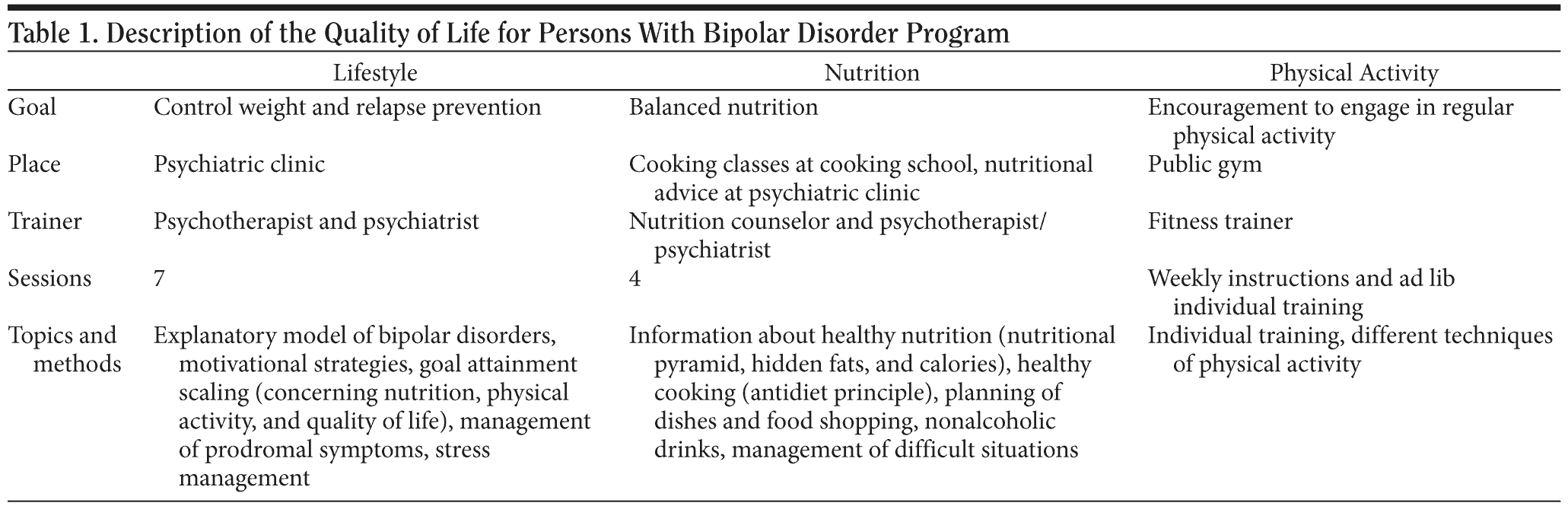
Effects of a Multimodal Lifestyle Intervention on Body Mass Index in Patients With Bipolar Disorder: A Randomized Controlled Trial
Objective: Patients with bipolar disorder are at increased risk of weight gain, which in turn increases the risk for somatic disease and nonadherence to maintenance therapy. Therefore, interventions addressing weight gain are expedient for the management of this disorder. We set out to evaluate the effects of a lifestyle intervention on body mass index (BMI) and cardiovascular and metabolic parameters in patients with bipolar disorder undergoing mood-stabilizing pharmacologic treatment.
Method: Fifty outpatients with bipolar disorder undergoing mood-stabilizing treatment participated in a randomized controlled trial (waiting control group: n = 24 and multimodal lifestyle intervention group: n = 26). Groups consisted of 2 cohorts (cohort 1: March 2005-February 2006; cohort 2: September 2005-August 2006). The intervention lasted 5 months and consisted of 11 group sessions and weekly fitness training. BMI and body weight as well as cardiovascular and metabolic parameters were determined at 3 assessment points: at pretreatment baseline, at 5 months (end of treatment), and at 11 months (6-month follow-up).
Results: Intention-to-treat analyses showed that the intervention significantly reduced BMI over time (P = .03), with significant and stable mean differences in BMI change between groups of 0.7 kg/m2 (95% CI, 0.2-1.3) at 5 months and 0.8 kg/m2 (95% CI, 0.1-1.6) at 11 months’ follow-up assessment. The lifestyle intervention had no significant effect on cardiovascular and metabolic parameters (all nonsignificant). The BMI reduction was only seen in female patients (P = .003).
Conclusions: BMI in patients with bipolar disorder can be reduced with a long-lasting effect by a multimodal lifestyle intervention. However, this effect was only seen in female participants, indicating the need for gender-specific interventions.
Trial Registration: clinicaltrials.gov Identifier: NCT00980863
Prim Care Companion J Clin Psychiatry 2010;12(5):e1-e8
© Copyright 2010 Physicians Postgraduate Press, Inc.
Submitted: October 5, 2009; accepted December 28, 2009.
Published online: September 9, 2010 (doi:10.4088/PCC.09m00906yel).
Corresponding author: Waldemar Greil, MD, Maximilians-Universität M×¼nchen, Department of Psychiatry, Nussbaumstrasse 7, D-80336 Munich ([email protected]).
Bipolar disorders are associated with a substantial risk of weight gain and the development of overweight and obesity, with up to 74% of bipolar patients being overweight or obese.1,2 Weight gain in patients with bipolar disorder is a multifactorial process with interacting biologic, psychological, sociodemographic, and behavior factors.3 Nevertheless, weight gain in bipolar disorder is most likely a secondary phenomenon since the premorbid weight of patients with bipolar disorder is within normal range.4 In particular, mood-stabilizing medication increases body weight, with differential effects of the respective psychotropic agent.5
There are several substantial consequences of weight gain. Next to the experience of considerable discrimination and a reduction in self-esteem, which can lead to further social withdrawal, weight gain affects compliance, which is of utmost importance for maintenance treatment.6,7 Also, weight gain increases the risk of somatic diseases (eg, metabolic syndrome, diabetes mellitus, or coronary heart disease) and mortality.8
Several pharmacologic and nonpharmacologic approaches to prevent and treat weight gain have been applied. The first approach comprises the initial choice of substance or a change in treatment strategy.9 However, adjustment of dose appears to have only little effect,10 and substances for weight reduction, for example, sibutramine and topiramate, effectively reduce weight but are associated with high discontinuation rates11 and need to be evaluated with respect to long-term effects in combination with mood-stabilizing medication.12
Clinical Points
- In patients with bipolar disorder, prevention and treatment of weight gain are essential, since weight gain is a serious adverse reaction to various mood-stabilizing drugs with the risk of metabolic syndrome.
- A lifestyle intervention including motivational support, nutrition counseling, and regular physical activity is feasible and efficacious in reducing body mass index (BMI).
- Since the BMI-reducing effect has been found in women only, a gender-specific approach may be meaningful.
The practicability and efficacy of nonpharmacologic programs, including nutritional counseling, encouragement of physical activity, and modification of behavior, in psychotropic drug-induced weight gain are supported by randomized controlled studies in patients with psychotic disorder.13 These effects encompass preventing weight gain after the initiation of antipsychotic medication14-17 and reducing weight in the context of long-term medication18-21 and after switching to a substance known to have a lower risk of weight gain.22
Since the majority of the relevant studies have been performed in patients with psychotic disorders,13,23 ie, predominantly schizophrenia patients, there is a lack of empirical support for the efficacy and practicability of similar approaches in patients with bipolar disorders.24,25 Therefore, we set out to evaluate the effects of a multimodal lifestyle intervention on weight, body mass index (BMI), and cardiovascular and metabolic parameters in outpatients with bipolar disorder undergoing treatment with mood-stabilizing medication by employing a randomized controlled trial with consecutive assessment of outcome variables over 11 months.
METHOD
Study Design
We conducted a randomized controlled trial over 11 months comparing the effects of a multimodal lifestyle intervention versus standard care. Groups consisted of 2 cohorts (cohort 1: March 2005-February 2006; cohort 2: September 2005-August 2006). Outcome variables were assessed at baseline (preintervention) as well as at 5 (end of intervention) and 11 months (follow-up). Ethics committee approval was obtained, and each participant gave written informed consent. This trial was registered in clinicaltrials.gov (Identifier: NCT00980863).
Participants
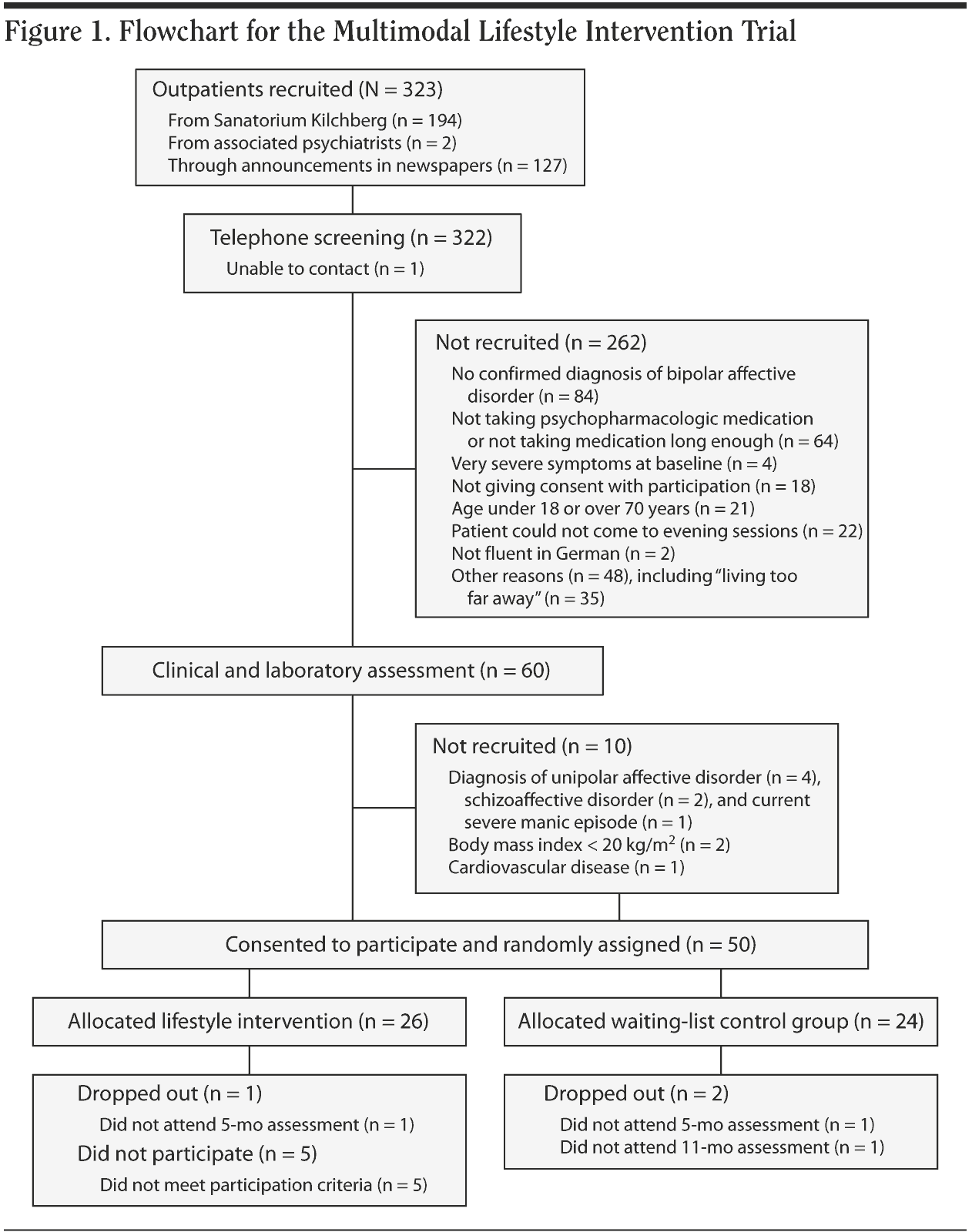
Recruitment began in January and ended in September 2005. Participants were recruited among outpatients of a psychiatric hospital (Sanatorium Kilchberg, Z×¼rich, Switzerland) and associated psychiatrists as well as through study advertisement in local newspapers.
All interested subjects were informed about the study and screened for eligibility in a telephone interview. Eligibility in the telephone interview was defined as stating to have received a diagnosis of bipolar disorder, being in the range of 18-70 years of age, and receiving psychopharmacologic treatment for at least 3 months. Eligible subjects were then invited to a clinical and laboratory examination, which encompassed examination to ensure eligibility and precondition for physical exercise, since the latter was a major component of the intervention. Clinical diagnosis of bipolar disorder was confirmed with the Mini International Neuropsychiatric Interview.26
Adults between 18 and 70 years of age were considered for inclusion if they (1) fulfilled the diagnostic criteria for bipolar disorder according to the DSM-IV-TR27; (2) were on treatment with medication for at least 3 months with 1 of the following substances: lithium, valproic acid, carbamazepine, oxcarbazepine, olanzapine, quetiapine, risperidone, or amisulpride (all have weight-increasing properties); and (3) were not underweight (BMI > 20 kg/m2).
Exclusion criteria were pregnancy; breast feeding; a diagnosis of anorexia nervosa or bulimia nervosa, diabetes type I or II, or another serious physical disease; and use of substances that reduce weight (eg, topiramate). Eligible subjects with comorbidity of acute psychosis, drug addiction, personality disorder, suicidal tendencies, or a current severe manic or depressive episode (even if inpatient treatment was necessary) were excluded only if attending the program were not possible due to the respective disorder.
Lifestyle Intervention
The program “Quality of Life for Persons With Bipolar Disorder” consists of 3 modules, with a total duration of 5 months (Table 1). The program was mainly administered outside the psychiatric setting (eg, gym) to alleviate the transfer into real life of the participants. All modules were free of charge for the patients.
Outcome Variables, Data Collection, and Measurement
At all assessment points, BMI and body weight were assessed and blood samples were obtained. Metabolic syndrome was defined by the National Cholesterol Education Program Adult Treatment Panel III (NCEP-ATP III) criteria28 (3 of the 5 following criteria have to be fulfilled): waist circumference: men > 102 cm, women > 88 cm; triglycerides: ≥ 150 mg/dL; high-density lipoprotein cholesterol: men < 40 mg/dL, women < 50 mg/dL; blood pressure ≥ 130/85 mm Hg; and glycemic index (since for practical reasons the fasting glucose concentration could not be determined in this study, we used HbA1c as a substitute estimation of glycemic status, with a cutoff score of 5.7%).29 Even if the blood pressure was in a normal range at the moment of measurement, patients were regarded to be hypertensive if they were taking long-term antihypertensive medication. To exclude acute infections and diseases of the thyroid gland, C-reactive protein levels of thyroid-stimulating hormone were assessed at baseline. Also, electrocardiograms were performed to screen for cardiovascular problems.
Statistical Analysis
We planned to recruit 60 patients with a diagnosis of bipolar affective disorder to provide 90% power (α = .05) to detect a medium to large multivariate effect size of f2 = 0.25. The actual sample size (N = 50) provided 87% power to detect this effect. We used SPSS 16 statistical software (SPSS Inc, Chicago, Illinois) for Apple OS X for all statistical analyses. Analyses of variance or Pearson χ2 tests were used to examine demographic and clinical variables at baseline. Multivariate time-by-group analysis of variance was used to investigate differences between groups in terms of treatment effects over time. To capture relative effects of the intervention, mean differences in change scores from baseline were calculated and analyzed by univariate analysis of variance. Mean differences in change scores were only considered in case of significant time-by-group interaction effects. Linear regression analysis was used to determine the influence of selected parameters on the primary outcome. Results were considered statistically significant if the P value was less than .05. All analyses were performed according to the principle of intention to treat with last observations carried forward when follow-up data were missing.
Two sets of intention-to-treat analyses were run. First, an analysis of data in all randomly assigned participants (N = 50; treatment group: n = 26, control group: n = 24). Second, an on-treatment analysis of subjects in the intervention group participating in the lifestyle intervention and controls (n = 42; treatment group: n = 20, control group: n = 22). “Not on treatment” was defined as at least 2 modules with more than two-thirds of all possible sessions in the respective module missed.
RESULTS
In total, 323 persons were interested in participation and 322 were screened with semistructured telephone interviews (Figure 1). Sixty people were invited for a clinical and laboratory examination; 10 of those did not fulfill the inclusion criteria and were thus excluded. After having provided written consent, the remaining 50 participants were randomly assigned to the 2 groups. Two participants did not attend the end-of-intervention assessment (intervention group: n = 1, control group: n = 1), and 1 participant (control group) did not attend the 6-month follow-up. In the intervention group, 5 participants attended fewer than one-third of all possible fixed dates in at least 2 modules and were thus regarded as not on treatment.
Characteristics of the Sample
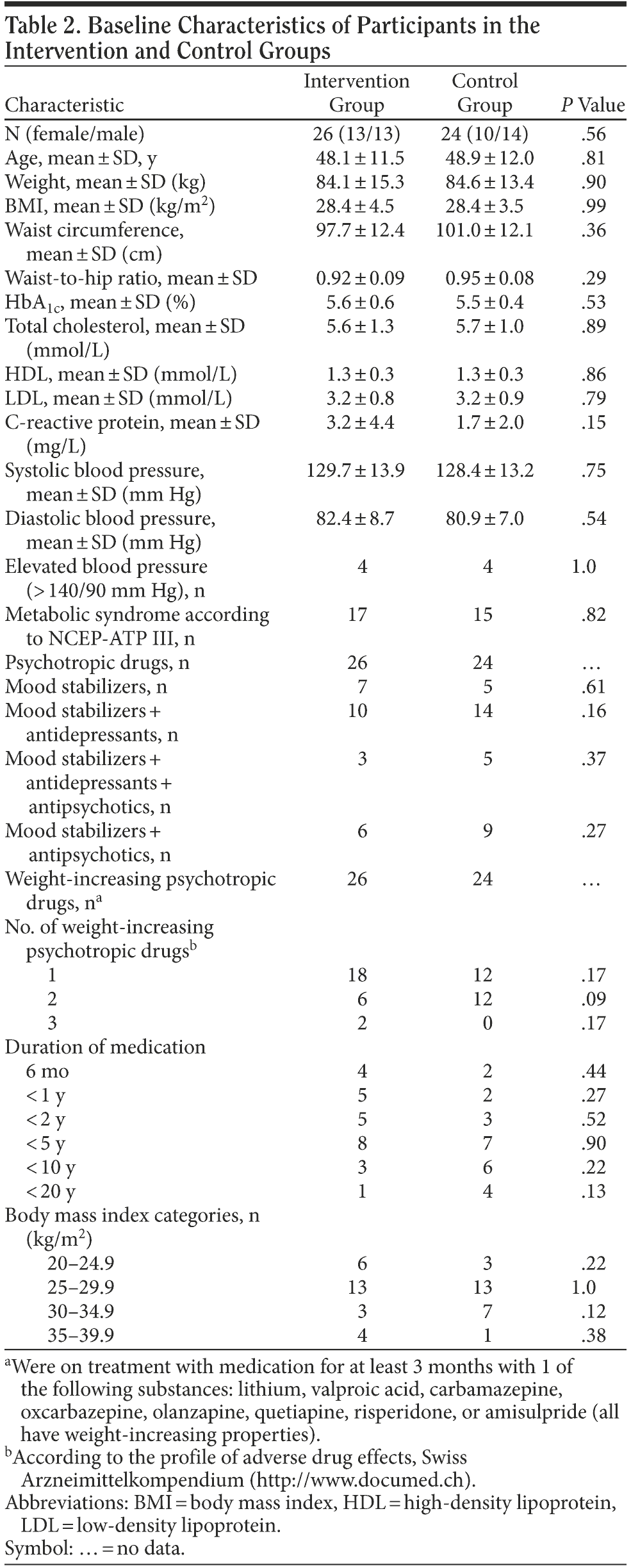
The mean age of participants in the randomly assigned sample was 48 years, with a range from 20 to 65 years. All participants had a diagnosis of bipolar disorder; 35 (70%) were bipolar I (intervention group: n = 20, control group: n = 15), and 15 (30%) fulfilled the criteria of a bipolar II disorder (intervention group: n = 6, control group: n = 9).
All participants were prescribed at least 1 psychotropic drug, and all participants (100%) received weight-increasing drugs. Of the sample, 74% (n = 38) took a combination of mood stabilizers, antidepressants, and/or antipsychotics. On average, the duration of psychopharmacologic medication was 5.5 years (ranging from 6 months to 18 years). During the course of the study, 2 patients changed from a weight-increasing medication to a weight-neutral medication (intervention group: n = 1, control group: n = 1). The majority of the participants (n = 26, 52%) were overweight (25-29.9 kg/m2), 15 participants (30%) were obese (30-39.9 kg/m2), and 9 participants (18%) had a weight within the normal range (20-24.9 kg/m2). Thirty-two patients (64%) fulfilled NCEP-ATP III criteria for metabolic syndrome. Table 2 shows characteristics of the patients at the baseline measurement point.
Effects of Intervention
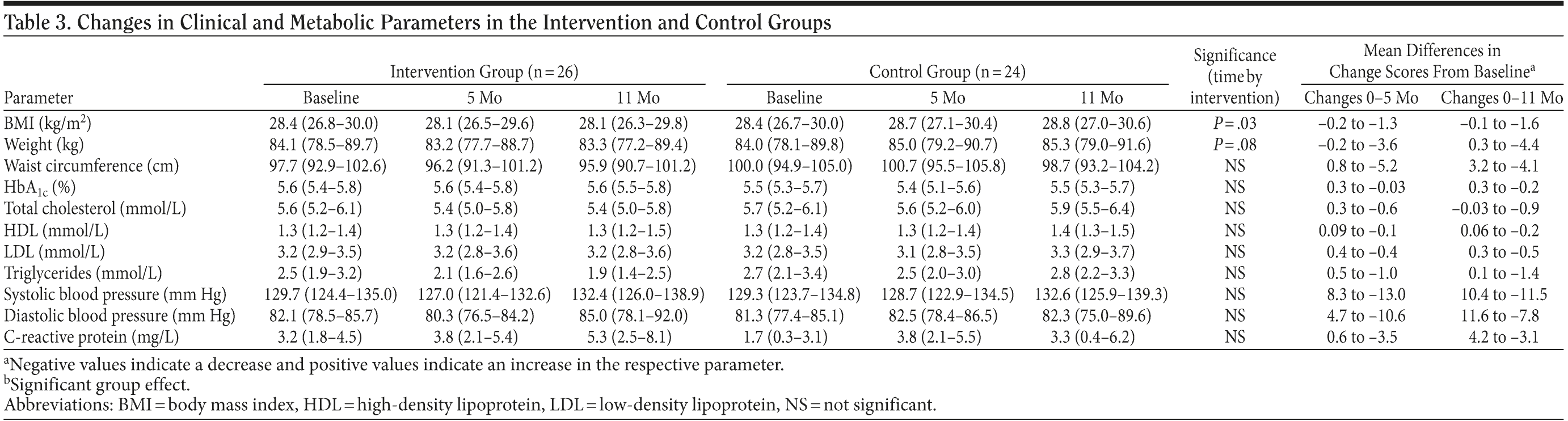
The participation rate in the intervention group, defined as not more than one module with fewer than one-third of all possible sessions in the respective module visited, was 77% (lifestyle: n = 21, 80.8%; nutrition: n = 20, 76.9%; and physical activity: n = 19, 73.1%). Individual overall participation rates were unrelated to any change in reported outcome variables (data not shown) but were significantly associated with age (r = 0.43, P = .03); thus, increasing age was associated with increased participation.
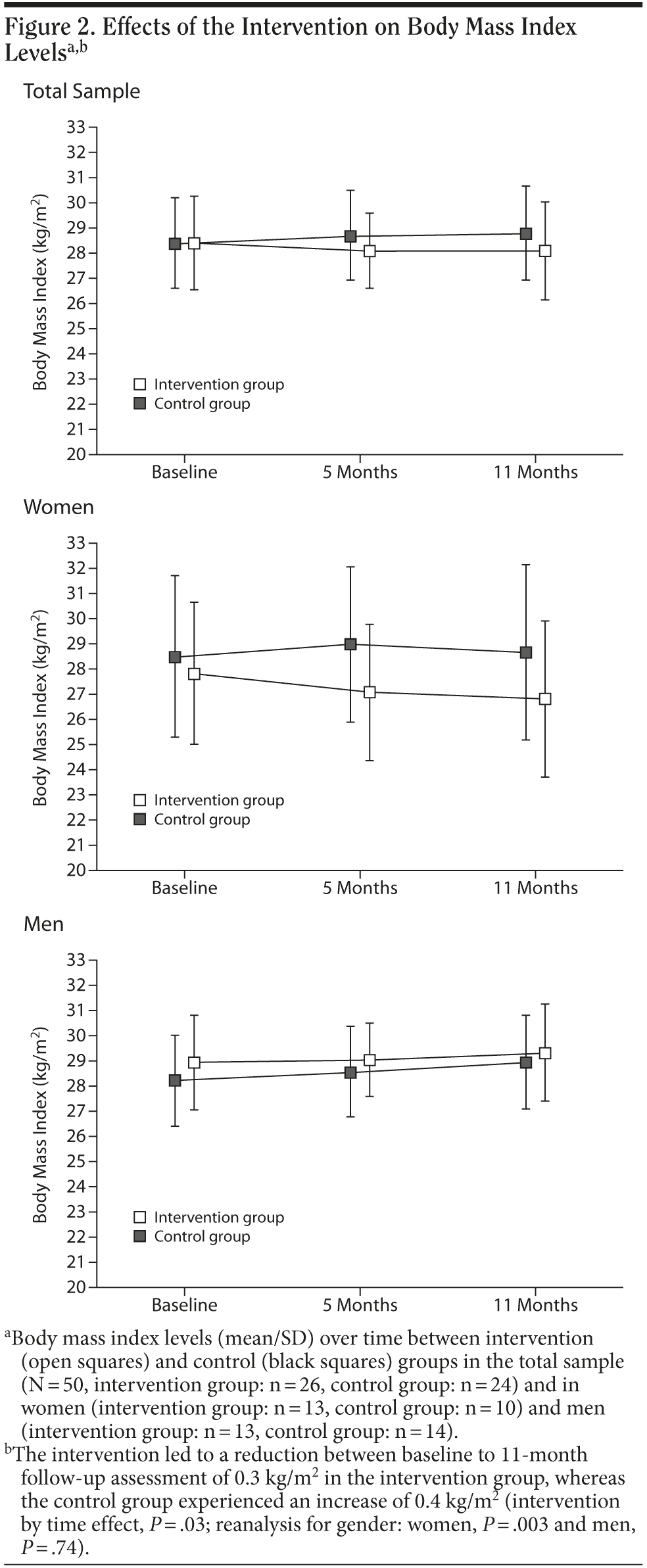
Table 3 shows the effects of the intervention on clinical and metabolic parameters. The intervention had a significant effect on the primary outcome of the study (P = .03), with a mean decrease of 0.3 kg/m2 (95% CI, −0.7 to 0.06) over time in the treatment group, whereas the control group showed an increase of 0.5 kg/m2 (95% CI, 0.01 to 0.8). Mean differences in change scores also differed significantly between groups, with a significant difference in BMI scores between groups of 0.7 and 0.8 kg/m2 at 5 and 11 months, respectively (Table 3). A similar trend was observed in body weight; however, this measure failed to reach statistical significance. With regard to cardiovascular and metabolic parameters, no significant interaction effects were observable. The total number of participants who fulfilled NCEP-ATP III criteria of metabolic syndrome did not differ between groups at any time (not significant, baseline assessment and 5 months and 11 months postassessment: intervention group: n = 17 [65%], n = 16 [62%], and n = 14 [54%]; control group: n = 15 [62%], n = 15 [62%], and n = 16 [67%]).
Exclusion of all subjects defined as not on treatment did not alter these results (data not shown).
Predictors of Effects of Intervention
Stepwise linear regression analysis (included predictors: treatment, baseline demographic data, baseline BMI, medication, and metabolic parameters; criterion: overall change in BMI from baseline to 11-month assessment) indicated that gender and treatment were the only significant predictors of BMI change (adjusted R2 = 0.18, P = .003; included predictors: treatment, β = .36, P = .009 and gender, β = .27, P = .04). Reanalysis of the effects of the randomized treatment for gender separately showed that the effects on BMI were observable in women but not in men (P = .003 and P = .74, respectively, Figure 2). Also, mean BMI differences from baseline differed significantly between groups in women (baseline to 5 months: 1.2 [0.6-1.9], P = .001; baseline to 11 months: 1.2 [0.04-2.3], P = .04) but not in men (baseline to 5 months: 0.3 [−0.5-1.1], P = .54; baseline to 11 months: 0.4 [−0.6-1.4], P = .43).
DISCUSSION
The purpose of this study was to evaluate the effects of a multimodal lifestyle intervention on BMI, body weight, and associated physiologic parameters in patients with bipolar disorders. The results show that the intervention led to a significant and lasting albeit moderate reduction of BMI of 0.7-0.8 kg/m2, approximating 2 kg. However, this effect was observable only in women.
To explain this moderate size of the effect of our intervention, characteristics of our study need to be taken into account. First, and in contrast to other studies, with the exception of weight-neutral lamotrigine as monotherapy, we did not have any restrictions regarding the substance that had been given as a mood stabilizer.18,20,22 Second, existing obesity or a prior distinct weight gain due to a weight-enhancing medication was not a criterion of inclusion. Therefore, the mean BMI at baseline of 28.4 kg/m2 in our study is lower than results reported in other studies (eg, 30.3-31.1,2,18 32.4,2,30 30.4,2,19 332,31). Also, the exclusion of patients with diabetes mellitus might have led to the exclusion of patients with high BMI scores. However, it needs to be noted that BMI and weight were not predictors of treatment outcome in our study.
In the following section, the observed effects will be discussed regarding their clinical significance. In our sample, the relative reduction (ie, mean differences in change scores from baseline) in the intervention group was approximately 2 kg (ie, less then 2.5%). Fontaine et al32 estimated the increase of death in patients with a drug-induced weight gain of only 2.5 kg in 10 years to be 257 per 100,000. Generally, the risk of mortality and morbidity is assumed to decrease significantly with a long-lasting weight reduction of 5%-10%.33 In order to achieve a comparable weight reduction, our intervention probably does not have the required intensity and duration. Successful programs of weight reduction often include nutrition counseling of higher frequency and a low-calorie diet.34 Little long-term effects on weight gain were shown in a recently published study on psychotic disorders.35
The intervention had no significant effect on cardiovascular and metabolic parameters. Since other interventions of similar duration have led to minor changes in these parameters as well,18 it seems plausible that observable and clinically relevant changes in physiologic parameters require a more distinct or enduring change of lifestyle, above all including a change of habits of physical activity.19
In the present study, the effect of a positive change of BMI was seen only in female participants. It may be speculated that this difference is due to social desirability or women showing a higher drive for thinness compared to men.36 In a randomized controlled trial of weight gain in the context of intake of olanzapine by Littrell et al,17 male subjects gained weight more quickly than female subjects. Yet, in another study, although not randomized and controlled, only male participants showed a reduction in weight.37 There is clearly a need for further studies on gender-specific effects of weight-reducing interventions.
The following limitations of our trial need to be considered, all related to the naturalistic design of our study. First, our choice of inclusion and exclusion criteria did not allow us to restrict our sample to overweight or obese subjects. Although baseline BMI was not associated with treatment effects on BMI, we cannot rule out that restricting the inclusion on overweight or obese patients might lead to different, presumably larger, effects. Second, due to the multimodality of our intervention, it is not possible to identify to what extent the single modules contribute to the observed effect. Additionally, pharmacologic treatment encompassed a multitude of different drug classes and substances, and the majority of our participants were taking more than 1 substance, averting the identification of possible specific drug-by-intervention interactions. Psychopharmacologic substances differ in their impact on weight,5 and it is possible that the type of pharmacologic agent has an influence on the effects of nonpharmacologic treatments to reduce weight. Finally, the majority of study participants were on long-term psychopharmacologic treatment of more than 5 years on average. It is possible that this factor could have confined the effects of the lifestyle intervention on weight. The impact of treatment duration on weight and weight reduction should be examined in future studies.
The study has the following strengths. First, the trial specifically addressed outpatients with a specific diagnosis on long-term mood-stabilizing treatment in the context of standard psychiatric care without restrictions of the medication or usual treatment conditions. Second, our randomized controlled trial included a longitudinal assessment of effects, encompassing an assessment of outcome variables 11 months after baseline assessment (ie, 6 months after postassessment). Finally, our intervention based on a manual was highly acceptable to patients as shown by a high participation rate (dropouts: n = 5, 19%).
Our results indicate that multimodal lifestyle interventions are a possible approach to address the important issue of weight and weight gain in patients with bipolar disorders taking medication.3
Drug names: carbamazepine (Carbatrol, Equetrol, and others), lamotrigine (Lamictal and others), lithium (Lithobid and others), olanzapine (Zyprexa), oxcarbazepine (Trileptal and others), quetiapine (Seroquel), risperidone (Risperdal and others), sibutramine (Meridia), topiramate (Topamax and others), valproic acid (Depakene, Stavzor, and others).
Author affiliations: Psychiatrische Privatklinik Sanatorium Kilchberg, Kilchberg (Drs Gillhoff, Tholuck, and Greil), and Clinical Psychology and Psychotherapy, Psychological Institute, University of Z×¼rich (Drs Gaab and Emini), Z×¼rich Switzerland; and Department of Psychiatry, University of Munich, Germany (Dr Greil).
Potential conflict of interest: None reported.
Funding/support: Funded by an unrestricted research grant from Eli Lilly, Geneva, Switzerland, and Migros-Genossenschaft, Z×¼rich, Switzerland, who provided free access to facilities for nutrition (“Klubschule Migros”); both were initiated by Dr Greil.
REFERENCES
1. Fagiolini A, Frank E, Scott JA, et al. Metabolic syndrome in bipolar disorder: findings from the Bipolar Disorder Center for Pennsylvanians. Bipolar Disord. 2005;7(5):424-430.PubMed doi:10.1111/j.1399-5618.2005.00234.x
2. McElroy SL, Frye MA, Suppes T, et al. Correlates of overweight and obesity in 644 patients with bipolar disorder. J Clin Psychiatry. 2002;63(3):207-213. PubMed
3. Wildes JE, Marcus MD, Fagiolini A. Obesity in patients with bipolar disorder: a biopsychosocial-behavioral model. J Clin Psychiatry. 2006;67(6):904-915.PubMed doi:10.4088/JCP.v67n0607
4. Shah A, Shen N, El-Mallakh RS. Weight gain occurs after onset of bipolar illness in overweight bipolar patients. Ann Clin Psychiatry. 2006;18(4):239-241.PubMed doi:10.1080/10401230600948423
5. Torrent C, Amann B, Sסnchez-Moreno J, et al. Weight gain in bipolar disorder: pharmacological treatment as a contributing factor. Acta Psychiatr Scand. 2008;118(1):4-18.PubMed doi:10.1111/j.1600-0447.2008.01204.x
6. Scott J, Pope M. Self-reported adherence to treatment with mood stabilizers, plasma levels, and psychiatric hospitalization. Am J Psychiatry. 2002;159(11):1927-1929.PubMed doi:10.1176/appi.ajp.159.11.1927
7. Weiden PJ, Mackell JA, McDonnell DD. Obesity as a risk factor for antipsychotic noncompliance. Schizophr Res. 2004;66(1):51-57.PubMed doi:10.1016/S0920-9964(02)00498-X
8. Newcomer JW. Antipsychotic medications: metabolic and cardiovascular risk. J Clin Psychiatry. 2007;68(suppl 4):8-13. PubMed
9. Meltzer HY. Putting metabolic side effects into perspective: risks versus benefits of atypical antipsychotics. J Clin Psychiatry. 2001;62(suppl 27):35-39, discussion 40-41. PubMed
10. Faulkner G, Cohn TA. Pharmacologic and nonpharmacologic strategies for weight gain and metabolic disturbance in patients treated with antipsychotic medications. Can J Psychiatry. 2006;51(8):502-511. PubMed
11. McElroy SL, Frye MA, Altshuler LL, et al. A 24-week, randomized, controlled trial of adjunctive sibutramine versus topiramate in the treatment of weight gain in overweight or obese patients with bipolar disorders. Bipolar Disord. 2007;9(4):426-434.PubMed doi:10.1111/j.1399-5618.2007.00488.x
12. Chue P, Kovacs CS. Safety and tolerability of atypical antipsychotics in patients with bipolar disorder: prevalence, monitoring and management. Bipolar Disord. 2003;5(suppl 2):62-79.PubMed doi:10.1111/j.1399-2406.2003.00063.x
13. Alvarez-Jiménez M, Hetrick SE, Gonzסlez-Blanch C, et al. Non-pharmacological management of antipsychotic-induced weight gain: systematic review and meta-analysis of randomised controlled trials. Br J Psychiatry. 2008;193(2):101-107.PubMed doi:10.1192/bjp.bp.107.042853
14. Alvarez-Jiménez M, Gonzסlez-Blanch C, Vסzquez-Barquero JL, et al. Attenuation of antipsychotic-induced weight gain with early behavioral intervention in drug-naive first-episode psychosis patients: a randomized controlled trial. J Clin Psychiatry. 2006;67(8):1253-1260.PubMed doi:10.4088/JCP.v67n0812
15. Scocco P, Longo R, Caon F. Weight change in treatment with olanzapine and a psychoeducational approach. Eat Behav. 2006;7(2):115-124. PubMed
16. Evans S, Newton R, Higgins S. Nutritional intervention to prevent weight gain in patients commenced on olanzapine: a randomized controlled trial. Aust N Z J Psychiatry. 2005;39(6):479-486. PubMed
17. Littrell KH, Hilligoss NM, Kirshner CD, et al. The effects of an educational intervention on antipsychotic-induced weight gain. J Nurs Scholarsh. 2003;35(3):237-241.PubMed doi:10.1111/j.1547-5069.2003.00237.x
18. Mauri M, Simoncini M, Castrogiovanni S, et al. A psychoeducational program for weight loss in patients who have experienced weight gain during antipsychotic treatment with olanzapine. Pharmacopsychiatry. 2008;41(1):17-23.PubMed doi:10.1055/s-2007-992148
19. Wu MK, Wang CK, Bai YM, et al. Outcomes of obese, clozapine-treated inpatients with schizophrenia placed on a six-month diet and physical activity program. Psychiatr Serv. 2007;58(4):544-550.PubMed doi:10.1176/appi.ps.58.4.544
20. Kwon JS, Choi JS, Bahk WM, et al. Weight management program for treatment-emergent weight gain in olanzapine-treated patients with schizophrenia or schizoaffective disorder: a 12-week randomized controlled clinical trial. J Clin Psychiatry. 2006;67(4):547-553.PubMed doi:10.4088/JCP.v67n0405
21. McKibbin CL, Patterson TL, Norman G, et al. A lifestyle intervention for older schizophrenia patients with diabetes mellitus: a randomized controlled trial. Schizophr Res. 2006;86(1-3):36-44.PubMed doi:10.1016/j.schres.2006.05.010
22. Brar JS, Ganguli R, Pandina G, et al. Effects of behavioral therapy on weight loss in overweight and obese patients with schizophrenia or schizoaffective disorder. J Clin Psychiatry. 2005;66(2):205-212.PubMed doi:10.4088/JCP.v66n0208
23. Ganguli R. Behavioral therapy for weight loss in patients with schizophrenia. J Clin Psychiatry. 2007;68(suppl 4):19-25. PubMed
24. Fagiolini A, Kupfer DJ, Houck PR, et al. Obesity as a correlate of outcome in patients with bipolar I disorder. Am J Psychiatry. 2003;160(1):112-117.PubMed doi:10.1176/appi.ajp.160.1.112
25. Vreeland B. Bridging the gap between mental and physical health: a multidisciplinary approach. J Clin Psychiatry. 2007;68(suppl 4):26-33. PubMed
26. Sheehan DV, Lecrubier Y, Sheehan KH, et al. The Mini-International Neuropsychiatric Interview (MINI): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59(suppl 20):22-33, quiz 34-57. PubMed
27. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
28. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA. 2001;285(19):2486-2497.PubMed doi:10.1001/jama.285.19.2486
29. Zhang P, Engelgau MM, Valdez R, et al. Efficient cutoff points for three screening tests for detecting undiagnosed diabetes and pre-diabetes: an economic analysis. Diabetes Care. 2005;28(6):1321-1325.PubMed doi:10.2337/diacare.28.6.1321
30. Khazaal Y, Fresard E, Rabia S, et al. Cognitive behavioural therapy for weight gain associated with antipsychotic drugs. Schizophr Res. 2007;91(1-3):169-177.PubMed doi:10.1016/j.schres.2006.12.025
31. Weber M, Wyne K. A cognitive/behavioral group intervention for weight loss in patients treated with atypical antipsychotics. Schizophr Res. 2006;83(1):95-101.PubMed doi:10.1016/j.schres.2006.01.008
32. Fontaine KR, Heo M, Harrigan EP, et al. Estimating the consequences of anti-psychotic induced weight gain on health and mortality rate. Psychiatry Res. 2001;101(3):277-288.PubMed doi:10.1016/S0165-1781(01)00234-7
33. Manson JE, Skerrett PJ, Greenland P, et al. The escalating pandemics of obesity and sedentary lifestyle: a call to action for clinicians. Arch Intern Med. 2004;164(3):249-258.PubMed doi:10.1001/archinte.164.3.249
34. Mauri M, Castrogiovanni S, Simoncini M, et al. Effects of an educational intervention on weight gain in patients treated with antipsychotics. J Clin Psychopharmacol. 2006;26(5):462-466.PubMed doi:10.1097/01.jcp.0000237943.57893.e6
35. Alvarez-Jiménez M, Mart×nez-Garc×a O, Pérez-Iglesias R, et al. Prevention of antipsychotic-induced weight gain with early behavioural intervention in first-episode psychosis: 2-year results of a randomized controlled trial. Schizophr Res. 2010;116(1):16-19. PubMed doi:10.1016/j.schres.2009.10.012
36. Lewinsohn PM, Seeley JR, Moerk KC, et al. Gender differences in eating disorder symptoms in young adults. Int J Eat Disord. 2002;32(4):426-440.PubMed doi:10.1002/eat.10103
37. Ball MP, Coons VB, Buchanan RW. A program for treating olanzapine-related weight gain. Psychiatr Serv. 2001;52(7):967-969.PubMed doi:10.1176/appi.ps.52.7.967