ABSTRACT
Objective: To leverage electronic health record (EHR) data to explore the relationship between weight gain and antipsychotic adherence among patients with schizophrenia and bipolar disorder (BD).
Methods: EHR data were used to identify individuals with at least 60 days of continuous antipsychotic use between 2005 and 2019. Patients were diagnosed with schizophrenia, schizoaffective disorder, BD, or neither diagnosis (psychiatric controls). We examined the association of weight gain in the first 90 days with the proportion of days covered (PDC) with an antipsychotic and with the frequency of medication switching or stopping.
Results: We identified 590 adults with schizophrenia or schizoaffective disorder, 819 adults with BD, and 642 psychiatric controls. In the first 90 days, the percentages of patients with a PDC ≥ 0.80 were 76.8% (schizophrenia), 77.1% (BD), and 70.7% (controls). Logistic regression models revealed that weight gain of ≥ 7% trended toward being significantly associated with greater adherence in the first 90 days (odds ratio = 1.29, P = .077) and was significantly associated with an increased likelihood of a medication switch in the first 180 days (odds ratio = 1.60, P = .003).
Discussion: Patients whose weight increased by 7% or more in the first 90 days were more adherent but were also more likely to switch medications during the first 180 days.
Prim Care Companion CNS Disord 2023;25(2):22m03310
To cite: Perkins AJ, Khandker R, Overley A, et al. Association of antipsychotic-related weight gain with treatment adherence and switching using electronic medical records data. Prim Care Companion CNS Disord. 2023;25(2):22m03310.
To share: https://doi.org/10.4088/PCC.22m03310
© 2023 Physicians Postgraduate Press, Inc.
aDepartment of Biostatistics and Health Data Science, Indiana University School of Medicine, Indianapolis, Indiana
bMerck & Co., Inc., Kenilworth, New Jersey
cEskenazi Health, Indianapolis, Indiana
dDepartment of Psychiatry, Indiana University School of Medicine, Indianapolis, Indiana
eSolid Research Group, LLC, St Paul, Minnesota
fRegenstrief Institute Inc, Indianapolis, Indiana
gIndiana University School of Medicine, Indianapolis, Indiana
*Corresponding author: Anthony J. Perkins, MS, Department of Biostatistics and Health Data Science, Indiana University, 410 W 10th St, Indianapolis, IN 46202 ([email protected]).
Globally, more than 65 million people are living with schizophrenia or bipolar disorder (BD).1 These conditions carry with them substantial individual and societal burden as well as premature mortality.2 Several classes of psychotropic medications are available to reduce the burden of these severe mental illnesses.3,4 Nevertheless, approximately 56% of patients living with schizophrenia and 44% of those living with BD are nonadherent to their psychotropic medications.4 Nonadherence to psychotropic medications increases the burden of these and other psychiatric disorders at both the individual and societal level by leading to relapse and poor symptom management, acute psychiatric and medical emergency department (ED) utilization and reduced quality of life, and higher overall health care expenditures.3–10 Unsurprisingly, antipsychotic treatment nonadherence has been identified as one of the main causes for antipsychotic treatment failure.6 Identification of factors associated with psychotropic medication nonadherence among patients living with schizophrenia and BD is crucial to reduce the burden of these devastating brain-based disorders.
A variety of studies and reviews have attempted to identify and describe factors associated with nonadherence in schizophrenia and BD.3,4,11,12 Common factors cited in these studies and reviews include poor insight into the illness or treatment, comorbid substance abuse, negative attitude toward medication, and side effects. Patients cite a number of side effects, including dizziness, fatigue, sleepiness, insomnia, restlessness, and sexual difficulties.4 Multiple studies13–18 have also associated poor adherence with identified weight gain, obesity, or perceived weight status related to taking antipsychotics in both schizophrenia and BD. However, typically weight (and often adherence) is self-reported in these studies. A current challenge for research on treatment nonadherence is a lack of consensus definition of adherence as well as the inherent limitations of available methods for assessing adherence. Currently, no gold standard for defining or measuring medication adherence in this population exists.3,19,20 We know of no studies that use electronic health record (EHR) or health information exchange data to investigate the relationship between weight gain and medication adherence to antipsychotic medications.
Given the importance of adherence in schizophrenia and BD, and the role of weight gain associated with antipsychotics, we sought to leverage EHR data as an efficient and cost-effective tool to explore the relationship between weight gain and antipsychotic adherence in these conditions.
METHODS
Cohort Selection
We identified patients using EHR data from 2 health systems (Eskenazi Health and Indiana University Health) in Indiana. Eligible patients included those identified from EHR records as having 90 days of antipsychotic use (oral or long-acting injectables), based on medication order data, between 2005 and 2018. A list of antipsychotics considered is provided in Supplementary Table 1. For these patients, we identified all dates wherein antipsychotics were ordered or prescribed and identified the first one as the “first antipsychotic date.” Patients were required to be aged ≥ 12 years and to have at least one encounter per year for at least 2 years following their index date; prisoners were excluded from the cohort. Patients whose “first antipsychotic date” was in 2005 were excluded from the analysis, as they may have been taking antipsychotics prior to the period of interest. We then limited the cohort to patients aged ≥ 18 years with at least one 60-day period of continuous use of antipsychotics and defined the “treatment date” for each patient as either:
- The “first antipsychotic date” if the 60-day continuous use period began within 6 months after that date or
- The beginning of the 60-day continuous use period if that period began more than 6 months after the “first antipsychotic date.”
Patients without a diagnosis of schizophrenia or BD at any time during the study period were categorized as psychiatric controls. Patients with diagnoses of schizophrenia including schizoaffective disorder (hereafter referred together as schizophrenia) (ICD-9 295X or ICD-10 F20 or F25X) or BD (ICD-9 296.0X, 296.1X, 296.4X-6X, 296.7, 296.80, 296.89 or ICD-10 F31X but not F31.81) were categorized as such. Patients with diagnoses for both schizophrenia and BD were categorized based on their most recent diagnosis, as it may take time to identify the correct diagnosis for these patients. If the most recent encounter included diagnoses from both categories, the patients were excluded from our cohort.
IRB
This study was approved by the Institutional Review Board of Indiana University (IRB no. 2011632512).
Measures
For each patient, we collected age as of the treatment date, race, gender, and insurance status. Using information from EHR records, we identified comorbidities of interest, prior utilization, and previous medication use during the 1-year period prior to the treatment date. We defined adherence to antipsychotics using the proportion of days covered (PDC). The PDC is calculated by dividing the number of days the medication is available (from prescription dates) by the number of days in the period of interest. Patients with a PDC ≥ 0.80 were considered adherent. Adherence reflected adherence to any antipsychotic medication: if a patient switched medications during the study period, both medications contributed to the PDC calculation. In addition to adherence, we identified medication switches and medication stoppages during the first 6 months of treatment. A medication switch was defined as a new antipsychotic prescription that was different than the initial antipsychotic medication. Medication stoppage was defined as the lack of an antipsychotic medication order or fill data in the EHR after the date when the previous supply would have been depleted.
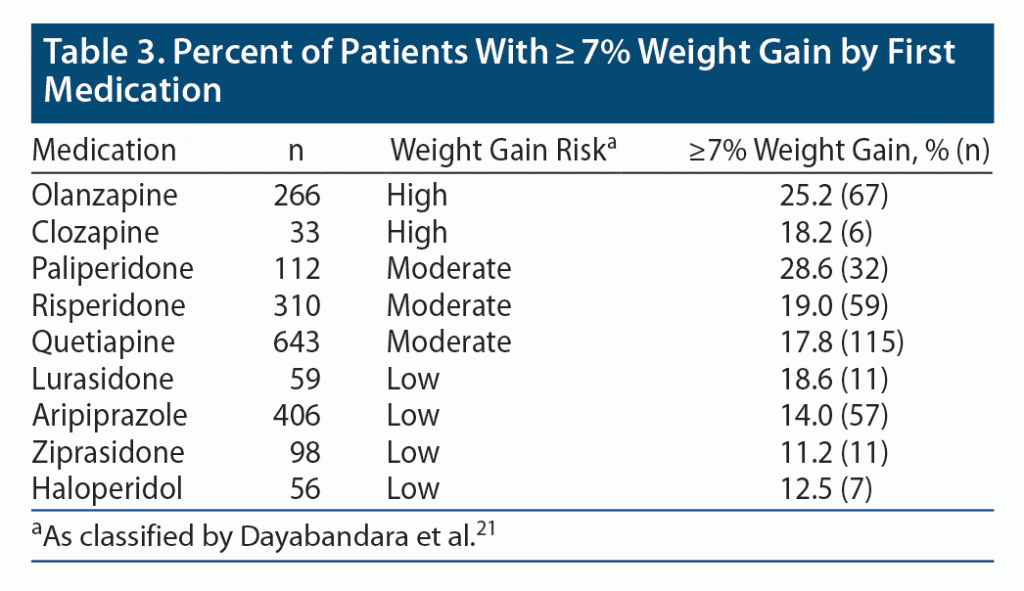
Weight gain was calculated as the difference between baseline weight and follow-up weight. Baseline weight was defined as the most recent weight recorded in the EHR prior to the treatment date, not more than 90 days prior to the treatment date. Follow-up weight was defined as the weight recorded in the EHR closest to the treatment date plus 90 days, not less than 60 days or more than 120 days after the treatment date. Significant weight gain was described as ≥ 7% increase in weight. Weight gain was also analyzed by therapeutic agent (for those with at least 30 patients) and the risk of weight gain associated with each agent as classified by Dayabandara et al.21
Statistical Analysis
We used χ2 tests and Kruskal-Wallis tests to compare demographic and clinical characteristics across the 3 cohorts (schizophrenia, BD, psychiatric controls). χ2 tests and Wilcoxon rank sum tests were used to compare clinical and demographic characteristics whether or not patients experienced significant weight gain. We used logistic regression to assess the relationship between weight gain and adherence (PDC ≥ 0.80), adjusting for demographic and clinical characteristics. A generalized logits model was used to model the association of weight gain with first medication use (continuance, switching, or stopping). Additional sensitivity analyses on the odds of switching or stopping were performed using alternative definitions of weight gain and antipsychotic start date.
RESULTS
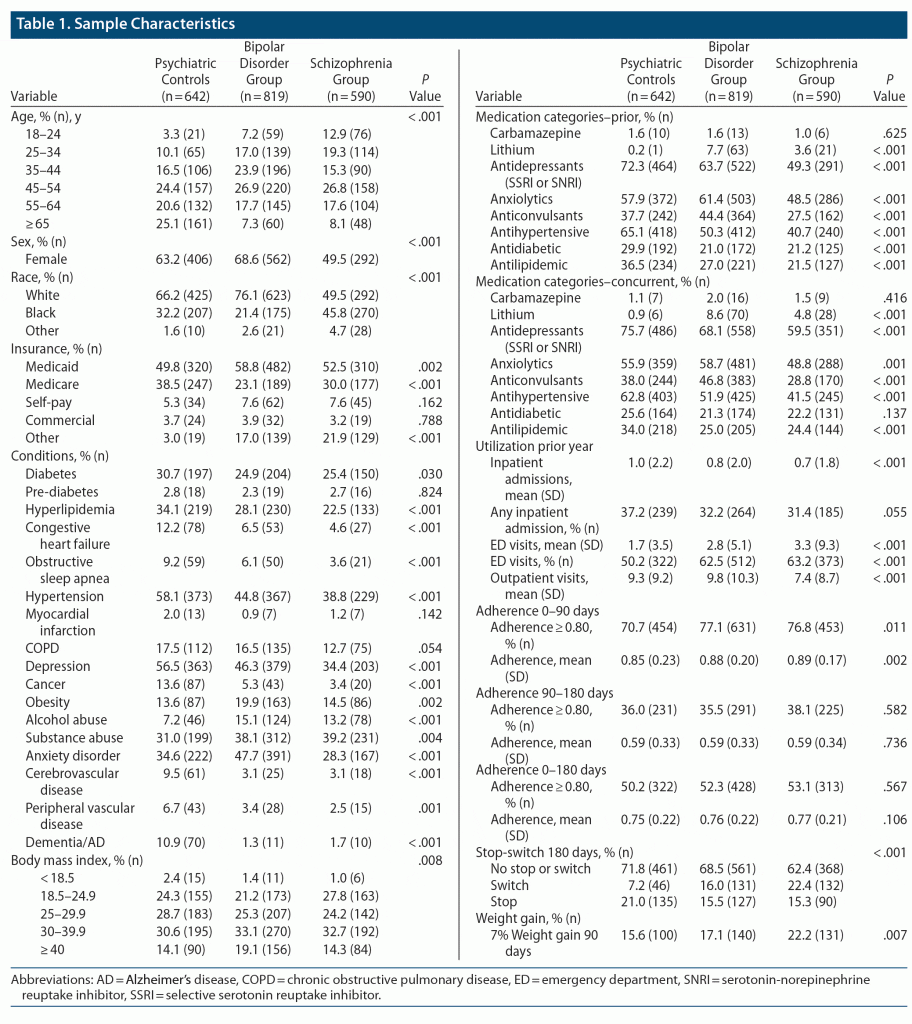
We identified a total of 7,012 individuals who met inclusion criteria regarding continuous antipsychotic use. There were 2,051 (29.3%) individuals who met our inclusion criteria when adding the requirement for multiple weights. Patients who did not have multiple weights were significantly younger and had less comorbidity than patients with multiple weights. Adherence between the 2 groups did not differ significantly. When classified by diagnosis, 28.8% were identified with schizophrenia, 39.9% were identified with BD, and 31.3% were psychiatric controls. Comparisons of the demographic and clinical characteristics by diagnostic cohort are presented in Table 1. Compared with the other cohorts, psychiatric controls were generally older and more likely to be enrolled in Medicare and to have chronic comorbid conditions, while schizophrenia patients were more likely to be Black and male. The BD participants were more often diagnosed with anxiety disorders compared to psychiatric controls and those with schizophrenia. Substance use disorders were significantly more common in the BD and schizophrenia groups than in psychiatric controls. Individuals with BD were more likely to be taking an anticonvulsant or anxiolytic the prior year. The percent of patients experiencing significant weight gain was highest in patients with schizophrenia (22.2%), followed by patients with BD (17.1%) and psychiatric controls (15.6%, P = .007). During the year prior to the treatment date, unsurprisingly, patients with schizophrenia and BD had more ED visits but fewer all-cause inpatient admissions than the psychiatric controls.
In the first 90 days after the treatment date, mean PDC ranged from 0.85 (psychiatric controls) to 0.89 (schizophrenia). During this period, the mean PDC and the percent with a PDC ≥ 0.80 were lower in psychiatric controls than the other cohorts, but there was no difference in mean PDC or the percent with a PDC ≥ 0.80 between 90 and 180 days. Patients with schizophrenia or BD were more likely to switch antipsychotic medications in the first 180 days, while psychiatric controls were more likely to stop taking antipsychotic medications.
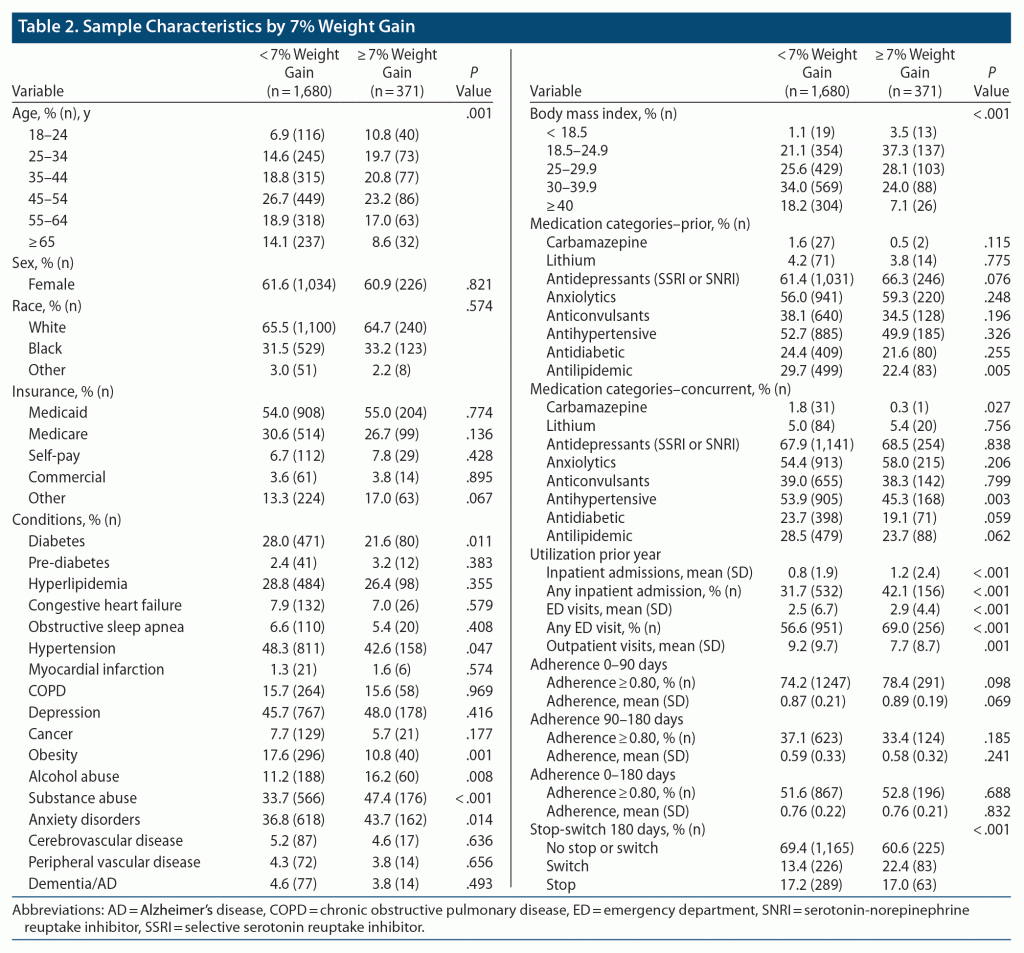
Comparison of demographic and clinical characteristics by significant weight gain are presented in Table 2. Patients who experienced significant weight gain were younger and more likely to have a diagnosis of alcohol abuse, substance abuse, or an anxiety disorder the year prior to treatment. They were also more likely to have more ED visits and inpatient admissions the prior year and were more likely to switch medications. Patients who did not experience a significant weight gain were more likely to have diabetes, hypertension, obesity, and a higher body mass index (BMI) at baseline. The percent of patients experiencing significant weight gain differed by therapeutic agent (Table 3). Medications classified as “low risk” had fewer patients experience significant weight gain (range, 11.2%–18.6% of patients) than did medications classified as either “moderate” (17.8%–28.6%) or “high risk” (18.2%–25.2%, Table 3).
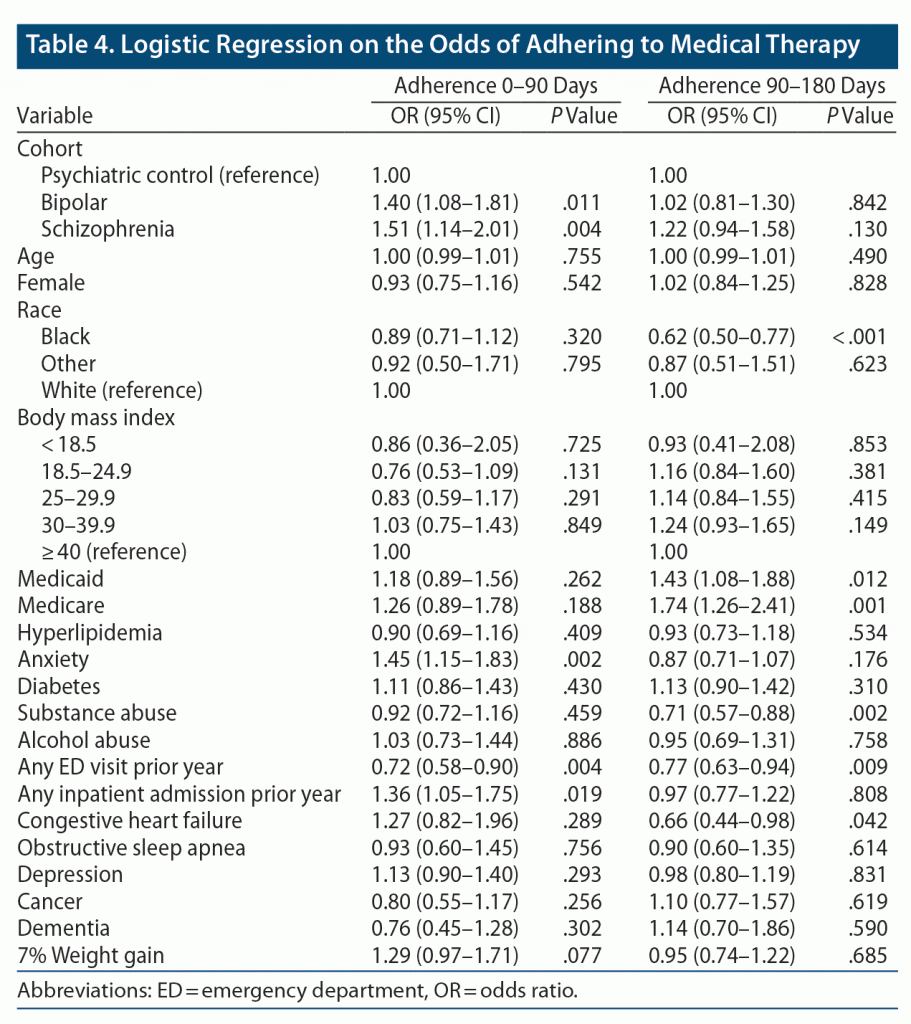
Logistic regressions on the odds of adherence (PDC ≥ 0.80) during the first 90 days and between 90 and 180 days identified several characteristics associated with adherence. Patients with BD and schizophrenia were more likely to be adherent in the first 90 days than psychiatric controls, even after adjustment (Table 4). Additionally, patients with significant weight gain tended to be more adherent (odds ratio [OR] = 1.29; 95% CI, 0.97–1.71; P = .077) in the first 90 days. However, significant weight gain was not associated with adherence from 90 to 180 days. Age, gender, and race were not significantly associated with adherence for either time period, with the exception that Black patients were less likely to be adherent from 90 to 180 days. A comorbidity of anxiety was associated with greater adherence during the first 90 days (OR = 1.45, P = .002), while substance abuse (OR = 0.71, P = .002) and heart failure (OR = 0.66, P = .042) were associated with lower adherence during days 90 to 180.
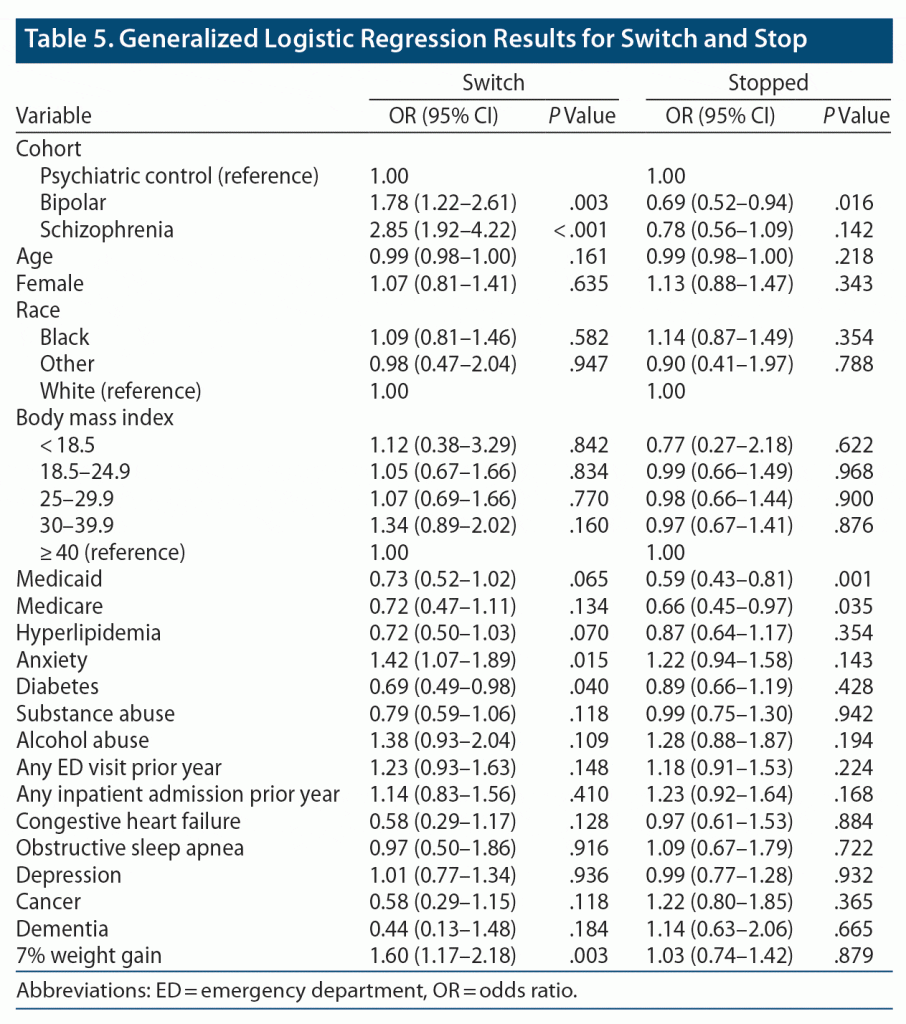
After adjustment, patients with BD or schizophrenia were more likely to switch antipsychotic medications than psychiatric controls, and patients with BD were less likely to stop antipsychotic medications than psychiatric controls (Table 5). Additionally, significant weight gain significantly increased the likelihood of a medication switch in the first 180 days (OR = 1.60; 95% CI, 1.17–2.18; P = .003). Results were similar when performing sensitivity analyses using varying weight calculations and treatment date restrictions (data not shown).
DISCUSSION
Using information from EHRs, we observed considerable frequencies of weight gain among antipsychotic users, even among those whose treatment was carrying “low weight gain risk” (Table 3). Among patients taking antipsychotics, those with significant weight gain were more adherent during the same period (the first 90 days). However, significant weight gain during the first 90 days was associated with a higher likelihood of switching antipsychotics during the first 180 days. Sensitivity analyses using different definitions of weight gain produce similar results. This finding is consistent with the recommendation of major treatment guidelines to consider switching antipsychotic treatment when significant weight gain occurs.22 It appears patients with BD and schizophrenia who benefit from the medications from a psychiatric perspective stayed on them, possibly in part via an anxiolytic effect, and therefore gained weight. Additionally, observed weight gain by therapeutic agent was consistent with the risk of weight gain previously reported.21 This pattern was not observed in the psychiatric control group, and we speculate that this group may not have functionally benefited from symptom control as robustly as those in the BD and schizophrenia groups. We note that comorbid substance use disorders also may have played a part in derailing compliance, particularly in the period of 90–180 days. In the current study, we chose to look at weight gain during the first 90 days of treatment. It has been shown the most significant weight gain often occurs in the first 6 to 12 weeks of treatment,23,24 although most antipsychotic agents have been linked to weight gain in both the short and long term.25,26
Previous studies have reported a negative association between weight gain and adherence to antipsychotics. A review from 201712 identified 3 studies (2 from the US13,14 and 1 from China18) reporting weight gain as a reason for nonadherence. In all 3 studies, reasons for nonadherence were investigated retrospectively; patients who were nonadherent reported reasons, and in each case included weight gain as a reason. None prospectively tracked weight gain and adherence simultaneously, nor was adherence measured as it is in the current study, but instead was self-reported. Additionally, the perception of weight gain was the key indicator in the Chinese study,18 although approximately half of participants experienced weight gain of 7% or more. As nicely summarized by Velligan et al,12(p461) “a perceived but not actual overweight status was associated with poor adherence.” In that same study,12 among those who perceived themselves as overweight and believed their antipsychotics were responsible, 72% reduced their dosages on their own.18
Switching antipsychotics is often advised when the current medication produces an inadequate clinical response or results in unacceptable side effects.27 Unfortunately, there is little guidance for when to switch therapy versus increase the dose of the current medication.28 Stopping therapy (under the supervision of a physician) is typically only justified if a patient has made a reasonable improvement and remained well for an extended period of time.27 Reviewing the risks of treatment discontinuation and assuring patients that viable alternatives will be sought if side effects become intolerable may help to mitigate the risk of patients stopping therapy on their own. Patients who intentionally stop taking their medication or who take less than prescribed may do so for a variety of reasons, including a negative attitude toward medication, side effects, or stigma.12 Patient-initiated discontinuation is not uncommon. An analysis of California Medicaid patients observed that 19% of participants “abandoned” their antipsychotic treatment within 6 months; this was approximately the same percentage who switched medications (20.9%).29 A review of 4 studies ranging in length from 24 weeks to 6 months observed that 53% of participants (schizophrenia or related disorder) stopped taking the prescribed antipsychotic before the end of the study.30
The consequences of poor adherence or stopping antipsychotic medication can be severe. Relapse of schizophrenia, which occurs within a year in as many as 80% of patients who discontinue their medication,31 can result in significant health care utilization and costs.32 Similarly, with BD, nonadherence to treatment increases symptoms, health care utilization, and suicides.33 However, weight gain among treated patients can lead to metabolic abnormalities (eg, high blood pressure, elevated triglyceride levels), which can increase the risk of type 2 diabetes and cardiovascular disease.34,35 Antipsychotic medication that minimized weight gain could reduce nonadherence, switching, or discontinuation while potentially mitigating the risk of adverse cardiometabolic outcomes associated with weight gain.
Limitations
While this study represents one of the first, to our knowledge, that leverages EHR data to examine the association between weight gain and antipsychotic adherence, our results should be viewed in light of some limitations. First, PDC is an inexact surrogate for medication adherence, and, therefore, we cannot know for certain whether study patients are truly adherent or not. It is possible that EHR data can misclassify adherence if, for instance, medication data were unavailable from the medical record or if patients experienced a long-term inpatient hospitalization (psychiatric or otherwise). Our algorithm allows patients to switch medications and remain adherent if their PDC across therapies is ≥ 0.80; given that weight gain may be a reason for switching, we also examined switching by weight gain status. Second, we did not examine changes in weight longitudinally; it is possible that some weight gain may be regression to the mean for patients who experienced significant weight loss prior to the start of therapy. Third, we did not examine weight gain by starting BMI. Some weight gain may be beneficial for patients with a low BMI, while those with a high BMI at baseline may be less likely to achieve the threshold of “significant” weight gain simply because it required more absolute change in weight. We observed that patients without significant weight gain had a higher baseline BMI and were more likely to be obese. Also, our study includes only antipsychotic-naive patients, who are known to be more susceptible to weight gain.23 Therefore, the results may not be generalizable to all patients receiving antipsychotics. Finally, this study does not consider antipsychotic dose, which may impact the results.
CONCLUSION
In summary, we observed that patients with significant weight gain were more adherent in the first 90 days and more likely to switch medications in the first 180 days. The association of weight gain to adherence during the same period may support the hypothesis that adherent people will likely gain weight. However, there is some evidence that significant weight gain may lead to switching from one antipsychotic to another. Despite several known limitations of EHR as a mechanism for assessing adherence, the demonstration of these expected associations supports the possibility that EHR may represent a promising tool to help associate adherence and other available data. This work suggests that efforts to mitigate the effects of weight gain are essential, given that weight gain is so tightly linked to efficacy and compliance with antipsychotic medication for patients with BD and schizophrenia.
Submitted: April 21, 2022; accepted August 8, 2022.
Published online: March 9, 2023.
Relevant financial relationships: Drs Khandker and Chekani are employees of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, New Jersey, who may own stock and/or hold stock options in Merck & Co., Inc., Kenilworth, New Jersey. Dr Solid receives consulting fees from Regenstrief. Drs Overly, Dexter, Boustani, and Hulvershorn; Mr Perkins; and Ms Roberts report no relevant financial relationships related to the subject of this article.
Funding/support: This study was funded by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, New Jersey.
Role of the sponsor: The funder of this study participated in study design, data collection, data analysis, data interpretation, and writing of the report. All authors had full access to all the data in the study and approved the decision to submit for publication.
Previous presentation: Results presented as a poster at the Professional Society for Health Economics and Outcomes Research (ISPOR) conference; Washington, DC; May 15, 2022.
Supplementary material: See accompanying pages.
Clinical Points
- A considerable amount of weight gain was observed among antipsychotic-naive users, even among those whose treatment was considered to have a low risk of weight gain.
- Significant weight gain was associated with increased likelihood of switching medications.
- It is important to manage early weight gain associated with antipsychotic medications to achieve long-term adherence.
References (35)

- GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1789–1858. PubMed CrossRef
- Laursen TM, Nordentoft M, Mortensen PB. Excess early mortality in schizophrenia. Annu Rev Clin Psychol. 2014;10(1):425–448. PubMed CrossRef
- El Abdellati K, De Picker L, Morrens M. Antipsychotic treatment failure: a systematic review on risk factors and interventions for treatment adherence in psychosis. Front Neurosci. 2020;14:531763. PubMed CrossRef
- Semahegn A, Torpey K, Manu A, et al. Psychotropic medication non-adherence and its associated factors among patients with major psychiatric disorders: a systematic review and meta-analysis. Syst Rev. 2020;9(1):17. PubMed CrossRef
- Burton WN, Chen CY, Conti DJ, et al. The association of antidepressant medication adherence with employee disability absences. Am J Manag Care. 2007;13(2):105–112. PubMed
- Goff DC, Hill M, Freudenreich O. Strategies for improving treatment adherence in schizophrenia and schizoaffective disorder. J Clin Psychiatry. 2010;71(suppl 2):20–26. PubMed CrossRef
- Higashi K, Medic G, Littlewood KJ, et al. Medication adherence in schizophrenia: factors influencing adherence and consequences of nonadherence, a systematic literature review. Ther Adv Psychopharmacol. 2013;3(4):200–218. PubMed CrossRef
- Lindström E, Bingefors K. Patient compliance with drug therapy in schizophrenia. Economic and clinical issues. Pharmacoeconomics. 2000;18(2):105–124. PubMed CrossRef
- McCutcheon R, Beck K, Bloomfield MA, et al. Treatment resistant or resistant to treatment? antipsychotic plasma levels in patients with poorly controlled psychotic symptoms. J Psychopharmacol. 2015;29(8):892–897. PubMed CrossRef
- Olivares JM, Sermon J, Hemels M, et al. Definitions and drivers of relapse in patients with schizophrenia: a systematic literature review. Ann Gen Psychiatry. 2013;12(1):32. PubMed CrossRef
- Lacro JP, Dunn LB, Dolder CR, et al. Prevalence of and risk factors for medication nonadherence in patients with schizophrenia: a comprehensive review of recent literature. J Clin Psychiatry. 2002;63(10):892–909. PubMed CrossRef
- Velligan DI, Sajatovic M, Hatch A, et al. Why do psychiatric patients stop antipsychotic medication? A systematic review of reasons for nonadherence to medication in patients with serious mental illness. Patient Prefer Adherence. 2017;11:449–468. PubMed CrossRef
- Baldessarini RJ, Perry R, Pike J. Factors associated with treatment nonadherence among US bipolar disorder patients. Hum Psychopharmacol. 2008;23(2):95–105. PubMed CrossRef
- DiBonaventura M, Gabriel S, Dupclay L, et al. A patient perspective of the impact of medication side effects on adherence: results of a cross-sectional nationwide survey of patients with schizophrenia. BMC Psychiatry. 2012;12(1):20. PubMed CrossRef
- Johnson FR, Ozdemir S, Manjunath R, et al. Factors that affect adherence to bipolar disorder treatments: a stated-preference approach. Med Care. 2007;45(6):545–552. PubMed CrossRef
- Kikkert MJ, Schene AH, Koeter MW, et al. Medication adherence in schizophrenia: exploring patients’, carers’ and professionals’ views. Schizophr Bull. 2006;32(4):786–794. PubMed CrossRef
- Weiden PJ, Mackell JA, McDonnell DD. Obesity as a risk factor for antipsychotic noncompliance. Schizophr Res. 2004;66(1):51–57. PubMed CrossRef
- Wong MM, Chen EY, Lui SS, et al. Medication adherence and subjective weight perception in patients with first-episode psychotic disorder. Clin Schizophr Relat Psychoses. 2011;5(3):135–141. PubMed CrossRef
- García S, Martínez-Cengotitabengoa M, López-Zurbano S, et al. Adherence to antipsychotic medication in bipolar disorder and schizophrenic patients: a systematic review. J Clin Psychopharmacol. 2016;36(4):355–371. PubMed CrossRef
- Prieto-Merino D, Mulick A, Armstrong C, et al. Estimating proportion of days covered (PDC) using real-world online medicine suppliers’ datasets. J Pharm Policy Pract. 2021;14(1):113. PubMed CrossRef
- Dayabandara M, Hanwella R, Ratnatunga S, et al. Antipsychotic-associated weight gain: management strategies and impact on treatment adherence. Neuropsychiatr Dis Treat. 2017;13:2231–2241. PubMed CrossRef
- American Diabetes Association; American Psychiatric Association; American Association of Clinical Endocrinologists; North American Association for the Study of Obesity. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;27(2):596–601. PubMed CrossRef
- Bak M, Fransen A, Janssen J, et al. Almost all antipsychotics result in weight gain: a meta-analysis. PLoS One. 2014;9(4):e94112. PubMed CrossRef
- Bretler T, Weisberg H, Koren O, et al. The effects of antipsychotic medications on microbiome and weight gain in children and adolescents. BMC Med. 2019;17(1):112. PubMed CrossRef
- Bushe CJ, Slooff CJ, Haddad PM, et al. Weight change from 3-year observational data: findings from the worldwide schizophrenia outpatient health outcomes database. J Clin Psychiatry. 2012;73(6):e749–e755. PubMed CrossRef
- Tek C, Kucukgoncu S, Guloksuz S, et al. Antipsychotic-induced weight gain in first-episode psychosis patients: a meta-analysis of differential effects of antipsychotic medications. Early Interv Psychiatry. 2016;10(3):193–202. PubMed CrossRef
- Keks N, Schwartz D, Hope J. Stopping and switching antipsychotic drugs. Aust Prescr. 2019;42(5):152–157. PubMed CrossRef
- Samara MT, Klupp E, Helfer B, et al. Increasing antipsychotic dose versus switching antipsychotic for non response in schizophrenia. Cochrane Database Syst Rev. 2018;5(5):CD011884. PubMed CrossRef
- Noordsy DL, Phillips GA, Ball DE, et al. Antipsychotic adherence, switching, and health care service utilization among Medicaid recipients with schizophrenia. Patient Prefer Adherence. 2010;4:263–271. PubMed CrossRef
- Liu-Seifert H, Adams DH, Kinon BJ. Discontinuation of treatment of schizophrenic patients is driven by poor symptom response: a pooled post-hoc analysis of four atypical antipsychotic drugs. BMC Med. 2005;3(1):21. PubMed CrossRef
- Emsley R, Chiliza B, Asmal L, et al. The nature of relapse in schizophrenia. BMC Psychiatry. 2013;13(1):50. PubMed CrossRef
- Ascher-Svanum H, Zhu B, Faries DE, et al. The cost of relapse and the predictors of relapse in the treatment of schizophrenia. BMC Psychiatry. 2010;10(1):2. PubMed CrossRef
- Busby KK, Sajatovic M. REVIEW: Patient, treatment, and systems-level factors in bipolar disorder nonadherence: A summary of the literature. CNS Neurosci Ther. 2010;16(5):308–315. PubMed CrossRef
- De Hert M, Detraux J, van Winkel R, et al. Metabolic and cardiovascular adverse effects associated with antipsychotic drugs. Nat Rev Endocrinol. 2011;8(2):114–126. PubMed CrossRef
- Mitchell AJ, Vancampfort D, Sweers K, et al. Prevalence of metabolic syndrome and metabolic abnormalities in schizophrenia and related disorders–a systematic review and meta-analysis. Schizophr Bull. 2013;39(2):306–318. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!