Dietary Restrictions and Drug Interactions With Monoamine Oxidase Inhibitors: An Update
Monoamine oxidase inhibitors (MAOIs) are effective treatments for depression that has atypical features or that has failed to respond to other antidepressants. However, MAOIs are underused because clinicians are concerned about dietary and drug interactions with this class of medication. Hypertensive crisis and serotonin syndrome can occur in rare cases due to interactions between MAOIs and foods containing tyramine as well as interactions with serotonergic and sympathomimetic agents. A better understanding of the foods and drugs that can cause adverse reactions, as well as knowledge of newer MAOIs with mechanisms of action and delivery methods that reduce these risks, may help clinicians to consider the use of these medications, when appropriate, in their patients with depression.
(J Clin Psychiatry 2012;73[suppl 1]:17-24)
From the Indiana Institute for Personalized Medicine and the Division of Clinical Pharmacology, Indiana University School of Medicine, Indianapolis.
This article is derived from the planning teleconference series “A Fresh Look at Monoamine Oxidase Inhibitors for Depression,” which was held December 2011 through February 2012 and supported by an educational grant from Mylan Specialty L.P. (formerly known as Dey Pharma, L.P.).
Dr Flockhart has no personal affiliations or financial relationships with any commercial interest to disclose relative to this article.
Corresponding author: David A. Flockhart, MD, PhD, Indiana University School of Medicine, Wishard Memorial Hospital, 1001 West 10th St, Indianapolis, IN 46202 ([email protected]).
doi:10.4088/JCP.11096su1c.03
© Copyright 2012 Physicians Postgraduate Press, Inc.
Monoamine oxidase inhibitors (MAOIs) are effective antidepressants, but their use has been limited by potentially fatal food and drug interactions. A clearer understanding of these interactions and the mechanism of action of these drugs, as well as knowledge of new formulations and delivery methods of MAOIs, could help clinicians make more use of this class of antidepressant.
The MAOIs are effective for depression with atypical features1 and depression that is treatment resistant.2 Although indicated by some guidelines3 to treat these conditions, MAOIs are still underused.4
A main reason for reluctance to use MAOIs is due to the dietary and drug restrictions, which are required because food and drug interactions can cause serious and potentially fatal adverse events, such as hypertensive crisis and serotonin syndrome. Newer formulations of MAOIs that have mechanisms of action different from those of older MAOIs may lessen the risk for these adverse effects and make this class of antidepressants worth reconsidering for some patients with depression.
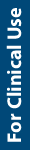
- Dietary restrictions are required with older, irreversible MAOIs and are lessened with low dosages of selective MAO-B inhibitors and with transdermal formulations.
- Drug interactions can occur with all MAOIs.
- Washout periods should be carefully observed when switching between an MAOI and a serotonergic agent.
- Counsel patients to inform other physicians that they are taking an MAOI and to carefully select over-the-counter drugs.
Mechanism of Action of MAOIs
The propensity for food and drug interactions of MAOIs is related to their mechanisms of action. The MAOIs inhibit monoamine oxidase (MAO), an enzyme that catalyzes the oxidative removal of monoamines, including serotonin, histamine, and the catecholamines dopamine, norepinephrine, and epinephrine, as well as trace amines.5 By covalently bonding to MAO enzymes, MAOIs prevent the removal of these amines, resulting in their increased synaptic availability. Some MAOIs bind to the enzyme for its lifetime (a duration of 2 to 4 weeks) and are, thus, classified as irreversible; reversible MAOIs can be displaced from the enzyme.
Two isoenzymes of MAO, MAO-A and MAO-B, are distributed unevenly throughout the body and brain. Monoamine oxidase-A predominates in the brain, gut, liver, placenta, and skin, while MAO-B predominates in the brain, platelets, and lymphocytes.4
Monoamine oxidase inhibitors can selectively inhibit either MAO-A or MAO-B enzymes, or they can be nonselective and inhibit both isomers; however, the inhibition of MAO-A appears to be necessary for an antidepressant effect to occur.6,7 The older MAOIs, isocarboxazid, phenelzine, and tranylcypromine, are nonselective and irreversible. Oral selegiline, which is also irreversible, is selective for MAO-B at low doses but loses its selectivity at higher doses, meaning that the higher doses are required to induce an antidepressant effect. The newer, transdermal formulation of selegiline can produce an antidepressant effect at low doses because it avoids first-pass metabolism in the liver and intestine. Moclobemide, a newer reversible MAOI, is selective for MAO-A but is currently unavailable in the United States.
Dietary Interactions With MAOIs
Tyramine and Hypertensive Crisis
Tyramine is a trace vasoactive amine that occurs naturally in a range of foods and in the gut. Usually, tyramine is metabolized by MAO-A in the intestinal wall and then in the liver. This process destroys excess tyramine before it can be absorbed and enter systemic circulation, where it is converted into synaptic norepineprine.4 In the synapse, excess norepinephrine is also destroyed by MAO-A.
When an MAOI irreversibly binds to MAO-A, the degradation of tyramine in the digestive system is prevented, allowing an excess of tyramine to enter the bloodstream and to be metabolized into norepinephrine. Norepinephrine stimulates postsynaptic α1– and other adrenergic receptors and the cardiovascular sympathetic nervous system and constricts the blood vessels.4 If too much tyramine is ingested, excess norepinephrine can lead to a rapid increase in blood pressure.
When blood pressure increase is large and sudden, a hypertensive crisis can occur. Hypertensive crisis is defined as having a systolic blood pressure greater than 180 mm Hg or a diastolic blood pressure greater than 120 mm Hg.8 This can cause permanent damage to bodily organs, stroke, aneurysm, and, potentially (although rarely), death. Hypertensive crises can be classified as hypertensive emergencies, in which patients have severe hypertension and acute end-organ damage, or hypertensive urgencies, in which patients have severe hypertension with no or minimal end-organ damage.8 Symptoms of a potentially fatal reaction include occipital headache that may radiate frontally, palpitations, neck stiffness or soreness, nausea, vomiting, sweating (sometimes with fever), dilated pupils, photophobia, and tachycardia or bradycardia associated with constricting chest pain.4
The first hypertensive crisis reported in a patient taking an MAOI was an adverse reaction to cheese.9 This was followed by reports of reactions to a wide range of other foods, then by attempts to assess the risks with particular food items.10,11
Foods Containing Tyramine and Other Vasoactive Amines
Tyramine. To reduce the risk of hypertensive crisis, patients taking irreversible MAOIs are required to restrict their intake of tyramine-rich foods and foods containing other vasoactive amines (Table 1).10,12,13 The average person can digest 200 mg to 800 mg of tyramine before experiencing an increase in blood pressure.11 However, a person who is taking an irreversible MAOI, in which MAO-A has compromised tyramine metabolization, can experience a mild reaction after ingesting only 6 mg to 10 mg of tyramine and a severe reaction with 10 mg to 25 mg.11 Clinicians should remember that individual response to tyramine and the levels in particular categories of food vary.10
Levels of tyramine that are too high (> 6 mg/serving) for people taking MAOIs are found in foods such as aged cheeses and meats, draft beer, soy products, and some yeast extracts, as well as spoiled foods (see Table 1).10 An active tyrosine decarboxylase (the action of which forms tyramine) is used in making, or is found in, many cheeses or fermented foods due to microbial contamination encountered during aging and storage.11 In general, fresher food is safer than food that has been stored.
Although some foods are restricted with MAOIs, many foods are allowed. Over time, closer examination of foods thought to have high tyramine levels has shown this not to be the case, in some instances. For example, avocados, banana pulp, raspberries, and chocolate were found to contain little or no tyramine, as were Chianti and many other alcoholic beverages.14,15 In addition, some reports of adverse interactions (eg, with liver and pickled herring) may have been due to spoilage of the food.14 Walker and colleagues15 examined the effect of storage on tyramine levels in some foods and confirmed that, while improper storage raised tyramine concentrations in chicken livers, improper or prolonged storage of raspberries, mozzarella cheese, and bananas did not increase tyramine to an unsafe level. More recently, tyramine levels in pizza and soybean products have been analyzed.16 While most commercial chain pizzas were found to have low amounts of tyramine (even those with double pepperoni and double cheese), high levels of tyramine were found in some soy products such as tofu and soy sauce.
Increased information about tyramine content in food has made adhering to a tyramine-restricted diet easier. Gardner et al10 used previous research and tyramine assays to provide a simplified “user friendly diet” that clarified the risk level of particular foods and beverages. The aim of this diet was to make the information about dietary restrictions easier to follow and adhere to. Continued improvements in food packaging and storage have also contributed to a safer food supply.11
In addition, the mechanism of action or delivery method of newer MAOIs, such as moclobemide and transdermal selegiline, may reduce the risk of hypertensive crisis with tyramine ingestion and, thus, lessen the need for diet modification. Reversible inhibitors of MAO-A (RIMAs) like moclobemide can be displaced from MAO-A and do not inhibit tyramine breakdown during digestion; therefore, dietary modifications are not needed with doses less than 900 mg/d of this medication.17 At the lowest dosage (6 mg/24 h), transdermal selegiline also has less need for dietary modification because it is not delivered directly into the liver and intestine like oral medications.
Other vasoactive amines. Pressor agents other than tyramine can be present in food.15 Vasogenic amines also include phenylethylamine and tryptamine.11 Histamine is a biogenic amine that is often found in foods, and excess intake can lead to hypertension.18 Dopamine may also contribute to hypertension via food interactions, although evidence is uncertain.19 For example, levodopa, rather than tyramine, may be responsible for the hypertensive risk that can occur when broad (fava) bean pods are ingested in those taking an MAOI.20
Drug Interactions With MAOIs
In addition to reactions with foods, both older and newer MAOIs can negatively interact with sympathomimetic and serotonergic agents (Table 2).12,13,21-25 Because of these possible interactions, agents that inhibit serotonin or norepinephrine reuptake (eg, selective serotonin reuptake inhibitors [SSRIs] and serotonin-norepinephrine reuptake inhibitors [SNRIs]) should not be combined with MAOIs, nor should agents that boost norepinephrine. To avoid mistakenly combining contraindicated medications, clinicians should always tell patients to inform their other physicians about their MAOI regimen. Further, patients should be counseled to check over-the-counter products for sympathomimetic agents.
The most serious, but rare, drug interactions that can result from the combination of sympathomimetic agents or serotonergic agents with MAOIs are hypertensive crisis and serotonin syndrome.
Sympathomimetic Agents and Hypertensive Crisis
Sympathomimetic agents imitate the effects of sympathetic nervous system neurotransmitters, such as norepinephrine. Sympathomimetic drugs can act directly or indirectly. Direct-acting sympathomimetics block the breakdown and reuptake of catecholamines and stimulate adrenergic and dopaminergic receptors. Indirect-acting sympathomimetics provoke the production and release of catecholamines and block norepinephrine transporter activity. The resulting effects of direct and indirect sympathomimetic action include increased cardiac output, vasoconstriction, regulation of body temperature, bronchial dilation, and relaxation of intestinal smooth muscle.
Sympathomimetic drugs can be used in the short-term to treat conditions such as hypotension, myocardial infarction, or congestion. These agents range from stimulants to illicit drugs to over-the-counter medications containing vasoconstrictors, like some cough and cold remedies and weight-reducing preparations (see Table 2).6
When coadministered with MAOIs, sympathomimetics that boost adrenergic stimulation by a mechanism other than MAO inhibition can dangerously elevate blood pressure, causing a hypertensive crisis.4 For example, when the decongestant phenylephrine, which constricts nasal blood vessels and directly stimulates α1-adrenergic postsynaptic vascular receptors, is combined with the pronoradrenergic actions of MAOIs, blood pressure can rise and cause hypertensive crisis in some patients.4
Other adrenergic stimulants that should be avoided include methylphenidate and amphetamine, which are used to treat attention-deficit/hyperactivity disorder (ADHD). Methylphenidate blocks the reuptake of norepinephrine, and amphetamine inhibits the reuptake of and also releases norepinephrine.4,26 Other treatments that block norepinephrine reuptake include some other antidepressants (eg, SNRIs), appetite suppressants, and the analgesic tramadol.4
Tryptophan. Tryptophan is converted into serotonin in the brain by tryptophan hydroxylase and decarboxylase.26 As a dietary supplement, tryptophan (6 to 18 mg/d) is contraindicated with MAOIs because it is a sympathomimetic compound.
Levodopa. Levodopa is also a contraindicated sympathomimetic compound, although oral selegiline is approved by the US Food and Drug Administration (FDA) as an adjunct to levodopa/carbidopa for Parkinson’s disease.21
Serotonergic Agents and Serotonin Syndrome
Part of the antidepressant effect of MAOIs includes inhibiting the MAO enzymes that deactivate serotonin, which in turn, enhances the synaptic availability of serotonin.26Agents that enhance serotonin’s effects, such as antidepressants that inhibit serotonin reuptake or amphetamines that release serotonin, should be avoided in combination with MAOIs because an accumulation of too much serotonin will result in serotonin syndrome, which can be fatal.
Serotonin syndrome is characterized by autonomic, neuromotor, and cognitive-behavioral symptoms.27,28 The most frequently reported symptoms are changes in mental status such as confusion and hypomania, restlessness, myoclonus, hyperreflexia, diaphoresis, shivering, and tremor. Diarrhea, incoordination, and fever can also occur. Severe hyperthermia with complications such as disseminated intravascular coagulation, rhabdomyolysis, and renal failure can sometimes precede death. Clinicians should be aware that serotonin syndrome has overlapping signs and symptoms with several other hyperthermic states or syndromes such as lethal catatonia, anticholinergic toxicity, malignant hyperthermia, and neuroleptic malignant syndrome.28,29
According to the Hunter Serotonin Toxicity Criteria,28 serotonin syndrome is present if a serotonergic agent has been taken and 1 of the following conditions is met:
- Spontaneous clonus
- Inducible clonus and agitation or diaphoresis
- Ocular clonus and agitation or diaphoresis
- Tremor and hyperreflexia
- Hypertonia, temperature greater than 38°C, and ocular clonus or inducible clonus.
In most cases, the syndrome resolves within 24 hours when the agents are discontinued.29 In severe cases, intensive care observation and treatment is necessary.
When prescribing MAOIs and advising patients about drug interactions, clinicians should remember that patients with several common medical conditions are at particular risk for serotonin syndrome because they may have a reduced ability to metabolize serotonin.29 These conditions include damaged vascular or pulmonary endothelium, atherosclerosis, hypertension, and hypercholesterolemia.
Serotonin syndrome can occur with many MAOI drug combinations, and new contraindications are occasionally added (see Table 2). The most toxic combination is an irreversible MAOI with another serotonergic agent.27,30 A relatively recent addition to the list of agents that can cause serotonin syndrome when combined with MAOIs is the antibiotic linezolid, which inhibits both MAO-A and MAO-B.4,24 The syndrome has also been reported between the newer MAOIs selegiline31,32 and moclobemide33 with other serotonergic agents. Although dietary restrictions are lessened with low dosages of transdermal selegiline and moclobemide, the drug interaction precautions remain in effect, as with all other MAOIs.
Because of the serious nature of serotonin syndrome, clinicians must prevent unnecessary excess serotonergic activity. When switching between serotonergic agents, clinicians should observe current manufacturers’ package insert recommendations. Generally, a washout period of at least 14 days is required after discontinuing an MAOI and before beginning a serotonergic agent, and vice versa.34 Because fluoxetine has a particularly long dynamic half-life,35 a washout period of more than 5 weeks may be needed.27
Efficacy and Safety of Newer MAOIs
MAOIs with varying mechanisms of action and methods of delivery have become available relatively recently. These medications are efficacious for depression or Parkinson’s disease.
Selegiline/l-Deprenyl
Oral and transdermal selegiline formulations are selective inhibitors of MAO-B at low doses. However, at higher doses (> 20 mg/d for oral selegiline and ≥ 9 mg/24 h for transdermal selegiline), selectivity is lost and both MAO-A and MAO-B are inhibited.
Oral selegiline. The oral formulation of selegiline is indicated for adjunctive use with levodopa/carbidopa for treatment of Parkinson’s disease,21 but is not indicated for the treatment of depression. Patients with Parkinson’s disease treated with this combination over 9 years showed better survival rates than those treated with l-dopa alone.36
The package insert21 warns that, even at the low dose of 10 mg/d, selectivity for MAO-B may not be complete and rare cases of hypertensive reaction associated with tyramine ingestion have been reported. Adverse reactions in patients treated with oral selegiline and amitriptyline, protriptyline, tricyclic antidepressants (TCAs), and SSRIs have also been reported. With oral selegiline, caution regarding diet should be given and a 14-day washout period for TCAs and SSRIs is advised because the mechanism of action of these combinations is not fully understood. In a small study (N = 8), the bioavailability of oral selegiline was increased 10- to 20-fold by the concomitant use of oral contraceptives and, thus, increased the risk of food and drug interactions.25 This finding also has implications for patients taking hormone replacement therapy, which contains female sex steroids.
Transdermal selegiline. Transdermal selegiline is indicated for treatment of major depressive disorder (MDD). It is delivered via a skin patch and is available in doses of 6, 9, and 12 mg/24 hours. Several double-blind, placebo-controlled studies have established the efficacy of selegiline for MDD.37-40
Bodkin and Amsterdam37 studied 177 outpatients with mostly moderate MDD who were treated with a 6 mg/24 hour dose (delivered by a 20 mg/20 cm2 patch) of selegiline for 6 weeks. Participants observed a tyramine-restricted diet. The selegiline patch produced greater improvement versus placebo at endpoint according to the Hamilton Depression Rating Scale-17 (HDRS17), the Hamilton Depression Rating Scale-28 (HDRS28), and the Montgomery-Asberg Depression Rating Scale (MADRS), with improvement occurring as early as week 1.
A larger study38 of outpatients with MDD (N = 365) over up to 8 weeks found a modest effect size for 6 mg/24 hour selegiline versus placebo. Ratings were statistically significant favoring selegiline on MADRS (P = .001) and HDRS28 (P = .039) scores, but nonsignificantly better on the HDRS17 and Clinical Global Impressions-Severity (CGI-S) ratings. Patients were not advised to adhere to a tyramine-restricted diet, and no significant differences in blood pressure occurred.
A long-term study39 of patients who had responded to 10 weeks of treatment with a 6 mg/24 hour patch found that, at 12 months, relapse rates were significantly lower with transdermal selegiline (16.8%) than placebo (30.7%) and time to relapse was significantly longer with the active agent than placebo (P = .0048).
A flexible-dose trial40 of the selegiline patch allowed use of the 2 higher doses if response was insufficient in 265 patients with MDD over 8 weeks. Significantly greater improvement compared with placebo was found on the HDRS28, MADRS, and Inventory for Depressive Symptomatology-Self Rated (IDS-SR) scales (P ≤ .05). No dietary restrictions were imposed, and no hypertensive crises resulted.
Moclobemide
Moclobemide is a RIMA that is available only outside the United States. Because moclobemide reversibly inhibits MAO-A, the need for dietary restrictions and the risk for hypertensive crisis are substantially lessened with this MAOI.
Meta-analyses17,41-43 of studies in patients with depression have found moclobemide to be more effective than placebo and to be comparable to TCAs and SSRIs.17 Moclobemide has a short half-life and, therefore, the drug is washed out fairly quickly after discontinuation.17
Rasagiline
Rasagiline is a selective inhibitor of MAO-B at recommended doses of 0.5 or 1 mg/d. Selectivity is lost progressively as dosage increases. This medication is indicated for initial monotherapy and adjunctive therapy with levodopa in patients with Parkinson’s disease,44 but is not indicated for depression.
For outpatients with Parkinson’s disease (N = 687), adjunctive rasagiline was similar to entacapone and better than placebo in reducing daily off-time and increasing daily on-time, and significantly improved CGI scores versus entacapone and placebo (P < .0001 and P = .0002, respectively).45
Rasagiline carries warnings for serotonin syndrome and hypertensive crisis. Foods containing high levels of tyramine and contraindicated drugs should clearly be avoided (see Tables 1 and 2).
Future Directions: Personalized Treatment for Depression
A large armamentarium of medications is available to treat depression, including MAOIs. One goal for the future in medicine is to be able to predict which patients are most likely to respond to particular agents, that is, personalizing therapy. In the past, when the armamentarium of depression treatments was beginning to grow, one way to personalize therapy was to determine the subtype of depression; for patients with atypical depression, MAOIs were believed to have superior efficacy.46 Better methods for treatment selection that incorporate pharmacogenetics may eventually become available.
Developing a predictive panel of pharmacogenetic biomarkers may eventually be able to guide the stratification of depression therapy for particular patients, which should improve their treatment outcomes and quality of life. A single, first genome-wide association study,47 which was based on samples from 1,948 participants in the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) study, has shown nonsignificant results. The study did identify single nucleotide polymorphisms associated with response and remission that deserve further research. Further, although the number of pharmacogenetic tests for cancer has grown in recent years, the same cannot be said for neuropsychiatric disorders.48,49 Genetic testing may, however, eventually be available to inform treatment selection in patients with depression.
Conclusions
Monoamine oxidase inhibitors are effective treatments for depression, but due to dietary restrictions and drug interactions, they are relatively underused. Despite the restriction of dietary tyramine, many foods are permitted with MAOIs, and some foods once thought to contain tyramine have been discovered to have no or little tyramine. Additionally, the need for a tyramine-restricted diet depends on the MAOI and its dosage. For example, transdermal delivery and MAO reversibility may avoid the need for dietary modifications. Although some MAOIs reduce the risk of dietary interactions, all MAOIs can have serious drug interactions with sympathomimetics, serotonergic agents, and other medications, potentially leading to hypertensive crisis or serotonin syndrome. In the future, a greater understanding of pharmacogenomics may assist clinicians in personalizing depression therapy and selecting the most effective antidepressant for individual patients.
Drug names: bupropion (Wellbutrin, Aplenzin, and others), carbamazepine (Carbatrol, Equetro, and others), citalopram (Celexa and others), clomipramine (Anafranil and others), cyclobenzaprine (Amrix, Flexeril, and others), desipramine (Norpramin and others), disulfiram (Anatabuse and others), doxepin (Zonalon and others), duloxetine (Cymbalta), entacapone (Comtan), fentanyl (Duragesic, Subsys, and others), fluoxetine (Prozac and others), fluvoxamine (Luvox and others), imipramine (Tofranil and others), isocarboxazid (Marplan), levodopa/carbidopa (Parcopa, Sinemet, and others), linezolid (Zyvox), meperidine (Demerol and others), methadone (Methadose and others), methylphenidate (Focalin, Daytrana, and others), milnacipran (Savella), mirtazapine (Remeron and others), morphine (Avinza, Kadian, and others), nortriptyline (Pamelor, Aventyl, and others), oxcarbazepine (Trileptal and others), paroxetine (Paxil, Pexeva, and others), pentazocine (Talwin), phenelzine (Nardil and others), procarbazine (Matulane), protriptyline (Vivactil and others), rasagiline (Azilect), reserpine (Serplan and others), selegiline oral formulation (Eldepryl, Zelapar, and others), selegiline transdermal system (EMSAM), sertraline (Zoloft and others), tramadol (Ryzolt, Ultram, and others), tranylcypromine (Parnate and others), trimipramine (Surmontil and others), venlafaxine (Effexor and others).
Disclosure of off-label usage: The author has determined that, to the best of his knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
References
1. Henkel V, Mergl R, Allgaier AK, et al. Treatment of depression with atypical features: a meta-analytic approach. Psychiatry Res. 2006;141(1):89-101. PubMed doi:10.1016/j.psychres.2005.07.012
2. Amsterdam JD, Shults J. MAOI efficacy and safety in advanced stage treatment-resistant depression: a retrospective study. J Affect Disord. 2005;89(1-3):183-188. PubMed doi:10.1016/j.jad.2005.06.011
3. American Psychiatric Association. Practice Guideline for the Treatment of Patients With Major Depressive Disorder, Third Edition. Washington, DC: American Psychiatric Association; 2010. http://psychiatryonline.org/content.aspx?bookid=28§ionid=1667485. Accessed May 17, 2012.
4. Stahl SM, Felker A. Monoamine oxidase inhibitors: a modern guide to an unrequited class of antidepressants. CNS Spectr. 2008;13(10):855-870. PubMed
5. Youdim MBH, Edmondson D, Tipton KF. The therapeutic potential of monoamine oxidase inhibitors. Nat Rev Neurosci. 2006;7(4):295-309. PubMed doi:10.1038/nrn1883
6. Wimbiscus M, Kostenko O, Malone D. MAO inhibitors: risks, benefits, and lore. Cleve Clin J Med. 2010;77(12):859-882. PubMed doi:10.3949/ccjm.77a.09103
7. VanDenBerg CM. The transdermal delivery system of monoamine oxidase inhibitors. J Clin Psychiatry. 2012;73(suppl 1):25-30.
8. Rodriguez MA, Kumar SK, De Caro M. Hypertensive crisis. Cardiol Rev. 2010;18(2):102-107. PubMed doi:10.1097/CRD.0b013e3181c307b7
9. Blackwell B, Marley E, Price J, et al. Hypertensive interactions between monoamine oxidase inhibitors and foodstuffs. Br J Psychiatry. 1967;113(497):349-365. PubMed doi:10.1192/bjp.113.497.349
10. Gardner DM, Shulman KI, Walker SE, et al. The making of a user friendly MAOI diet. J Clin Psychiatry. 1996;57(3):99-104. PubMed
11. McCabe-Sellers BJ, Staggs CG, Bogle ML. Tyramine in foods and monoamine oxidase inhibitor drugs: a crossroad where medicine, nutrition, pharmacy, and food industry converge. J Food Compost Anal. 2006;19(suppl):S58-S65. doi:10.1016/j.jfca.2005.12.008
12. EMSAM (selegiline patch) [package insert]. Napa, CA: Dey Pharma LP; 2010. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=b891bd9f-fdb8-4862-89c5-ecdd700398a3. Accessed May 17, 2012.
13. Nardil (phenelzine sulfate) [package insert]. New York, NY: Pfizer, Inc; 2011. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=513a41d0-37d4-4355-8a6d-a2c643bce6fa. Accessed May 17, 2012.
14. Shulman KI, Walker SE, MacKenzie S, et al. Dietary restriction, tyramine, and the use of monoamine oxidase inhibitors. J Clin Psychopharmacol. 1989;9(6):397-402. PubMed doi:10.1097/00004714-198912000-00002
15. Walker SE, Shulman KI, Tailor SA, et al. Tyramine content of previously restricted foods in monoamine oxidase inhibitor diets. J Clin Psychopharmacol. 1996;16(5):383-388. PubMed doi:10.1097/00004714-199610000-00007
16. Shulman KI, Walker SE. Refining the MAOI diet: tyramine content of pizzas and soy products. J Clin Psychiatry. 1999;60(3):191-193. PubMed doi:10.4088/JCP.v60n0308
17. Bonnet U. Moclobemide: therapeutic use and clinical studies. CNS Drug Rev. 2003;9(1):97-140. PubMed doi:10.1111/j.1527-3458.2003.tb00245.x
18. Naila A, Flint S, Fletcher G, et al. Control of biogenic amines in food: existing and emerging approaches. J Food Sci. 2010;75(7):R139-R150. PubMed doi:10.1111/j.1750-3841.2010.01774.x
19. Gillman PK. Advances pertaining to the pharmacology and interactions of irreversible nonselective monoamine oxidase inhibitors. J Clin Psychopharmacol. 2011;31(1):66-74. PubMed doi:10.1097/JCP.0b013e31820469ea
20. Jefferson JW. Who put the tyramine in Mrs. Murphy’s fava bean [letter]? J Clin Psychiatry. 2008;69(8):1336-1337. PubMed doi:10.4088/JCP.v69n0819b
21. Eldepryl (selegiline) [package insert]. Napa, CA: Dey Pharma LP; 2011. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=106429ad-859a-4b29-babf-42cb85f7236e. Accessed May 17, 2012.
22. Marplan (isocarboxazid) [package insert]. Parsippany, NJ: Validus Pharmaceuticals, Inc; 2012. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=ac387aa0-3f04-4865-a913-db6ed6f4fdc5. Accessed May 17, 2012.
23. Parnate (tranylcypromine) [package insert]. Research Triangle Park, NC: GlaxoSmithKline LLC; 2012. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=b9f8ba41-3f86-4633-3e8e-fcf1cffa9756. Accessed May 17, 2012.
24. Lawrence KR, Adra M, Gillman PK. Serotonin toxicity associated with the use of linezolid: a review of postmarketing data. Clin Infect Dis. 2006;42(11):1578-1583. PubMed doi:10.1086/503839
25. Laine K, Anttila M, Helminen A, et al. Dose linearity study of selegiline pharmacokinetics after oral administration: evidence for strong drug interaction with female sex steroids. Br J Clin Pharmacol. 1999;47(3):249-254. PubMed doi:10.1046/j.1365-2125.1999.00891.x
26. Stahl MS. Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications. 3rd ed. New York, NY: Cambridge University Press; 2008.
27. Sternbach H. The serotonin syndrome. Am J Psychiatry. 1991;148(6):705-713. PubMed
28. Dunkley EJ, Isbister GK, Sibbritt D, et al. The Hunter Serotonin Toxicity Criteria: simple and accurate diagnostic decision rules for serotonin toxicity. QJM. 2003;96(9):635-642. PubMed doi:10.1093/qjmed/hcg109
29. Sternback H. Serotonin syndrome: how to avoid, identify, and treat. J Fam Pract Online. 2003;2(5). http://www.jfponline.com/Pages.asp?AID=636#2. Accessed May 16, 2012.
30. Rapaport MH. Dietary restrictions and drug interactions with monoamine oxidase inhibitors: the state of the art. J Clin Psychiatry. 2007;68(suppl 8):42-46. PubMed
31. Robinson DS. Transdermal selegiline: a new-generation MAOI. Prim Psychiatry. 2006;13(5):33-35.
32. Jessen L, Kovalick LJ, Azzaro AJ. The selegiline transdermal system (EMSAM): a therapeutic option for the treatment of major depressive disorder. P T. 2008;33(4):212-246. PubMed
33. Neuvonen PJ, Pohjola-Sintonen S, Tacke U, et al. Five fatal cases of serotonin syndrome after moclobemide-citalopram or moclobemide-clomipramine overdoses. Lancet. 1993;342(8884):1419. PubMed doi:10.1016/0140-6736(93)92774-N
34. Marangell LB. Switching antidepressants for treatment-resistant major depression. J Clin Psychiatry. 2001;62(suppl 18):12-17. PubMed
35. Gitlin MJ. Venlafaxine, monoamine oxidase inhibitors, and the serotonin syndrome [letter]. J Clin Psychopharmacol. 1997;17(1):66-67. PubMed doi:10.1097/00004714-199702000-00022
36. Birkmayer W, Knoll J, Riederer P, et al. Increased life expectancy resulting from addition of l-deprenyl to Madopar treatment in Parkinson’s disease: a longterm study. J Neural Transm. 1985;64(2):113-127. PubMed doi:10.1007/BF01245973
37. Bodkin JA, Amsterdam JD. Transdermal selegiline in major depression: a double-blind, placebo-controlled, parallel-group study in outpatients. Am J Psychiatry. 2002;159(11):1869-1875. PubMed doi:10.1176/appi.ajp.159.11.1869
38. Amsterdam JD. A double-blind, placebo-controlled trial of the safety and efficacy of selegiline transdermal system without dietary restrictions in patients with major depressive disorder. J Clin Psychiatry. 2003;64(2):208-214. PubMed doi:10.4088/JCP.v64n0216
39. Amsterdam JD, Bodkin JA. Selegiline transdermal system in the prevention of relapse of major depressive disorder: a 52-week, double-blind, placebo-substitution, parallel-group clinical trial. J Clin Psychopharmacol. 2006;26(6):579-586. PubMed doi:10.1097/01.jcp.0000239794.37073.70
40. Feiger AD, Rickels K, Rynn MA, et al. Selegiline transdermal system for the treatment of major depressive disorder: an 8-week, double-blind, placebo-controlled, flexible-dose titration trial. J Clin Psychiatry. 2006;67(9):1354-1361. PubMed doi:10.4088/JCP.v67n0905
41. Lotufo-Neto F, Trivedi M, Thase ME. Meta-analysis of the reversible inhibitors of monoamine oxidase type A moclobemide and brofaromine for the treatment of depression. Neuropsychopharmacology. 1999;20(3):226-247. PubMed doi:10.1016/S0893-133X(98)00075-X
42. Silverstone T. Moclobemide: placebo-controlled trials. Int Clin Psychopharmacol. 1993;7(3-4):133-136. PubMed doi:10.1097/00004850-199300730-00002
43. Papakostas GI, Fava M. A metaanalysis of clinical trials comparing moclobemide with selective serotonin reuptake inhibitors for the treatment of major depressive disorder. Can J Psychiatry. 2006;51(12):783-790. PubMed
44. AZILECT (rasagiline mesylate) [package insert]. Kansas City, MO: TEVA Neuroscience, Inc; 2012. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=a40d0e73-3f9f-4b01-979d-402c9cdaeb60. Accessed May 17, 2012.
45. Rascol O, Brooks DJ, Melamed E, et al, for the LARGO study group. Rasagiline as an adjunct to levodopa in patients with Parkinson’s disease and motor fluctuations (LARGO, Lasting effect in Adjunct therapy with Rasagiline Given Once daily, study): a randomised, double-blind, parallel-group trial. Lancet. 2005;365(9463):947-954. PubMed doi:10.1016/S0140-6736(05)71083-7
46. Perry PJ, Alexander B. Rational use of antidepressants. Prim Care. 1987;14(4):773-783. PubMed
47. Garriock HA, Kraft JB, Shyn SI, et al. A genomewide association study of citalopram response in major depressive disorder. Biol Psychiatry. 2010;67(2):133-138. PubMed doi:10.1016/j.biopsych.2009.08.029
48. Lawrence S. Diagnostics investors think positive. Nat Biotechnol. 2006;24(8):884. PubMed doi:10.1038/nbt0806-884
49. Gerretsen P, M×¼ller DJ, Tiwari A, et al. The intersection of pharmacology, imaging, and genetics in the development of personalized medicine. Dialogues Clin Neurosci. 2009;11(4):363-376. PubMed