Background: Recent studies suggest that risperidone long-acting injection (RLAI) may be considered for controlling mood episodes in bipolar disorder patients who have relapsed due to medication nonadherence or failure to respond to standard therapies. Currently, no study has reported the usefulness of RLAI in youths with bipolar disorder. The aim of this study was to evaluate short-term effects of RLAI in the naturalistic treatment of early-onset bipolar disorder and its role in symptomatic remission and adherence to treatment.
Method: Nineteen early-onset bipolar disorder outpatients receiving RLAI were observed in a 6-month naturalistic study at the outpatient clinic of the Child and Adolescent Affective Disorders Program at the Institute of Psychiatry of the University of S×£o Paulo, S×£o Paulo, Brazil. All patients met DSM-IV criteria for bipolar disorder. Clinical response to RLAI was evaluated using the Children’s Global Assessment Scale (CGAS) and Clinical Global Impressions scale (CGI) across 3 time periods: index time (T0), 8 weeks after (T1), and 24 weeks after (T2). These subjects were recruited from May 2008 to December 2009.
Results: Patients receiving RLAI presented considerable improvement in global functioning (CGAS: T0 = 20.6; T1 = 42.9; and T2 = 49.2) and clinical severity (CGI: T0 = 5.9; T1 = 3.9; and T2 = 3.4). Global CGI mean scores of clinical improvement were 2.2 at T1 and 2.4 at T2. There were no significant changes in laboratory measurements and weight throughout follow-up.
Conclusions: RLAI was shown to be an alternative treatment for youths with bipolar disorder failing to respond to prior medication trials or with adherence problems. Further blind, randomized controlled studies are necessary to confirm these initial findings.
Trial registration: Sistema Nacional de InformaçÅes Sobre ׉tica em Pesquisa Envolvendo Seres Humanos-Commis×£o Nacional de ׉tica em Pesquisa identifier: CAAE 0709.0.015.000-06
Prim Care Companion CNS Disord 2013;15(3):doi:10.4088/PCC.12m01368
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: February 23, 2012; accepted May 11, 2012 (doi:10.4088/PCC.12m01368).
Published online: May 2, 2013.
Corresponding author: Miguel A. Boarati, MD, Childhood and Adolescence Affective Disorders Program, Institute and Department of Psychiatry, School of Medicine of the University of S×£o Paulo, R. Dr Ov×dio Pires de Campos, 785, 3°. andar, ala norte, sala 12, CEP 01060-970, S×£o Paulo—SP, Brazil ([email protected]).
Six-Month Open-Label Follow-Up of Risperidone
Long-Acting Injection Use in Pediatric Bipolar Disorder
ABSTRACT
Background: Recent studies suggest that risperidone long-acting injection (RLAI) may be considered for controlling mood episodes in bipolar disorder patients who have relapsed due to medication nonadherence or failure to respond to standard therapies. Currently, no study has reported the usefulness of RLAI in youths with bipolar disorder. The aim of this study was to evaluate short-term effects of RLAI in the naturalistic treatment of early-onset bipolar disorder and its role in symptomatic remission and adherence to treatment.
Method: Nineteen early-onset bipolar disorder outpatients receiving RLAI were observed in a 6-month naturalistic study at the outpatient clinic of the Child and Adolescent Affective Disorders Program at the Institute of Psychiatry of the University of São Paulo, São Paulo, Brazil. All patients met DSM-IV criteria for bipolar disorder. Clinical response to RLAI was evaluated using the Children’s Global Assessment Scale (CGAS) and Clinical Global Impressions scale (CGI) across 3 time periods: index time (T0), 8 weeks after (T1), and 24 weeks after (T2). These subjects were recruited from May 2008 to December 2009.
Results: Patients receiving RLAI presented considerable improvement in global functioning (CGAS: T0 = 20.6; T1 = 42.9; and T2 = 49.2) and clinical severity (CGI: T0 = 5.9; T1 = 3.9; and T2 = 3.4). Global CGI mean scores of clinical improvement were 2.2 at T1 and 2.4 at T2. There were no significant changes in laboratory measurements and weight throughout follow-up.
Conclusions: RLAI was shown to be an alternative treatment for youths with bipolar disorder failing to respond to prior medication trials or with adherence problems. Further blind, randomized controlled studies are necessary to confirm these initial findings.
Trial registration: Sistema Nacional de Informaçōes Sobre Ética em Pesquisa Envolvendo Seres Humanos-Commisão Nacional de Ética em Pesquisa identifier: CAAE 0709.0.015.000-06
Prim Care Companion CNS Disord 2013;15(3):doi:10.4088/PCC.12m01368
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: February 23, 2012; accepted May 11, 2012 (doi:10.4088/PCC.12m01368).
Published online: May 2, 2013.
Corresponding author: Miguel A. Boarati, MD, Childhood and Adolescence Affective Disorders Program, Institute and Department of Psychiatry, School of Medicine of the University of São Paulo, R. Dr Ovídio Pires de Campos, 785, 3°. andar, ala norte, sala 12, CEP 01060-970, São Paulo—SP, Brazil ([email protected]).
Recent studies have demonstrated that long-acting formulations of second-generation antipsychotics may be considered for controlling mood episodes in adults with bipolar disorder who have relapsed due to medication nonadherence or failed to respond to standard therapies.1,2 Researchers suggest that the depot formulation offers the advantages of the atypical antipsychotics as well as stabilization of serum drug level.3
Psychiatric misdiagnosis, changeable atypical picture, and adverse pharmacologic response are common situations that render pediatric bipolar disorder more cumbersome to manage. One of the major challenges for practitioners is tracking repeated medication nonadherence of children and adolescents with bipolar disorder; such nonadherence can impede the treatment response and lead to an unsatisfactory outcome.4
Risperidone long-acting injection (RLAI) is the first long-acting atypical antipsychotic agent to become available as a treatment option. Its safety and maintenance effectiveness have been demonstrated through clinical trials in adults with stable bipolar disorder,5,6 and Fu-I and colleagues7 reported 3 cases of youths with bipolar disorder who had adherence problems that benefited from the use of RLAI. Although the US Food Drug and Administration has approved oral risperidone for use in pediatric bipolar disorder patients,8 few studies have reported the usefulness of RLAI in youths with bipolar disorder.
The present study aims to describe the effects of RLAI as a short-term treatment agent in patients with severe bipolar disorder symptomatology and nonadherence to prescribed oral medications. The tolerability, side effects, and 6-month mood-stabilization effects of RLAI are also reported.
METHOD
The target population was drawn from an extensive pediatric phenomenology study funded by the São Paulo Research Support Foundation, São Paulo, Brazil. The trial was registered at Sistema Nacional de Informaçōes Sobre Ética em Pesquisa Envolvendo Seres Humanos-Commisão Nacional de Ética em Pesquisa (identifier: CAAE 0709.0.015.000-06). Institutional review boards at the respective institutions approved the data collection as part of a clinical research registry.
The subjects of the present study were children and adolescents with bipolar disorder meeting criteria for nonadherence or therapeutic failure in the Children and Adolescents Affective Disorder Program’s outpatient clinic at the Institute of Psychiatry of the University of São Paulo, São Paulo, Brazil. These subjects were recruited from May 2008 to December 2009.
Subjects’ current bipolar disorder diagnoses were ascertained by 2 child and adolescent psychiatrists (M.A.B. and A.P.F-M.) in face-to-face clinical interviews and 2 structured interviews: the Child Behavior Check List and the Diagnostic Interview for Children and Adolescents10 were applied. In addition, all psychiatric comorbidities were assessed, and clinical records were routinely reviewed. The final best-estimate diagnosis was reached after a consensus panel meeting between research staff and the senior psychiatrist (L.F.-I.).
The Children’s Global Assessment Scale (CGAS)11,12 and Clinical Global Impressions,13 both Severity of Illness (CGI-S) and Improvement (CGI-I) scales, were periodically used as guidance to assess global functioning, clinical improvement, and treatment response (eg, change of doses, combination with other medications, or need for suspending RLAI).
Weight gain, extrapyramidal symptoms, and adverse events were closely monitored in each clinical visit, and the Systematic Assessment for Treatment Emergent Effects scale14 was applied to document the presence and the intensity of side effects of RLAI.
The final sample was composed of children and adolescents who (1) fulfilled Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, criteria for bipolar I disorder, bipolar II disorder, or bipolar disorder not otherwise specified (NOS); (2) were currently presenting severe pediatric bipolar disorder symptomatology (CGI-S > 5) with severe functional impairment (CGAS < 31); and (3) had previously used an oral formulation of risperidone with good tolerability and nonadherence to prescribed oral medications. In this study, nonadherence was defined as the failure to follow the recommended time schedules (eg, missing doses, not taking medication according to the recommended time schedule, and lack of adequate supervision by caregivers).
The exclusion criteria were (1) comorbidity with pervasive developmental disorders, schizophrenia, or mental retardation (IQ < 70); (2) reported past hypersensitivity, intolerance, or clinical worsening with the use of risperidone oral formulation; (3) previous history of severe extrapyramidal symptoms or neuroleptic malignant syndrome; (4) reported past intolerance to intramuscular injection; and (5) current use of depot antipsychotics.
Laboratory workup (blood count and kidney and liver functions, thyroid hormones, blood glucose, lipids, and prolactin), electroencephalogram, and electrocardiogram were routinely performed before starting medication or at baseline. Periodic biochemical tests were repeated each 3 months for monitoring metabolic and hormonal profiles. Patients were evaluated initially every 2 weeks. From the second month of treatment on, the evaluation was performed every 1 or 2 months, depending on the stabilization of the patients’ clinical picture. A consultation could be arranged at any time in cases of clinical worsening or side effects of medication.
A subject was considered a dropout when the patient failed to receive 2 consecutive injections of RLAI. Good acceptability of treatment was considered if the patient or family did not hinder the use of RLAI. However, if any resistance to the medication was detected, our RLAI application–trained nurse provided specific orientation about treatment benefits (eg, clinical improvement and not taking medication every day). No refusal occurred after this approach was initiated.
All participants and responsible persons were extensively informed about the proposed treatment and possible side effects and adverse events of RLAI, as well as the similarity of the oral risperidone with depot preparation. Patients were specifically warned about the possibility of new adverse events emerging during the use of RLAI and encouraged to contact emergency medical services and to communicate side effects. The treatment started only after patients and their legal guardians signed informed consent forms. The institutional ethics board authorized the prescription of RLAI for these patients fulfilling conditions described above. The Institute of Psychiatry at the University of Sao Paulo supported the RLAI treatment of all cases.
During the observation period, 20 children and adolescents met the inclusion criteria at the index time. One boy could not complete the protocol due to his parent’s inability to attend periodic evaluations. The final sample (N = 19) was composed of 16 boys and 3 girls, with a mean age of 12.1 years (standard deviation [SD] = 2.2). The mean age at onset of the first affective episode was 6.9 years (SD = 3.3), and the mean age at onset of the first manic episode was 8.0 years (SD = 3). Most of the subjects were diagnosed with bipolar I disorder (n = 17, 89.5%) and 2 (10.5%) patients with bipolar disorder NOS. Eighteen participants reported familial history of psychiatric disorders (94.7%).
In this sample, all subjects were severely impaired (initial mean CGAS = 20.6, SD = 9.7; and initial mean CGI-S = 5.9 SD = 1.0), and 6 patients (31.6%) had been previously hospitalized on psychiatric wards. Nine patients (47.4%) presented 1 or more psychiatric comorbidities, and 4 (21.1%) had attention-deficit/hyperactive disorder (ADHD), 2 (10.5%) had Tourette syndrome, 2 (10.5%) were diagnosed with conduct disorder, and 1 had enuresis (5.3%).
All patients had previously tried psychopharmacologic treatments and had used a mean of 8.6 (SD = 4.2) different types of medications throughout their treatments. Among the final sample, 11 patients (57.9%) were included in this study for adherence problems, and the most common reasons for nonadherence were inadequate supervision by caregivers (eg, low parental support and failure of the family in monitoring medical treatment) and difficulties inherent to bipolar disorder (eg, patient’s refusal to take medication). Eight patients (42.1%) were included because they failed to respond to other therapeutic regimens.
Only 3 patients (15.8%) simultaneously took another medication while taking RLAI. One patient used carbamazepine as a mood stabilizer and chlorpromazine for adjunctive anxiety control, 1 took chlorpromazine as an adjunctive to address sleep complaints, and 1 took divalproex as a mood stabilizer and methylphenidate for ADHD when starting RLAI. The subjects continued using these drugs after initiation of RLAI. No other drugs were associated with controlling mood symptoms or adverse effects during the period of observation for any other patients.
Data Analysis
Three time periods were considered for the data analysis.
- Time 0 (T0) or baseline was the occasion of the first RLAI injection. On this occasion, most patients were still using oral risperidone and/or other medications. At T0, patients were evaluated and laboratory tests were performed.
- Time 1 (T1) or the short-term treatment endpoint, ie, 2 months after the beginning of treatment. At this point, patients who had significant global functioning improvement (CGAS > 31) or clinical severity improvement (CGI-S < 3) and preferred to return to the oral formulation treatment could leave the protocol. The patients who did not improve significantly could also change their therapeutic regimen.
- Time 2 (T2) or the long-term treatment endpoint,
ie, 6 months after beginning treatment, was the point to observe the maintenance of improvement and tolerability of RLAI for a long-term period.
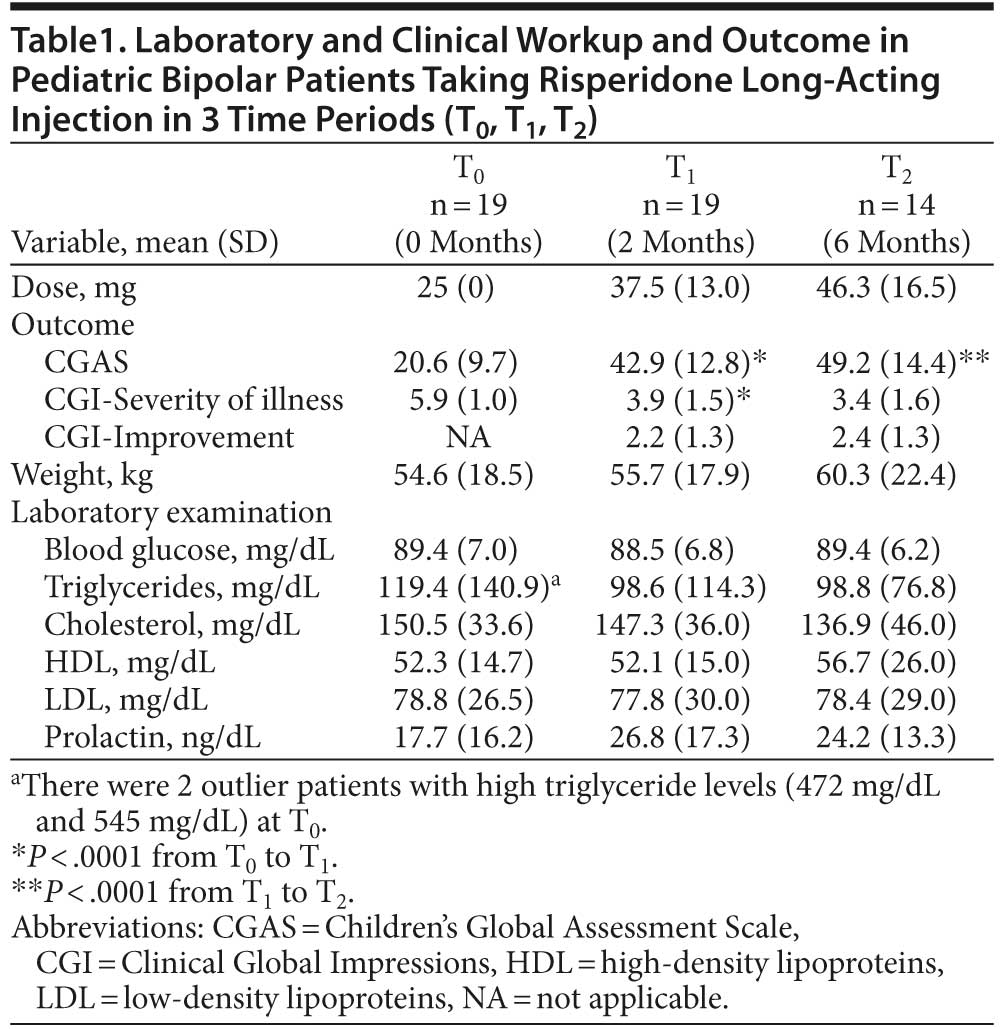
The initial dose of RLAI was 25 mg (1 ampoule) every 15 days. The patients continued to take the oral dose for 3 weeks after beginning the RLAI. At T1, the mean dose administered was 37.5 mg and at T2 the mean dose administered was 46.3 mg per injection (Table 1).
Statistical Methods
Descriptive analyses of means, SDs, frequencies, and percentages were derived from data. Paired-sample t test within-subject measure of time (T0, T1, and T2) was used to examine response status, main effects, and interactions between factors. Associations were examined between several demographic and clinical factors in the 3 different periods using Fisher exact tests. Analysis was performed using SPSS for Windows, version 17.0 (IBM, Armonk, New York).15
RESULTS
Short-Term Treatment
All 19 patients completed the short-term treatment (T0 to T1). During this period, global functioning and symptom severity improvement were shown by the significant changes in CGAS (20.6 vs 42.9, P < .0001) and CGI-S scores (5.9 vs 3.9, P < .0001). The mean CGI-I score was 2.2 at this endpoint (Table 1).
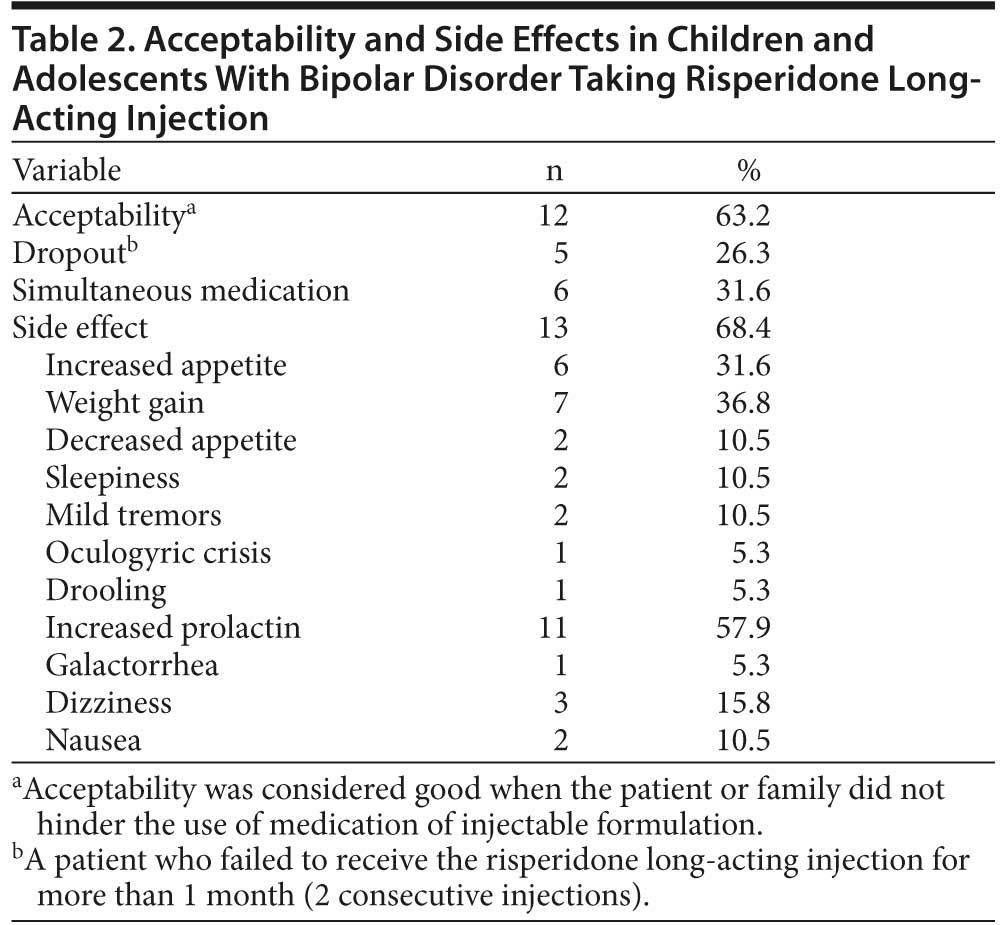
After completing the short-term treatment, 5 patients left the protocol. Three patients left the study after deciding to return to the oral formulation, 1 moved away and was referred to another treatment center, and 1 patient stopped RLAI due to the lack of significant improvement. No one demonstrated worsening of clinical symptoms (Table 2).
Long-Term Treatment Endpoint
Fourteen patients completed the long-term treatment, from T1 to T2 or 6 months after T0. The trend of sustained improvement is reflected in the change in the CGAS mean score from T1 to T2 (42.9 vs 49.2, P < .0001). The steady improvement from T1 to T2 was shown in similar mean scores of the CGI-S (3.9 vs 3.4, P = .21) and CGI-I (2.2 vs 2.4, P = .834) (Table 1).
Tolerability and Side Effects
The mean weight gain was 1.1 kg (SD = 0.6) at T1 and 5.7 kg (SD = 4.1) at T2. Since most patients had been taking oral antipsychotic medications before starting RLAI, the mean level of prolactin was high at T0 (see Table 1). However, there was no significant change in the measurement of prolactin throughout the treatment with RLAI. Also, no significant change in lipids (cholesterol and triglycerides) and blood glucose was recorded during the treatment period (Table 1).
During the treatment period (T0 to T2), 13 patients (68.4%) presented some other laboratory measurements and symptoms attributable as direct side effects of RLAI. The most significant were high prolactin levels (11 or 57.9%), weight gain (7 or 36.8%), and increased appetite (6 or 31.6%).
Two patients reported mild transient tremors in the T0 to T1 periods, and 1 reported a single transient episode of an oculogyric crisis in the T1 to T2 period. None presented neuroleptic malignant syndrome, severe extrapyramidal symptoms, or hypersensitivity to RLAI (Table 2).
Some children and adolescents who initially resisted the injectable formulation changed their minds after being provided psychoeducation and guidelines.
DISCUSSION
In this study, we describe the safety and efficacy of RLAI in pediatric bipolar disorder, evaluating its effectiveness in real-world practice. The findings for this 6-month naturalistic follow-up indicate that RLAI in children and adolescents is generally well tolerated and efficacious and that RLAI enhanced adherence of pediatric bipolar disorder patients who had previously used multiple drug treatments with no significant improvement and poor adherence.
Several studies have pointed out the safety and efficacy of risperidone oral formulation for pediatric bipolar disorder,16,17 and recent trials in adults with bipolar disorder suggested that RLAI is a good treatment option.6 However, as RLAI is a medication whose vehicle (intramuscular depot) has unprecedented use in children and adolescents, evaluating its safety and efficacy in real-life conditions is essential for obtaining useful results in routine clinical situations.18
The severe and dysfunctional conditions of patients at T0 positively changed at the first endpoint (T1) of response analysis, and clinical severity, measured by the CGI-S score, passed from the state of severely ill at T0 to moderately ill at T1 (see Table 1). These results are consistent with those found in similar studies of adults with bipolar disorder treated with RLAI2,19–21 and encourage the use of a depot regimen as a possible treatment option for severely impaired pediatric bipolar disorder patients with poor adherence to oral medication.
The nature of mood instability and the prolonged course of the disease in young bipolar disorder patients may make adherence to any treatment difficult.4 In this sample, the positive evolution observed at T1 was maintained at T2 (see Table 1), indicating that the improvement in overall functioning of these children and adolescents observed in the short-term also remained in the long-term treatment. These findings are consistent with a report of pediatric bipolar disorder patients who benefited from long-term use of RLAI with satisfactory clinical response and tolerability over time.7
Regarding safety and tolerability, the results of this naturalistic observation suggest that RLAI may be well tolerated for both short- and long-term use, with low metabolic changes (eg, glucose and lipid profile), and patients with previous use of oral formulation risperidone reported no difference in side effects with RLAI. In our sample, the most important side effects were increased appetite, weight gain, and high plasma levels of prolactin (see Table 1), which are typical concerns when using atypical antipsychotics.
In this study, the weight gain with RLAI was similar to that found in trials with risperidone oral formulation in youths with bipolar disorder.22 This result is also consistent with results found in recent studies with RLAI in adolescents and young adults with schizophrenia and schizoaffective disorder.23,24 The increase of prolactin level was also similar to that found in trials with risperidone oral formulation.25 Galactorrhea was not observed in short-term (2 months) treatment, and only 1 female adolescent presented this side effect at endpoint T2.
Taking into account the profile of the population (children and adolescents) who generally fear injectable medications, the relatively low dose that was applied allowed for a good acceptability of the vehicle of administration (intramuscular), indicating that intramuscular injection formulation may not be a problem for continued use.
Different factors may lead to noncompliance with treatment, and previous studies have shown that the incidence of nonadherence in adults and young patients with bipolar disorder is highly significant, increasing the incidence of hospitalizations, cycling, and financial costs.4,26,27 For youths with bipolar disorder, there are several types of nonadherence to a prescribed treatment in a clinical setting. In this sample, the main component of nonadherence was that the family failed to supervise the treatment, possibly due to chaotic family conditions (ie, relatives with psychiatric illness). Taken together, inadequate family functioning, in addition to the characteristics of the disorder itself, may exert negative influence on pediatric bipolar disorder patients’ treatment adherence27,28; therefore, the alliance between the care provider and the young bipolar disorder patient should be continuously monitored even though a new resource is available.
There are several limitations to our study, and the results should be read with caution and not hastily extrapolated to larger populations. First, our study was limited by the small sample of children with specific characteristics (eg, nonadherence, severely impaired). Secondly, as this was a naturalistic observational study, as opposed to a controlled study, it had a small sample and broad inclusion criteria. However, these limitations and the lack of a blind control group or wash-out of previous medication before RLAI application were due to the severity of cases and reflect the real-world practice that was the focus of this follow-up observational study.
Since this clinical subject sample was recruited in a university-based teaching hospital, the selected patients might be more clinically impaired than the general population, but, otherwise, this selection bias might ensure the homogeneity of the sample. Nevertheless, the present study indicates the need for further randomized controlled studies in order to better quantify and qualify the results obtained here.
CONCLUSION
In summary, this study demonstrated that RLAI may be an efficacious and relatively well-tolerated alternative for the treatment of pediatric bipolar disorder. This long-acting antipsychotic could help clinicians to manage the compliance status of patients, preventing the occurrence of more severe complications (eg, suicide attempts or hospitalizations) and dramatically reversing the therapeutic failure of patients who did not follow the recommended oral medication treatment.
Drug names: carbamazepine (Carbatrol, Equetro, and others), divalproex (Depakote and others), methylphenidate (Focalin, Daytrana, and others), risperidone (Risperdal and others).
Author affiliations: The Childhood and Adolescence Affective Disorders Program (Drs Boarati, Ferreira-Maia, Cavalcanti, and Fu-I) and the Section of Psychiatric Epidemiology (LIM-23) (Dr Wang), Institute and Department of Psychiatry, School of Medicine, University of São Paulo, São Paulo, Brazil.
Author contributions: Statistical expertise for this study was provided by Yuan-Pang Wang, PhD.
Potential conflicts of interest: Dr Boarati has received support to participate in a medical meeting from Janssen-Cilag. Dr Wang has received grant/research support from the São Paulo Research Support Foundation (FAPESP; grant 2008/11415-9). Dr Fu-I is a consultant for AstraZeneca; has received grant/research support from the FAPESP (grant 2008/55402-8); and has received support to participate in a medical meeting from Janssen-Cilag and Abbott. Drs Ferreira-Maia and Cavalcanti have no conflicts of interest to report.
Funding/support: This study was partially (clinical assessment and phenomenology study) funded by the São Paulo Research Support Foundation (Grant 2008/55402-8) and the University of São Paulo, São Paulo, Brazil.
Role of sponsor: No funding sources were involved in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the paper for publication.
Previous presentation: Preliminary findings from this trial were presented at the 57th Annual Meeting of the American Academy of Child and Adolescent Psychiatry; October 26–31, 2010; New York, New York.
REFERENCES
1. Fagiolini A, Casamassima F, Mostacciuolo W, et al. Risperidone long-acting injection as monotherapy and adjunctive therapy in the maintenance treatment of bipolar I disorder. Expert Opin Pharmacother. 2010;11(10):1727–1740. doi:10.1517/14656566.2010.490831 PubMed
2. Quiroz JA, Yatham LN, Palumbo JM, et al. Risperidone long-acting injectable monotherapy in the maintenance treatment of bipolar I disorder. Biol Psychiatry. 2010;68(2):156–162. doi:10.1016/j.biopsych.2010.01.015 PubMed
3. Kane JM, Eerdekens M, Lindenmayer JP, et al. Long-acting injectable risperidone: efficacy and safety of the first long-acting atypical antipsychotic. Am J Psychiatry. 2003;160(6):1125–1132. doi:10.1176/appi.ajp.160.6.1125 PubMed
4. Coletti DJ, Leigh E, Gallelli KA, et al. Patterns of adherence to treatment in adolescents with bipolar disorder. J Child Adolesc Psychopharmacol. 2005;15(6):913–917. doi:10.1089/cap.2005.15.913 PubMed
5. Vieta E, Nieto E, Autet A, et al. A long-term prospective study on the outcome of bipolar patients treated with long-acting injectable risperidone. World J Biol Psychiatry. 2008;9(3):219–224. doi:10.1080/15622970701530917 PubMed
6. Macfadden W, Alphs L, Haskins JT, et al. A randomized, double-blind, placebo-controlled study of maintenance treatment with adjunctive risperidone long-acting therapy in patients with bipolar I disorder who relapse frequently. Bipolar Disord. 2009;11(8):827–839. doi:10.1111/j.1399-5618.2009.00761.x PubMed
7. Fu-I L, Boarati MA, Stravogiannis A, et al. Use of risperidone long-acting injection to support treatment adherence and mood stabilization in pediatric bipolar patients: a case series. J Clin Psychiatry. 2009;70(4):604–606. doi:10.4088/JCP.08l04487 PubMed
8. Risperdal [package insert]. Titusville, New Jersey: Ortho-McNeil-Janssen Pharmaceuticals, Inc; 2007. http://risperdal.com/sites/default/files/shared/pi/risperdal_uspi.pdf. Updated November 2011. Accessed June 14, 2012.
9. Achenbach TM, Edelbrock CS. Behavioral problems and competencies reported by parents of normal and disturbed children aged four through sixteen. Monogr Soc Res Child Dev. 1981;46(1):1–82. doi:10.2307/1165983 PubMed
10. Reich W. Diagnostic Interview for Children and Adolescents (DICA). J Am Acad Child Adolesc Psychiatry. 2000;39(1):59–66. doi:10.1097/00004583-200001000-00017 PubMed
11. Green B, Shirk S, Hanze D, et al. The Children’s Global Assessment Scale in clinical practice: an empirical evaluation. J Am Acad Child Adolesc Psychiatry. 1994;33(8):1158–1164. doi:10.1097/00004583-199410000-00011 PubMed
12. Shaffer D, Gould MS, Brasic J, et al. A Children’s Global Assessment Scale (CGAS). Arch Gen Psychiatry. 1983;40(11):1228–1231. doi:10.1001/archpsyc.1983.01790100074010 PubMed
13. Guy W. ECDEU Assessment Manual for Psychopharmacology. US Dept Health, Education, and Welfare publication (ADM) 76-338. Rockville, Md: National Institute of Mental Health; 1976:218–222.
14. Levine J, Schooler NR. SAFTEE: a technique for the systematic assessment of side effects in clinical trials. Psychopharmacol Bull. 1986;22(2):343–381. PubMed
15. Levesque R. SPSS Programming and Data Management: A Guide for SPSS and SAS Users. 4th ed. Chicago, IL: SPSS Inc; 2007.
16. American Academy of Child and Adolescent Psychiatry. Practice parameter on the use of psychotropic medication in children and adolescents. J Am Acad Child Adolesc Psychiatry. 2009;48(9):961–973. doi:10.1097/CHI.0b013e3181ae0a08 PubMed
17. Biederman J, Mick E, Wozniak J, et al. An open-label trial of risperidone in children and adolescents with bipolar disorder. J Child Adolesc Psychopharmacol. 2005;15(2):311–317. doi:10.1089/cap.2005.15.311 PubMed
18. Stroup TS, McEvoy JP, Swartz MS, et al. The National Institute of Mental Health Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) project: schizophrenia trial design and protocol development. Schizophr Bull. 2003;29(1):15–31. doi:10.1093/oxfordjournals.schbul.a006986 PubMed
19. Bobo WV, Shelton RC. Risperidone long-acting injectable (Risperdal Consta) for maintenance treatment in patients with bipolar disorder. Expert Rev Neurother. 2010;10(11):1637–1658. doi:10.1586/ern.10.143 PubMed
20. Kemp DE, Canan F, Goldstein BI, et al. Long-acting risperidone: a review of its role in the treatment of bipolar disorder. Adv Ther. 2009;26(6):588–599. doi:10.1007/s12325-009-0039-0 PubMed
21. Yatham LN, Fallu A, Binder CE. A 6-month randomized open-label comparison of continuation of oral atypical antipsychotic therapy or switch to long acting injectable risperidone in patients with bipolar disorder. Acta Psychiatr Scand suppl. 2007;116(434):50–56. doi:10.1111/j.1600-0447.2007.01059.x PubMed
22. Bishop JR, Pavuluri MN. Review of risperidone for the treatment of pediatric and adolescent bipolar disorder and schizophrenia. Neuropsychiatr Dis Treat. 2008;4(1):55–68. PubMed
23. Emsley R, Medori R, Koen L, et al. Long-acting injectable risperidone in the treatment of subjects with recent-onset psychosis: a preliminary study. J Clin Psychopharmacol. 2008;28(2):210–213. doi:10.1097/JCP.0b013e318167269d PubMed
24. Lasser RA, Bossie CA, Zhu Y, et al. Long-acting risperidone in young adults with early schizophrenia or schizoaffective illness. Ann Clin Psychiatry. 2007;19(2):65–71. doi:10.1080/10401230701332931 PubMed
25. Rosenbloom AL. Hyperprolactinemia with antipsychotic drugs in children and adolescents [published online ahead of print August 24, 2010]. Int J Pediatr Endocrinol. 2010. PubMed
26. Bagalman E, Yu-Isenberg KS, Durden E, et al. Indirect costs associated with nonadherence to treatment for bipolar disorder. J Occup Environ Med. 2010;52(5):478–485. doi:10.1097/JOM.0b013e3181db811d PubMed
27. Drotar D, Greenley RN, Demeter CA, et al. Adherence to pharmacological treatment for juvenile bipolar disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(7):831–839. doi:10.1097/chi.0b013e31805c7421 PubMed
28. Colom F, Lam D. Psychoeducation: improving outcomes in bipolar disorder. Eur Psychiatry. 2005;20(5–6):359–364. doi:10.1016/j.eurpsy.2005.06.002 PubMed
Enjoy this premium PDF as part of your membership benefits!


