This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.
Background: Public health considerations require that clinical trials address the complex "real-world" needs of patients with chronic illnesses. This is particularly true for persons with schizophrenia, whose management is frequently complicated by factors such as comorbid substance abuse, homelessness, and contact with the criminal justice system. In addition, barriers to obtaining health care in the United States often prevent successful community reentry and optimal patient management. Further, nonadherence to treatment is common, and this reinforces cycles of relapse and recidivism.
Long-acting injectable antipsychotic therapy may facilitate continuity of treatment and support better outcomes, particularly in patients who face these challenges. Clinical trials with classical explanatory designs may not be the best approaches for evaluating these considerations. We describe the design and rationale of a novel trial that combines both explanatory and pragmatic design features and studies persons with schizophrenia who face these challenges.
Design and Rationale: The Paliperidone Palmitate Research in Demonstrating Effectiveness (PRIDE) study is a prospective, open-label, randomized, 15-month study conducted between May 5, 2010, and December 9, 2013, comparing long-acting injectable paliperidone palmitate and oral antipsychotic medications in subjects with schizophrenia (according to DSM-IV criteria). Investigators and subjects had broad flexibility for treatment decision-making, thus making it a model that better reflects real-world practice. The primary end point was time to treatment failure, defined as arrest/incarceration psychiatric hospitalization; suicide; treatment discontinuation or supplementation due to inadequate efficacy, safety, or tolerability; or increased psychiatric services to prevent hospitalization. This end point was adjudicated by a blinded event monitoring board. Patients were followed to the 15-month end point, regardless of whether they were maintained on their initial randomized treatment. This article provides some of the reasoning behind the authors’ choices when combining features from both explanatory and pragmatic approaches to this trial’s design.
Conclusions: The PRIDE study incorporates real-world design features in a novel, prospective, comparative study of long-acting injectable and oral antipsychotics in persons with schizophrenia who have had recent contact with the criminal justice system. Insights provided should help the reader to better understand the need for more real-world approaches for clinical studies and how a broader approach can better aid clinical treatment and public health decision-making.
Trial Registration: ClinicalTrials.gov identifier: NCT01157351
Design and Rationale of the Paliperidone Palmitate Research in Demonstrating Effectiveness (PRIDE) Study: A Novel Comparative Trial of Once-Monthly Paliperidone Palmitate Versus Daily Oral Antipsychotic Treatment for Delaying Time to Treatment Failure in Persons With Schizophrenia

ABSTRACT
Background: Public health considerations require that clinical trials address the complex "real-world" needs of patients with chronic illnesses. This is particularly true for persons with schizophrenia, whose management is frequently complicated by factors such as comorbid substance abuse, homelessness, and contact with the criminal justice system. In addition, barriers to obtaining health care in the United States often prevent successful community reentry and optimal patient management. Further, nonadherence to treatment is common, and this reinforces cycles of relapse and recidivism.
Long-acting injectable antipsychotic therapy may facilitate continuity of treatment and support better outcomes, particularly in patients who face these challenges. Clinical trials with classical explanatory designs may not be the best approaches for evaluating these considerations. We describe the design and rationale of a novel trial that combines both explanatory and pragmatic design features and studies persons with schizophrenia who face these challenges.
Design and Rationale: The Paliperidone Palmitate Research in Demonstrating Effectiveness (PRIDE) study is a prospective, open-label, randomized, 15-month study conducted between May 5, 2010, and December 9, 2013, comparing long-acting injectable paliperidone palmitate and oral antipsychotic medications in subjects with schizophrenia (according to DSM-IV criteria). Investigators and subjects had broad flexibility for treatment decision-making, thus making it a model that better reflects real-world practice. The primary end point was time to treatment failure, defined as arrest/incarceration psychiatric hospitalization; suicide; treatment discontinuation or supplementation due to inadequate efficacy, safety, or tolerability; or increased psychiatric services to prevent hospitalization. This end point was adjudicated by a blinded event monitoring board. Patients were followed to the 15-month end point, regardless of whether they were maintained on their initial randomized treatment. This article provides some of the reasoning behind the authors’ choices when combining features from both explanatory and pragmatic approaches to this trial’s design.
Conclusions: The PRIDE study incorporates real-world design features in a novel, prospective, comparative study of long-acting injectable and oral antipsychotics in persons with schizophrenia who have had recent contact with the criminal justice system. Insights provided should help the reader to better understand the need for more real-world approaches for clinical studies and how a broader approach can better aid clinical treatment and public health decision-making.
Trial Registration: ClinicalTrials.gov identifier: NCT01157351
J Clin Psychiatry
© Copyright 2014 Physicians Postgraduate Press, Inc.
Submitted: December 20, 2013; accepted May 28, 2014.
Online ahead of print: October 28, 2014 (doi:10.4088/JCP.13m08965).
Corresponding author: Larry Alphs, MD, PhD, Therapeutic Team Leader Psychiatry, Janssen Scientific Affairs, LLC, 1125 Trenton-Harbourton Rd-A32404, Titusville, NJ 08560 ([email protected]).
To help patients with schizophrenia lead more productive lives and better fulfill their potential, their complex "real-world" needs must be better understood. Clinical trials that study these needs must inform an ever-widening group of stakeholders, including patients, clinicians, regulatory authorities, and health care payers. To address these diverse expectations, trials with real-world (pragmatic) designs are necessary that build upon foundational studies establishing treatment safety and efficacy (explanatory trials). Pragmatic trials require attention to distinctive methodological considerations. This article focuses on design considerations as they relate to a comparative study of antipsychotics in schizophrenia.
Trials designed for regulatory approval typically focus on explanatory considerations; that is, they concentrate on whether the particular drug under study is safe and effective. Such trials require careful selection of a study population that is otherwise medically healthy and is likely to be adherent to treatment.1,2 Teams conducting these studies must be highly trained to provide reliable safety and efficacy information. To adequately capture the required information, assessment measures are often highly specialized and are applied more frequently than related assessments used in standard clinical practice.2 Considerable attention is also given to reinforcing treatment adherence so as to enhance the likelihood that results can be attributed to the study drug.2
Such explanatory trials often leave many questions related to clinical practice unanswered. Pragmatic or real-world trials seek to address these questions. To do this, they require a different approach. Exclusionary criteria should be limited, so that study subjects better represent the diverse population expected to be exposed to the treatment.1 Treatment providers should fully reflect the range of clinicians and staff with skill sets customarily available when the treatment is delivered. Comparative interventions should include the range of options that are available to the treating clinician.2 Outcome assessments should be unambiguous measures of response that are meaningful to both the patient and clinician.2 In practice, few trials are purely explanatory or pragmatic, and many prospective clinical trials include a range of pragmatic and explanatory features.2 These considerations were used to design a study comparing daily oral and long-acting injectable treatments for schizophrenia.
RATIONALE
Symptoms of schizophrenia can be treated effectively with antipsychotic medication; however, poor adherence to prescribed treatment is one of the biggest challenges of managing the symptoms of schizophrenia and delaying time to relapse.3,4 Long-acting injectable antipsychotics deliver therapeutic concentrations over several weeks, eliminating the need for daily dosing5 and providing clinicians with certain knowledge of adherence or nonadherence. As a result, these agents increase the likelihood of continuous and effective treatment and may reduce patients’ risk for relapse. This, in turn, could decrease the likelihood of institutionalization in hospitals and incarceration. Studies comparing long-acting injectable versus oral antipsychotic treatment have provided inconsistent results,6-15 with some indication that demonstrating a differential effectiveness among these formulations is better established with a more pragmatic clinical trial design than with one that is more explanatory.16-19 With this in mind, we designed the Paliperidone Palmitate Research in Demonstrating Effectiveness (PRIDE) study (ClinicalTrials.gov identifier: NCT01157351) to compare once-monthly paliperidone palmitate and daily oral antipsychotics in real-world schizophrenia, as defined by subject inclusion criteria, treatment, and outcomes. It was hypothesized that paliperidone palmitate would be more effective than oral antipsychotics. We provide a description of that trial’s design and the reasoning behind it, addressing the selection of patients, outcome measures, and study end points.
STUDY OBJECTIVES
The primary objective of the PRIDE study was to compare the effectiveness of paliperidone palmitate treatment with daily oral antipsychotic treatment in delaying time to treatment failure (as defined by several real-world outcomes) over 15 months in subjects with schizophrenia.

- Paliperidone palmitate may provide benefits over oral antipsychotics, but explanatory-designed trials have not characterized the real-world outcomes resulting from use of these agents.
- Pragmatic trials, with their limited exclusionary criteria, wide range of treatment providers, broad treatment options, and naturalistic outcome assessments, are designed to better reflect real-world situations and may lead to more practical understanding of treatment decision choices.
- The Paliperidone Palmitate Research in Demonstrating Effectiveness (PRIDE) study combines both explanatory and pragmatic design elements to examine the relative effectiveness of paliperidone palmitate, a once-monthly antipsychotic, and daily oral antipsychotics in patients with schizophrenia and a history of recent criminal justice system contact.
Key secondary objectives were to compare paliperidone palmitate with oral antipsychotic treatment in (1) time to first psychiatric hospitalization or arrest/incarceration; (2) overall patient functioning, as measured by the Personal and Social Performance Scale20; (3) time to first psychiatric hospitalization; and (4) overall symptom improvement, as measured by the Clinical Global Impressions-Severity of Illness scale.21 An additional objective was to examine the safety and tolerability of paliperidone palmitate treatment compared with oral antipsychotic treatment.
STUDY DESIGN
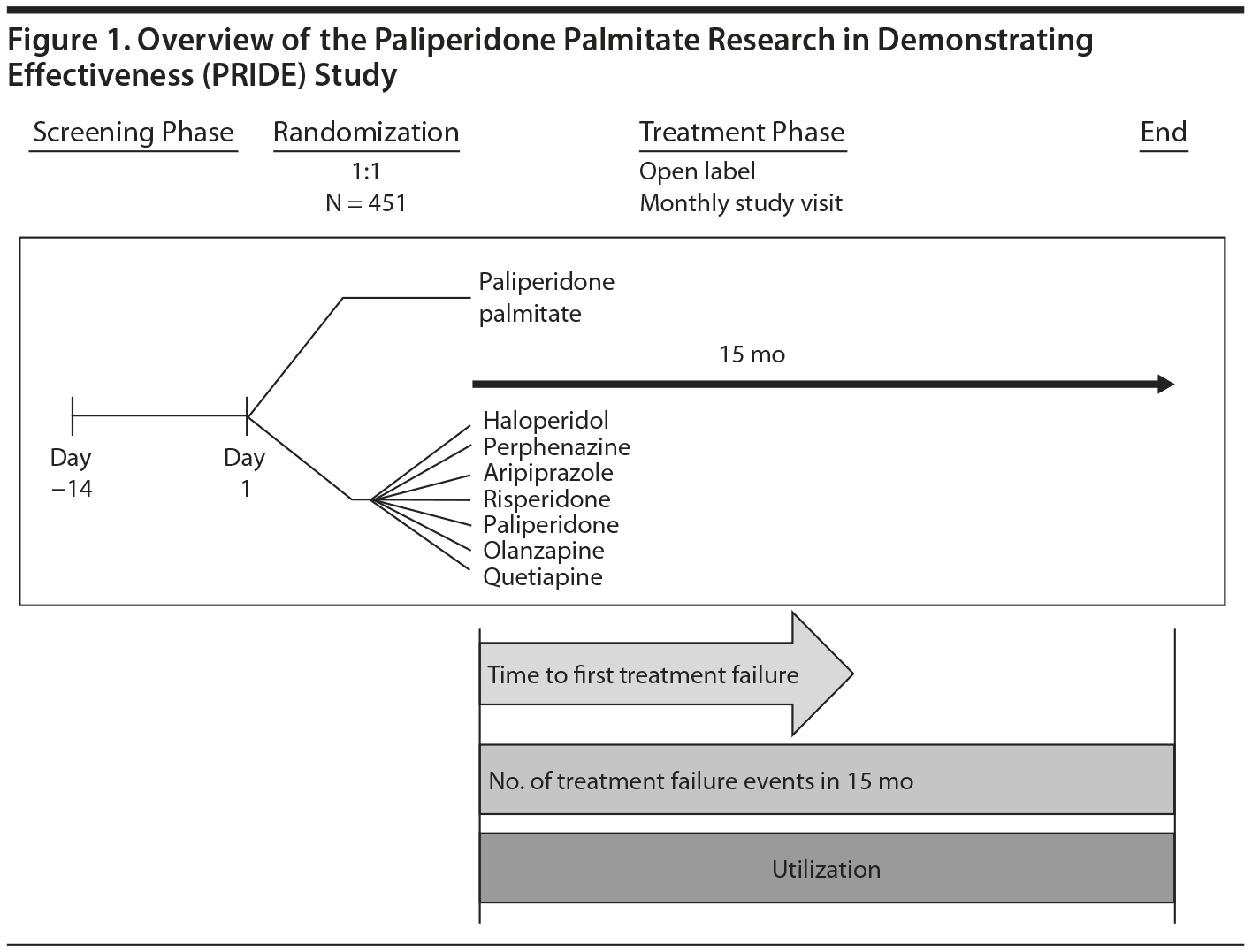
The PRIDE study was a randomized, prospective, open-label, active-controlled, parallel-group comparative efficacy and effectiveness study of paliperidone palmitate versus oral antipsychotic treatment in adults with schizophrenia. The study was conducted between May 5, 2010, and December 9, 2013. It consisted of a screening phase of up to 2 weeks, followed by a 15-month randomized, open-label treatment phase (Figure 1). An independent event monitoring board, blinded to individual subject treatment assignment, certified the occurrence and time of the first treatment failure (the primary end point) for each randomly assigned subject.
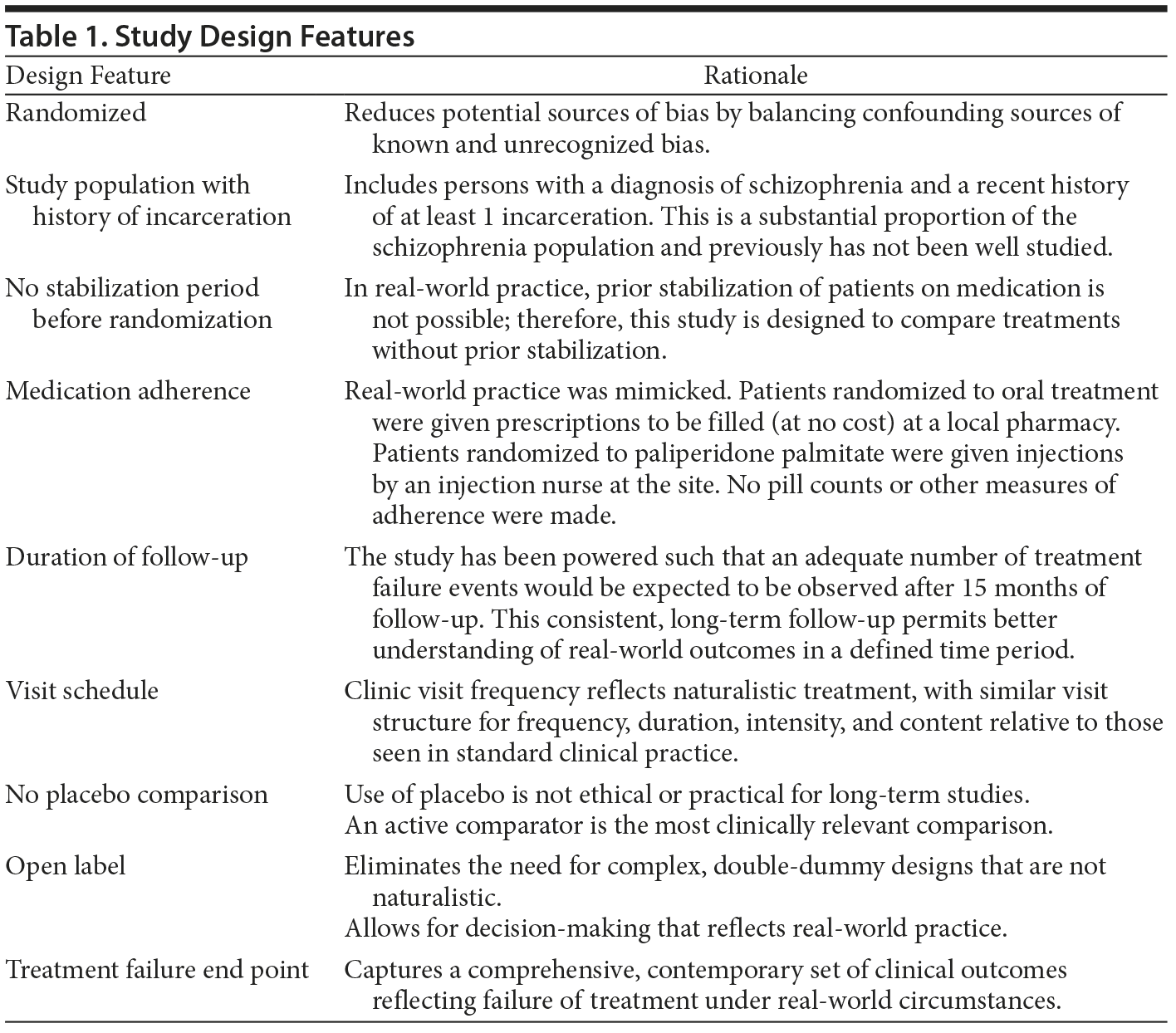
Incorporating both explanatory (efficacy) and pragmatic (effectiveness) design elements, the study was randomized and controlled but was conducted in the context of a naturalistic treatment setting rather than in a highly controlled clinical trial environment. This required balancing often competing considerations regarding pragmatic or explanatory choices for study design elements. The final design allowed information to be gathered on both efficacy and effectiveness (see Table 1 for detail) outcomes. Design domains that characterize a study along the pragmatic-explanatory continuum include patient selection criteria, site and investigator selection criteria, flexibility in dosing and use of concomitant medications, outcome selection, intensity of follow-up, and practices related to treatment adherence and patient retention.2,12 Study sites and investigators were selected on the basis of access to subjects who fit the entry criteria and ability to follow subjects in a clinical trial setting.
Reasoning behind the choices surrounding some of the more important aspects of the trial design is discussed in more detail below.
Study End Points
The PRIDE study included some end points that were more explanatory in nature and others that were more pragmatic. Because the trial was designed to meet standards for regulatory submission, it could not be purely pragmatic. Patients enrolled in the PRIDE study were encouraged to continue in the study to their predefined, 15-month completion date, even after a change from their initial, randomized treatment assignment because, as in real-life, events that occur after early discontinuation may be extremely relevant to the patient’s predefined outcome.
The primary end point and the key secondary end points examined treatment response only while subjects were taking their randomly assigned medication. This condition represented an explanatory approach and permits specific understanding of the relative safety and efficacy of the assigned treatments. The exploratory pragmatic end points examined treatment to the 15-month end point or the final recorded observation, regardless of whether subjects were maintained on the initial randomized treatment. This permitted understanding of the longer term consequences of the choice among treatments assigned at randomization.
The primary study end point, treatment failure, was developed as a pragmatic construct that is more relevant to the experience of patients with schizophrenia than other assessments. In particular, it incorporated arrest or incarceration as a component of treatment failure—an important pragmatic aspect of contemporary mental illness outcomes in the United States. Treatment failure was defined as 1 of the following: arrest/incarceration, psychiatric hospitalization, or suicide; or, as determined by the study physician, discontinuation of antipsychotic treatment due to inadequate efficacy, treatment supplementation with another antipsychotic due to inadequate efficacy, discontinuation of antipsychotic treatment due to safety or tolerability concerns, or an increase in the level of psychiatric services to prevent imminent psychiatric hospitalization.
Precise definitions were developed for all major elements of the treatment failure end point. An arrest was defined as the taking of a subject into custody by a legal authority for any reason. The definition did not include times when a subject was stopped, questioned, or temporarily detained by a law enforcement officer or by a preplanned, probation-associated, or court-ordered contact with the criminal justice system.
Study Subjects
Because a more pragmatic approach was used to better reflect the broad range of patients found in regular clinical practice, few inclusionary or exclusionary entry criteria were applied beyond the primary intention to focus on persons with schizophrenia who had had recent contact with the criminal justice system. The few additional inclusion/exclusion criteria included were those required by the ethics of conducting a clinical trial and those required to capture specific efficacy and safety data (some of which are required by regulatory authorities) in an effort to understand the relative response to comparator treatments.
Selection criteria for investigators included their knowledge and connections with their local criminal justice system. Field-based medical staff from the sponsor worked with investigators to further develop an understanding of their local criminal justice system landscape.
Upon study start, it was found that traditional clinical trial recruitment efforts were ineffective. As a result, a recruitment outreach strategy was developed focusing on streets, homeless shelters, and single-residence units. Additional alternative stakeholders, such as law enforcement, case managers, and behavioral health departments, were included in the outreach process.
It was planned that approximately 442 male and female subjects between 18 and 65 years of age with schizophrenia (diagnosed according to Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition criteria,22 confirmed by using the Mini-International Neuropsychiatric Interview, version 6.023) who had been taken into custody at least twice in the previous 2 years would be enrolled in this study. At least 1 instance of custody must have led to an incarceration, and the most recent release from custody must have occurred within 90 days of the screening visit.
Subjects were excluded if they had been actively abusing intravenous drugs within the past 3 months or had an opiate dependence disorder. Otherwise, substance abuse was not an exclusionary factor. This approach permitted a compromise between being highly inclusive and ensuring the subjects would actually be available later for data collection. It also allowed many subjects to be enrolled in the PRIDE study who would have been excluded from more traditional explanatory studies.
Treatment
For subjects taking more than 1 oral antipsychotic at screening, 1 was chosen by the investigator as the primary oral antipsychotic. Any additional antipsychotics were tapered and discontinued during the first 3 days of screening so that all subjects received only 1 oral antipsychotic on the day before randomization.
Before randomization, the treating clinician and the subject reviewed the 7 oral antipsychotics available for use in this study (aripiprazole, haloperidol, olanzapine, paliperidone, perphenazine, quetiapine, and risperidone) to determine their acceptability on the basis of prior experience with these medications. One or more (up to 6) could be deselected. Thereafter, subjects with the same set of prespecified oral antipsychotics were placed within similar randomization strata. At visit 1, subjects were randomly assigned within their stratum in a 1:1 ratio to paliperidone palmitate or oral antipsychotic treatment. If the subject was randomly assigned to receive oral antipsychotic treatment, the specific oral medication was randomly selected from the prespecified oral antipsychotics. This randomization approach was chosen over a simpler clinician choice design to reduce treatment selection bias among the oral antipsychotic treatments available.24
In general, dosing followed label instructions for the assigned treatments, but monotherapy was required between day 8 and day 15 so as to ensure that this approach was tried. Although supplemental oral antipsychotic treatment was allowed on or after day 15, the investigator was asked to first consider increasing the randomly assigned study drug dosage or adding adjunctive nonantipsychotic psychotropic therapy (ie, anxiolytics, antidepressants, or mood stabilizers) to manage worsening symptoms. This strategy encouraged use of optimal monotherapy for the primary assigned treatment.
Subjects in the paliperidone palmitate arm who were not taking 234 mg of paliperidone palmitate could receive supplemental oral paliperidone (if the investigator deemed a higher dose to be necessary) until the dose of paliperidone palmitate could be increased at the next injection day without declaration of a treatment failure. The questions addressed by this study related to relative comparisons of paliperidone palmitate and oral antipsychotic medications and hypothesized superiority for paliperidone palmitate. Therefore, subjects were allowed to switch from once-monthly paliperidone palmitate to oral antipsychotics, as it would have allowed the study hypotheses to be evaluated for both the explanatory and pragmatic outcomes. However, subjects were not allowed to switch from a daily oral antipsychotic to once-monthly paliperidone palmitate and still remain in the study because, given the study hypothesis that paliperidone palmitate was superior to oral antipsychotic, this would not have allowed the pragmatic question regarding relative superiority of paliperidone palmitate to oral antipsychotic to be fully addressed.
Clinical Assessments and Evaluations
During the screening period, psychiatric/medical histories were obtained, diagnostic criteria for schizophrenia ascertained, safety screening procedures performed, and other eligibility criteria evaluated. Throughout the 15-month treatment period, visits occurred on a similar schedule for both treatment arms on a monthly basis. Subjects were assessed at each study visit for occurrence of treatment failure.
To the extent possible, all subjects were followed for 15 months if they consented to ongoing participation. Subjects who experienced treatment failure had the option of continuing to take their randomly assigned treatment or changing to a new oral treatment if they did not find the randomized medication tolerable or adequately effective. Such subjects returned for regularly scheduled visits and underwent all assessments, including assessments for treatment failure. If a subject discontinued drug treatment, end-of-treatment procedures were completed as soon as possible thereafter. Subjects who left the study before reaching their 15-month end point were allowed to reenter the study any number of times until 15 months from randomization had elapsed.
Statistical Analysis
Sample size determination was based on testing the primary null explanatory hypothesis that there is no difference between paliperidone palmitate and oral antipsychotic treatment in distribution of time to first treatment failure. Assuming time to first treatment failure follows an exponential distribution, the primary null and alternative hypothesis could be expressed in terms of the hazard ratio of the 2 treatment groups. For detecting treatment differences measured by a hazard ratio of 0.516 with 80% power, at a 2-sided .05 significance level, using an exponential maximum likelihood test of equality of survival curves, at least 72 first-treatment failures would have been needed. If it is assumed that the maximum follow-up time is 15 months and 30% of the randomly assigned subjects would drop out by 15 months before experiencing a treatment failure event (ie, common exponential dropout rate of 0.0238), a total of 442 subjects were required (221 per group). The hazard ratio of 0.516 corresponds to event rate differences ranging from 10% to 20%. This difference was judged to be clinically relevant.
For this study, the intent-to-treat (ITT) population was defined as all randomly assigned subjects who received at least 1 dose of their assigned study treatment. The explanatory ITT (eITT) analysis set for the primary efficacy end point was defined as time to first treatment failure observed before the eITT end point (last injection date + 28 days, or last prescription date of the randomly assigned oral medication + number of days’ supply + 1 day) for all ITT subjects. First treatment failure times for subjects who did not experience any treatment failure before the eITT end point were censored at the eITT end point, and treatment differences were compared using a log rank test based on the eITT analysis set.
CONCLUSIONS
The PRIDE study is a 15-month, prospective, randomized, active-controlled, open-label comparative trial, conducted in a naturalistic, real-world setting. It is designed to compare once-monthly paliperidone palmitate with daily oral antipsychotic treatment in delaying time to treatment failure in adults with schizophrenia who have had recent contact with the criminal justice system. The primary end point for the PRIDE study is novel, as it encompasses criminal justice contact as a component of treatment failure. The design allows both explanatory and pragmatic questions to be addressed in the context of a single study. The explanatory end points should support prior findings with paliperidone palmitate in an important segment of the population of persons with schizophrenia relative to alternative oral treatments. The choice of time to treatment failure and the more pragmatic aspects of the trial permit a broad understanding of the effectiveness of paliperidone palmitate when used under standard practice conditions compared with a range of treatment alternatives. Indeed, the inclusion of time to arrest as part of the primary end point incorporates a measure that is relevant to the real-world experience of many persons with schizophrenia. This end point is usually neglected but has important implications for individuals, families, and broader public health considerations.
The findings from this study will permit a deeper understanding of differences between the 2 primary treatments studied (paliperidone palmitate and oral antipsychotics) and the longer term consequences of making treatment decisions, regardless of the resultant treatment conditions. This knowledge should help inform better treatment decisions and generate better public health policy.
Drug names: aripiprazole (Abilify), haloperidol (Haldol and others), olanzapine (Zyprexa and others), paliperidone (Invega), paliperidone palmitate (Invega Sustenna), quetiapine (Seroquel and others), risperidone (Risperdal and others).
Author affiliations: Scientific Affairs, Janssen Scientific Affairs, LLC (Drs Alphs and Starr and Mr Rodriguez), Titusville; Biostatistics and Reporting Department, Janssen R & D, LLC (Dr Mao), Titusville; and Global Medical Affairs, Janssen Global Services, LLC (Dr Hulihan), Raritan, New Jersey.
Potential conflicts of interest: Drs Alphs and Starr and Mr Rodriguez are employees of Janssen Scientific Affairs and Johnson & Johnson stockholders. Dr Mao is an employee of Janssen Research & Development and a Johnson & Johnson stockholder. Dr Hulihan is an employee of Janssen Global Services and is a Johnson & Johnson stockholder.
Funding/support: This research was funded by Janssen Scientific Affairs, LLC, Titusville, New Jersey (primary study protocol number R092670-SCH3006).
Previous presentations: 13th International Congress on Schizophrenia Research; April 2-6, 2011; Colorado Springs, Colorado ▪ American Society for Experimental NeuroTherapeutics 13th Annual Meeting; February 24-26, 2011; Bethesda, Maryland ▪ American College of Neuropsychopharmacology 49th Annual Meeting; December 5-9, 2010; Miami Beach, Florida ▪ International Society for CNS Clinical Trials and Methodology Autumn Conference; October 13-14, 2010; Baltimore, Maryland ▪ and National Conference on Correctional Health Care Annual Meeting; October 9-13, 2010; Las Vegas, Nevada.
Acknowledgments: The authors wish to acknowledge Reuven Ferziger, MD (employee of Janssen Scientific Affairs, LLC, at the time of the study); Ed Crumbley, BA (employee of Janssen Scientific Affairs, LLC, at the time of the study); and Norris Turner, PharmD, PhD (employee of Johnson & Johnson Health Care Systems), for their participation during the development of the study design. The authors also thank Cynthia Bossie, PhD (employee of Janssen Scientific Affairs, LLC), and Matthew Grzywacz, PhD, of ApotheCom, LLC, Yardley, Pennsylvania (funded by Janssen Scientific Affairs, LLC), for providing writing and editorial assistance. Drs Ferziger, Turner, and Bossie and Mr Crumbly are Johnson & Johnson stockholders.
REFERENCES
1. Roland M, Torgerson DJ. What are pragmatic trials? BMJ. 1998;316(7127):285. PubMed doi:10.1136/bmj.316.7127.285
2. Thorpe KE, Zwarenstein M, Oxman AD, et al. A pragmatic-explanatory continuum indicator summary (PRECIS): a tool to help trial designers. CMAJ. 2009;180(10):E47-E57. PubMed doi:10.1503/cmaj.090523
3. Morken G, Widen JH, Grawe RW. Non-adherence to antipsychotic medication, relapse and rehospitalisation in recent-onset schizophrenia. BMC Psychiatry. 2008;8(1):32. PubMed doi:10.1186/1471-244X-8-32
4. Kane JM. Improving treatment adherence in patients with schizophrenia. J Clin Psychiatry. 2011;72(9):e28. PubMed doi:10.4088/JCP.9101tx2c
5. Gopal S, Hough DW, Xu H, et al. Efficacy and safety of paliperidone palmitate in adult patients with acutely symptomatic schizophrenia: a randomized, double-blind, placebo-controlled, dose-response study. Int Clin Psychopharmacol. 2010;25(5):247-256. PubMed doi:10.1097/YIC.0b013e32833948fa
6. Chue P, Eerdekens M, Augustyns I, et al. Comparative efficacy and safety of long-acting risperidone and risperidone oral tablets. Eur Neuropsychopharmacol. 2005;15(1):111-117. PubMed doi:10.1016/j.euroneuro.2004.07.003
7. Bai YM, Chen TT, Wu B, et al. A comparative efficacy and safety study of long-acting risperidone injection and risperidone oral tablets among hospitalized patients: 12-week randomized, single-blind study. Pharmacopsychiatry. 2006;39(4):135-141. PubMed doi:10.1055/s-2006-946703
8. Emsley R, Oosthuizen P, Koen L, et al. Oral versus injectable antipsychotic treatment in early psychosis: post hoc comparison of two studies. Clin Ther. 2008;30(12):2378-2386. PubMed doi:10.1016/j.clinthera.2008.12.020
9. Weiden PJ, Schooler NR, Weedon JC, et al. A randomized controlled trial of long-acting injectable risperidone vs continuation on oral atypical antipsychotics for first-episode schizophrenia patients: initial adherence outcome. J Clin Psychiatry. 2009;70(10):1397-1406. PubMed doi:10.4088/JCP.09m05284yel
10. Macfadden W, Ma YW, Thomas Haskins J, et al. A prospective study comparing the long-term effectiveness of injectable risperidone long-acting therapy and oral aripiprazole in patients with schizophrenia. Psychiatry (Edgmont). 2010;7(11):23-31. PubMed
11. Gaebel W, Schreiner A, Bergmans P, et al. Relapse prevention in schizophrenia and schizoaffective disorder with risperidone long-acting injectable vs quetiapine: results of a long-term, open-label, randomized clinical trial. Neuropsychopharmacology. 2010;35(12):2367-2377. PubMed doi:10.1038/npp.2010.111
12. Rosenheck RA, Krystal JH, Lew R, et al; CSP 555 Research Group. Challenges in the design and conduct of controlled clinical effectiveness trials in schizophrenia. Clin Trials. 2011;8(2):196-204. PubMed doi:10.1177/1740774510392931
13. Schooler NR, Buckley PF, Mintz J, et al. PROACTIVE: initial results of an RCT comparing long-acting injectable risperidone to 2nd generation oral antipsychotics. Presented at the 50th Annual Meeting of the American College of Neuropsychopharmacology; December 4-8, 2011; Kona, Hawaii: S104-S105
14. Zhornitsky S, Stip E. Oral versus long-acting injectable antipsychotics in the treatment of schizophrenia and special populations at risk for treatment nonadherence: a systematic review. Schizophr Res Treatment. 2012;2012:407171. PubMed doi:10.1155/2012/407171
15. Barrio P, Batalla A, Castellv× P, et al. Effectiveness of long-acting injectable risperidone versus oral antipsychotics in the treatment of recent-onset schizophrenia: a case-control study. Int Clin Psychopharmacol. 2013;28(4):164-170. PubMed
16. Kirson NY, Weiden PJ, Yermakov S, et al. Efficacy and effectiveness of depot versus oral antipsychotics in schizophrenia: synthesizing results across different research designs. J Clin Psychiatry. 2013;74(6):568-575. PubMed doi:10.4088/JCP.12r08167
17. Kishimoto T, Nitta M, Borenstein M, et al. Long-acting injectable versus oral antipsychotics in schizophrenia: a systematic review and meta-analysis of mirror-image studies. J Clin Psychiatry. 2013;74(10):957-965. PubMed doi:10.4088/JCP.13r08440
18. Kishimoto T, Robenzadeh A, Leucht C, et al. Long-acting injectable vs oral antipsychotics for relapse prevention in schizophrenia: a meta-analysis of randomized trials. Schizophr Bull. 2014;40(1):192-213. PubMed
19. Fusar-Poli P, Kempton MJ, Rosenheck RA. Efficacy and safety of second-generation long-acting injections in schizophrenia: a meta-analysis of randomized-controlled trials. Int Clin Psychopharmacol. 2013;28(2):57-66. PubMed doi:10.1097/YIC.0b013e32835b091f
20. Morosini PL, Magliano L, Brambilla L, et al. Development, reliability and acceptability of a new version of the DSM-IV Social and Occupational Functioning Assessment Scale (SOFAS) to assess routine social functioning. Acta Psychiatr Scand. 2000;101(4):323-329. PubMed
21. Guy W. ECDEU Assessment Manual for Psychopharmacology. US Dept Health, Education, and Welfare publication (ADM) 76-338. Rockville, MA: National Institute of Mental Health;1976:218-222.
22. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition. Washington, DC: American Psychiatric Association; 1994.
23. Sheehan DV, Lecrubier Y, Sheehan KH, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59(suppl 20):22-33, quiz 34-57. PubMed
24. Lavori PW, Rush AJ, Wisniewski SR, et al. Strengthening clinical effectiveness trials: equipoise-stratified randomization. Biol Psychiatry. 2001;50(10):792-801. PubMed doi:10.1016/S0006-3223(01)01223-9
This PDF is free for all visitors!