ABSTRACT
Objective: To analyze all current literature related to different transcranial magnetic stimulation (TMS) modalities in the management of schizophrenia symptoms and compare the main controversies in the outcome measures.
Data Sources: A comprehensive search in Ovid MEDLINE, APA PsycINFO, and PubMed was conducted (1996 to present) to identify all articles using the search terms schizophrenia and single pulse TMS, schizophrenia and paired pulse TMS, schizophrenia and repetitive TMS, schizophrenia and deep TMS, and schizophrenia and TBS.
Data Selection: Search results were limited to the English language and human subjects. Unrelated articles were excluded after the initial review. Nineteen studies fulfilled the eligibility criteria. These studies included 531 schizophrenia patients and 283 healthy controls.
Results: While some adverse effects of TMS are reported, the process is generally considered safe. However, discrepancies exist regarding the length of the cortical silent period. The cortical silent period is thought to be mediated by γ-aminobutyric acid (GABA) receptors and is considered a useful probe to assess motor cortical inhibition. It is generally believed that patients with schizophrenia present changes in the cortical silent period due to GABA abnormalities. A few physiologic studies utilized TMS to study motor cortical excitability in schizophrenia and reported longer, and some shorter, cortical silent periods compared with healthy controls. Also, some studies reported a shorter cortical silent period for unmedicated versus medicated patients, while others reported no differences.
Conclusion: TMS treatment is commonly reported to improve auditory hallucination in schizophrenia. Advantage of use of deep TMS over repetitive TMS, however, is controversial.
Prim Care Companion CNS Disord 2022;24(4):21r03144
To cite: Soltani E, Bateni H, Shad MU. Different modalities of transcranial magnetic stimulation to manage schizophrenia. Prim Care Companion CNS Disord. 2022;24(4):21r03144.
To share: https://doi.org/10.4088/PCC.21r03144
© 2022 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, The Valley Health System, Las Vegas, Nevada
bPhysical Therapy Program, Northern Illinois University, DeKalb, Illinois
cDepartment of Psychiatry, University of Nevada Las Vegas, Las Vegas, Nevada
dDepartment of Psychiatry, Touro University Nevada College of Osteopathic Medicine, Las Vegas, Nevada
*Corresponding author: Elham Soltani, MD, The Valley Health System, 2075 E Flamingo Rd, Las Vegas, NV 08119 ([email protected]).
Schizophrenia is a chronic debilitating condition that has life-long complications in terms of stress, cost, and quality of life for patients and their families. Approximately 1% of the world population is diagnosed with schizophrenia, generally during their early 20s.1 Schizophrenia is one of the most complex syndromes of the brain and involves multiple symptom domains with different neurobiological underpinnings, which explains why there is so much interindividual variability in clinical presentation and treatment response in these patients. The core symptom domains that are globally presented in this patient population include psychosis or positive symptoms (ie, hallucinations and delusions), negative symptoms (such as avolition, amotivation, anhedonia, asociality), and cognitive dysfunction (such as deficits in attention, memory, and executive function).2–5 Unfortunately, the existing antipsychotic medications were developed to primarily address psychosis with no clinically meaningful effects on other symptom domains. A major reason for partial efficacy of antipsychotic medications is due to the lack of an etiobiological understanding of the illness.5 The antipsychotic medications provide only symptomatic relief in positive symptoms rather than a complete remission in most patients. The results are even more disappointing in patients with treatment-refractory schizophrenia (TRS), so much so that only about 30% of patients with TRS respond to clozapine, which is still the gold standard to manage such cases.6 Clearly, there is a need to develop more effective and comprehensive treatments to manage schizophrenia with an ability to address treatment refractoriness as well as other symptom domains besides psychosis.
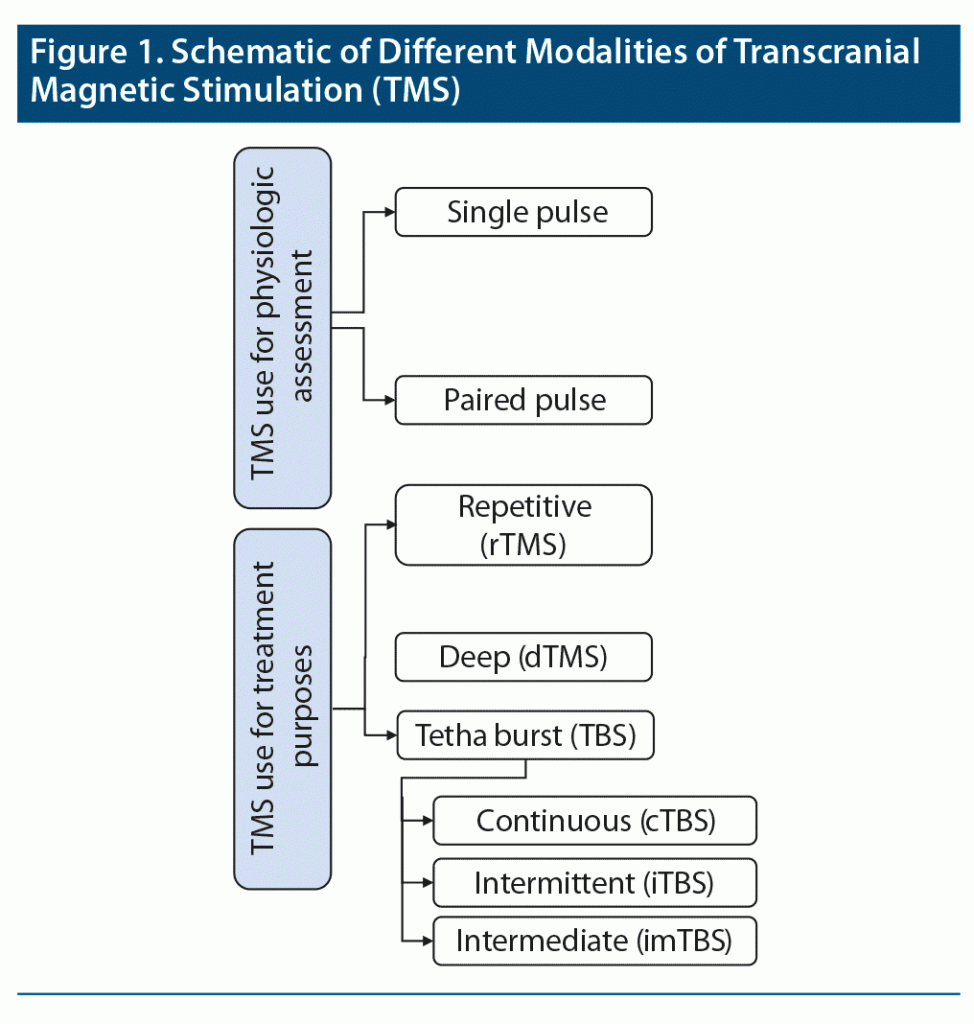
Transcranial magnetic stimulation (TMS) is noninvasive brain neuromodulation by induction of a magnetic field through a magnetic coil. In this procedure, a strong magnetic pulse is delivered to the brain cortex, which in turn creates an inverted electric charge and depolarizes neurons in brain tissue.7 Given its potential as an alternative to psychopharmacologic treatment, TMS has attracted considerable attention from researchers and therapists within the past 2 decades. Different types of repetitive magnetic stimulation have been used to achieve this goal. However, single pulse, paired pulse, and repetitive pulse are the main approaches that have been extensively used and studied previously.8 In a single-pulse stimulation, 1 pulse is delivered with a frequency of less than 1 Hz. In a paired TMS, 2 pulses are delivered to inhibit or excite neurons in either the same or the opposite hemisphere. In a repetitive TMS (rTMS), a series of pulses are delivered either in low frequency (inhibitory, eg, 1 Hz) or high frequency (excitatory, eg, 10 Hz). A newer form of rTMS, express TMS or theta burst TMS (TBS), delivers 3 bursts of high-frequency stimulation (50 Hz) at a burst interval of 200 ms (ie, 5 Hz). TBS appears to have greater effect in a shorter period and is offered in 3 patterns: continuous (cTBS), intermittent (iTBS), and intermediate (imTBS).9 In the cTBS paradigm, uninterrupted trains of TBS signals are delivered for a period of 40 seconds (total of 600 pulses). In iTBS, on the other hand, a total of 600 pulses of TBS stimulation (each pulse is 2 seconds long) are delivered with 8-second intervals (ie, pulses are delivered every 10 seconds, total of 190 seconds). In the imTBS paradigm, a 5-second TBS signal is repeated every 15 seconds (total 600 pulse, 110 seconds).9
Huang et al9 found that an intermittent TBS protocol would result in facilitation, whereas a continuous TBS protocol would result in an inhibitory effect on motor-evoked potentials. An alternative to TMS and TBS is deep TMS (eg, Brainsway), in which pulses reach deeper into the brain (H-coil, 6 cm), and it is often used to treat drug-resistant major depressive disorder.10
TMS application in psychiatry has been reviewed previously; however, much of the research focused on 1 type of TMS or 1 outcome measure or symptom.10 The objective of this review was to analyze all current literature related to different TMS modalities in the management of schizophrenia symptoms and to compare main controversies in outcome measures.
METHODS
A comprehensive search in Ovid MEDLINE, APA PsycINFO, and PubMed was conducted to identify all articles (1996 to present) related to transcranial magnetic stimulation and schizophrenia. The search was conducted using the following terms: schizophrenia and single pulse TMS, schizophrenia and paired pulse TMS, schizophrenia and repetitive TMS, schizophrenia and deep TMS, and schizophrenia and TBS. When search results were limited to English and human subjects and unrelated articles were excluded after our initial review, a total of 43 articles remained in the pool. All articles were downloaded and reviewed for further assessment. Review articles and those that recruited only healthy participants were excluded. The remaining 19 articles were reviewed and discussed further.11–29
RESULTS
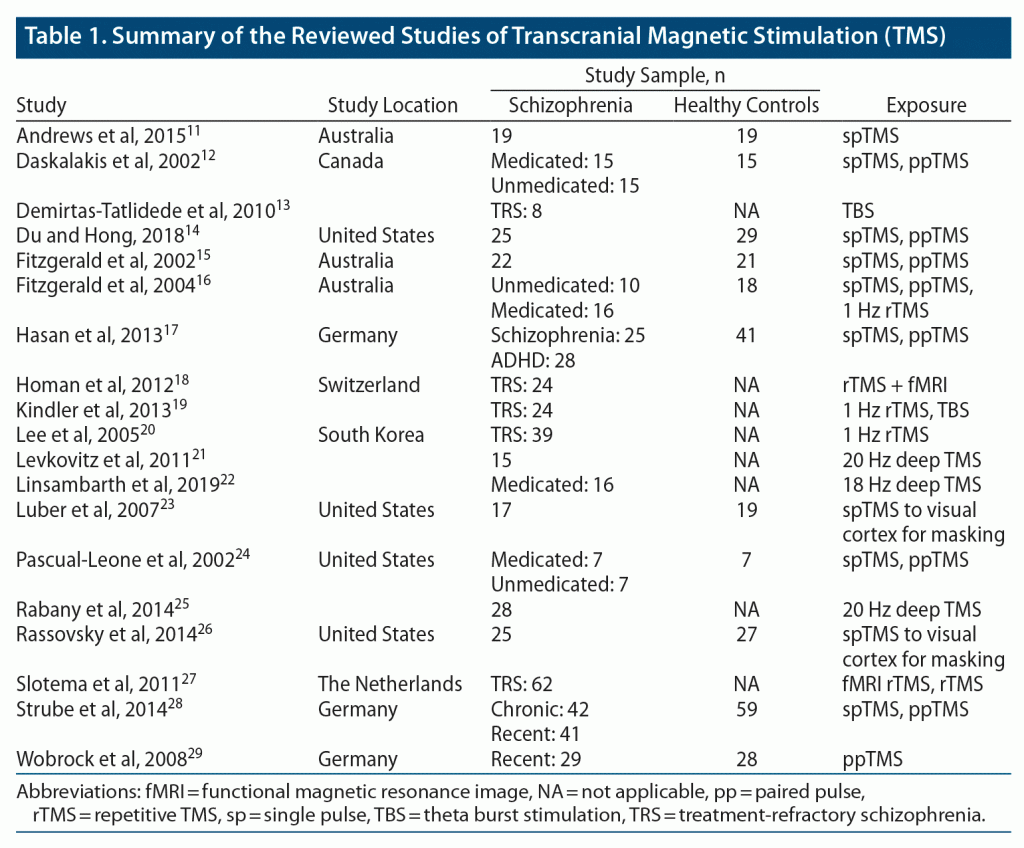
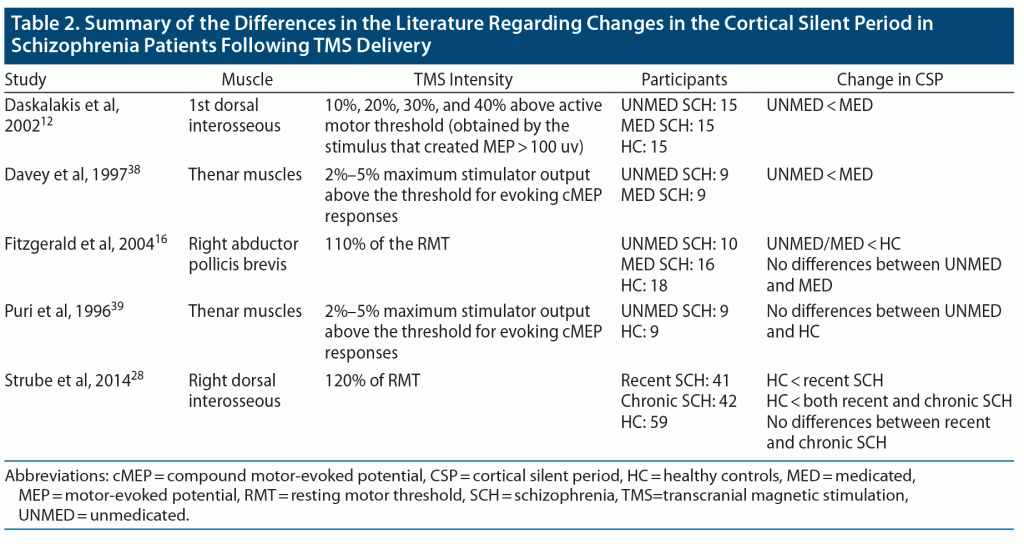
The 19 studies11–29 that fulfilled the eligibility criteria for this review were mostly conducted in North America and Europe. Overall, these studies tested 531 patients with schizophrenia or schizophrenia-like symptoms (including medicated, unmedicated, treatment refractory) and 283 healthy controls (Table 1). These studies administered various TMS modalities, including single-pulse TMS, paired-pulse TMS, low-frequency rTMS, high-frequency deep TMS, and TBS (Table 2).
Use of TMS for Physiologic Assessment
The included studies highlighted the use of different modalities of TMS for both physiologic assessment and treatment purposes. In this context, single- and paired-pulse TMS were often used to identify and compare motor-evoked potential, silence periods following motor-evoked potential, short-interval intercortical inhibition (SICI), and intercortical facilitation. On the other hand, other modalities of TMS were used for treatment and comparison of treatment effectiveness. Several studies used single-pulse TMS to document characteristics of schizophrenia patients.11,23,26 The study by Andrews et al11 explored the potential dysfunctionality of mirror neurons in schizophrenia patients and reported similar suppression of μ and motor resonance as they observed in the control group, suggesting the intactness of the function of mirror neurons in the stable schizophrenia outpatients.
It is well known that patients with schizophrenia demonstrate visual backward masking impairment.30,31 On the other hand, previous studies have shown that transcranial magnetic stimulation affects visual processing in the primary visual cortex.32,33 Luber et al23 and Rassovsky et al26 both utilized single-pulse TMS on the visual cortex to study the effect of forward and backward masking. Luber et al23 speculated that if the visual backward masking impairment in schizophrenia is localized to the primary visual cortex, then TMS neural stimulation may simulate similar effects as visual backward masking for schizophrenia patients, and target recognition will be further challenged in these patients when compared to healthy controls. Luber et al23 indicated that recovery from occipital TMS application between schizophrenia patients and healthy controls is not significantly different. Therefore, it was concluded that schizophrenia patients have no deficit in first visual processing in the occipital cortex (V1).23
Additionally, deficits of social cognition, which lead to social dysfunction and inability to perceive emotions, is a well-known characteristic of individuals with schizophrenia. With the use of single-pulse TMS, Rassovsky et al26 studied facial emotion perception in this patient group. Four basic facial emotions (happy, sad, angry, and afraid) were displayed by photographs to healthy controls and schizophrenia patients. To manipulate temporal processing by visual suppression, single-pulse TMS was delivered to the visual cortex either before or after photograph presentation (forward and backward masking). The study26 found that, at the level of the primary visual cortex, both control and patient participants were affected similarly by visual suppression via single-pulse TMS. The study26 suggested that deficits in facial emotion identification in patients with schizophrenia occur at later integrative processing stages (posterior primary visual cortex).
Several studies12,14–17,24,28,29 utilized single- and paired-pulse TMS to study characteristics of schizophrenia patients or schizophrenia-like symptoms. Du and Hong14 conducted a methodological study to determine the test-retest reliability of SICI as evoked by paired-pulse TMS. Results showed acceptable reliability for both case and control (> 0.6 intraclass correlation coefficient) for SICI elicited by 1- and 3-ms interstimulus intervals. In contrast to healthy controls, who showed a poor intraclass correlation coefficient (ICC), schizophrenia patients showed acceptable ICC-based reliability of intracortical facilitation at 9- and 15-ms interstimulus intervals.
Other studies12,15,17,24,28,29 looked at changes in cortical inhibition in schizophrenia patients. Daskalakis et al12 concluded that schizophrenia patients show a deficit of cortical inhibition, and antipsychotic medication may increase cortical inhibition. Fitzgerald et al15 found that both cortical inhibition and silent period length is reduced in schizophrenia patients, while Strube et al28 reported a reduced SICI and longer cortical silent period in schizophrenia patients compared with healthy controls. However, Strube et al28 also reported a longer cortical silent period in recent-onset patients compared to healthy controls. Similarly, Wobrock et al29 also reported a reduced SICI in first-episode schizophrenia patients. In another study, Hasan et al17 compared patterns of cortical disinhibition and abnormal hemispheric balance of intracortical excitability networks among medicated schizophrenia patients, wherein patients showed a prolonged contralateral silent period and cortical disinhibition as well as a trend toward a reduced SICI compared to healthy controls. Additionally, Pascual-Leone et al24 showed that medicated patients present higher motor thresholds (~5%) in both hemispheres and significantly decreased intracortical inhibition compared to unmedicated patients and control subjects.
Use of TMS for Treatment Purposes
Other modalities of TMS (ie, deep TMS,21,22,25 rTMS,16,18–20,27 and TBS13,19) were utilized to explore the effect of magnetic stimulation on the schizophrenic-like symptoms. Among those studies that explored deep TMS, Levkovitz et al21 and Rabany et al25 studied the safety and effectiveness of deep TMS in conjunction with antipsychotic medication for the treatment of negative symptoms and cognitive deficits in schizophrenia. Both studies applied deep TMS daily for 20 days and concluded that H1 coil deep TMS (20 Hz) as an additional treatment to antipsychotic medication improves both negative symptoms and cognitive deficits of schizophrenia. In a more recent study, Linsambarth et al22 attempted to clarify whether the improvement in negative symptoms in schizophrenia achieved through deep TMS is specific to negative symptoms or attributable to antidepressant effects of transcranial stimulation. The authors22 administered H2 coil deep TMS (18 Hz) on bilateral prefrontal regions of medicated schizophrenia patients and concluded that improvement of negative symptoms might partially result from prefrontal cortex–related functions contributing to negative symptoms in schizophrenia.
Fitzgerald et al16 studied potential abnormal responses to rTMS in patients with schizophrenia and its relation to cortical inhibition deficits. Low-frequency (1 Hz) rTMS was induced. Although participants showed similar motor cortical excitability at baseline, rTMS did not reduce cortical excitability or increase the cortical silent period in the patient population, regardless of whether they were medicated or not, while it did decrease in healthy controls. Others utilized low-frequency (1 Hz) rTMS to explore its effect on auditory hallucination in schizophrenia patients. Homan et al18 concluded that only patients with higher superior temporal gyrus perfusion benefited from rTMS as an additional therapy to their ongoing treatment. In a double-blind randomized controlled study, Lee et al20 reported that a 10-day daily low-frequency (1 Hz) rTMS over the left or right temporoparietal cortex will significantly reduce auditory hallucination in schizophrenia patients. In contrast, in a large randomized controlled trial, Slotema et al27 reported no significant changes in the severity of general psychotic symptoms (eg, auditory hallucination) between the 3 groups of functional magnetic resonance image–guided (to the site of maximum hallucinatory activation) low-frequency rTMS, left temporoparietal rTMS, and sham rTMS.
Two studies in our review utilized TBS.13,19 In a single-blind randomized controlled study, Kindler et al19 compared the effectiveness of continuous TBS and 10-day, low-frequency (1 Hz) rTMS in the treatment of auditory verbal hallucination and reported a similar outcome. Demirtas-Tatlidede et al13 induced intermittent magnetic resonance imaging–guided TBS to schizophrenia patients. While iTBS created insignificant side effects (ie, neck pain, headache, a mild drop of diastolic blood pressure for 5 minutes following TBS), it was effective to improve cognition, reduce negative symptoms, and elevate mood.
DISCUSSION
Transcranial magnetic stimulation is a noninvasive method that has been used for both therapeutic and physiologic assessment purposes.34,35 While some adverse effects of TMS are reported in the literature,36,37 the process is generally considered safe.21 TMS has been found effective in the evaluation and treatment of several psychiatric disorders including schizophrenia.37 Figure 1 provides a schematic of common TMS modalities.
Use of TMS for Physiologic Assessment
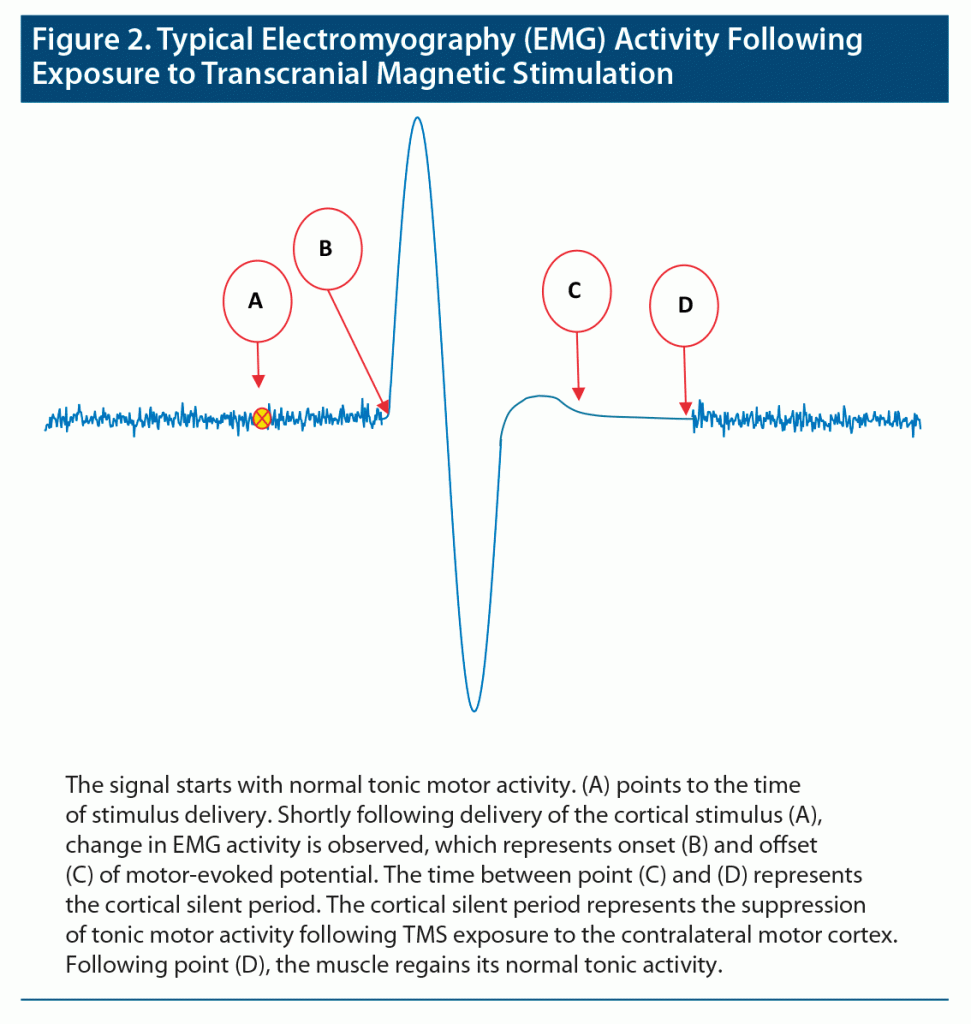
Figure 2 depicts a typical change in electromyography (EMG) signal following delivery of TMS. The cortical silent period is reflected on EMG activity of muscles (typically dorsal interosseous muscles) as is referred to the suppression of tonic motor activity following TMS exposure to the contralateral motor cortex. While changes in the cortical silent period among schizophrenia patients have attracted the attention of many researchers, the alteration is still controversial. Several studies compared the cortical silent period of medicated and unmedicated patients with schizophrenia with that of healthy controls. Daskalakis et al12 reported that the cortical silent period is shorter for unmedicated schizophrenia patients compared to medicated patients. This finding is in agreement with that of Davey et al38 and in contrast with that of Puri et al39 and Fitzgerald et al,16 who reported no difference in the cortical silent period between medicated and unmedicated patients. While Fitzgerald et al16 reported a shorter cortical silent period for schizophrenia patients, both Strube et al28 and Hasan et al17 reported that patients with schizophrenia have a significantly longer cortical silent period compared with healthy participants. We anticipate that the discrepancies in the literature on the cortical silent period are due to the variations in study protocols and TMS intensities. Table 2 summarizes the difference found in the literature regarding changes in the cortical silent period in schizophrenia patients following TMS delivery.
Use of TMS for Treatment Purposes
Auditory hallucination is a common characteristic of several psychiatric disorders, including schizophrenia, in which patients experience a false perception of sound. Among the studies that we reviewed, several focused on the effect of TMS administration on auditory hallucination.18–20,27 Homan et al18 reported that auditory hallucination in medication-refractory schizophrenia patients with higher superior temporal gyrus perfusion is reduced by 50% or more following 10 days of rTMS therapy. Similarly, Lee et al20 reported that 10 days of daily rTMS at 1 Hz benefits treatment-resistant patients with schizophrenia in reduction of auditory hallucinations. In contrast, however, Kindler et al19 reported that exposure of patients with schizophrenia or schizoaffective disorder to either cTBS or 1 Hz TMS similarly improves auditory verbal hallucinations. Additionally, Slotema et al27 reported no significant changes in the severity of auditory verbal hallucination between the 3 groups of fMRI-guided low-frequency rTMS, left temporoparietal rTMS, and sham rTMS.
While the underlying mechanism of different types of TMS delivery is similar, there are different US Food and Drug Administration–approved variations. Deep TMS is a variation wherein an H-coil is utilized to create a synergistic magnetic field that reaches deeper structures in the brain and requires a shorter length of treatment. Although several studies21,40,41 have reported good efficacy of deep TMS, Lonergan et al42 delineated a lower efficacy and higher rate of side effects for deep TMS compared with surface TMS machines. However, in our review all studies21,22,25 that utilized deep TMS reported that deep TMS as an additional treatment to antipsychotic medication improves both negative symptoms and cognitive deficits of schizophrenia.
CONCLUSION
Our review compared studies with a focus on the use of different modalities of TMS and schizophrenia. The main controversies that we found in our review were related to silent periods following exposure to TMS and auditory hallucination in patients with schizophrenia that are compared and discussed.
Overall, it appears that use of TMS for physiologic assessment or treatment of schizophrenia is beneficial. To establish a robust conclusion and resolve controversies however, further studies are needed. In summary, from the clinical perspective, it is generally believed that deep TMS stimulates a deeper and larger area and therefore takes less time to achieve the same results compared with rTMS. Additionally, TBS appears to achieve results much faster than deep TMS.
In regard to treatment of schizophrenia symptoms, although many controversies exist in use and benefits of rTMS and deep TMS, it appears that use of low-frequency rTMS (1 Hz) and deep TMS benefits patients with schizophrenia by reducing auditory hallucination. Deep TMS (20 Hz) appears to improve both negative symptoms and cognitive deficits of schizophrenia when added as an additional treatment to antipsychotic medication. Furthermore, use of cTBS improves hallucination, which is reported to create similar outcomes as low-frequency (1 Hz) rTMS. iTBS, on the other hand, is used to improve negative symptoms, cognition, and mood in schizophrenia.
Submitted: September 15, 2021; accepted February 4, 2022.
Published online: August 25, 2022.
Relevant financial relationships: None.
Funding/support: None.
Previous presentations: Partial content presented in poster format at the virtual 21st World Psychiatric Association World Congress of Psychiatry; October 18–21, 2021 and the 4th International Brain Stimulation Conference; December 6–9, 2021; Charleston, South Carolina.
Clinical Points
- TMS treatment seems promising to address symptoms of schizophrenia, particularly in treatment-resistant patients.
- Intermittent theta burst TMS and deep TMS potentially provide benefit in the treatment of negative symptoms of schizophrenia.
References (42)

- Kahn RS, Sommer IE, Murray RM, et al. Schizophrenia. Nat Rev Dis Primers. 2015;1(1):15067. PubMed CrossRef
- Sellgren CM, Gracias J, Watmuff B, et al. Increased synapse elimination by microglia in schizophrenia patient-derived models of synaptic pruning. Nat Neurosci. 2019;22(3):374–385. PubMed CrossRef
- Wang M, Zhang L, Gage FH. Microglia, complement and schizophrenia. Nat Neurosci. 2019;22(3):333–334. PubMed CrossRef
- Yang Q, Wang Y, Zhang S, et al. Biomarker discovery for immunotherapy of pituitary adenomas: enhanced robustness and prediction ability by modern computational tools. Int J Mol Sci. 2019;20(1):151. PubMed CrossRef
- Walker E, Kestler L, Bollini A, et al. Schizophrenia: etiology and course. Annu Rev Psychol. 2004;55(1):401–430. PubMed CrossRef
- Elkis H. Treatment-resistant schizophrenia. Psychiatr Clin North Am. 2007;30(3):511–533. PubMed CrossRef
- Hallett M. Transcranial magnetic stimulation and the human brain. Nature. 2000;406(6792):147–150. PubMed CrossRef
- Galletta EE, Rao PR, Barrett AM. Transcranial magnetic stimulation (TMS): potential progress for language improvement in aphasia. Top Stroke Rehabil. 2011;18(2):87–91. PubMed CrossRef
- Huang YZ, Edwards MJ, Rounis E, et al. Theta burst stimulation of the human motor cortex. Neuron. 2005;45(2):201–206. PubMed CrossRef
- Bersani FS, Minichino A, Enticott PG, et al. Deep transcranial magnetic stimulation as a treatment for psychiatric disorders: a comprehensive review. Eur Psychiatry. 2013;28(1):30–39. PubMed CrossRef
- Andrews SC, Enticott PG, Hoy KE, et al. No evidence for mirror system dysfunction in schizophrenia from a multimodal TMS/EEG study. Psychiatry Res. 2015;228(3):431–440. PubMed CrossRef
- Daskalakis ZJ, Christensen BK, Chen R, et al. Evidence for impaired cortical inhibition in schizophrenia using transcranial magnetic stimulation. Arch Gen Psychiatry. 2002;59(4):347–354. PubMed CrossRef
- Demirtas-Tatlidede A, Freitas C, Cromer JR, et al. Safety and proof of principle study of cerebellar vermal theta burst stimulation in refractory schizophrenia. Schizophr Res. 2010;124(1-3):91–100. PubMed CrossRef
- Du X, Hong LE. Test-retest reliability of short-interval intracortical inhibition and intracortical facilitation in patients with schizophrenia. Psychiatry Res. 2018;267:575–581. PubMed CrossRef
- Fitzgerald PB, Brown TL, Daskalakis ZJ, et al. A transcranial magnetic stimulation study of inhibitory deficits in the motor cortex in patients with schizophrenia. Psychiatry Res. 2002;114(1):11–22. PubMed CrossRef
- Fitzgerald PB, Brown TL, Marston NA, et al. Reduced plastic brain responses in schizophrenia: a transcranial magnetic stimulation study. Schizophr Res. 2004;71(1):17–26. PubMed CrossRef
- Hasan A, Schneider M, Schneider-Axmann T, et al. A similar but distinctive pattern of impaired cortical excitability in first-episode schizophrenia and ADHD. Neuropsychobiology. 2013;67(2):74–83. PubMed CrossRef
- Homan P, Kindler J, Hauf M, et al. Cerebral blood flow identifies responders to transcranial magnetic stimulation in auditory verbal hallucinations. Transl Psychiatry. 2012;2(11):e189. PubMed CrossRef
- Kindler J, Homan P, Flury R, et al. Theta burst transcranial magnetic stimulation for the treatment of auditory verbal hallucinations: results of a randomized controlled study. Psychiatry Res. 2013;209(1):114–117. PubMed CrossRef
- Lee SH, Kim W, Chung YC, et al. A double-blind study showing that two weeks of daily repetitive TMS over the left or right temporoparietal cortex reduces symptoms in patients with schizophrenia who are having treatment-refractory auditory hallucinations. Neurosci Lett. 2005;376(3):177–181. PubMed CrossRef
- Levkovitz Y, Rabany L, Harel EV, et al. Deep transcranial magnetic stimulation add-on for treatment of negative symptoms and cognitive deficits of schizophrenia: a feasibility study. Int J Neuropsychopharmacol. 2011;14(7):991–996. PubMed CrossRef
- Linsambarth S, Jeria A, Avirame K, et al. Deep transcranial magnetic stimulation for the treatment of negative symptoms in schizophrenia: beyond an antidepressant effect. J ECT. 2019;35(4):e46–e54. PubMed CrossRef
- Luber B, Stanford AD, Malaspina D, et al. Revisiting the backward masking deficit in schizophrenia: individual differences in performance and modeling with transcranial magnetic stimulation. Biol Psychiatry. 2007;62(7):793–799. PubMed CrossRef
- Pascual-Leone A, Manoach DS, Birnbaum R, et al. Motor cortical excitability in schizophrenia. Biol Psychiatry. 2002;52(1):24–31. PubMed CrossRef
- Rabany L, Deutsch L, Levkovitz Y. Double-blind, randomized sham controlled study of deep-TMS add-on treatment for negative symptoms and cognitive deficits in schizophrenia. J Psychopharmacol. 2014;28(7):686–690. PubMed CrossRef
- Rassovsky Y, Lee J, Nori P, et al. Exploring facial emotion perception in schizophrenia using transcranial magnetic stimulation and spatial filtering. J Psychiatr Res. 2014;58:102–108. PubMed CrossRef
- Slotema CW, Blom JD, de Weijer AD, et al. Can low-frequency repetitive transcranial magnetic stimulation really relieve medication-resistant auditory verbal hallucinations? negative results from a large randomized controlled trial. Biol Psychiatry. 2011;69(5):450–456. PubMed CrossRef
- Strube W, Wobrock T, Bunse T, et al. Impairments in motor-cortical inhibitory networks across recent-onset and chronic schizophrenia: a cross-sectional TMS Study. Behav Brain Res. 2014;264:17–25. PubMed CrossRef
- Wobrock T, Schneider M, Kadovic D, et al. Reduced cortical inhibition in first-episode schizophrenia. Schizophr Res. 2008;105(1-3):252–261. PubMed CrossRef
- Rund BR, Egeland J, Sundet K, et al. Early visual information processing in schizophrenia compared to recurrent depression. Schizophr Res. 2004;68(2-3):111–118. PubMed CrossRef
- Schechter I, Butler PD, Silipo G, et al. Magnocellular and parvocellular contributions to backward masking dysfunction in schizophrenia. Schizophr Res. 2003;64(2-3):91–101. PubMed CrossRef
- Corthout E, Uttl B, Ziemann U, et al. Two periods of processing in the (circum)striate visual cortex as revealed by transcranial magnetic stimulation. Neuropsychologia. 1999;37(2):137–145. PubMed CrossRef
- Miller MB, Fendrich R, Eliassen JC, et al. Transcranial magnetic stimulation: delays in visual suppression due to luminance changes. Neuroreport. 1996;7(11):1740–1744. PubMed CrossRef
- Groppa S, Oliviero A, Eisen A, et al. A practical guide to diagnostic transcranial magnetic stimulation: report of an IFCN committee. Clin Neurophysiol. 2012;123(5):858–882. PubMed CrossRef
- Perera T, George MS, Grammer G, et al. The Clinical TMS Society Consensus Review and treatment recommendations for TMS therapy for major depressive disorder. Brain Stimul. 2016;9(3):336–346. PubMed CrossRef
- Loo CK, McFarquhar TF, Mitchell PB. A review of the safety of repetitive transcranial magnetic stimulation as a clinical treatment for depression. Int J Neuropsychopharmacol. 2008;11(1):131–147. PubMed CrossRef
- Rossi S, Hallett M, Rossini PM, et al; Safety of TMS Consensus Group. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol. 2009;120(12):2008–2039. PubMed CrossRef
- Davey NJ, Puri BK, Lewis HS, et al. Effects of antipsychotic medication on electromyographic responses to transcranial magnetic stimulation of the motor cortex in schizophrenia. J Neurol Neurosurg Psychiatry. 1997;63(4):468–473. PubMed CrossRef
- Puri BK, Davey NJ, Ellaway PH, et al. An investigation of motor function in schizophrenia using transcranial magnetic stimulation of the motor cortex. Br J Psychiatry. 1996;169(6):690–695. PubMed CrossRef
- Carmi L, Tendler A, Bystritsky A, et al. Efficacy and safety of deep transcranial magnetic stimulation for obsessive-compulsive disorder: a prospective multicenter randomized double-blind placebo-controlled trial. Am J Psychiatry. 2019;176(11):931–938. PubMed CrossRef
- Levkovitz Y, Isserles M, Padberg F, et al. Efficacy and safety of deep transcranial magnetic stimulation for major depression: a prospective multicenter randomized controlled trial. World Psychiatry. 2015;14(1):64–73. PubMed CrossRef
- Lonergan B, Nguyen E, Lembo C, et al. Patient- and technician-oriented attitudes toward transcranial magnetic stimulation devices. J Neuropsychiatry Clin Neurosci. 2018;30(3):242–245. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!