Objective: To assess the effect of dose increase in adult patients with schizophrenia who demonstrate inadequate initial response to standard-dose lurasidone and to evaluate the efficacy of low-dose lurasidone in adult patients with schizophrenia.
Methods: In this randomized, double-blind, placebo-controlled study conducted between May 2013 and June 2014, hospitalized patients with acute schizophrenia (DSM-IV-TR criteria) were randomly assigned to double-blind treatment with lurasidone 20 mg/d (n = 101), lurasidone 80 mg/d (n = 199), or placebo (n = 112). Nonresponders to lurasidone 80 mg/d (Positive and Negative Syndrome Scale [PANSS] score decrease < 20%) at 2 weeks were re-randomized to lurasidone 80 mg/d or 160 mg/d for the remaining 4 weeks of the study. The primary outcome measure was change from baseline to week 6 in PANSS total score.
Results: In nonresponders to lurasidone 80 mg/d (n = 95), dose increase to 160 mg/d at week 2 significantly reduced PANSS total score at week 6 study endpoint compared with continuing 80 mg/d (-16.6 vs -8.9; P < .05 [effect size = 0.52]). While a comparable magnitude of improvement was observed in Clinical Global Impression-Severity (CGI-S) score from week 2 to week 6 endpoint for lurasidone 160 mg/d versus 80 mg/d (-1.0 vs -0.6; effect size = 0.44), the difference was not statistically significant (P = .052). Patients receiving lurasidone 20 mg/d did not demonstrate significant improvement compared with placebo at week 6 in PANSS total (-17.6 vs -14.5; P = .26) or CGI-S (-0.93 vs -0.73; P = .17) scores. Few dose-related adverse effects associated with lurasidone were observed.
Conclusions: In adult patients with schizophrenia demonstrating nonresponse to 2 weeks of treatment with lurasidone 80 mg/d, dose increase to 160 mg/d resulted in significant symptom improvement compared with continuing lurasidone 80 mg/d. Lurasidone 20 mg/d was not associated with significant improvement in psychotic symptoms in adult patients with schizophrenia.
Trial Registration: ClinicalTrials.gov identifier: NCT01821378
This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.
Lurasidone Dose Escalation in Early Nonresponding Patients With Schizophrenia:
A Randomized, Placebo-Controlled Study

ABSTRACT
Objective: To assess the effect of dose increase in adult patients with schizophrenia who demonstrate inadequate initial response to standard-dose lurasidone and to evaluate the efficacy of low-dose lurasidone in adult patients with schizophrenia.
Methods: In this randomized, double-blind, placebo-controlled study conducted between May 2013 and June 2014, hospitalized patients with acute schizophrenia (DSM-IV-TR criteria) were randomly assigned to double-blind treatment with lurasidone 20 mg/d (n = 101), lurasidone 80 mg/d (n = 199), or placebo (n = 112). Nonresponders to lurasidone 80 mg/d (Positive and Negative Syndrome Scale [PANSS] score decrease < 20%) at 2 weeks were re-randomized to lurasidone 80 mg/d or 160 mg/d for the remaining 4 weeks of the study. The primary outcome measure was change from baseline to week 6 in PANSS total score.
Results: In nonresponders to lurasidone 80 mg/d (n = 95), dose increase to 160 mg/d at week 2 significantly reduced PANSS total score at week 6 study endpoint compared with continuing 80 mg/d (-16.6 vs -8.9; P < .05 [effect size = 0.52]). While a comparable magnitude of improvement was observed in Clinical Global Impression-Severity (CGI-S) score from week 2 to week 6 endpoint for lurasidone 160 mg/d versus 80 mg/d (-1.0 vs -0.6; effect size = 0.44), the difference was not statistically significant (P = .052). Patients receiving lurasidone 20 mg/d did not demonstrate significant improvement compared with placebo at week 6 in PANSS total (-17.6 vs -14.5; P = .26) or CGI-S (-0.93 vs -0.73; P = .17) scores. Few dose-related adverse effects associated with lurasidone were observed.
Conclusions: In adult patients with schizophrenia demonstrating nonresponse to 2 weeks of treatment with lurasidone 80 mg/d, dose increase to 160 mg/d resulted in significant symptom improvement compared with continuing lurasidone 80 mg/d. Lurasidone 20 mg/d was not associated with significant improvement in psychotic symptoms in adult patients with schizophrenia.
Trial Registration: ClinicalTrials.gov identifier: NCT01821378
J Clin Psychiatry 2016;77(12):1672-1680
dx.doi.org/10.4088/JCP.16m10698
© Copyright 2016 Physicians Postgraduate Press, Inc.
aSunovion Pharmaceuticals Inc, Fort Lee, New Jersey, and Marlborough, Massachusetts
bDepartment of Psychiatry & Behavioral Sciences, New York Medical College, Valhalla
cDepartment of Psychiatry, The Zucker Hillside Hospital, Glen Oaks; and Department of Psychiatry, Hofstra North Shore-Long Island Jewish School of Medicine, Hempstead, New York
*Corresponding author: Antony Loebel, MD, Sunovion Pharmaceuticals Inc, One Bridge Plaza North, Ste 510, Fort Lee, NJ 07024 ([email protected]).
Lack of adequate improvement early in the course of antipsychotic therapy has been established as a predictor of later nonresponse in multiple short-term studies of patients with acute schizophrenia.1-10 Strategies for addressing early nonresponse have not been well studied, however, resulting in little empirical evidence to inform clinical decision-making for patients with schizophrenia who demonstrate an inadequate initial response to treatment. Potential management strategies for early nonresponse include continuation of the initial treatment (ie, waiting for further improvement), dose escalation, augmentation with adjunctive treatments, or a switch to another antipsychotic medication.11,12
Lurasidone is an atypical antipsychotic agent with high binding affinity for D2, 5-HT2A, and 5-HT7 receptors (antagonist), moderate affinity for 5-HT1A receptors (partial agonist), and no appreciable affinity for H1 and M1 receptors.13 Lurasidone was approved for the treatment of adult patients with schizophrenia based on 5 short-term, placebo-controlled studies that evaluated the dose range of 40-160 mg/d.14-18 The current study had 2 main objectives: to evaluate the efficacy of lurasidone dose increase in patients with an inadequate response after 2 weeks of standard-dose lurasidone therapy and to determine the potential efficacy of low-dose lurasidone (20 mg/d) in patients with schizophrenia.
METHODS
Patients
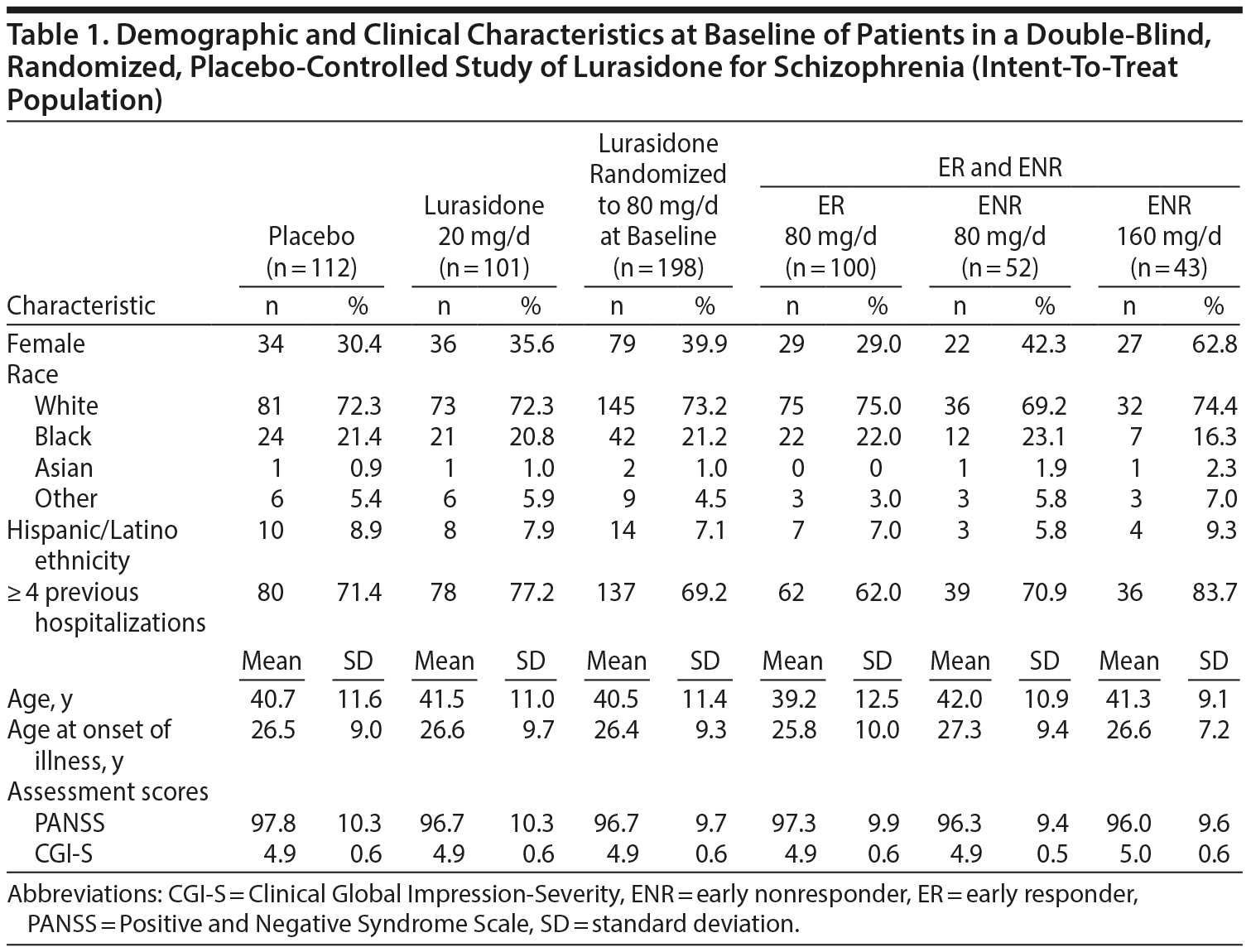
This study enrolled patients 18-75 years of age diagnosed with schizophrenia for ≥ 6 months (DSM-IV-TR criteria) and experiencing an acute exacerbation (< 2 months in duration), as indicated by a PANSS total score ≥ 80; a PANSS item score ≥ 4 (moderate) on ≥ 2 of the following items: delusions, conceptual disorganization, hallucinations, and unusual thought content; and a Clinical Global Impression-Severity (CGI-S) score ≥ 4 (moderately ill). Patients were excluded if another DSM-IV diagnosis had been the primary focus of treatment within the previous 3 months or if they had a history of alcohol or substance abuse (previous 3 months), showed evidence of a severe movement disorder, or were considered by the investigator to be at imminent risk of suicide or injury to self or others.
The study was approved by an institutional review board or ethics committee at each investigational site and was conducted in accordance with the International Conference on Harmonization Good Clinical Practices guidelines and with the ethical principles of the Declaration of Helsinki. After complete description of the study to the patients, written informed consent was obtained. The study was registered at ClinicalTrials.gov (identifier: NCT01821378).
Study Design
This multiregional, 6-week, randomized, double-blind, placebo-controlled, parallel-group study was conducted between May 2013 and June 2014. The rationale and design of this study are discussed in greater detail in a separate report.19
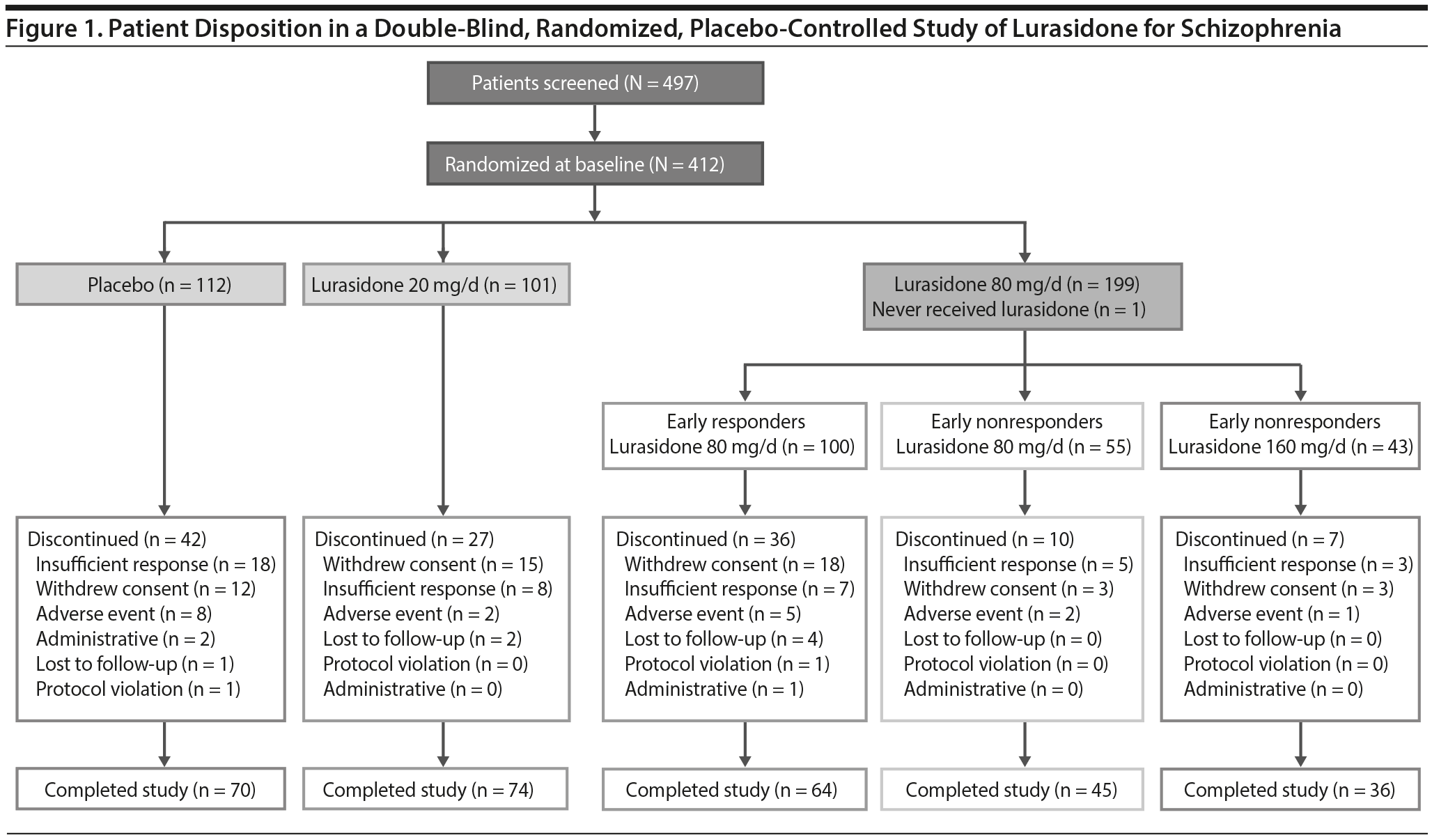
A total of 412 patients underwent random assignment in a 1:2:1 ratio via an interactive voice/web response system to receive fixed-dose lurasidone 20 mg/d, lurasidone 80 mg/d, or placebo. Study medication was provided in blister packs of identically matched lurasidone 20 mg, lurasidone 40 mg, or placebo tablets. A central randomization center used a computer-generated list of random numbers to allocate study treatments. None of the investigators, study staff, or patients had access to randomization codes or list. Study medication was taken in the evening with a meal or within 30 minutes after eating. Patients assigned to lurasidone 20 mg/d or placebo received the same medication throughout the study. Patients assigned to lurasidone 80 mg/d received this dose for the first 2 weeks, at which time they were classified as early responders (≥ 20% improvement from baseline in PANSS total score) or early nonresponders (< 20% improvement). Early responders continued to take lurasidone 80 mg/d, while early nonresponders were re-randomized in a 1:1 ratio either to continue lurasidone 80 mg/d or to receive 160 mg/d for the remaining 4 weeks of the study. Exposure to lurasidone was evaluated using pharmacokinetic analysis of blood samples taken at weeks 2, 4, and 6.

- Limited evidence is available to guide clinicians in selecting appropriate treatment strategies for patients with schizophrenia who demonstrate early nonresponse to antipsychotic therapy.
- This study provides controlled evidence that early nonresponders with schizophrenia may benefit from an increase in lurasidone dose from 80 mg/d to 160 mg/d. Significantly greater improvement in schizophrenia symptoms was found after lurasidone dose escalation compared with continuing the initial dose.
- Lurasidone 20 mg/d was not efficacious for the treatment of adult patients with acute schizophrenia.
Hospitalization was mandatory for all patients through week 3, after which patients deemed clinically stable and appropriate for discharge (including CGI-S score ≤ 3) were followed as outpatients.
Concomitant Medications
Treatment with anticholinergic agents or propranolol was permitted for movement disorders as needed (but not prophylactically). Lorazepam, temazepam, and zolpidem (or their equivalents) were permitted as needed for anxiety or insomnia but not within 8 hours of any psychiatric assessment.
Efficacy Assessments
Efficacy and safety assessments were performed by qualified site-based raters. The primary and key secondary efficacy endpoints were the change from baseline to week 6 in PANSS total score and CGI-S score, respectively, for the lurasidone 20 mg/d group compared with the placebo group. Secondary efficacy endpoints included change from baseline to week 6 on the PANSS and CGI-S for patients randomized to 80 mg/d compared with the placebo group and change from week 2 to week 6 for early nonresponders who received lurasidone 160 mg/d versus 80 mg/d.
Safety and Tolerability Evaluation
Safety and tolerability were assessed by the incidence and severity of adverse events during the study. Movement disorders were assessed by the Simpson-Angus Scale, the Barnes Akathisia Rating Scale, and the Abnormal Involuntary Movement Scale. Additional safety evaluations included vital signs, laboratory tests, 12-lead electrocardiogram (ECG), and physical examination. Suicidal ideation and behavior were assessed using the Columbia-Suicide Severity Rating Scale (C-SSRS).
Statistical Analysis
The intent-to-treat (ITT) population consisted of randomly assigned patients who received at least 1 dose of study medication and had a least 1 postbaseline efficacy assessment. The early nonresponder (ENR) ITT population consisted of patients in the lurasidone 80 mg/d group who were re-randomized at week 2 (to lurasidone 80 mg/d or 160 mg/d), received at least 1 dose of study medication after randomization, and had at least 1 subsequent efficacy assessment. For early nonresponders who continued on lurasidone 80 mg/d (ENR 80 mg) or increased to 160 mg/d (ENR 160 mg), efficacy results are presented from week 2 (time of re-randomization) to week 6. Efficacy endpoints were evaluated using a mixed model for repeated measures (MMRM) analysis that included treatment, visit, pooled center, baseline scores, and a treatment-by-visit interaction term, using an unstructured covariance matrix for within-patient correlation. To preserve the type I error rate, a sequential testing procedure was employed, with the primary efficacy variable (change in PANSS total score for lurasidone 20 mg/d vs placebo) tested first. Interpretation of the result for the key secondary variable (CGI-S for lurasidone 20 mg/d vs placebo) could be made only if the result for the primary variable was significant. There were no adjustments for multiplicity for other secondary efficacy analyses.
The proportion of responders at study endpoint (defined as ≥ 20% or ≥ 30% reduction from baseline PANSS total score [with 30 points subtracted from each score to account for the scale range]) was compared between the lurasidone and placebo groups using logistic regression with the last observation carried forward (LOCF). The number needed to treat (NNT) to attain 1 additional responder was derived for the lurasidone groups as follows: NNT = 1/(lurasidone responders – placebo responders).
The safety population included all patients who were randomly assigned and received at least 1 dose of study medication. For all patients, including early nonresponders re-randomized at week 2, safety data were summarized from study baseline through endpoint.
Sample size was determined based on 2-sample t tests and powered at 80%. One hundred fifty patients (75 patients for each of the placebo and lurasidone 20 mg/d arms) were estimated to be required to detect an 8.8-point difference in mean change from baseline to week 6 in PANSS total score between lurasidone 20 mg/d and placebo (with a pooled standard deviation [SD] of 19). It was estimated that 92 patients (46 patients for each of the lurasidone ENR 80 mg/d and ENR 160 mg/d arms) were required to detect an 11.3-point treatment difference in mean change from week 2 to week 6 (with a pooled SD of 19). The total estimated sample size of 400 patients (placebo: 100 patients, lurasidone 20 mg/d: 100 patients, and lurasidone 80 mg/d: 200 patients) included an additional 86 patients based on expected attrition.
RESULTS
A total of 412 patients met all eligibility criteria and were randomly assigned to double-blind treatment with lurasidone 20 mg/d (n = 101), lurasidone 80 mg/d (n = 199), or placebo (n = 112) (Figure 1). Demographic and clinical characteristics were similar for the 3 treatment groups randomized at study baseline (Table 1). After 2 weeks of treatment with lurasidone 80 mg/d, 98 patients (49.5%) were classified as early nonresponders (< 20% improvement in PANSS total score) and re-randomized to continued treatment with lurasidone 80 mg/d (n = 55) or lurasidone 160 mg/d (n = 43).
Efficacy
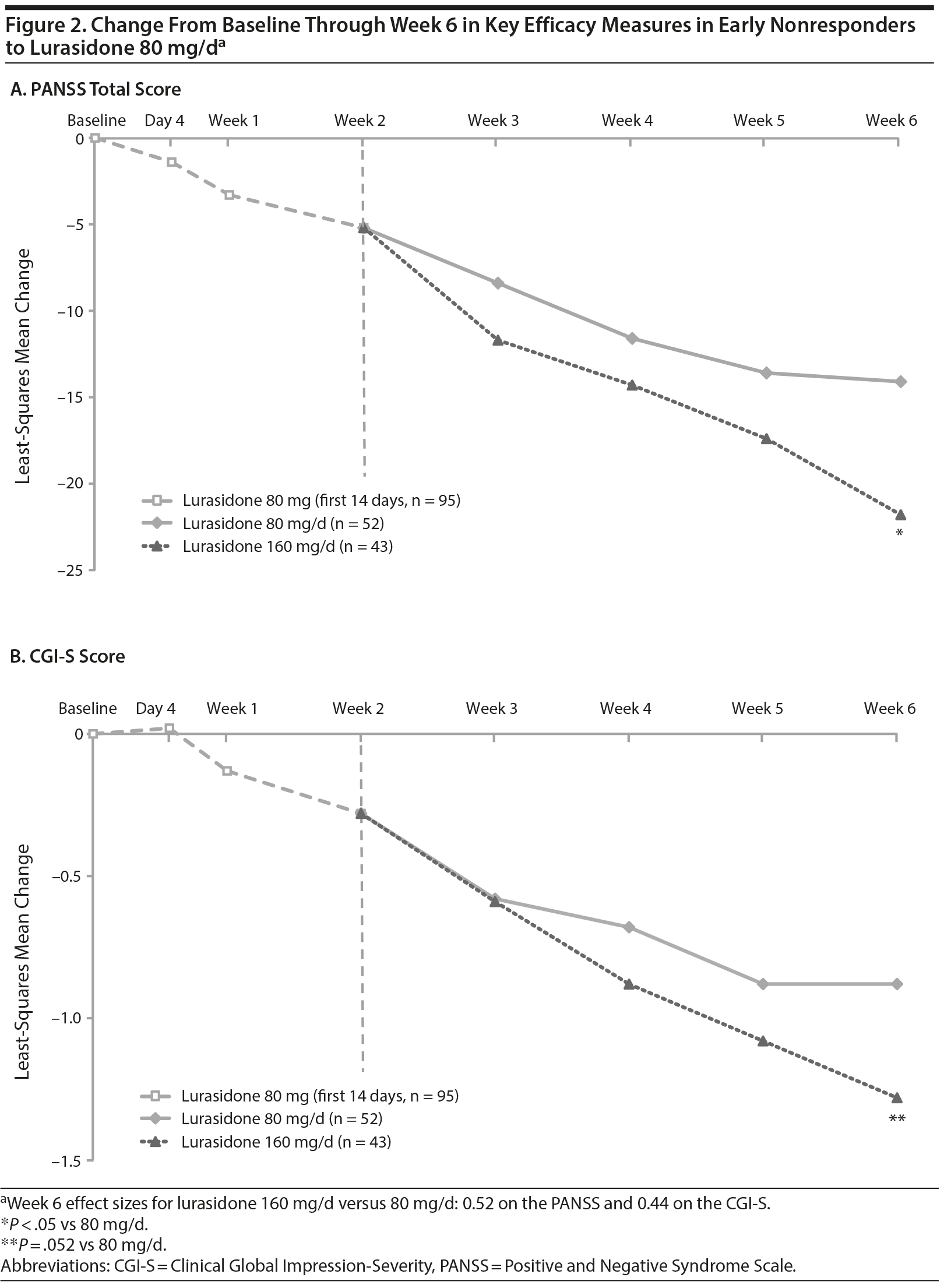
In patients classified as early nonresponders at week 2, least squares (LS) mean change from week 2 to week 6 in PANSS total score was significantly greater for patients whose lurasidone dose was increased to 160 mg/d compared with that for patients who continued receiving 80 mg/d (-16.6 vs -8.9 [difference, 7.7]; P < .05 [effect size = 0.52]; Figure 2A). While a comparable magnitude of improvement was observed in CGI-S score from week 2 to week 6 for early nonresponders re-randomized to lurasidone 160 mg/d versus 80 mg/d (LS mean change, -1.0 vs -0.6 [difference, 0.4]; effect size = 0.44), the difference was not statistically significant (P = .052; Figure 2B). Treatment response (defined as ≥ 20% decrease in PANSS total score from study baseline to week 6, LOCF) was demonstrated by 74.4% and 59.6% of early nonresponders re-randomized to lurasidone 160 mg/d and 80 mg/d, respectively (P = .164 [NNT, 7]); treatment response defined as ≥ 30% PANSS total score decrease was demonstrated by 53.5% and 38.5% of patients, respectively (P = .177 [NNT, 7]). Results for other secondary efficacy outcomes in early nonresponders are reported in Supplementary eTable 1 (available at PSYCHIATRIST.COM).
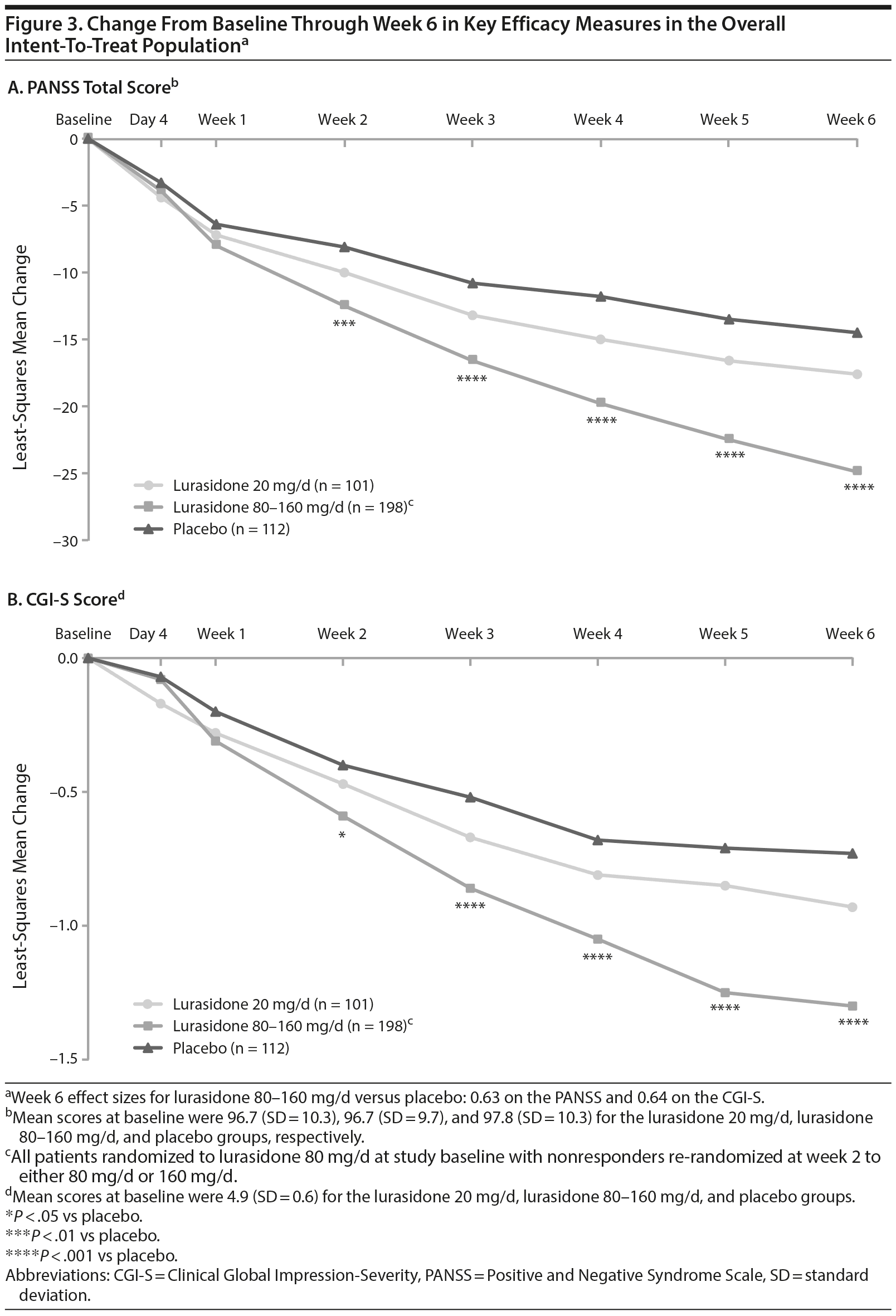
In patients receiving lurasidone 20 mg/d, LS mean change from baseline to week 6 in PANSS total score (the primary study endpoint, MMRM analysis) was not significantly different compared with patients receiving placebo (-17.6 vs -14.5; P = .26 [effect size = 0.19]). Similarly, LS mean change on the CGI-S was not significantly different in patients receiving lurasidone 20 mg/d compared with placebo (-0.9 vs -0.7; P = .17 [effect size = 0.22]). Patients initially randomized to lurasidone 80 mg/d demonstrated significant improvement overall compared with the placebo group at week 6 on both the PANSS (LS mean change, -24.9; P < .001 [effect size = 0.63]) and CGI-S (LS mean change, -1.3; P < .001 [effect size = 0.64]) scores (Figure 3A and 3B). These findings supported the assay sensitivity of the study.
Pharmacokinetic analysis evaluated the relationship between lurasidone exposure and early response/nonresponse (eAppendix 1).
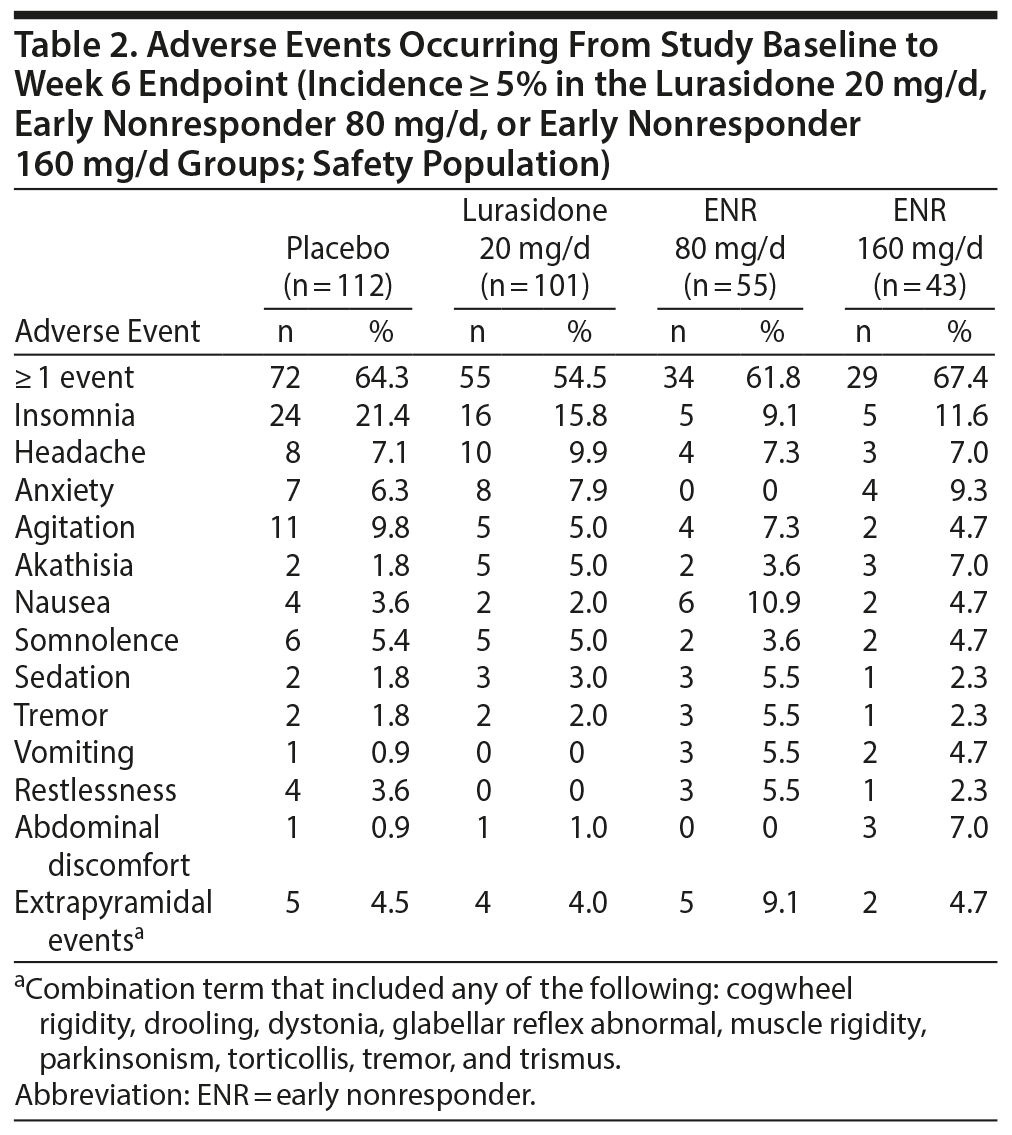
Safety
Adverse events with incidence ≥ 5% from study baseline to endpoint are listed in Table 2 for the following groups: lurasidone 20 mg/d, early nonresponders re-randomized to lurasidone 80 mg/d, and early nonresponders re-randomized to lurasidone 160 mg/d. Serious treatment-emergent adverse events were reported in 3 patients in the lurasidone 20 mg/d group, 4 patients in the early nonresponder 80 mg/d group, 1 patient in the early nonresponder 160 mg/d group, and 8 patients in the placebo group. No deaths occurred during the study. In early nonresponders, patients whose dose was increased to lurasidone 160 mg/d reported a greater incidence of anxiety, abdominal discomfort, akathisia, insomnia, and somnolence compared with patients who continued on lurasidone 80 mg/d (Table 2). The adverse event profile was similar for early responders to lurasidone 80 mg/d (Supplementary eTable 2) and early nonresponders continued on lurasidone 80 mg/d, except for akathisia (16.0% vs 3.6%) and parkinsonism (5.0% vs 0.0%), which were more common in early responders. Adverse events with onset after re-randomization of early nonresponders at week 2 are shown in Supplementary eTable 3.
The proportion of patients with treatment-emergent suicidal ideation, per the C-SSRS, was 2.0% in the lurasidone 20 mg/d group, 3.6% in the early nonresponder 80 mg/d group, 4.7% in the early nonresponder 160 mg/d group, and 0% in the placebo group. There were no suicidal behavior reports or completed suicides during the study.
The incidence of extrapyramidal symptom-related adverse events is reported in Table 2. The proportion of patients who received anticholinergic medication at any time during treatment with study medication was 5.0% in the lurasidone 20 mg/d group, 9.0% in the early nonresponder 80 mg/d group, 9.3% in the early nonresponder 160 mg/d group, and 4.5% in the placebo group. LS mean change from baseline to LOCF endpoint in Simpson-Angus Scale, Barnes Akathisia Scale, and Abnormal Involuntary Movement Scale scores was small and not clinically meaningful in all treatment groups.
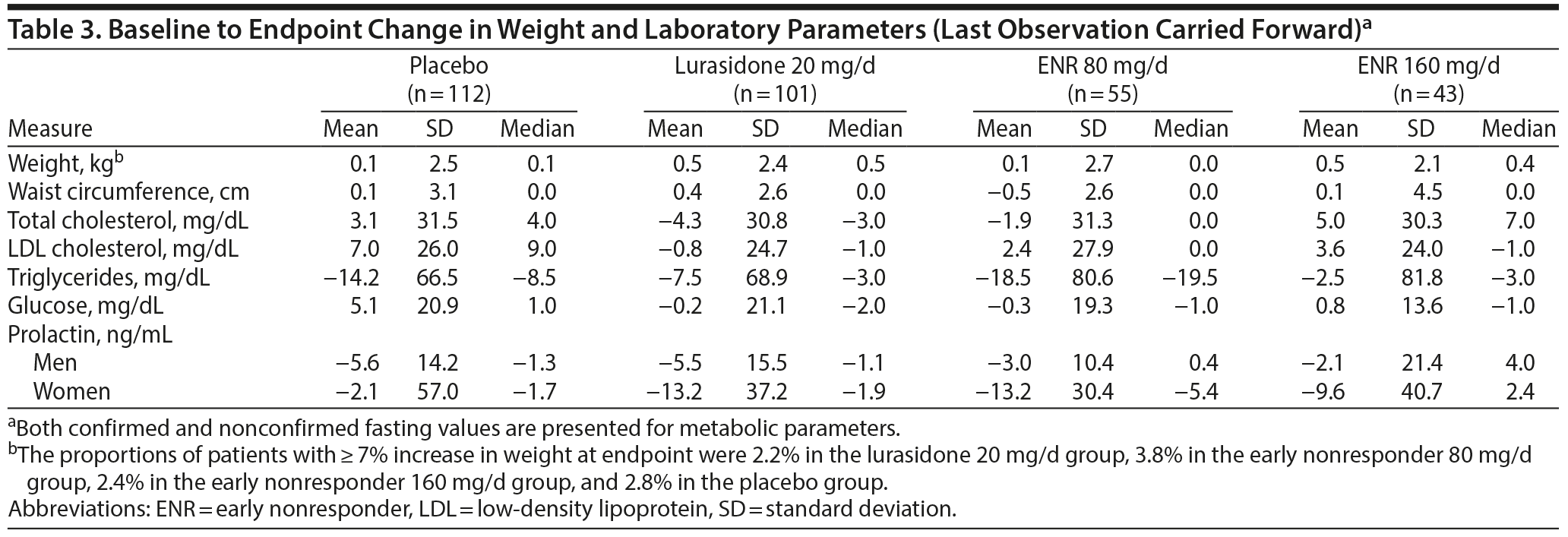
There were no clinically meaningful differences in the effect of treatment on laboratory parameters or vital signs (Table 3) or on ECG assessments between the lurasidone groups and the placebo group. Minimal changes in weight were observed across all groups, with no significant differences for the lurasidone groups (20 mg/d and 80 mg/d at initial randomization) compared with placebo. No patients in any lurasidone group had a postbaseline change in QTc Fridericia > 60 ms or a postbaseline QTc Fridericia > 500 ms. Vital signs, laboratory tests, and ECG assessments were generally similar for early responders and early nonresponders to lurasidone 80 mg/d (Supplementary eTable 2).
DISCUSSION
This study utilized a rigorous, placebo-controlled design to evaluate the effect of dose escalation in adult patients with schizophrenia who demonstrated an inadequate initial response to lurasidone. The results reported here support the safety and efficacy of dose escalation as a strategy for addressing early nonresponse to a standard dose of lurasidone in patients with schizophrenia. Dose increase to lurasidone 160 mg/d was associated with statistically significant and clinically meaningful improvement in symptoms of schizophrenia, as assessed by the PANSS total score, compared with continuation of the initial dose in early nonresponders to lurasidone 80 mg/d (P < .05; week 6 effect size = 0.52). In addition, clinically meaningful but statistically nonsignificant improvement was observed on the CGI-S (P = .052; week 6 effect size = 0.44).
Early nonresponse to antipsychotic therapy in patients with schizophrenia has been identified as a robust predictor of poor response at short-term trial endpoints.1 In this study, response/nonresponse was assessed after 2 weeks of treatment based on converging evidence from schizophrenia studies that evaluated receiver operating characteristics (eg, sensitivity, specificity, and positive and negative predictive values) and found that a 2-week decision time point may be ideal for prediction of short-term outcomes.2,6-9
PANSS total score reduction < 20% was utilized to define nonresponse in this study. This change in PANSS total score is comparable to a rating of minimally improved or worse on the CGI-Improvement scale20 and consistent with response thresholds utilized in previous research.4,7,21,22 Using this threshold of response, the present study showed that increasing the lurasidone dose to 160 mg/d at week 2 in nonresponders to lurasidone 80 mg/d was associated with a week 6 response rate of 74.4% compared with 59.6% for patients who continued the 80 mg/d dose. The placebo-adjusted difference in response rates (or treatment effect size) for lurasidone 160 mg/d versus 80 mg/d was essentially unchanged when the response threshold was increased to a ≥ 30% decrease from baseline PANSS total score (14.8%, NNT = 7 vs 15.0%, NNT = 7), suggesting that this finding was not dependent on PANSS response threshold. Increasing the lurasidone dose from 80 mg/d to 160 mg/d was associated with modest increases in the incidence of anxiety, abdominal discomfort, akathisia, insomnia, and somnolence, relative to continuing lurasidone 80 mg/d. Consistent with previous studies,23,24 short-term treatment with lurasidone in this study showed no clinically meaningful effects on weight, lipid parameters, and glucose levels.
We note that an appreciable proportion of patients classified as nonresponders at week 2 in this study went on to demonstrate response after 6 weeks of treatment in the absence of dose escalation (ie, on lurasidone 80 mg/d). Therefore, the increased probability of response in early nonresponders after dose escalation to lurasidone 160 mg/d should be considered in the context of the risk-versus-benefit findings associated with this strategy. These considerations include a modest increase in the frequency of certain adverse events and the clinical need to enhance the rate of response.
Study designs for evaluating the effectiveness of dose escalation in early nonresponding patients with schizophrenia have been proposed by Correll et al.25 However, few randomized, controlled studies have evaluated dose escalation as a strategy for addressing early nonresponse to antipsychotic therapy. A recent meta-analysis26 included 5 trials (348 patients) investigating dose escalation with quetiapine (2 studies), ziprasidone, haloperidol, or fluphenazine. There was no evidence that patients with early nonresponse benefited from dose escalation; no significant differences were found in mean PANSS/Brief Psychiatric Rating Scale score change between the dose-increase and control groups, and the pooled effect size for the primary outcome was 0.14.26 Sample sizes were relatively small, however, and nonresponse was assessed after 3 or 4 weeks of treatment in most studies. None of the 5 studies included a placebo-only treatment arm, and the 3 more recent studies that tested dose escalation of quetiapine and ziprasidone examined supratherapeutic doses that exceeded the maximum recommended limit contained in product labeling.
In contrast to prior studies, dose escalation was found to be efficacious in this study of patients with schizophrenia treated with lurasidone. Several factors may have contributed to the ability to detect an effect of lurasidone dose escalation in this study. First, inclusion of a placebo-control group may have reduced expectation bias toward improvement on the part of both patients and investigators, thereby enhancing signal detection in this study. Second, it is possible that D2 receptor occupancy was more optimal in early nonresponders treated with lurasidone 160 mg/d compared with 80 mg/d. Imaging studies have shown a strong relationship between serum lurasidone concentration and D2 receptor occupancy.27,28 A PET study of patients with schizophrenia randomly assigned to receive lurasidone 80, 120, or 160 mg/d for 1 week indicated that doses greater than 80 mg/d may be necessary to achieve therapeutic threshold levels of D2 receptor occupancy in some patients.28 In support of these observations, in this study, serum lurasidone concentration increased in a dose-related fashion, with an approximately 2-fold increase observed in patients treated with lurasidone 160 mg/d compared with patients continued on 80 mg/d. Notably, serum lurasidone concentration in patients receiving lurasidone 80 mg/d did not differentiate treatment responders from nonresponders at week 2. Third, in a prior short-term, placebo-controlled, fixed-dose study in patients with schizophrenia, a significantly higher rate of response was observed in patients treated with lurasidone 160 mg/d compared with 80 mg/d.18,29 The observed effects of lurasidone dose escalation in the current study are therefore consistent with previous observations of dose-response in association with lurasidone treatment.30
In this study, lurasidone 20 mg/d was not found to be efficacious in the treatment of patients with an acute exacerbation of schizophrenia. In contrast, the patient group initially randomized to 80 mg/d showed substantial improvement on the PANSS and CGI-S compared with the placebo group (week 6 effect sizes of 0.63 and 0.64, respectively). Given the efficacy demonstrated in patients randomized to lurasidone 80 mg/d, it appears that the absence of significant improvement in psychotic symptoms associated with lurasidone 20 mg/d represents a true negative result (because an effect of treatment was demonstrated in the group exposed to standard-dose lurasidone) and not a failed study (in which known effective treatment[s] or dose[s] fail to separate from placebo). This study, taken together with the findings from previous lurasidone efficacy trials, indicates that the lowest effective dose of lurasidone for the treatment of schizophrenia is 40 mg/d, as noted in current prescribing information.31
Several study limitations should be noted. The efficacy of dose escalation represented a secondary assessment in this study (the efficacy of lurasidone 20 mg/d was the primary endpoint). Treatment duration was relatively brief, and it is not known whether the differences between lurasidone 160 mg/d and 80 mg/d observed in this short-term study would be maintained beyond 4 weeks of treatment. The effect of increase to an intermediate lurasidone dose between 80 mg/d and 160 mg/d (ie, 120 mg/d) in patients with early nonresponse was not evaluated in this study. It is possible that determination of nonresponse at a different time point (particularly later than 2 weeks) may reduce observed differences between the lurasidone dose groups. Assessment of adverse effect frequencies associated with standard versus high lurasidone dose was limited due to relatively small sample sizes and number of adverse event reports.
In this study, dose increase of lurasidone in early nonresponders resulted in superior efficacy compared with continuing the initial dose, with modest increases in the incidence of some adverse events. Lurasidone 20 mg/d was not found to be an efficacious treatment for patients with schizophrenia, supporting the currently recommended lurasidone dose range of 40-160 mg/d. Dose escalation may be a preferred treatment approach for patients with schizophrenia who demonstrate an inadequate initial response to lurasidone.
Submitted: January 21, 2016; accepted June 20, 2016.
Online first: July 19, 2016.
Drug names: lorazepam (Ativan and others), lurasidone (Latuda), propranolol (Inderal and others), quetiapine (Seroquel and others), temazepam (Restoril and others), ziprasidone (Geodon and others), zolpidem (Ambien, Edluar, and others).
Potential conflicts of interest: Drs Loebel, Silva, Goldman, and Cucchiaro and Ms Watabe are employees of Sunovion Pharmaceuticals Inc. Dr Citrome has engaged in collaborative research with or received consulting or speaking fees from Acadia, Alexza, Alkermes, Allergan (Actavis, Forest), AstraZeneca, Avanir, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly and Company, Forum, Genentech, Intra-Cellular Therapies, Janssen, Jazz, Lundbeck, Medivation, Inc, Merck & Co, Inc, Mylan, Novartis, Noven, Otsuka, Pfizer, Reckitt Benckiser, Reviva, Shire, Sunovion, Takeda, Teva, Valeant, and Vanda. Dr Kane has been a consultant for Alkermes, Bristol-Myers Squibb, Eli Lilly, Forest, Forum, Genentech, Lundbeck, Intra-Cellular Therapies, Janssen, Johnson & Johnson, Novartis, Otsuka, Reviva, Roche, Sunovion, and Teva; has received honoraria from Alkermes, Bristol-Myers Squibb, Eli Lilly, Forest, Forum, Genentech, Lundbeck, Intra-Cellular Therapies, Janssen, Johnson & Johnson, Otsuka, Reviva, Roche, Sunovion, and Teva; has been on the speakers/advisory boards for Bristol-Myers Squibb, Genentech, and Otsuka; and has been a stock shareholder in MedAvante.
Funding/support: This study was sponsored by Sunovion Pharmaceuticals Inc.
Role of the sponsor: The sponsor was involved in the study design, collection, and analysis of data. The interpretation of results and the decision to submit this manuscript for publication in The Journal of Clinical Psychiatry were made by the authors independently.
Previous presentations: Presented in part at the 53rd Annual Meeting of the American College of Neuropsychopharmacology, December 7-11, 2014, Phoenix, Arizona; 15th International Congress on Schizophrenia Research, March 28-April 1, 2015, Colorado Springs, Colorado; the 168th Annual Meeting of the American Psychiatric Association, May 16-20, 2015, Toronto, Canada; and the US Psychiatric and Mental Health Congress, September 10-13, 2015, San Diego, California.
Acknowledgments: The authors thank the study investigators for their participation and Michael Tocco, PhD, for scientific review of this article. Nancy Holland, PhD, Synchrony Medical Communications, LLC, provided medical writing and editorial assistance for this manuscript under the direction of the authors. Financial support for this writing and editorial assistance from Synchrony Medical Communications, LLC, was provided by Sunovion Pharmaceuticals Inc, Marlborough, Massachusetts. Clinical research was sponsored by Sunovion Pharmaceuticals Inc.
Supplementary material: Available at PSYCHIATRIST.COM.
REFERENCES
1. Samara MT, Leucht C, Leeflang MM, et al. Early improvement as a predictor of later response to antipsychotics in schizophrenia: a diagnostic test review. Am J Psychiatry. 2015;172(7):617-629. PubMed doi:10.1176/appi.ajp.2015.14101329
2. O’ Gorman C, Kapur S, Kolluri S, et al. Early improvement on antipsychotic treatment as a predictor of subsequent response in schizophrenia: analyses from ziprasidone clinical studies. Hum Psychopharmacol. 2011;26(4-5):282-290. PubMed doi:10.1002/hup.1200
3. Stauffer VL, Case M, Kinon BJ, et al. Early response to antipsychotic therapy as a clinical marker of subsequent response in the treatment of patients with first-episode psychosis. Psychiatry Res. 2011;187(1-2):42-48. PubMed doi:10.1016/j.psychres.2010.11.017
4. Kinon BJ, Chen L, Ascher-Svanum H, et al. Early response to antipsychotic drug therapy as a clinical marker of subsequent response in the treatment of schizophrenia. Neuropsychopharmacology. 2010;35(2):581-590. PubMed doi:10.1038/npp.2009.164
5. Kinon BJ, Chen L, Stauffer VL, et al. Early onset of antipsychotic action in schizophrenia: evaluating the possibility of shorter acute efficacy trials. J Clin Psychopharmacol. 2010;30(3):286-289. PubMed doi:10.1097/JCP.0b013e3181dcb7c3
6. Ascher-Svanum H, Nyhuis AW, Faries DE, et al. Clinical, functional, and economic ramifications of early nonresponse to antipsychotics in the naturalistic treatment of schizophrenia. Schizophr Bull. 2008;34(6):1163-1171. PubMed doi:10.1093/schbul/sbm134
7. Kinon BJ, Chen L, Ascher-Svanum H, et al. Predicting response to atypical antipsychotics based on early response in the treatment of schizophrenia. Schizophr Res. 2008;102(1-3):230-240. PubMed doi:10.1016/j.schres.2008.02.021
8. Leucht S, Busch R, Kissling W, et al. Early prediction of antipsychotic nonresponse among patients with schizophrenia. J Clin Psychiatry. 2007;68(3):352-360. PubMed doi:10.4088/JCP.v68n0301
9. Chang YC, Lane HY, Yang KH, et al. Optimizing early prediction for antipsychotic response in schizophrenia. J Clin Psychopharmacol. 2006;26(6):554-559. PubMed doi:10.1097/01.jcp.0000246211.95905.8c
10. Correll CU, Malhotra AK, Kaushik S, et al. Early prediction of antipsychotic response in schizophrenia. Am J Psychiatry. 2003;160(11):2063-2065. PubMed doi:10.1176/appi.ajp.160.11.2063
11. Hatta K, Ito H. Strategies for early non-response to antipsychotic drugs in the treatment of acute-phase schizophrenia. Clin Psychopharmacol Neurosci. 2014;12(1):1-7. PubMed doi:10.9758/cpn.2014.12.1.1
12. Buckley PF, Correll CU. Strategies for dosing and switching antipsychotics for optimal clinical management. J Clin Psychiatry. 2008;69(suppl 1):4-17. PubMed
13. Ishibashi T, Horisawa T, Tokuda K, et al. Pharmacological profile of lurasidone, a novel antipsychotic agent with potent 5-hydroxytryptamine 7 (5-HT7) and 5-HT1A receptor activity. J Pharmacol Exp Ther. 2010;334(1):171-181. PubMed doi:10.1124/jpet.110.167346
14. Ogasa M, Kimura T, Nakamura M, et al. Lurasidone in the treatment of schizophrenia: a 6-week, placebo-controlled study. Psychopharmacology (Berl). 2013;225(3):519-530. PubMed doi:10.1007/s00213-012-2838-2
15. Nakamura M, Ogasa M, Guarino J, et al. Lurasidone in the treatment of acute schizophrenia: a double-blind, placebo-controlled trial. J Clin Psychiatry. 2009;70(6):829-836. PubMed doi:10.4088/JCP.08m04905
16. Meltzer HY, Cucchiaro J, Silva R, et al. Lurasidone in the treatment of schizophrenia: a randomized, double-blind, placebo- and olanzapine-controlled study. Am J Psychiatry. 2011;168(9):957-967. PubMed doi:10.1176/appi.ajp.2011.10060907
17. Nasrallah HA, Silva R, Phillips D, et al. Lurasidone for the treatment of acutely psychotic patients with schizophrenia: a 6-week, randomized, placebo-controlled study. J Psychiatr Res. 2013;47(5):670-677. PubMed
18. Loebel A, Cucchiaro J, Sarma K, et al. Efficacy and safety of lurasidone 80 mg/day and 160 mg/day in the treatment of schizophrenia: a randomized, double-blind, placebo- and active-controlled trial. Schizophr Res. 2013;145(1-3):101-109. PubMed doi:10.1016/j.schres.2013.01.009
19. Loebel A, Citrome L, Correll CU, et al. Treatment of early non-response in patients with schizophrenia: assessing the efficacy of antipsychotic dose escalation. BMC Psychiatry. 2015;15(1):271. PubMed doi:10.1186/s12888-015-0629-0
20. Leucht S, Kane JM, Kissling W, et al. What does the PANSS mean? Schizophr Res. 2005;79(2-3):231-238. PubMed doi:10.1016/j.schres.2005.04.008
21. Liu-Seifert H, Adams DH, Kinon BJ. Discontinuation of treatment of schizophrenic patients is driven by poor symptom response: a pooled post-hoc analysis of four atypical antipsychotic drugs. BMC Med. 2005;3(1):21. PubMed doi:10.1186/1741-7015-3-21
22. Leucht S, Zhao J. Early improvement as a predictor of treatment response and remission in patients with schizophrenia: a pooled, post-hoc analysis from the asenapine development program. J Psychopharmacol. 2014;28(4):387-394. PubMed doi:10.1177/0269881113517956
23. Citrome L. Lurasidone for the acute treatment of adults with schizophrenia: what is the number needed to treat, number needed to harm, and likelihood to be helped or harmed? Clin Schizophr Relat Psychoses. 2012;6(2):76-85. PubMed doi:10.3371/CSRP.6.2.5
24. Citrome L. Lurasidone for schizophrenia: a review of the efficacy and safety profile for this newly approved second-generation antipsychotic. Int J Clin Pract. 2011;65(2):189-210. PubMed doi:10.1111/j.1742-1241.2010.02587.x
25. Correll CU, Kishimoto T, Kane JM. Randomized controlled trials in schizophrenia: opportunities, limitations, and trial design alternatives. Dialogues Clin Neurosci. 2011;13(2):155-172. PubMed
26. Dold M, Fugger G, Aigner M, et al. Dose escalation of antipsychotic drugs in schizophrenia: a meta-analysis of randomized controlled trials. Schizophr Res. 2015;166(1-3):187-193. PubMed doi:10.1016/j.schres.2015.04.024
27. Wong DF, Kuwabara H, Brašić JR, et al. Determination of dopamine D₂ receptor occupancy by lurasidone using positron emission tomography in healthy male subjects. Psychopharmacology (Berl). 2013;229(2):245-252. PubMed doi:10.1007/s00213-013-3103-z
28. Potkin SG, Keator DB, Kesler-West ML, et al. D2 receptor occupancy following lurasidone treatment in patients with schizophrenia or schizoaffective disorder. CNS Spectr. 2014;19(2):176-181. PubMed doi:10.1017/S109285291300059X
29. Loebel A, Cucchiaro J, Silva R, et al. Efficacy of lurasidone across five symptom dimensions of schizophrenia: pooled analysis of short-term, placebo-controlled studies. Eur Psychiatry. 2015;30(1):26-31. PubMed doi:10.1016/j.eurpsy.2014.08.001
30. Chapel S, Chiu YY, Hsu J, et al. Lurasidone dose response in bipolar depression: a population dose-response analysis. Clin Ther. 2016;38(1):4-15. PubMed doi:10.1016/j.clinthera.2015.11.013
31. Latuda (lurasidone hydrochloride) tablets, for oral use [package insert]. Marlborough, MA: Sunovion Pharmaceuticals, Inc; 2013.
This PDF is free for all visitors!