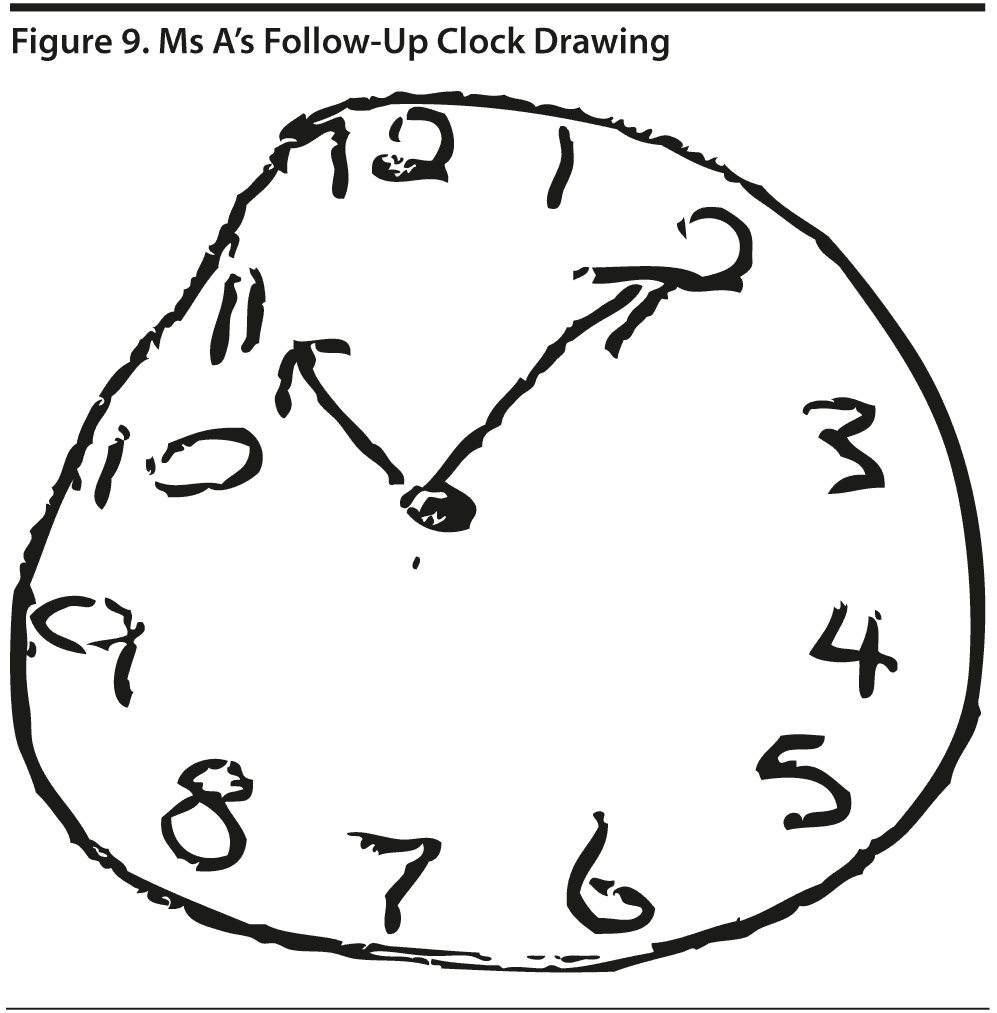Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
CASE CONFERENCE
The Banner Alzheimer’s Institute Case Conference is a weekly event in which physicians and staff discuss challenging and/or teaching cases of patients seen at the Institute’s Memory Disorders Clinic. These conferences are attended by a multidisciplinary group that includes Banner Alzheimer’s Institute dementia specialists, community physicians (internal medicine, family medicine, and radiology), physician assistants, social workers, nurses, medical students, residents, and fellows.
BANNER ALZHEIMER’ S INSTITUTE
The Banner Alzheimer’s Institute located in Phoenix, Arizona, has an unusually ambitious mission: to end Alzheimer’s disease without losing a generation, set a new standard of care for patients and families, and forge a model of collaboration in biomedical research. The Institute provides high-level care and treatment for patients affected by Alzheimer’s disease, dementia, and related disorders. In addition, the Institute offers extensive support services for families and many unique and rewarding research opportunities.
Prim Care Companion CNS Disord 2012;14(6):doi:10.4088/PCC.12alz01491
© Copyright 2012 Physicians Postgraduate Press, Inc.
Received: November 9, 2012; accepted November 9, 2012.
Published online: December 27, 2012.
AUTHORS
Roy Yaari, MD, MAS, a neurologist, is associate director of the Memory Disorders Clinic of Banner Alzheimer’s Institute and a clinical professor of neurology at the College of Medicine, University of Arizona, Tucson.
Helle Brand, PA, is a physician assistant at the Memory Disorders Clinic of Banner Alzheimer’s Institute.
James D. Seward, PhD, ABPP, is a clinical neuropsychologist at Banner Alzheimer’s Institute.
Anna D. Burke, MD, is a geriatric psychiatrist and dementia specialist at the Memory Disorders Clinic of Banner Alzheimer’s Institute.
Adam S. Fleisher, MD, MAS, is associate director of Brain Imaging at the Banner Alzheimer’s Institute, a neurologist at the Institute’s Memory Disorders Clinic, and an associate professor in the Department of Neurosciences at the University of California, San Diego.
Pierre N. Tariot, MD, a geriatric psychiatrist, is director of Banner Alzheimer’s Institute and a research professor of psychiatry at the College of Medicine, University of Arizona, Tucson.
Corresponding author: Roy Yaari, MD, MAS, Banner Alzheimer’s Institute, 901 E. Willetta St, Phoenix, AZ 85006 ([email protected]).
CME Background
Original material is selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME activities on a variety of topics from volume to volume. This special series of case reports about dementia was deemed valuable for educational purposes by the Publisher, Editor in Chief, and CME Institute Staff. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the material and go to PrimaryCareCompanion.com to complete the Posttest and Evaluation online.
CME Objective
After studying this case, you should be able to:
- Conduct a differential diagnosis in a patient who presents with behavioral and memory problems and a history of substance abuse and suicide attempt
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1.0 hour of Category I credit for completing this program.
Date of Original Release/Review
This educational activity is eligible for AMA PRA Category 1 Creditâ„¢ through December 31, 2015. The latest review of this material was December 2012.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Larry Culpepper, MD, MPH, Editor in Chief, has been a consultant for AstraZeneca, Pfizer, Takeda, and Trovis and has been a member of the speakers/advisory boards for Forest and Merck. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears at the end of the article.
HISTORY OF PRESENT ILLNESS
Ms A, a 43-year-old left-handed woman with impaired cognition, was evaluated on the inpatient psychiatric unit. Clinical history was obtained from Ms A’s husband of 17 years and her 24-year-old son from a prior relationship. Her premorbid personality is described as “fiery.” She is a person who “speaks her mind,” is always “in control,” and always gets “her way.” At baseline, she is reported to have “spotty” short-term memory and is quick to anger.
About 4 years ago, Ms A experienced significant stress not associated with cognitive changes. Her husband’s mother had developed Alzheimer’s disease and moved in with them so that they could provide care. Concurrently, Ms A’s son, who struggled with substance abuse, impregnated his girlfriend, who subsequently gave birth to Ms A’s second grandchild. Additionally, Ms A’s adoptive father, who lived out of state, was diagnosed with Alzheimer’s disease while undergoing treatment for disseminated prostate cancer. During this time, Ms A continued to be “in charge.” She was able to perform all household chores, including cleaning, shopping, cooking, and finances, while traveling out of state frequently to visit her ailing father. However, during this time, she drank “excessive amounts” of alcohol. She was cited for driving under the influence. Her husband stated that Ms A was drinking “anywhere from 1 glass of wine to 2 bottles of wine almost every day.”
During one of her visits with her father, Ms A confronted him about alleged physical and sexual abuse by him when she was a child. He denied this accusation and made light of it. Ms A’s alcohol consumption continued, and her anger intensified. Ms A’s husband was not aware of her abusive childhood until she told him about confronting her father. After that, Ms A’s husband no longer wanted her to travel out of state to care for her father. Ms A was resentful, arguments escalated, and the couple’s relationship deteriorated. Her husband stated that she was “irrational,” and after 6 months, they finally agreed to marital counseling. When their first counseling appointment was drawing near, Ms A traveled to visit her father without telling anyone. When she returned from this trip, she appeared more “irrational” and arguments continued. The next day, Ms A’s son found her unresponsive in her bedroom with an empty bottle of oxycodone by her, which she had taken from her father’s house. The family reports that they were unable to find a pulse. When paramedics arrived, they found Ms A awake and able to answer questions “slowly but appropriately.” Her blood pressure was 150/100 mm Hg, her heart rate was 150 bpm, and her oxygen saturation was 80% on room air. With administration of oxygen, her oxygen saturation improved to 100%. Upon arrival to the emergency department, Ms A was noted to be “awake and alert,” but she had acute renal failure and elevated hepatic liver function tests. Ms A admitted that she took 13 oxycodone pills “looking for an escape” from her marital problems. Over 4 days in the hospital, she had a remarkable medical recovery and improved to baseline cognition and health. Ms A was then discharged home where she functioned at her baseline level “as if nothing had happened.” The family had removed alcohol from the house and observed her closely.
Approximately 10 unremarkable days following her discharge from the hospital, Ms A began to display “bizarre” behaviors. For example, she parked her car in the garden causing damage to her car and insisted that she parked appropriately. She also parked her car in the middle of the driveway at her son’s workplace. When confronted, she would say, “I can park where I want.” Each day she became progressively “weirder” per description of her husband. She would talk on the phone holding it upside down. She became disoriented to time. She began scratching her arms excessively. She squeezed toothpaste on door hinges. She believed that people were in the shower spraying her with water, and she claimed to see other people in the house (who were not there). She poured sugar into a candle thinking it was a cup. She caused a stove fire. The family thought that Ms A might be abusing drugs or alcohol, although they did not witness drug or alcohol use. Her bizarre behaviors continued to worsen.
By about 2 weeks after her hospital discharge, Ms A was unable to dress herself and was geographically disoriented and agitated. She put a cup in the microwave with no water in it. Her attempts to shower took 2 to 3 hours. She caused another kitchen fire to which she was oblivious. Ms A’s husband brought her back to the emergency department 2½ weeks after her discharge from the hospital. She was admitted, and once medically cleared, was transferred to a psychiatric unit. At that time, divalproex sodium 500 mg daily was initiated for presumptive underlying bipolar disorder.
PAST MEDICAL HISTORY
Ms A was sexually and physically abused as a child per her husband’s report. This abuse included head injuries without known history of concussion. She had no prior medical diagnoses. There was no history of prior suicide attempts. There was no prior history of psychiatric illness.
ALLERGIES
Ms A had no known drug allergies.
MEDICATIONS
Prior to this hospitalization, Ms A was not prescribed any medications. She was started on divalproex sodium 500 mg daily during this hospitalization.
SOCIAL HISTORY
Ms A did not complete high school. She worked “odd jobs.” In the months before her overdose, Ms A was not employed and was described as a “homemaker.” She had her first son at the age of 19 years. Her boyfriend at that time was reported to be verbally and physically abusive toward her. Ms A and her husband have been married for 17 years, and they have 1 son together. There have been significant social stressors as mentioned above. Ms A currently lives with her husband, 1 of her 2 sons, and her husband’s mother, who has Alzheimer’s disease.
SUBSTANCE USE
Ms A did not drink alcohol excessively until recently and had 1 citation for driving under the influence, as mentioned previously. She stopped smoking cigarettes about 8 years ago after a 20-year history of smoking 1 pack per day. Despite family suspicions, there was no known history of illicit substance use.
FAMILY HISTORY
Ms A was adopted. Her biological mother and maternal grandmother are alive and cognitively intact. Her biological father is also alive and presumed to be cognitively intact. Her adoptive father, as mentioned above, has Alzheimer’s disease. Ms A has 2 half siblings from the same biological mother who are both cognitively intact. There was no known psychiatric history in Ms A’s biological family.
PHYSICAL EXAMINATION
Ms A’s vital signs were blood pressure: 118/80 mm Hg, pulse: 102 bpm, temperature: 97.9°F (36.6°C), and respiratory rate: 18 breaths/minute. Ms A’s general physical examination was unremarkable.
NEUROLOGIC EXAMINATION
The neurologic examination was normal except for a glabellar reflex. Deep tendon reflexes were symmetric but quite brisk in the bilateral lower extremities, and the plantar reflexes were both down-going.
Different dementias may be associated with various physical examination findings. However, most often the physical examination is normal in the early stages. Some subtle general findings can include frontal release signs such as a positive snout, glabellar, or palmomental reflex (Links et al, 2010).
Based on the clinical history, what can be eliminated from the differential diagnosis?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Substance abuse | 0% |
| B. Alcohol withdrawal | 0% |
| C. Psychiatric disease | 0% |
| D. FTD | 0% |
| E. Prion disease | 0% |
| F. Dementia (other) | 0% |
| G. Anoxic brain injury | 0% |
| H. Encephalitis (bacterial, viral, or fungal) | 0% |
| I. Autoimmune vasculitis | 0% |
| J. Focal brain lesion (tumor, stroke) | 0% |
| K. Paraneoplastic syndrome | 0% |
| L. Nonconvulsive status epilepticus | 0% |
| M. All of the above should be included in the differential | 100% |
Of the conference attendees, 100% chose option M (all of the above). Although some of the conditions may be more likely than others, at this time, the differential diagnosis is quite broad, and Ms A will require further testing. Some conference attendees felt that, although alcohol withdrawal should be kept within the differential diagnosis, the time frame of 10 days without alcohol (if the family report is accurate) would be longer than expected for alcohol withdrawal. With regard to option C (psychiatric disease), possible specific diagnoses proposed by attendees were depression with psychosis, bipolar depression, schizoaffective disorder, or personality disorder. The attendees felt that option D (FTD) was not likely given the acute course of the cognitive illness but agreed that it should be kept within the differential diagnosis until more information is obtained. Likewise, option E (prion disease) would be less likely given the absence of motor findings, but it was felt that it should remain within the differential diagnosis given Ms A’s rapid cognitive decline. Many conference attendees believed that anoxic brain injury was possible but unlikely given the 10-day lucid interval after Ms A’s cardiac arrest.
A Mini-Mental State Examination (MMSE) score generally correlates with disease severity. Scores ≤ 9 points can indicate severe dementia, scores between 10-20 points can indicate moderate dementia, and a score > 20 can indicate mild dementia (Mungas, 1991). Although MMSE scores must be interpreted in light of both the patient’s age and education, education is the primary demographic factor that affects scores. Therefore, whereas a cutoff of < 23 is widely used in distinguishing between normal and abnormal performance, this cutoff may have less predictive ability in poorly educated individuals (Folstein et al, 1975).
Based on the clinical history, what do you think the MMSE score is?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 26-30 | 0% |
| B. 21-25 | 23% |
| C. 16-20 | 15% |
| D. 11-15 | 54% |
| E. < 11 8% |
Most conference attendees predicted a moderately impaired MMSE score. Ms A scored 17/30 on the MMSE. Figure 1 shows Ms A’s pentagon drawing and sentence from the MMSE.
In the Category Retrieval Test, the examiner asks the patient to name as many animals as possible in 1 minute. The examiner records the responses. Performance on this measure is influenced by age; unimpaired people in their 60s should name about 18 animals, whereas people in their 80s should name about 15 (Mitrushina et al, 2005). There is no hard-and-fast cutoff for impairment; however, patients who name 4 or more animals less than expected raise concerns. Note that bilingual individuals are at a disadvantage on this and other measures of verbal fluency (Gollan et al, 2002).
Based on the clinical history, what do you think the Category Retrieval Test score is?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 21-25 | 0% |
| B. 16-20 | 0% |
| C. 11-15 | 31% |
| D. 6-10 | 69% |
| E. 0-5 | 0% |
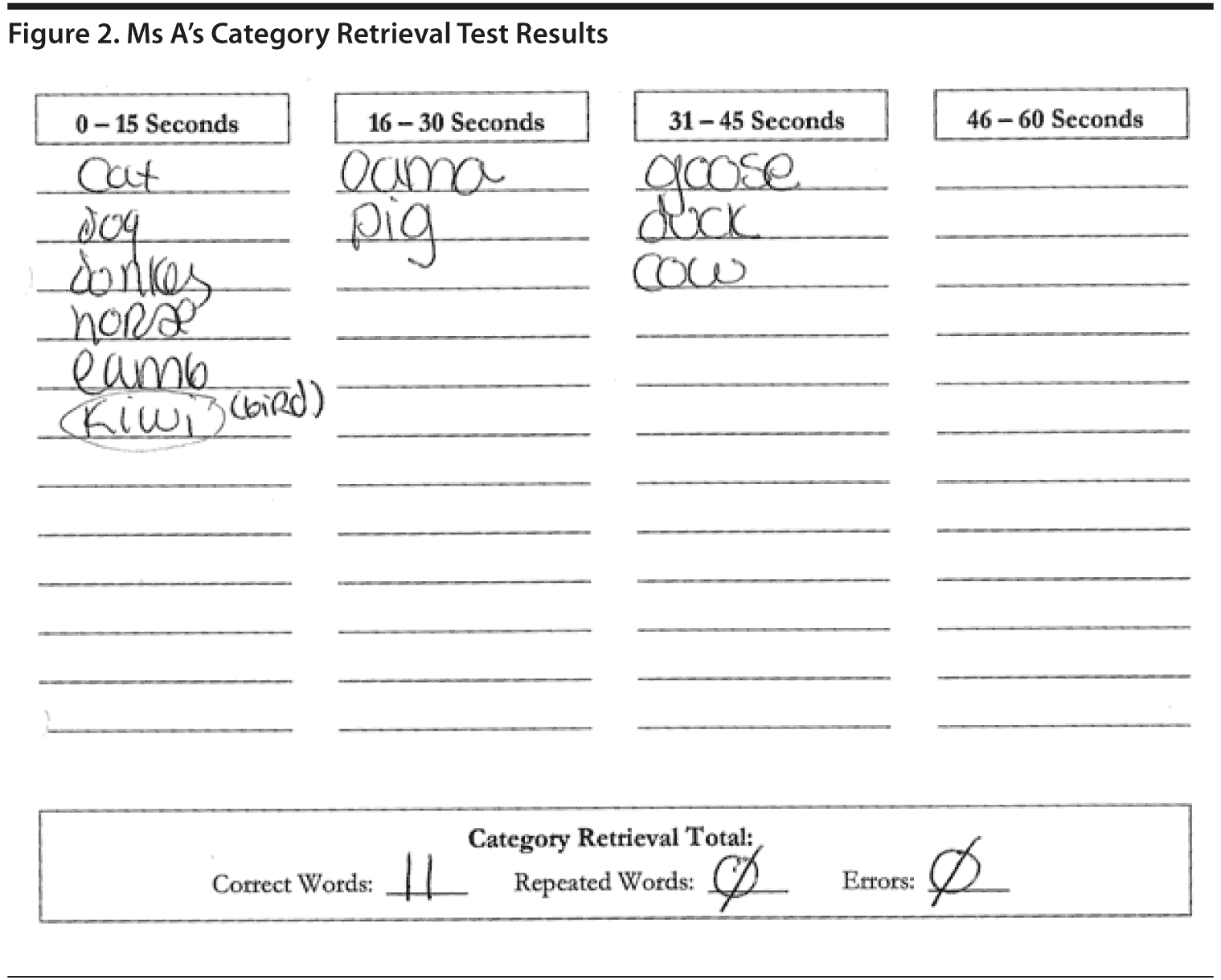
Ms A listed 11 animals with no repetitions. Figure 2 shows Ms A’s Category Retrieval Test results.
The Montreal Cognitive Assessment is a 30-point test that assesses several cognitive domains. Because it is more challenging than the Mini-Mental State Examination, it has greater sensitivity for mild cognitive impairment and early stages of dementia. With a cutoff score < 26, the sensitivity for detecting mild cognitive impairment (N = 94) was 90% and the specificity was 87% (Nasreddine et al, 2005). This test is available at http://mocatest.org/.
Based on the clinical history, what do you think the MoCA score is?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 26-30 | 0% |
| B. 21-25 | 0% |
| C. 16-20 | 21% |
| D. 11-15 | 71% |
| E. < 11 | 7% |
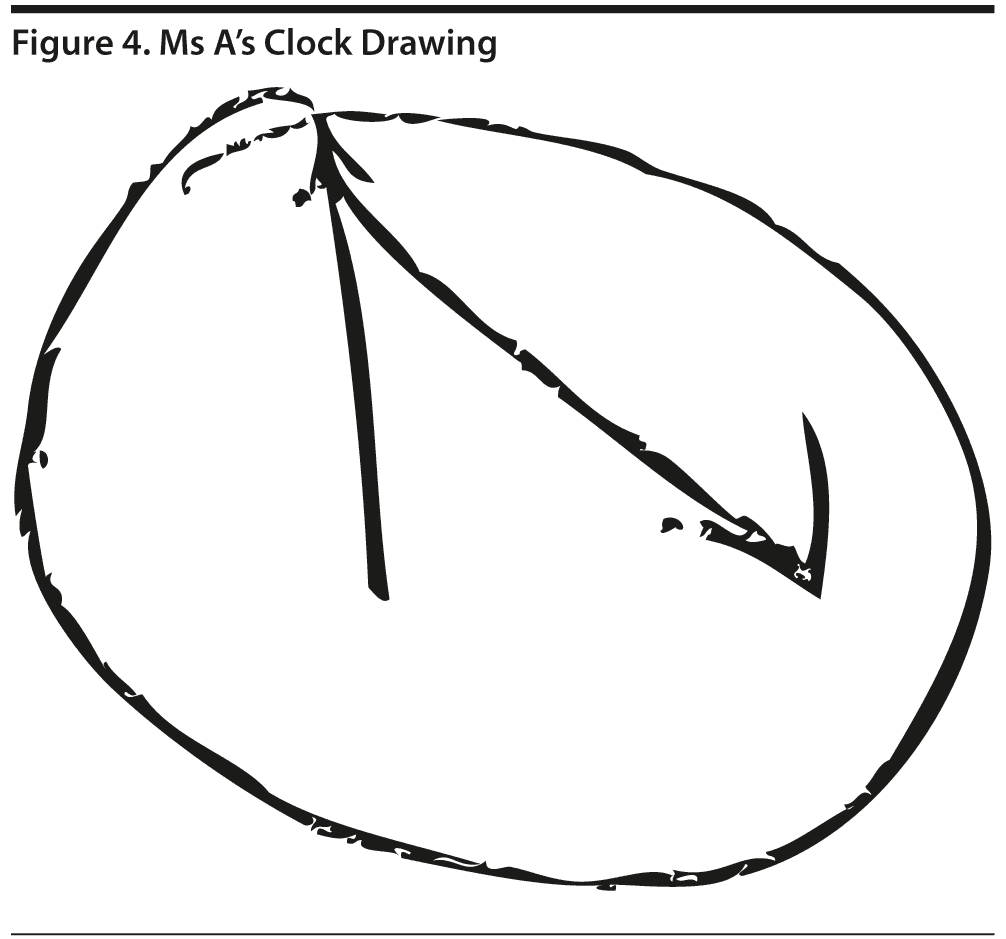
Ms A scored 9/30 on the MoCA. This score includes 1 additional point for education ≤ 12 years. Figure 3 shows Ms A’s MoCA test results, and Figure 4 shows her clock drawing.
What test(s) are not clinically indicated at this time?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Lumbar puncture (cell count, glucose, protein, fungal screen, viral screen, bacterial, etc) | 0% |
| B. Brain MRI | 0% |
| C. Brain FDG-PET | 100% |
| D. EEG | 0% |
| E. CBC and CMP, ammonia level | 0% |
| F. TSH | 0% |
| G. Urinalysis | 0% |
| H. Toxicology screen | 0% |
| I. Human immunodeficiency virus | 0% |
| J. ESR | 0% |
| K. All tests are indicated | 0% |
Of the conference attendees, 100% felt that FDG-PET was not indicated. All attendees felt that all other tests listed are indicated. If the workup is otherwise negative, an FDG-PET may be useful in the future.
TEST RESULTS
Ms A’s CMP was unremarkable as was the CBC (very mild anemia); her urinalysis and ESR as well as ammonia, vitamin B12, and C-reactive protein levels were normal; her TSH level was mildly elevated at 5.01 mIU/mL; and her human chorionic gonadotropin screen was not consistent with pregnancy. Ms A’s drug screens were negative for acetaminophen, acetone, salicylate, amphetamine, barbiturate, benzodiazepine, cannabinoid, cocaine, ethanol, isopropanol, methadone, methanol, opiate, oxycodone, phencyclidine, propoxyphene, and tricyclic antidepressants. The only drug/chemical detected by gas chromatography/mass spectrometry was caffeine.
Ms A’s EEG was unremarkable. Results of her cerebrospinal fluid (CSF) lumbar puncture were red blood cell count: 1 and white blood cell count: 1 (normal), CSF glucose: 61 mmol/L (normal), and CSF protein: 56 g/L (mild elevation, not clinically significant). The CSF was negative for bacteria, herpes simplex virus, and West Nile virus; VDRL and fungal tests were also negative.
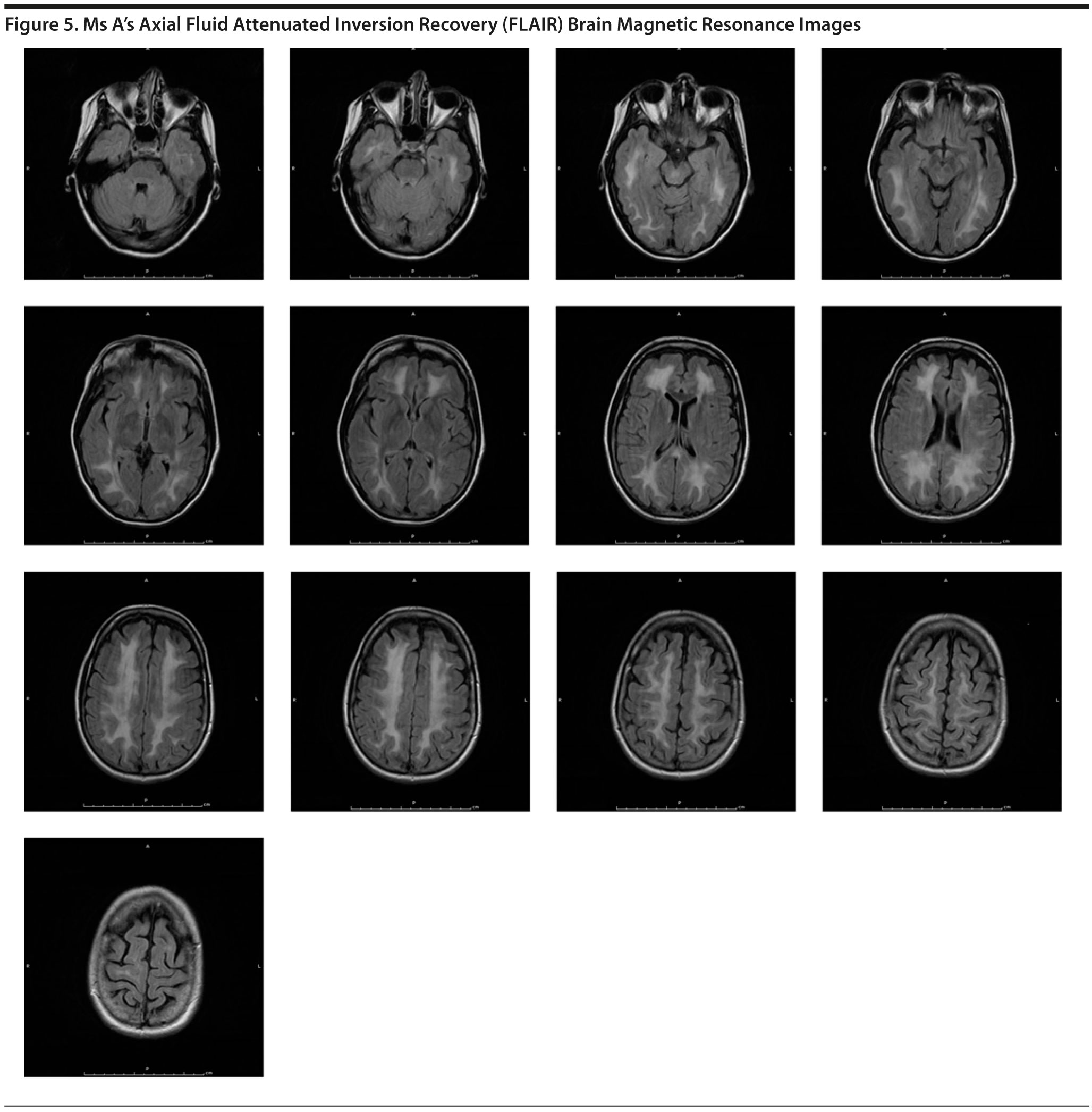
MRI of the brain showed a diffuse leukoencephalopathy (Figure 5). After viewing the MRI, many of the conference participants suggested adding leukodystrophies, such as metachromatic leukodystrophy, to the differential diagnosis. It was thought that Ms A may have a leukodystrophy that led to her behavior changes.
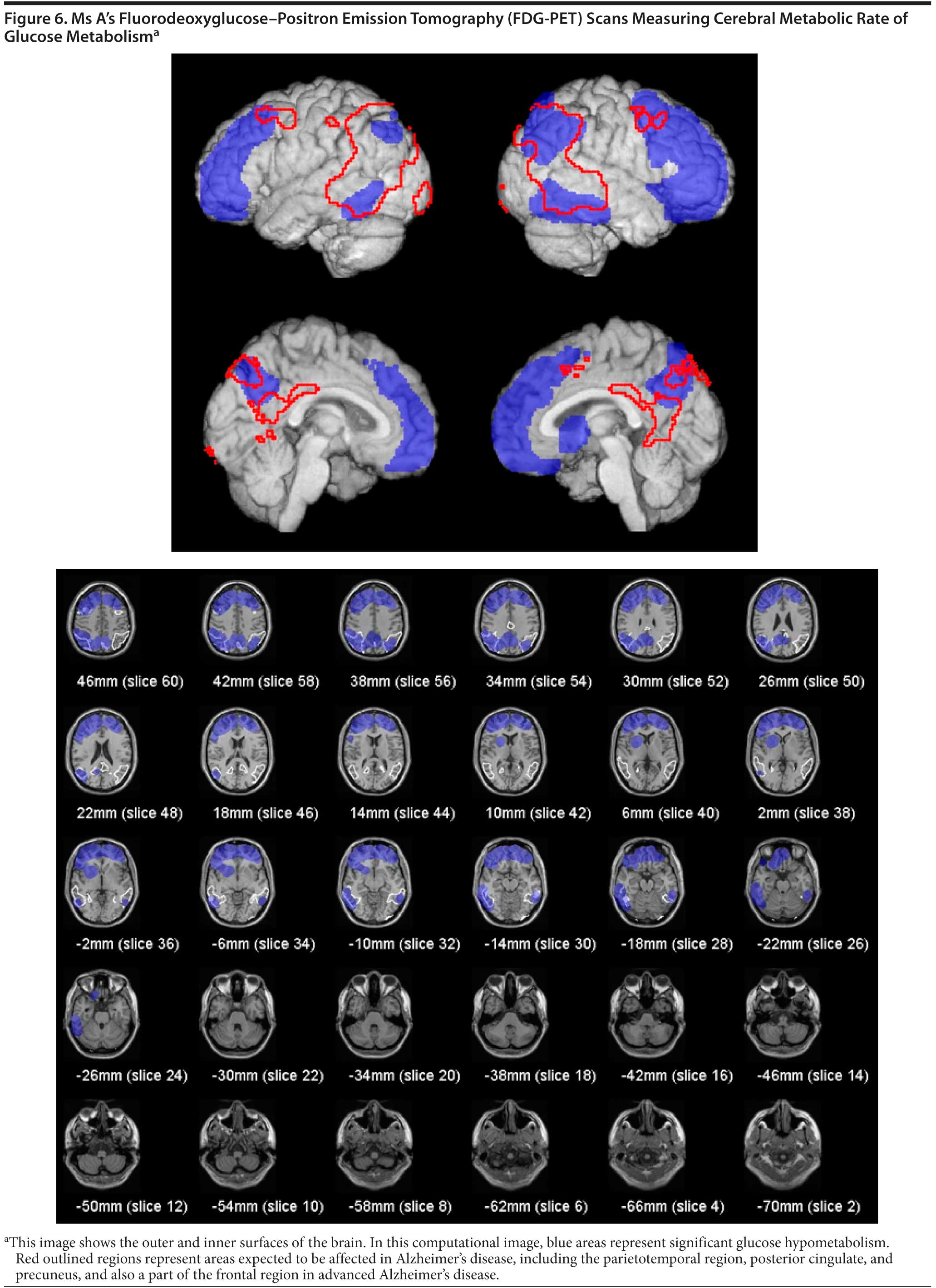
An FDG-PET scan was ordered by the inpatient hospital team and was not recommended by the Banner Alzheimer’s Institute treating physician. The clinical indication for FDG-PET is to help distinguish between Alzheimer’s disease and FTD. Given the results of the MRI, there was clearly a disease process affecting the white matter. The FDG-PET results are shown for interest (Figure 6). The results concluded that extensive cortical hypometabolism was present. The pattern is most suggestive of frontotemporal dementia.
THE BANNER ALZHEIMER’ S INSTITUTE TREATING PHYSICIAN’ S IMPRESSION AND PLAN
Impression
Ms A is a 43-year-old woman who is an inpatient on the psychiatric ward following a suicide attempt with oxycodone overdose. She suffered a cardiac arrest for an unknown period of time with renal and liver impairment and improved to baseline. She experienced a 10-day lucid interval and then developed progressively worsening changes in behavior and psychosis. Laboratory tests thus far have been generally unremarkable except for an MRI of the brain that showed diffuse leukoencephalopathy. Her premorbid personality was “fiery,” and Ms A was recently abusing alcohol amid a troubled marriage. The most likely etiology of her current behavioral and cognitive changes is due to the syndrome of delayed posthypoxic leukoencephalopathy. Note, none of the conference attendees were familiar with this syndrome. Although the FDG-PET findings are presumed to be a result of the delayed posthypoxic leukoencephalopathy, the question arises as to whether she may have an underlying incipient FTD. Given the clinical history, it is highly unlikely that Ms A may have had a preexisting leukoencephalopathy or progressive neurodegenerative condition.
Plan
- Monitor cognition for improvement.
- Once stable, if Ms A does not return to baseline, she may need rehabilitation.
- Continue treatment of underlying psychiatric illness.
- Follow up as an outpatient for continued observation.
8-WEEK FOLLOW-UP WITH THE Banner Alzheimer’s Institute TREATING PHYSICIAN
Ms A presented for follow-up with a close friend who supplemented the clinical history. Ms A was discharged from the hospital after a 2-week inpatient psychiatry admission with significant and rapid cognitive improvement. At the time of discharge, Ms A states that she was about 70%-75% back to her baseline level of functioning. Within the month after the discharge from the hospital, she states that she was about 95% back to her baseline level and feels that she is almost 100% at the current time. Ms A’s friend corroborates her impression of improvement.
Ms A states that she has no suicidal ideations and that she is in very good spirits, as she feels that she was given a second chance at life. Since discharge from the hospital, however, issues with her husband have further deteriorated. Ms A gives a very good history today with clear historical detail. She continues taking divalproex sodium 500 mg daily since her hospitalization. Divalproex sodium levels are being followed by Ms A’s primary care physician. No further brain imaging has been performed since her hospitalization.
Ms A’s neurologic examination was normal. No frontal release signs were present.
Based on the clinical history, what do you think the MMSE score is at this follow-up visit?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 26-30 | 100% |
| B. 21-25 | 0% |
| C. 16-20 | 0% |
| D. 11-15 | 0% |
| E. < 11 | 0% |
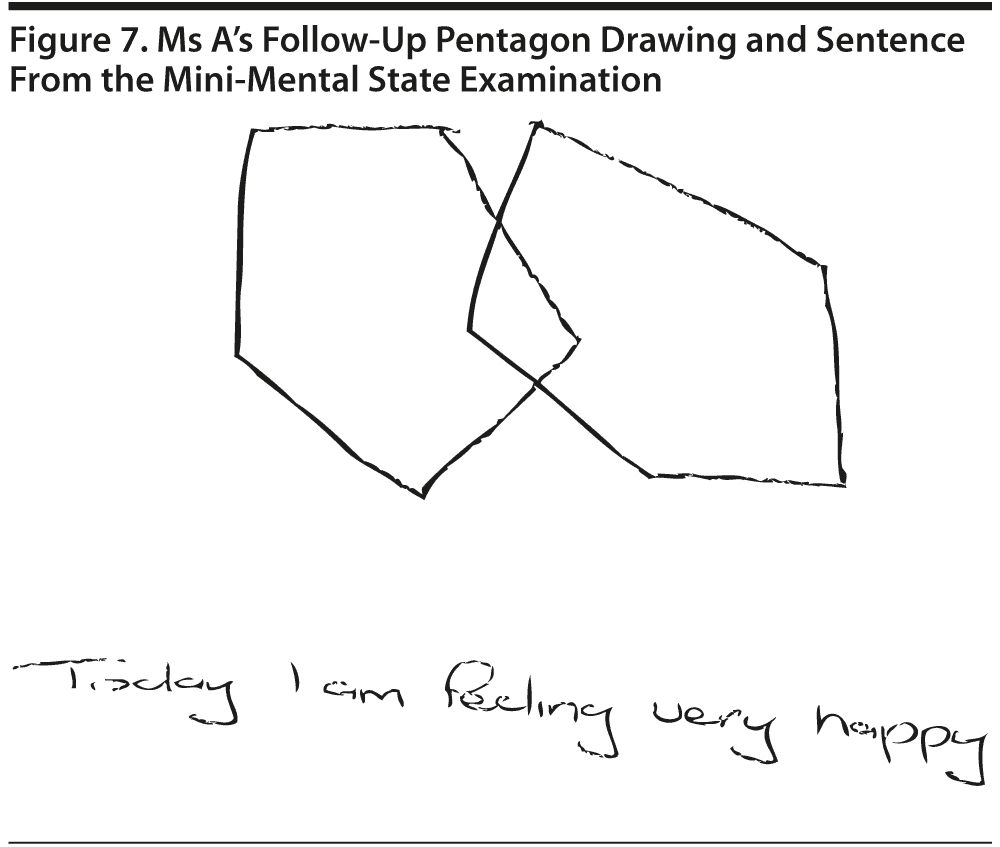
All conference attendees believed that her MMSE normalized (MMSE score: 30/30). Figure 7 shows Ms A’s follow-up pentagon drawing and sentence from the MMSE.
Based on the clinical history, what do you think the Category Retrieval Test score is at this follow-up visit?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 21-25 | 46% |
| B. 16-20 | 54% |
| C. 11-15 | 0% |
| D. 6-10 | 0% |
| E. 0-5 | 0% |
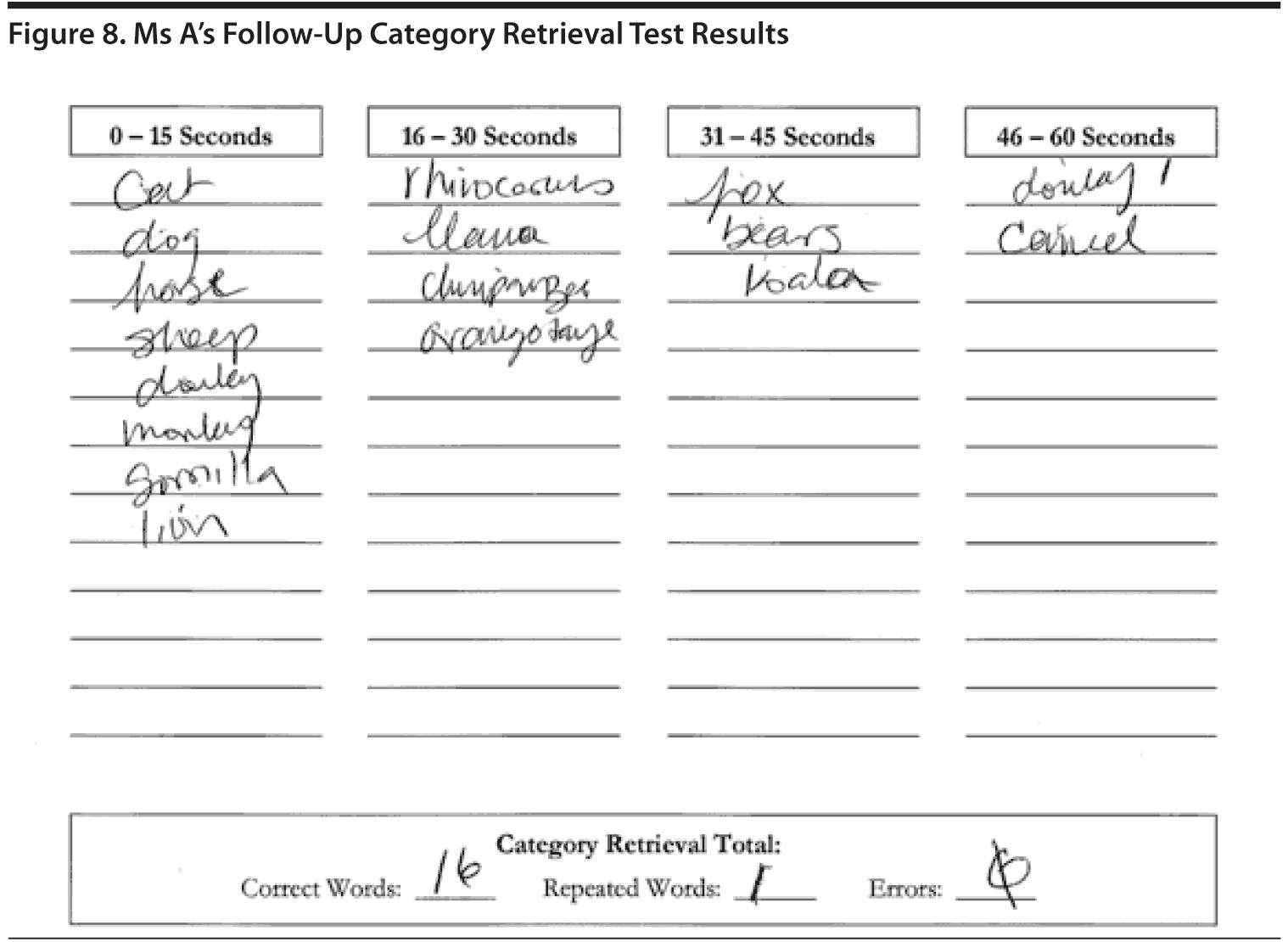
Figure 8 shows Ms A’s Category Retrieval Test results.
Based on the clinical history, what do you think the MoCA score is at this follow-up visit?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 26-30 | 79% |
| B. 21-25 | 21% |
| C. 16-20 | 0% |
| D. 11-15 | 0% |
| E. < 11 | 0% |
Figures 9 and 10 show Ms A’s follow-up clock drawing and MoCA results, respectively.
THE Banner Alzheimer’s Institute TREATING PHYSICIAN’ S IMPRESSION AT 8-WEEK FOLLOW-UP
Ms A is a 43-year-old woman with a status of post-delayed posthypoxic leukoencephalopathy from which it appears that she has nearly recovered. She gives a very clear and detailed history and is fully appropriate; she denies suicidal ideation. She does not appear depressed at this time and appears to be coping well. Ms A does not have dementia.
At this time, is a repeat MRI of the brain indicated?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Yes | 93% |
| B. No | 7% |
Nearly all conference attendees answered “yes”; however, after a discussion, all were in agreement that an MRI would be indicated only for the purposes of curiosity. Although the treating physician was indeed quite curious, it was not felt that a repeat MRI was indicated at this time.
THE Banner Alzheimer’s Institute TREATING PHYSICIAN’ S PLAN AT 8-WEEK FOLLOW-UP
- Continue divalproex sodium 500 mg daily.
- Encourage follow-up with psychiatric outpatient care
- On the basis of the assessment today, it appears that Ms A has the capacity to make decisions for herself regarding personal matters including medical and financial decisions.
- On the basis of the testing today, there is no specific reason why Ms A should not be driving; however, a formal on-road driving safety test is recommended to make sure that her driving is safe.
- Consider a repeat MRI in the future if clinically indicated, but at this time, a repeat MRI would not affect clinical management.
- Follow up on an as-needed basis.
12-WEEK FOLLOW-UP WITH THE Banner Alzheimer’s Institute TREATING PHYSICIAN
Ms A presents with her husband who supplemented the clinical history. Since the last visit, Ms A feels that she is 100% back to herself. Her husband corroborates this report. She is seeing a psychiatrist and counselor and continues to deny suicidal ideation. There continues to be significant marital strife. There are marital discordance issues at this time as well as financial stressors. Ms A passed a formal driving assessment and has been driving without complication.
Impression
Ms A is a 43-year-old woman diagnosed with delayed posthypoxic leukoencephalopathy who has made a remarkable recovery. Ms A continues to deny suicidal ideation and does not appear to be depressed at this time. She does not have dementia. She appears to have recovered from her delayed posthypoxic leukoencephalopathy and seems to be doing quite well at this time. There are significant marital and financial stressors.
Plan
- Continue follow up with the psychiatrist and counselor.
- Continue divalproex sodium 500 mg daily for now; Ms A’s psychiatrist will determine whether it can be discontinued.
- Discussed the possibility of repeating an MRI. If indicated in the future, we can consider this, but at the current time, we will defer.
- Recommended continued counseling.
- Strongly recommended that Ms A find employment to help with self-esteem and her psychiatric care as well.
- Follow up on an as-needed basis.
DISCUSSION
Delayed posthypoxic leukoencephalopathy is a rare complication resulting from any event that causes prolonged cerebral hypooxygenation. Often, there is a full recovery from the initial comatose state, followed days to weeks later by acute neuropsychiatric findings such as disorientation, amnesia, hyperreflexia, frontal release signs, parkinsonism, akinetic-mutism, or psychosis. MRI of the brain demonstrates diffuse cerebral demyelination. Both the pathophysiologic mechanism and the reason why it affects only a subset of hypoxicischemic patients remain unclear. It is hypothesized that prolonged moderate hypooxygenation of subcortical white matter can disrupt adenosine-5′ ²-triphosphate-dependent enzymatic pathways involved in myelin turnover, resulting in delayed demyelination (Ginsberg, 1979). For a comprehensive overview of delayed posthypoxic leukoencephalopathy, the article by Shprecher and Mehta (2010) is recommended.
Ms A’s premorbid history did not include any psychiatric diagnosis, and she did not experience depression following the delayed posthypoxic leukoencephalopathy. However, given the complexity of her course, not altering the divalproex was reasonable pending a diagnostic reevaluation by her psychiatrist once she was stable, as she may not have met diagnostic criteria for bipolar disorder, which was the presumptive diagnosis given by the inpatient hospital team. Her ongoing counseling will also address alcohol/substance abuse, for which she may be at risk, as well as marital issues.
Disclosure of off-label usage
The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
FINANCIAL DISCLOSURE
Dr Yaari is a consultant for Amedisys Home Health. Dr Tariot has served as a consultant for Acadia, AC Immune, Allergan, Eisai, Epix, Forest, Genentech, MedAvante, Memory Pharmaceuticals, Myriad, Novartis, Sanofi-Aventis, Schering-Plough, and Worldwide Clinical Trials; has received consulting fees and grant/research support from Abbott, AstraZeneca, Avid, Baxter, Bristol-Myers Squibb, Elan, Eli Lilly, GlaxoSmithKline, Medivation, Merck, Pfizer, Toyama, and Wyeth; has received educational fees from Alzheimer’s Foundation of America; has received other research support from Alzheimer’s Association, Arizona Department of Health Services, GE, Institute for Mental Health Research, Janssen, National Institute of Mental Health, and National Institute on Aging; has received honoraria from AstraZeneca, Eisai, Eli Lilly, and Pfizer; is a stock shareholder in Adamas and MedAvante; and holds a patent for “Biomarkers of Alzheimer’s Disease.” Drs Seward, Burke, and Fleisher and Ms Brand have no personal affiliations or financial relationships with any commercial interest to disclose relative to the activity.
FUNDING/SUPPORT
None reported.
DISCLAIMER
The opinions expressed are those of the authors, not of Banner Health or Physicians Postgraduate Press.
Clinical Points
- Delayed posthypoxic leukoencephalopathy is a rare complication resulting from any event that causes prolonged cerebral hypooxygenation.
- On rare occasions, a full recovery from an initial comatose state can be followed days to weeks later by acute neuropsychiatric findings such as disorientation, amnesia, hyperreflexia, frontal release signs, parkinsonism, akinetic-mutism, or psychosis.
This CME activity is expired. For more CME activities, visit cme.psychiatrist.com.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
References
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state.” A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198. doi:10.1016/0022-3956(75)90026-6 PubMed
Ginsberg MD. Delayed neurological deterioration following hypoxia. Adv Neurol. 1979;26:21-44. PubMed
Gollan TH, Montoya RI, Werner GA. Semantic and letter fluency in Spanish-English bilinguals. Neuropsychology. 2002;16(4):562-576. doi:10.1037/0894-4105.16.4.562 PubMed
Links KA, Merims D, Binns MA, et al. Prevalence of primitive reflexes and parkinsonian signs in dementia. Can J Neurol Sci. 2010;37(5):601-607. PubMed
Mitrushina M, Boone KB, Razani J, et al. Handbook of Normative Data for Neuropsychological Assessment. 2nd ed. New York, NY: Oxford University Press; 2005.
Mungas D. In-office mental status testing: a practical guide. Geriatrics. 1991;46(7):54-58, 63, 66. PubMed
Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699. doi:10.1111/j.1532-5415.2005.53221.x PubMed
Shprecher D, Mehta L. The syndrome of delayed post-hypoxic leukoencephalopathy. NeuroRehabilitation. 2010;26(1):65-72. PubMed
Enjoy this premium PDF as part of your membership benefits!