Objective: To examine the short- and long-term effect of electroconvulsive therapy on verbal, visual, and autobiographical memory functions in patients treated for a severe depressive episode. Patients were compared with healthy controls undergoing neurocognitive assessments at the same time points to account for normal forgetfulness and potential learning effects.
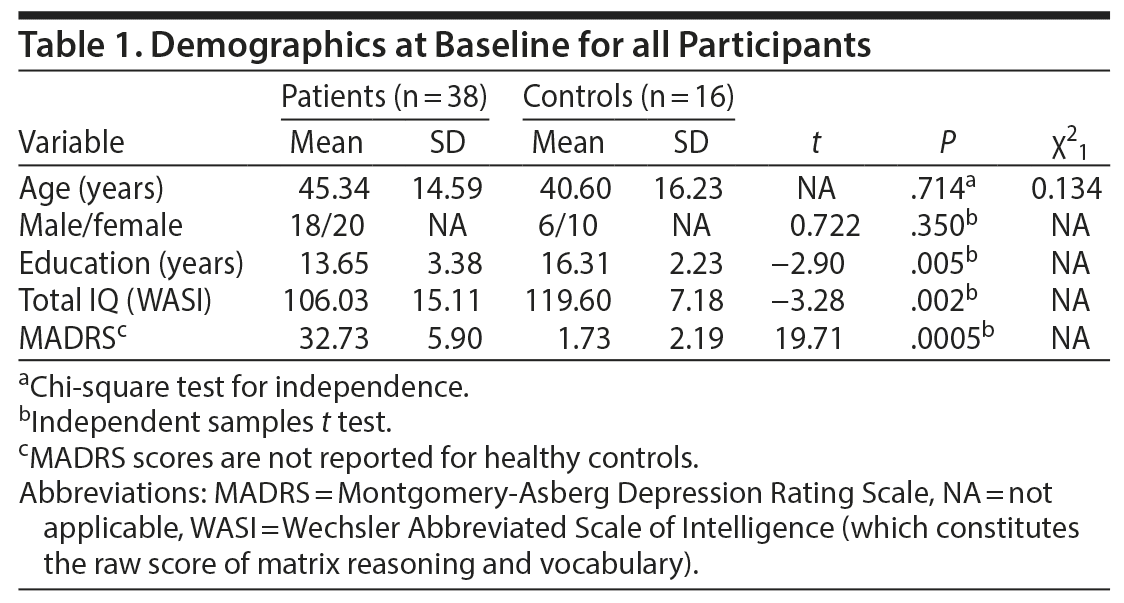
Methods: A pre-post intervention design included patients (n = 38) and controls (n = 16) referred to Haukeland University Hospital for electroconvulsive therapy (ECT) from September 2013 to September 2018. Patients diagnosed with a major depressive episode (according to ICD-10 criteria) underwent right unilateral ECT with brief-pulse, square-wave, constant current. Neurocognitive assessments were administered pretreatment and, on average, 19 days and 6 months posttreatment. Performance on the California Verbal Learning Test Second Edition, Rey Osterrich Complex Figure, and Autobiographical Memory Interview-Short Form were the main outcome measures, examining verbal, visual, and autobiographical memory, respectively.
Results: Patients performed significantly worse compared to controls on all measures of verbal and visual memory at every assessment (P ≤ .001). Within-group analyses showed no impaired visual or verbal memory function due to ECT. However, autobiographical consistency was significantly decreased for patients (70.30%) compared to controls (82.03%) 6 months posttreatment (P = .0005).
Conclusions: Patients’ ability to acquire new general knowledge is considered as unaffected by ECT. Deficits in autobiographic memory were found 6 months posttreatment, indicating both an iatrogenic effect of treatment and an effect of depression on retrograde memory functions. For patients, the risk of this iatrogenic effect of treatment must be evaluated against the symptomatic and potential functional recovery due to ECT.
Trial registration: Clinicaltrials.gov identifier: NCT04348825.
A Longitudinal Comparison Between Depressed Patients Receiving Electroconvulsive Therapy and Healthy Controls on Specific Memory Functions
ABSTRACT
Objective: To examine the short- and long-term effect of electroconvulsive therapy on verbal, visual, and autobiographical memory functions in patients treated for a severe depressive episode. Patients were compared with healthy controls undergoing neurocognitive assessments at the same time points to account for normal forgetfulness and potential learning effects.
Methods: A pre-post intervention design included patients (n = 38) and controls (n = 16) referred to Haukeland University Hospital for electroconvulsive therapy (ECT) from September 2013 to September 2018. Patients diagnosed with a major depressive episode (according to ICD-10 criteria) underwent right unilateral ECT with brief-pulse, square-wave, constant current. Neurocognitive assessments were administered pretreatment and, on average, 19 days and 6 months posttreatment. Performance on the California Verbal Learning Test Second Edition, Rey Osterrich Complex Figure, and Autobiographical Memory Interview-Short Form were the main outcome measures, examining verbal, visual, and autobiographical memory, respectively.
Results: Patients performed significantly worse compared to controls on all measures of verbal and visual memory at every assessment (P ≤ .001). Within-group analyses showed no impaired visual or verbal memory function due to ECT. However, autobiographical consistency was significantly decreased for patients (70.30%) compared to controls (82.03%) 6 months posttreatment (P = .0005).
Conclusions: Patients’ ability to acquire new general knowledge is considered as unaffected by ECT. Deficits in autobiographic memory were found 6 months posttreatment, indicating both an iatrogenic effect of treatment and an effect of depression on retrograde memory functions. For patients, the risk of this iatrogenic effect of treatment must be evaluated against the symptomatic and potential functional recovery due to ECT.
Trial registration: Clinicaltrials.gov identifier: NCT04348825.
Prim Care Companion CNS Disord 2020;22(3):19m02547
To cite: Blomberg MO, Semkovska M, Kessler U, et al. A longitudinal comparison between depressed patients receiving electroconvulsive therapy and healthy controls on specific memory functions. Prim Care Companion CNS Disord. 2020;22(3):19m02547.
To share: https://doi.org/10.4088/PCC.19m02547
© Copyright 2020 Physicians Postgraduate Press, Inc.
aDepartment of Biological and Medical Psychology, University of Bergen, Bergen, Norway
bDepartment of Psychology, University of Limerick, Limerick, Ireland
cNorment, Division of Psychiatry, Haukeland University Hospital, University of Bergen, Bergen, Norway
dDepartment of Psychiatry, University of Bergen, Bergen, Norway
eDepartment of Clinical Medicine, University of Bergen, Bergen, Norway
fMohn Medical Imaging and Visualization Centre, Department of Radiology, Haukeland University Hospital, Bergen, Norway
*Corresponding author: Åsa Hammar, PhD, Department of Biological and Medical Psychology, University of Bergen, Jonas Lies vei 91, 5009 Bergen, Norway ([email protected]).
Electroconvulsive therapy (ECT) is a highly effective treatment for major depressive disorder (MDD).1 However, the extent of treatment is often limited by concerns regarding adverse effects on memory.2
Cognitive impairment is often seen in patients with MDD and can be one of the most disabling symptoms. The main neurocognitive domains affected in MDD have been shown to be memory, executive functions, processing speed, and attention.3-6 Cognitive impairments observed during an MDD episode may persist in phases of symptom reduction.4
Regarding cognitive functions post-ECT, an extensive meta-analysis7 found that most cognitive deficits associated with ECT are limited to the first 3 days posttreatment. Overall cognitive performance recovered to pretreatment levels within 2 weeks. However, baseline measures, retrograde memory tasks, autobiographical memory, and subjective reports were not included.7 A systematic review2 concluded that autobiographical memory impairment occurs post-ECT. Objective measures were relatively short term (< 6 months), whereas the subjective experiences of amnesia were more persistent (> 6 months). Perceived autobiographical memory impairments were considered a major concern with ECT. Cognitive dysfunction has been identified in the acute disorientation phase and in assessments immediately following ECT.8,9 However, this phase is usually brief.10,11
The severity, persistence, and extent of memory impairments due to ECT remain controversial.12 Some of the heterogeneity observed in earlier research might be attributable to the use of the nonspecific term memory,13 as opposed to more specific categories of memory functions. Results of verbal and nonverbal measures are often amalgamated, with no clear distinction of the different stages in the memory process,7 which precludes the study of specific iatrogenic effects on the different types of memory.
Contradicting results in previous research of memory function after ECT may be explained by many factors. First, various neuropsychological tests have been applied with the intention to measure the same function. For instance, one study14 used the California Verbal Learning Test-Second Edition (CVLT-II)15 to assess verbal memory, while another16used the Rey Auditory Verbal Learning Test.17 Tests may differ in sensitivity, design, and function being assessed. Second, results of the same neuropsychological tests have been interpreted differently. Wechsler digit span forward18 has been cited as a measure of verbal memory,19 working memory,20 and executive function.3 Third, previous research is characterized by divergent populations of patients. The patients included may have received different kinds of ECT and subsequently were measured on treatment outcome at different times elapsed since intervention. Fourth, some studies do not include a control group or baseline measures, affecting how results are interpreted.
There is currently a need to rectify the methodological shortcomings of earlier research on memory functions post-ECT. The absence of healthy controls, baseline measures, and long-term follow-up assessments is seen in some studies. Memory is one of the core domains within cognitive functioning. In the present study, memory is not regarded as one unitary construct but is seen as different specific aspects within the memory domain. Due to earlier reports2 of autobiographical memory impairment post-ECT, assessment of this function was included.
Objectives and Research Questions
The main aim of the present study is to examine how ECT affects objective measures of verbal, visual, and autobiographical memory functions in patients treated for a major depressive episode. Patients are compared with healthy controls undergoing neurocognitive assessments following the protocol for patients to account for normal forgetfulness and potential learning effects.
The following 5 questions were addressed:
- Do patients perform worse on measures of verbal and visual memory functions at baseline compared to controls?
- Will measurements of verbal and visual memory functions in patients significantly differ from controls 7-14 days and 6 months posttreatment?
- Do patients perform worse on verbal and visual memory tasks 7-14 days post-ECT compared to baseline?
- At 6 months posttreatment, will patients improve their verbal and visual memory performance or perform at the same level as 7-14 days posttreatment?
- Will measurements of autobiographical memory be affected by ECT when comparing patients to controls?
Hypotheses
On the basis of available research, we expect to find differences between the groups in all variables at baseline and 7-14 days and 6 months posttreatment. No significant decline is predicted for patients in verbal and visual memory functions 7-14 days posttreatment, considering that the acute disorientation phase should be stabilized. At 6 months posttreatment, we foresee patients performing approximately the same or improving on tasks requiring verbal and visual memory functions compared to their performance 7-14 days posttreatment. Patients are presumed to show greater decline in autobiographical memory compared to controls.
METHODS
Participants
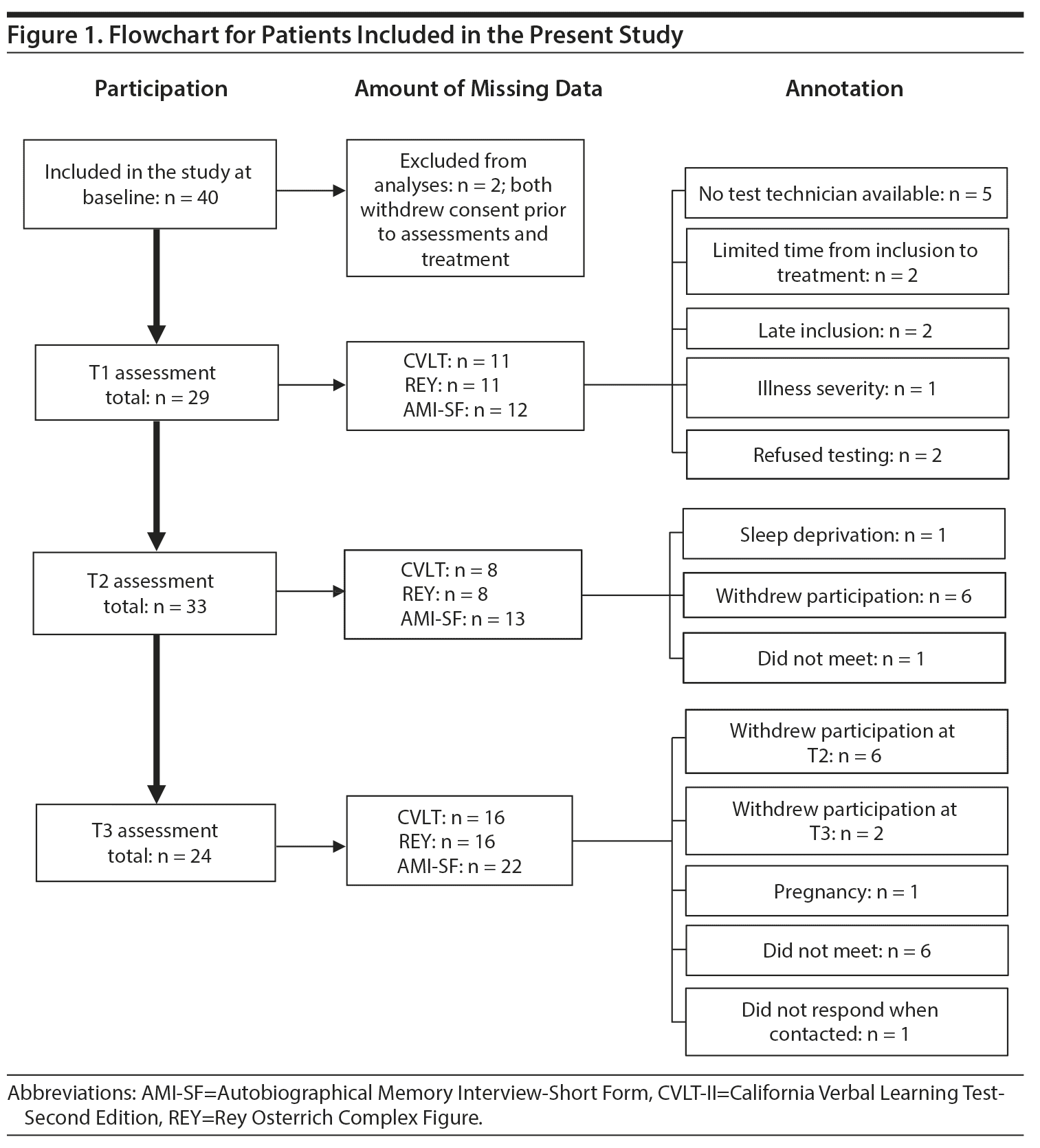
The patient group consisted of 40 patients referred to Haukeland University Hospital, Bergen, Norway, for ECT. Two patients withdrew consent prior to assessment and are therefore excluded from the analyses.

- Patients’ ability to acquire new general knowledge does not seem to be affected by electroconvulsive therapy (ECT); however, the severe depressive episode itself seems to affect these memory functions.
- Results indicate a potential side effect of ECT with reduced autobiographical memory; risk of reduced autobiographical memory must be evaluated against the expected symptomatic recovery from depression.
- A possible barrier to successful treatment with use of ECT is clinician and patient misconception of potential side effects, which may lead to reduced extent of treatment.
Eligibility criteria included patients aged > 18 years referred to ECT for a moderate or severe depressive episode. There was no upper age limit for participation. Patients fulfilled the criteria of 1 of the following ICD-10 diagnoses21: F31.3 (n = 4), F31.4 (n = 3), F32.1 (n = 1), F32.3 (n = 2), F33.1 (n = 5), F33.2 (n = 21), or F33.3 (n = 2). The diagnoses were confirmed according to the protocol of the Mini-International Neuropsychiatric Interview.22 ECT is shown to be efficient for both bipolar and unipolar depression.23,24 Therefore, both bipolar and unipolar groups were included. Patients were diagnosed based on a clinical interview. Additional information was obtained from hospital records, and the inclusion criterion was a score > 25 on the Montgomery-Asberg Depression Rating Scale (MADRS).25
A trained clinician considered all patients eligible for treatment on the basis of their capability to give informed written consent. Exclusion criteria were pregnancy and receiving ECT in the last year.
The control group consisted of 16 healthy volunteers (n = 16) of equivalent mean age and sex distribution as the patients (Table 1). The control group underwent repeated neuropsychological testing at the same timepoints as the patients.
ECT Treatment
ECT was administrated as recommended by the Norwegian Health Department.26 Additional information about ECT in the current study was previously published.27
Stimulation and electrode placement. A Thymatron System IV (Somatics LLC, Lake Bluff, Illinois) provided brief-pulse, square-wave, constant current. Patients underwent treatment with right unilateral electrode placement. There were 3 sessions per week until remission or the maximum of 18 sessions was reached or terminated when no further improvement was anticipated.
Stimulus. The duration of the stimulus pulse was ordinarily set to 0.5 ms. The initial stimulus energy was calculated based on age. Energy was calculated as the patient’s age in years ×— 5 @ stimulus charge in mC.28
Anesthesia. Anesthesia was given as thiopental or propofol. Patients were hyperoxygenated with oxygen-enriched air minutes before and during anesthesia to optimize the induction of seizures.
Seizure adequacy. The adequacy of each seizure was evaluated by an ECT clinician based on duration, reorientation time, clinical effect, and d-waves. For seizures evaluated as not effective, the stimulus parameter was adjusted during the next session.
Design, Procedure, Neurocognitive Assessment, and Clinical and Cognitive Measures
A pre-post intervention design was conducted from September 2013 to September 2018. Neuropsychological assessments were conducted within 7 days pretreatment (T1) and, on average, 19 days (range, 1-53 days; median: 16 days) (T2) and 6 months (T3) posttreatment. Assessments were carried out by a trained test technician.
Symptom severity was monitored by the MADRS.25 Verbal memory was assessed with the CVLT-II.15 Three variables were included as follows: the first repetition of list A measuring verbal attention span, immediate recall of list A measuring verbal short-term memory, and delayed recall of list A measuring verbal long-term memory.
Visual memory was examined with the Rey Osterrich Complex Figure (REY).29 To assess visual long-term memory, participants were asked to replicate the complex figure as seen in the initial learning trial 30 minutes earlier.
Autobiographical memory was measured with the Autobiographical Memory Interview-Short Form (AMI-SF).30 The AMI-SF requests information about 6 autobiographical events examined by 30 questions. Unfortunately, a mistake was made during administration, and the sixth part of the interview was not administered to 11 of the participants. Therefore, this part of the interview was excluded for all participants in calculation of raw scores. Follow-up scores from T2 and T3 are participants’ consistency in answers with baseline answers. Consistency scores were calculated at T2 and T3 based on the percentage of the participants’ answers that were consistent with their answers at T1. The sixth part of the interview was not a source of bias in this variable. Hence, 2 variables were included: raw scores to assess within-group variations and consistency scores to examine between-group differences.
Ethical Considerations
Participants provided informed written consent before participation. This study was approved by the Regional Committee for Medical and Health Research Ethics, REC, South East, ID: 2013/1032 ECT and neurology (Clinicaltrials.gov identifier: NCT04348825.).
Statistics
All statistical analyses were performed in IBM SPSS Statistics version 25 (IBM Corp, Armonk, New York). A mixed between-within subjects analysis of variance (ANOVA) was conducted for each main variable as recommended with repeated testing and multilevel data.31 Post hoc tests were conducted as 1-way repeated measures ANOVA for within-group variables and independent sample t tests for between-group differences. Given the number of statistical testing, all analyses were Bonferroni corrected with a significance level of P < .002. Missing data were substituted with the group mean score (see Figure 1 for missing data). Effect sizes are reported as partial eta square (ηp2) and interpreted as recommended by Cohen.32
RESULTS
Clinical Measures
A 1-way repeated measures ANOVA was conducted to examine the effect of treatment on mood. The significant decrease of mean MADRS scores from T1 to T2 was 17.11 points (P = .0005). There was no significant difference between T2 and T3 (P = .359). A large effect size was seen (ηp2 = 0.82).
Cognitive Measures
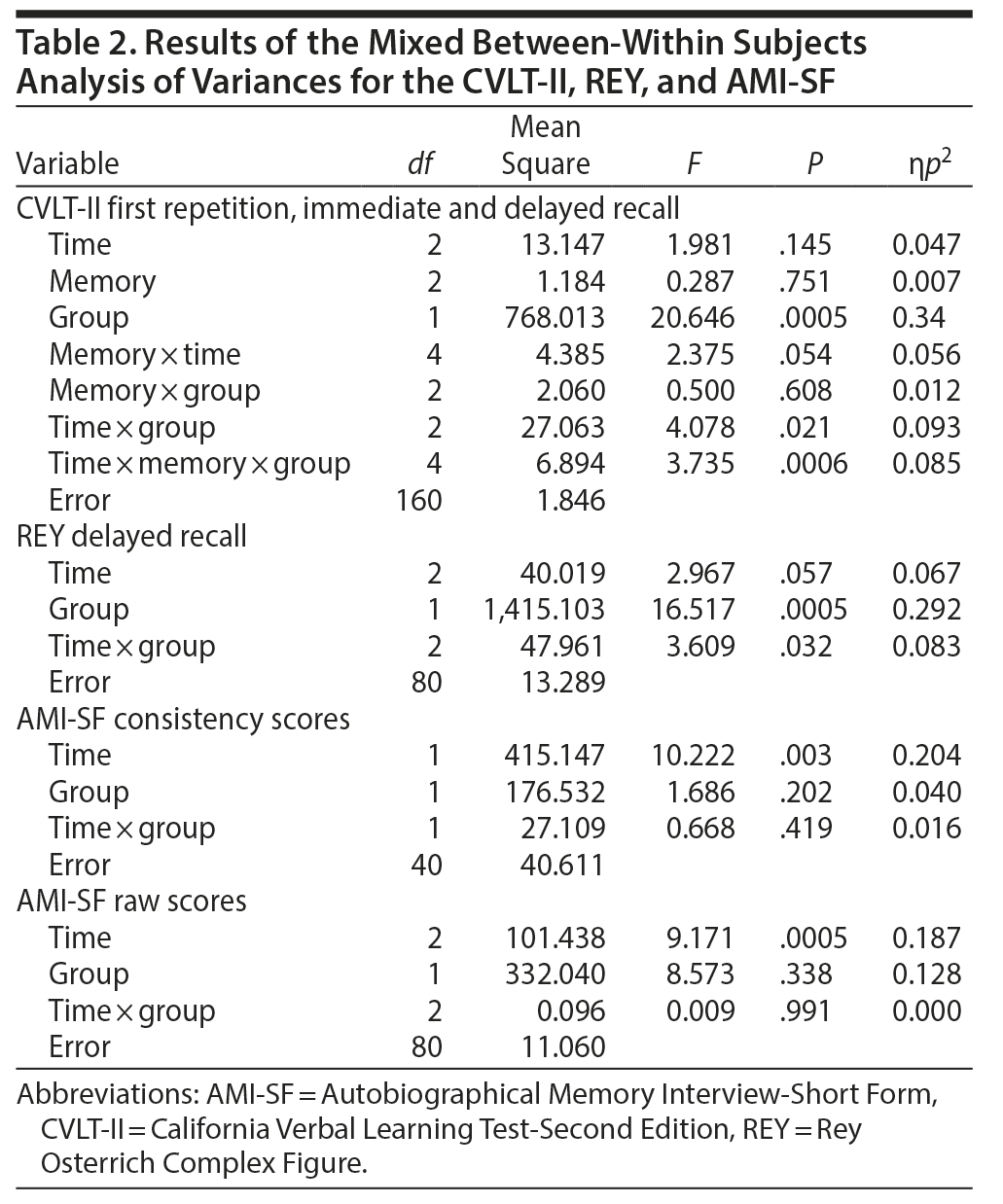
Mixed between-within ANOVAs. In sum, a significant interaction effect was found for memory, time, and group on the CVLT-II, in addition to a significant main effect for group. Likewise, there was a significant main effect for group on the REY. There was a significant main effect for time in AMI-SF raw scores. Similar results showed a nearly significant main effect for time in the consistency scores. Moderate to large effect sizes were found for all significant results. Post hoc testing was conducted on the basis of these results. Table 2 provides the results.
Years of education and WASI scores were applied as covariates. Years of education did not affect the group differences observed in CVLT-II scores (P = .596), REY scores (P = .656), AMI-SF raw scores (P = .338), or consistency scores (P = .878). There were no significant results between groups when controlling for WASI scores in regard to CVLT-II scores (P = .163), REY scores (P = .458), AMI-SF raw scores (P = .005), or consistency scores (P = .006).
Sensitivity analyses were conducted to control for the variety of days at T2. These analyses were restricted to the patients assessed within 21 days posttreatment (n = 20); all other patients were excluded. Results for the CVLT-II and REY were similar to those of the original analyses. A significant interaction effect was seen for time, memory, and group (P = .046), in addition to a significant main effect for group (P = .001), for the CVLT-II. There was a significant main effect for group (P = .001) for the REY. Relative to the AMI-SF variable, sensitivity analyses did not replicate the main results: neither the effect for time (P = .603) nor the effect of group (P = .020) remained significant; the latter, however, approached significance.
Post hoc t tests for between-group differences. CVLT-II. Significant differences were found between the groups at T1 (t52 = −3.46, P = .001), T2 (t52 = −5.69, P = .0005), and T3 (t52 = −7.92, P = .0005) for the first repetition of list A. Results of immediate recall of list A also showed statistical differences between the groups at T1 (t52 = −4.25, P = .0005), T2 (t52 = −3.94, P = .0005), and T3 (t52 = −5.41, P = .0005). Similar results were found for delayed recall of list A, with significant differences between the groups at T1 (t52 = −4.30, P = .0005), T2 (t52 = −3.51, P = .001), and T3 (t52 = −5.25, P = .0005).
REY. Results showed significant differences between the groups at T1 (t52 = −3.93, P = .0005), T2 (t52 = −5.98, P = .0005), and T3 (t52 = −5.65, P = .0005).
AMI-SF consistency scores. There was no significant between-group difference at T2 (t52 = −1.89, P = .065). However, results showed a significant difference at T3 (t52 = −3.96, P = .0005). Mean consistency scores were significantly decreased for patients (70.30%) compared to controls (82.03%).
In sum, significant between-group differences were found, with patients performing worse compared to controls on nearly every variable at every assessment.
Post hoc 1-way ANOVAs for within-group differences. Patients. Patients displayed stability in results of all CVLT-II and REY variables (P ≥ .07). However, there was a significant decrease in AMI-SF raw scores from T1 to T2 (P = .0005) and T2 to T3 (P = .0005).
Controls. Indications of learning effects were found for the first repetition of list A of the CVLT-II. A significant within-group difference was seen from T1 to T2 (P = .002). Indications of learning effects were identified for the REY, although results did not reach statistical significance from T1 to T2 (P = .004). Like patients, controls displayed stability in performance on CVLT-II immediate and delayed recall (P ≥ .01). Regarding AMI-SF raw scores, a significant decrease in scores from T1 to T2 (P = .0005), but not from T2 to T3, was found (P = .005).
In sum, patients showed stability in performance on the CVLT-II and REY over time. Results indicated learning effects for controls in 2 variables. Forgetfulness in AMI-SF was found for both patients and controls.
DISCUSSION
Memory Functions
Verbal and visual memory functions. As expected, patients performed significantly worse on all measures of verbal and visual memory functions at baseline and 7-14 days and 6 months post-ECT compared to healthy controls. Hence, memory impairments outlasting the specific depressive episode were revealed. These results support earlier conclusions that patients may have cognitive dysfunction despite recovering from the depressive episode.4,33
Nonetheless, measures of visual and verbal memory functions were stable for patients at every timepoint. These results are in accordance with the majority of studies7,9,34 showing stability of cognitive performance post-ECT. Stabilization or an improvement in these functions was expected. Previous research shows improvement in verbal8,14 and visual7,35,36 memory functions posttreatment.
Autobiographical memory. Forgetfulness was identified at every assessment for patients and controls. As anticipated, patients displayed a larger decrease in consistency than controls 6 months posttreatment, indicating both an iatrogenic effect of treatment and an effect of depression. This finding coincides with research comparing an ECT group with a group receiving pharmacologic treatment for depression. In comparison, ECT patients had significantly reduced autobiographical memory consistency 6 weeks posttreatment.37 However, no difference was found in autobiographical memory consistency when comparing patients receiving ECT with patients receiving pharmacologic treatment at 6 months posttreatment.38
AMI-SF, as used in the above-mentioned studies, is strongly criticized.39 Patients could potentially provide answers at T2 and T3 that are correct but regarded as incorrect if not in line with the baseline answer acquired during the period of severe depressive symptoms. As noted by others,2 patients could consistently report incorrect information, resulting in a difficulty obtaining an exact measure of their experiences and memories.A larger number of items target older memories, although ECT-induced amnesia is known to be densest for recent memories.13 Overlearned and often repeated facts are targeted but regarded as unlikely to be affected by ECT.13 The ethical attributes of AMI-SF should be addressed. The events examined are personal and require details of events that severely depressed patients may not engage in.
Limitations
Some limitations should be considered when interpreting these results. The dataset included, unfortunately, a substantial amount of missing data. This issue was corrected in the statistical analysis by substituting the missing variables with the group mean.
We cannot dismiss the possibility that the significant between-group difference in WASI scores may have influenced the results on AMI-SF scores. Results were nearly significant for these variables when applying WASI as covariate.
There were indications of learning effects interfering with the results of the first repetition for list A in CVLT-II and REY. Patients did not display as large or significant learning effects as controls.
Since no subjective reports of memory functions were included, it cannot be guaranteed that the neuropsychological tests target all possible memory deficits patients may experience post-ECT.
Sensitivity analyses were conducted to determine if the variety of time intervals between T1 and T2 might have affected the results. Obtained results remain robust for the CVLT-II and REY variables. However, effect of time was no longer significant for the AMI-SF variable, which may be explained by the decrease of the overall sample size.
Clinical Implications
Informing patients about potential adverse effects of ECT is as important as conveying the clinical effect.13 The present results reveal an antidepressant effect of ECT maintained 6 months posttreatment. On the basis of our results, patients can be informed that everyday memory functions are presumed to be unaffected by treatment. The ability to acquire new information and knowledge is not reduced.
Conversely, previously acquired personal memories may be affected negatively by treatment. The extent of these retrograde memory impairments remains uncertain. The risk of this iatrogenic effect must be considered in relation to the potential positive effects of treatment. Reduced autobiographical memory might be the price severely depressed patients have to pay for symptom reduction and, conceivably, a functional recovery and increased quality of life. An earlier literature review40 estimated that approximately 60% of patients would undergo ECT again. However, research41 implies that most patients (86%) would undergo ECT again despite 60% of the same patients reporting cognitive impairment posttreatment. Furthermore, it is possible that, as previous evidence has suggested,42 these autobiographical memory issues actually affect only some (and not all) individuals. This interpretation is supported by the decreased significance in the results observed on the AMI-SF variable following the sensitivity analyses: if only a few patients present autobiographical amnesia, then their reduced performance would dissolve in the group mean.
Implications for Future Research
Several uncertainties regarding the effect of ECT on memory functions remain, demanding further investigation. The potential iatrogenic effect on autobiographical memory needs further examination. Development of a sensitive objective measure for autobiographical memory is needed. Collecting normative data and examining reliability and validity should be prioritized. Qualitative research regarding memory dysfunction could provide interesting results considering the subjective nature of autobiographical memory. In long-term follow-up studies, autobiographical memory functions should be examined over a period extending 6 months posttreatment to determine if memories recover over time.
Mapping of other cognitive functions influencing memory is crucial when results indicate memory dysfunction. The present results revealed a lasting deficit in verbal attention span for patients. Hence, attentional resources in patients with MDD should be examined further.
CONCLUSION
No short- or long-term effects of ECT were found on verbal and visual memory functions. Patients’ ability to acquire new knowledge was unaffected by ECT. However, patients exhibited long-lasting verbal and visual memory impairments compared to healthy controls despite a significant reduction of depressive symptoms. Patients displayed decreased autobiographical consistency 6 months posttreatment, indicating a potential iatrogenic effect of treatment. The extent and clinical importance of these retrograde memory impairments remain uncertain and should be examined further based on the present study. The risk of potential iatrogenic effects of treatment must be evaluated against the symptomatic and potential functional recovery posttreatment.
Submitted: September 17, 2019; accepted December 20, 2019.
Published online: May 14, 2020.
Potential conflicts of interest: None.
Funding/support: Support was received from Western Norway Regional Health Authority (no. 911986 to KJO and no. 912238 to Dr Oltedal) and the University of Bergen (to Dr Oltedal).
Role of the sponsor: The sponsor had no role in the design and conduct of the study; collection, management, analysis, and interpretation of data; or preparation, review, or approval of the manuscript.
Acknowledgments: The authors thank Leila Frid, BA, Ingunn Samn׸y, MA, and Heidi Pedrero Waage, BA (all from Haukeland University Hospital, Bergen, Norway), for assistance, as well as all participants in the study. Mss Frid, Samn׸y, and Waage report no conflicts of interest related to the subject of this article.
REFERENCES
1.UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet. 2003;361(9360):799-808. PubMed CrossRef
2.Fraser LM, O’ Carroll RE, Ebmeier KP. The effect of electroconvulsive therapy on autobiographical memory: a systematic review. J ECT. 2008;24(1):10-17. PubMed CrossRef
3.McDermott LM, Ebmeier KP. A meta-analysis of depression severity and cognitive function. J Affect Disord. 2009;119(1-3):1-8. PubMed CrossRef
4.Hammar A, Ardal G. Cognitive functioning in major depression: a summary. Front Hum Neurosci. 2009;3:26. PubMed
5.Rock PL, Roiser JP, Riedel WJ, et al. Cognitive impairment in depression: a systematic review and meta-analysis. Psychol Med. 2014;44(10):2029-2040. PubMed CrossRef
6.Marazziti D, Consoli G, Picchetti M, et al. Cognitive impairment in major depression. Eur J Pharmacol. 2010;626(1):83-86. PubMed CrossRef
7.Semkovska M, McLoughlin DM. Objective cognitive performance associated with electroconvulsive therapy for depression: a systematic review and meta-analysis. Biol Psychiatry. 2010;68(6):568-577. PubMed CrossRef
8.Bodnar A, Krzywotulski M, Lewandowska A, et al. Electroconvulsive therapy and cognitive functions in treatment-resistant depression. World J Biol Psychiatry. 2016;17(2):159-164. PubMed CrossRef
9.Vasavada MM, Leaver AM, Njau S, et al. Short- and long-term cognitive outcomes in patients with major depression treated with electroconvulsive therapy. J ECT. 2017;33(4):278-285. PubMed CrossRef
10.Lisanby SH, Luber B, Schlaepfer TE, et al. Safety and feasibility of magnetic seizure therapy (MST) in major depression: randomized within-subject comparison with electroconvulsive therapy. Neuropsychopharmacology. 2003;28(10):1852-1865. PubMed CrossRef
11.Sobin C, Sackeim HA, Prudic J, et al. Predictors of retrograde amnesia following ECT. Am J Psychiatry. 1995;152(7):995-1001. PubMed CrossRef
12.Mohn C, Rund BR. Significantly improved neurocognitive function in major depressive disorders 6 weeks after ECT. J Affect Disord. 2016;202:10-15. PubMed CrossRef
13.Robertson H, Pryor R. Memory and cognitive effects of ECT: informing and assessing patients. Adv Psychiatr Treat. 2006;12(3):228-237. CrossRef
14.Biedermann SV, Bumb JM, Demirakca T, et al. Improvement in verbal memory performance in depressed in-patients after treatment with electroconvulsive therapy. Acta Psychiatr Scand. 2016;134(6):461-468. PubMed CrossRef
15.Woods SP, Delis DC, Scott JC, et al. The California Verbal Learning Test, Second Edition: test-retest reliability, practice effects, and reliable change indices for the standard and alternate forms. Arch Clin Neuropsychol. 2006;21(5):413-420. PubMed CrossRef
16.Loo CK, Sainsbury K, Sheehan P, et al. A comparison of RUL ultrabrief pulse (0.3 ms) ECT and standard RUL ECT. Int J Neuropsychopharmacol. 2008;11(7):883-890. PubMed CrossRef
17.Frasca TAA, Iodice A, McCall WV. The relationship between changes in learning and memory after right unilateral electroconvulsive therapy. J ECT. 2003;19(3):148-150. PubMed
18.Kaufman AS. Test Review: Wechsler, D. Manual for the Wechsler Adult Intelligence Scale, Revised. New York: Psychological Corporation, 1981. J Psychoed Assess. 1983;1(3):309-313. CrossRef
19.Cronin D, Bodley P, Potts L, et al. Unilateral and bilateral ECT: a study of memory disturbance and relief from depression. J Neurol Neurosurg Psychiatry. 1970;33(5):705-713. PubMed CrossRef
20.Ziegelmayer C, Hajak G, Bauer A, et al. Cognitive performance under electroconvulsive therapy (ECT) in ECT-naive treatment-resistant patients with major depressive disorder. J ECT. 2017;33(2):104-110. PubMed CrossRef
21.World Health Organization. ICD-10: International Classification of Diseases. Direktoratet for e-helse website. https://finnkode.ehelse.no/#icd10/0/0/0/-1.
22.Sheehan DV, Lecrubier Y, Sheehan KH, et al. The Mini-International Neuropsychiatric Interview (MINI): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59(suppl 20):22-33, quiz 34-57. PubMed
23.Dierckx B, Heijnen WT, van den Broek WW, et al. Efficacy of electroconvulsive therapy in bipolar versus unipolar major depression: a meta-analysis. Bipolar Disord. 2012;14(2):146-150. PubMed CrossRef
24.Bailine S, Fink M, Knapp R, et al. Electroconvulsive therapy is equally effective in unipolar and bipolar depression. Acta Psychiatr Scand. 2010;121(6):431-436. PubMed CrossRef
25.Montgomery SA, Åsberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134(4):382-389. PubMed CrossRef
26.Electroconvulsive treatment (ECT): National recommendations, advice and packages. The Norwegian Directorate of Health website. https://www.helsedirektoratet.no/tema/elektrokonvulsiv-behandling-ect. Updated June 26, 2017. Accessed March 17, 2019.
27.Oltedal L, Kessler U, Ersland L, et al. Effects of ECT in treatment of depression: study protocol for a prospective neuroradiological study of acute and longitudinal effects on brain structure and function. BMC Psychiatry. 2015;15(1):94. PubMed CrossRef
28.Abrams R. Electroconvulsive Therapy. 4th ed. New York, NY: Oxford University Press; 2002.
29.Shin MS, Park SY, Park SR, et al. Clinical and empirical applications of the Rey-Osterrieth Complex Figure Test. Nat Protoc. 2006;1(2):892-899. PubMed CrossRef
30.McElhiney MC, Moody BJ, Sackheim HA. The Autobiographical Memory Interview-Short Form. New York, NY: Department of Biological Psychiatry: New York State Psychiatric Institute; 2001.
31.Misangyi VF, LePine JA, Algina J, et al. The adequacy of repeated-measures regression for multilevel research: comparisons with repeated-measures ANOVA, multivariate repeated-measures ANOVA, and multilevel modeling across various multilevel research designs. Organ Res Methods. 2006;9(1):5-28. CrossRef
32.Cohen JW. Statistical Power Analysis for the Behavioral Sciences. 2nd ed. Hillsdale, NJ: Lawrence Erlbaum Associates; 1988.
33.Hihn H, Baune BT, Michael N, et al. Memory performance in severely depressed patients treated by electroconvulsive therapy. J ECT. 2006;22(3):189-195. PubMed CrossRef
34.Maric NP, Stojanovic Z, Andric S, et al. The acute and medium-term effects of treatment with electroconvulsive therapy on memory in patients with major depressive disorder. Psychol Med. 2016;46(4):797-806. PubMed CrossRef
35.Kalogerakou S, Oulis P, Anyfandi E, et al. Episodic visual learning/memory and attentional flexibility in patients with major depressive disorder after clinically effective electroconvulsive therapy. J ECT. 2015;31(4):246-252. PubMed CrossRef
36.Fernie G, Bennett DM, Currie J, et al. Detecting objective and subjective cognitive effects of electroconvulsive therapy: intensity, duration and test utility in a large clinical sample. Psychol Med. 2014;44(14):2985-2994. PubMed CrossRef
37.Kessler U, Schoeyen HK, Andreassen OA, et al. The effect of electroconvulsive therapy on neurocognitive function in treatment-resistant bipolar disorder depression. J Clin Psychiatry. 2014;75(11):e1306-e1313. PubMed CrossRef
38.Bjoerke-Bertheussen J, Schoeyen H, Andreassen OA, et al. Right unilateral electroconvulsive therapy does not cause more cognitive impairment than pharmacologic treatment in treatment-resistant bipolar depression: a 6-month randomized controlled trial follow-up study. Bipolar Disord. 2018;20(6):531-538. PubMed CrossRef
39.Semkovska M, McLoughlin DM. Measuring retrograde autobiographical amnesia following electroconvulsive therapy: historical perspective and current issues. J ECT. 2013;29(2):127-133. PubMed CrossRef
40.Chakrabarti S, Grover S, Rajagopal R. Electroconvulsive therapy: a review of knowledge, experience and attitudes of patients concerning the treatment. World J Biol Psychiatry. 2010;11(3):525-537. PubMed CrossRef
41.Rush G, McCarron S, Lucey JV. Patient attitudes to electroconvulsive therapy. Psychiatr Bull. 2007;31(6):212-214. CrossRef
42.Semkovska M, O’ Grady T. Unravelling autobiographical retrograde amnesia following bitemporal electroconvulsive therapy: effect of treatment versus effect of time. Psychology (Irvine). 2017;8(4):611-626. CrossRef
Enjoy this premium PDF as part of your membership benefits!