Objective: Catatonia is a motor dysregulation syndrome often accompanied by deep vein thrombosis (DVT) and pulmonary embolism (PE). Although electroconvulsive therapy (ECT) is effective for catatonia, it is unknown whether ECT contributes to the onset of a PE from a residual DVT. The objective of this case series is to examine and propose safety methods for ECT in catatonia patients with a DVT.
Methods: Data were obtained retrospectively via chart review for 5 psychiatric inpatients diagnosed with catatonia based on DSM-IV-TR or DSM-5 criteria from April 2010 to March 2017 who underwent ECT after developing a DVT.
Results: All 5 patients received anticoagulation therapy after the onset of DVT and underwent subsequent ECT. Three patients had distal DVT (thromboses located below the knee in the calf veins) before ECT, which did not result in an onset of PE in the course of ECT. One had a proximal DVT (thromboses in the popliteal vein and above), and the ECT session was completed without the occurrence of PE. In the fifth patient, a proximal DVT developed into a PE after an ECT session.
Conclusions: These results suggest that it is important to determine the location of a DVT and to continue anticoagulation therapy until a proximal DVT disappears before ECT is performed.
Electroconvulsive Therapy for Catatonia
With Deep Venous Thrombosis:
A Case Series
ABSTRACT
Objective: Catatonia is a motor dysregulation syndrome often accompanied by deep vein thrombosis (DVT) and pulmonary embolism (PE). Although electroconvulsive therapy (ECT) is effective for catatonia, it is unknown whether ECT contributes to the onset of a PE from a residual DVT. The objective of this case series is to examine and propose safety methods for ECT in catatonia patients with a DVT.
Methods: Data were obtained retrospectively via chart review for 5 psychiatric inpatients diagnosed with catatonia based on DSM-IV-TR or DSM-5 criteria from April 2010 to March 2017 who underwent ECT after developing a DVT.
Results: All 5 patients received anticoagulation therapy after the onset of DVT and underwent subsequent ECT. Three patients had distal DVT (thromboses located below the knee in the calf veins) before ECT, which did not result in an onset of PE in the course of ECT. One had a proximal DVT (thromboses in the popliteal vein and above), and the ECT session was completed without the occurrence of PE. In the fifth patient, a proximal DVT developed into a PE after an ECT session.
Conclusions: These results suggest that it is important to determine the location of a DVT and to continue anticoagulation therapy until a proximal DVT disappears before ECT is performed.
Prim Care Companion CNS Disord 2018;20(4):18m02286
To cite: Inagawa Y, Saito S, Okada T, et al. Electroconvulsive therapy for catatonia with deep venous thrombosis: a case series. Prim Care Companion CNS Disord. 2018;20(4):18m02286.
To share: https://doi.org/10.4088/PCC.18m02286
© Copyright 2018 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Jichi Medical University, Tochigi, Japan
bDepartment of Mental Health, Saitama Medical Center, Jichi Medical University, Saitama, Japan
*Corresponding author: Yuta Inagawa, MD, Department of Psychiatry, Jichi Medical University, 3311-1 Yakushiji, Shimotsuke, Tochigi, 329-0498, Japan ([email protected]).
Catatonia is a motor dysregulation syndrome due to different causes that often develops into immobility. Immobility in catatonia patients is accompanied by blood stasis that increases the risk of venous thromboembolism (ie, a blood clot in a vein). Venous thromboembolisms can be a deep vein thrombosis (DVT) or a pulmonary embolism (PE). Ishida and colleagues1 reported that the incidence of DVT in catatonia patients was significantly higher than that of noncatatonia patients with physical restraints (35.7% and 10.6%, respectively). The trigger of DVT is postulated to be involved with immobility due to stupor, as well as medical practices such as physical restraints and sedation with antipsychotics.2–4 Other complications often occur in catatonia patients (eg, aspiration pneumonia, contracture, decubitus ulcer),5 resulting in a longer duration of illness. Several studies6–9 have addressed the question of whether a longer duration of catatonic symptoms could cause poor treatment responses to not only benzodiazepines but also electroconvulsive therapy (ECT). In this regard, it is important to resolve catatonic symptoms as soon as possible before the development of complications and treatment resistance.
ECT has been reported to be the most effective therapy for catatonic patients.6,8,10 Although ECT is generally considered a safe procedure,11 a few studies12–14 reported that PEs have occurred during ECT sessions and that the PEs probably led to sudden death. Mamah and colleagues15 suggested that the pathogenesis of the onset of a PE during an ECT session could lie in strenuous muscle contraction due to a generalized seizure, resulting in the detachment of a clot and embolization from residual DVT. However, the risk of PE during ECT sessions remains controversial; different lines of evidence showed that ECT sessions could be successfully completed after the development of a venous thromboembolism with no complications.10,16–19
The risk of developing a PE is considered to be higher in patients with a proximal DVT (ie, a DVT located in veins above the knee) compared to patients with a distal DVT (ie, a DVT below the knee).20 Nevertheless, to our knowledge, there is no report elucidating the association between the location of a residual DVT and the new onset of a PE during an ECT session. Thus, no consensus regarding the safe introduction and procedures of ECT in patients with a residual DVT has been established. In this case series, we describe 5 catatonia patients with residual DVTs who underwent ECT sessions. Our study is unique in that we focused on the association between the location of the residual DVT and the new onset of a PE.
METHODS
Through chart review, we identified patients diagnosed with catatonia based on the DSM-IV-TR or DSM-5 criteria who were admitted to the psychiatric ward of Jichi Medical University Hospital between April 2010 and March 2017. The clinical data of the patients including the diagnosis, clinical manifestations of catatonia, laboratory data, computed tomography (CT) images, and clinical course during ECT sessions were obtained. Anesthesia was induced through intravenous propofol 50 mg or thiopental 100–275 mg. Muscle relaxation was induced through intravenous rocuronium 20–30 mg or suxamethonium 30–40 mg. Informed consents were obtained from all patients to publish the case reports. The patient information has been de-identified to protect anonymity. Ethical approval for this retrospective study was granted by the Ethics Committee at Jichi Medical University.
RESULTS
Over a period of 8 years, 5 patients with catatonia had undergone ECT after developing a DVT. The patients’ clinical features are summarized in Table 1. All 5 patients were women and Japanese, with a mean age of 55.2 years. Three of the 5 patients had schizophrenia, 1 patient had bipolar I disorder, and 1 patient had major depressive disorder. All of the patients were administered anticoagulant medications during their ECT sessions, and no bleeding-related complications occurred.
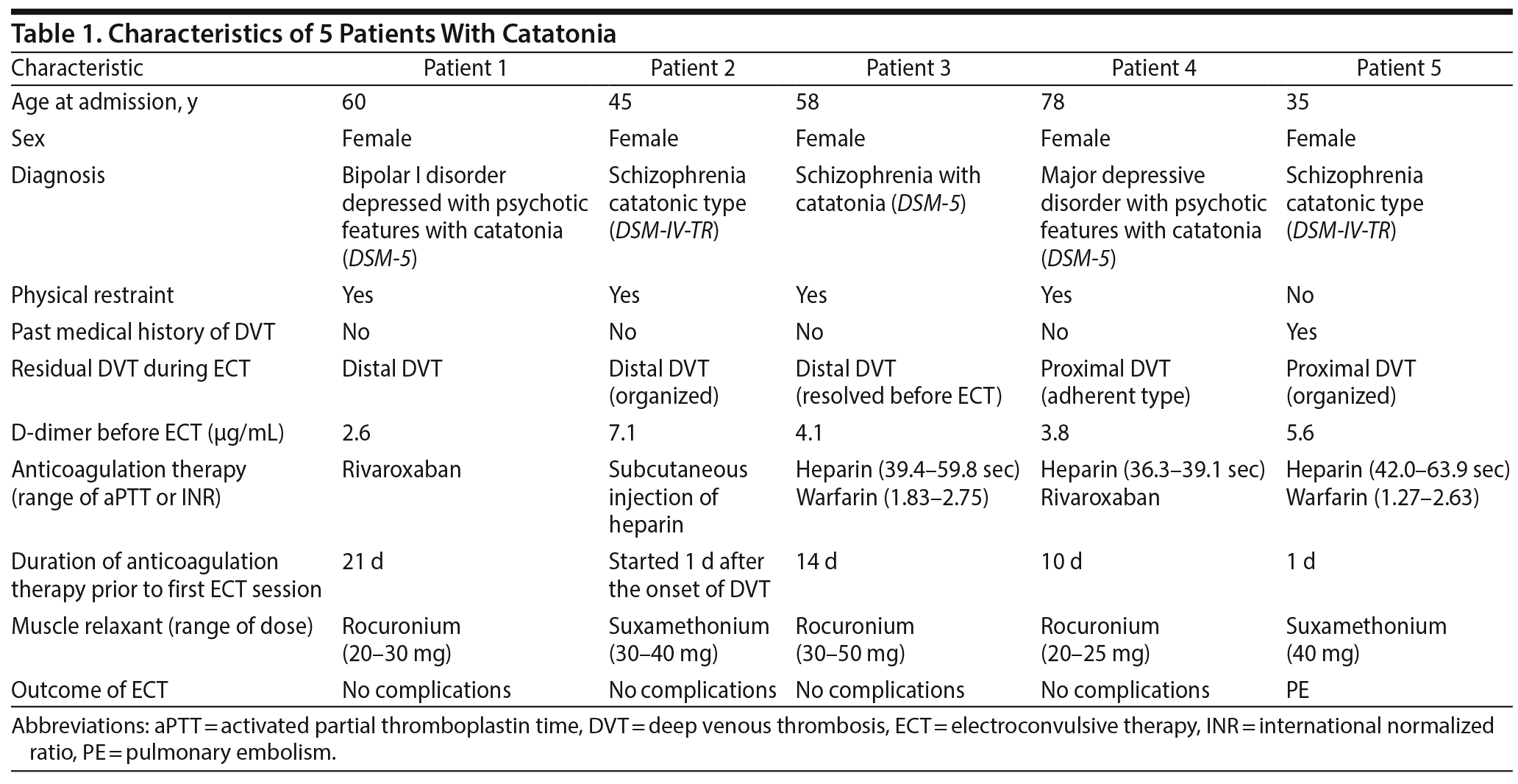
DVTs are generally classified into proximal DVTs (a thrombosis located in the popliteal, femoral, or iliac veins) and distal DVTs (a thrombosis located in the veins of the calf, including the anterior tibial vein, posterior tibial vein, and peroneal veins). Four of the patients, who needed to be physically restrained due to psychomotor excitement or self-removal of tubes and devices, developed a distal DVT. The remaining patient (patient 5), who had a past medical history of DVT, developed proximal DVT and subsequent PE without physical restraint. Additionally, in patient 5, ultrasonography was ineffective for evaluating the existence of a DVT above the popliteal vein.
In the following section, the cases of patients 1, 4, and 5 are presented (patient numbers are as noted in Table 1). Since the clinical course of patients 2 and 3 were similar to that of patient 1, we do not describe those cases to avoid redundancy.
CASES
Patient 1
A 60-year-old Japanese woman with a history of multiple hospitalizations due to recurrent bipolar disorder exhibited insomnia, hypochondriac symptoms, and depression. One month later, she was admitted to a psychiatric hospital. At admission, the laboratory evaluation showed dehydration with hypernatremia and mild renal dysfunction, and she was transferred to a general hospital. For the treatment of the patient’s dehydration, hypernatremia, and mild renal dysfunction, fluid replacement was conducted, and lithium was discontinued. Several months after discharge, she presented with insomnia and depressive symptoms again, resulting in readmission to the psychiatric hospital. She gradually showed marked psychomotor excitement, which was treated with an intravenous administration of flunitrazepam and haloperidol. Nine days later, her arterial oxygen saturation dropped to 85% on room air. Laboratory test results revealed elevations of D-dimer (30.2 μg/mL) and fibrinogen degradation products (43.2 μg/mL), thus new onset of a PE was suspected. She was referred to the intensive care unit in our hospital.
Contrast-enhanced CT demonstrated bilateral pulmonary infarction and DVTs in the left popliteal vein and bilateral tibial veins (ie, proximal and distal DVTs). She was initially treated with 5,000 units of heparin and 800,000 units of monteplase, followed by a continuous intravenous heparin infusion. On day 5, reexamination by CT with contrast revealed reduction of the PE, and residual DVT was found only in bilateral tibial veins (distal DVT). The heparin infusion was replaced by oral rivaroxaban treatment at a dose of 15 mg/d. However, she refused to take this medicine and presented with pica (she ate wooden chopsticks). Physical restraint was used to prevent the self-removal of intravenous lines and the nasal tube, and flunitrazepam and lorazepam were started. On day 10, she was transferred to the psychiatric ward.
Due to marked mutism, stupor, and posturing, she could not eat or take medication voluntarily. She was diagnosed with catatonia, and the administration of medication and feeding via a nasal tube was initiated. On day 13, she pulled her nasal tube out despite the physical restraints. Since the treatment with benzodiazepines was found to be ineffective, the initiation of ECT was considered. On day 16, a laboratory investigation revealed a decrease in D-dimer (2.6 μg/mL), which indicated a low risk of a new onset of a PE. The patient’s family gave written informed consent, and a course of bilateral ECT was started. After the third ECT session, the patient was able to take food and medication orally. Since her catatonic features and delusions resolved, the ECT was discontinued (6 sessions in total), and the use of physical restraint was stopped. Lithium was restarted at 400 mg and gradually increased to 1,000 mg (0.58 mEq/L). Three months after discharge, oral rivaroxaban treatment was discontinued. The patient remained in remission for 2 years.
Patient 4
A Japanese woman developed Parkinson’s disease at the age of 73 years. Antiparkinson medications had kept her medically stable for 5 years. At the age of 78 years, she presented to the hospital with visual hallucinations. Several months later, she was admitted to the neurologic ward in our hospital because she refused to eat due to her delusion of guilt, and she became bedridden. On hospital day 3, physical restraint was performed to prevent her from slipping down or falling from the bed. On hospital day 4, she was diagnosed with major depressive disorder with psychotic features; mirtazapine 15 mg/d and quetiapine 37.5 mg/d were started. On hospital day 7, laboratory results showed an increase in D-dimer (17.0 μg/mL) and fibrinogen degradation products (21.7 μg/mL). CT with contrast revealed the new onset of a venous thromboembolism, including bilateral PEs and DVTs in the right popliteal vein and left soleal vein (proximal and distal DVTs). Continuous intravenous heparin infusion was initiated (10,000–15,000 units/d). However, catatonic features including mutism, negativism, and stupor were gradually apparent. On hospital day 11, she was transferred to our psychiatric ward.
The continuous intravenous heparin infusion was switched to an administration of rivaroxaban via a nasal tube on hospital day 11. Two days later, reexamination by CT with contrast revealed that the bilateral PEs had reduced, whereas the DVT located in the right popliteal vein had not diminished. Echocardiography showed normal cardiac function. On hospital day 14, the patient presented with a high fever (101.3°F) and a decrease in oxygen saturation (90% in room air) because she had developed aspiration pneumonia due to dysphagia. Oxygen supplementation and ampicillin/sulbactam 3.0 g every 8 hours were started.
Although a reexamination by CT with contrast showed residual DVT in the right popliteal vein (a proximal DVT), the benefits of ECT were weighed against the risk of the onset of PE and bleeding since the patient’s persistent depressive symptoms and other complications had caused muscle weakness, leading to a poor prognosis. ECT was proposed with those risks, and written informed consent was obtained from her family. On hospital day 17, the first ECT session was initiated. After the fourth ECT session, the patient’s depressive symptoms and delusion of guilt dramatically resolved. ECT was discontinued after a total of 6 sessions due to the patient’s clinical stability. She was transferred to another hospital on day 60 because she needed a continuation of rehabilitation for dysphagia.
Patient 5
The patient was a 35-year-old Japanese woman. At the age of 15 years, she developed schizophrenia with persecutory delusions and insomnia, which required 4 months of hospitalization in our psychiatric ward. At the age of 32 years, she decided to stop taking antipsychotic medication because of her pregnancy. One day after her delivery, she suddenly presented with delusional perception, severe confusion, and psychomotor excitement. She was immediately transferred to our psychiatric ward, and haloperidol and chlorpromazine were started. When the patient complained of pain in her left leg, CT with contrast was performed. The CT results revealed a DVT below her knee (a distal DVT), but anticoagulant medication was not started due to the risk of obstetric bleeding, which was weighed against the benefit of treating the DVT. Two months after the hospitalization, she remitted and was discharged with a regimen of olanzapine 5 mg/d.
At the age of 35 years, the patient exhibited insomnia after a huge earthquake occurred off the east coast of Japan. Seven days after the earthquake, she was admitted to our psychiatric ward with a recurrence of persecutory delusions and severe confusion. At admission, she presented with severe psychomotor excitement and negativism. The laboratory examination was notable for the elevation of creatine phosphokinase (9,467 U/L) with a high fever (> 100.4°F). For her catatonic symptoms, intravenous diazepam 10 mg/d was started. On the following day, her excitement and high fever resolved. Intravenous haloperidol 15 mg/d and oral olanzapine 10 mg/d were then initiated. On day 6, the patient developed a high fever (> 100.4°F), tachycardia (> 180 beats/min), muscle rigidity, catalepsy, and stupor. A laboratory examination revealed a decrease in creatine phosphokinase (1,559 U/L). Since malignant catatonia was suspected, the antipsychotic medication was withdrawn. On day 8, a course of bilateral ECT was initiated after written informed consent was obtained from her family. One day after the first ECT session, a laboratory investigation showed an elevation of D-dimer (5.6 μg/mL). Ultrasonography revealed an organized DVT in the left popliteal vein (a proximal DVT), but we could not evaluate the existence of DVT above the popliteal vein because the patient was unable to cooperate with the test and enhanced CT. A continuous intravenous heparin infusion was started immediately. Since her catatonia symptoms were persistent, the continuation of ECT was considered. After the risk of the onset of PE and cerebral hemorrhage in the course of ECT was explained, the patient’s family provided written consent to continue the treatment. On hospital day 10, the second ECT session was performed. The continuous administration of heparin was discontinued 6 hours before the ECT session to prevent subsequent brain hemorrhage.19 One day after the second session, the patient’s oxygen saturation was decreased to 86% in room air, and CT with contrast detected the new onset of bilateral PEs. The ECT session was then discontinued.
DISCUSSION
All 5 of the catatonia patients in this case series received anticoagulation therapy for distal or proximal DVTs. Although the administration of anticoagulation therapy for a distal DVT remains controversial because of the bleeding risk, it is reported that anticoagulation therapy reduces the risk of the onset of PE. For example, Utter et al21 reported that anticoagulation therapy reduced the rate of onset of both proximal DVT and PE from distal DVT. Since proximal DVT is considered to be a higher risk factor for PE, it is necessary to continue anticoagulant treatment even for a distal DVT for at least 6 weeks to 3 months to prevent the subsequent development of a proximal DVT.22,23
On the other hand, anticoagulation therapy is generally thought to induce bleeding tendency.21 There are several reports that anticoagulation therapy did not increase the risk of bleeding during ECT. Mehta et al24 reported no ECT-related complications due to warfarin therapy among 35 patients. Our findings are also in line with a study25 reporting no ECT-related complications due to rivaroxaban therapy. However, Suzuki et al19 suggested that the discontinuation of heparin before each ECT session prevented brain hemorrhage induced by transient hypertension due to the procedure. These findings indicate that anticoagulation therapy is a beneficial treatment for patients with distal DVTs during ECT to prevent the propagation of the DVT and a new onset of PE.
There are several reports indicating that the courses of ECT were safely completed after the development of a venous thromboembolism. The case reports by Dean and Coconcea,16 Pandya et al,17 and Singh and Wahi18 showed no recurrence of PE during ECT sessions after the development of PE. Medda et al10 reported that a catatonia patient with DVT in a popliteal vein (a proximal DVT) safely underwent ECT after anticoagulation therapy. However, in those cases, it was unknown whether the residual DVT vanished with anticoagulation therapy just before the initiation of ECT. Suzuki et al19 showed that 2 patients with PE safely underwent ECT sessions after the absence of residual DVT was observed. To our knowledge, there have been no reports of the location of residual DVT just before the initiation of ECT. Taken together, the past and present reports highlight that the association of the onset of PE and the location of residual DVT remains unclear.
Patients 1–3 developed a distal DVT and received anticoagulation therapy. Although in patient 3 the DVT resolved before the initiation of ECT, residual distal DVT existed in patients 1 and 2. Additionally, patient 4 developed a DVT in the popliteal vein (a proximal DVT) just before her ECT sessions. There were no complications in these cases, suggesting that the benefits of ECT outweighed the risk of embolization from residual DVT and other complications, including aspiration pneumonia, contracture, or decubitus ulcer. Moreover, the anticoagulation therapy might have prevented the new development of DVT in all 4 patients.
In patient 5, however, anticoagulant treatment was ineffective in the prophylaxis against PE. We could not confirm the absence of a proximal DVT above the popliteal vein by ultrasonography. A possible explanation is that the new onset of PE was derived from the proximal vein, which we could not assess. In addition, it was reported that the embolization from residual DVT could result from strenuous muscle contraction due to a generalized seizure.15 Since the onset of PE occurred 1 day after the second session of ECT and the patient had received muscle relaxants to prevent strenuous muscle contraction during each ECT session, ECT might not directly contribute to embolization from residual DVT.
Although it is generally necessary to continue anticoagulation therapy for 6 weeks to 3 months, a longer duration of catatonic symptoms could cause a poor response to medication or ECT. As noted, it is important to initiate ECT immediately in patients with catatonia to prevent subsequent complications. On the basis of these findings, we suggest that anticoagulation therapy should be continued until a proximal DVT disappears before safely initiating ECT. In cases in which ultrasonography does not reveal whether a DVT exists in proximal veins, clinicians should be cautioned to perform subsequent CT with contrast to detect the existence of DVT in whole veins. In short, ECT for patients with a DVT might be possible after the absence or disappearance of a proximal DVT is confirmed.
The benefits of initiating ECT should always be weighed against the risks of PE from residual DVT. If the risks are weighed against the benefits of ECT, anticoagulant treatment for a residual DVT should take precedence over the initiation of ECT. In contrast, given that the benefits of ECT are weighed against the risks of PE (especially in patients with catatonia), ECT sessions in patients with a distal versus proximal DVT would contribute to better outcomes. Although more studies with larger numbers of patients are warranted to evaluate the association between the onset of PE and the location of residual DVT, the cases described here suggest that ECT could be considered as a safe treatment for individuals with a distal DVT.
Submitted: February 18, 2018; accepted April 3, 2018.
Published online: July 5, 2018.
Potential conflicts of interest: None.
Funding/support: None.
Previous presentation: Presented at the World Psychiatric Association (WPA) XVII World Congress; Berlin, Germany; October 8–12, 2017.
REFERENCES
1. Ishida T, Sakurai H, Watanabe K, et al. Incidence of deep vein thrombosis in catatonic patients: a chart review. Psychiatry Res. 2016;241:61–65. PubMed CrossRef
2. Barbui C, Conti V, Cipriani A. Antipsychotic drug exposure and risk of venous thromboembolism: a systematic review and meta-analysis of observational studies. Drug Saf. 2014;37(2):79–90. PubMed CrossRef
3. Chandele PH, Cholera R, Kale S, et al. Theoretical and practical issues related to the management of severe and refractory psychotic illness complicated by pulmonary embolism. Indian J Psychiatry. 2015;57(4):414–418. PubMed CrossRef
4. Ishiguro C, Wang X, Li L, et al. Antipsychotic drugs and risk of idiopathic venous thromboembolism: a nested case-control study using the CPRD. Pharmacoepidemiol Drug Saf. 2014;23(11):1168–1175. PubMed CrossRef
5. Clinebell K, Azzam PN, Gopalan P, et al. Guidelines for preventing common medical complications of catatonia: case report and literature review. J Clin Psychiatry. 2014;75(6):644–651. PubMed CrossRef
6. Luchini F, Medda P, Mariani MG, et al. Electroconvulsive therapy in catatonic patients: efficacy and predictors of response. World J Psychiatry. 2015;5(2):182–192. PubMed CrossRef
7. Narayanaswamy JC, Tibrewal P, Zutshi A, et al. Clinical predictors of response to treatment in catatonia. Gen Hosp Psychiatry. 2012;34(3):312–316. PubMed CrossRef
8. Raveendranathan D, Narayanaswamy JC, Reddi SV. Response rate of catatonia to electroconvulsive therapy and its clinical correlates. Eur Arch Psychiatry Clin Neurosci. 2012;262(5):425–430. PubMed CrossRef
9. Sienaert P, Dhossche DM, Vancampfort D, et al. A clinical review of the treatment of catatonia. Front Psychiatry. 2014;5:181. PubMed CrossRef
10. Medda P, Fornaro M, Fratta S, et al. A case of deep venous thrombosis following protracted catatonic immobility recovered with electroconvulsive therapy: the relevance for an early intervention. Gen Hosp Psychiatry. 2012;34(2):209.e5–209.e7. PubMed CrossRef
11. Dennis NM, Dennis PA, Shafer A, et al. Electroconvulsive therapy and all-cause mortality in Texas, 1998–2013. J ECT. 2017;33(1):22–25. PubMed CrossRef
12. Hamdieh M, Safaie N, Abbasinejad M. A psychiatric patient with catatonia and pulmonary thromboemboli. J Neuropsychiatry Clin Neurosci. 2014;26(1):E19–E20. PubMed CrossRef
13. Kursawe HK, Schmikaly R. Fatal pulmonary embolism following mitigated electroconvulsive therapy: a case report from the intensive care viewpoint. Psychiatr Neurol Med Psychol Beih. 1988;40:107–108. PubMed
14. Weber DL, Ruvolo C, Cashin P. Sudden death following electroconvulsive therapy. N Y State J Med. 1973;73(8):1000–1001. PubMed
15. Mamah D, Lammle M, Isenberg KE. Pulmonary embolism after ECT. J ECT. 2005;21(1):39–40. PubMed CrossRef
16. Dean J, Coconcea C. Electroconvulsive therapy in a patient with pulmonary embolism: a case report. J ECT. 2016;32(3):e12. PubMed CrossRef
17. Pandya HN, Keyes MJ, Christenson BC. Electroconvulsive therapy in a schizophrenic patient with neuroleptic malignant syndrome and pulmonary embolism: a case report. Psychiatry (Edgmont). 2007;4(4):21. PubMed
18. Singh G, Wahi S. Pulmonary embolism in the ECT patient: a case report and discussion. Gen Hosp Psychiatry. 2008;30(1):87–89. PubMed CrossRef
19. Suzuki K, Takamatsu K, Takano T, et al. Safety of electroconvulsive therapy in psychiatric patients shortly after the occurrence of pulmonary embolism. J ECT. 2008;24(4):286–288. PubMed CrossRef
20. Kearon C. Natural history of venous thromboembolism. Circulation. 2003;107(suppl 1):I22–I30. PubMed CrossRef
21. Utter GH, Dhillon TS, Salcedo ES, et al. Therapeutic anticoagulation for isolated calf deep vein thrombosis. JAMA Surg. 2016;151(9):e161770. PubMed CrossRef
22. Li AY, Woulfe T, Rolfe-Vyson V, et al. Management and outcomes of axial isolated distal deep vein thrombosis at North Shore Hospital, New Zealand: a retrospective audit. Intern Med J. 2015;45(2):177–182. PubMed CrossRef
23. Streiff MB, Agnelli G, Connors JM, et al. Guidance for the treatment of deep vein thrombosis and pulmonary embolism. J Thromb Thrombolysis. 2016;41(1):32–67. PubMed CrossRef
24. Mehta V, Mueller PS, Gonzalez-Arriaza HL, et al. Safety of electroconvulsive therapy in patients receiving long-term warfarin therapy. Mayo Clin Proc. 2004;79(11):1396–1401. PubMed CrossRef
25. Shuman M, Hieber R, Moss L, et al. Rivaroxaban for thromboprophylaxis in a patient receiving electroconvulsive therapy. J ECT. 2015;31(1):e19–e20. PubMed CrossRef
This PDF is free for all visitors!


