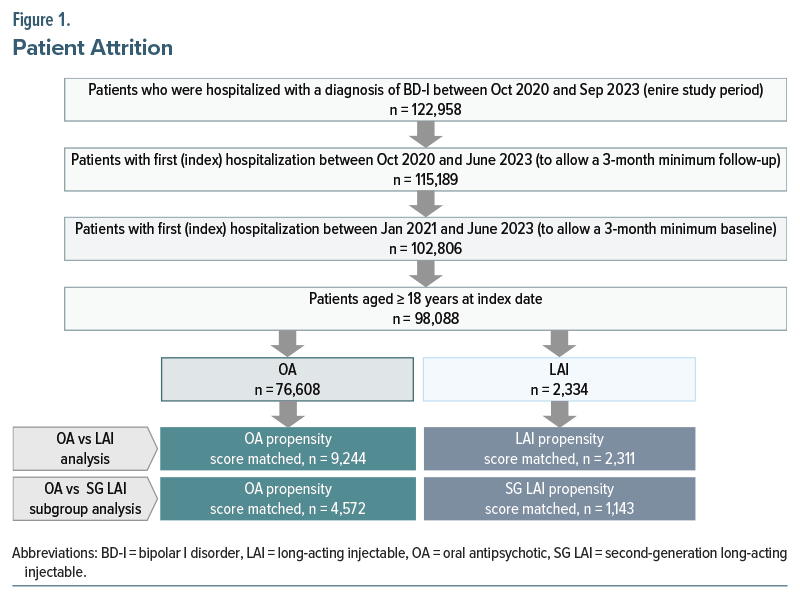
Eligible patients were aged ≥18 years with a primary or admitting diagnosis of BD-I (International Statistical Classification of Diseases and Related Health Problems, Tenth Revision [ICD-10] diagnosis code of F31.0, F31.8, or F31.9) and had data available ≥3 months pre index date (baseline) and ≥3 months post index date (follow-up) during the study period. The date of first hospitalization with a BD-I diagnosis was set as the index date. Patients were grouped based on discharge medication at index hospitalization, which included (1) OA only; (2) LAI only; (3) mood stabilizer (MS); (4) OA and LAI; (5) OA and MS; (6) LAI and MS; (7) OA, LAI, and MS; and (8) a group with none of these medications at discharge (see Supplementary Table 1 for the specific LAIs used).
To examine rehospitalization rates and risk of rehospitalization between patients treated with LAIs compared with OAs, 2 main groups were derived from the cohorts described above. The OA group included patients from groups 1 and 5 above, with discharge on an OA defined as receiving an OA on the day of or the day before discharge. The LAI group included patients from groups 2, 4, 6, and 7, with discharge on an LAI defined as receiving an LAI during the hospital stay. A subgroup of patients discharged on an SG LAI was also identified from the overall LAI group.
Outcomes and Measures
Patient characteristics captured at index date or over the 3-month baseline period included sex, age groups, marital status, payer type (defined as Commercial, Medicare, Medicaid, or other), admission source, physician specialty, length of stay at index hospitalization, proportion of other psychiatric comorbidities, baseline antipsychotic or mood disorder use, and suicide attempt/ideation diagnosis, Charlson comorbidity index score,30 and hospital profile (urban vs rural, teaching vs nonteaching hospitals, hospital size [by number of beds], and geographic region).
The primary outcomes evaluated during the follow-up included rehospitalizations within 30, 60, and 90 days following index hospitalization, risk of rehospitalization, and length of stay of first rehospitalization. Exploratory outcomes included the number and proportion of patients continuing vs switching medication at first rehospitalization and who had a subsequent rehospitalization. Analysis of hospitalizations included those that were BD-I related (ie, diagnosis present in primary or admitting position) and all-cause hospitalizations.
Statistical Analysis
Patients in the LAI cohort and SG LAI subgroup were propensity-score matched (PSM) 1:4 to the patients in the OA cohort. The matching was based on demographics, comorbidities, baseline OA use, payer type, admission and discharge sources, length of stay, underlying mental health disorder diagnosis, and hospital profile measures. Balance after PSM was assessed with the standardized mean difference (SMD).
Rehospitalization rates within 30, 60, and 90 days after index hospitalization were calculated and compared between groups using χ2 tests. The mean length of hospitalization stay between groups was compared with t tests. Rehospitalization risk between patients discharged on LAIs and patients discharged on OAs was assessed using a Cox proportional hazards model, adjusting for confounding variables, which were not balanced after PSM. Kaplan-Meier curves and log-rank testing were also used to assess time to rehospitalization for the OA and LAI groups. Results were considered statistically significant at P<.05.
Ethics
This was a retrospective study that used data from de-identified medical charts, which were documented during routine clinical practice. Institutional review board approval or waiver was not required.
RESULTS
Patient Characteristics
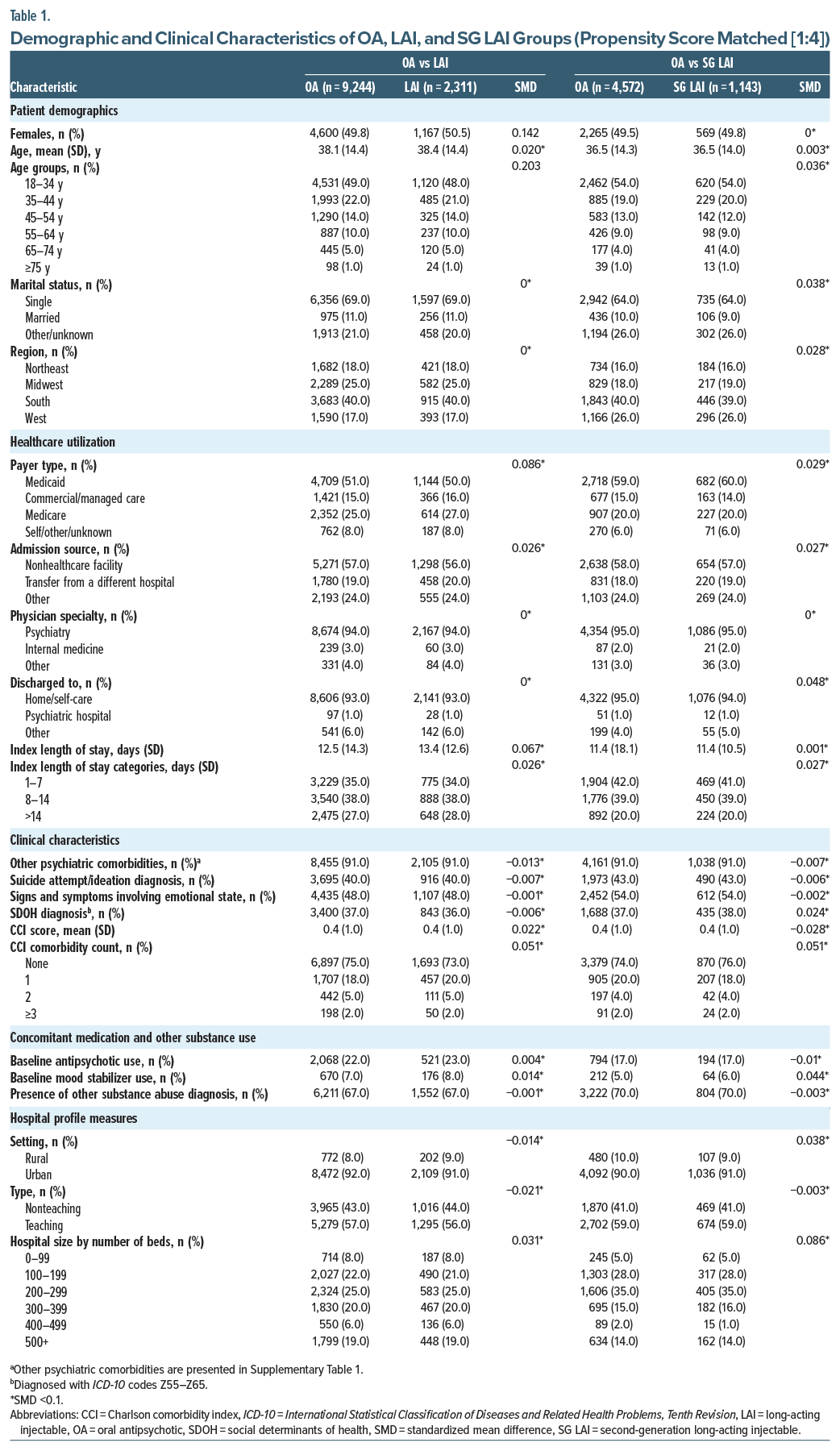
Of 98,088 eligible patients, 76,608 (78.1%) received OA at discharge; 2,334 (2.4%) of all eligible patients received LAI, 9,105 (9.3%) received MS, and 10,041 (10.2%) did not receive any of these medications. PSM included 9,244 and 2,311 patients discharged on OA and LAI, and 4,572 and 1,143 patients discharged on OA and SG LAI, respectively (Figure 1). Of the 2,311 patients prescribed LAIs and 1,143 patients prescribed SG LAIs, 1,981 and 1,143 patients were also discharged on OAs, respectively. Balance between the OA and LAI groups was achieved after PSM (SMD <0.10) for all variables except for patients’ sex and age distribution. All variables were balanced between the SG LAI subgroup and the OA group.
Within PSM cohorts, approximately 50% of patients were women, with a mean age of 36–38 years across groups. Approximately 91% of patients presented with other psychiatric comorbidities (Table 1), including mental and behavioral disorders due to psychoactive substance use (73%–77%), anxiety and other nonmood psychotic disorders (49%–60%), and schizophrenia and other nonmood psychotic disorders (28%–40%) were most prevalent across all groups (Supplementary Table 2). During the baseline period, 17%–23% of patients were using antipsychotics and 5%–8% were using MSs to treat their BD-I. Approximately 40%–43% of patients had attempted suicide or presented with suicidal ideation (Table 1).
Rehospitalization Rates
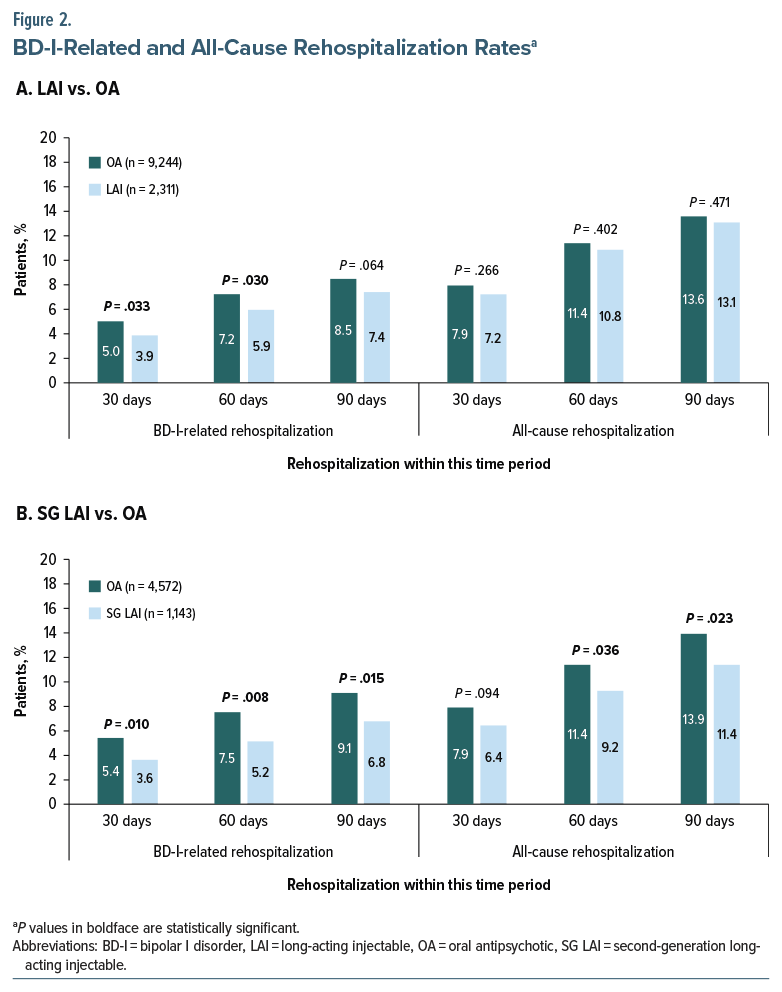
BD-I–related rehospitalization rates were lower for the LAI group than for the OA group within 30 (3.9% vs 5.0%; P=.033), 60 (5.9% vs 7.2%; P= .030), and 90 (7.4% vs 8.5%; P =.064) days, although the 90-day difference was not statistically significant (Figure 2A). The SG LAI subgroup had a lower proportion of patients rehospitalized for BD-I within 30 (3.6% vs 5.4%; P=.010), 60 (5.2% vs 7.5%; P=.008), and 90 (6.8% vs 9.1%; P =.015) days than the OA group (Figure 2B). All-cause rehospitalization rates were not significantly different between the LAI and OA groups. However, in the SG LAI subgroup vs the OA group, the proportion of patients rehospitalized within 30 days was numerically lower (6.4% vs 7.9%; P = .094). Lower rehospitalization rates between the SG LAI and OA groups within 60 (9.2% vs 11.4%; P = .036) and 90 (11.4% vs 13.9%; P = .023) days reached statistical significance (Figure 2B).
Risk of Rehospitalization
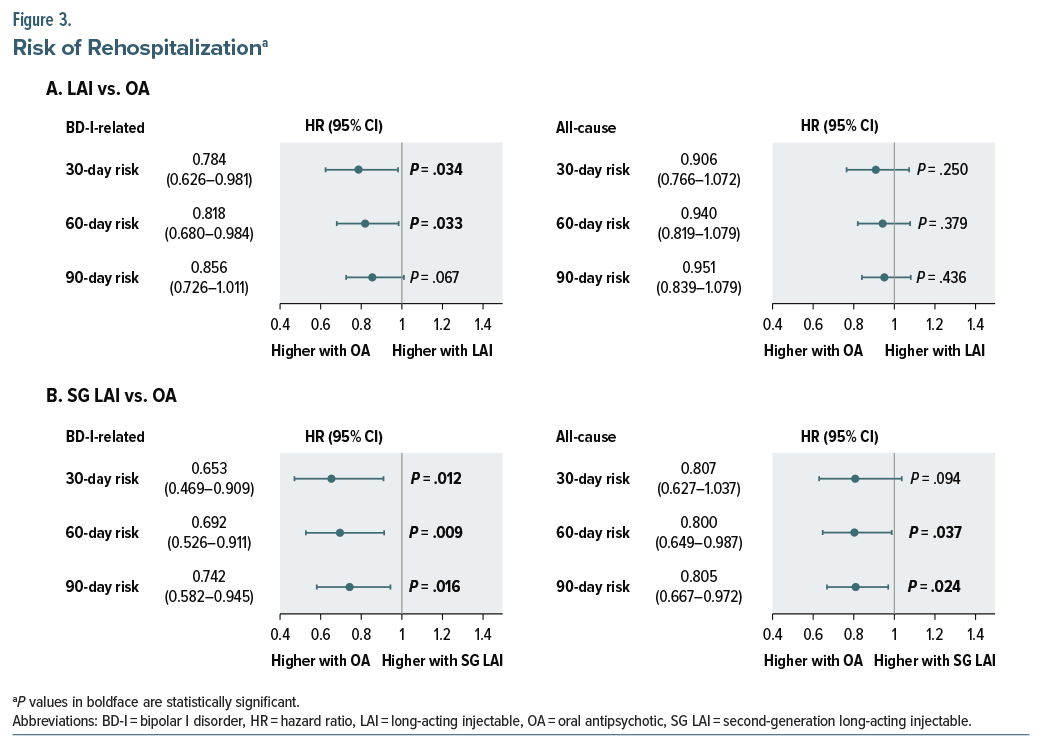
Patients in the LAI group were at lower risk of BD-I–related rehospitalization within 30 (HR: 0.784; 95% CI: 0.626–0.981) and 60 days (HR: 0.818; 95% CI: 0.680–0.984) compared to the OA group. There was also a lower risk (statistically nonsignificant) of BD-I–related rehospitalization for patients in the LAI group within 90 days (HR: 0.856; 95% CI: 0.726–1.011) compared to the OA group (Figure 3A). Patients in the SG LAI subgroup had a lower risk of rehospitalization within 30 (HR: 0.653; 95% CI: 0.469–0.909), 60 (HR: 0.692; 95% CI: 0.526–0.911), and 90 days (HR: 0.742; 95% CI: 0.582–0.945) than patients in the OA group (Figure 3A). For all-cause rehospitalizations, risk was lower in patients in the LAI group than in patients in the OA group within 30, 60, and 90 days; however, these differences were not statistically significant. For patients in the SG LAI subgroup, the risk of rehospitalization within 30 days (HR: 0.807; 95% CI: 0.627–1.037) was numerically lower than in patients in the OA group. Within 60 (HR: 0.800; 95% CI: 0.649–0.987) and 90 (HR: 0.805; 95% CI: 0.667–0.972) days, the lower risk of rehospitalization reached statistical significance for the SG LAI subgroup compared with the OA group (Figure 3B). Kaplan-Meier cumulative incidence plots showed statistically significant differences in risk for BD-I–related rehospitalization between the LAI and OA groups (Supplementary Figures 1A, 1B).
Length of Rehospitalization Stay
The average length of stay of BD-I–related and all-cause rehospitalization within 30, 60, and 90 days was statistically similar between LAI, SG LAI, and OA groups. The average length of stay in days was 10.8 vs 11.0 within 30 days, 10.1 vs 10.4 within 60 days, and 9.9 vs 10.1 within 90 days of BD-I–related rehospitalization for LAI vs OA groups, respectively. When comparing LAI vs OA groups for all-cause rehospitalizations, average length of stay was 10.5 vs 11.6 within 30 days, 10.0 vs 10.7 within 60 days, and 9.9 vs 10.4 within 90 days, respectively.
When comparing SG LAI vs OA groups, average length of stay was 8.5 vs 9.9 within 30 days, 8.6 vs 9.7 within 60 days, and 8.3 vs 9.6 within 90 days of BD-I–related rehospitalization. For all-cause rehospitalizations, average length of stay for SG LAI vs OA was 7.6 vs 10.4, 7.8 vs 9.8, and 7.9 vs 9.7 within 30, 60, and 90 days, respectively.
Continuation of Medication vs Switching at Rehospitalization
While the majority of patients discharged on OA at index (84.4%–86.4%) remained on OA, only 38.9%–41.4% of those discharged on LAIs at rehospitalization within 30–90 days continued with LAI treatment. Among patients discharged on SG LAIs, 35.4%–37.0% were maintained on LAIs at rehospitalization within the same timeframe (Supplementary Figure 2). Exploratory analyses suggested lower rates of second rehospitalization among patients who continued LAIs (and SG LAIs) at first rehospitalization compared with those who switched to OAs, although most differences were not statistically significant. Detailed proportions and timepoints are shown in Supplementary Figures 3B–3E.
DISCUSSION
In this large observational US study of hospital discharges among patients with BD-I, patients discharged on LAIs had lower rates of BD-I–related rehospitalizations within 30 and 60 days than those who were discharged on OAs. The results showed a marked reduction in rehospitalization risk for patients receiving SG LAIs, suggesting that these formulations may provide sustained symptom control, ultimately reducing the likelihood of early relapse and rehospitalization, which is a critical factor in managing chronic psychiatric conditions.7,16
Nonadherence to oral treatments is a significant barrier to effective long-term management of BD-I, leading to increased hospitalizations and poorer overall outcomes.6,31 The results of this study aligned with findings from similar research conducted in schizophrenia populations, where LAIs have similarly been used to address nonadherence and have been shown to reduce hospitalization rates and improve adherence compared to OAs.9–11,13,14 For instance, a meta-analysis found that patients with schizophrenia treated with LAIs experienced significantly fewer relapses and hospitalizations than those on OAs.15 Other studies reported that LAI use, SG LAIs in particular, was associated with a reduced risk of rehospitalization and improved treatment adherence in patients with schizophrenia.12 Newer SG LAIs are better tolerated, which in turn leads to better treatment adherence.21 Among patients with BD, schizophrenia, and schizoaffective disorder, 56.9% of patients taking OA were rehospitalized compared with only 43.1% in the LAI group within 1 year.16
The reduced rehospitalization rates observed in this study among patients discharged on an LAI compared with those discharged on an OA suggest that increasing the use of LAIs could potentially decrease acute care needs, which in turn could alleviate some of this financial burden associated with BD-I (estimated at $202 billion annually).5 Previous findings in patients with schizophrenia have shown that LAIs can reduce hospital readmissions and related healthcare expenditures, supporting their cost-effectiveness.24
Although LAIs were associated with statistically significant reductions in BD-I–related rehospitalization compared with OAs, the absolute effects were modest. At 30 days, the absolute risk rate (ARR) for all LAIs was 1.1% (yielding a number needed to treat [NNT] of approximately 91), whereas SG LAIs showed an ARR of 1.8% (NNT of approximately 56). By 60 days, the ARR for SG LAIs increased to 2.3% (NNT=43), indicating a more clinically meaningful benefit. Given the substantial economic and psychosocial burden of rehospitalization in BD-I,29,31 even these modest absolute reductions may be relevant, particularly in high-risk groups or when combined with adherence-enhancing strategies.
Our findings should be interpreted alongside prior evidence showing limited differences between LAIs and OAs in BD, including meta-analytic and real-world studies reporting comparable effectiveness and high discontinuation rates.32–34 The modest reductions in early rehospitalization observed in our cohort potentially reflect the fact that LAIs confer their greatest benefit during the high-risk postdischarge period, when adherence challenges are most pronounced.1,2,28 Differences from earlier work may stem from variations in study design, patient characteristics, and adherence patterns, as well as real-world factors such as greater clinical expertise among clinicians who prescribe LAIs or more structured discharge planning and follow-up for patients initiated on LAIs.22,23 Additionally, clinicians’ concerns about LAI-induced depression may lead to preferential use in patients with manic-predominant polarity,19 which can influence comparability across treatment groups. These contextual factors may help explain the differences in rehospitalization observed across the various follow-up periods.
To our knowledge, there are no published retrospective US-based studies that have assessed the downstream effects of continuing vs switching the antipsychotic type at rehospitalization in patients with BD-I. Most patients in this study who were discharged on OAs or LAIs continued their medication regimen. A notable proportion of those initially prescribed LAIs at hospitalization were switched to an OA upon rehospitalization. Our analysis found that those who continued with LAIs following rehospitalization demonstrated a lower likelihood of subsequent rehospitalizations, which may help inform clinical decisions on whether to continue or switch medications for patients with BD-I. These findings underscore the need for further exploration of the motivations behind switching medications, as well as the implications of these decisions on patient outcomes and healthcare resource utilization.
In the management of BD-I, recent guidelines emphasized the importance of individualized treatment plans that consider both pharmacologic and psychosocial interventions.8 LAIs were not generally used in the treatment of BD-I, unless a patient presented with severe symptoms, suboptimal adherence to OAs, or experienced multiple relapses.19 Even in patients with schizophrenia, LAI use in early-phase schizophrenia was low (4% among commercially insured patients) and was generally not the first-line therapy.35 A recent consensus panel has also reported that the primary barriers to effective LAI use in BD-I are the attitudes and knowledge or experience of clinicians. Physician perceptions that patients prefer OAs over LAIs limited treatment discussions with their patients.19 Underutilization of LAIs in the management of BD-I warrants further investigation, and future research should focus on understanding the dynamics of treatment decision-making and exploring real-world applications of these findings to ensure that all patients receive effective, personalized care tailored to their unique needs.
Several limitations of the study need to be acknowledged. Risk of potential misdiagnosis was possible, as this study captured patients with no confirmatory second diagnosis of BD-I. Treatment with BD medications was based on routine clinical practice, which may have introduced bias. Although rigorous statistical methods, including PSM, were used to adjust for baseline differences between patients, the study design and the nature of the database, particularly unmeasured factors, such as disease severity, prior treatment history, or comorbidity profile, limited the ability to fully control for all confounders. Even after matching, a slight imbalance remained between the OA and LAI groups in terms of patient sex and age, even though the variables were adjusted for in the analysis of rehospitalization risk and time to rehospitalization. These residual imbalances and unmeasured factors should be considered when interpreting the study findings. The matching process was limited to variables available during encounters within the same hospital with no information on services received outside the hospital. Furthermore, the reliance on hospital discharge data may not have fully captured the complexity of patient experiences outside the hospital setting, including community-based care, medication formulary restrictions, medication utilization, and medication adherence. It must be noted that the observation period of this study (2020–2023) coincided with the COVID-19 pandemic, which might have impacted the availability and administration of LAIs due to social distancing and travel restrictions. The study also did not account for variations in clinician practices or patient preferences, which could have significantly impacted treatment continuation and effectiveness. For instance, the stigma surrounding mental health treatment and the perception of injectable therapies could have influenced patient acceptance and adherence to LAI regimens.2 There was also no information on LAI use in the 3 months before index hospitalization (ie, the baseline period). The definition of the LAI group as any patient receiving an LAI during hospitalization, in contrast to the OA group defined at discharge, may have led to misclassification and potential confounding by indication. Specifically, some patients classified as receiving LAIs may have received OAs at discharge. Nonetheless, restricting the group to prescriptions given immediately before discharge could exclude patients who began an LAI earlier and continued postdischarge, which would not accurately represent the full population exposed to an LAI during inpatient care. Because multiple comparisons were conducted, the risk of type I errors is increased, which increases the potential for false-positive findings. Although no formal correction was applied, the primary analyses of BD-I–related rehospitalization were prespecified. Secondary and descriptive analyses were supportive only and should be interpreted with appropriate caution. Lastly, a subgroup analysis of patients who received first-generation LAIs was not conducted, which made the results less generalizable to settings where these treatments were still used.
CONCLUSION
This study indicated that LAIs, particularly second-generation formulations, were associated with lower rates of rehospitalization among patients with BD-I compared with OAs. The findings underscored the potential of LAIs in reducing the burden on healthcare resources and their potential in improving BD-I management. Underutilization of LAIs for BD-I treatment in clinical practice highlights a critical gap that warrants further investigation. As healthcare systems strive to optimize management strategies for chronic psychiatric conditions, integrating LAIs into standard treatment protocols may improve individual patient outcomes.
Article Information
Published Online: March 2, 2026. https://doi.org/10.4088/JCP.25m16154
© 2026 Physicians Postgraduate Press, Inc.
Submitted: September 23, 2025; accepted January 21, 2026.
To Cite: Kaplan S, Dotiwala Z, Casciano J, et al. Outcomes and practice patterns of long-acting injectable versus oral antipsychotics among patients with bipolar I disorder in the United States: a hospital database analysis. J Clin Psychiatry 2026;87(2):25m16154.
Author Affiliations: Teva Pharmaceutical Industries Ltd., Netanya, Israel (Kaplan); eMAX Health Systems, LLC, Delray Beach, Florida (Dotiwala, Casciano); Teva UK Limited, Harlow, United Kingdom (Suett); Teva Branded Pharmaceutical Products R&D LLC, Parsippany, New Jersey (Phatak); Teva Branded Pharmaceutical Products R&D LLC, West Chester, Pennsylvania (Zhang, Krasenbaum); University Hospitals Cleveland Medical Center, Case Western Reserve University School of Medicine, Cleveland, Ohio (Sajatovic).
Corresponding Author: Sigal Kaplan, PhD, BPharm, Teva Pharmaceutical Industries Ltd, 12 Hatrufa St, Netanya 42504, Israel ([email protected]).
Relevant Financial Relationships: Drs Kaplan, Suett, Phatak, and Zhang and Ms Krasenbaum are employees and/or shareholders of Teva Pharmaceuticals. Ms Dotiwala and Mr Casciano are employees of eMAX Health Systems, which has received funding from Teva Pharmaceuticals for data and analytics in relation to this project. Dr Sajatovic has been awarded grants from Neurelis, Intra-Cellular Therapies, Merck, Otsuka, Alkermes, and Teva within the past 3 years; has been a consultant for Alkermes, Otsuka, Lundbeck, Janssen, and Teva Pharmaceuticals over the past year; has received royalties from Springer Press, Johns Hopkins University Press, Oxford Press, and UpToDate over the past year; and has been compensated for preparation of and/or participation in CME activities over the past year for the American Physician Institute (CMEtoGO), Psychopharmacology Institute, American Epilepsy Society, and Clinical Care Options.
Funding/Support: Funded by Teva Branded Pharmaceutical Products R&D LLC.
Role of the Sponsor: The sponsor participated in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and funded the preparation of the manuscript. All authors, including those affiliated with the sponsor, fulfilled all authorship criteria and participated in the review and approval of the manuscript and in the decision to submit the manuscript for publication.
Previous Presentation: Part of this work was previously presented at the Academy of Managed Care Pharmacy (AMCP) Annual Meeting; March 31–April 3, 2025; Houston, Texas, and at NEI Saturday Morning Live; April 26, 2025; virtually.
Data Sharing Statement: Qualified researchers may request access to patient-level data and related study documents, including the study protocol and the statistical analysis plan. Requests will be reviewed for scientific merit, product approval status, and conflicts of interest. Patient level data will be de-identified, and study documents will be redacted to protect the privacy of participants and to protect commercially confidential information. Please email [email protected] to make your request.
Acknowledgments: Medical writing and editorial support were provided by Jean-Paul Fouche, PhD; Alison Adams, PhD, CMPP; and Kelsey Gribbon, MS, of Ashfield MedComms, an Inizio company, and were funded by Teva Branded Pharmaceutical Products R&D, LLC.
ORCID: Sigal Kaplan: https://orcid.org/0000-0002-3352-8480
Supplementary Material: Available at Psychiatrist.com.
Clinical Points
- Evidence on real-world rehospitalization outcomes with long-acting injectable antipsychotics (LAIs) in bipolar I disorder is limited.
- Second-generation LAIs may reduce early rehospitalization in bipolar I disorder, particularly for patients with poor adherence or high early relapse risk.
- Incorporating LAIs into discharge and follow-up planning may support adherence and reduce short-term readmissions.
References (35)

- Bobo WV. The diagnosis and management of bipolar I and II disorders: clinical practice update. Mayo Clin Proc. 2017;92(10):1532–1551. PubMed CrossRef
- Carvalho AF, Firth J, Vieta E. Bipolar disorder. N Engl J Med. 2020;383(1):58–66. PubMed CrossRef
- GBD 2019 Mental Disorders Collaborators. Global, regional, and national burden of 12 mental disorders in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Psychiatry. 2022;9(2):137–150. PubMed CrossRef
- Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the World Mental Health Survey Initiative. Arch Gen Psychiatry. 2011;68(3):241–251. PubMed CrossRef
- Yan T, Greene M, Chang E, et al. Hospitalization risk factors in antipsychotic-treated schizophrenia, bipolar I disorder or major depressive disorder. J Comp Eff Res. 2019;8(4):217–227. PubMed CrossRef
- Peters AT, West AE, Eisner L, et al. The burden of repeated mood episodes in bipolar I disorder: results from the National Epidemiological Survey on Alcohol and Related Conditions. J Nerv Ment Dis. 2016;204(2):87–94. PubMed CrossRef
- Tohen M, Goldberg JF, Hassoun Y, et al. Identifying profiles of patients with bipolar I disorder who would benefit from maintenance therapy with a long-acting injectable antipsychotic. J Clin Psychiatry. 2020;81(4):OT19046AH1. PubMed CrossRef
- Oliva V, Fico G, De Prisco M, et al. Bipolar disorders: an update on critical aspects. Lancet Reg Health Eur. 2025;48:101135. PubMed CrossRef
- Dickson MC, Nguyen MM, Patel C, et al. Adherence, persistence, readmissions, and costs in Medicaid members with schizophrenia or schizoaffective disorder initiating paliperidone palmitate versus switching oral antipsychotics: a real-world retrospective investigation. Adv Ther. 2023;40(1):349–366. PubMed CrossRef
- Patel C, Emond B, Lafeuille MH, et al. Real-world analysis of switching patients with schizophrenia from oral risperidone or oral paliperidone to once-monthly paliperidone palmitate. Drugs Real World Outcomes. 2020;7(1):19–29. PubMed CrossRef
- Rubio JM, Taipale H, Tanskanen A, et al. Long-term continuity of antipsychotic treatment for schizophrenia: a nationwide study. Schizophr Bull. 2021;47(6):1611–1620. PubMed CrossRef
- Kane JM, Rubio JM, Casciano J, et al. Real-world outcomes and practice patterns among patients with schizophrenia when switched from oral antipsychotics to long-acting injectable formulations after hospitalization. Psychiatry Res. 2025;348:116455. PubMed CrossRef
- Kane JM, Schooler NR, Marcy P, et al. Effect of long-acting injectable antipsychotics vs usual care on time to first hospitalization in early-phase schizophrenia: a randomized clinical trial. JAMA Psychiatry. 2020;77(12):1217–1224. PubMed CrossRef
- Kishimoto T, Hagi K, Kurokawa S, et al. Long-acting injectable versus oral antipsychotics for the maintenance treatment of schizophrenia: a systematic review and comparative meta-analysis of randomised, cohort, and pre-post studies. Lancet Psychiatry. 2021;8(5):387–404. PubMed CrossRef
- Leucht S, Cipriani A, Spineli L, et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet. 2013;382(9896):951–962. PubMed CrossRef
- Maestri TJ, Mican LM, Rozea H, et al. Do long-acting injectable antipsychotics prevent or delay hospital readmission?. Psychopharmacol Bull. 2018;48(3):8–15. PubMed CrossRef
- Abilify Maintena. Product monograph. Otsuka Pharmaceutical Co., Ltd.; 2021.
- Risperdal Consta. Package insert. Janssen Pharmaceutical Companies; 2021.
- Vieta E, Tohen M, McIntosh D, et al. Early use of long-acting injectable antipsychotics in bipolar disorder type I: an expert consensus. Bipolar Disord. 2025;27(1):7–16. PubMed CrossRef
- Llorca PM, Abbar M, Courtet P, et al. Guidelines for the use and management of long-acting injectable antipsychotics in serious mental illness. BMC Psychiatry. 2013;13:340. PubMed CrossRef
- Correll CU, Citrome L, Haddad PM, et al. The use of long-acting injectable antipsychotics in schizophrenia: evaluating the evidence. J Clin Psychiatry. 2016;77(suppl 3):1–24. PubMed CrossRef
- Sajatovic M, Ross R, Legacy SN, et al. Initiating/maintaining long-acting injectable antipsychotics in schizophrenia/schizoaffective or bipolar disorder - expert consensus survey part 2. Neuropsychiatr Dis Treat. 2018;14:1475–1492. PubMed CrossRef
- Sajatovic M, Ross R, Legacy SN, et al. Identifying patients and clinical scenarios for use of long-acting injectable antipsychotics - expert consensus survey part 1. Neuropsychiatr Dis Treat. 2018;14:1463–1474. PubMed CrossRef
- Patel R, Liman C, Oyesanya M, et al. Retrospective cohort study of long-acting injectable (LAI) antipsychotic initiation in the inpatient setting: impact of LAI characteristics on transition and continuation of care among patients with schizophrenia in the USA. BMJ Open. 2025;15(3):e092216. PubMed CrossRef
- Jackson JW, Fulchino L, Rogers J, et al. Impact of drug-reimbursement policies on prescribing: a case-study of a newly marketed long-acting injectable antipsychotic among relapsed schizophrenia patients. Pharmacoepidemiol Drug Saf. 2018;27(1):95–104. PubMed CrossRef
- Fu AZ, Pesa JA, Lakey S, et al. Healthcare resource utilization and costs before and after long-acting injectable antipsychotic initiation in commercially insured young adults with schizophrenia. BMC Psychiatry. 2022;22(1):250. PubMed CrossRef
- Lohman MC, Scott V, Verma M, et al. Distribution and correlates of long-acting injectable antipsychotic use among community mental health center patients. Psychiatry Res. 2025;345:116378. PubMed CrossRef
- Greene M, Yan T, Chang E, et al. Medication adherence and discontinuation of long-acting injectable versus oral antipsychotics in patients with schizophrenia or bipolar disorder. J Med Econ. 2018;21(2):127–134. PubMed CrossRef
- Bessonova L, Ogden K, Doane MJ, et al. The economic burden of bipolar disorder in the United States: a systematic literature review. Clinicoecon Outcomes Res. 2020;12:481–497. PubMed CrossRef
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383. PubMed CrossRef
- Dembek C, Mackie D, Modi K, et al. The economic and humanistic burden of bipolar disorder in adults in the United States. Ann Gen Psychiatry. 2023;22(1):13. PubMed CrossRef
- Prajapati AR, Wilson J, Song F, et al. Second-generation antipsychotic long-acting injections in bipolar disorder: systematic review and meta-analysis. Bipolar Disord. 2018;20(8):687–696. PubMed CrossRef
- Gentile S. Discontinuation rates during long-term, second-generation antipsychotic long-acting injection treatment: a systematic review. Psychiatry Clin Neurosci. 2019;73(5):216–230. PubMed CrossRef
- Korkmaz SA, Gurler S. Real-world effectiveness of long-acting injectable vs. oral antipsychotics in patients with bipolar I disorder: a 1-year retrospective observational study. Curr Med Res Opin. 2024;40(5):855–861. PubMed CrossRef
- Kane JM, Mychaskiw MA, Lim S, et al. Treatment journey from diagnosis to the successful implementation of a long-acting injectable antipsychotic agent in young adults with schizophrenia. J Clin Psychiatry. 2023;84(3):22m14544. PubMed CrossRef
This PDF is free for all visitors!