Prim Care Companion CNS Disord 2022;24(6):21cr03220
To cite: Varghese R, Czuma R, Cohen-Oram A, et al. A case of lurasidone-induced urinary retention. Prim Care Companion CNS Disord. 2022;24(6):21cr03220.
To share: https://doi.org/10.4088/PCC.21cr03220
© 2022 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry and Behavioral Neurosciences, Tampa General Hospital, University of South Florida, Tampa, Florida
*Corresponding author: Rojan Varghese, MD, Tampa General Hospital, 1 Tampa General Cir, Tampa, FL 33606 ([email protected]).
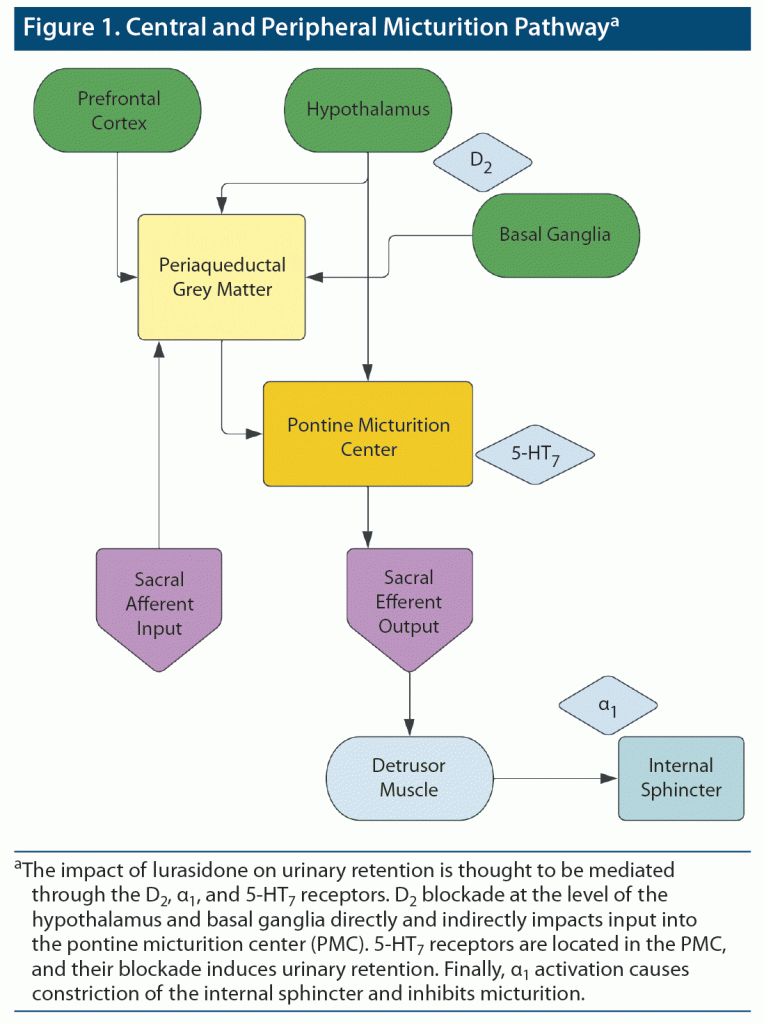
Urinary retention is the inability to completely empty the bladder.1 Micturition is a complex process, involving several receptors in central neural pathways in the brain and spinal cord as well as peripheral smooth muscle activity in the bladder and urethra (Figure 1).2 As such, a variety of medications have been implicated in cases of urinary retention, estimated to play a role in 2%–10% of all cases.3,4 In the literature, antipsychotics, particularly ziprasidone, haloperidol, amisulpride, trebenzomine, thioridazine, and quetiapine, have been associated with urinary retention.4 Lurasidone is a newer second-generation antipsychotic, and to date there has been only 1 case report5 associating its use with urinary retention; however, notably there were 2 other potential causative agents involved. We now report a case of a woman who developed urinary retention during the course of lurasidone monotherapy.
Case Report
Ms P was a 58-year-old woman with a history of bipolar I disorder (DSM-5 criteria) who presented for management of depressive symptoms. Notably, she had no prior history of urinary retention or other urinary pathology, though she had a history of stable diabetes mellitus with associated peripheral neuropathy. She had failed multiple previous psychotropic trials and so was started on oral lurasidone 20 mg/d for bipolar depression (DSM-5 criteria). At her follow-up visit 1 month later, she noted moderate response but not remission of her depressive symptoms, and lurasidone was increased to 40 mg/d. After this dose increase, she began to report difficulty with urination, which resolved over days on return to her previous 20-mg dose. This difficulty had not previously occurred with any psychotropics or medications in general. However, at this dose, she continued to describe her mood as “depressed.” She was amenable to a repeat trial of lurasidone 40 mg/d. However, after 3 days, the patient again reported difficulty urinating, and once again her symptoms resolved after reducing the lurasidone dose to 20 mg/d. No other comorbid medical factors were diagnosed or exacerbated during these encounters.
Discussion
This case represents an occurrence highly suggestive of lurasidone-induced acute urinary retention. The patient had no prior history of urinary pathology or symptoms before the initiation of lurasidone. Diabetic neuropathy has been associated with cases of urinary retention, though our patient had no prior urinary symptoms, nor any progression of her diabetes.6 There is also a lower prevalence of organic causes of urinary retention in females.6 The case also presents a strong chronological connection between the up-titration and down-titration of lurasidone with the development and resolution, respectively, of urinary retention. The monotherapeutic use of lurasidone also distinguishes this case from a previous report5 of urinary retention that occurred in combination with other agents.
Lurasidone is a second-generation antipsychotic first approved for use in schizophrenia in 2010 and later approved for monotherapy in bipolar depression.7 Lurasidone’s most notable receptor property in comparison to other antipsychotics is its strong antagonism of the serotonin 5-HT7 receptor. There is growing evidence that this receptor is involved in the promotion of micturition and voiding efficiency; thus, lurasidone’s antagonism at this receptor provides a relatively unique mechanism for development of urinary retention.8 Another possible contributory mechanism is D2 receptor blockade, as activation of D2 central receptors promotes micturition. However, of note, no association between affinity for the D2 receptor and incidence of urinary retention has been formally studied.4 Lurasidone also shows moderate affinity for α1 receptors, activation of which may contribute to internal urethral sphincter contraction and urinary retention.6,7
Ms P’s presentation is the only documented case of urinary retention associated with lurasidone monotherapy, and the repeated chronological link between medication titration and symptoms is suggestive of causality. Given increased use of lurasidone after its relatively recent entry into the market, its status as only 1 of 3 US Food and Drug Administration–approved medications for bipolar depression, and the launch of a generic in 2023, clinicians should be aware of and consider screening for this side effect more routinely.
Published online: December 13, 2022.
Relevant financial relationships: Drs Varghese, Czuma, Cohen-Oram, and Jiang have no disclosures of funding/support or conflicts of interest to report.
Funding/support: None.
Additional information: Information has been de-identified to protect anonymity.
References (8)

- Yoon PD, Chalasani V, Woo HH. Systematic review and meta-analysis on management of acute urinary retention. Prostate Cancer Prostatic Dis. 2015;18(4):297–302. PubMed CrossRef
- Yoshimura N, Chancellor MB. Neurophysiology of lower urinary tract function and dysfunction. Rev Urol. 2003;5(suppl 8):S3–S10. PubMed
- Trinchieri M, Perletti G, Magri V, et al. Urinary side effects of psychotropic drugs: A systematic review and metanalysis. Neurourol Urodyn. 2021;40(6):1333–1348. PubMed CrossRef
- Faure Walker N, Brinchmann K, Batura D. Linking the evidence between urinary retention and antipsychotic or antidepressant drugs: a systematic review. Neurourol Urodyn. 2016;35(8):866–874. PubMed CrossRef
- Kahn B, Boazak M, Ragazino J, et al. An additive mix? acute urinary retention in a patient with benign prostatic hyperplasia treated with suboxone, lurasidone, and trazodone. Focus Am Psychiatr Publ. 2018;16(3):292–298. PubMed CrossRef
- Verhamme KMC, Sturkenboom MCJM, Ch Stricker BH, et al. Drug-Induced Urinary Retention Incidence. Management and Prevention; 2008:31.
- Riva MA. An update of the preclinical profile of lurasidone. Evidence-Based Psychiatric Care. 2015;1:67–72.
- Norouzi-Javidan A, Javanbakht J, Barati F, et al. Effect of 5-HT7 receptor agonist, LP-211, on micturition following spinal cord injury in male rats. Am J Transl Res. 2016;8(6):2525–2533. PubMed
Enjoy this premium PDF as part of your membership benefits!