Objective: Individuals with binge-eating disorder (BED) experience psychiatric and somatic comorbidities and obesity, but the nature and magnitude of prescription medication utilization is unclear. We investigated utilization using Swedish registry data and a case-control design.
Methods: Cases were identified from Riksät and Stepwise longitudinal registers and were individuals diagnosed with BED per DSM-IV-TR criteria between July 1, 2006, and December 31, 2009, at eating disorder clinics (n = 238, 96% female, mean age = 22.8 years). For each case, 10 controls were matched on sex and year, month, and county of birth (n = 2,380). An index date was derived for each control, which was the date of diagnosis of BED in the corresponding case. The association between BED and prescription medication utilization was investigated before and within 12 months after diagnosis.
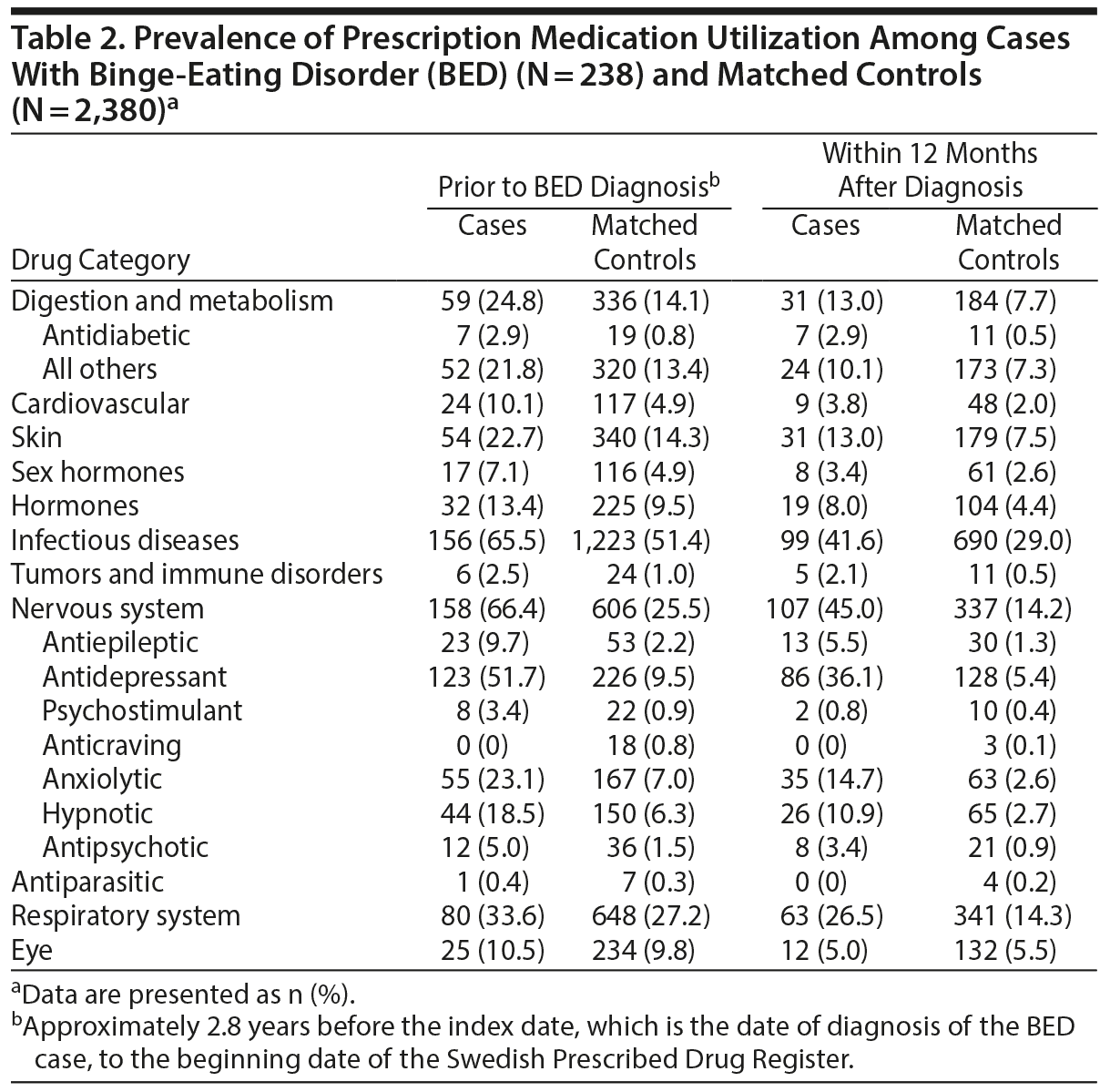
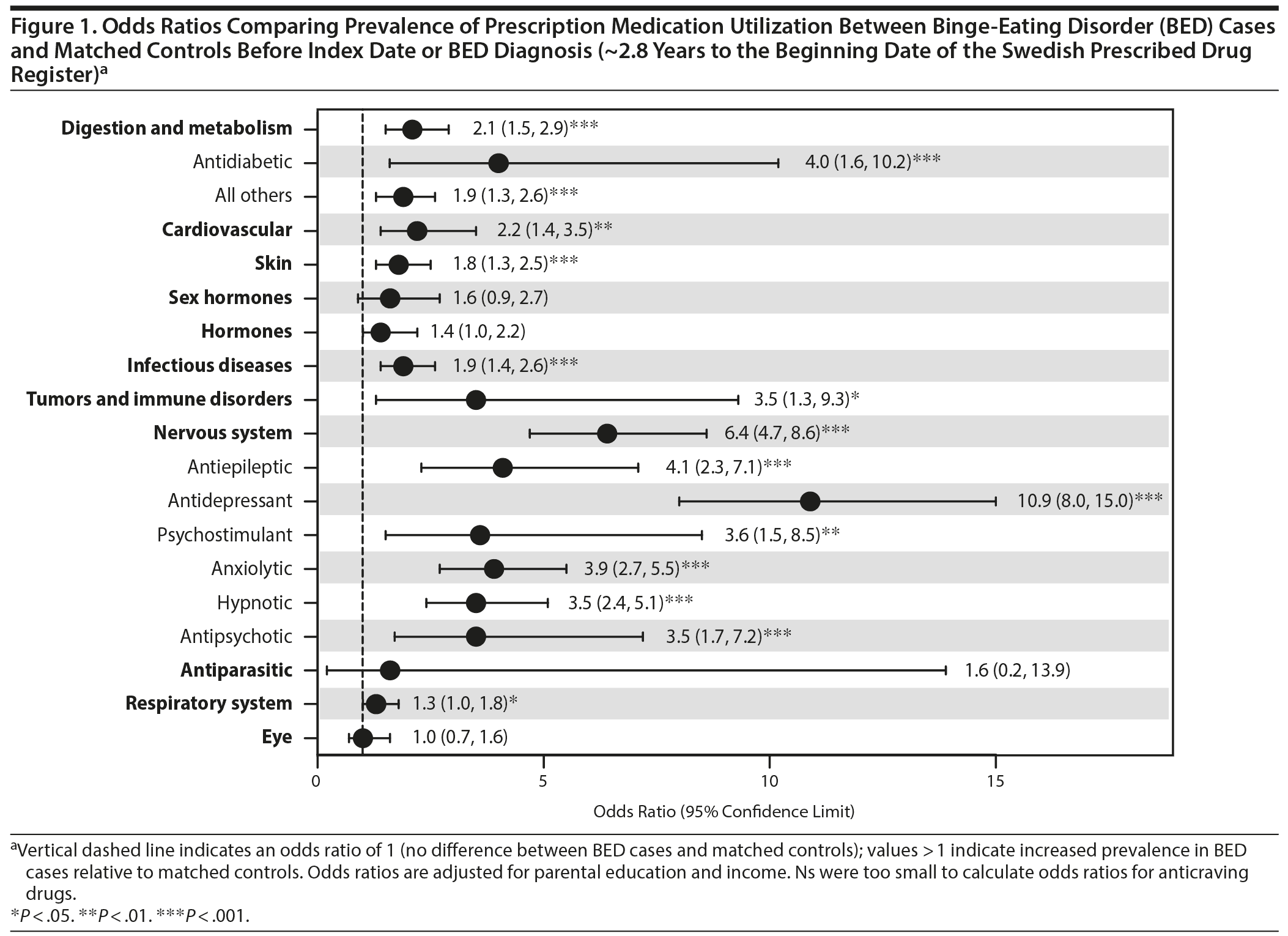
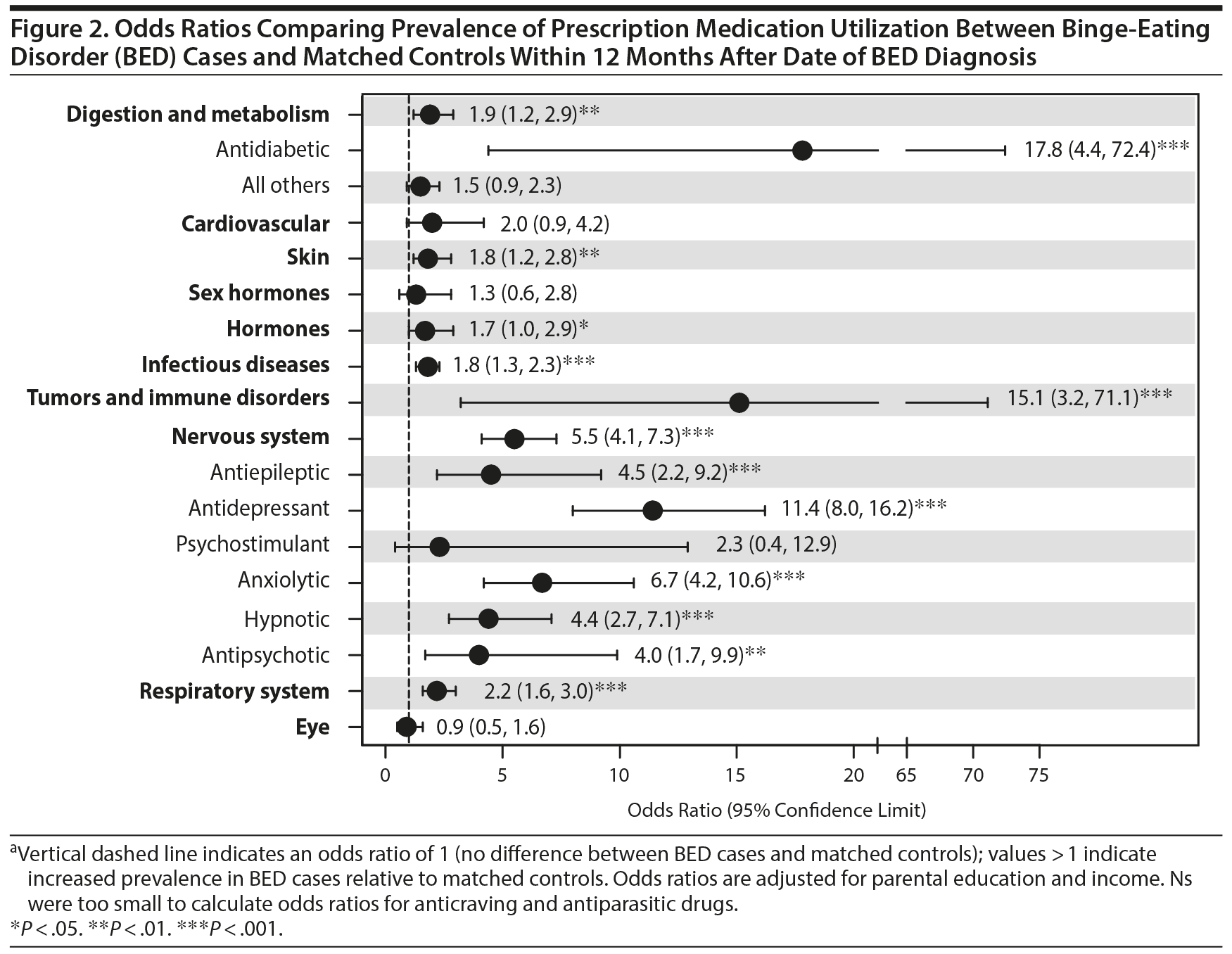
Results: Before diagnosis, cases were significantly more likely than matched controls to have been prescribed nervous system (odds ratio = 6.4; 95% confidence limit = 4.7, 8.6), tumors and immune disorders (3.5; 1.3, 9.3), cardiovascular (2.2; 1.4, 3.5), digestion and metabolism (2.1; 1.5, 2.9), infectious diseases (1.9; 1.4, 2.6), skin (1.8; 1.3, 2.5), and respiratory system (1.3; 1.0, 1.8) medications. Cases also had higher odds of prescription use than controls across most categories within 12 months after diagnosis. Several associations were significant after accounting for lifetime psychiatric comorbidity and obesity.
Conclusions: Individuals with BED had increased utilization of psychiatric and nonpsychiatric medications compared with matched controls. Findings confirm that the illness burden of BED extends to high medication utilization and underscore the importance of thorough medication reviews when treating individuals with BED.
A Register-Based Case-Control Study of
Prescription Medication Utilization in Binge-Eating Disorder
ABSTRACT
Objective: Individuals with binge-eating disorder (BED) experience psychiatric and somatic comorbidities and obesity, but the nature and magnitude of prescription medication utilization is unclear. We investigated utilization using Swedish registry data and a case-control design.
Methods: Cases were identified from Riksät and Stepwise longitudinal registers and were individuals diagnosed with BED per DSM-IV-TR criteria between July 1, 2006, and December 31, 2009, at eating disorder clinics (n = 238, 96% female, mean age = 22.8 years). For each case, 10 controls were matched on sex and year, month, and county of birth (n = 2,380). An index date was derived for each control, which was the date of diagnosis of BED in the corresponding case. The association between BED and prescription medication utilization was investigated before and within 12 months after diagnosis.
Results: Before diagnosis, cases were significantly more likely than matched controls to have been prescribed nervous system (odds ratio = 6.4; 95% confidence limit = 4.7, 8.6), tumors and immune disorders (3.5; 1.3, 9.3), cardiovascular (2.2; 1.4, 3.5), digestion and metabolism (2.1; 1.5, 2.9), infectious diseases (1.9; 1.4, 2.6), skin (1.8; 1.3, 2.5), and respiratory system (1.3; 1.0, 1.8) medications. Cases also had higher odds of prescription use than controls across most categories within 12 months after diagnosis. Several associations were significant after accounting for lifetime psychiatric comorbidity and obesity.
Conclusions: Individuals with BED had increased utilization of psychiatric and nonpsychiatric medications compared with matched controls. Findings confirm that the illness burden of BED extends to high medication utilization and underscore the importance of thorough medication reviews when treating individuals with BED.
Prim Care Companion CNS Disord 2016;18(4):doi:10.4088/PCC.16m01939
© Copyright 2016 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, University of North Carolina at Chapel Hill
bSchool of Psychology and Speech Pathology, Curtin University, Perth, Western Australia
cSchool of Paediatrics and Child Health, The University of Western Australia, Perth
dDepartment of Medical Epidemiology and Biostatistics, Karolinska Institutet, Stockholm, Sweden
eStockholm Centre for Eating Disorders, Sweden
fDepartment of Clinical Neuroscience, Karolinska Institutet, Stockholm, Sweden
gShire, Lexington, Massachusetts
hDepartment of Nutrition, University of North Carolina at Chapel Hill
*Corresponding author: Hunna J. Watson, PhD, Department of Psychiatry, University of North Carolina at Chapel Hill, CB #7160, 101 Manning Dr, Chapel Hill, NC 27599-7160 ([email protected]).
Binge-eating disorder (BED), characterized by recurrent binge eating without regular compensatory behaviors, was only recently codified as a psychiatric disorder in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). BED affects 2%–3.5% of individuals over the lifetime and is associated with substantial psychiatric comorbidity, obesity, and disease burden.1,2 To further study medical and psychiatric morbidity in BED, we investigated prescription medication utilization before and after BED diagnosis using Swedish national register data.
BED is associated with depression, anxiety disorders, substance use disorders, and obesity.1,2 Evidence suggests elevated rates of hypertension,2–4 type 2 diabetes,2,5–7 autoimmune diseases,7 gastrointestinal disorders,2,8–11 and respiratory problems4,11,12 in patients with BED. Knowledge about prescription medication utilization may extend insight into the spectrum of morbidity and aid in gauging the medical and therapeutic needs of this population. Also of interest is the extent to which psychiatric comorbidities and obesity may account for any observed elevations in utilization. Some studies13,14 on clinical samples have reported the prevalence of prescription medication use; however, they are limited because they only report on antidepressants, rely on particular collection methodologies (ie, self-report), lack controls, and use small, nongeneralizable samples.
Given the dearth of information on a wide range of prescription medications from population-based samples, we linked Swedish national prescription drug and eating disorder register records to characterize medication utilization in individuals with BED. Our focus was intentionally broad and explored all prescribed medications as an index of both health and utilization. We hypothesized increased utilization in BED across both psychiatric (ie, psychotropic) and somatic drug categories before and in the 12 months following diagnosis of BED. This hypothesis was derived from data on psychiatric and obesity comorbidity,1,2 somatic comorbidity,2,3,5–10,12 and delayed help-seeking in eating disorders.15 A secondary objective was to explore the role of psychiatric comorbidities and obesity in any observed association between BED and medication utilization. Results have the potential to provide critical insight for health care beyond what has been reported in prior clinical studies.
METHODS
Study Population
The study sample was derived from a total population cohort of individuals born in Sweden between 1979 and 1993. Using the unique national personal identification number, we linked data in Sweden’s Total Population Register, Riksät16 and Stepwise17 eating disorder quality registers, the Swedish Prescribed Drug Register, and other population-based registers. Inclusion in the Swedish population registers does not require informed consent. For Stepwise, research participation is elective via an opt-out procedure (~3% decline participation18). The University of North Carolina Biomedical Institutional Review Board and the Regional Ethics Committee of Karolinska Institutet approved this study.
Cases were identified from Riksät and Stepwise longitudinal registers and met DSM-IV-TR19 criteria for BED between July 1, 2006, and December 31, 2009 (in 1994, BED was introduced into the DSM-IV as a provisional disorder). The date of BED diagnosis is subsequently referred to as the index date. These registers provide quality assurance for over 35 specialist treatment clinics and capture nearly all individuals receiving inpatient, day patient, or outpatient specialized eating disorder treatment in Sweden. The index BED diagnosis reflected the first diagnosis of BED recorded and could occur at the initial clinic presentation or at an annual follow-up evaluation following initial presentation for another eating disorder. Follow-up assessments occur annually for the duration of the individual’s engagement in treatment and are available for ~69% of registrants. To be included in the registers, the following criteria must have been met: (1) medical or self-referral to a participating treatment program, (2) diagnosed with an eating disorder by a medical provider, and (3) intent to treat in the program.
We ascertained 10 controls for each case using the Multi-Generation Register.20 Controls were matched to cases on sex and year, month, and county of birth. If a case was born outside of Sweden, controls were additionally matched on immigration status and time of migration (controls could not immigrate later than their respective cases), regardless of origin. Controls had to be alive and a resident in Sweden for an equivalent time period: from birth or immigration until the end of study follow-up of their index case. Controls were required not to have received a BED diagnosis in Riksät or Stepwise, but they could have had another eating disorder (which was detected in 1.0% of controls) recorded in Riksät, Stepwise, or the National Patient Register (NPR) (includes all inpatient care since 1987 and hospital-based outpatient visits since 2001).
Measures
Medication prescriptions. Medication prescriptions were obtained from the Swedish Prescribed Drug Register, which contains complete data (> 99%) for all medications prescribed and dispensed to the entire Swedish population since July 1, 2005.21 Prescription records included all those available for each participant from registry inception to up to 12 months after the index date. Medications are classified in the register according to the Anatomic Therapeutic Chemical (ATC) classification system, which classifies drugs based on the anatomic system on which they act and their therapeutic, pharmacologic, or chemical properties.
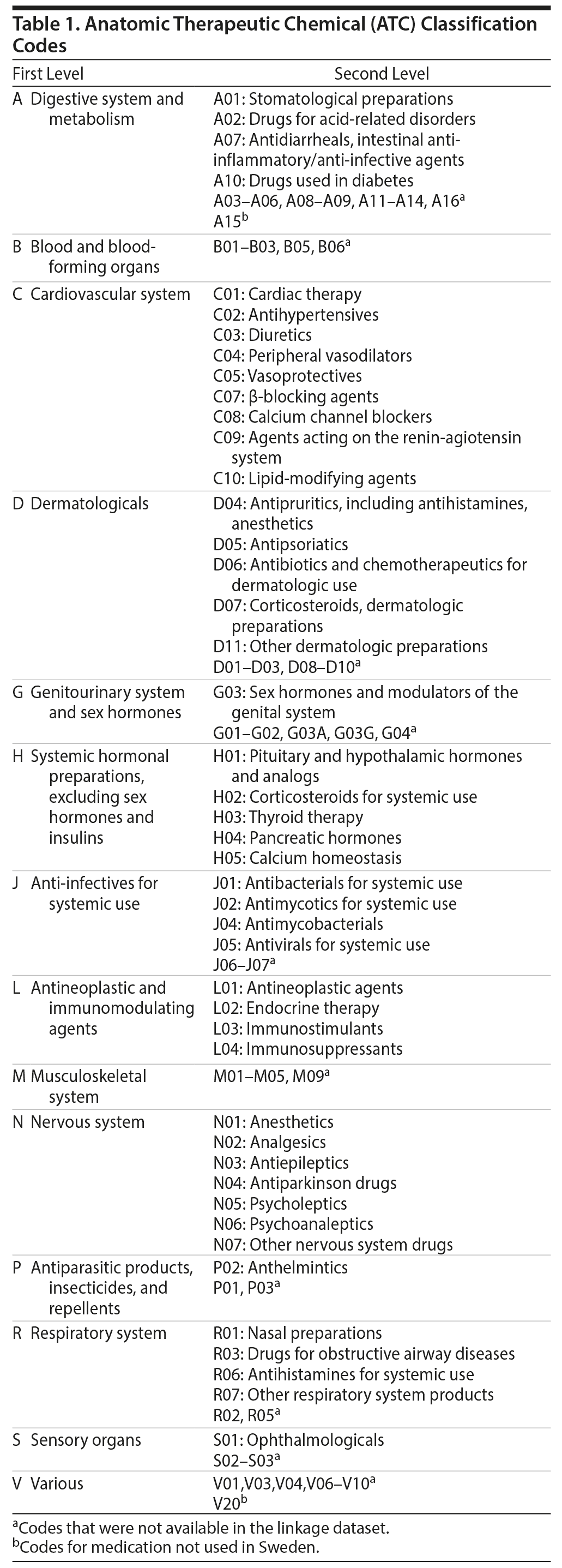
The first level of the code identifies the anatomic main group and consists of 1 letter. The second level of the code identifies the therapeutic main group and consists of 2 digits, and the third level identifies the therapeutic and pharmacologic subgroup and consists of 1 letter. There are 5 levels altogether. We included overarching categories (digestion and metabolism [ATC code A01, A02, A07, A10], cardiovascular [ATC code C], skin [ATC code D04, D05, D06, D07, D11], sex hormones [ATC code G03], hormones [ATC code H], infectious diseases [ATC code J01, J02, J04, J05], tumors and immune disorders [ATC code L], nervous system [ATC code N], antiparasitic [ATC code P02], respiratory system [ATC code R01, R03, R06, R07], eye [ATC code S01]) and specific nervous system subcategories to capture more detail on psychiatric prescriptions (antiepileptic [ATC code N03], antipsychotic [ATC code N05A], anxiolytic [ATC code N05B], hypnotic [ATC code N05C], antidepressant [ATC code N06A], psychostimulant [ATC code N06B], anticraving [ATC code N07B]). Some categories were unavailable in the Swedish Prescribed Drug Register (Table 1). We included all ATC codes available to us in the data linkage; unfortunately, this linkage does not cover all classes of medication used in Sweden (see Table 1 for information on codes and availability).
Psychiatric comorbidity. Psychiatric comorbidity was coded as the presence versus absence of any lifetime psychiatric disorder or suicide-related or intentional self-harm injury and was used in the secondary analysis as a covariate. Comorbidity was obtained from the NPR and was based on the World Health Organization (WHO) International Classification of Diseases, Ninth Revision22 (ICD-9: years 1987–1996) or the ICD-1023 (1997–present). The psychiatric disorders included schizophrenia (ICD-9: 295A–295E, 295G, 295W, 295X; ICD-10: F20, F20.0–F20.6, F20.8, F20.9), schizoaffective disorder (295H; F23.1, F23.2, F25, F25.0–F25.2, F25.8, F25.9), bipolar disorder (296A, 296C–296H, 296W, 296X; F30, F30.1, F30.2, F30.8, F30.9, F31, F31.0–F31.9, F34.0), major depressive disorder (MDD) (296B, 300E, 311; F32, F32.0–F32.3, F32.8, F32.9, F33, F33.0–F33.4, F33.8, F33.9, F34.1, F34.8, F34.9, F38, F38.0, F38.1, F38.8, F39), anxiety disorder (minus obsessive-compulsive disorder [OCD] and posttraumatic stress disorder [PTSD] [300A, 300C; F40, F40.0–F40.2, F40.8, F40.9, F41, F41.0–F41.3, F41.8, F41.9]), OCD (300D; F42, F42.0–F42.2, F42.8, F42.9), PTSD (308, 309A, 309B, 309W, 309X; F43, F43.0–F43.2, F43.8, F43.9), attention-deficit/hyperactivity disorder (ADHD) (314J, 314W, 314X; F90, F90.0, F90.1, F90.8), autism (299A; F84.0, F84.1, F84.5), alcohol use disorder (303, 303A, 303X, 305A; F10, F10.0–F10.9), illicit drug use disorder (304, 304A–304H, 304W, 304X, 305X; F11, F11.0–F11.9, F12, F12.0–F12.9, F13, F13.0–F13.9, F14, F14.0–F14.9, F15, F15.0–F15.9, F16, F16.0–F16.9, F18, F18.0–F18.9, F19, F19.0–F19.9), and suicide attempts/intentional self-harm (E95A–E95H, E95W, E95X; X60–X84).
Obesity. Obesity (body mass index [BMI] ≥ 30 kg/m2) for cases only was derived from height and weight in Riksät/Stepwise at the time of diagnosis. As height and weight are not regularly recorded in the patient and population registers, BMI was unavailable for matched controls.
Parental education and income. Education and income were obtained from the Longitudinal Integration Database for Health Insurance and Labour Market Studies (LISA: Longitudinell Ingegrationsdatabas för Sjukförsäkrings- och Arbetsmarknadsstudier) for the year of the index date. Parental education was measured as the highest attained level of either parent (categorized as primary school [≤ 9 years], secondary school, or tertiary education). Income was measured as the individual share of disposable family income, obtained by calculating the sum of family members’ disposable income multiplied by individual consumption weights (0.96 for adults), then divided by the total family consumption weight. This calculation gives net values in Swedish kronor, which were categorized into quartiles for analysis.
Statistical Analysis
Analyses were conducted with R 3.1.1. Given the exploratory nature of the study, we did not perform familywise error rate correction.24 All analyses included parental education and income as covariates and used exact likelihood methods. First, we examined the association between BED status and prescription medication utilization prior to the index date using conditional logistic regression models. Second, we examined these associations in the 12 months after the index date using conditional logistic regression models. Third, we examined the extent to which psychiatric comorbidity explained the association between BED and prescription medication utilization within 12 months after the index date by adding lifetime psychiatric comorbidity as a covariate to the conditional logistic regression models. Finally, we investigated the role of obesity in 3 related analyses: (1) cases with comorbid obesity were compared with matched controls using conditional logistic regression (case OB+/control), (2) cases without comorbid obesity were compared with matched controls using conditional logistic regression (case OB–/control), and (3) cases with comorbid obesity were compared with cases without comorbid obesity using logistic regression (case OB+/case OB–). These comparisons were conducted because BMI data were unavailable for controls and could not be added as a covariate to the models.
RESULTS
A total of 238 cases (96% female) were identified. The mean age at the time of BED diagnosis was 22.8 years (SD = 3.6, range, 14.9–29.7). Approximately 73.5% of BED cases were identified at initial assessment and 26.5% at an annual follow-up (13.9% had bulimia nervosa at initial assessment, 7.1% had anorexia nervosa, 5.5% had eating disorder not otherwise specified). Of cases, 71.8% had a comorbid lifetime psychiatric disorder compared with 12.8% of controls. Specifically, the prevalence estimates for cases versus controls were schizophrenia (0.4% vs 0.1%), schizoaffective disorder (0% vs < 0.01%), bipolar disorder (4.2% vs 0.7%), MDD (23.5% vs 4.2%), anxiety disorder (19.3% vs 3.9%), OCD (1.3% vs 0.5%), PTSD (6.3% vs 2.2%), ADHD (1.3% vs 0.8%), autism (0% vs < 0.01%), alcohol use disorder (1.7% vs 2.1%), illicit drug use disorder (0.4% vs 0.8%), suicide attempts/intentional self-harm (6.7% vs 2.3%), and non-BED eating disorder (54.2% vs 1.4%).
Prescription Medication Use Prior to BED Diagnosis
Prescription medication utilization was observed for a mean of 2.8 years (SD = 1.0) before the index date or BED diagnosis (ie, the maximum availability of the Swedish Prescribed Drug Register records). Table 2 shows the prevalence of medication use and Figure 1 shows the case-control comparisons. As shown in Figure 1, cases were significantly more likely to be prescribed digestion and metabolism (reported as odds ratio [OR]; 95% confidence limit [CL]) (2.1; 1.5, 2.9) including antidiabetic medications (4.0; 1.6, 10.2) and all others (1.9; 1.3, 2.6), cardiovascular (2.2; 1.4, 3.5), skin (1.8; 1.3, 2.5), infectious diseases (1.9; 1.4, 2.6), tumors and immune disorders (3.5; 1.3, 9.3), nervous system (6.4; 4.7, 8.6) including all psychotropic classes medications examined, and respiratory system (1.3; 1.0, 1.8) medications.
Prescription Medication Use Within
12 Months After BED Diagnosis
Table 2 shows the prevalence of medication use among the 2 groups within 12 months after the index date of BED diagnosis, and Figure 2 presents the associations between BED and prescription medication utilization for this timeframe. Cases generally had higher odds of prescription medication utilization compared with controls, especially for psychotropic medications. Cases were also more likely than controls to be prescribed various nonpsychotropics, including digestion and metabolism (1.9; 1.2, 2.9), skin (1.8; 1.2, 2.8), hormones (1.7; 1.0, 2.9), infectious diseases (1.8; 1.3, 2.3), tumors and immune disorders (15.1; 3.2, 71.1), and respiratory system (2.2; 1.6, 3.0) medications.

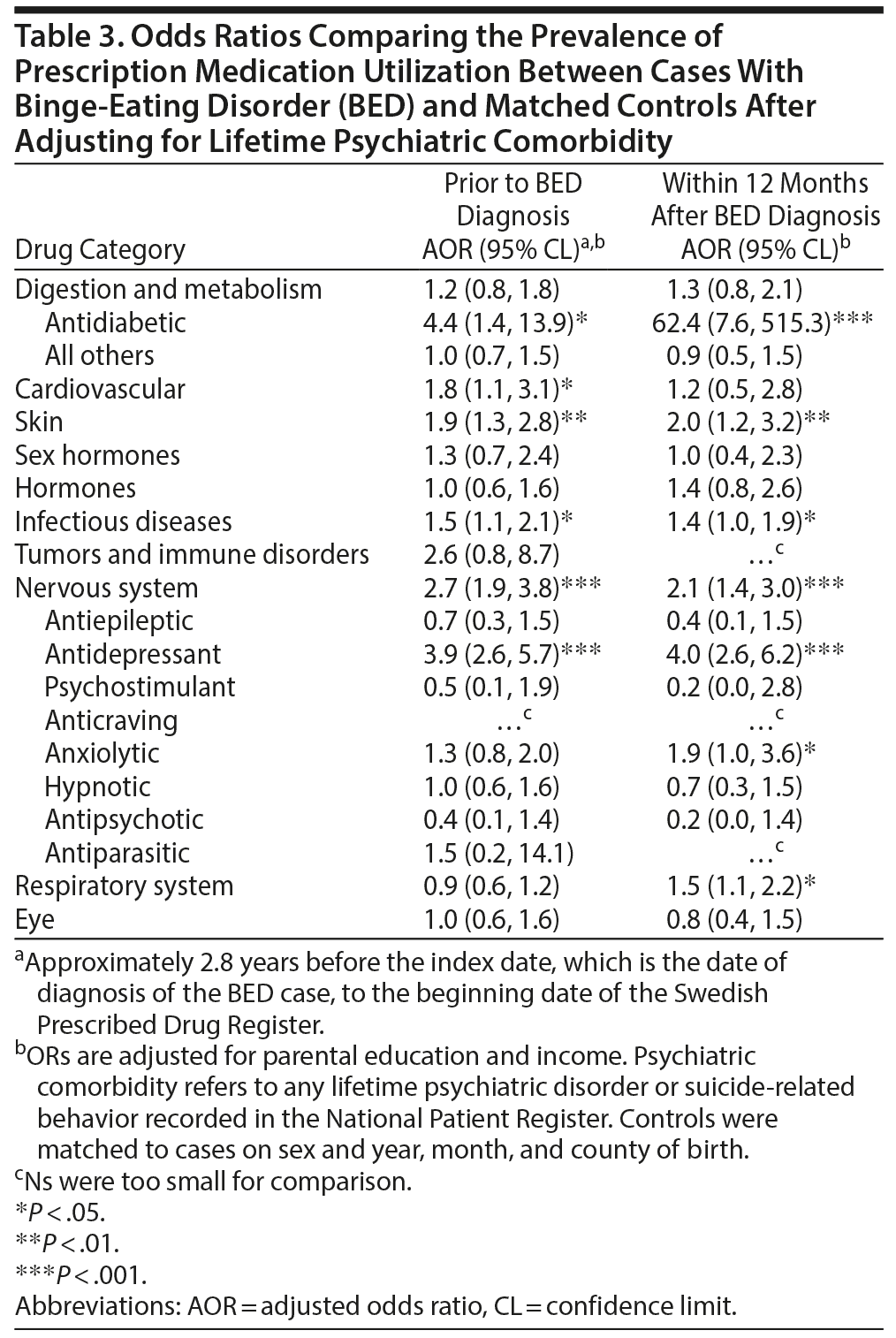
The Impact of Psychiatric Comorbidity
The associations between BED and prescription medication status were evaluated using lifetime psychiatric comorbidity as a covariate to partial out the variance in prescription medication utilization due to lifetime psychiatric comorbidity (Table 3). Similar patterns were observed in analyses both prior to BED diagnoses and within 12 months after BED diagnosis. The association between BED and antidiabetic medication strengthened. The associations between BED and skin, infectious diseases, nervous system overall (and antidepressant medication specifically), and respiratory system medications remained statistically significant: with the exception of skin medications, these ORs decreased (Table 3). The associations between BED and digestion and metabolism medications besides antidiabetics, and several specific nervous system medications, including antiepileptics, hypnotics, and antipsychotics, decreased and became statistically nonsignificant.
The Role of Comorbid Obesity in
Prescription Medication Utilization
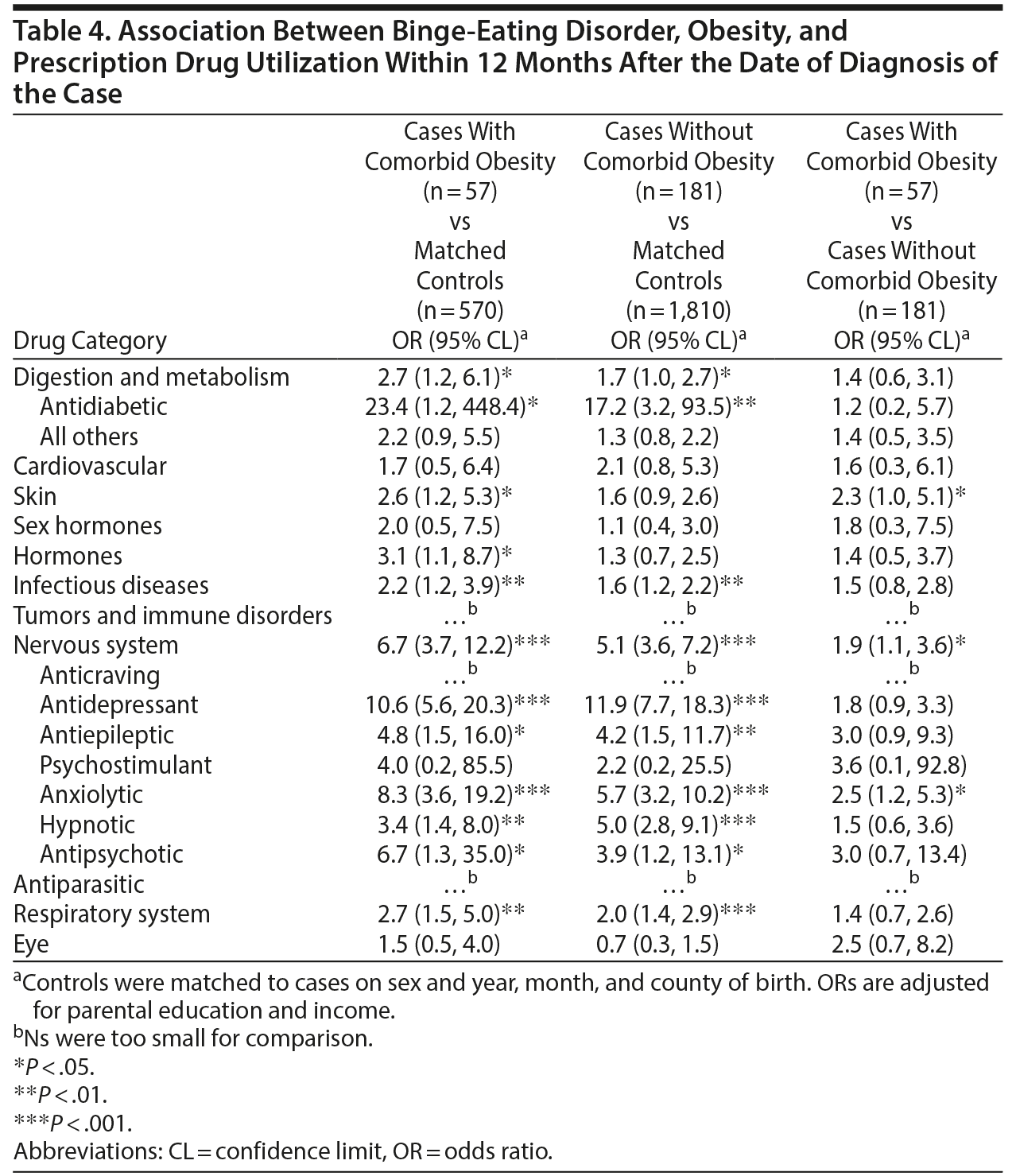
The association between BED and prescription medication utilization within 12 months after diagnosis was investigated with respect to comorbid obesity (Table 4). Given the caveat that we did not have BMI measures in controls, we performed group comparisons. Overall, cases with and without comorbid obesity had higher odds of prescription medication utilization for both psychotropic and nonpsychotropic medication than matched controls. When we directly compared utilization in BED cases with and without comorbid obesity, those with comorbid obesity were significantly more likely to be prescribed anxiolytics (2.5; 1.2, 5.3) and skin agents (2.3; 1.0, 5.1).
DISCUSSION
We compared treatment-seeking individuals with BED identified in national eating disorders quality registers across Sweden to matched controls and found striking differences in prescription medication utilization, before and in the 12 months following BED diagnosis. Differences were not limited to psychotropic medication but also spanned somatic medication categories including digestion and metabolism, cardiovascular, skin, hormones, infectious diseases, tumors and immune disorders, and respiratory system drugs.
The elevated medication utilization in cases supports and augments previous findings that BED is associated with high disease burden, such as depression, anxiety disorders, and somatic conditions including cardiovascular, type 2 diabetes, and respiratory problems.1–12 This study expands previous research by demonstrating that burden extends to prescription medication utilization, both before and after diagnosis of BED, and possibly skin, infectious disease, hormone, and immune-related comorbidity. These findings are consistent with the general observations of role impairment in people with BED.25,26
The overall finding of higher prescription medication utilization before BED diagnosis is relevant because stigma, poor self-recognition of problem eating, shame, and embarrassment impede help-seeking for BED.27,28 Further, primary care physicians rarely screen for binge eating and often fail to recognize BED,29 which contributes to low detection and treatment rates (< 10%).2 Individuals with BED often come to the attention of providers by seeking treatment for physical and other mental health conditions (ie, depression) or for weight loss.2,15 Primary health care professionals including psychiatrists can contribute to earlier detection and treatment of BED by inquiring about eating behaviors and disordered eating when treating patients with a risk profile for BED (ie, higher current BMI, childhood obesity, lifetime mood disorders, seeking weight loss, elevated psychiatric and nonpsychiatric prescription medication utilization).2,30 Accurate, timely diagnosis and treatment may lead to a reduced rate of onset of associated somatic and psychiatric problems and better long-term outcomes.
Our results show that BED was associated with higher utilization regardless of the presence of lifetime psychiatric comorbidity and obesity; however, both increased the odds of utilization. Similar to present findings, in a nationally representative survey of US adults,2 the association between chronic physical conditions and BED tempered when adjusting for preexisting psychiatric disorders. Compared with controls, prescription medication utilization for somatic problems was most elevated among cases with comorbid obesity, which may be attributable to the health consequences associated with obesity.11 Nevertheless, BED may confer risk of increased medication utilization independent of psychiatric history and obesity.
Several limitations to this study must be considered. First, we are unable to determine if any of the medications were prescribed to treat the symptoms of BED. Although evidence exists for the short-term efficacy of selective serotonin reuptake inhibitors (SSRIs), antiepileptics, and, more recently, lisdexamfetamine dimesylate (Vyvanse), the first medication approved for adults with BED by the US Food and Drug Administration,31–34 as in many other countries, there are no approved medications for the treatment of BED in Sweden. We know strikingly little about utilization of pharmacologic treatments for BED in the wider community. Second, the absence of BMI data for controls limited the extent to which we could evaluate obesity as a confounder. Third, although the medication register lists all medications prescribed and dispensed, it cannot verify that patients took the medications as prescribed. Fourth, the data linkage available to us did not include all ATC codes so we could not explore utilization as exhaustively as would be optimal or compare utilization in some ATC classes of interest, such as antiobesity. Fifth, given typical delays in treatment seeking, the time of first diagnosis of BED is unlikely to reflect age at onset. Sixth, selection bias is likely. BED is a diagnosis that is often either not elicited by the practitioner or volunteered by the patient, making its detection dependent on factors other than the presence of the disorder itself. Moreover, individuals with more severe presentations or circumstances favoring access to treatment may be more likely to access specialist services. Finally, our results cannot determine direction of causality. BED may be more likely to emerge in individuals with multiple medical and psychiatric conditions. Alternatively, BED may itself confer risk for developing comorbidities. Another possibility is that an unmeasured variable may exist that increases risk for both BED and multiple somatic and psychiatric problems leading to the observed pattern of increased medication utilization.
BED was associated with elevated prescription medication utilization before and after diagnosis, confirming that the psychiatric and somatic needs of individuals with BED are substantial. These findings have implications for identifying and treating BED earlier and understanding and preventing burden. The failure to screen for and detect BED, and perhaps the reluctance of patients to discuss BED with their providers,27,35 could lead to pharmacologic management of an array of symptoms that could be avoided were an accurate diagnosis and disposition for BED treatment made. BED is significantly underrecognized and underdiagnosed in clinical practice. Primary care and mental health providers should be alert for a constellation of risks and correlates of BED (obesity, fluctuating weight, depression, anxiety, childhood obesity, high medication utilization) and screen for BED accordingly. Rapid screening is possible36,37 and can facilitate accurate diagnosis and treatment referral. Further, mental health professionals should remain vigilant for somatic conditions in their BED patients and encourage thorough medical review and medication monitoring.
Submitted: February 3, 2016; accepted May 13, 2016.
Published online: July 7, 2016.
Drug names: lisdexamfetamine (Vyvanse).
Potential conflicts of interest: Dr Thornton is an investigator on a research grant from Shire. Dr von Hausswolff-Juhlin is a consultant on a research grant from Shire. Dr Norring is a consultant on a research grant from Shire. Dr Herman is an employee of Shire and holds stock and/or stock options in Shire. Dr Larsson has served as a speaker for Eli-Lilly and Shire and has received a research grant from Shire. Dr Bulik has received a research grant from Shire and a consulting fee from Ironshore. Drs Watson, Munn-Chernoff, and Welch; Mr Jangmo; and Ms Wiklund report no financial or other relationships relevant to the subject of this article.
Funding/support: This work was made possible by a research grant from Shire to the University of North Carolina at Chapel Hill. The Riksät quality register is financially supported by the Swedish State and the Swedish Association of Local Authorities and Regions. The Stepwise database is financially supported by Stockholm County Council. Dr Bulik acknowledges support from the Swedish Research Council (VR Dnr: 538-2013-8864).
Role of the sponsor: Research was funded by the sponsor, Shire Development LLC (Wayne, Pennsylvania). The sponsor was involved in the design and conduct of the study, planning the analysis and interpretation of data, and preparation, review, and approval of the manuscript. The sponsor was not involved in performing the statistical analysis.
Acknowledgments: Under the direction of the authors, Craig Slawecki, PhD, of Complete Healthcare Communications, LLC (Chadds Ford, Pennsylvania) provided assistance with the manuscript figures; this support was funded by Shire. Dr Slawecki reports no financial or other relationships relevant to the subject of this article.
REFERENCES
1. Hudson JI, Hiripi E, Pope HG Jr, et al. The prevalence and correlates of eating disorders in the National Comorbidity Survey Replication. Biol Psychiatry. 2007;61(3):348–358. doi:10.1016/j.biopsych.2006.03.040 PubMed
2. Kessler RC, Berglund PA, Chiu WT, et al. The prevalence and correlates of binge eating disorder in the World Health Organization World Mental Health Surveys. Biol Psychiatry. 2013;73(9):904–914. doi:10.1016/j.biopsych.2012.11.020 PubMed
3. Stein DJ, Aguilar-Gaxiola S, Alonso J, et al. Associations between mental disorders and subsequent onset of hypertension. Gen Hosp Psychiatry. 2014;36(2):142–149. doi:10.1016/j.genhosppsych.2013.11.002 PubMed
4. Scott KM, Lim C, Al-Hamzawi A, et al. Association of mental disorders with subsequent chronic physical conditions: World Mental Health Surveys from 17 countries. JAMA Psychiatry. 2016;73(2):150–158. doi:10.1001/jamapsychiatry.2015.2688 PubMed
5. Herpertz S, Albus C, Lichtblau K, et al. Relationship of weight and eating disorders in type 2 diabetic patients: a multicenter study. Int J Eat Disord. 2000;28(1):68–77. doi:10.1002/(SICI)1098-108X(200007)28:1<68::AID-EAT8>3.0.CO;2-R PubMed
6. Crow S, Kendall D, Praus B, et al. Binge eating and other psychopathology in patients with type II diabetes mellitus. Int J Eat Disord. 2001;30(2):222–226. doi:10.1002/eat.1077 PubMed
7. Raevuori A, Suokas J, Haukka J, et al. Highly increased risk of type 2 diabetes in patients with binge eating disorder and bulimia nervosa. Int J Eat Disord. 2015;48(6):555–562. doi:10.1002/eat.22334 PubMed
8. Javaras KN, Pope HG, Lalonde JK, et al. Co-occurrence of binge eating disorder with psychiatric and medical disorders. J Clin Psychiatry. 2008;69(2):266–273. doi:10.4088/JCP.v69n0213 PubMed
9. Johnson JG, Spitzer RL, Williams JB. Health problems, impairment and illnesses associated with bulimia nervosa and binge eating disorder among primary care and obstetric gynaecology patients. Psychol Med. 2001;31(8):1455–1466. doi:10.1017/S0033291701004640 PubMed
10. Peat CM, Huang L, Thornton LM, et al. Binge eating, body mass index, and gastrointestinal symptoms. J Psychosom Res. 2013;75(5):456–461. doi:10.1016/j.jpsychores.2013.08.009 PubMed
11. Thornton LM, Watson H, Jangmo A, et al. Binge eating disorder in the Swedish national registers: somatic comorbidity. Presented at the International Conference on Eating Disorders. May 5–7, 2016; San Francisco, CA.
12. Bulik CM, Sullivan PF, Kendler KS. Medical and psychiatric morbidity in obese women with and without binge eating. Int J Eat Disord. 2002;32(1):72–78. doi:10.1002/eat.10072 PubMed
13. Pagoto S, Bodenlos JS, Kantor L, et al. Association of major depression and binge eating disorder with weight loss in a clinical setting. Obesity (Silver Spring). 2007;15(11):2557–2559. doi:10.1038/oby.2007.307 PubMed
14. Schag K, Teufel M, Junne F, et al. Impulsivity in binge eating disorder: food cues elicit increased reward responses and disinhibition. PLoS ONE. 2013;8(10):e76542. doi:10.1371/journal.pone.0076542 PubMed
15. Mond JM, Hay PJ, Rodgers B, et al. Health service utilization for eating disorders: findings from a community-based study. Int J Eat Disord. 2007;40(5):399–408. doi:10.1002/eat.20382 PubMed
16. Swedish Association of Local Authorities and Regions. National Healthcare Quality Registries in Sweden. Stockholm, Sweden: KLF Grafisk Producation; 2007.
17. Birgegård A, Björck C, Clinton D. Quality assurance of specialised treatment of eating disorders using large-scale Internet-based collection systems: methods, results and lessons learned from designing the Stepwise database. Eur Eat Disord Rev. 2010;18(4):251–259. doi:10.1002/erv.1003 PubMed
18. Runfola CD, Thornton LM, Pisetsky EM, et al. Self-image and suicide in a Swedish national eating disorders clinical register. Compr Psychiatry. 2014;55(3):439–449. doi:10.1016/j.comppsych.2013.11.007 PubMed
19. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Fourth Edition Text Revision. Washington, DC: American Psychiatric Press; 2000.
20. Statistics Sweden. Statistics Sweden Web site. http://www.scb.se/en_/. Accessed May 11, 2015.
21. Wettermark B, Hammar N, Fored CM, et al. The new Swedish Prescribed Drug Register—opportunities for pharmacoepidemiological research and experience from the first six months. Pharmacoepidemiol Drug Saf. 2007;16(7):726–735. doi:10.1002/pds.1294 PubMed
22. World Health Organization (WHO). International Classification of Diseases, Ninth Revision (ICD-9). Geneva, Switzerland: World Health Organization; 1977.
23. World Health Organization. International Classification of Diseases, Tenth Revision. Geneva, Switzerland: World Health Organization; 1992.
24. Rothman KJ. No adjustments are needed for multiple comparisons. Epidemiology. 1990;1(1):43–46. doi:10.1097/00001648-199001000-00010 PubMed
25. Preti A, Girolamo G, Vilagut G, et al; ESEMeD-WMH Investigators. The epidemiology of eating disorders in six European countries: results of the ESEMeD-WMH project. J Psychiatr Res. 2009;43(14):1125–1132. doi:10.1016/j.jpsychires.2009.04.003 PubMed
26. Kessler RC, Shahly V, Hudson JI, et al. A comparative analysis of role attainment and impairment in binge-eating disorder and bulimia nervosa: results from the WHO World Mental Health Surveys. Epidemiol Psychiatr Sci. 2014;23(1):27–41. doi:10.1017/S2045796013000516 PubMed
27. Evans EJ, Hay PJ, Mond J, et al. Barriers to help-seeking in young women with eating disorders: a qualitative exploration in a longitudinal community survey. Eat Disord. 2011;19(3):270–285. doi:10.1080/10640266.2011.566152 PubMed
28. Mond J, Hay P, Rodgers B, et al. Self-recognition of disordered eating among women with bulimic-type eating disorders: a community-based study. Int J Eat Disord. 2006;39(8):747–753. doi:10.1002/eat.20306 PubMed
29. Crow SJ, Peterson CB, Levine AS, et al. A survey of binge eating and obesity treatment practices among primary care providers. Int J Eat Disord. 2004;35(3):348–353. doi:10.1002/eat.10266 PubMed
30. Striegel-Moore RH, Fairburn CG, Wilfley DE, et al. Toward an understanding of risk factors for binge-eating disorder in black and white women: a community-based case-control study. Psychol Med. 2005;35(6):907–917. doi:10.1017/S0033291704003435 PubMed
31. McElroy SL, Guerdjikova AI, Blom TJ, et al. A placebo-controlled pilot study of the novel opioid receptor antagonist ALKS-33 in binge eating disorder. Int J Eat Disord. 2013;46(3):239–245. doi:10.1002/eat.22114 PubMed
32. McElroy SL, Guerdjikova AI, Winstanley EL, et al. Acamprosate in the treatment of binge eating disorder: a placebo-controlled trial. Int J Eat Disord. 2011;44(1):81–90. doi:10.1002/eat.20876 PubMed
33. National Institute for Clinical Excellence (NICE). Eating Disorders: Core Interventions in the Treatment and Management of Anorexia Nervosa, Bulimia Nervosa, and Related Eating Disorders. Clinical Guideline 9. London, UK: NICE; 2004.
34. Reas DL, Grilo CM. Pharmacological treatment of binge eating disorder: update review and synthesis. Expert Opin Pharmacother. 2015;16(10):1463–1478. doi:10.1517/14656566.2015.1053465 PubMed
35. Herman BK, Safikhani S, Hengerer D, et al. The patient experience with DSM-5-defined binge eating disorder: characteristics, barriers to treatment, and implications for primary care physicians. Postgrad Med. 2014;126(5):52–63. doi:10.3810/pgm.2014.09.2800 PubMed
36. Herman BK, Deal L, DiBenedetti DB, et al. Development of the Binge-Eating Disorder Screener. Presented at the 168th Annual Meeting of the American Psychiatric Association. May 16–20, 2015; Toronto, Canada.
37. Luck AJ, Morgan JF, Reid F, et al. The SCOFF questionnaire and clinical interview for eating disorders in general practice: comparative study. BMJ. 2002;325(7367):755–756. doi:10.1136/bmj.325.7367.755 PubMed
This PDF is free for all visitors!


