Objective: To study the prevalence of premenstrual dysphoric disorder (PMDD) symptom patterns among women in the United Arab Emirates and to measure the debilitating nature of PMDD symptoms and sociodemographic correlates.
Methods: This cross-sectional sample study used the Mini-International Neuropsychiatric Interview-Plus (MINI-Plus) and Premenstrual Symptoms Screening Tool (PSST) to screen for presence and severity of PMDD symptoms in Arab women attending ambulatory health services in Alain city, Emirate of Abu Dhabi, United Arab Emirates, for routine health care between May 2005 and September 2005.
Results: The study participants include 508 women (76% Emiratis, 15% Omanis, and 8% other Arabs) of childbearing age. In total, 94 women (18.6%) met MINI-Plus criteria for PMDD; of these, 21 (4.1%) met PSST criteria for severe symptoms, 29 (5.7%) for moderate symptoms, and 44 (8.7%) for mild or less symptoms. One woman (0.2%) with severe symptoms and 12 women (2.4%) with moderate symptoms had negative MINI-Plus scores. Presence of PMDD symptoms was significantly associated with higher education (P = .000), single marital status (P = .001), major life stressors (P = .001), and personal/family use of psychotropic medications (P = .000/P = .006), personal/family psychological problems (P = .000/P = .001), irregular/painful menses (P = .043/P = .001), and functional impairment on the Sheehan Disability Scale (P = .000). Multilogistic regression analysis showed higher education, major life stressor, personal use of psychotropic medications, personal/family psychological problem, and painful menses were independent predictors of PMDD symptoms.
Conclusions: PMDD symptoms were common among the Arab women in our study. The cyclically triggered mood disturbances were clustered in women with personal/familial psychological problems, perhaps linking biologic constitution to genetic predisposition for the development of PMDD symptomatology.
Prevalence, Severity, and Correlates of Premenstrual Dysphoric Disorder Symptoms Among Women in the Arabian Peninsula
ABSTRACT
Objective: To study the prevalence of premenstrual dysphoric disorder (PMDD) symptom patterns among women in the United Arab Emirates and to measure the debilitating nature of PMDD symptoms and sociodemographic correlates.
Methods: This cross-sectional sample study used the Mini-International Neuropsychiatric Interview–Plus (MINI-Plus) and Premenstrual Symptoms Screening Tool (PSST) to screen for presence and severity of PMDD symptoms in Arab women attending ambulatory health services in Alain city, Emirate of Abu Dhabi, United Arab Emirates, for routine health care between May 2005 and September 2005.
Results: The study participants include 508 women (76% Emiratis, 15% Omanis, and 8% other Arabs) of childbearing age. In total, 94 women (18.6%) met MINI-Plus criteria for PMDD; of these, 21 (4.1%) met PSST criteria for severe symptoms, 29 (5.7%) for moderate symptoms, and 44 (8.7%) for mild or less symptoms. One woman (0.2%) with severe symptoms and 12 women (2.4%) with moderate symptoms had negative MINI-Plus scores. Presence of PMDD symptoms was significantly associated with higher education (P = .000), single marital status (P = .001), major life stressors (P = .001), and personal/family use of psychotropic medications (P = .000/P = .006), personal/family psychological problems (P = .000/P = .001), irregular/painful menses (P = .043/P = .001), and functional impairment on the Sheehan Disability Scale (P = .000). Multilogistic regression analysis showed higher education, major life stressor, personal use of psychotropic medications, personal/family psychological problem, and painful menses were independent predictors of PMDD symptoms.
Conclusions: PMDD symptoms were common among the Arab women in our study. The cyclically triggered mood disturbances were clustered in women with personal/familial psychological problems, perhaps linking biologic constitution to genetic predisposition for the development of PMDD symptomatology.
Prim Care Companion CNS Disord 2017;19(4):17m02112
https://doi.org/10.4088/PCC.17m02112
© Copyright 2017 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry and Behavioral Sciences, College of Medicine and Health Sciences, UAE University, Alain, United Arab Emirates
bStrategic Planning Division, Abu Dhabi Municipality, United Arab Emirates
cDepartment of Statistics, College of Medicine and Health Sciences, UAE University, Alain, United Arab Emirates
dDepartment of Family Medicine, Tawam Hospital, Alain, United Arab Emirates
eDepartment of Obstetrics and Gynecology, Arab Gulf University, Bahrain
fDepartment of Pediatrics, College of Medicine and Health Sciences, UAE University, Alain, United Arab Emirates
*Corresponding author: Ossama T. Osman, MD, College of Medicine and Health Sciences, UAE University, PO Box 17666, Alain, UAE ([email protected]).
Many women of reproductive age (15–49 years) experience premenstrual symptoms of varying severity. Premenstrual dysphoric disorder (PMDD), however, describes the severe cyclical mood disorder and psychosocial impairment that precede menses.1 The symptoms occur with most menses and are mainly limited to the luteal phases of ovulatory cycles.2,3 At least 2%–8% of women meet the strict criteria of the disorder4 as defined in the DSM-IV and DSM-5, a prevalence that is higher than many DSM Axis I disorders.1
Pathogenicity of PMDD has been recently illustrated. Allopregnanolone (a main derivative of progesterone) is known to affect mood and mood disorders via the action of γ-aminobutyric acid.5 This neuroactive steroid peaks in the luteal phase and declines sharply after the onset of menses; this cyclicity contributes to the etiology of PMDD.5–7 Another reported predisposing factor is the polymorphism methionine allele (G196A) in the brain-derived neurotrophic factor gene, which conveys lower frontocingulate cortex activation during the luteal phase.8 Other potential variables include increased gray matter volume in the posterior cerebellum, increased inflammatory biomarkers, and exposure to significant emotional or physical trauma.8–13
With respect to therapy, serotonergic antidepressants are considered the drugs of choice. The reported rapid response to luteal or continuous dosing of selective serotonin reuptake inhibitors suggests the mechanism of PMDD differs from that of major depressive disorder.14,15 This hasty therapeutic gain may reflect a reversal of the underlying mechanism of PMDD or simply an alleviation of its symptomatology.16 Other useful interventions include cognitive-behavioral therapy, skills training, and nutritional recommendations.17
Prevalence and severity of PMDD symptoms in Arab women have not been previously investigated. This community-based study combined 2 instruments to screen for the presence and severity of the disorder symptoms in women in the region.
METHODS
Women presenting in 5 ambulatory health services in Alain city (Emirate of Abu Dhabi, United Arab Emirates) for routine health care between May 2005 and September 2005 were recruited for this cross-sectional sample study. The study was approved by the Alain Medical District Human Research Ethics Committee Review Board. Informed consent was obtained from each participant.
The study used validated Arabic versions of the Mini-International Neuropsychiatric Interview–Plus (MINI-Plus),18,19 Premenstrual Symptoms Screening Tool (PSST),20 and Sheehan Disability Scale (SDS).21 The surveys were completed by participating women in the presence of trained female staff who were fluent in the Arabic language and well equated to the Arabian culture. Participation was voluntary, and the questionnaire answers were anonymous and confidential.
The MINI-Plus and PSST were used as screening instruments for the presence and severity of PMDD symptomatology.1 The PSST is a 1-page test that transforms categorical DSM-IV criteria into a rating scale with degrees of severity.20 The PSST scores 14 symptoms that start before onset of bleeding and resolve within a few days of bleeding on a 4-point rating scale (not at all [0], mild [1], moderate [2], or severe [3]); the maximum score was 42.
Perceived functional disability was evaluated on the 4 SDS responsibility domains: work/school, social life, family/home, and religious duties.21 This 10-point/domain Visual Analog Scale measured self-reported impairments. The domains were analyzed separately and summed into a single functional impairment score that ranged from zero (unimpaired) to 40 (highly impaired). A score ≥ 5 on any domain or a sum score of 20 necessitated monitoring.21
Statistical Analysis
Simple and multiple logistic regression analyses were performed using the statistical package IBM SPSS (version 20) to investigate the association of several variables with PMDD symptoms. A significance level (α) < .05 was used.
RESULTS
The study included 508 women; their characteristics are shown in Table 1. Thirty-seven percent of the participants had a college degree, 78% had reasonable income, 54% had excess body fat, 85% reported work-related problems, and 60% had illness in the family. Medical problems (≤ 17%) and functional disabilities (≤ 11%, as screened by SDS) were infrequent, while psychological problems (32%), irregular (onset of menses varied > 7 days each month) menstrual cycles (28%), and low back/pelvic pain during menses (73%) were relatively common (Table 1).

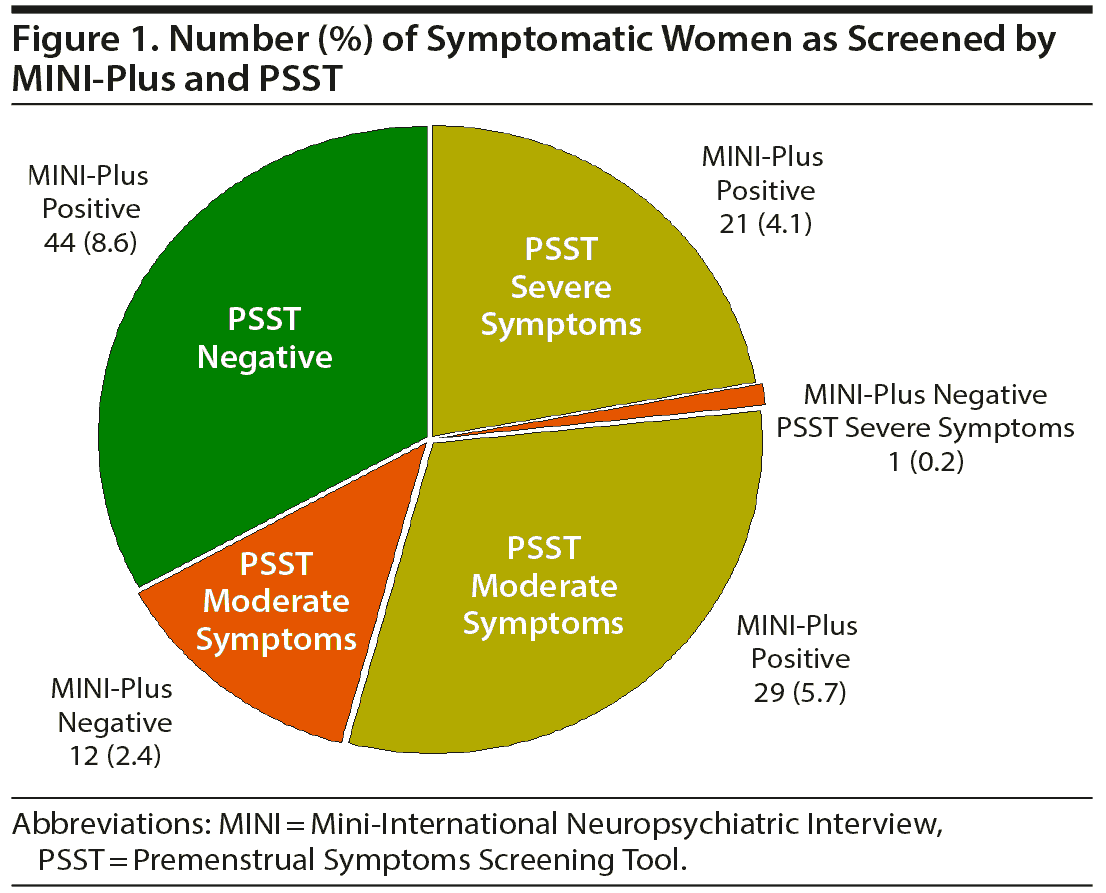
Ninety-four women (18.6%) met MINI-Plus criteria for positive PMDD symptomatology. Of these, 21 met PSST criteria for severe symptoms (4.1%), 29 for moderate symptoms (5.7%), and 44 for mild or less symptoms (8.7%). One woman with severe symptoms (0.2%) and 12 women with moderate symptoms (2.4%) had negative MINI-Plus scores.
One of the 22 women with severe symptoms (4.5%) and 12 of the 41 women with moderate symptoms (29.3%) (as screened by PSST) had negative MINI-Plus scores. Forty-four of the 94 women with positive MINI-Plus scores (46.8%), on the other hand, had negative PSST scores. Using both instruments, 107 of the 508 women (21.1%) had PMDD symptoms (22 had severe symptoms by PSST, 41 had moderate symptoms by PSST, and 44 had positive MINI-Plus scores only).
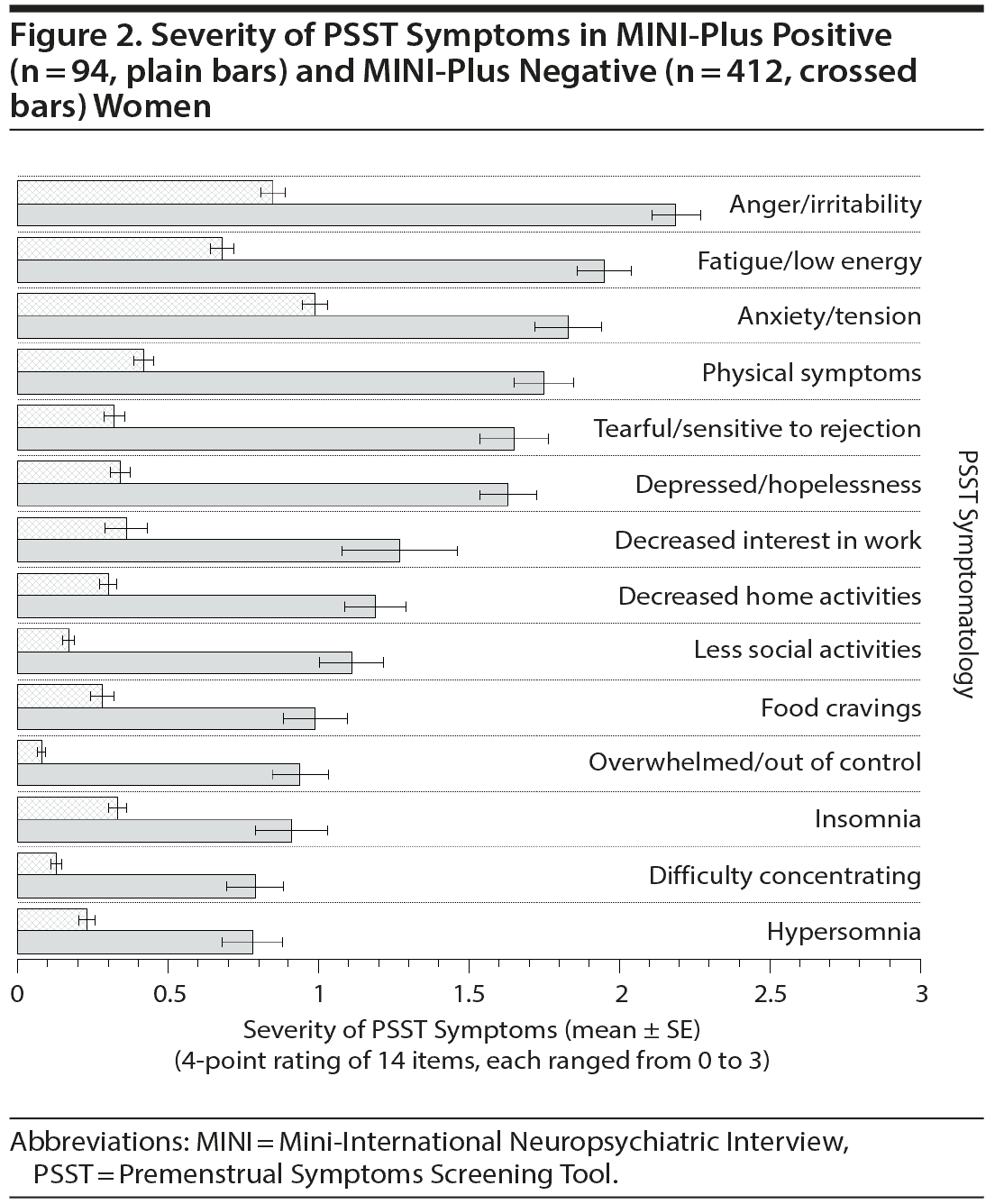
Figures 1 and 2 compare the severity of PSST symptoms in MINI-positive (n = 94) and MINI-negative (n = 412) women. Women who screened positive by MINI-Plus had more severe symptomatology for the entire 14 items of the PSST, suggesting all of these symptoms are relevant to PMDD. Anger/irritability (mean ± SE score: 2.19 ± 0.08, maximum score per item: 3.0), fatigue/low energy (score: 1.95 ± 0.09), anxiety/tension (score: 1.83 ± 0.11), somatic symptoms (score: 1.75 ± 0.10), and tearful/sensitivity to rejection (score: 1.65 ± 0.12) were particularly dominant complaints.
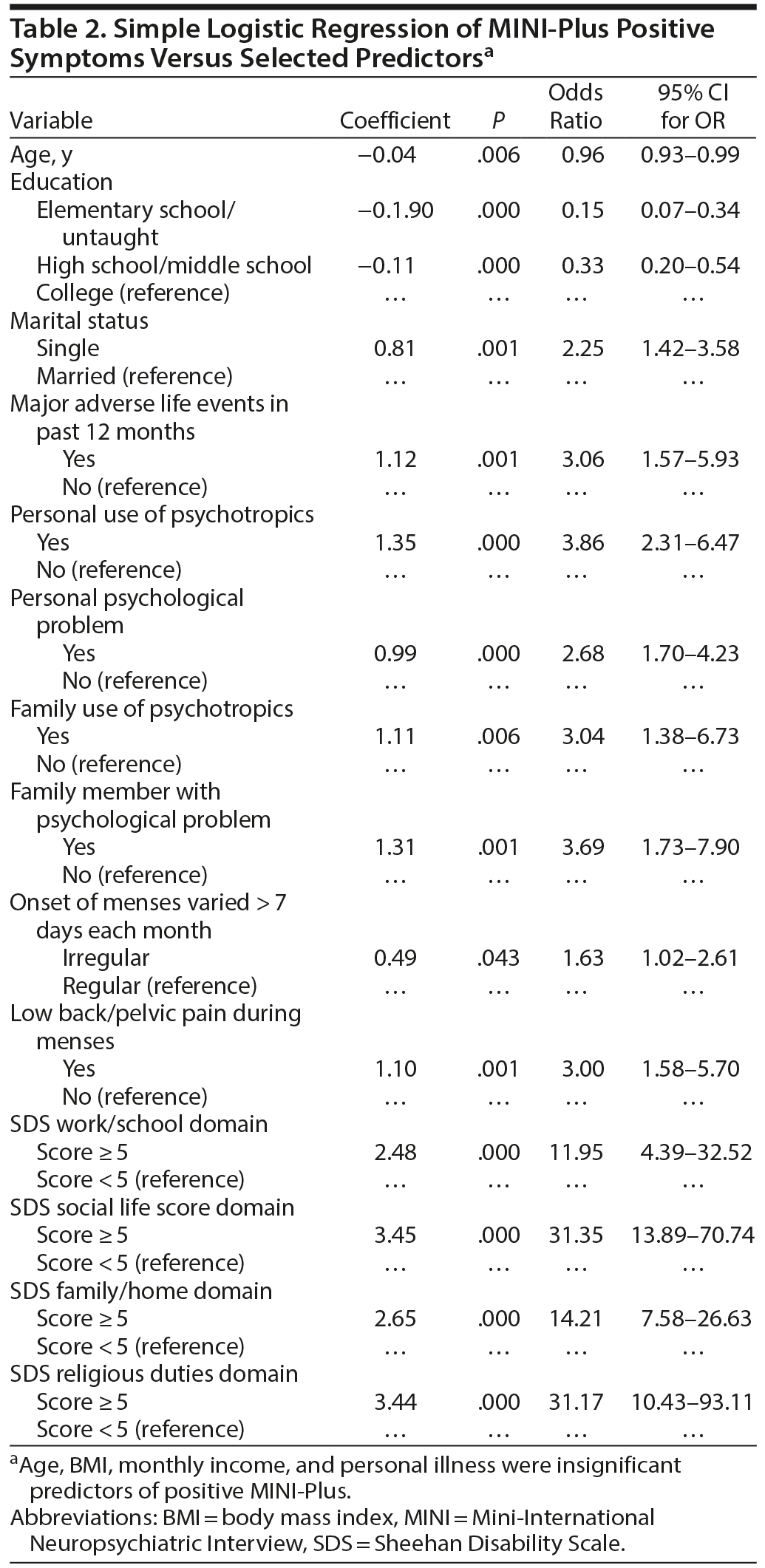
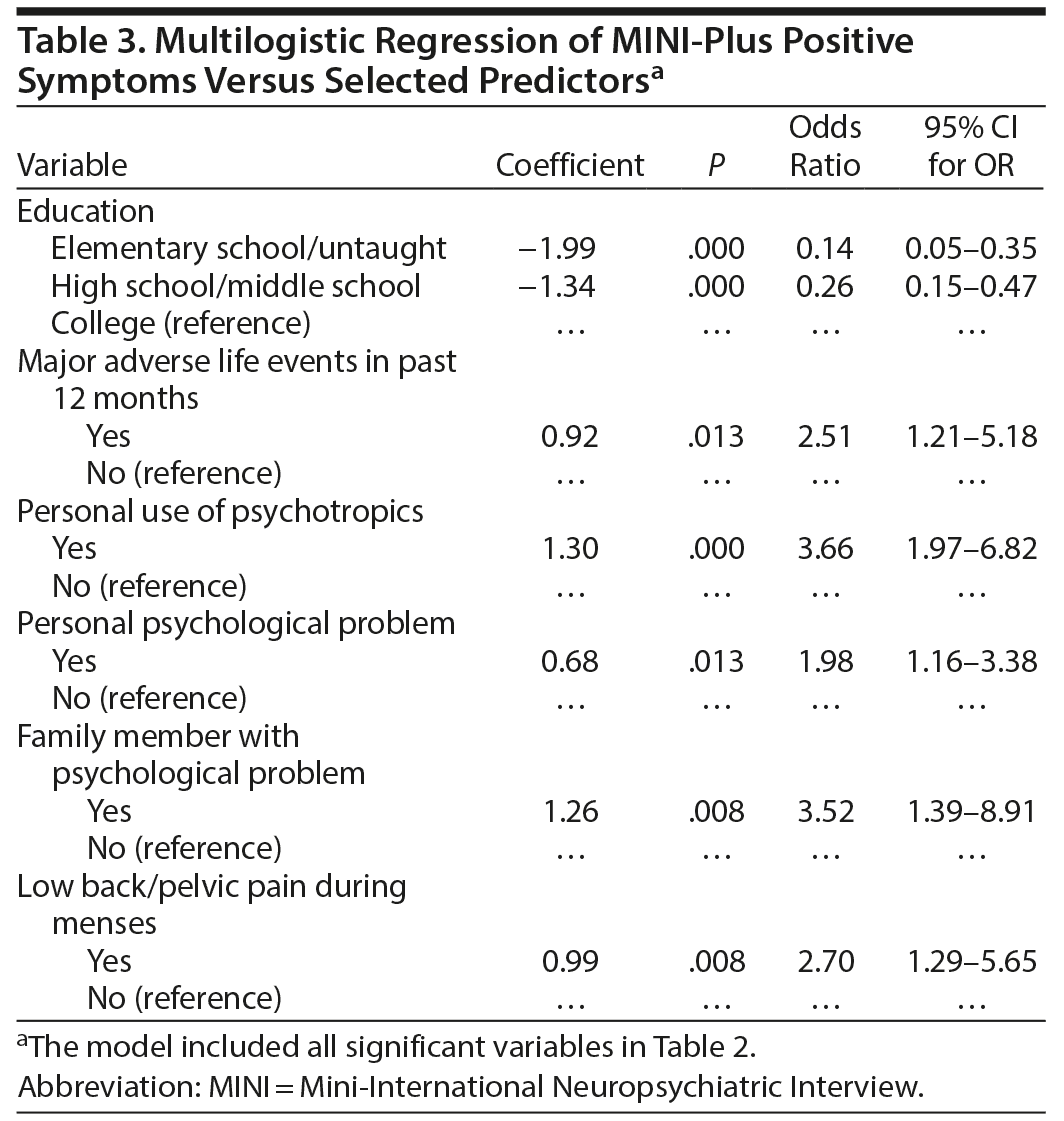
Simple logistic regression model showed the presence of PMDD symptoms (as screened by MINI-Plus) was associated with higher education, single marital status, major life stressors, personal or family use of psychotropic medications, personal or family psychological problem, irregular or painful menses, and SDS functional disability (P ≤ .006, Table 2). Multilogistic regression showed higher education, major life stressor, personal use of psychotropic medications, personal or family psychological problem, and painful menses were independent predictors of PMDD symptoms as screened by the MINI-Plus (P ≤ .013, Table 3). Similarly, personal use of psychotropic medications, family member with psychological problem, and single marital status were independent predictors of severe PMDD symptoms as screened by the PSST (P ≤ .046, Table 4).

DISCUSSION
PMDD symptomatology is founded on the presence of premenstrual affective disorder (severe depressed mood and anxiety) associated with cognitive impairment (difficulty concentrating), increased interpersonal conflicts, and somatic complaints.1–3 Diagnosis is confirmed by prospective daily rating of 2 or more consecutive symptomatic cycles, which represents a limitation in our study.22–24 Our study instead used 2 scales as screening instruments to estimate prevalence and severity of PMDD symptoms in Arab women. The results show that 18.6% of women had PMDD symptoms and 4.3% had severe symptoms. These values are comparable to previously reported prevalence in other populations (about 8%).4
The PSST appeared to apply necessary measures for assessing severity and impact of the symptoms on quality of life. The test was quick and practical.20 Although PMDD is classified in the DSM-IV as a mood disorder (depression not otherwise specified), anxiety was equally representative. As previously reported, psychological symptoms were more prominent than somatic complaints.22–24 Thus, unlike other culturally sensitive psychiatric syndromes, our culture had inconsequential influences on the PMDD symptoms.
Presence of PMDD symptoms was more frequent in educated, single women with life stressors and personal or family history of psychological problem. These findings suggest both acquired and inherited predispositions contribute to the development of symptoms. PMDD symptoms should be recognized by physicians who are involved with women’s health.23 Prompt referral of symptomatic women is essential, as intervention is often necessary.24
Although we used DSM-IV criteria for the disorder, both DSM-IV and DSM-5 diagnoses required the premenstrual pattern to include at least 5 physical, affective, or behavioral symptoms, with a requirement of specific qualities for the key affective symptoms, which are all satisfied in our study.2 The extended form of the MINI-Plus has a full module for PMDD with questions designed to inquire about these criteria. We have used this extended MINI-Plus module in our study. In complement, we also used another scale (SDS) to measure the severity of the impairment in functioning in the domains listed in the DSM-5 criteria (work/school, social/interpersonal, and family responsibilities).
Studies involving the natural history and long-term outcome of PMDD in different societies are needed. This information helps to illustrate the complex interactions between the rapidly evolving predisposing factors (genetic, biological, and psychological variables) and the sociocultural contributors to the disease. National campaigns (written and media) aiming to improve public awareness of PMDD symptoms are also needed.22 Future research should explore genes (eg, using whole exome sequencing) that could predispose women to developing premenstrual dysphoric disorder.
Submitted: February 11, 2017; accepted March 20, 2017.
Published online: July 6, 2017.
Potential conflicts of interest: None.
Funding/support: This study was solely funded by a grant from the UAE University (#01-08-8-11/05). There was no external funding for this research.
Role of the sponsor: The sole supporter of this study was United Arab Emirates University Research Affairs Department, which had no role in the design, analysis, interpretation, or publication of this study.
Acknowledgments: We are grateful to Tariq Mohamed Jabber, MD (Abu Dhabi Ambulatory Health Services); Hanan Al-Raesi, MD; and Sania Al-Hamad, BS (Dr Al-Raesi and Ms Al-Hamad are with the College of Medicine and Health Sciences, United Arab Emirates University) for their valuable contributions. Drs Jabber and Al-Raesi and Ms Al-Hamad report no conflicts of interested related to the subject of this article.
REFERENCES
1. American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders. Fifth Edition. Washington, DC: American Psychiatric Association; 2013.
2. Hantsoo L, Epperson CN. Premenstrual dysphoric disorder: epidemiology and treatment. Curr Psychiatry Rep. 2015;17(11):87. PubMed doi:10.1007/s11920-015-0628-3
3. Robinson LL, Ismail KM. Clinical epidemiology of premenstrual disorder: informing optimized patient outcomes. Int J Womens Health. 2015;7:811–818. PubMed
4. Sepede G, Sarchione F, Matarazzo I, et al. Premenstrual dysphoric disorder without comorbid psychiatric conditions: a systematic review of therapeutic options. Clin Neuropharmacol. 2016;39(5):241–261. PubMed doi:10.1097/WNF.0000000000000173
5. Schüle C, Nothdurfter C, Rupprecht R. The role of allopregnanolone in depression and anxiety. Prog Neurobiol. 2014;113:79–87. PubMed doi:10.1016/j.pneurobio.2013.09.003
6. Bäckström T, Bixo M, Strömberg J. GABAA Receptor-modulating steroids in relation to women’s behavioral health. Curr Psychiatry Rep. 2015;17(11):92. PubMed doi:10.1007/s11920-015-0627-4
7. Schiller CE, Johnson SL, Abate AC, et al. Reproductive steroid regulation of mood and behavior. Compr Physiol. 2016;6(3):1135–1160. PubMed doi:10.1002/cphy.c150014
8. Comasco E, Hahn A, Ganger S, et al. Emotional fronto-cingulate cortex activation and brain derived neurotrophic factor polymorphism in premenstrual dysphoric disorder. Hum Brain Mapp. 2014;35(9):4450–4458. PubMed doi:10.1002/hbm.22486
9. Berman SM, London ED, Morgan M, et al. Elevated gray matter volume of the emotional cerebellum in women with premenstrual dysphoric disorder. J Affect Disord. 2013;146(2):266–271. PubMed doi:10.1016/j.jad.2012.06.038
10. Ossewaarde L, van Wingen GA, Rijpkema M, et al. Menstrual cycle-related changes in amygdala morphology are associated with changes in stress sensitivity. Hum Brain Mapp. 2013;34(5):1187–1193. PubMed doi:10.1002/hbm.21502
11. Wium-Andersen MK, Ørsted DD, Nielsen SF, et al. Elevated C-reactive protein levels, psychological distress, and depression in 73, 131 individuals. JAMA Psychiatry. 2013;70(2):176–184. PubMed doi:10.1001/2013.jamapsychiatry.102
12. Bertone-Johnson ER, Whitcomb BW, Missmer SA, et al. Early life emotional, physical, and sexual abuse and the development of premenstrual syndrome: a longitudinal study. J Womens Health (Larchmt). 2014;23(9):729–739. PubMed doi:10.1089/jwh.2013.4674
13. Gingnell M, Bannbers E, Wikström J, et al. Premenstrual dysphoric disorder and prefrontal reactivity during anticipation of emotional stimuli. Eur Neuropsychopharmacol. 2013;23(11):1474–1483. PubMed doi:10.1016/j.euroneuro.2013.08.002
14. Dimmock PW, Wyatt KM, Jones PW, et al. Efficacy of selective serotonin-reuptake inhibitors in premenstrual syndrome: a systematic review. Lancet. 2000;356(9236):1131–1136. PubMed doi:10.1016/S0140-6736(00)02754-9
15. Marjoribanks J, Brown J, O’Brien PM, et al. Selective serotonin reuptake inhibitors for premenstrual syndrome. Cochrane Database Syst Rev. 2013;(6):CD001396. PubMed
16. Lovick T. SSRIs and the female brain–potential for utilizing steroid-stimulating properties to treat menstrual cycle-linked dysphorias. J Psychopharmacol. 2013;27(12):1180–1185. PubMed doi:10.1177/0269881113490327
17. Maharaj S, Trevino K. A comprehensive review of treatment options for premenstrual syndrome and premenstrual dysphoric disorder. J Psychiatr Pract. 2015;21(5):334–350. PubMed doi:10.1097/PRA.0000000000000099
18. Sheehan D, Janavus J, Baker R, et al. 2001. M.I.N.I. Plus. (Mini International Neuropsychiatric Interview) Version 5.0. http://www.medical‐outcomes.com/minisvite.asp. Accessed June 7, 2017.
19. Sheehan DV, Lecrubier Y, Harnett‐Sheehan K, et al. The validity of the Mini International Neuropsychiatric Interview (MINI) according to the SCID‐P and its reliability. Eur Psychiatry. 1997;12(5):232–241. doi:10.1016/S0924-9338(97)83297-X
20. Steiner M, Macdougall M, Brown E. The Premenstrual Symptoms Screening Tool (PSST) for clinicians. Arch Women Ment Health. 2003;6(3):203–209. PubMed doi:10.1007/s00737-003-0018-4
21. Sheehan KH, Sheehan DV. Assessing treatment effects in clinical trials with the discan metric of the Sheehan Disability Scale. Int Clin Psychopharmacol. 2008;23(2):70–83. PubMed doi:10.1097/YIC.0b013e3282f2b4d6
22. Hardy C, Sillence E. What are women being exposed to? a review of the quality, content and ownership of websites on premenstrual dysphoric disorder. Womens Health Issues. 2016;26(2):183–189. PubMed doi:10.1016/j.whi.2015.11.007
23. Bosman RC, Jung SE, Miloserdov K, et al. Daily symptom ratings for studying premenstrual dysphoric disorder: a review. J Affect Disord. 2016;189:43–53. PubMed doi:10.1016/j.jad.2015.08.063
24. Hall E, Steiner M. Psychiatric symptoms and disorders associated with reproductive cyclicity in women: advances in screening tools. Womens Health (Lond). 2015;11(3):397–415. PubMed doi:10.2217/whe.15.1
This PDF is free for all visitors!

