Abstract
Objective: This exploratory post hoc analysis examined the safety/efficacy of the aripiprazole once-monthly 400 mg (AOM 400) long-acting injectable (LAI) in Black/African American patients diagnosed with bipolar I disorder (BP-I).
Methods: Data were from a 52-week, open-label trial of AOM 400 maintenance treatment in patients diagnosed with DSM-IV-TR–defined BP-I. Outcomes included treatment-emergent adverse events (TEAEs), clinician-rated extrapyramidal symptoms (EPS), patient stability, Young Mania Rating Scale (YMRS), Montgomery–Asberg Depression Rating Scale (MADRS), and Clinical Global Impression–Bipolar Version–Severity (CGI-BP-S) scores, functioning, and quality of life (QoL). Data analyses comprised descriptive statistics and a mixed model for repeated measures.
Results: Outcomes were analyzed in 464 patients (Black/African American, n=104; White, n=255; Asian, n=94; other racial groups, n=11). No notable increase in TEAEs, serious TEAEs, or TEAEs leading to discontinuation were observed in Black/African American patients vs those in other racial groups. Rates of akathisia, tremor, increased weight, or hypertension were lower/similar in Black/African American patients vs other racial groups; changes in EPS scores were minimal in all groups. At last visit, 90.1% of Black/African American patients were stable, similar to other racial groups. Small changes in YMRS total score occurred in all groups, with MADRS total score and CGI-BP-S scores largely unchanged. Functioning and QoL improved in Black/African American patients, to a similar/greater degree than other racial groups. Limitations include the open-label design, prior aripiprazole stabilization, and sparse metabolic laboratory data, constraining causal inference and metabolic conclusions.
Conclusion: The safety and efficacy of AOM 400 is comparable between Black/African American and non-Black/African American patients with BP-I. The data provide valuable evidence supporting second-generation LAI antipsychotic use in these patients.
Trial Registration: ClinicalTrials.gov identifier: NCT01710709.
J Clin Psychiatry 2026;87(2):25m16114
Author affiliations are listed at the end of this article.
Racial disparities are evident in the management of bipolar disorder, including notable differences in antipsychotic prescription frequency, dosage, and type.1,2 Specifically, Black/African American people diagnosed with bipolar disorder are more likely than their White counterparts to receive an antipsychotic1,2 and to receive them at higher doses independent of any comorbid schizophrenia or depression.1 In addition, Black/African American patients diagnosed with bipolar disorder are more often prescribed a first-generation antipsychotic (FGA) than White patients, including higher-potency FGAs (ie, haloperidol or fluphenazine).1–3
There is an apparent lack of clinical trial data on the efficacy and tolerability of newer, second-generation antipsychotics (SGAs), particularly SGA long-acting injectables (LAIs), in Black/African American patients. While a clinical trial subpopulation analysis of an SGA LAI has been performed in Black/African American patients diagnosed with schizophrenia,4 to the best of the authors’ knowledge, no comparable analyses exist in bipolar disorder. As such, it is not known to what extent race-based antipsychotic prescribing practices in bipolar disorder reflect the efficacy and safety of SGA LAIs in Black/African American patients or are the result of other factors.
Aripiprazole is an SGA indicated in the United States for the treatment of patients diagnosed with bipolar I disorder (BP-I).5–7 It is available as an orally administered tablet,6 or an LAI maintenance treatment administered either once-monthly (aripiprazole once-monthly 400 mg [AOM 400])7 or once every 2 months (aripiprazole 2-month ready-to-use 960 mg [Ari 2MRTU 960]).5 In a double-blind, placebo-controlled, randomized withdrawal study, AOM 400 was effective and well tolerated for the maintenance treatment of BP-I, delaying mood episode recurrence and maintaining symptom control and functioning.8–10 A related extension study demonstrated the long-term safety and efficacy of AOM 400.11 The large dataset of the extension study and inclusion of notable numbers of Black/African American patients provides an opportunity to examine outcomes with AOM 400 in Black/African American individuals, who have historically been under-represented in clinical trials.12 Thus, the current analysis examined the safety and efficacy of AOM 400 in Black/African American patients diagnosed with BP-I.
METHODS
Study Overview and Design
This was an exploratory post hoc analysis of outcomes in Black/African American adults diagnosed with BP-I treated with AOM 400. Data were from a 52-week multicenter study (ClinicalTrials.gov identifier: NCT01710709),11 which was an open-label extension to a 52-week double-blind, randomized withdrawal study of AOM 400 vs placebo in the maintenance treatment of BP-I (NCT01567527).9 The extension study aim was to assess long-term safety (primary objective) and efficacy (secondary objective) of maintenance treatment with AOM 400, and it was conducted between November 2012 and November 2016.11 It comprised a screening phase, oral aripiprazole cross-titration and stabilization phases, and an AOM 400 maintenance phase. The requirement to complete each study phase was determined by patient cohort (ie, de novo [newly enrolled into the extension study] or rollover [recruited from the double-blind, placebo-controlled lead-in study]) and prior antipsychotic treatment11 (Supplementary Figure 1).
The study protocol was approved by an institutional review board or independent ethics committee, with informed consent obtained from all patients (or their legal representatives, where relevant).11
Eligibility Criteria
Key inclusion criteria for the de novo cohort were as follows: male and female outpatients aged 18–65 years; a diagnosis of BP-I per the criteria of the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision,13 confirmed using the Mini-International Neuropsychiatric Interview14; and ≥1 previous manic or mixed episode, with manic symptoms that necessitated hospitalization, or treatment with a mood stabilizer or an antipsychotic. Key exclusion criteria included a current depressive episode or ≥9 episodes in the past year.11 Patients in the rollover cohort must have completed the 52-week maintenance phase of the double-blind, placebo-controlled lead-in study (AOM 400 or placebo arm) without recurrence of a mood episode.11
Study Treatment and Endpoints
During the extension study maintenance phase, patients received AOM 400 on an open-label basis; treatment was a single gluteal intramuscular injection every 4 weeks, for a maximum of 52 weeks.11 For rollover patients, per a protocol amendment, the open-label phase was reduced from 52 to 28 weeks.
For the current analysis, selected outcomes were analyzed in Black/African American patients and compared with those in other racial groups. Safety and tolerability endpoints included treatment-emergent adverse events (TEAEs), with a focus on extrapyramidal symptoms (EPS) and metabolic TEAEs; clinician-rated EPS (using the Abnormal Involuntary Movement Scale, the Barnes Akathisia Rating Scale, and the Simpson–Angus Scale15–17); weight gain; and metabolic laboratory assessments. The main efficacy endpoint was the proportion of stable patients at baseline (ie, start of the maintenance phase) who remained stable at the last visit.11 Stability was defined as (1) outpatient status, (2) Young Mania Rating Scale (YMRS)18 total score of ≤12, (3) Montgomery–Asberg Depression Rating Scale (MADRS)19 total score of ≤12, and (4) no active suicidality (with active suicidality defined as MADRS item 10 score of ≥4, or “yes” on question 4 or 5 of the Columbia-Suicide Severity Rating Scale).11 Other endpoints included individual clinical stability indicators (ie, YMRS total score, MADRS total score, and Clinical Global Impression–Bipolar Version–Severity [CGI-BP-S] scores [overall, mania, and depression])20; patient quality of life (QoL), assessed using the Brief Quality of Life in Bipolar Disorder (Brief QoL.BD) questionnaire21; and patient functioning, assessed using the Functioning Assessment Short Test (FAST) questionnaire.22
Race was self-reported by trial participants, according to the following predefined response options: White; Black or African American; American Indian or Alaska Native; Asian; Native Hawaiian or other Pacific Islander; or other (to be specified). This information was recorded by site personnel in a demographic questionnaire that formed part of the case report form.
Statistical Analysis
Safety endpoints were examined using descriptive statistics. Changes from baseline were analyzed using a mixed model for repeated measures (MMRM), including fixed effects for race, region, study week, and race-by-week interaction, as well as baseline score and baseline-by-week interaction as covariates. The model was consistent with that used in the lead-in study9 and in a previous post hoc analysis.23 The model was estimated using restricted maximum likelihood with Kenward–Roger degrees of freedom. An unstructured (UN) covariance matrix was used as the default within-subject covariance structure. If the UN structure failed to converge, progressively simpler covariance structures, including compound symmetry–heterogeneous, Toeplitz–heterogeneous, autoregressive(1)–heterogeneous, compound symmetry, variance components, autoregressive(1), Toeplitz, factor analytic, and Huynh–Feldt, were evaluated sequentially. For each alternative structure, empirical (sandwich) robust standard errors were applied. The first covariance structure that achieved stable convergence and produced interpretable estimates was selected for the final analysis. Consistent with the prespecified MMRM model for efficacy outcomes, it was assumed that data were missing at random, with analyses based on observed data without imputation.
A correlation test was undertaken to determine whether there was a significant relationship between clinical stability and functioning endpoints. Nominal P values were reported, with Cohen d used to calculate effect sizes.
Although no formal hypotheses were specified, a post hoc power analysis was conducted to help contextualize the efficacy and safety findings, with power calculations based on a 2-sided α of .05. Various sensitivity analyses were conducted to assess the robustness of the findings under alternative analytical assumptions. These included the following: inclusion of baseline body mass index (BMI) as a model term; application of alternative imputation methods for missing data (ie, last observation carried forward to account for nonsystematic early dropout and multiple imputation combined using Rubin’s rule); exposure-adjusted discontinuations per 100 patient-years; and exposure-adjusted TEAE incidence rates.
RESULTS
Patients
Data were analyzed for 464 patients (Black/African American, n = 104; White, n = 255; Asian, n = 94; other racial groups, n = 11), of whom 85 were rollover patients.11 The study completion rate was 62.7% (291/464). Reasons for discontinuation according to self-reported race are shown in Supplementary Table 1, while a Kaplan–Meier curve depicting time to discontinuation according to race is shown in Supplementary Figure 2. The frequency of all-cause discontinuation was higher in Black/African American patients (51.0%) vs that in White patients (37.6%), Asian patients (23.4%), and patients of other racial groups (18.2%), driven by proportionately more Black/African American patients meeting protocol-specified withdrawal criteria (Supplementary Table 2). Further analysis showed that, of the 18 Black/African American patients who met the predefined withdrawal criteria, 15 were because of a positive test for drug use; this was compared with 8/11 White patients, 0/3 Asian patients, and 1/1 patients of other races who met the predefined withdrawal criteria. Discontinuation rates for other reasons (eg, loss to follow-up, adverse events, subject withdrawal of consent) were generally similar among the different racial groups (Supplementary Table 2). A Cox regression model that adjusted for region and baseline BMI showed no statistically significant differences in time to treatment discontinuation between Black/African American patients and patients of other races after controlling for multiplicity using simultaneous inference (all adjusted P values >.05).
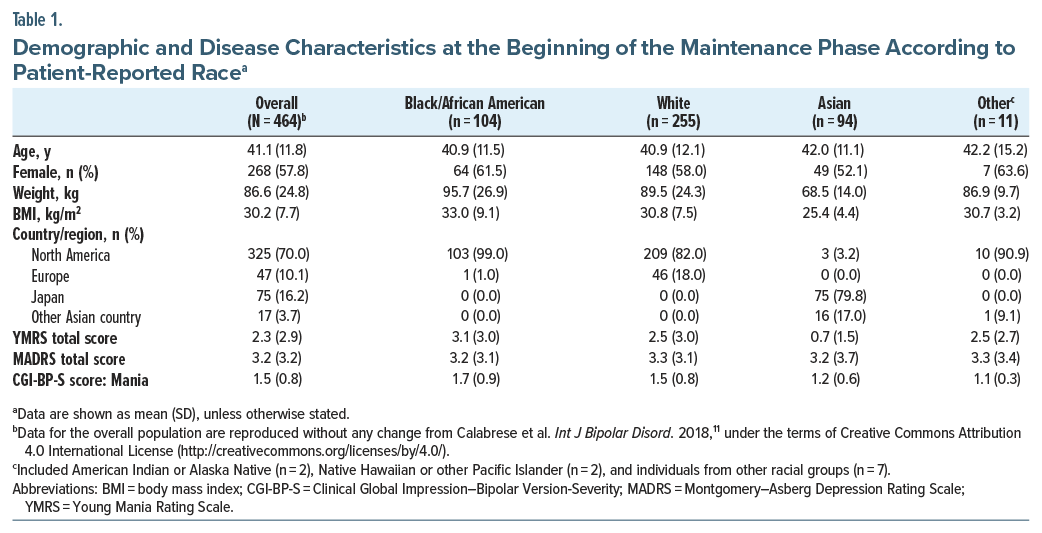
Demographic and disease characteristics at the start of the maintenance phase are shown in Table 1. Almost all Black/African American participants were enrolled in North America, while almost all Asian participants were enrolled in Japan or another country in Asia. YMRS scores at baseline were lower in Asian patients vs Black/African American and White patients, and variation was observed across the different racial groups in the proportion of female patients (52.1–63.6%) and mean BMI (25.4–33.0 kg/m2). For the difference in BMI, Cohen d was 0.48, representing a small-to-medium difference for Black/African American patients vs those of other races (t test, P = .0003).
Safety
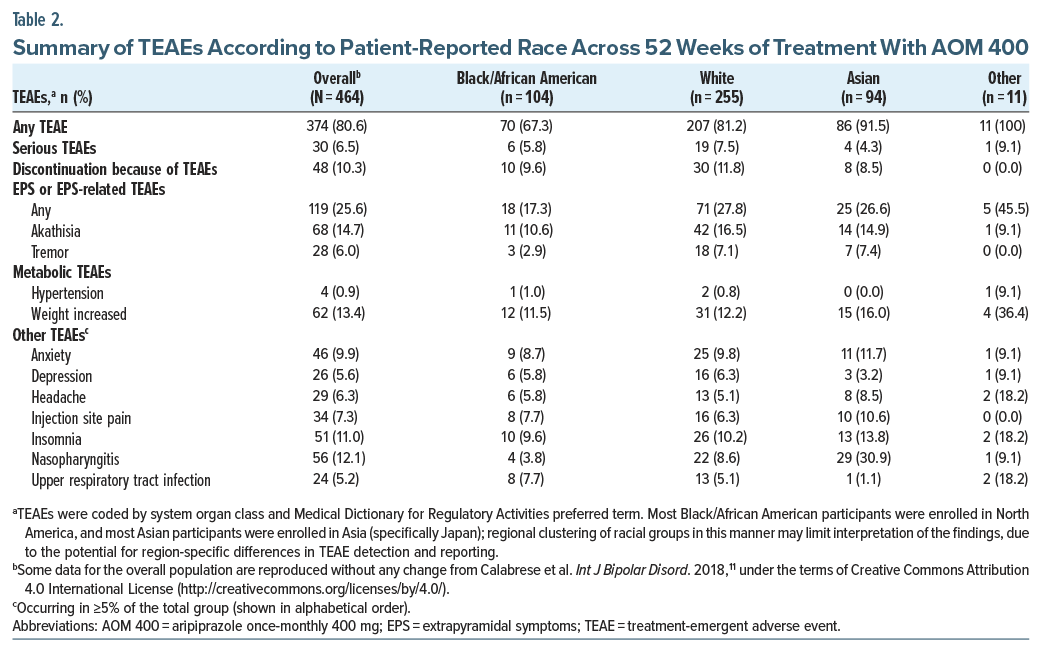
Treatment-emergent adverse events. The incidence of TEAEs was lower in Black/African American patients (67.3%) vs non-Black/African American patients (White, 81.2%; Asian, 91.5%; other racial groups, 100%) (Table 2). Rates of serious TEAEs and discontinuations due to TEAEs were similar across all racial groups (Table 2).
There was a tendency for lower or similar rates of akathisia, tremor, increased weight, or hypertension in Black/African American patients vs those of other races (Table 2). Lower rates of TEAEs, including akathisia, in Black/African American patients were not driven by a lower dose of AOM; compared with White or Asian study participants, fewer Black/African American patients received a reduced AOM dose of 300 mg (Supplementary Table 3). Also, lower rates of akathisia in Black/African American patients vs White or Asian patients were not driven by increased use of concomitant anticholinergics or benzodiazepine derivatives (Supplementary Table 4).
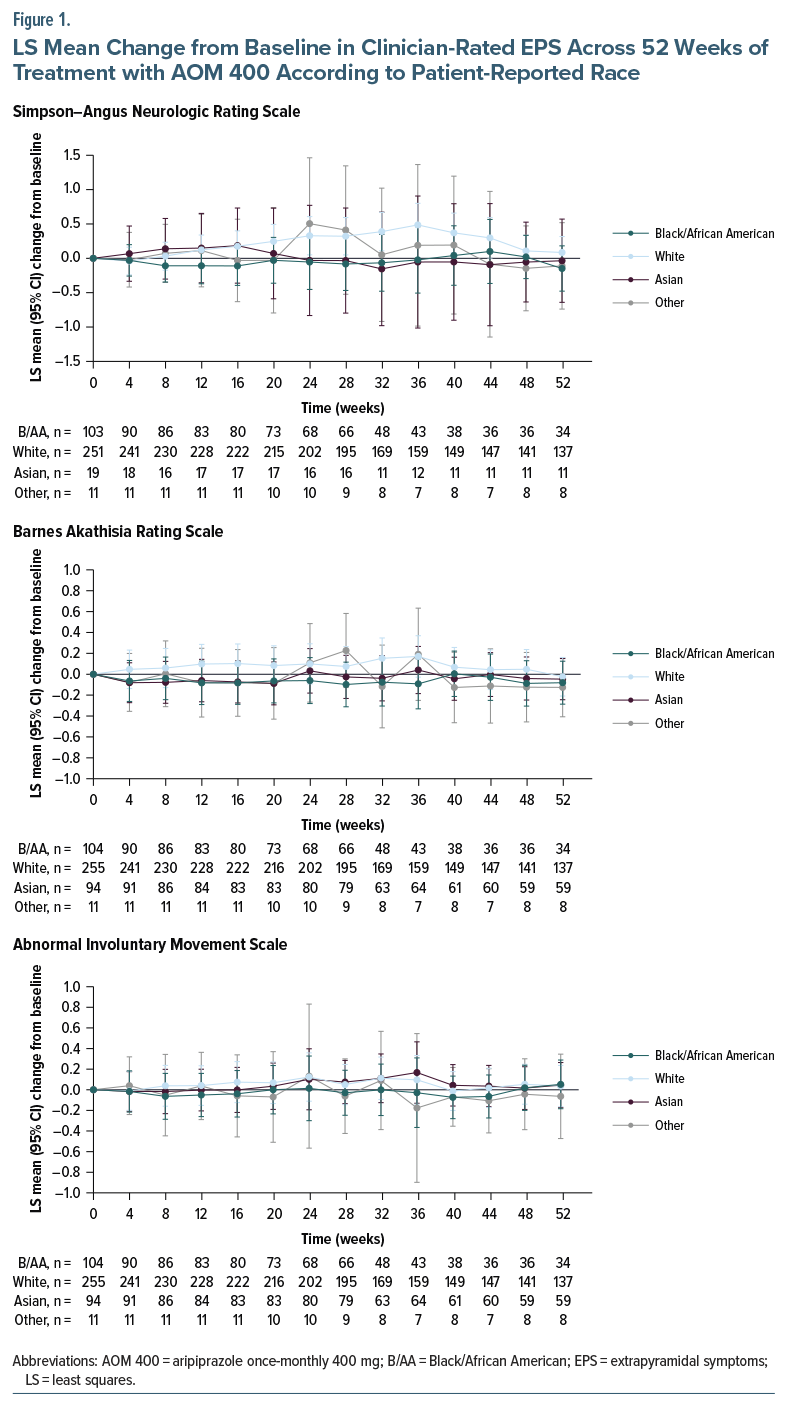
Clinician-rated extrapyramidal symptoms. Changes in EPS rating scale scores during treatment with AOM 400 were minimal and not clinically meaningful across the different racial groups (Figure 1).
Weight gain of ≥7%. The proportion of Black/African American patients with a ≥7% increase from baseline in body weight at any time point was 16.3% (n/N = 17/104), similar to that in Asian patients (17.0% [16/94]) and lower than that in White patients (21.6% [55/255]) or patients of other races (45.5% [5/11]).
Metabolic laboratory parameters. Data for glycosylated hemoglobin (HbA1c) at the last study visit were available for only 6.9% (32/464) of the overall study population, precluding any meaningful analysis according to race. The proportion of patients with a potentially clinically relevant laboratory value at any time point for fasting triglycerides was lower in Black/African American patients (29.2% [21/72]) vs those of other races (White, 40.4% [69/171]; Asian, 33.0% [29/88]; other racial groups, 71.4% [5/7]). For glucose and lipids other than triglycerides, the small number of patients with potentially clinically relevant values prevented a meaningful analysis according to race.
Prolactin levels. At week 52, 4 of 236 patients (1.7%) in the overall study population had prolactin levels above the upper limit of normal, but no clinically meaningful shifts in prolactin were observed during the study. One patient, a 46-year-old Black/African American female, was withdrawn because of a serious adverse event of moderate hyperprolactinemia; this was identified on the day her first and only AOM 400 dose was administered and was considered unrelated to AOM 400. At the early termination visit, the patient’s prolactin level had recovered to within the normal range.
Efficacy
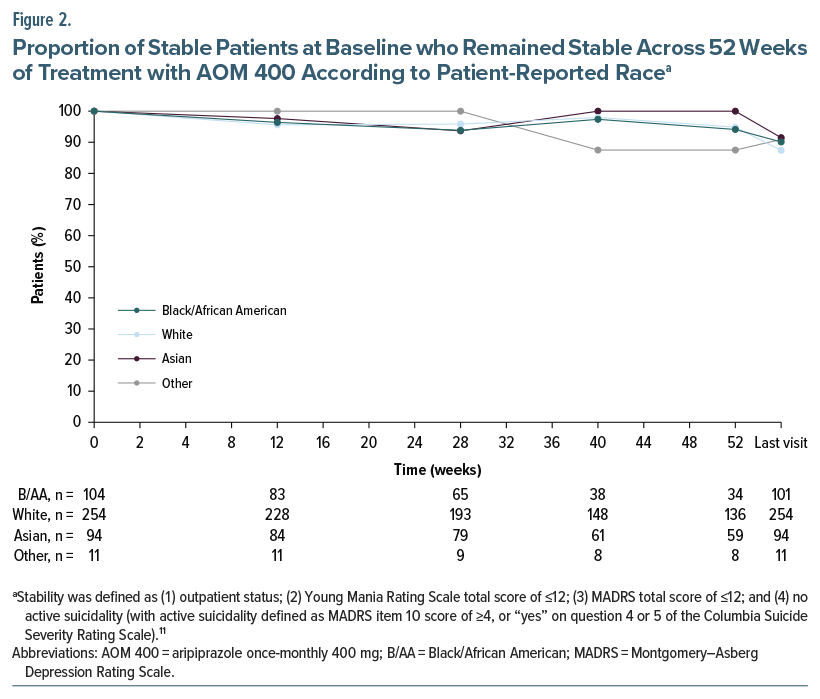
A high proportion (90.1%) of Black/African American patients remained stable at the last visit, similar to patients of other races (White, 87.4%; Asian, 91.5%; other racial groups, 90.9%) (Figure 2).
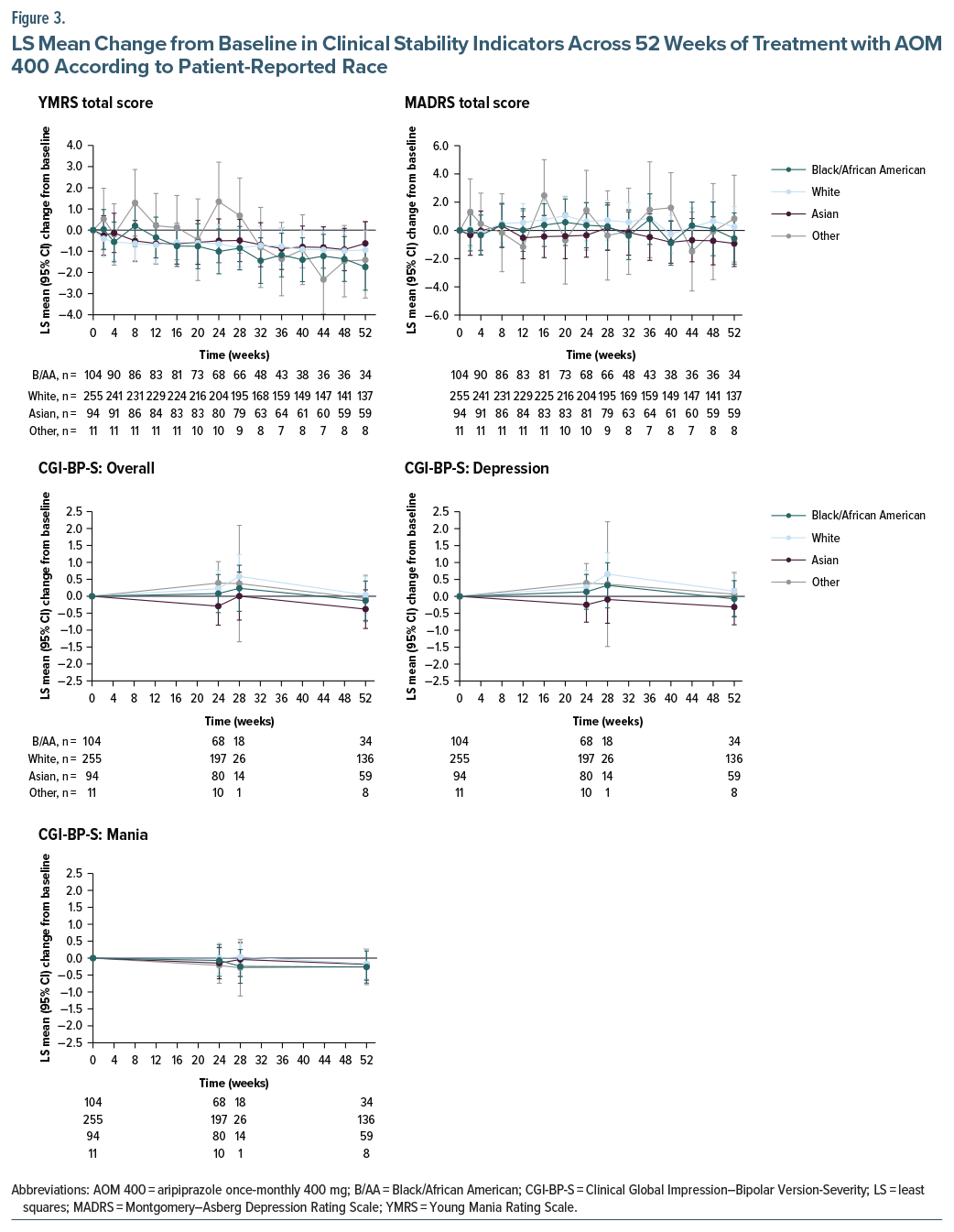
A small improvement in YMRS total score from baseline to week 52 was observed across all racial groups, while MADRS total score and CGI-BP-S scores remained largely unchanged from baseline to week 52 across all groups, indicating that patients remained stable (Figure 3).
The least squares (LS) mean change in FAST total score from baseline to week 52 was greater in Black/African American patients vs those of other races (Black/African American [n=31], –5.97 [95% confidence interval {CI}: –8.82 to –3.13]; White [n =134], –1.89 [95% CI: –3.24 to –0.53], P=.0113, Cohen d=0.30); (Asian [n =59], –1.93 [95% CI: –3.97 to 0.12], P= .0241, Cohen d= 0.33; other racial groups [n=7], 0.15 [95% CI: –5.81 to 6.10], P=.0679, Cohen d=0.61). Additionally, a statistically significant positive correlation was observed between improvements at week 52 in FAST total score and change in mood scores in Black/African American patients; the correlation was 0.22 (P=.0010) between FAST total score and YMRS total score and 0.34 (P<.0001) between FAST total score and MADRS total score.
The LS mean change in Brief QoL.BD total score from baseline to week 52 was generally similar in Black/African American patients vs those of other races (Black/African American [n=34], 2.50 [95% CI: –0.15 to 5.15]; White [n =137], 1.93 [95% CI: 0.60 to 3.26], P=.7034, Cohen d =0.04; Asian [n =59], –1.02 [95% CI: –3.10 to 1.06], P=.0412, Cohen d=0.29; other racial groups [n=8], 2.96 [95% CI: –2.53 to 8.45], P=.8822, Cohen d=0.05). LS mean changes in certain Brief QoL.BD line items improved to a greater extent in Black/African American patients vs non-Black/African American patients. This included “Felt Physically Well” (Black/African American [n =34], 0.49 [95% CI: 0.18 to 0.80]; White [n =137], 0.20 [95% CI: 0.04 to 0.36], P=.0999, Cohen d=0.19; Asian [n= 59], –0.13 [95% CI: –0.38 to 0.11], P=.0024, Cohen d=0.44; other racial groups [n=8], –0.04 [95% CI: –0.69 to 0.61], P=.1485, Cohen d=0.46); and “Woken Up Feeling Refreshed” (Black/African American [n =34], 0.51 [95% CI: 0.17 to 0.85]; White [n =137], –0.01 [95% CI: –0.18 to 0.17], P=.0087, Cohen d=0.31; Asian [n= 59], –0.13 [95% CI: –0.39 to 0.13], P=.0038, Cohen d=0.42; other racial groups [n=8], 0.47 [95% CI: –0.24 to 1.18], P=.9214, Cohen d=0.03).
Post Hoc Power Analysis
Based on a subgroup size of n=104 for Black/African American patients (representing 22.4% of the overall population), a post hoc power analysis provided ≥80% power to detect a ≥9% increase in discontinuations due to TEAEs (from 10% in the overall population), an 11.6% decrease in overall TEAE incidence (from 80.6% in the overall population), and a 9% decrease in the proportion of stable patients at baseline who remained stable across 52 weeks of treatment (from 90% in the overall population). Effect sizes were between 0.26 and 0.28, representing a small effect that could be reasonably detected with the subgroup size.
Sensitivity Analyses
The results of sensitivity analyses incorporating BMI as an additional model term and using alternative methods for handling missing data were consistent with the main analysis. A sensitivity analysis of exposure-adjusted discontinuations per 100 patient-years showed a higher incidence rate of all-cause discontinuation in Black/African American patients vs those of other races, consistent with the main analysis (Supplementary Figure 2). A sensitivity analysis of exposure-adjusted TEAE incidence rates provided results that were consistent with the main analysis.
DISCUSSION
The current post hoc analysis used data from a 52-week extension study11 to examine the safety and efficacy of AOM 400 in Black/African American patients diagnosed with BP-I. Results demonstrated that AOM 400 was associated with maintenance of stability and functional improvement in Black/African American patients in an open-label maintenance cohort previously stabilized on aripiprazole, with safety outcomes that were comparable to patients of other races. In addition, QoL and functioning improved with AOM 400 in Black/African American patients to a similar or greater extent than in patients of other races.
Theoretically, genetic and metabolic factors may influence antipsychotic dose or tolerability in Black/African American individuals, including a higher prevalence of CYP2D6 poor metabolizers and a greater degree of insulin resistance/metabolic dysregulation compared with certain other racial groups.24–26 Despite this, the current analysis provided no indication that AOM 400 had a worse tolerability profile in Black/African American patients. Rates of akathisia and tremor with AOM 400 in Black/African American patients were lower than or similar to those in other racial groups, while changes in EPS rating scale scores during treatment were minimal and not clinically meaningful. This is in contrast to historical data indicating a greater risk of EPS in Black/African American vs White patients, seemingly driven by increased use of FGAs. In a study that examined racial disparities in FGA prescriptions in adult patients with psychiatric and neurological conditions, including bipolar disorder, those of Black/African American race were more than twice as likely as their White counterparts to receive an FGA.3 Related to this, Black/African American patients were more than twice as likely as White patients to receive concurrent benztropine for extrapyramidal side effects, with most of this difference driven by FGA use.3 No such finding was observed in the current study; in fact, concurrent use of benzodiazepine derivatives was lower in Black/African American patients. This indicates that the nominally lower rates of akathisia and tremor in the Black/African American group compared with the White and Asian groups were not an artifact of increased use of medications to manage EPS. Similarly, the lower rate of akathisia and the lack of signal for weight gain were not attributable to reduced drug exposure, since Black/African American patients were less likely to receive a dose reduction to 300 mg than patients from other racial groups. Regardless, these safety observations remain descriptive in nature and must be confirmed in adequately designed studies with prospectively collected metabolic panels.
Sparse data for HbA1c, glucose, and lipids (excluding triglycerides) limit any meaningful metabolic conclusions in the current analysis. Broadly speaking, the risk of metabolic side effects among Black/African American patients exposed to SGAs is varied, with a general trend toward an increased risk, compared with other racial groups.27 This may be driven by higher rates of metabolic comorbidities in this population (eg, obesity, insulin resistance, diabetes, and hypertension),24,26,28 which may discourage clinicians from prescribing SGAs for Black/African American patients.29 While this decision may be clinically appropriate, for example, in a patient with a high metabolic side-effect risk, it does not entirely explain the pattern of greater use of FGAs in Black/African American vs White patients. Historical data show a trend for greater use of olanzapine (an SGA with a worse metabolic profile than comparatively newer agents such as aripiprazole)30 in Black vs White patients,3 along with a persistent difference in race-based prescribing of FGAs even after accounting for differences in metabolic factors.3
In the current analysis, efficacy outcomes with AOM 400 in Black/African American patients were comparable to those in patients of other races; across all racial groups, patients remained stable after prior aripiprazole stabilization—indicated by small or negligible changes in YMRS and MADRS total scores. QoL and functioning improved in Black/African American patients to a similar or greater degree than in patients from other racial groups. Of note, the 5.97-point improvement in FAST total score in Black/African American patients approached the estimated minimal clinically important difference (MCID) of 8 points31 and was significantly correlated with improvements in mood scores, illustrating a link between mood regulation and better functioning. Regarding the Brief QoL.BD, an MCID or responder threshold score is yet to be established.32
While data from the current analysis indicate that AOM 400 maintains stability and functional improvement and is not associated with specific safety concerns in Black/African American individuals, racial disparities in mental health care may be a barrier to its use. Such disparities are attributable to implicit bias, and to historical and existing structural racism, in which societal systems and institutions, including healthcare, unfairly disadvantage non-White individuals.33 For example, Black/African American psychiatric patients are less likely than their White counterparts to receive adequate treatment, outpatient follow-up care, or psychotherapy,34–37 even after variously controlling for socioeconomic factors such as health insurance status and household income.34,36,37 Black/African American individuals with new mental health disturbances, including bipolar disorder, are also less likely than their White counterparts to receive care before the manifestation of psychosis, resulting in fewer opportunities for early diagnosis and intervention.37 In addition, as mentioned previously, Black/African American individuals with psychiatric and neurological conditions, including bipolar disorder, are more likely than their White counterparts to be treated with an FGA and/or are less likely to be treated with an SGA.1–3,38,39 Beyond concerns about metabolic side effects with SGAs,3,30 researchers have suggested that increased use of FGAs in Black/African American vs White patients, despite the greater risk of EPS with FGAs,3,40,41 may be driven by medication cost3,29,39; indeed, FGA prescriptions are particularly elevated in Black/African American patients living in higher-poverty neighborhoods.3 Insurance coverage is also proposed as a contributing factor,29 with significantly lower SGA use observed in Black/African American vs White patients with Medicare, no insurance, or uncompensated care.38 Provider type may also be relevant; Black/African American individuals with mental health concerns are more likely to be treated by a primary care physician than a mental health specialist,42 and primary care physicians may be slower to prescribe newer antipsychotics.43 Additional contributors to greater FGA use in Black/African American patients may include perceptions of lower adherence to SGAs and assumptions about reduced SGA efficacy in this population.44 Beyond racial disparities, barriers to the use of AOM 400 in Black/African American patients may reflect those seen more broadly in a BP-I setting, especially in early disease. These include a lack of awareness of the benefits of LAIs for treating BP-I, the limited presence of LAIs in treatment guidelines, issues with healthcare reimbursement, and a lack of additional resources and infrastructure to support LAI use.45
An additional barrier to the use of AOM 400 for the treatment of bipolar disorder may be medical mistrust on the part of Black/African American individuals, which can include skepticism of or hesitancy in using modern medicines, including COVID-19 vaccines.46,47 Regarding psychiatric medications, data indicate that, compared with White individuals, those of Black/African American race have higher levels of mistrust, reducing their willingness to use such drugs.48 This highlights the need for healthcare environments that address the needs of Black/African American patients, including strengthening provider–patient relationships, promoting shared decision-making, reducing mental health stigma, ensuring culturally competent care, and increasing Black/African American representation in clinical trials. Additionally, based on the results of the current analysis, clinicians should assess patients individually and consider newer SGA medications, like AOM 400, wherever possible, in line with current treatment guidelines that recommend the use of SGAs for the first-and second-line treatment of bipolar disorder.49 This approach is supported by a recent expert consensus statement advocating early treatment with LAIs in patients with BP-I, within a shared decision-making framework involving clinicians, patients, and their supporters (eg, family).45 This is based on evidence for improved outcomes with LAI vs oral antipsychotics in randomized controlled trials (RCTs), cohort studies, and mirror-image studies, including a reduction in psychiatric and all-cause hospitalizations and visits to the emergency room,50,51 a reduced amount of time spent in hospital,50 and a reduction in relapse, notably in patients with rapid cycling or with a high frequency of relapse.50,52 Such outcomes, however, have not been universally observed, with an older meta-analysis of 4 RCTs showing no benefit of an SGA LAI over oral therapy or treatment as usual in terms of study-defined relapse.53
It is noteworthy that the impact of drug use on overall discontinuation rates was more pronounced in Black/African American patients vs patients of other races. The reasons for this are not known but may be driven by social determinants of health, including racial/ethnic segregation and discrimination, exposure to community violence, neighborhood instability, and negative peer influences, all of which have been shown to increase the risk of substance use in Black/African American individuals.54 The findings may also reflect recruitment bias, as Black/African American patients were enrolled almost exclusively from sites in North America where rates of illicit drug use are higher than in many other regions.55 In everyday practice, the presence of comorbid substance use disorder presents a clinical complexity that may make LAIs more suitable than daily oral treatment, due to their potential to enhance treatment adherence.56 This may extend to other complex BP-I presentations, including those with comorbid obsessive-compulsive disorder, in which the LAIs AOM 400 and paliperidone palmitate have demonstrated efficacy and tolerability.57
This exploratory post hoc analysis has limitations, including that it was not fully powered for some outcome measures and subgroups. Further, inferential statistics were exploratory and nominal with no adjustment for multiple comparisons. As such, the findings are hypothesis-generating and require confirmation in adequately powered, prospective studies. Because patients were stabilized on aripiprazole before the extension study, efficacy changes were expected to be minimal; this prior stabilization likely enriched the population with aripiprazole responders and tolerators, limiting the generalizability of the efficacy and safety findings. For rollover patients, per a protocol amendment, the open-label phase was reduced from 52 to 28 weeks (excluding patients at Japanese sites) due to sufficient safety data from the lead-in and other trials; this explains the lower patient numbers and changes in some efficacy outcomes near week 28 of the study. This protocol amendment may have altered the study population during the open-label phase, although only a small portion were rollover patients (85/464).11 Because most Black/African American participants were enrolled in North America and most Asian participants in Asia, the results may be confounded by regional and healthcare system factors; the inclusion of race as a model covariate may reduce but not eliminate residual confounding. Finally, small sample sizes for some metabolic parameters, particularly HbA1c, which was available for only 6.9% of patients, precluded an analysis according to race, limiting any meaningful conclusion on the relative metabolic effects of AOM 400 in Black/African American patients vs those of other races. Strengths of the analysis include that data were derived from a relatively large sample of patients, including more than 100 of Black/African American race.
In conclusion, the findings, from what is believed to be the first clinical trial subpopulation analysis of an SGA LAI in Black/African American patients diagnosed with BP-I, support the use of maintenance treatment with AOM 400 in this population. These findings reduce the evidence gap for SGA LAIs in Black/African American individuals living with BP-I and may help to shape data-informed treatment strategies. Given the similar pharmacokinetic profiles of AOM 400 and the newer Ari 2MRTU 960,58 it is expected that the safety and efficacy of Ari 2MRTU 960 will be similar to that of AOM 400 in Black/African American patients.
Article Information
Published Online: March 9, 2026. https://doi.org/10.4088/JCP.25m16114
© 2026 Physicians Postgraduate Press, Inc.
Submitted: August 26, 2025; accepted January 5, 2026.
To Cite: Bell Lynum KS, Such P, Zhang Z, et al. Safety and efficacy of maintenance treatment with aripiprazole once-monthly in Black/African American adults diagnosed with bipolar I disorder: post hoc analysis of a 52-week, open-label study.
J Clin Psychiatry 2026;87(2):25m16114.
Author Affiliations: Otsuka Pharmaceutical Development & Commercialization, Inc., Princeton, New Jersey (Bell Lynum, Zhang, Simmons-Yon, Atkins); H. Lundbeck A/S, Valby, Denmark (Such); Department of Psychiatry and Behavioral Sciences, George Washington University, Washington, DC (Lawson); Department of Psychiatry, Icahn School of Medicine at Mount Sinai, New York, New York (Goldberg).
Corresponding Author: Karimah S. Bell Lynum, PharmD, MBA, BCPP, Otsuka Pharmaceutical Development & Commercialization, Inc, 508 Carnegie Center, Princeton, NJ 08540 ([email protected]).
Author Contributions: All authors were involved in data collection and/or analysis. All authors participated in the drafting or the critical review of the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Relevant Financial Relationships: Drs Bell Lynum, Zhang, and Simmons-Yon are full-time employees of Otsuka Pharmaceutical Development & Commercialization, Inc. Dr Such is a full-time employee of H. Lundbeck A/S, Valby, Denmark. Dr Atkins is a consultant for Otsuka Pharmaceutical Development & Commercialization, Inc. Prof. Goldberg has received honoraria as a consultant or speakers bureau participant from AbbVie, Alkermes, Alvogen, Axsome, Bristol Meyers Squibb, Intracellular Therapies, Luye Pharmaceuticals, Otsuka, and Sunovion and has received royalties from American Psychiatric Publishing and Cambridge University Press. Prof. Lawson has no relevant disclosures to declare.
Funding/Support: The work described here was sponsored by Otsuka Pharmaceutical Development & Commercialization, Inc. and Lundbeck LLC.
Role of the Funders/Sponsors: Otsuka Pharmaceutical Development & Commercialization Inc. and Lundbeck LLC were involved in the design of the analysis; the collection, analysis, and interpretation of data; the writing and reviewing of this article; and the decision to submit the article for publication.
Previous Presentation: Data included in this manuscript were previously presented at the Psych Congress, October 29–November 2, 2024, Boston, Massachusetts, and at the Neuroscience Education Institute Congress, November 7–10, 2024, Colorado Springs, Colorado.
Acknowledgments: Writing and editorial support was provided by Lyndal Staples, BSc, and colleagues of the Prime Group of Companies (Knutsford, UK), funded by Otsuka Pharmaceutical Development & Commercialization, Inc. and H. Lundbeck A/S.
ORCID: Karimah S. Bell Lynum: https://orcid.org/0000-0002-2045-9578;
Pedro Such: https://orcid.org/0000-0003-1905-1142;
Zhen Zhang: https://orcid.org/0000-0001-8448-5663;
Norman Atkins, Jr: https://orcid.org/0000-0002-0802-6032;
William Lawson: https://orcid.org/0000-0002-9324-7090;
Joseph F. Goldberg: https://orcid.org/0000-0002-7160-7286
Supplementary Material: Available at Psychiatrist.com.
Clinical Points
- This post hoc analysis examined safety, efficacy, functional, and quality-of-life (QoL) outcomes in Black/African American patients diagnosed with bipolar I disorder (BP-I) treated with the aripiprazole once-monthly 400 mg (AOM 400) long-acting injectable (LAI) for 52 weeks.
- Safety outcomes with AOM 400 in Black/African American patients were broadly similar to those observed in patients from other racial groups. AOM 400 was associated with maintenance of clinical stability in Black/African American patients and with improvements in functioning and QoL that were similar to or greater than those in other racial groups.
- These findings can inform discussions about the use of AOM 400 or the once-every-2-months formulation of aripiprazole in Black/African American patients diagnosed with BP-I, within a shared decision-making framework. They may also be used to address race-based barriers to second-generation LAI use in Black/African American patients diagnosed with BP-I and barriers to LAI use more broadly in a BP-I setting.
References (58)

- Medina C, Akinkunmi A, Bland N, et al. Differences in schizophrenia treatments by race and ethnicity-analysis of electronic health records. Schizophr (Heidelb). 2024;10(1):48. PubMed CrossRef
- Tchikrizov V, Ladner ME, Caples FV, et al. Health disparities in the treatment of bipolar disorder. Pers Med Psychiatry. 2023:37–38:100101.
- Cook TB, Reeves GM, Teufel J, et al. Persistence of racial disparities in prescription of first-generation antipsychotics in the USA. Pharmacoepidemiol Drug Saf. 2015;24(11):1197–1206. PubMed CrossRef
- Bell Lynum KS, Henderson DC, Wright HJ, et al. Treatment effect with paliperidone palmitate compared with oral antipsychotics in Black/African American Patients with schizophrenia and a history of criminal justice system involvement: a post hoc analysis of the PRIDE study. J Clin Psychiatry. 2021;82(2):20m13356. PubMed CrossRef
- Abilify Asimtufii (aripiprazole). Prescribing information. Otsuka America Pharmaceutical. Inc.; 2025.
- Abilify (aripiprazole). Prescribing information. Otsuka America Pharmaceutical. Inc.; 2025.
- Abilify Maintena (aripiprazole). Prescribing information. Otsuka America Pharmaceutical. Inc.; 2025.
- Calabrese JR, Sanchez R, Jin N, et al. Symptoms and functioning with aripiprazole once-monthly injection as maintenance treatment for bipolar I disorder. J Affect Disord. 2018;227:649–656. PubMed CrossRef
- Calabrese JR, Sanchez R, Jin N, et al. Efficacy and safety of aripiprazole once-monthly in the maintenance treatment of bipolar I disorder: a double-blind, placebo-controlled, 52-week randomized withdrawal study. J Clin Psychiatry. 2017;78(3):324–331. PubMed CrossRef
- Calabrese JR, Sanchez R, Jin N, et al. The safety and tolerability of aripiprazole once-monthly as maintenance treatment for bipolar I disorder: a double-blind, placebo-controlled, randomized withdrawal study. J Affect Disord. 2018;241:425–432. PubMed CrossRef
- Calabrese JR, Jin N, Johnson B, et al. Aripiprazole once-monthly as maintenance treatment for bipolar I disorder: a 52-week, multicenter, open-label study. Int J Bipolar Disord. 2018;6(1):14. PubMed CrossRef
- Akinhanmi MO, Biernacka JM, Strakowski SM, et al. Racial disparities in bipolar disorder treatment and research: a call to action. Bipolar Disord. 2018;20(6):506–514. PubMed CrossRef
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. American Psychiatric Publishing Inc; 2000.
- Sheehan DV, Lecrubier Y, Sheehan KH, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59(suppl 20):22–33.
- Barnes TR. A rating scale for drug-induced akathisia. Br J Psychiatry. 1989;154:672–676. PubMed CrossRef
- Guy W. ECDEU Assessment Manual for Psychopharmacology: Revised. US Department of Health, Education and Welfare, Public Health Service, Alcohol, Drug Abuse and Mental Health Administration, NIMH Psychopharmacology Research Branch. Division of Extramural Research Programs; 1976.
- Simpson GM, Angus JW. A rating scale for extrapyramidal side effects. Acta Psychiatr Scand Suppl. 1970;212:11–19. PubMed CrossRef
- Young RC, Biggs JT, Ziegler VE, et al. A rating scale for mania: reliability, validity and sensitivity. Br J Psychiatry. 1978;133:429–435. PubMed CrossRef
- Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134:382–389. PubMed CrossRef
- Spearing MK, Post RM, Leverich GS, et al. Modification of the Clinical Global Impressions (CGI) Scale for use in bipolar illness (BP): the CGI-BP. Psychiatry Res. 1997;73(3):159–171. PubMed CrossRef
- Michalak EE, Murray G, Collaborative RESearch Team to Study Psychosocial Issues in Bipolar Disorder CREST.BD. Development of the QoL.BD: a disorder-specific scale to assess quality of life in bipolar disorder. Bipolar Disord. 2010;12(7):727–740. PubMed CrossRef
- Rosa AR, Sánchez-Moreno J, Martínez-Aran A, et al. Validity and reliability of the Functioning Assessment Short Test (FAST) in bipolar disorder. Clin Pract Epidemiol Ment Health. 2007;3:5. PubMed CrossRef
- Bell Lynum KS, Castro CF, Zhang Z, et al. Aripiprazole once-monthly for the treatment of adult patients with earlier-stage bipolar I disorder: a post hoc analysis of data from a double-blind, placebo-controlled, 52-week randomized withdrawal trial. Int J Bipolar Disord. 2024;12(1):37. PubMed CrossRef
- Akinyemiju T, Moore JX, Judd S, et al. Metabolic dysregulation and cancer mortality in a national cohort of Blacks and Whites. BMC Cancer. 2017;17(1):856. PubMed CrossRef
- Gaedigk A, Sangkuhl K, Whirl-Carrillo M, et al. Prediction of CYP2D6 phenotype from genotype across world populations. Genet Med. 2017;19(1):69–76. PubMed CrossRef
- Raygor V, Abbasi F, Lazzeroni LC, et al. Impact of race/ethnicity on insulin resistance and hypertriglyceridaemia. Diab Vasc Dis Res. 2019;16(2):153–159. PubMed CrossRef
- Jerome RN, Pulley JM, Sathe NA, et al. Exploring biologic predictors response disparities to atypical antipsychotics among blacks: a quasi-systematic review. Ethn Dis. 2020;30(suppl 1):229–240. PubMed CrossRef
- Cabassa LJ, Blanco C, Lopez-Castroman J, et al. Racial and ethnic differences in diabetes mellitus among people with and without psychiatric disorders: results from the National Epidemiologic Survey on Alcohol and Related Conditions. Gen Hosp Psychiatry. 2011;33(2):107–115. PubMed CrossRef
- Puyat JH, Daw JR, Cunningham CM, et al. Racial and ethnic disparities in the use of antipsychotic medication: a systematic review and meta-analysis. Soc Psychiatry Psychiatr Epidemiol. 2013;48(12):1861–1872. PubMed CrossRef
- Pillinger T, McCutcheon RA, Vano L, et al. Comparative effects of 18 antipsychotics on metabolic function in patients with schizophrenia, predictors of metabolic dysregulation, and association with psychopathology: a systematic review and network meta-analysis. Lancet Psychiatry. 2020;7(1):64–77. PubMed CrossRef
- Roux P, Brunet-Gouet E, Ehrminger M, et al. Minimum clinically important differences for the Functioning Assessment Short Test and a battery of neuropsychological tests in bipolar disorders: results from the FACE-BD cohort. Epidemiol Psychiatr Sci. 2020;29:e144. PubMed CrossRef
- CREST BD. QoL.BD – Frequently Asked Questions. https://www.crestbd.ca/wp-content/uploads/2016/02/QoLBD_FAQ.pdf. Accessed November 24, 2025.
- Ruiz-White I, Kramer L, Philips L, et al. Racial and ethnic disparities in physical and mental health care and clinical trials. J Clin Psychiatry. 2023;84(4):23ah14887. PubMed CrossRef
- Gonzalez JM, Thompson P, Escamilla M, et al. Treatment characteristics and illness burden among European Americans, African Americans, and Latinos in the first 2,000 patients of the Systematic Treatment Enhancement Program for Bipolar Disorder. Psychopharmacol Bull. 2007;40(1):31–46. PubMed
- Johnson KR, Johnson SL. Inadequate treatment of Black Americans with bipolar disorder. Psychiatr Serv. 2014;65(2):255–258. PubMed CrossRef
- Marcus SC, Chuang CC, Ng-Mak DS, et al. Outpatient follow-up care and risk of hospital readmission in schizophrenia and bipolar disorder. Psychiatr Serv. 2017;68(12):1239–1246. PubMed CrossRef
- Heun-Johnson H, Menchine M, Axeen S, et al. Association between race/ ethnicity and disparities in health care use before first-episode psychosis among privately insured young patients. JAMA Psychiatry. 2021;78(3):311–319. PubMed CrossRef
- Herbeck DM, West JC, Ruditis I, et al. Variations in use of second-generation antipsychotic medication by race among adult psychiatric patients. Psychiatr Serv. 2004;55(6):677–684. PubMed CrossRef
- Lawson W, Johnston S, Karson C, et al. Racial differences in antipsychotic use: claims database analysis of Medicaid-insured patients with schizophrenia. Ann Clin Psychiatry. 2015;27(4):242–252. PubMed CrossRef
- Scherk H, Pajonk FG, Leucht S. Second-generation antipsychotic agents in the treatment of acute mania: a systematic review and meta-analysis of randomized controlled trials. Arch Gen Psychiatry. 2007;64(4):442–455. PubMed CrossRef
- Tohen M, Goldberg JF, Gonzalez-Pinto Arrillaga AM, et al. A 12-week, double-blind comparison of olanzapine vs haloperidol in the treatment of acute mania. Arch Gen Psychiatry. 2003;60(12):1218–1226. PubMed
- Pingitore D, Snowden L, Sansone RA, et al. Persons with depressive symptoms and the treatments they receive: a comparison of primary care physicians and psychiatrists. Int J Psychiatry Med. 2001;31(1):41–60. PubMed CrossRef
- Morrens M, Destoop M, Cleymans S, et al. Evolution of first-generation and second-generation antipsychotic prescribing patterns in Belgium between 1997 and 2012: a population-based study. J Psychiatr Pract. 2015;21(4):248–258. PubMed CrossRef
- Kelly DL, Kreyenbuhl J, Dixon L, et al. Clozapine underutilization and discontinuation in African Americans due to leucopenia. Schizophr Bull. 2007;33(5):1221–1224. PubMed CrossRef
- Vieta E, Tohen M, McIntosh D, et al. Early use of long-acting injectable antipsychotics in bipolar disorder type I: an expert consensus. Bipolar Disord. 2025;27(1):7–16. PubMed CrossRef
- Cueva KL, Marshall AR, Snyder CR, et al. Medical mistrust among Black patients with serious illness: a mixed methods study. J Gen Intern Med. 2024;39(14):2747–2754. PubMed CrossRef
- Bogart LM, Ojikutu BO, Tyagi K, et al. COVID-19 related medical mistrust, health impacts, and potential vaccine hesitancy among Black Americans living with HIV. J Acquir Immune Defic Syndr. 2021;86(2):200–207. PubMed CrossRef
- André Christie-Mizell C, Blount SA, Laster Pirtle WN, et al. Psychiatric medication, African Americans and the paradox of mistrust. J Natl Med Assoc. 2015;107(2):51–59.
- Keramatian K, Chithra NK, Yatham LN. The CANMAT and ISBD guidelines for the treatment of bipolar disorder: summary and a 2023 update of evidence. Focus (Am Psychiatr Publ). 2023;21(4):344–353. PubMed CrossRef
- Bartoli F, Cavaleri D, Nasti C, et al. Long-acting injectable antipsychotics for the treatment of bipolar disorder: evidence from mirror-image studies. Ther Adv Psychopharmacol. 2023;13:20451253231163682. PubMed CrossRef
- Lähteenvuo M, Tanskanen A, Taipale H, et al. Real-world effectiveness of pharmacologic treatments for the prevention of rehospitalization in a Finnish nationwide cohort of patients with bipolar disorder. JAMA Psychiatry. 2018;75(4):347–355. PubMed CrossRef
- Kishi T, Oya K, Iwata N. Long-acting injectable antipsychotics for prevention of relapse in bipolar disorder: a systematic review and meta-analyses of randomized controlled trials. Int J Neuropsychopharmacol. 2016;19(9):pyw038. PubMed CrossRef
- Prajapati AR, Wilson J, Song F, et al. Second-generation antipsychotic long-acting injections in bipolar disorder: systematic review and meta-analysis. Bipolar Disord. 2018;20(8):687–696. PubMed CrossRef
- Lin C, Cousins SJ, Zhu Y, et al. A scoping review of social determinants of health’s impact on substance use disorders over the life course. J Subst Use Addict Treat. 2024;166:209484. PubMed CrossRef
- Peacock A, Leung J, Larney S, et al. Global statistics on alcohol, tobacco and illicit drug use: 2017 status report. Addiction. 2018;113(10):1905–1926. PubMed CrossRef
- Goldberg JF, Achtyes ED, Sajatovic M, et al. Clinical application of aripiprazole monohydrate long-acting injectables for the treatment of bipolar type I disorder: a consensus panel report. J Clin Psychiatry. 2025;86(3):plunlai2424ah3. PubMed CrossRef
- Martiadis V, Pessina E, Raffone F, et al. Efficacy and safety of adjunctive aripiprazole LAI or paliperidone LAI for the management of patients suffering from bipolar I disorder with comorbid obsessive-compulsive disorder. J Clin Med. 2025;14(3):954. PubMed CrossRef
- Harlin M, Yildirim M, Such P, et al. A randomized, open-label, multiple-dose, parallel-arm, pivotal study to evaluate the safety, tolerability, and pharmacokinetics of aripiprazole 2-month long-acting injectable in adults with schizophrenia or bipolar I disorder. CNS Drugs. 2023;37(4):337–350. PubMed CrossRef
This PDF is free for all visitors!