ABSTRACT
Objective: To compare outcomes among newborns of opioid-using and nonopioid drug–using mothers with those of control mothers who did not report substance use.
Methods: Using the Rochester Epidemiology Project, newborns diagnosed with drug withdrawal syndrome (per ICD-9 or ICD-10 codes) from January 2010 through June 2017 were identified. For mothers, data collected included age, race, drug use, number of prenatal visits, and results of the urinary drug abuse survey, meconium test, and self-report survey. Demographic and perinatal data collected for newborns included birth date; sex; Apgar scores at 1, 5, and 10 minutes; neonatal intensive care stay; and vital status. Controls (n = 771) were similarly selected in regard to sex, birth date, and county.
Results: Of 328 infants identified, 168 were born with opioid neonatal abstinence syndrome and 160 with a nonopioid withdrawal syndrome. Control mothers had more prenatal visits than mothers in the nonopioid and opioid groups. Newborns of control mothers had higher Apgar scores at 1 and 5 minutes than both substance-using groups. Opioid-using mothers were almost twice as likely to have newborns requiring intensive care and 3 times as likely to use benzodiazepines compared to the other substance-using mothers. Substance-using mothers had more premature babies than controls.
Conclusions: Prenatal opioid use is a substantial risk factor for prematurity. Newborns diagnosed with neonatal abstinence syndrome are at risk of perinatal complications. Mothers using opioids during pregnancy also tend to use other substances. Longitudinal research should clarify how prenatal substance use interacts with other risk factors during a child’s first years.
Prim Care Companion CNS Disord 2021;23(3):20m02737
To cite: Gandhi KD, Schak KM, Vande Voort JL, et al. The negative impact of maternal perinatal opioid use on neonatal outcomes. Prim Care Companion CNS Disord. 2021;23(3):20m02737.
To share: https://doi.org/10.4088/PCC.20m02737
© Copyright 2021 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry and Psychology, Mayo Clinic, Rochester, Minnesota
bDivision of Community Pediatric and Adolescent Medicine, Mayo Clinic, Rochester, Minnesota
cDivision of Biomedical Statistics and Informatics, Mayo Clinic, Rochester, Minnesota
dDepartment of Psychiatry and Behavioral Science, Mayo Clinic, Rochester, Minnesota
eUniversity of North Dakota, Grand Forks, North Dakota
*Corresponsing author: Kathryn M. Schak, MD, Department of Psychiatry and Psychology, Mayo Clinic, 200 First St SW, Rochester, MN 55905 ([email protected]).
Opioid use in pregnancy has increased more than 5-fold during the opioid epidemic in the United States, and approximately 50% of women who abuse opioids also abuse at least 1 other drug.1 The consequences of opioid use in pregnancy, both for mother and infant, are a public health crisis. Maternal opioid use may lead to substantial complications for newborns including premature birth, feeding difficulties, autonomic instability, neurologic impairment, respiratory distress, and prolonged stays in the neonatal intensive care unit (NICU).2 Maternal polysubstance use further exacerbates perinatal outcomes and increases vulnerability for the newborn to develop neonatal abstinence syndrome.3 Polysubstance use in pregnancy has a complex impact on newborns that, at present, is poorly understood.
A study by Irner et al3 showed that newborns of mothers who were polysubstance abusers had poorer perinatal outcomes than newborns who were exposed only to alcohol or to no substances. Few prior studies have examined the concurrent use of alcohol, tobacco, or other substances for mothers with opioid use disorder despite the high prevalence of alcohol, tobacco, and opioid use during pregnancy.4,5 This lack of data is surprising given that almost 50% of women with opioid use disorder struggle with alcohol use in pregnancy according to the National Survey of Drug Use and Health.6 A mother’s concurrent use of opioids and alcohol puts her newborn at risk for developing neonatal abstinence syndrome (NAS) and fetal alcohol syndrome and should be part of the regular screening process for mothers who describe opioid use during pregnancy.7 Even more infants are at risk due to nicotine exposure in utero: 88% of pregnant women on opioid maintenance treatment smoke during pregnancy.8 The prevalence of nicotine addiction in pregnant women who are also struggling with opioid use disorder has been estimated as high as 97%.8
The primary objective of this study was to examine differences in perinatal outcomes of infants whose mothers primarily used opioids, a group of mothers who used substances other than opioids, and a control group of mothers who did not use any substances. We hypothesized that newborns of mothers with primary opioid use would have more medical complications and longer lengths of NICU stay than newborns of mothers who used other substances or did not use any substance. The exploratory objective of this study was to examine patterns of other drugs of abuse used in addition to opioids.
METHODS
Study Setting
Data were collected from the expanded Rochester Epidemiology Project (REP), which is a collaboration of clinics, hospitals, and other medical facilities over a 27-county region in Minnesota and Wisconsin. The REP includes data for community members who have agreed to share their health records for research and links records of persons across different providers.9 This study was approved by the Mayo Clinic Institutional Review Board, and informed consent was waived for those providing research authorization.
Identification of Newborns
We identified all newborns with birth dates between January 1, 2010, and June 30, 2017, and ICD-9 or ICD-10 codes specifically related to newborn drug withdrawal. To ensure that all newborns with drug withdrawal were included, current procedural terminology (CPT) codes were also searched. Supplementary Table 1 provides all ICD-9, ICD-10, and CPT codes. Patient information was validated by results from meconium and urinary drug abuse survey (UDAS) testing of the mother or newborn or both when available.
Data Collection
For mothers, collected data included age, race, number of prenatal visits, details of drug use, results of UDAS and meconium testing, and responses to self-report surveys at admission to the labor and delivery ward, including tobacco use (yes/no), drug use (yes/no), and alcohol use (yes/no). Stimulant or benzodiazepine use during pregnancy was also recorded. For the respective newborns, collected demographic and perinatal data included birth date; sex; Apgar scores at 1, 5, and 10 minutes after birth; NICU stay (yes/no); and death within the first 2 years after birth. Neonatal death was defined as death after live birth within 30 days of birth. In our study, we recorded all children who died within the first 2 years after live birth. Of 7 deaths, only 1 patient met criteria for neonatal death, 5 patients died within the first year of life, and 1 died within the first 2 years of life. Data regarding newborn medical conditions were divided into 5 groups: prematurity (defined as born before 37 weeks), failure to thrive, jaundice, feeding issues, and respiratory distress.
On the basis of information abstracted from the records, mothers were assigned to 3 categories: those who used opioids (opioid group), those who used drugs other than opioids (nonopioid drug group), and those who did not have a history of drug, alcohol, or tobacco use, including during pregnancy (nondrug control group). To make our study population more generalizable, we included mothers using opioids with or without marijuana, tobacco, alcohol, and benzodiazepines.
Statistical Analysis
Data were summarized using mean (SD) and range for continuous variables and counts and percentages for categorical variables. We then analyzed the association of maternal drug use classification (ie, opioid group, nonopioid drug group, and nondrug control group) and outcomes. Outcomes and group (opioid or nonopioid drug) were coded as binary indicator variables (0/1), except in initial comparisons of demographic and clinical variables across all 3 groups, using analysis of variance models assuming nominal group relationship. The nondrug control group was used as a baseline for comparisons. Linear regression models were used for continuous outcomes. Logistic regression models were used for binary outcome variables. Multivariable logistic regression models were used, with adjustments for maternal age, prenatal visits, and other drug use. P values were unadjusted for multiple comparisons; however, we considered 12 outcomes as primary interests, of which many were highly associated. Application of a strict (Bonferroni) correction led to a significance cutoff of P < .05/12 = .004, which retained our significant findings for all 12 outcomes. The groups were initially matched for newborn sex, newborn birth date (± 0.5 years), and geographic county region, However, based on changes in research authorization and data availability, some matches were excluded; thus, an exact paired case:control design was unattainable. Analyses were conducted using SAS v9.4 (SAS Institute Inc, Cary, North Carolina).
RESULTS
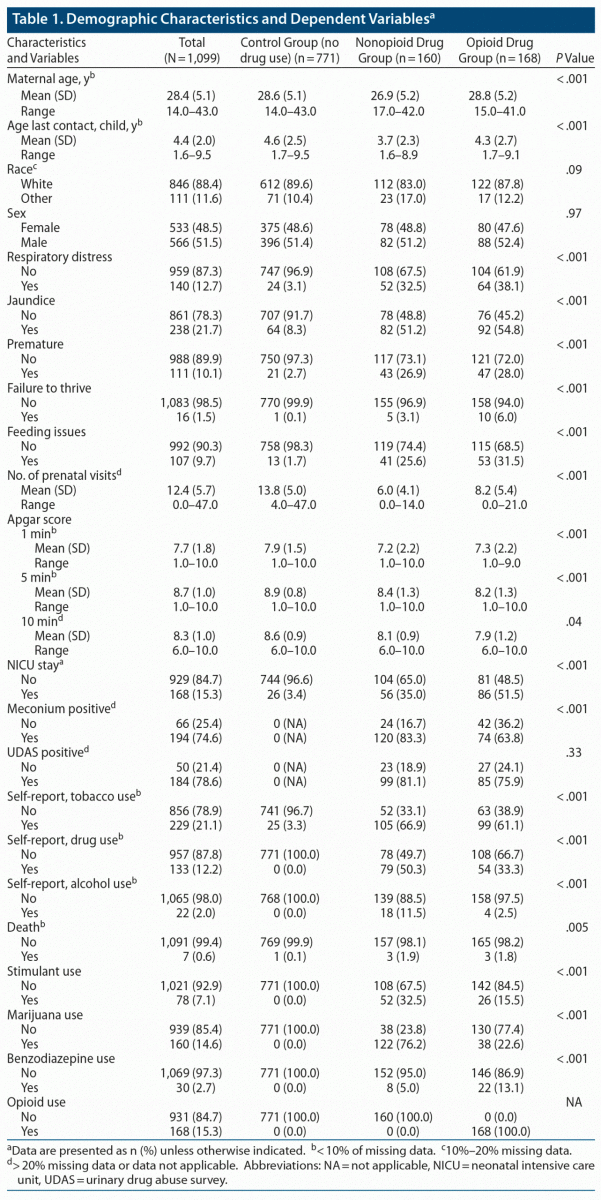
A total of 328 newborns with ICD and CPT codes related to postnatal drug withdrawal were identified. Of those 328 patients, 168 were identified as being born with opioid withdrawal and 160 with withdrawal from various drugs other than opioids, such as methamphetamines, barbiturates, phencyclidine, and cocaine. Most newborns were white: 89.6% in the control group, 83.0% in the nonopioid drug control group, and 87.8% in the opioid group (Table 1).
Logistic Regression Analysis
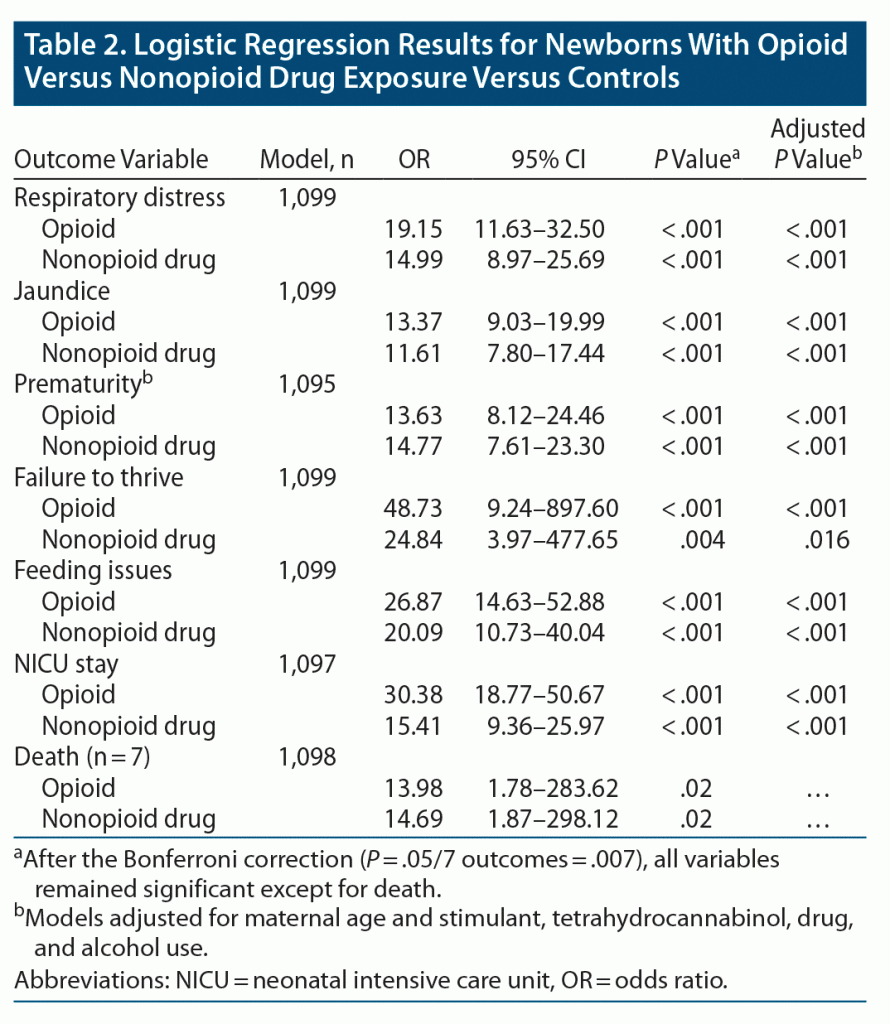
Newborns with opioid and nonopioid drug withdrawal had substantially higher odds than controls of having respiratory distress, jaundice, prematurity, failure to thrive, feeding issues, NICU stay, and death than newborns of mothers who did not use drugs (Table 2). These results remained consistent when accounting for maternal age and other drug use.
Linear Regression Results
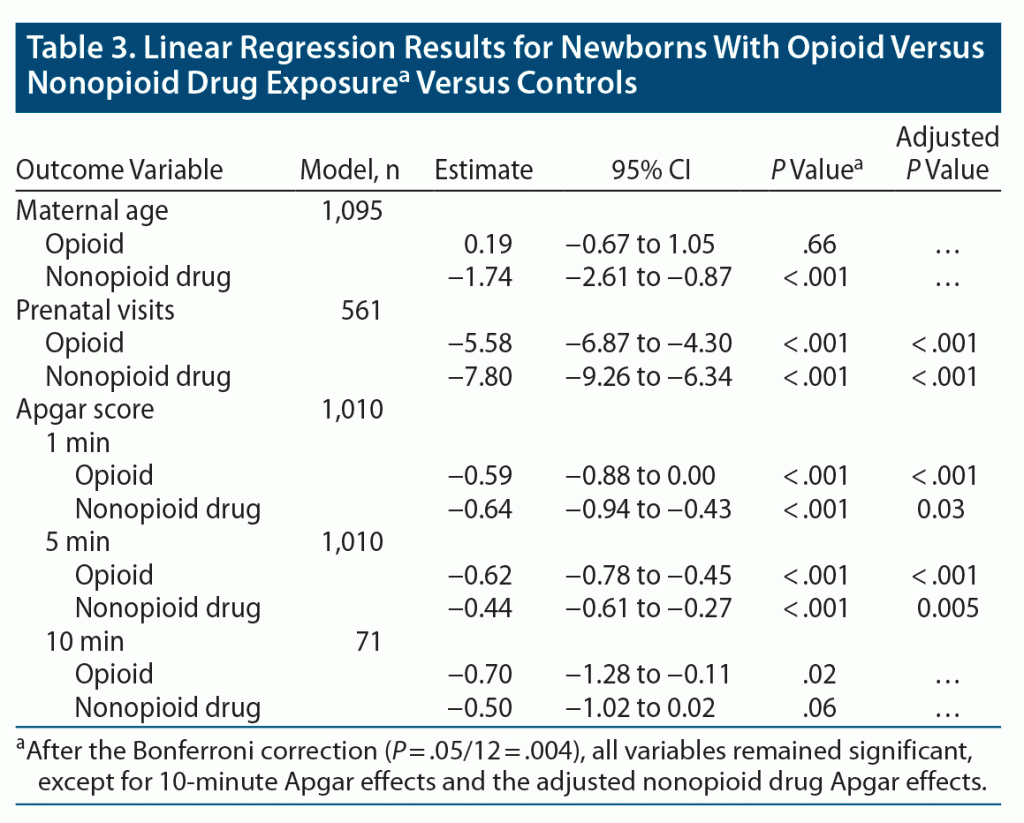
Mothers in the nonopioid drug group were younger (mean age of 26.9 years) than those in the nondrug control group (mean age of 28.8 years) (P < .001). Mothers in the nondrug group, on average, had more prenatal visits than opioid-using mothers and nonopioid drug–using mothers (mean of 13.81, 6.02, and 8.23, respectively; P < .001). As shown in Table 3, newborns of nondrug controls had higher Apgar scores at both 1 and 5 minutes than both drug-using groups (all P < .001). Prenatal and 1-minute Apgar results remained consistent when accounting for maternal age and other drug use.
Logistic Regression Results
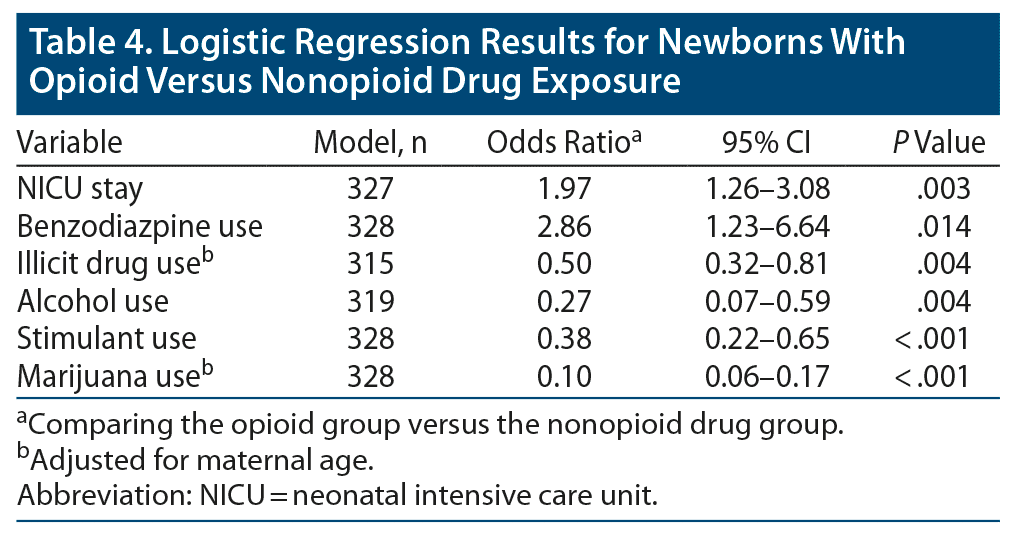
Mothers in the opioid group were almost twice as likely to have newborns who required a NICU stay (odds ratio [OR] = 1.97, P = .003) and almost 3 times more likely to use benzodiazepines (OR = 2.86, P = .01) than mothers in the nonopioid drug group (Table 4). However, opioid-using mothers were less likely than mothers in the nonopioid drug group to self-report the use of drugs (OR = 0.49, P = .001) or alcohol (OR = 0.27, P = .003) and to have a history of stimulant (OR = 0.38, P < .001) or marijuana use (OR = 0.09, P < .001).
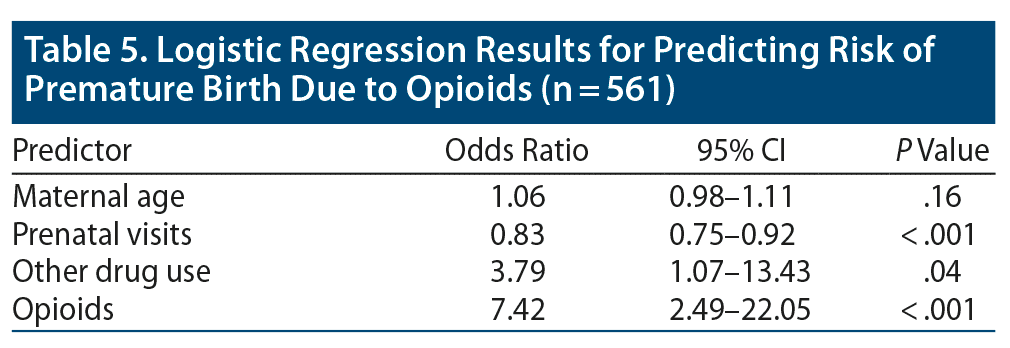
Mothers who used drugs had more premature babies: nonopioid drug group, 27%; opioid group, 28%; nondrug control group, 2.7% (P < .001). To determine risk factors for premature birth, we considered all drug-using mothers as 1 group and compared them with the control group of nonusers. Drug use increased the odds of having a premature baby, even when we adjusted for maternal age, tobacco use, and number of prenatal visits (OR = 3.79, P = .04). Opioid use further increased risk (OR = 7.42, P < .001) (Table 5).
DISCUSSION
In this study, we found that opioid use during pregnancy may substantially increase the risk for premature birth even more than the risk associated with drugs other than opioids. We found that nonopioid drug use in pregnancy was a significant risk factor for premature birth (OR = 3.79) and that opioid use during pregnancy increased the risk of premature delivery by 7.42 times compared with no substance use in pregnancy, even after adjusting for known risk factors of premature birth including maternal age, tobacco use, and number of prenatal visits.
The relationship of nonopioid drugs to prematurity has been described by others. In a population-based study, Baer et al10 examined all singleton live births in California between 2007 and 2012 and found that mothers with a history of polysubstance or cocaine use were at highest risk of premature birth, and marijuana was the only drug not associated with preterm birth.10 Prematurity is a main risk factor for infant mortality and morbidity.11 Infants born prematurely are more likely to experience respiratory distress syndrome,12 infections,13 necrotizing enterocolitis,14 and blindness.15 Prematurity is also a known risk factor for long-term consequences such as academic challenges, language difficulties,16 attention-deficit/hyperactivity disorder,17 nonaffective psychosis, and other mental health issues.18
In this study, perinatal events for newborns with withdrawal from opioids or other drugs occurred at rates similar or slightly higher than those reported in the literature. Over half of infants were treated for jaundice in both the opioid group and nonopioid drug group (54.8% and 51.2%, respectively). Infants exposed to opioids and other drugs were more than 3 times as likely to experience significant feeding issues as well as failure to thrive. According to the literature, hyperphagia attributable to increased metabolic needs can also occur in infants with opioid withdrawal.19 Our retrospective review did not include the diagnosis of hyperphagia, which is a limitation. Furthermore, both drug-exposed groups were nearly 3 times more likely to have significant respiratory distress than the nondrug control group. This finding is comparable to that of previous studies described in a review article20 that showed a number of respiratory issues including tachypnea in infants with NAS.
Polysubstance use occurred frequently in the opioid group. Newborns with opioid withdrawal had more frequent additional exposure to benzodiazepines than newborns in the nonopioid drug group, who were more likely to be exposed to marijuana and alcohol. This finding agrees with published data suggesting that more than 30% of opioid overdoses occur in combination with benzodiazepines.21,22 Patients frequently use the 2 drugs simultaneously (benzodiazepines to control withdrawal symptoms), which may explain this use pattern. Tobacco use was comparable for the 2 drug-exposed groups (> 60%) and much higher than in the control group (3%). This was an important finding because tobacco exposure is one of the major preventable risk factors of impaired child growth and development.23 Tobacco use by mothers is also a known risk factor for more severe opioid withdrawal symptoms in newborns.24–26 In the nonopioid drug group, 11.5% of mothers reported alcohol use, whereas 2.5% of mothers in the opioid group reported alcohol use, which is substantially less than 50% reported for mothers using opioids by the National Survey of Drug Use and Health.6 In our study, both groups may have substantially underreported substance use. Regardless, this finding emphasizes the importance of screening and finding new ways to address polysubstance use in pregnancy, especially for mothers who report opioid use.
Because of the retrospective design, this study had a number of limitations. It was difficult to estimate the drug type (whether it was prescribed or illicit use), quantity of drugs, and length of use during pregnancy. This difficulty is most likely a result of the mothers’ reluctance to disclose information about drug use to health care professionals. Although we had access to UDAS or meconium results or both for most mothers and infants, this information was not consistently available for every case. We used the clinical diagnosis of NAS, but its cause was limited to health record documentation. Not every infant with opioid withdrawal met NAS criteria, and we were unable to differentiate because of terminology differences between ICD-9 and ICD-10 codes. Original design included exact matches on birth year, sex, and county; however, due to research authorization and data availability limitations, we were unable to maintain the optimal 1:4 case/control matched structure. Additionally, we did not have access to the infants’ Finnegan scores, a quantitative measure of severity of opioid withdrawal, and we relied on indirect proxies for withdrawal severity, including NICU stay. It is possible that mild withdrawal cases were not included in our database. The use of NICU stay and length of stay is complicated because this may have been affected by factors unrelated to drug withdrawal severity such as other medical conditions, presence or absence of breastfeeding (information that we did not have), and the mother’s condition. We also did not have information about medication used to treat withdrawal during the hospitalization. Tobacco use and alcohol use were based only on mothers’ self-reports, which may not have been reliable. In addition to the newborn birth date used in matching, there also may have been temporal effects as therapies evolved over the 12 years reflected in the report experience.
CONCLUSION
Opioid use during pregnancy was a significant risk factor for many problems in newborns, including premature birth. Infants with NAS were at risk of major perinatal complications such as premature birth, jaundice, and prolonged NICU stay. Mothers who used opioids in pregnancy tended to abuse other substances including tobacco, benzodiazepines, stimulants, and marijuana and may underreport alcohol use. Prospective, longitudinal research is needed to further identify the pattern of polysubstance use in pregnancy, and new screening and intervention methods are needed for this patient population.
Submitted: June 29, 2020; accepted March 15, 2021.
Published online: June 10, 2021.
Potential conflicts of interest: Dr Vande Voort receives equipment in-kind support from Assurex Health for an investigator-initiated study and is a site investigator for a multicenter study funded by Neuronetics. Dr Croarkin has received research grant support from Pfizer, equipment support from Neuronetics, and supplies and genotyping services from Assurex Health for investigator-initiated studies; is the primary investigator for a multicenter study funded by Neuronetics and a site primary investigator for a study funded by NeoSync; and is a consultant for Procter & Gamble Company and Myriad Neuroscience. Dr Romanowicz receives grant funding from the Mayo Foundation Departmental Small Grant Program and the Palix Foundation. Drs Gandhi, Schak, Shekunov, Lynch, Rummans, Pease, and Desai and Mss Geske and Limbeck report no conflicts of interest related to the subject of this article.
Funding/support: This study was made possible using the resources of the Rochester Epidemiology Project, which is supported by the National Institute on Aging of the National Institutes of Health under award no. R01AG034676. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Role of the sponsor: The sponsor had no role in the design and conduct of the study; collection, management, analysis, and interpretation of data; or preparation, review, or approval of the manuscript.
Previous presentation: Presented at the 66th Annual Meeting of the American Academy of Child and Adolescent Psychiatry; Chicago, Illinois; October 14–19, 2019. Portions of this manuscript have been published in abstract form: J Am Acad Child Adolesc Psychiatry. 2019 Oct;58(10) suppl:S174–175.
Supplementary material: See accompanying pages.
References (26)

- Patrick SW, Schumacher RE, Benneyworth BD, et al. Neonatal abstinence syndrome and associated health care expenditures: United States, 2000–2009. JAMA. 2012;307(18):1934–1940. PubMed CrossRef
- Kocherlakota P. Neonatal abstinence syndrome. Pediatrics. 2014;134(2):e547–e561. PubMed CrossRef
- Irner TB, Teasdale TW, Nielsen T, et al. Substance use during pregnancy and postnatal outcomes. J Addict Dis. 2012;31(1):19–28. PubMed CrossRef
- Bakhireva LN, Shrestha S, Garrison L, et al. Prevalence of alcohol use in pregnant women with substance use disorder. Drug Alcohol Depend. 2018;187:305–310. PubMed CrossRef
- Heberlein A, Leggio L, Stichtenoth D, et al. The treatment of alcohol and opioid dependence in pregnant women. Curr Opin Psychiatry. 2012;25(6):559–564. PubMed CrossRef
- Kozhimannil KB, Graves AJ, Jarlenski M, et al. Non-medical opioid use and sources of opioids among pregnant and non-pregnant reproductive-aged women. Drug Alcohol Depend. 2017;174:201–208. PubMed CrossRef
- Roberts SC, Pies C. Complex calculations: how drug use during pregnancy becomes a barrier to prenatal care. Matern Child Health J. 2011;15(3):333–341. PubMed CrossRef
- Jones HE, Heil SH, Tuten M, et al. Cigarette smoking in opioid-dependent pregnant women: neonatal and maternal outcomes. Drug Alcohol Depend. 2013;131(3):271–277. PubMed CrossRef
- Rocca WA, Grossardt BR, Brue SM, et al. Data Resource Profile: Expansion of the Rochester Epidemiology Project medical records-linkage system (E-REP). Int J Epidemiol. 2018;47(2):368–368j. PubMed CrossRef
- Baer RJ, Chambers CD, Ryckman KK, et al. Risk of preterm and early term birth by maternal drug use. J Perinatol. 2019;39(2):286–294. PubMed CrossRef
- Ray JG, Park AL, Fell DB. Mortality in infants affected by preterm birth and severe small-for-gestational age birth weight. Pediatrics. 2017;140(6):e20171881. PubMed CrossRef
- Condò V, Cipriani S, Colnaghi M, et al. Neonatal respiratory distress syndrome: are risk factors the same in preterm and term infants? J Matern Fetal Neonatal Med. 2017;30(11):1267–1272. PubMed CrossRef
- Stoll BJ, Hansen N, Fanaroff AA, et al. Late-onset sepsis in very low birth weight neonates: the experience of the NICHD Neonatal Research Network. Pediatrics. 2002;110(2 pt 1):285–291. PubMed CrossRef
- Stoll BJ, Hansen NI, Bell EF, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics. 2010;126(3):443–456. PubMed CrossRef
- Chen J, Smith LE. Retinopathy of prematurity. Angiogenesis. 2007;10(2):133–140. PubMed CrossRef
- You J, Yang HJ, Hao MC, et al. Late preterm infants’ social competence, motor development, and cognition. Front Psychiatry. 2019;10:69. PubMed
- Franz AP, Bolat GU, Bolat H, et al. Attention-deficit/hyperactivity disorder and very preterm/very low birth weight: a meta-analysis. Pediatrics. 2018;141(1):e20171645. PubMed CrossRef
- Nosarti C, Reichenberg A, Murray RM, et al. Preterm birth and psychiatric disorders in young adult life. Arch Gen Psychiatry. 2012;69(6):E1–E8. PubMed CrossRef
- Corr TE, Schaefer EW, Paul IM. Growth during the first year in infants affected by neonatal abstinence syndrome. BMC Pediatr. 2018;18(1):343. PubMed CrossRef
- Jones HE, Fielder A. Neonatal abstinence syndrome: Historical perspective, current focus, future directions. Prev Med. 2015;80:12–17. PubMed CrossRef
- Bachhuber MA, Hennessy S, Cunningham CO, et al. Increasing benzodiazepine prescriptions and overdose mortality in the United States, 1996–2013. Am J Public Health. 2016;106(4):686–688. PubMed CrossRef
- Sun EC, Dixit A, Humphreys K, et al. Association between concurrent use of prescription opioids and benzodiazepines and overdose: retrospective analysis. BMJ. 2017;356:j760. PubMed CrossRef
- Quelhas D, Kompala C, Wittenbrink B, et al. The association between active tobacco use during pregnancy and growth outcomes of children under five years of age: a systematic review and meta-analysis. BMC Public Health. 2018;18(1):1372. PubMed CrossRef
- Choo RE, Huestis MA, Schroeder JR, et al. Neonatal abstinence syndrome in methadone-exposed infants is altered by level of prenatal tobacco exposure. Drug Alcohol Depend. 2004;75(3):253–260. PubMed CrossRef
- Hudak ML, Tan RC; Committee on Drugs; Committee on Fetus and Newborn; American Academy of Pediatrics. Neonatal drug withdrawal. Pediatrics. 2012;129(2):e540–e560. PubMed CrossRef
- Sutter MB, Leeman L, Hsi A. Neonatal opioid withdrawal syndrome. Obstet Gynecol Clin North Am. 2014;41(2):317–334. PubMed
Enjoy this premium PDF as part of your membership benefits!