Abstract
Objective: Depression and cardiovascular disease (CVD) commonly co-occur in adults aged ≥50 years and older, yet the relationship between depressive symptom frequency and specific cardiovascular outcomes remains underexplored. The objective of this study was to investigate the association between symptom frequency and CVD across demographic and behavioral subgroups.
Methods: We analyzed data from 16,257 participants aged ≥50 years in the National Health and Nutrition Examination Survey (2010–2020). Depressive symptoms were categorized by self-reported frequency. CVD outcomes included coronary heart disease (CHD), myocardial infarction (MI), angina, and congestive heart failure (CHF). Survey-weighted logistic regression models estimated adjusted odds ratios (ORs) for cardiovascular outcomes by depressive symptom frequency, stratified by sex, race/ethnicity, body mass index (BMI), and smoking status.
Results: Depressive symptoms were reported by 24% of participants, with women more represented in moderate categories and men predominating in the highest-frequency group. Non-Hispanic black and Mexican American participants showed greater symptom burden. CVD prevalence increased stepwise with symptom frequency: CHD=7.3%–18.2% and MI=7.0%–13.6% (P<.001). Median BMI was significantly higher among those with depressive symptoms across all CVD subtypes. Co-occurring depressive symptoms with obesity(BMI ≥30, ORs= 3.2 and 3.1) or current smoking (ORs= 3.6 and 3.5) tripled the odds of CHD or angina and CHF or MI, respectively, compared to counterparts without these risk factors. Co-occurrence of CVD subtypes was more common in those with depressive symptoms, particularly in adults aged ≥65 years.
Conclusion: Depressive symptom frequency was independently associated with elevated CVD risk, especially in the presence of obesity or smoking. These findings support integrated screening and prevention strategies targeting both mental health and cardiometabolic risk in adults aged ≥50 years.
Prim Care Companion CNS Disord 2026;28(2):25m04116
Author affiliations are listed at the end of this article.
Depression and cardiovascular disease (CVD) are two of the most frequently encountered conditions in primary care settings, each associated with substantial morbidity, mortality, and economic burden.1,2 Mounting evidence suggests that these conditions often co-occur, with depression functioning as both a risk factor for, and a consequence of, various cardiovascular outcomes.3 In clinical practice, the detection of depression in patients with comorbid CVD remains challenging, yet crucial, due to its impact on adherence, functional status, and prognosis.4 Recent meta-analyses and longitudinal studies confirm that depressive symptoms are independently associated with increased risk of coronary heart disease (CHD), myocardial infarction (MI), heart failure, and all-cause cardiovascular mortality.3,5 Several biological and behavioral mechanisms have been proposed, including systemic inflammation, autonomic nervous system dysfunction, hypothalamic-pituitary-adrenal axis dysregulation, and maladaptive health behaviors such as poor medication adherence, smoking, and sedentary lifestyle.6–8 These pathways are particularly relevant in older adults, who often present with overlapping somatic symptoms that may mask underlying psychological distress.9,10
Importantly, most studies examining the depression-CVD relationship have focused on clinical depression diagnoses or aggregate psychological distress scores. Fewer have explored the role of depressive symptom frequency, which may capture gradations of mood disturbance and offer insights into risk stratification within real-world populations. Symptom frequency may serve as a more actionable signal for primary care providers, who routinely assess mood symptoms during time-limited visits and must prioritize interventions based on severity and risk.11,12 The National Health and Nutrition Examination Survey (NHANES) provides an opportunity to address this gap using standardized assessments in a nationally representative US cohort. NHANES data have been extensively used to inform public health policy and clinical research across a broad range of cardiometabolic and psychiatric conditions.13,14
This study aims to examine the association between depressive symptom burden and CVD among US adults aged ≥50 years using this nationally representative dataset, with a primary focus on symptom frequency across clinically relevant categories. In addition to evaluating gradient effects by symptom frequency, we compare individuals with and without depressive symptoms to contextualize frequency-based patterns across composite and subtype-specific cardiovascular outcomes. We also examine whether these associations are modified by obesity and smoking status. Analyses are further stratified by demographic factors, including age, sex, and race/ethnicity, to explore potential disparities in risk patterns across population subgroups. The goal is to inform clinically actionable risk stratification and prevention in primary care settings serving older adults.
METHODS
Study Design and Population
This cross-sectional analysis employed data from the NHANES, conducted by the Centers for Disease Control and Prevention. NHANES utilizes a stratified, multistage probability sampling design to yield estimates representative of the civilian, noninstitutionalized US population. We pooled data from survey cycles spanning 2010 to March 2020 pre–COVID-19 pandemic.
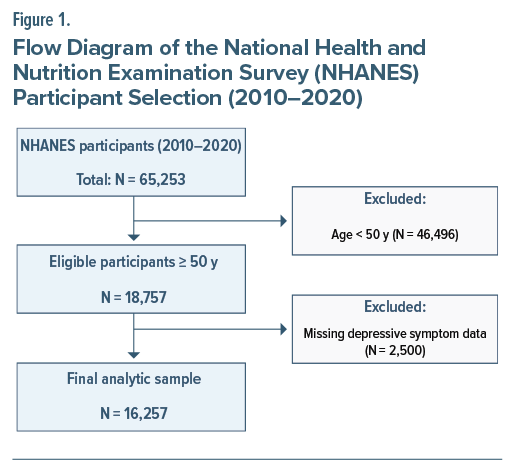
From an initial combined sample of 65,253 participants, eligibility was restricted to individuals aged ≥50 years. Participants were sequentially excluded for the following reasons: age <50 years (n=46,496), missing mental health data (n=2,500), or incomplete cardiovascular or demographic information (number suppressed to preserve anonymity). After applying all exclusion criteria, the final analytic cohort comprised 16,257 participants (Figure 1).
The NHANES protocol was approved by the National Center for Health Statistics Institutional Review Board, and all participants provided written informed consent. Because the public-use dataset is deidentified, the secondary analyses conducted here were exempt from additional institutional review at the authors’ institutions.
Measurement of Depressive Symptoms and CVD
Depressive symptoms were assessed using a single self-report item administered in NHANES: “Over the past two weeks, how often have you felt down, depressed, or hopeless?” Response options included the following: not at all, several days, more than half the days, nearly every day, don’t know, and refused. We retained the “don’t know” and “refused” categories to examine whether nonresponse itself carried cardiovascular risk associations.
These categories were treated as an ordinal measure of depressive symptom frequency, reflecting increasing symptom burden. Symptom frequency was the primary exposure of interest; however, for descriptive and comparative purposes, a secondary binary categorization was also used, grouping participants as having depressive symptoms (several days or more frequent) vs no depressive symptoms (not at all).
CVD history was defined by participants’ self-report of physician diagnoses for CHD, MI, congestive heart failure (CHF), or angina pectoris. Each condition was coded as a binary variable (presence = 1 and absence = 0). For analyses of overall CVD burden, a composite “any CVD” indicator was also constructed.
Covariates
We included demographic and behavioral covariates known to confound associations between mood, obesity, and CVD. Demographic variables comprised age (continuous), sex, and race/ethnicity (non-Hispanic white, non-Hispanic black, Mexican American, other Hispanic, other race). Body mass index (BMI) was calculated (kg/m2) based on measured weight and height; participants were categorized as normal (<25), overweight (25–29.9), or obese (≥30). Smoking status was classified as current, former, or never smoker.
Statistical Analysis
All analyses incorporated the NHANES complex sampling structure, including sampling weights, primary sampling units, and strata, using the survey package in R (version 4.0 or later). Descriptive statistics were reported as medians with interquartile ranges (IQRs) for continuous variables and frequencies with weighted proportions for categorical variables. Differences across depressive symptom groups were tested using survey-adjusted χ² tests (for categorical variables) and survey-weighted Kruskal-Wallis tests (for continuous variables).
To examine the association between depressive symptoms and cardiovascular risk, survey-weighted multivariable logistic regression models were used to estimate adjusted odds ratios (ORs). Separate models were constructed for 2 composite cardiovascular outcomes: (1) CHD or angina and (2) CHF or MI. The reference group for depressive symptoms was “not at all.” Covariates in the models included age, sex, race/ ethnicity, BMI category, and smoking status. Predictor categories of “don’t know” and “refused” were retained for completeness but not emphasized in interpretation. A 2-sided P value <.05 was considered statistically significant.
RESULTS
Participant Characteristics by Depressive Symptom Frequency
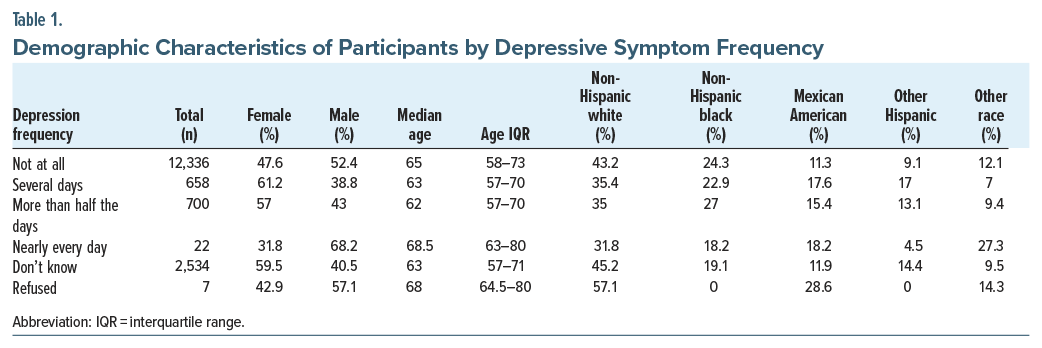
In the analytic cohort of 16,257 participants aged ≥50 years, depressive symptoms were distributed as follows: not at all (n = 12,336), several days (n = 658), more than half the days (n = 700), nearly every day (n = 22), don’t know (n = 2,534), and refused (n = 7). Women comprised 50.4% of the overall sample and were more frequently represented among those reporting symptoms on several days (61.2%) and more than half the days (57.0%). Men predominated in the “nearly every day” group (68.2%) and among those without symptoms (52.4%). Median age differed significantly across categories (P < .001), ranging from 62.0 years (IQR = 57.0–70.0) in the “more than half the days” group to 68.5 years (IQR = 63.0–80.0) in the “nearly every day” group. Racial and ethnic distributions also varied significantly (P < .001), with non-Hispanic whites comprising the largest subgroup overall (42.8%). Non-Hispanic blacks accounted for 23.5% of the overall cohort, with slightly higher representation among those with moderate symptom frequency. Mexican Americans represented 11.8% overall, with increased proportions in groups reporting more frequent symptoms. Individuals identifying as other Hispanic or other race, including multiracial, showed smaller but variable representation across categories.
Prevalence of CVD by Symptom Frequency
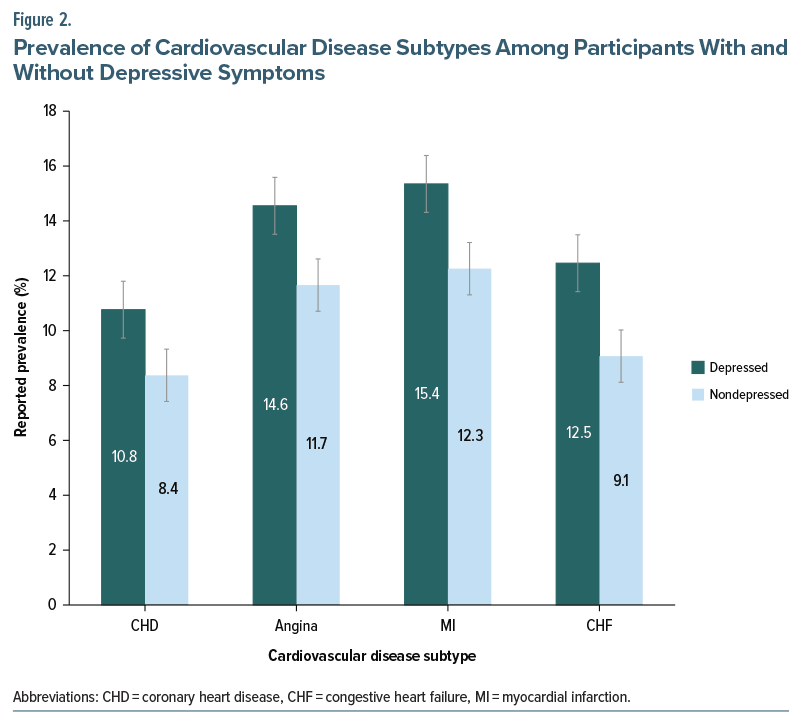
Overall, 8.5% reported a history of CVD, inclusive of CHD, MI, CHF, or angina pectoris. Overall, participants categorized as having depressive symptoms (ie, reporting symptoms on several days, more than half the days, or nearly every day) exhibited higher prevalence of CVD subtypes than their nondepressed counterparts: CHD (10.8% vs 8.4%), angina (14.6% vs 11.7%), MI (15.4% vs 12.3%), and CHF (12.5% vs 9.1%) (all P <.001) (Figure 2).
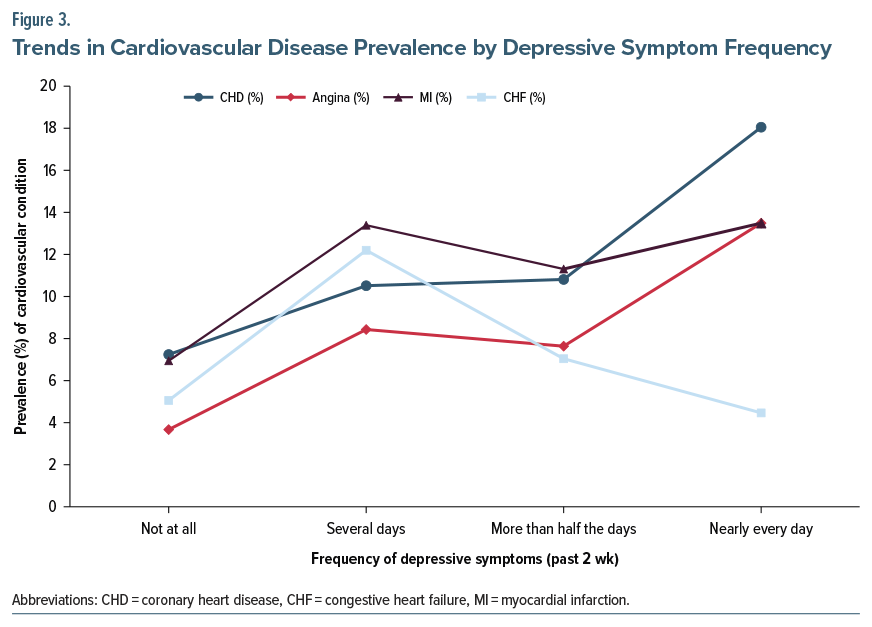
When stratified by depressive symptom frequency, a stepwise increase in prevalence was evident for each CVD subtype across the response categories (Table 1, Figure 3). For CHD, prevalence rose from 7.3% in the “not at all” group to 10.6% (“several days”), 10.9% (“more than half the days”), and 18.2% (“nearly every day”). For angina, corresponding prevalence values were 3.7%, 8.5%, 7.7%, and 13.6%, respectively. MI prevalence was 7.0%, 13.5%, 11.4%, and 13.6%, respectively, while CHF prevalence was 5.1%, 12.3%, 7.1%, and 4.5%, respectively. All trend tests across frequency categories were statistically significant (P < .001) (Figure 2). Notably, co-occurrence of CVD subtypes was more frequent among participants with depressive symptoms: Among those with CHD, 68% of depressed individuals also reported angina compared to 57% of nondepressed participants (P < .001). Similarly, among CHF patients, 71% of depressed participants also reported a prior MI vs 62% without depression. These overlaps were most pronounced in individuals aged ≥65 years.
Association Between Depression and BMI
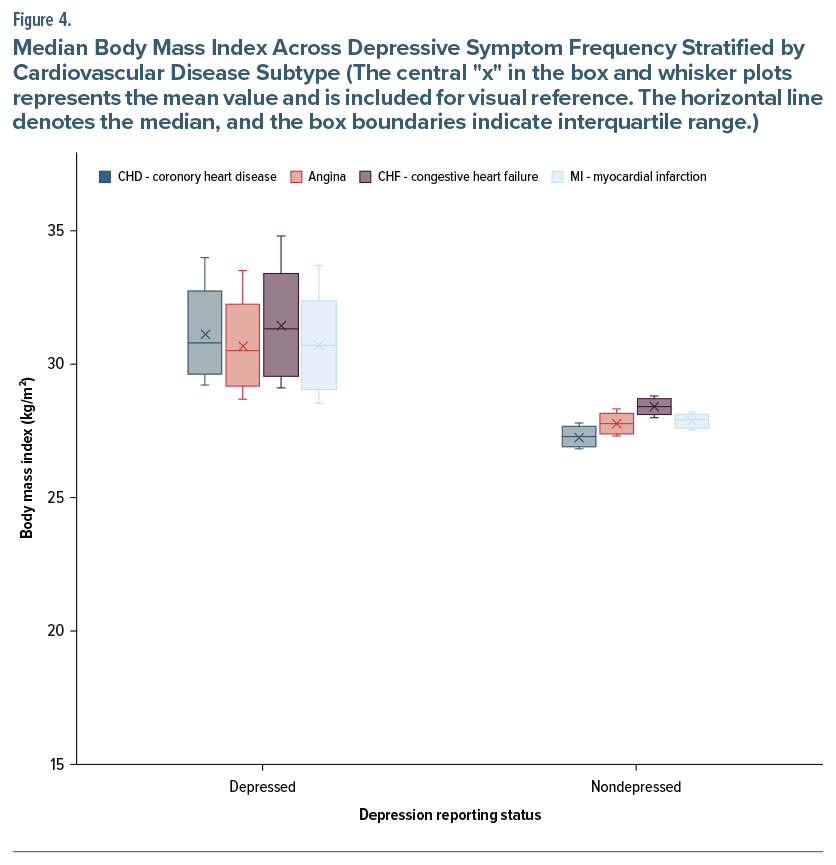
Box-and-whisker plot analysis demonstrated a consistent relationship between depressive symptoms and elevated BMI among participants with CVD. Individuals with both CHD and depressive symptoms exhibited a median BMI of 30.8 kg/m2 (IQR=29.2–34.0), whereas those without depressive symptoms had a median BMI of 27.3 kg/m2. Among participants with angina pectoris, the median BMI was 30.5 kg/m2 (IQR= 28.7–33.5) for those with depressive symptoms compared to 27.8 kg/m2 in those without. In the case of CHF, the median BMI was 31.3 kg/m2 (IQR=29.1–34.8) for depressed participants and 28.4 kg/m2 for those who were not depressed. For MI, the median BMI was 30.7 kg/m2 (IQR=28.5–33.7) vs 27.9 kg/m2, respectively. All comparisons were statistically significant (P <.001) (Figure 4).
Combined Impact of Depression, Obesity, and Smoking on CVD Risk
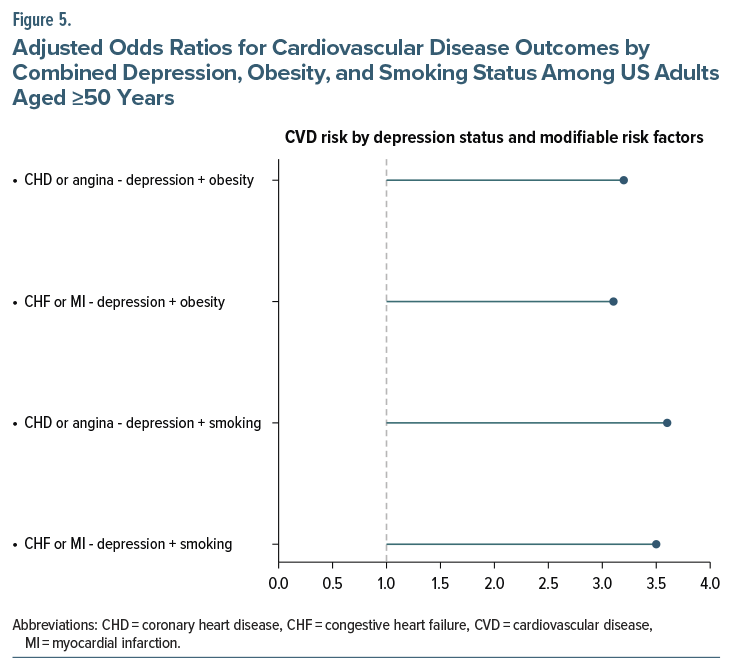
In addition to individual BMI differences, the co-occurrence of depressive symptoms with obesity and smoking was associated with significantly increased odds of CVD. Participants with depressive symptoms and a BMI ≥30 kg/m2 had 3.2 times higher odds of having CHD or angina and 3.1 times higher odds of having CHF or MI, when compared with individuals without depressive symptoms and with normal weight. Similarly, current smokers with depressive symptoms demonstrated a 3.6-fold increase in the odds of CHD or angina and a 3.5-fold increase in the odds of CHF or MI, relative to never-smokers without depressive symptoms. All associations were statistically significant (P <.001) (Figure 5).
DISCUSSION
In this nationally representative cohort of adults aged ≥50 years, we observed a graded, frequency-dependent relationship between depressive symptoms and CVD. Higher symptom frequency corresponded to increased prevalence of CHD, angina, MI, and CHF. The combined presence of depressive symptoms with either obesity or current smoking substantially elevated the odds of CVD, suggesting interactive and compounding risk factors.
These findings reinforce existing literature linking depression to poor cardiovascular outcomes. A 2023 meta-analysis of nearly 2 million individuals found that depression increased the risk of MI (hazard ratio [HR]=1.28), stroke (HR =1.13), and overall CVD (HR= 1.16), as well as all-cause and cardiovascular mortality.1,15
Approximately 1 in 5 patients with CVD meet criteria for major depression, especially those with heart failure or coronary artery disease, where depression predicts recurrent events and higher mortality.16,17 Importantly, this association is bidirectional, with CVD itself increasing the risk of depression through mechanisms such as inflammation, reduced mobility, and psychological stress.18,19
While prior studies have relied on clinical diagnoses or screening scores, our use of a single-item, frequency-based assessment reflects a practical and scalable approach common in primary care, enabling evaluation of depressive symptoms along a continuum that may better capture subthreshold states relevant to cardiovascular risk. Emerging evidence suggests that subthreshold or infrequent depressive symptoms are also associated with increased cardiovascular risk, underscoring the need to assess symptom frequency along a continuum. Approximately 11% of US adults report ≥4 depressive symptoms, and this subgroup faces a 14% increased risk of incident CVD and a 17% higher all-cause mortality.15 Our findings mirror this gradient: Participants reporting symptoms “nearly every day” had CHD and MI prevalence of 18.2% and 13.6%, respectively, compared with 7.3% and 7.0% among those without symptoms. Participants with moderate symptom frequency showed intermediate risk (CHD: 10.6%, MI: 13.5%), highlighting a dose-response association between mood severity and cardiac morbidity. This symptom-based approach also reflects how depression often presents in primary care, wherein patients may express subclinical emotional distress and brief screening may guide clinical decisions.
The association varied across sex, race, and ethnicity. Women were more represented in moderate symptom categories, which may reflect greater help-seeking or symptom disclosure, while men were overrepresented among those reporting daily symptoms, consistent with delayed engagement until severity is high.20,21 Non-Hispanic black individuals comprised 23.5% of the cohort, with increased representation in moderate symptom groups. Mexican Americans, representing 11.8% of the sample, were more prevalent among those with frequent symptoms. These disparities are consistent with prior evidence suggesting that depression in minority groups is often underrecognized yet biologically consequential, reflecting cumulative psychosocial stress and systemic inequities.22–24
The observed amplification of CVD risk among participants with both depressive symptoms and obesity reflects established pathophysiologic links through inflammatory, neuroendocrine, behavioral, and genetic pathways.25,26 In our study, individuals with depressive symptoms and obesity (BMI≥30 kg/m2) had 3.2 times higher odds of CHD or angina and 3.1 times higher odds of CHF or MI compared to normal-weight individuals without depressive symptoms. Median BMI values were consistently higher among participants with depressive symptoms across all CVD subtypes. These findings align with prior data showing a bidirectional relationship between depression and obesity (risk ratio =1.37), with co-occurrence nearly doubling the risk of coronary events (HR =1.88) and conferring even greater risk among men (HR =2.32).27,28 Current guidelines advocate for routine metabolic screening in psychiatric settings, including glucose, lipid profiles, and waist circumference, while recommending depression screening in primary care for older adults with high cardiometabolic burden. Metformin has shown modest benefits in reducing weight and insulin resistance in psychiatric populations.29,30 In addition, glucagon-like peptide 1 receptor agonists such as semaglutide are gaining attention for their cardiovascular benefits and potential mood-enhancing effects.31 For older adults with depressive symptoms and elevated cardiometabolic risk, adopting these dual-pathway strategies may support improved mental health and cardiovascular outcomes.
Smoking further compounded cardiovascular risk. In our study, current smokers with depressive symptoms had over 3-fold higher odds of CHD or angina (OR =3.6) and CHF or MI (OR=3.5) compared to never-smokers without depressive symptoms. This pattern reflects prior longitudinal evidence demonstrating additive effects of tobacco use and depression on cardiac outcomes, driven by shared behavioral vulnerabilities and physiologic dysregulation such as inflammation, autonomic imbalance, and poor adherence.32,33 Additionally, smoking may predispose individuals to depression, reinforcing a bidirectional and self-perpetuating risk cycle.34,35 The American College of Cardiology recommends combining behavioral counseling with pharmacologic treatments such as varenicline, which has shown the highest cessation rates in patients with depression.36,37 In older adults with depressive symptoms, smoking history should prompt both cardiometabolic screening and tailored cessation strategies, serving as an opportunity for integrated care.
Despite growing evidence linking depressive symptoms with CVD, their comanagement remains suboptimal in clinical practice due to intersecting barriers at the patient, provider, and system levels. Stigma and fears about medication side effects contribute to underreporting of symptoms, while providers may under-recognize depression due to overlapping somatic complaints or time constraints in primary care. Polypharmacy concerns and limited access to psychiatric services further complicate treatment. Our findings suggest that even subclinical depressive symptom frequency may be a useful clinical signal warranting attention in cardiovascular risk assessment. These gaps highlight the need for integrated, proactive strategies that prioritize early recognition, patient-centered education, and collaborative care models tailored to the aging population.
Limitations
This study has several limitations. First, the cross-sectional design precludes determination of causal directionality between depressive symptoms and CVD. Second, both depression and cardiovascular outcomes were based on self-report and may be subject to recall or reporting bias. The depression measure relied on a single-item frequency question rather than a validated scale or clinical diagnosis, which may limit granularity. Third, the analysis did not account for antidepressant use, duration of depressive symptoms, or comorbid psychiatric conditions that may confound the observed associations.
Clinical Implications and Future Directions
For physicians managing aging populations, our findings underscore the importance of integrating mental health and cardiometabolic risk assessment into routine care. While diagnostic thresholds remain essential for identifying clinical depression, incorporating symptom frequency may support earlier risk stratification and enhanced clinical vigilance. Evidence from randomized trials demonstrates that primary care–based collaborative care models targeting depression alongside cardiometabolic conditions can improve depressive symptoms, cardiovascular risk factors, and overall medical outcomes.38–42 Future research should examine the long-term cardiovascular outcomes and implementation effectiveness of such integrated care models in diverse aging populations, particularly across varied health care settings. Pragmatic trials embedded in primary care and chronic disease management settings could evaluate collaborative care models that combine behavioral, pharmacologic, and nonpharmacologic strategies. Special focus is warranted on high-risk subgroups such as individuals with obesity or nicotine use and on culturally responsive approaches that address the under-recognition of depression in minority populations.
Article Information
Published Online: March 3, 2026. https://doi.org/10.4088/PCC.25m04116
© 2026 Physicians Postgraduate Press, Inc.
Submitted: October 22, 2025; accepted December 19, 2025.
To Cite: Ahmed F, Awan AI, Kamel O, et al. Depression and cardiovascular burden in aging populations: insights from the National Health and Nutrition Examination Survey 2010–2020 on symptom frequency, sociodemographic disparities, and modifiable risk factors. Prim Care Companion CNS Disord 2026;28(2):25m04116.
Author Affiliations: Jersey Shore University Medical Center, Neptune City, New Jersey (Ahmed); Maimonides Medical Center, Brooklyn, New York (Awan); South Valley University, Egypt (Kamel); King Edward Medical University, Lahore, Pakistan (Waheed); Ameer-ud-Din Medical College, Lahore, Pakistan (Afzaal); Batterjee Medical College, Jeddah, Saudi Arabia (Aman).
Corresponding Author: Ali Imam Awan, MD, Maimonides Medical Center, Brooklyn, New York ([email protected]).
Financial Disclosure: None.
Funding/Support: None.
Additional Information: This study utilized publicly available data from the National Health and Nutrition Examination Survey, conducted by the Centers for Disease Control and Prevention. All data are accessible at https://www.cdc.gov/nchs/nhanes/.
Clinical Points
- Even infrequent depressive symptoms are associated with elevated cardiovascular risk in adults aged ≥50 years; assessing symptom frequency, rather than relying solely on diagnostic thresholds, can support early risk stratification in primary care.
- Patients who report both depressive symptoms and either obesity or current smoking have substantially higher odds of cardiovascular disease; this pattern underscores the importance of integrated screening for both mood and modifiable cardiometabolic risks.
- Incorporating brief and scalable depression screening tools into routine cardiovascular risk assessments may help clinicians identify high-risk patients and initiate timely, collaborative care interventions.
References (42)

- Krittanawong C, Maitra NS, Qadeer YK, et al. Association of depression and cardiovascular disease. Am J Med. 2023;136(9):881–895. PubMed CrossRef
- GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396(10258):1204–1222. PubMed CrossRef
- Zeng J, Qiu Y, Yang C, et al. Cardiovascular diseases and depression: a meta-analysis and Mendelian randomization analysis. Mol Psychiatry. 2025;30(9):4234–4246. PubMed CrossRef
- Smith SM, Wallace E, O’Dowd T, et al. Interventions for improving outcomes in patients with multimorbidity in primary care and community settings. Cochrane Database Syst Rev. 2021;2021(1):CD006560.
- Harshfield EL, Pennells L, Schwartz JE, et al. Association between depressive symptoms and incident cardiovascular diseases. JAMA. 2020;324(23):2396–2405. PubMed CrossRef
- Slusher AL, Acevedo EO. Stress induced proinflammatory adaptations: plausible mechanisms for the link between stress and cardiovascular disease. Front Physiol. 2023;14:1124121. PubMed CrossRef
- Pan A, Sun Q, Okereke OI, et al. Depression and the risk of stroke morbidity and mortality: a meta-analysis and systematic review. JAMA. 2011;306(11):1241–1249. PubMed CrossRef
- Penninx BWJH. Depression and cardiovascular disease: epidemiological evidence on their linking mechanisms. Neurosci Biobehav Rev. 2017;74(Pt B):277–286. PubMed CrossRef
- Levine GN, Cohen BE, Commodore-Mensah Y, et al. Psychological health, well-being, and the mind-heart-body connection: a scientific statement from the American Heart Association. Circulation. 2021;143(10):e763–e783. PubMed CrossRef
- Hou R, Westbury L, Fuggle N, et al. Immune-endocrine biomarkers associated with mental health: a 9-year longitudinal investigation from the Hertfordshire Ageing Study. Brain Behav Immun. 2022;101:146–152. PubMed CrossRef
- Park LT, Zarate CA. Depression in the primary care setting. New Engl J Med. 2019;380(6):559–568. PubMed CrossRef
- Castellini G, Pecchioli S, Cricelli I, et al. How to early recognize mood disorders in primary care: a nationwide, population-based, cohort study. Eur Psychiatry. 2016;37:63–69. PubMed CrossRef
- Gou R, Cao H, Dou D, et al. Association of Health Related Social Needs (HRSN) with depression in NHANES (2003-2023): mediating effects of frailty. J Affect Disord. 2025;386:119410. PubMed CrossRef
- Liang X, Or B, Tsoi MF, et al. Prevalence of metabolic syndrome in the United States national health and nutrition examination survey 2011-18. Postgrad Med J. 2023;99(1175):985–992. PubMed CrossRef
- Rajan S, McKee M, Rangarajan S, et al. Association of symptoms of depression with cardiovascular disease and mortality in low-middle-and high-income countries. JAMA Psychiatry. 2020;77(10):1052–1063. PubMed CrossRef
- Subramaniapillai M, Chen VCH, McIntyre RS, et al. Added burden of major depressive disorder on cardiovascular morbidity and mortality among patients with cardiovascular disease and the modifying effects of antidepressants: a national retrospective cohort study. J Affect Disord. 2021;294:580–585. PubMed CrossRef
- Feng Z, Tong WK, Tang Z. Longitudinal trends in the prevalence and treatment of depression among adults with cardiovascular disease: an analysis of national health and nutrition examination survey 2009-2020. Front Psychiatry. 2022;13:943165. PubMed CrossRef
- Halaris A. Inflammation-associated Co-morbidity between depression and cardiovascular disease. Curr Top Behav Neurosci. 2017;31:45–70. PubMed CrossRef
- Huang L, Zhang L, Liu C, et al. Interaction and mechanisms of depression and cardiovascular disease: a mini-review. PeerJ. 2025;13:e20148. PubMed CrossRef
- Roxo L, Silva M, Perelman J. Gender gap in health service utilisation and outcomes of depression: a cross-country longitudinal analysis of European middle-aged and older adults. Prev Med. 2021;153:106847. PubMed CrossRef
- Wang K, Lu H, Cheung EFC, et al. “Female preponderance” of depression in non-clinical populations: a meta-analytic study. Front Psychol. 2016;7:1398. PubMed CrossRef
- Jester DJ, Kohn JN, Tibiriçá L, et al. Differences in social determinants of health underlie racial/ethnic disparities in psychological health and well-being: study of 11,143 older adults. Am J Psychiatry. 2023;180(7):483–494. PubMed CrossRef
- Webb EK, Carter SE, Ressler KJ, et al. The neurophysiological consequences of racism-related stressors in Black Americans. Neurosci Biobehav Rev. 2024;161:105638. PubMed CrossRef
- Vyas CM, Donneyong M, Mischoulon D, et al. Association of race and ethnicity with late-life depression severity, symptom burden, and care. JAMA Netw Open. 2020;3(3):e201606. PubMed CrossRef
- Capuron L, Lasselin J, Castanon N. Role of adiposity-driven inflammation in depressive morbidity. Neuropsychopharmacology. 2017;42(1):115–128. PubMed CrossRef
- Stapel B, Jelinic M, Drummond GR, et al. Adipose tissue compartments, inflammation, and cardiovascular risk in the context of depression. Front Psychiatry. 2022;13:831358. PubMed CrossRef
- Liu S, Luo J, Zhang T, et al. The combined role of obesity and depressive symptoms in the association with ischaemic heart disease and its subtypes. Sci Rep. 2022;12(1):14419. PubMed CrossRef
- Wang X, Hu Y, Qin LQ, et al. Combined association of central obesity and depressive symptoms with risk of heart disease: a prospective cohort study. J Affect Disord. 2022;297:360–365. PubMed CrossRef
- Apovian CM, Aronne LJ, Bessesen DH, et al. Pharmacological management of obesity: an endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2015;100(2):342–362. PubMed CrossRef
- Elmaleh-Sachs A, Schwartz JL, Bramante CT, et al. Obesity management in adults: a review. JAMA. 2023;330(20):2000–2015. PubMed CrossRef
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):2221–2232. PubMed CrossRef
- Carroll AJ, Huffman MD, Zhao L, et al. Associations between depressive symptoms, cigarette smoking, and cardiovascular health: longitudinal results from CARDIA. J Affect Disord. 2020;260:583–591. PubMed CrossRef
- Carroll AJ, Huffman MD, Zhao L, et al. Evaluating longitudinal associations between depressive symptoms, smoking, and biomarkers of cardiovascular disease in the CARDIA study. Psychosom Med. 2019;81(4):372–379. PubMed CrossRef
- Yan B, Hu J, Xi J, et al. The role of C-reactive proteins in tobacco smoke exposure and the risk of depression. Psychol Health Med. 2025:1–15. PubMed CrossRef
- Wu Z, Yue Q, Zhao Z, et al. A cross-sectional study of smoking and depression among US adults: NHANES (2005-2018). Front Public Health. 2023;11:1081706. PubMed CrossRef
- Cinciripini PM, Kypriotakis G, Green C, et al. The effects of varenicline, bupropion, nicotine patch and placebo on smoking cessation among smokers with major depression: a randomized clinical trial. Depress Anxiety. 2022;39(5):429–440. PubMed CrossRef
- Benowitz NL, Pipe A, West R, et al. Cardiovascular safety of varenicline, bupropion, and nicotine patch in smokers: a randomized clinical trial. JAMA Intern Med. 2018;178(5):622–631. PubMed CrossRef
- Kennedy SH, Lam RW, McIntyre RS, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder: section 3. Pharmacological treatments. Can J Psychiatry. 2016;61(9):540–560. PubMed CrossRef
- Bogner HR, Morales KH, De Vries HF, et al. Integrated management of type 2 diabetes mellitus and depression treatment to improve medication adherence: a randomized controlled trial. Ann Fam Med. 2012;10(1):15–22. PubMed CrossRef
- Khambaty T, Stewart JC, Gupta SK, et al. Association between depressive disorders and incident acute myocardial infarction in human immunodeficiency virus–infected adults. JAMA Cardiol. 2016;1(8):929–937. PubMed CrossRef
- Lin EHB, Von Korff M, Ciechanowski P, et al. Treatment adjustment and medication adherence for complex patients with diabetes, heart disease, and depression: a randomized controlled trial. Ann Fam Med. 2012;10(1):6–14. PubMed CrossRef
- Katon WJ, Lin EHB, Korff MV, et al. Collaborative care for patients with depression and chronic illnesses. New Engl J Med. 2010;363(27):2611–2620. PubMed
Enjoy this premium PDF as part of your membership benefits!