Satisfaction With Medication Is Correlated With Outcome but Not Persistence in Patients Treated With Placebo, Escitalopram, or Serotonin-Norepinephrine Reuptake Inhibitors: A Post Hoc Analysis
Objective: To test whether satisfaction with taking medication, assessed using item 15 of the Quality of Life Enjoyment and Satisfaction Questionnaire (Q-LES-Q), is associated with clinical outcome and persistence with treatment.
Method: In this post hoc analysis, data were analyzed from 4 randomized placebo-controlled studies of patients with major depressive disorder treated with escitalopram (650 patients taking escitalopram and 534 taking placebo), together with data from 2 randomized trials of escitalopram versus venlafaxine or duloxetine (235 patients taking escitalopram and 233 taking a serotonin-norepinephrine reuptake inhibitor). The studies were published between 2002 and 2007. Instruments included the Q-LES-Q, which was assessed at baseline and week 8, and the Montgomery-Asberg Depression Rating Scale (MADRS), which was assessed at baseline and weeks 1, 2, 4, 6, and 8.
Results: At baseline, the mean ± SD MADRS total score was 30.0 ± 4.6, and the mean Q-LES-Q item 15 score was 2.9 ± 0.9. At week 8, the MADRS response rates of placebo-treated patients with a low, moderate, or high satisfaction with medication at baseline were 30%, 37%, and 46%, respectively (mixed model repeated measures [MMRM]). The corresponding MADRS response rates for escitalopram-treated patients with a low, moderate, or high satisfaction at baseline were 56%, 60%, and 67%, respectively (MMRM). Baseline satisfaction with medication was not significantly correlated with time to withdrawal (all reasons). The change in satisfaction with medication from baseline to endpoint was significantly correlated with symptomatic improvement on the MADRS (P < .001).
Conclusions: Baseline satisfaction with medication after 1 week of placebo run-in is a moderator of treatment outcome but not of persistence of treatment in the acute treatment phase of depressed outpatients. Patient attitude toward medication should be taken into account before treatment is initiated.
Prim Care Companion CNS Disord 2011;13(4):doi:10.4088/PCC.10m01080
© Copyright 2011 Physicians Postgraduate Press, Inc.
Submitted: September 6, 2010; accepted January 31, 2011.
Published online: July 28, 2011 (doi:10.4088/PCC.10m01080).
Corresponding author: Koen Demyttenaere, PhD, Department of Psychiatry, University Hospital Gasthuisberg, Herestraat 49, B-3000 Leuven, Belgium ([email protected]).
Efforts to make clinical trials with antidepressants more informative have mainly focused on clinical and methodological aspects. This includes taking into account higher or lower baseline severity of depression, first-episode or recurrent-episode patients, and 2-arm or 3-arm treatment designs, as well as whether the study included placebo run-in or not, whether the number of visits or the content of the visits were limited, and whether the study prohibited concurrent formal psychotherapy or merely assessed symptoms and adverse events. Less attention has been paid to patient adherence, although this is often suboptimal, even in clinical trials, and adherence influences outcome in acute, continuation, and maintenance phases of treatment with antidepressants.1-4
Clinical Points
- Baseline attitude toward antidepressant medication is only moderately positive, even in patients enrolled in randomized, controlled trials.
- Compared to a more negative baseline attitude toward antidepressants, a more positive attitude results in a better outcome; this effect is more pronounced in patients taking placebo than in patients taking antidepressants.
- Baseline attitude toward antidepressant medication does not seem to influence medication persistence.
Adherence refers to how well a patient follows the physician’s instructions within a designated timeframe,5 whereas medication persistence refers to the act of continuing the treatment for the prescribed duration. It may be defined as “the duration of time from initiation to discontinuation of therapy.”6(p44) Poor persistence with pharmacologic treatment in depression is common in general practice and in clinical trial settings, where adherence is monitored more closely; about 30% of patients do not complete treatment, inevitably diminishing the effectiveness of antidepressants.7
It is thought that persistence is influenced by patient beliefs, which can vary widely.8 Patient persistence seems to be lower when perceived harm exceeds perceived need and higher when perceived need exceeds perceived harm.8,9 Many patients are unenthusiastic about pharmacotherapy (as well as psychotherapy) for the treatment of depression.10,11 However, it has also been suggested that the fear and uncertainty about stopping antidepressants are sometimes stronger than the fear and uncertainty about continuing the medication.12
The perceived need can be seen as the product of the subjective need for treatment and the belief in the efficacy of the antidepressant. The perceived harm can be influenced by the fear of adverse events or by inadequate beliefs about depression and its treatment.13
Patients often have erroneous beliefs about the etiology of a depressive disorder,14 and about what antidepressants can and cannot do.15 Skepticism about antidepressants seems to be stronger among younger patients who have never taken antidepressants, who view their symptoms as mild and transient, and who are unclear about the factors affecting their depression.16 The influence of basic patient attitudes and beliefs on outcome and tolerability has been investigated mainly in observational studies and less frequently in randomized clinical trials.17
The aim of the present investigation was to determine the following:
- If baseline satisfaction with medication differs between patients with and without previous treatment for depression;
- If satisfaction with medication, assessed using the Quality of Life Enjoyment and Satisfaction Questionnaire (Q-LES-Q),18 changes during the acute phase of treatment with antidepressants; and
- If baseline satisfaction with medication is associated with clinical outcome and persistence with treatment.
METHOD
Studies Analyzed
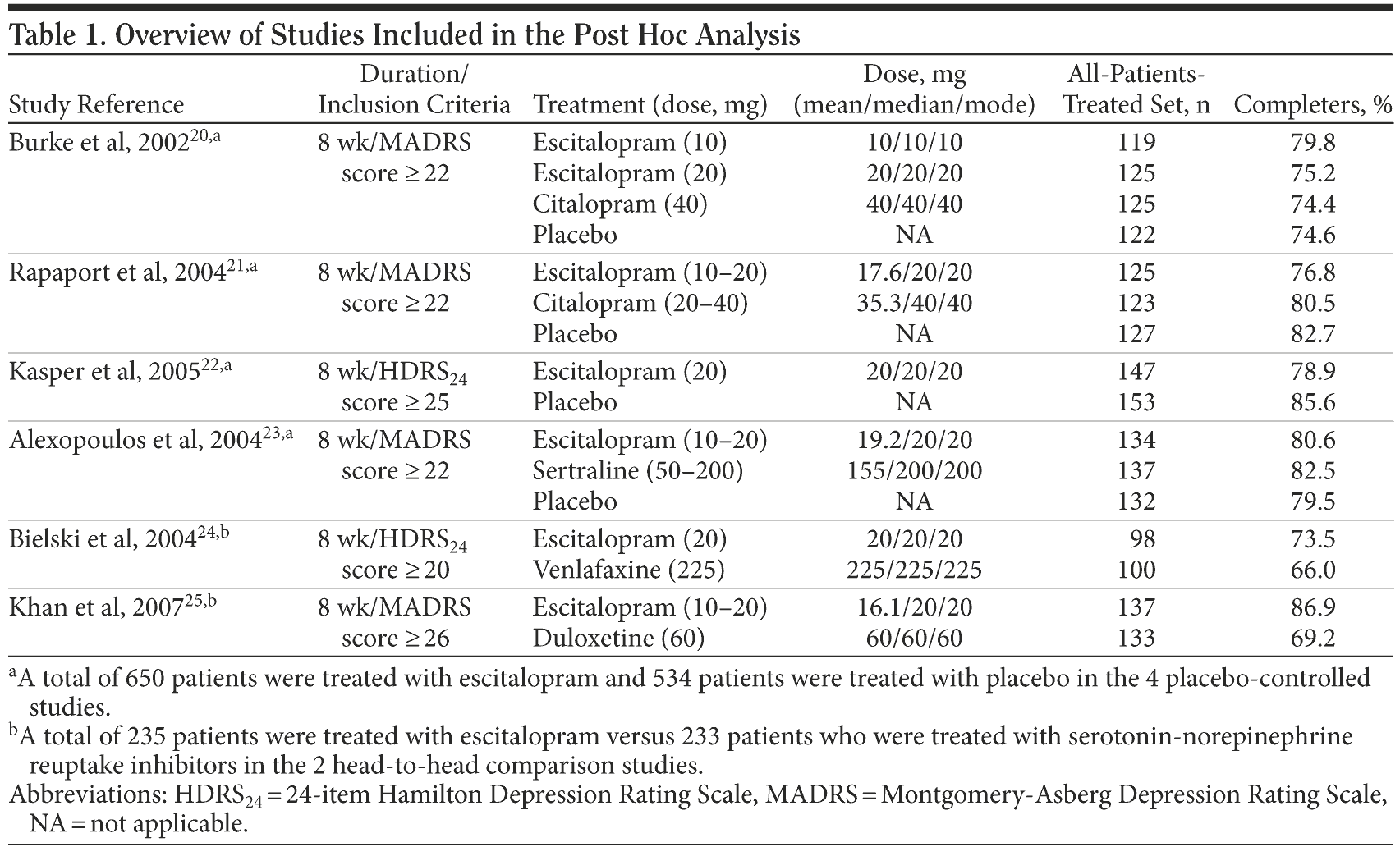
In the present investigation, the following variables were analyzed: Montgomery-Asberg Depression Rating Scale (MADRS)19; Q-LES-Q item 15 (satisfaction with medication); remission, defined as a MADRS score ≤ 12; and response, defined as a 50% reduction from baseline in MADRS score. Safety analyses are based on the all-patients-treated set, comprising all patients who took at least 1 dose of study medication. Efficacy analyses are based on the full analysis set, comprising all treated patients with at least 1 valid postbaseline MADRS assessment. Four placebo-controlled studies of patients with major depressive disorder (MDD) treated with escitalopram assessed the 16-item Q-LES-Q.20-23 The Q-LES-Q was also used in 2 head-to-head comparisons, one of escitalopram and the serotonin-norepinephrine reuptake inhibitor (SNRI) venlafaxine24 and a second of escitalopram and the SNRI duloxetine.25 The studies are summarized in Table 1. The studies were published between 2002 and 2007.
At baseline, all patients had experienced 1 week of single-blind placebo run-in. The MADRS was assessed at baseline and weeks 1, 2, 4, 6, and 8 in all studies. The Q-LES-Q was assessed at baseline and at last assessment in all studies. Item 15 is phrased, “Taking everything into consideration, during the past week how satisfied have you been with your medication? (If not taking any, leave item blank).” For each study, persistence with treatment was inferred from the overall withdrawal rate in each treatment group. The correlation between Q-LES-Q item 15 at baseline with outcome and persistence was calculated. The Q-LES-Q analysis was restricted by having assessments only at baseline and at the end of the study, so the observed change from baseline needs to be interpreted with care.
Statistical Methodology
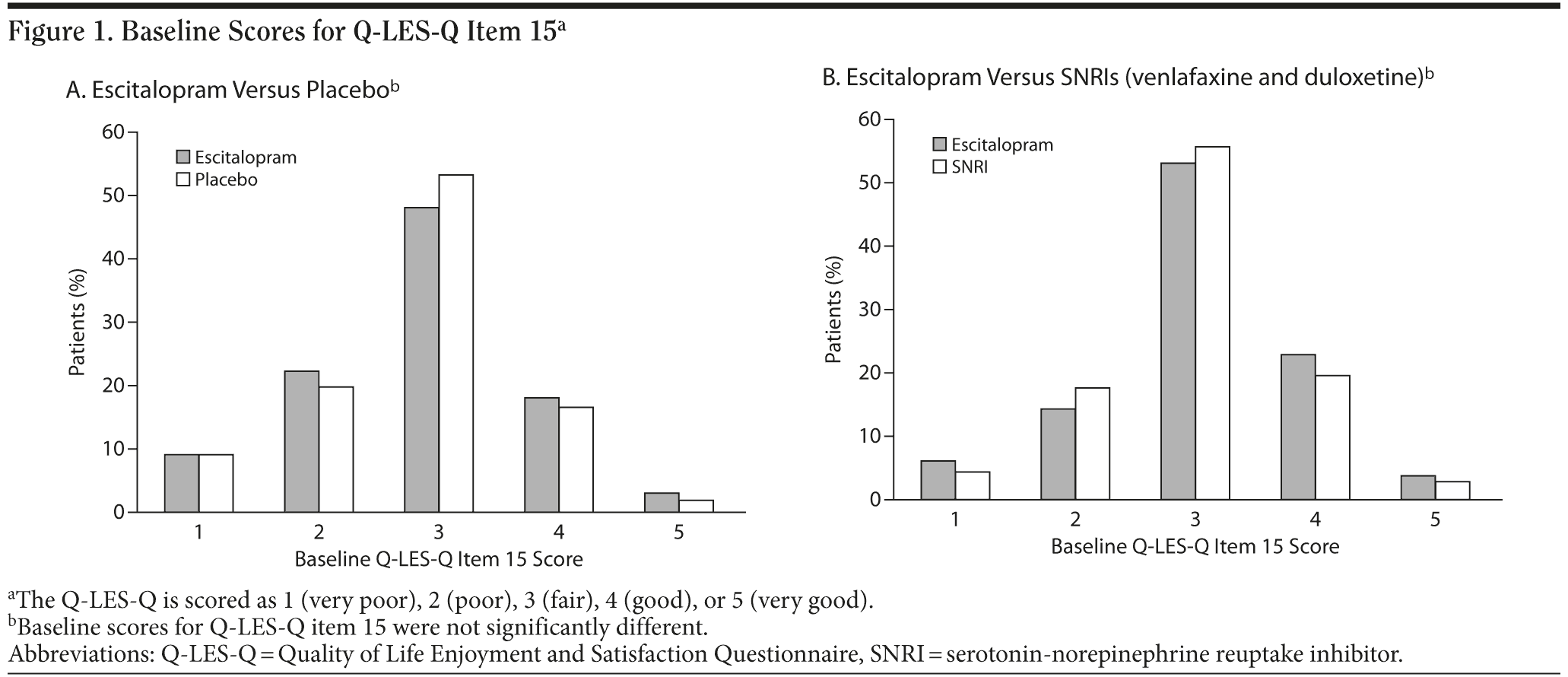
The Q-LES-Q is scored as 1 (very poor), 2 (poor), 3 (fair), 4 (good), or 5 (very good) for each item of the questionnaire. The distribution of scores for item 15 was symmetric around the midpoint, and the proportion of patients scoring 1 and 5 was low (Figure 1A and 1B). In addition, it is questionable if a change from 1 to 2 is the same as a change from 2 to 3 or from 3 to 4; thus, in order to simplify the interpretation of the results, and due to the low number of patients scoring 1 and 5, patients were grouped into 3 classes on the basis of their baseline Q-LES-Q item 15 score (satisfaction with medication): low (scoring 1-2), fair (scoring 3), and high (scoring 4-5). This was then treated as a class variable when included in the analysis. All statistical analyses used SAS version 9.2 (SAS Institute, Inc, Cary, North Carolina).
The mean change in MADRS score was estimated for each baseline item 15 class using mixed model repeated measures (MMRM) and the MIXED procedure. In these analyses, baseline MADRS score was included as a covariate and study center, week, treatment, and grouped baseline item 15 were factors. Interactions between week and treatment and baseline item 15 were also included in the model to test if baseline item 15 was associated with the outcome. An unstructured covariance structure was assumed, and the Kenward-Roger approximation was used to estimate the denominator degrees of freedom.
The proportions of remitters and responders were estimated using last observation carried forward (LOCF) and observed cases (OC), as well as MMRM (for which no imputation of missing data is required). For LOCF and OC, a logistic regression model was used to test for differences between the baseline item 15 classes within each treatment group; baseline MADRS score was included as a covariate, and study center, treatment, and baseline Q-LES-Q group were included as factors together with interaction between treatment and baseline Q-LES-Q group. A comparable model was used in the MMRM approach, wherein week is also included as a factor together with the interaction term between week, treatment, and baseline Q-LES-Q group. The GENMOD procedure (SAS Institute, Inc, Cary, North Carolina) was used for the analysis of all binary outcome variables. To investigate if the findings were consistent between the subgroups of previously treated and not previously treated patients, an interaction between baseline Q-LES-Q score and earlier treatment of MDD (defined as a binary variable) was included and tested for significance. In the latter analysis and in the analyses where all active groups are pooled together, a correction for study instead of centers was made.
The Cochran-Armitage test for trends in proportions was used to test whether the baseline Q-LES-Q score followed a different distribution for the previously treated patients compared with those who were not previously treated.
The withdrawal patterns over time for the different groups were tabulated. The time course of withdrawal was investigated using survival analysis methods to test whether different factors, such as baseline Q-LES-Q item 15 score or treatment group, had any impact on the time to withdrawal. A patient can only withdraw once, so a patient who withdraws for one reason cannot withdraw for another reason, thus, a comparison of rates is not valid.
RESULTS
Patient Characteristics
Six studies, all in specialist settings in the United States, were included (see Table 1). Over all studies, a total of 1,652 patients had a mean ± SD age of 40.1 ± 12.1 years, 60% were women, and 80% were white. The mean ± SD MADRS total score was 30.0 ± 4.6. There were no significant differences between the treatment groups at baseline. Almost half the patients (48%) had been treated previously with antidepressants for MDD. In the 6 trials, all patients received single-blind lead-in treatment with placebo for 1 week.
Baseline Q-LES-Q Item 15 Scores
The Q-LES-Q item 15 score reflected the patient’s satisfaction with the medication (single-blind placebo) that had been received during the 7 days between screening and baseline. During this period, the mean ± SD MADRS score decreased from 30.9 ± 4.4 at screening to 30.0 ± 4.6 at baseline. There were 41 (2.5%) patients with no baseline item 15 score (27 in the escitalopram group and 10 in the corresponding placebo group, and 1 in the SNRI group and 3 in the corresponding escitalopram group). At baseline, the mean ± SD Q-LES-Q item 15 score was 2.9 ± 0.9 for all treatment groups. The distribution of Q-LES-Q item 15 scores at baseline is shown in Figure 1A and B.
The baseline Q-LES-Q item 15 score was not correlated with patient age or sex. There was a small, but significant, negative correlation between the baseline Q-LES-Q item 15 score and the baseline MADRS score (Pearson’s correlation coefficient of −0.2, P < .001). For patients with a low baseline satisfaction with medication (n = 442), 54% had previously been treated; for patients with a fair satisfaction (n = 830), 48% had previously been treated; and for patients with a high satisfaction (n = 339), 44% had previously been treated. The tendency for fewer patients to have been previously treated for depression with increasing baseline satisfaction with medication was significant (Cochran-Armitage test, P < .01).
Baseline Q-LES-Q Item 15 Scores (satisfaction with medication) and Outcome
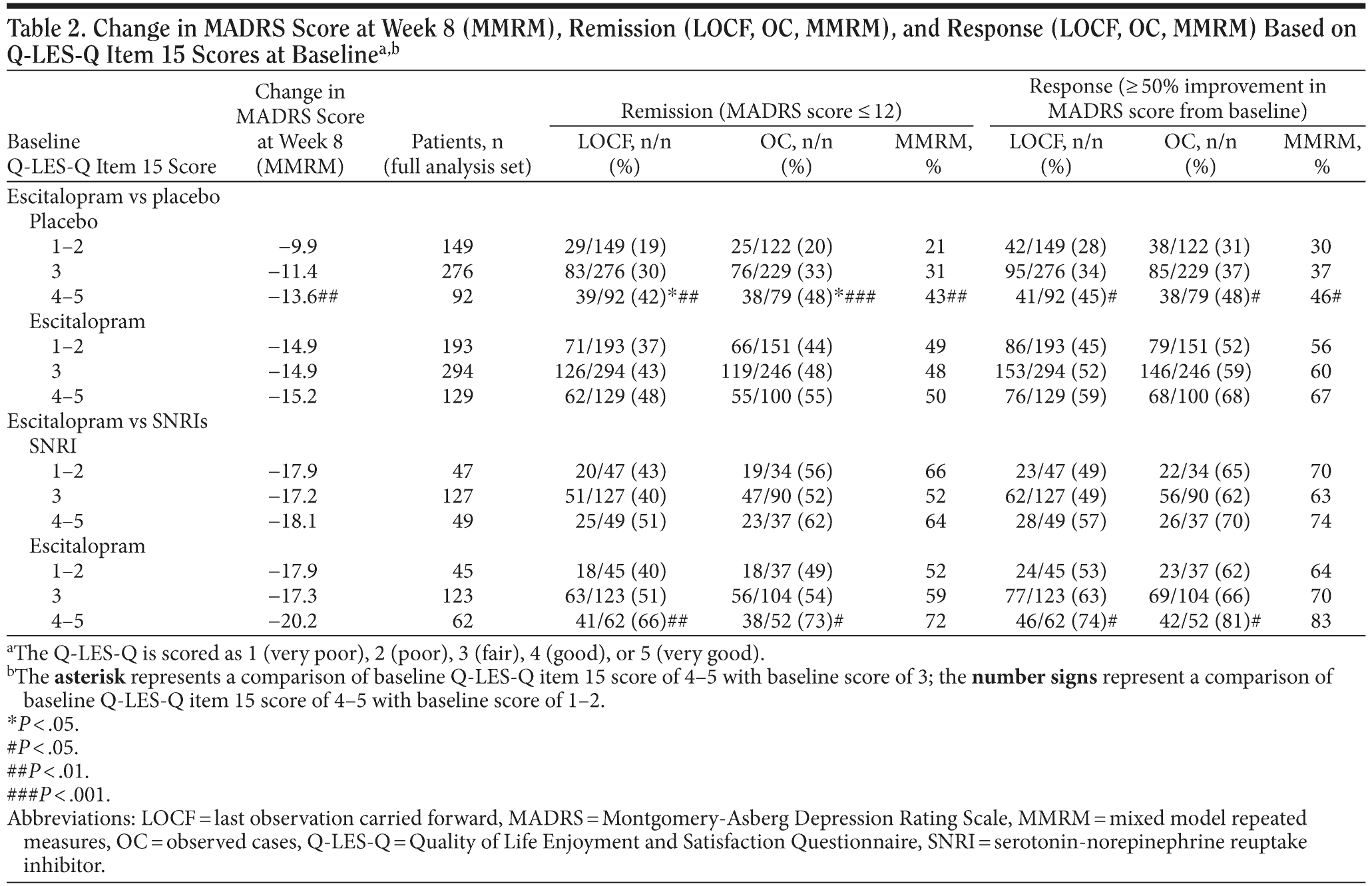
The effect of baseline item 15 scores on outcome was investigated using continuous (mean change in MADRS scores using MMRM) as well as categorical outcome measures (response and remission rates using OC, LOCF, and MMRM). Table 2 provides the results for the individual comparisons (escitalopram versus placebo and escitalopram versus SNRIs). In Table 3, the data for all active treatments were pooled and stratified by previous treatment. Tables 2 and 3 show that patients with a more positive baseline satisfaction with medication had a better outcome than patients with a more negative baseline satisfaction with medication, based on continuous outcomes. This was true both for patients taking placebo as well as for patients on active treatment (escitalopram or SNRIs).
Regarding the categorical endpoints, placebo patients with a positive baseline satisfaction with medication had significantly higher response and remission rates than placebo patients with a negative baseline attitude. The same trend was seen in patients receiving active treatment, although it was not as strong as in placebo patients.
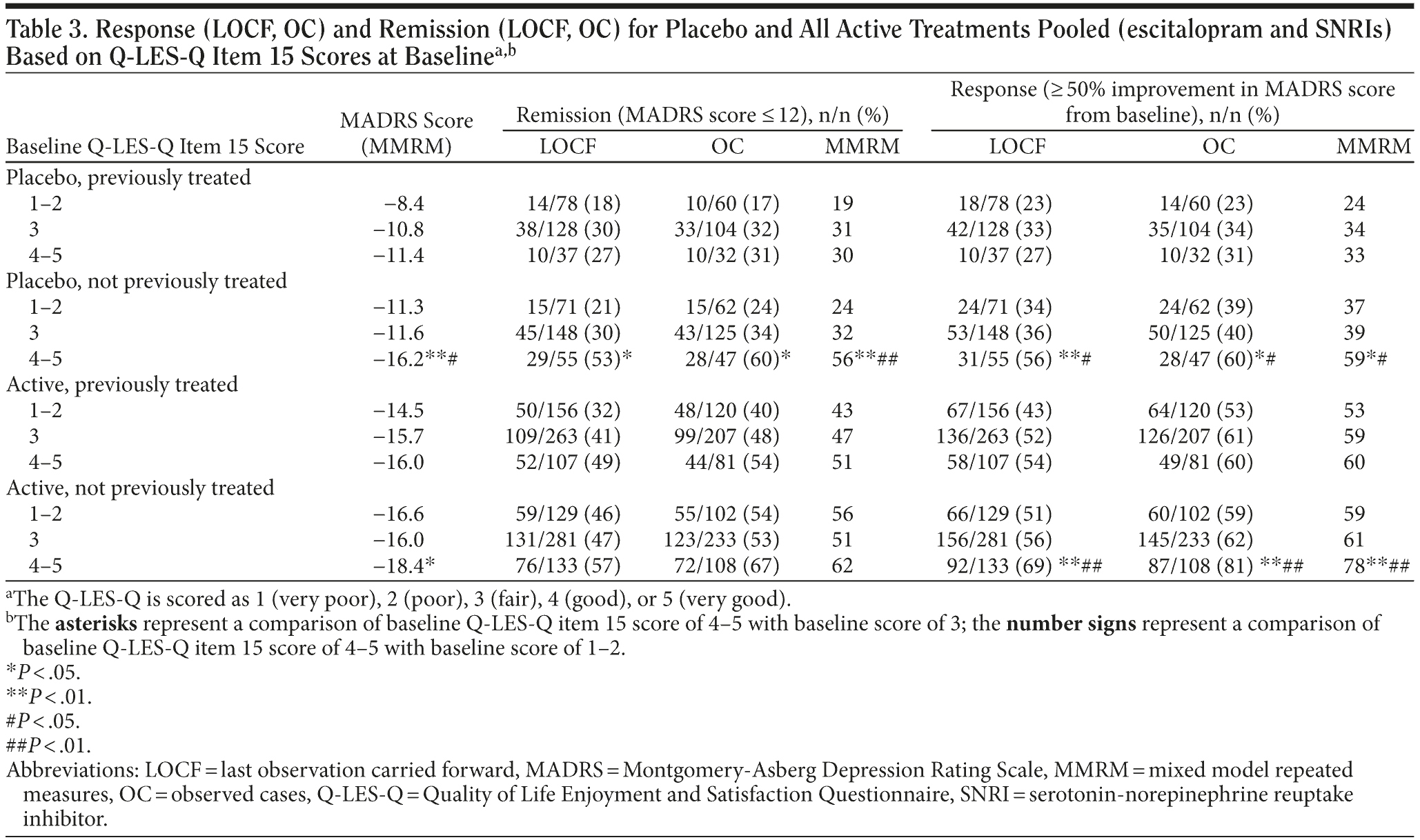
The association of baseline satisfaction with medication with outcome remained statistically significant, even after controlling for patients having been treated previously with an antidepressant (Table 3). However, this association appears to be even greater for patients not previously treated. Thus, patients with a more positive attitude toward medication and not previously treated for depression show a large response to placebo (Table 3).
Baseline Q-LES-Q Item 15 Scores (satisfaction with medication) and Persistence
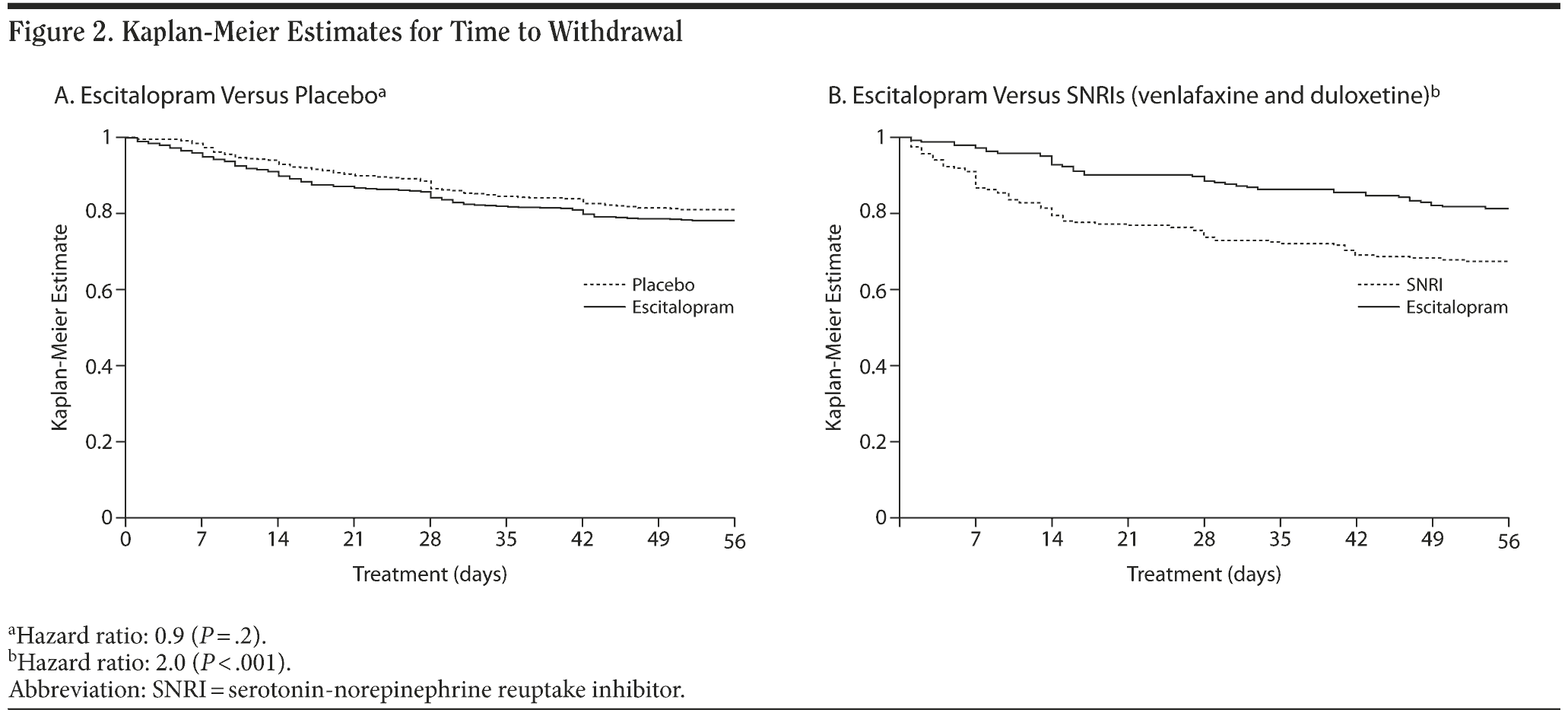
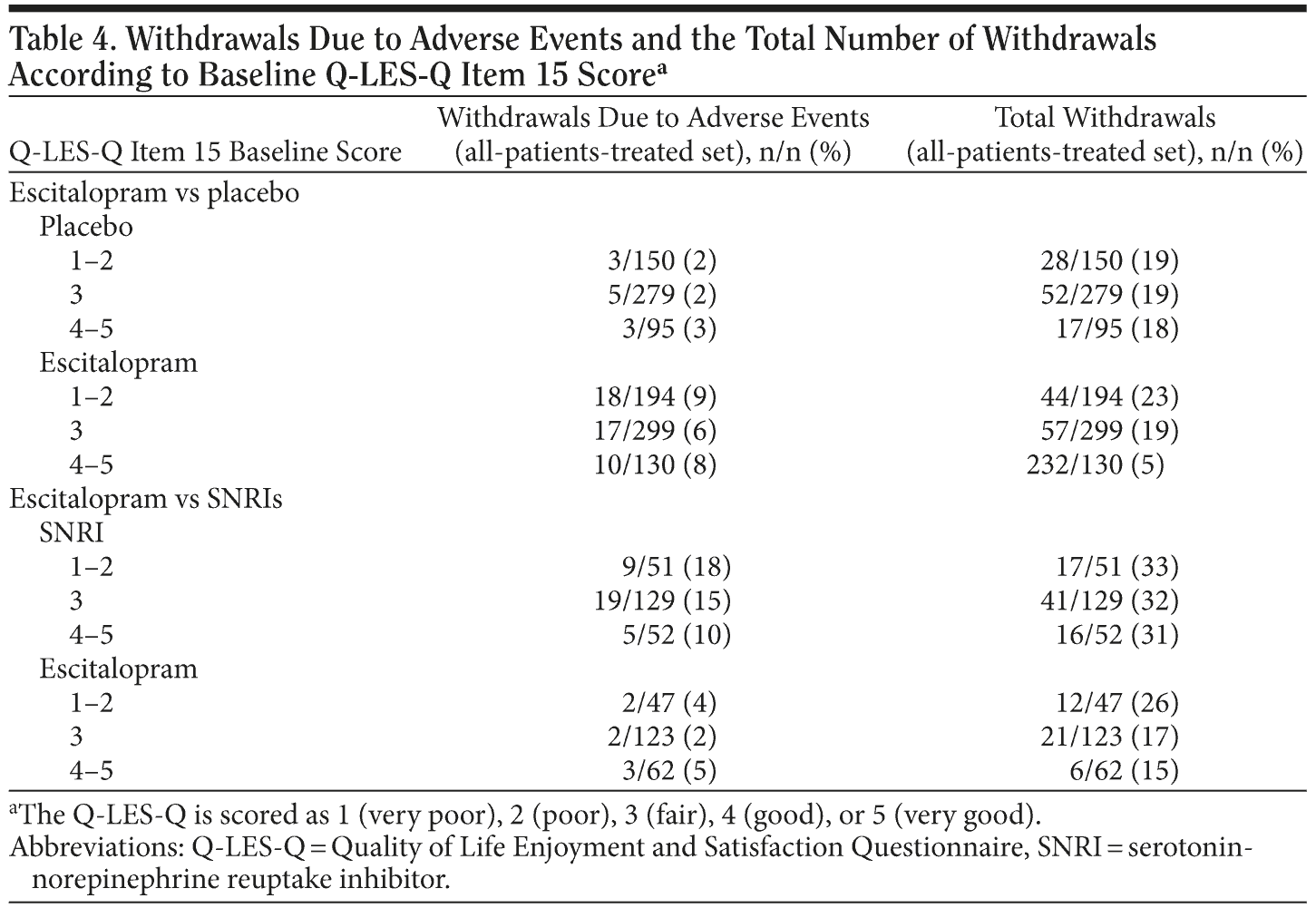
Persistence with treatment (Figure 2A and 2B) was similar in placebo patients and escitalopram patients, while it was lower in SNRI patients than in escitalopram patients in the same trials (P < .001, log rank test). Withdrawal rates are shown in Table 4 and illustrate that there was no significant association with the baseline Q-LES-Q item 15 score.
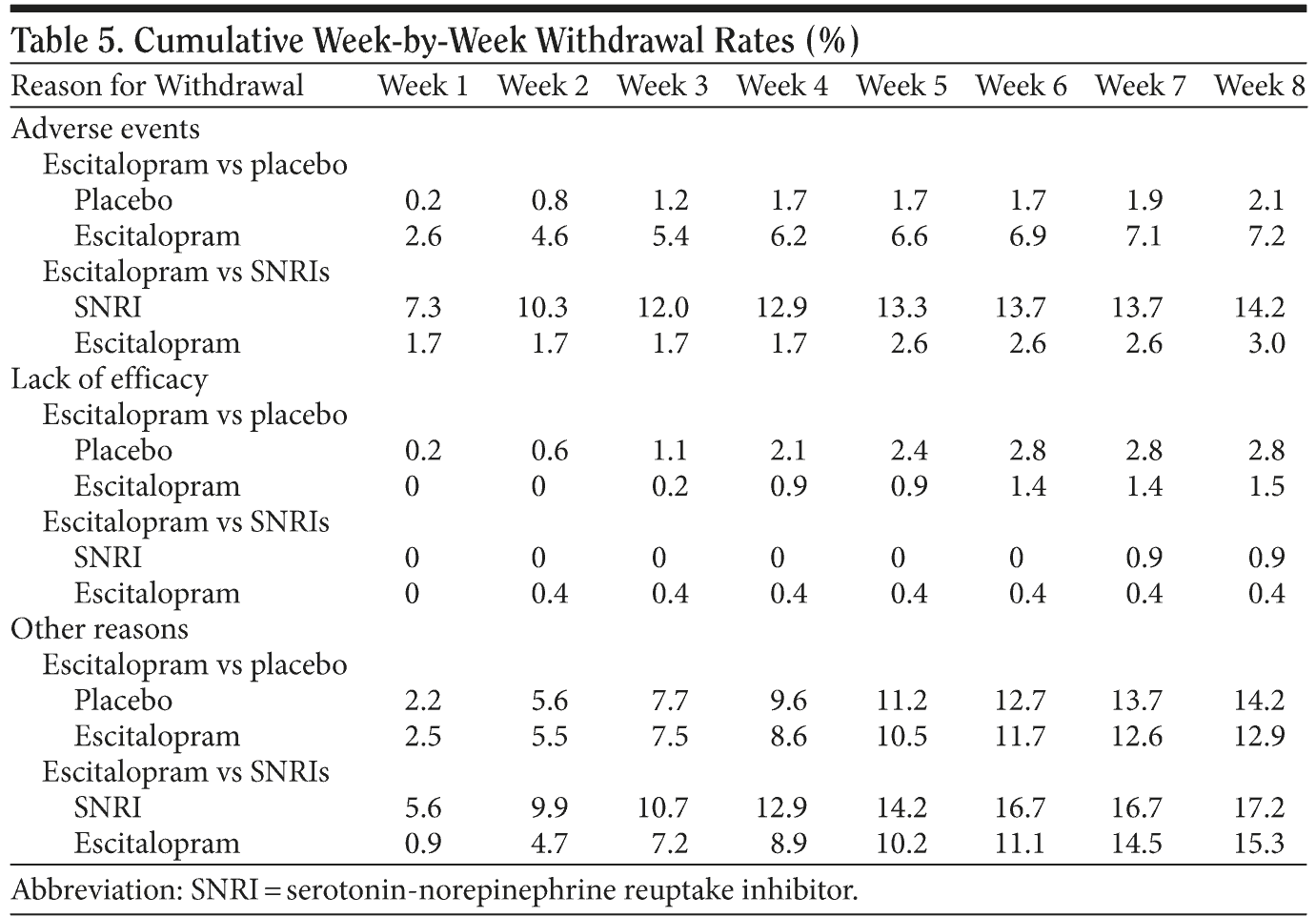
As shown in Figure 2A, there was no significant difference in the time to withdrawal between escitalopram and placebo. There was a significant difference (P < .001, log rank test) in the time to withdrawal between escitalopram and SNRIs in favor of escitalopram (Figure 2B), as well as the time to withdrawal due to adverse events (Table 5) (P < .001, log rank test). The baseline item 15 score had no significant association with the time to withdrawal for any reason, or the withdrawal rate due to adverse events. In the studies comparing escitalopram with placebo, a larger proportion of the withdrawals were due to adverse events (P < .001) in the escitalopram group compared with the placebo group.
Change From Baseline in Q-LES-Q Item 15 Scores (observed cases)
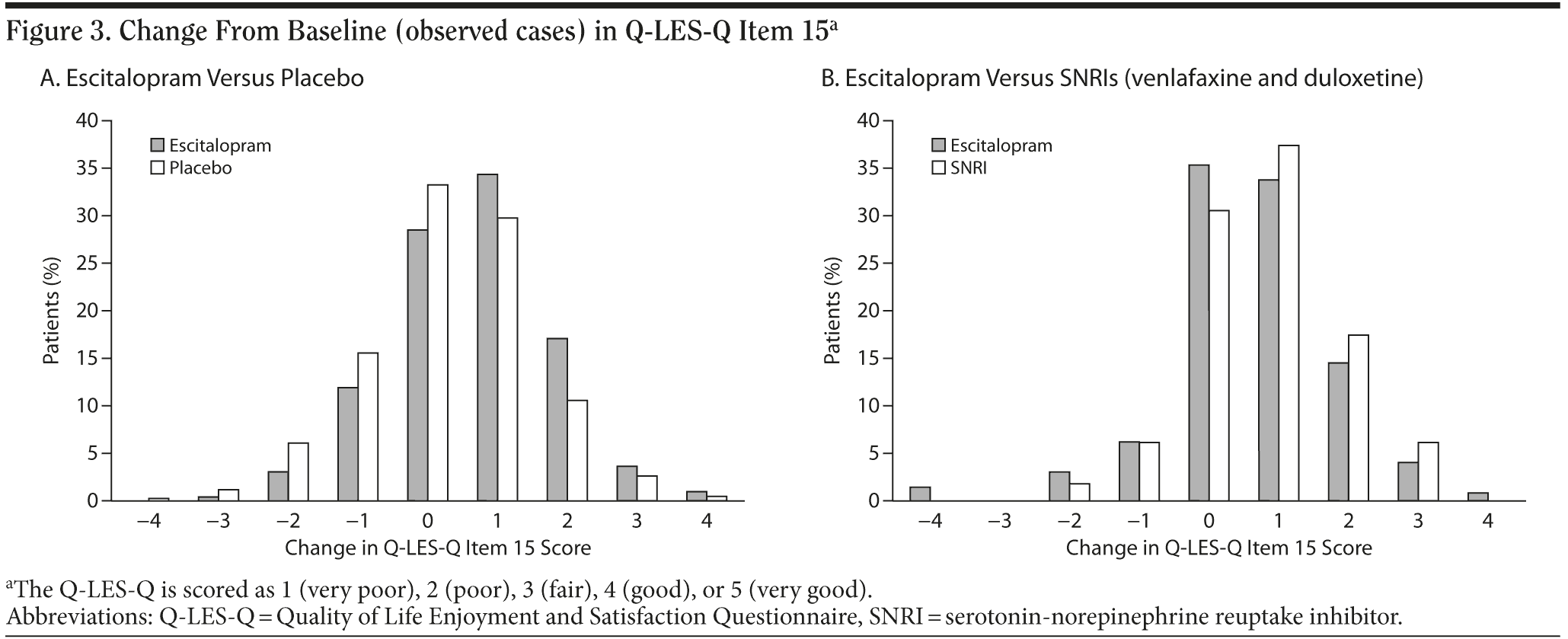
The Q-LES-Q item 15 scores improved for the majority of patients after 8 weeks of treatment (Figure 3A and 3B). An improvement (increase) in the item 15 score was correlated with an improvement (decrease) in the MADRS score. The Pearson correlation coefficient was −0.5 in the escitalopram and placebo analyses and −0.4 in the escitalopram and SNRI analyses (both correlations are significant, P < .001).
In the placebo-controlled studies, patients whose Q-LES-Q item 15 score worsened at week 8 (n = 173) had an estimated mean ± SD change from baseline of 5.5 ± 7.8 MADRS points. Patients with no change in the Q-LES-Q item 15 score at week 8 (n = 279) had an estimated mean ± SD change from baseline of 11.2 ± 8.8 MADRS points. Patients with an improvement in the Q-LES-Q item 15 score at week 8 (n = 455) had an estimated mean ± SD change from baseline of 18.3 ± 9.2 MADRS points.
In the comparator studies of escitalopram versus SNRIs, patients whose Q-LES-Q item 15 score worsened at week 8 (n = 34) had an estimated mean ± SD change from baseline of 9.7 ± 8.2 MADRS points. Patients with no change in the Q-LES-Q item 15 score at week 8 (n = 116) had an estimated mean ± SD change from baseline of 16.0 ± 8.9 MADRS points. Patients with an improvement in the Q-LES-Q item 15 score at week 8 (n = 200) had an estimated mean ± SD change from baseline of 20.9 ± 7.9 MADRS points.
DISCUSSION
The baseline satisfaction with medication was only moderately positive, since about 50% of the included patients considered their attitude toward medication as fair, while between 20% and 30% considered their attitude as very poor or poor. Compared to depressed patients in the community, there was probably a selection bias, because all of these depressed patients consented to treatment with medication. It is indeed known that patients as well as the general population have rather negative opinions about antidepressants.10,11 Since all patients entered these trials with a single-blind placebo run-in week, item 15 of this scale was probably assessing patients’ satisfaction with placebo; however, there were no differences between treatment groups at randomization.
The most important findings of this investigation are that baseline satisfaction with medication significantly influences outcome, but not persistence with treatment. Indeed, patients with a more positive baseline satisfaction with medication (good or very good) had a better outcome with placebo as well as with active treatment. This is not only true for the continuous endpoint measures (change in MADRS score) but also for the categorical endpoint measures (response and remission). The effect of satisfaction with medication on outcome seemed to be more pronounced in the placebo group than in the active treatment groups. The data show that the difference in outcome between patients with a higher and patients with a lower baseline satisfaction with medication is almost as pronounced as the difference between placebo patients and active treatment patients. For example, the response rates (MMRM) for patients taking placebo with a higher versus lower satisfaction with medication were 46% versus 30%, and the response rates for fair attitude patients taking placebo versus fair attitude patients on active treatment were 37% versus 60%.
This robust effect of satisfaction with medication on outcome is probably a contributor to the well-documented large placebo effect in patients treated for depression; for example, the placebo effect is estimated to be about 50% of the response to pain medication and about 80% of the improvement observed with antidepressants.26,27 This is comparable to the results from studies looking at the effect of psychotherapy; no distinct differences have been found between differing psychotherapies, and it has been shown that about 20% of the effect is due to specific psychotherapeutic techniques, the rest being “common” or “aspecific” factors.28,29 This suggests that, in depression, the so-called placebo effect is large and due to the caring doctor-patient relationship, as well as to specific factors such as hope, expectations, and beliefs. The more frequently patients are seen by their treating physician, the larger the effect of placebo and of antidepressants. It has, for example, been shown that the effect of an antidepressant and of a placebo is greater in 6-week clinical trials when patients are seen weekly than when they are seen less frequently.30 Only a robust improvement in symptomatology seems to increase the satisfaction with medication. Indeed, in placebo-treated or in active medication-treated patients, response (defined as ≥ 50% improvement from baseline) generally results in improved satisfaction. Nonresponse (defined as < 20% improvement from baseline) generally results in a worsened satisfaction, probably because these improvements are below initial expectations.
The present study does not show any relation between satisfaction with medication at baseline after 1 week of single-blind placebo treatment and persistence with treatment. This is in contrast with the existing literature, wherein it has been documented that persistence with treatment (during maintenance phase) with antidepressants depends on the balance between perception of need and harmfulness of antidepressant medication concerns.8 The present investigation differs from the latter study in that the present investigation includes data from acute phase studies and that the single item measure of satisfaction with medication in the present investigation does not sufficiently differentiate between necessity of treatment and concerns about medication. It has indeed been documented that reasons for dropout in the earlier phases of treatment are mainly due to lack of efficacy and adverse events, while the reasons for dropout in the later phases are due more to psychological reasons and attitudes.1,2
In the present placebo-controlled studies, it is interesting that patients with a high baseline satisfaction with medication randomized to placebo have a significantly better outcome than those with a low baseline satisfaction with medication. This result was not seen for patients randomized to active treatment in the present analysis. However, this may be a way to identify at baseline patients who respond just as well to placebo as to active treatment. The inclusion of placebo responders is thought to be a factor in failed clinical trials, and at least 1 patient-rated screening instrument has been suggested to identify such patients in generalized anxiety disorder trials.31 Several of the 20 items included in this scale are related to patients’ attitudes toward medication, and excluding the approximately 37% of patients with a cutoff score of 50 or more decreased the mean change from baseline on the Hamilton Anxiety Scale, but increased the difference to placebo from −3.2 to −6.1 points.31
The present investigation has several limitations. Satisfaction with medication was assessed with a single item of the Q-LES-Q, which is not a validated instrument for the assessment of compliance with medication. The results of these post hoc analyses from existing datasets should be considered as correlations, with no causality implied.
In conclusion, baseline satisfaction with medication is only fair in patients enrolled in clinical trials. Only a robust improvement in symptomatology seems to make this satisfaction greater. Thus, baseline satisfaction with medication is significantly associated with outcome but not with persistence with treatment.
Drug names: citalopram (Celexa and others), duloxetine (Cymbalta), escitalopram (Lexapro and others), sertraline (Zoloft and others), venlafaxine (Effexor and others).
Author affiliations: Department of Psychiatry, University Hospital Gasthuisberg, Leuven, Belgium (Dr Demyttenaere); Clinical Psychopharmacology, King’s College London, Institute of Psychiatry, United Kingdom (Dr Lader); and H. Lundbeck A/S, International Clinical Research, Copenhagen Valby, Denmark (Drs Reines and Lönn).
Potential conflicts of interest: Drs Demyttenaere and Lader are members of the escitalopram advisory board and have received honoraria from H. Lundbeck A/S. Drs Reines and Lönn are employees of H. Lundbeck A/S, the manufacturer of escitalopram.
Funding/support: The data are from studies that were sponsored by Forest Laboratories, Inc.
Previous presentation: Presented in part as a poster at the 162nd Annual Meeting of the American Psychiatric Association; May 16-21, 2009; San Francisco, California.
Acknowledgment: The authors gratefully acknowledge the technical assistance of David John Simpson, PhD, during the preparation of the manuscript. Dr Simpson is an employee of H. Lundbeck A/S, the manufacturer of escitalopram, and reports no other conflicts of interest related to the subject of this article.
REFERENCES
1. Demyttenaere K, Enzlin P, Dewé W, et al. Compliance with antidepressants in a primary care setting, 1: beyond lack of efficacy and adverse events. J Clin Psychiatry. 2001;62(suppl 22):30-33. PubMed
2. Demyttenaere K, Enzlin P, Dewé W, et al. Compliance with antidepressants in a primary care setting, 2: the influence of gender and type of impairment. J Clin Psychiatry. 2001;62(suppl 22):34-37. PubMed
3. von Knorring L, Akerblad AC, Bengtsson F, et al. Cost of depression: effect of adherence and treatment response. Eur Psychiatry. 2006;21(6):349-354. PubMed doi:10.1016/j.eurpsy.2006.04.005
4. Melfi CA, Chawla AJ, Croghan TW, et al. The effects of adherence to antidepressant treatment guidelines on relapse and recurrence of depression. Arch Gen Psychiatry. 1998;55(12):1128-1132. PubMed doi:10.1001/archpsyc.55.12.1128
5. Cramer J, Rosenheck R, Kirk G, et al; VA Naltrexone Study Group 425. Medication compliance feedback and monitoring in a clinical trial: predictors and outcomes. Value Health. 2003;6(5):566-573. PubMed doi:10.1046/j.1524-4733.2003.65269.x
6. Cramer JA, Roy A, Burrell A, et al. Medication compliance and persistence: terminology and definitions. Value Health. 2008;11(1):44-47. doi: 10.1111/j.1524-4733.2007.00213.x PubMed
7. Bollini P, Pampallona S, Kupelnick B, et al. Improving compliance in depression: a systematic review of narrative reviews. J Clin Pharm Ther. 2006;31(3):253-260. PubMed doi:10.1111/j.1365-2710.2006.00733.x
8. Aikens JE, Nease DE Jr, Nau DP, et al. Adherence to maintenance-phase antidepressant medication as a function of patient beliefs about medication. Ann Fam Med. 2005;3(1):23-30. PubMed doi:10.1370/afm.238
9. Russell J, Kazantzis N. Medication beliefs and adherence to antidepressants in primary care. N Z Med J. 2008;121(1286):14-20. PubMed
10. Angermeyer MC, Matschinger H. Public attitude towards psychiatric treatment. Acta Psychiatr Scand. 1996;94(5):326-336. PubMed doi:10.1111/j.1600-0447.1996.tb09868.x
11. Benkert O, Graf-Morgenstern M, Hillert A, et al. Public opinion on psychotropic drugs: an analysis of the factors influencing acceptance or rejection. J Nerv Ment Dis. 1997;185(3):151-158. PubMed doi:10.1097/00005053-199703000-00004
12. Verbeek-Heida PM, Mathot EF. Better safe than sorry—why patients prefer to stop using selective serotonin reuptake inhibitor (SSRI) antidepressants but are afraid to do so: results of a qualitative study. Chronic Illn. 2006;2(2):133-142. PubMed
13. Horne RE. Treatment perception and self-regulation. In: Cameron LD, Leventhal H, eds. The Self-Regulation of Health and Illness Behaviour. London, UK: Routledge; 2003:138-153.
14. Priest RG, Vize C, Roberts A, et al. Lay people’s attitudes to treatment of depression: results of opinion poll for Defeat Depression Campaign just before its launch. BMJ. 1996;313(7061):858-859. PubMed
15. Demyttenaere K, Bruffaerts R, Albert A, et al. Development of an antidepressant compliance questionnaire. Acta Psychiatr Scand. 2004;110(3):201-207. PubMed doi:10.1111/j.1600-0447.2004.00350.x
16. Aikens JE, Nease DE Jr, Klinkman MS. Explaining patients’ beliefs about the necessity and harmfulness of antidepressants. Ann Fam Med. 2008;6(1):23-29. PubMed doi:10.1370/afm.759
17. Brown C, Battista DR, Bruehlman R, et al. Beliefs about antidepressant medications in primary care patients: relationship to self-reported adherence. Med Care. 2005;43(12):1203-1207. PubMed doi:10.1097/01.mlr.0000185733.30697.f6
18. Endicott J, Nee J, Harrison W, et al. Quality of Life Enjoyment and Satisfaction Questionnaire: a new measure. Psychopharmacol Bull. 1993;29(2):321-326. PubMed
19. Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134:382-389. PubMed
20. Burke WJ, Gergel I, Bose A. Fixed-dose trial of the single isomer SSRI escitalopram in depressed outpatients. J Clin Psychiatry. 2002;63(4):331-336. PubMed
21. Rapaport MH, Bos A, Zheng H. Escitalopram continuation treatment prevents relapse of depressive episodes. J Clin Psychiatry. 2004;65(1):44-49.
22. Kasper S, Ninan P, Ventura D, et al. Escitalopram in the treatment of severe depression. Eur Psychiatry. 2005;20(suppl 1):S143. http://www.forestclinicaltrials.com/CTR/CTRController/CTRViewPdf?_file_id=scsr/SCSR_SCT-MD-26_final.pdf. Accessed December 14, 2009.
23. Alexopoulos GS, Gordon J, Zhang D. A placebo-controlled trial of escitalopram and sertraline in the treatment of major depressive disorder. Neuropsychopharmacology. 2004;29(suppl):S87. http://www.forestclinicaltrials.com/CTR/CTRController/CTRViewPdf?_file_id=scsr/SCSR_SCT-MD-27_final.pdf. Accessed December 14, 2009.
24. Bielski RJ, Ventura D, Chang C-C. A double-blind comparison of escitalopram and venlafaxine extended release in the treatment of major depressive disorder. J Clin Psychiatry. 2004;65(9):1190-1196. PubMed doi:10.4088/JCP.v65n0906
25. Khan A, Bose A, Alexopoulos GS, et al. Double-blind comparison of escitalopram and duloxetine in the acute treatment of major depressive disorder. Clin Drug Investig. 2007;27(7):481-492. PubMed doi:10.2165/00044011-200727070-00005
26. Kirsch I, Deacon BJ, Huedo-Medina TB, et al. Initial severity and antidepressant benefits: a meta-analysis of data submitted to the Food and Drug Administration. PLoS Med. 2008;5(2):e45. PubMed doi:10.1371/journal.pmed.0050045
27. Benedetti F, Vighetti S, Ricco C, et al. Pain threshold and tolerance in Alzheimer’s disease. Pain. 1999;80(1-2):377-382. PubMed doi:10.1016/S0304-3959(98)00228-0
28. Wampold BE, Mondin G, Moody M, et al. A meta-analysis of outcome studies comparing bona fide psychotherapies: empirically, ‘ all must have prizes.’ Psychol Bull. 1997;122(3):203-215. doi:10.1037/0033-2909.122.3.203
29. Lambert MJ. Early response in psychotherapy: further evidence for the importance of common factors rather than “placebo effects.” J Clin Psychol. 2005;61(7):855-869. PubMed doi:10.1002/jclp.20130
30. Posternak MA, Zimmerman M. Therapeutic effect of follow-up assessments on antidepressant and placebo response rates in antidepressant efficacy trials: metaanalysis. Br J Psychiatry. 2007;190(4):287-292. PubMed doi:10.1192/bjp.bp.106.028555
31. Feltner D, Hill C, Lenderking W, et al. Development of a patient-reported assessment to identify placebo responders in a generalized anxiety disorder trial. J Psychiatr Res. 2009;43(15):1224-1230. PubMed doi:10.1016/j.jpsychires.2009.04.001