Efficacy of Add-On Deep Transcranial Magnetic Stimulation in Comorbid Alcohol Dependence and Dysthymic Disorder: Three Case Reports

ABSTRACT
Background: Craving for alcohol is associated with abnormal activation in the dorsolateral prefrontal cortex. Deep transcranial magnetic stimulation (dTMS) has shown promise in the treatment of depression. There are few treatment options for treatment-resistant dysthymic disorder comorbid with alcohol use disorder.
Objective: To investigate the possible anticraving efficacy of bilateral dorsolateral prefrontal cortex high-frequency dTMS in 3 patients with comorbid long-term DSM-IV-TR dysthymic disorder and alcohol use disorder.
Method: Three patients with alcohol use disorder with dysthymic disorder in their detoxification phase (abstaining for > 1 month) underwent twenty 20-minute sessions of 20 Hz dTMS over the dorsolateral prefrontal cortex over 28 days between 2011 and 2012. Alcohol craving was rated with the Obsessive Compulsive Drinking Scale and depressive symptoms with the Hamilton Depression Rating Scale.
Results: All 3 patients responded unsatisfactorily to initial intravenous antidepressant and antianxiety combinations but responded after 10 dTMS sessions, improving on both anxiety-depressive symptoms and craving. This improvement enabled us to reduce antidepressant dosages after dTMS cycle completion.
Discussion: High-frequency bilateral dorsolateral prefrontal cortex dTMS with left prevalence was found to produce significant anticraving effects in alcohol use disorder comorbid with dysthymic disorder. The potential of dTMS for reducing craving in patients with substance use disorder deserves to be further investigated.
Prim Care Companion CNS Disord 2013;15(1):doi:10.4088/PCC.12m01438
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: July 10, 2012; accepted November 27, 2012.
Published online: February 7, 2013.
Corresponding author: Georgios D. Kotzalidis, MD, Neurosciences, Mental Health, and Sensory Organs Department, Sapienza University, School of Medicine and Psychology, Psychiatry Unit, Sant’ Andrea Hospital, Via di Grottarossa 1035-1039, 00189 Rome, Italy ([email protected]).
Alcohol craving is an irresistible urge to drink or intense thoughts about alcohol1 and is a primary factor in generating and maintaining alcohol dependence and alcohol-taking behavior, as well as relapse.2 Craving in alcohol use disorder is associated with hyperactivation of areas related to emotional responses to alcohol-related cues,3,4 in particular, of the dorsolateral prefrontal cortex.5
Mood and substance use disorders are each associated with significant societal and personal costs,6 which increase in the case of comorbidity.7 The presence of dysthymia induces a 5-fold increase in costs of substance use disorders.8
The lifetime prevalence of dysthymic disorder in the Epidemiologic Catchment Area survey was 3.1%, with greater risk among women younger than age 65 years and in unmarried, young, and low-income individuals.9 Treatment options for dysthymic disorder and comorbid alcohol use disorder are limited to antidepressant drugs, but their effect size is rather weak.10
Although several anticraving drugs for alcohol dependence are available, their effectiveness is limited,11 hence the need to develop new therapeutic interventions. Several studies showed the potential anticraving effects of repetitive transcranial magnetic stimulation (rTMS) in substance dependence.12-15 TMS appears to be a promising candidate for treating addictive behaviors16,17 and other brain diseases.18 In the drug addiction domain, the therapeutic potential of rTMS has been tested in nicotine dependence,14,15 cocaine addiction,13 and alcoholism.12 TMS variants and direct electric stimulation proved to be effective in reducing alcohol craving in patients with alcohol use disorder when stimulation was carried bilaterally to the dorsolateral prefrontal cortex,4,12,19,20 but reduction is only transient when stimulation is delivered to the dorsal anterior cingulate21 and is absent when it is delivered to the dorsolateral prefrontal cortex only on the right side.22 TMS was shown to improve depressive and anxiety symptoms in patients with alcohol use disorder.23 Deep TMS (dTMS), which differs from rTMS in that it uses an H coil that is able to concentrate electrical fields in deeper brain structures than rTMS, improved depressive symptoms in patients with unipolar24-28 and bipolar depression.29,30 However, the value of dTMS has not been tested in substance or alcohol use disorders.
CASE REPORTS
We admitted 3 patients with comorbid long-term DSM-IV-TR dysthymic disorder and alcohol use disorder in their detoxification phase (abstaining for > 1 month) to the alcohology service of the day hospital of Villa Rosa, Suore Hospitaliere of the Sacred Heart of Jesus, Viterbo, Italy. For all 3 patients, dysthymic disorder was temporally related to adverse life events and preceded the onset of alcohol use disorder. They were all treated with 100 mg/d of intravenous trazodone plus other intravenous and oral medications, and all had shown unsatisfactory clinical response before being offered add-on dTMS, which has been found to benefit depressive symptoms and alcohol addiction-related mood symptomatology.
The DSM-IV-TR diagnoses were established after structured interviews with all patients (Structured Clinical Interviews for DSM-IV Axis I and II Disorders31,32). Patients were evaluated for depression with the Hamilton Depression Rating Scale33 and for alcohol craving with the Obsessive Compulsive Drinking Scale.34 All patients had experienced craving when abstaining from alcohol in the past.
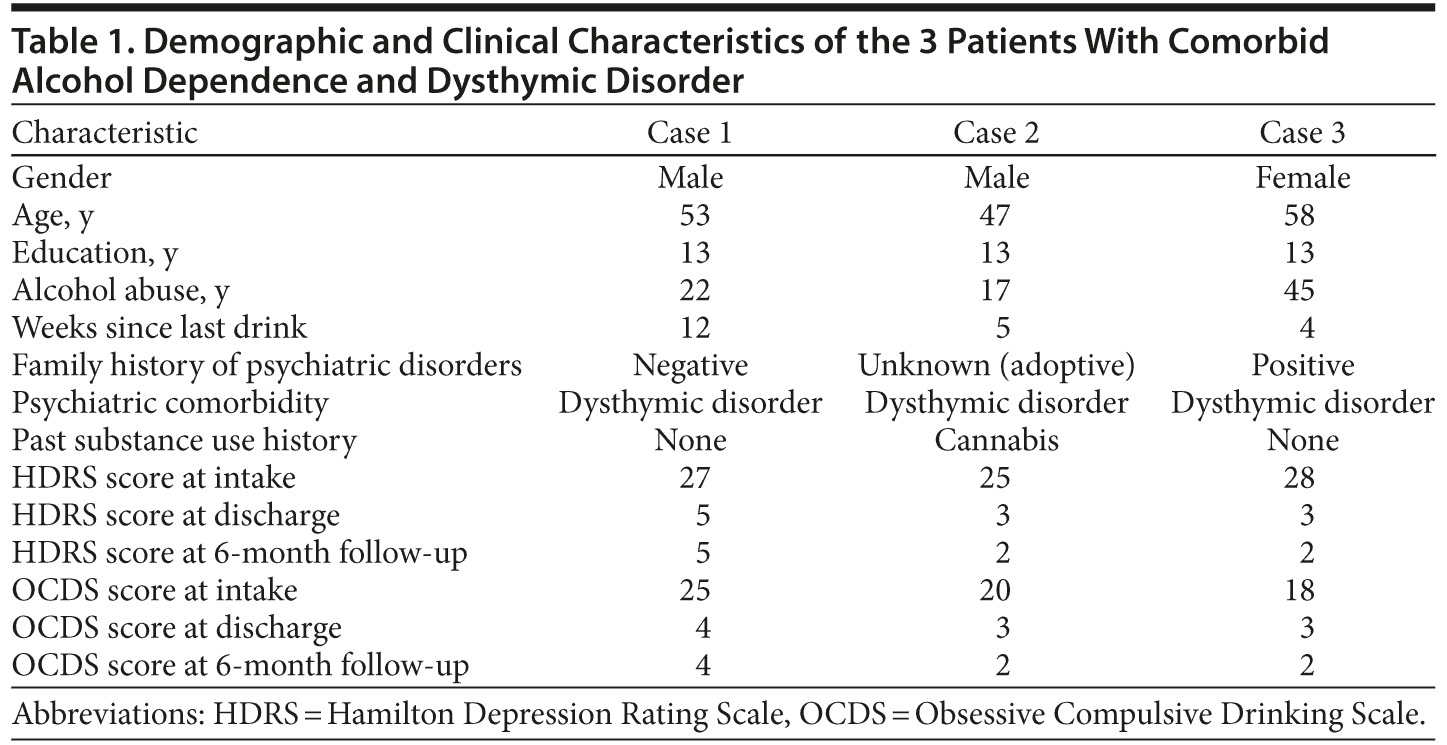
All patients provided written informed consent for the collection of their data for research, participation in the study, and subsequent publication of the study results. Their sociodemographic and clinical characteristics, including their scores on the 2 assessment tools across the hospitalization and follow-up, are shown in Table 1.
Case 1
Mr A, a 53-year-old man with dysthymic disorder and a 20-year history of alcohol addiction with no family history of psychiatric disorders, came to our attention in September 2011 due to the development of Wernicke’s encephalopathy. At hospitalization, he manifested confusion, confabulation, and gait disturbances (ataxia). Mr A also manifested sadness, depressed mood, insomnia, anxiety, and weakness.

- Deep transcranial magnetic stimulation (dTMS) is a technique that is increasingly reported to benefit psychiatric conditions such as mood disorders and substance use disorders.
- When patients with concurrent alcohol use disorder and dysthymic disorder respond poorly to otherwise appropriate treatment, add-on dTMS may prove successful.
- If dTMS is made more available, this new, currently little-used technique may contribute to integrated treatment of an increasing number of patients with mood and substance use disorders.
Mr A worked in a hotel as a concierge, was a moderate smoker, and was the father of a 31-year-old son with whom he had few contacts. Mr A lost his mother at age 10 years and had a tense relationship with his father since that time. He had been experiencing dysthymic symptoms since the age of 22 years, characterized by depressed mood, feelings of inadequacy, guilt, irritability, and impairment in role and social functioning. His alcohol use history started at the age of 30 years when he was working in cinema production and was fired. Alcohol use progressed to addiction within 3 years and continued to worsen progressively, eventually leading to Wernicke’s encephalopathy. Mr A required hospitalization for 3 months, during which time he received intravenous diazepam 20 mg/d, trazodone 100 mg/d, and tiapride 150 mg/d; he also received oral multivitamin complexes and abstained from alcohol. With this treatment, his neurologic symptoms soon subsided, and his electroencephalogram was normal; however, residual depressive symptoms persisted.
Mr A underwent a full cycle of dTMS and improved in depression and craving (deflected mood, irritability, and severe anxiety) starting by the second week of treatment. At the end of the dTMS sessions, his clinical condition had strongly improved (Table 1), allowing us to reduce trazodone to 50 mg/d, tiapride to 100 mg/d, and diazepam to 4 mg/d. At 6-month follow-up, Mr A was well with no depressive symptoms and minimum alcohol craving.
Case 2
Mr B, a 47-year-old man with dysthymic disorder and panic attacks since the age of 20 years and a history of alcohol abuse since the age of 30 years, presented to our day hospital in December 2012 with generalized anxiety, somatic symptoms, irritability, and dysphoric and depressed mood. His dysthymia manifested as hopelessness and social withdrawal. He had no organic disease other than hiatal hernia. Mr B had lived in an orphanage until he was adopted at the age of 8 years. His relationship with his adoptive family had always been poor. At age 19 years, he was sued by his adoptive mother for extortion and spent 3 months in jail. During adolescence, he occasionally used cannabis. Mr B married at age 25 years and had 2 healthy sons. He had worked in a hospital until the age of 30 years, when he was fired. He then started drinking about 2 L/d of wine until age 44 years; for the following 2 years, he gradually increased alcohol consumption to 5 L/d. Mr B received intravenous clomipramine 100 mg/d, diazepam 20 mg/d, trazodone hydrochloride 100 mg/d, and tiapride hydrochloride 150 mg/d for 3 months, but his mood and anxiety symptoms responded poorly. He underwent a full cycle of dTMS and showed symptom improvement after about 2 weeks of treatment (10 sessions). After completion of the dTMS cycle, Mr A’s improvement allowed us to adjust his medication to oral clomipramine 175 mg/d, olanzapine 10 mg/d, and diazepam 4 mg/d. At 6-month follow-up, depressive symptoms and alcohol craving were absent.
Case 3
Ms C, a 58-year-old woman with early-onset dysthymic disorder (at age 13 years) and a history of alcohol abuse since the age of 15 years, was referred to our service in January 2012 complaining of increased alcohol intake and worsening of anxiety and depressive symptoms. The daughter of 2 divorced parents, Ms C suffered physical abuse by her father when she was a child. Her brother committed suicide when he was 26 years old. Ms C is married, is the mother of a 31-year-old son, and works as an antiquarian. She received intravenous diazepam 10 mg/d and trazodone 50 mg/d and oral olanzapine 10 mg/d and duloxetine 60 mg/d with no apparent benefit. She underwent a full cycle of dTMS and showed improvements in anxiety, depression, and craving by the second week of treatment. After completion of dTMS sessions, Ms C’s marked clinical improvement allowed us to adjust her medication to maintenance oral olanzapine 10 mg/d, duloxetine 30 mg/d, and diazepam 4 mg/d. At 6-month follow-up, depressive symptoms had subsided and alcohol craving was absent.
dTMS Treatment
All 3 patients were abstaining from alcohol for more than 1 month before the first dTMS session. The dTMS sessions were conducted using the Brainsway’s H1 coil Deep TMS System (Brainsway, Har Hotzvim, Jerusalem, Israel). The H1 coil is designed to elicit neuronal activation in medial and lateral prefrontal regions, including the orbitofrontal cortex, with a preference for the left hemisphere.35 The H1 coils were positioned over the patient’s scalp. The optimal spot on the scalp for stimulation of the right abductor pollicis brevis muscle was located, and the motor threshold was established by delivering single stimulations to the motor cortex. The motor threshold was measured by gradually increasing the stimulation intensity (using single pulse mode, applying 1 pulse every 5 seconds [0.2 Hz]) and recording electrical activity in the abductor pollicis brevis using surface electrodes. Motor threshold was defined as the lowest stimulation intensity required to evoke motor potential of at least 50 μ V in 5 of 10 stimulations. The coil was then placed 5.5 cm anterior to the motor spot (ie, corresponding to the prefrontal cortex), and spatial coordinates were recorded with markings on a cap placed on the subject’s head to ensure placement reproducibility. Deep TMS treatment was delivered by an expert physician in trains of 20 Hz at 120% of the measured motor threshold. Each dTMS session consisted of 55 trains with a 2-second duration for each, and a 20-second intertrain interval. The complete cycle of the dTMS treatment consisted of 5 consecutive session days in a week for 4 consecutive weeks for a total of 20 sessions. The treatment was well tolerated by the patients.
DISCUSSION
Patients with alcohol use disorder continue using alcohol despite experiencing alcohol-associated role impairment and adverse health consequences. Alcohol abuse and dependence are associated with neuropsychological impairment,36 poor medication adherence,37 economic costs, and lost productivity.38 Alcohol use disorder represents a widespread and serious personal and public health problem in the United States.39 Mood and alcohol use disorder often co-occur, each complicating the course and outcome of the other.40,39 Although causality is difficult to establish,41 mood disorder is believed to be most often the consequence of alcohol use disorder.42 In our case series, dysthymic disorder always preceded the onset of alcohol use and abuse. Dorsolateral prefrontal cortex stimulation through dTMS was followed by decreased alcohol craving in our patients, but the mechanism through which this result was obtained remains to be established. All 3 patients showed a similar pattern in their response to the dTMS add-on, with improvements in both depressive symptoms and reduced craving by the tenth session. Improvement continued through the rest of the cycle in all 3 patients and persisted at follow-up despite dose reduction and change of formulation of antidepressant treatment.
This is the first report of the usefulness of stimulating the dorsolateral prefrontal cortex in alcohol craving. We found a decrease in alcohol craving in 3 consecutive patients with comorbid dysthymic disorder and alcohol use disorder who received add-on bilateral dTMS similar to the rTMS study by Mishra and colleagues.12 However, we may not infer as to the usefulness of dTMS in patients who are not treated with medications or other somatic treatment or who are not comorbid for dysthymic disorder because we could not compare our results with those of controls with alcohol use disorder only or those of untreated patients receiving dTMS only. Our case study showed improvement in depressive symptoms of patients with dysthymic disorder with comorbid alcohol use disorder; this finding parallels observations with rTMS in patients with alcoholic withdrawal syndrome23 and in depressive populations with dTMS.24-30 As mentioned previously, we are currently unable to speculate as to the mechanism of action of dTMS and with regard to the actual value of the technique in obtaining our results. Our report indicates the possible usefulness of add-on dTMS in patients with comorbid dysthymic disorder and alcohol use disorder. Further studies using sham comparisons are needed to assess the real value of this new technique in reducing depressive-anxiety symptoms and craving in populations with mood disorders comorbid with alcohol use disorder.
Drug names: clomipramine (Anafranil and others), diazepam (Diastat, Valium, and others), duloxetine (Cymbalta), olanzapine (Zyprexa), trazodone (Oleptro and others).
Author affiliations: Department of Neurosciences, Mental Health, and Sensory Organs (NESMOS), Sapienza University, School of Medicine and Psychology, Sant’ Andrea Hospital, Rome, Italy (Drs Rapinesi, Kotzalidis, Serata, Del Casale, Solfanelli, Brugnoli, Tatarelli, Angeletti, Ferracuti, and Girardi); Department of Neuropsychiatry, Villa Rosa, Suore Hospitaliere of the Sacred Heart of Jesus, Viterbo, Italy (Drs Rapinesi, Serata, Scatena, Digiacomantonio, Carbonetti, Fensore, and Girardi); Department of Neurology and Psychiatry, Sapienza University, Rome, Italy (Dr Bersani); and ATID Ltd Advanced Technology Innovation Distribution, Rome, Italy (Dr Raccah).
Potential conflicts of interest: Dr Raccah is a scientific consultant to ATID Ltd, distributor of deep rTMS (Brainsway) technology in Italy. In the past 2 years, Dr Girardi has received research support from Eli Lilly, Janssen, and Springer Healthcare; has served on advisory boards for Eli Lilly, Otsuka, Pfizer, Schering, and Springer Healthcare; and has received honoraria from Eli Lilly and Springer Healthcare. Drs Rapinesi, Kotzalidis, Serata, Del Casale, Bersani, Solfanelli, Scatena, Brugnoli, Digiacomantonio, Carbonetti, Fensore, Tatarelli, Angeletti, and Ferracuti report no conflicts of interest related to the subject of this article.
Funding/support: None reported.
Acknowledgments: The authors acknowledge the contribution of Mimma Ariano, MS, Felicia Proietti, MS, and the late Tiziana Mattei, MS, librarians at the School of Medicine and Psychology, Sapienza University, Rome, Italy, as well as secretary Lucilla Martinelli, MS (Sapienza University, Rome, Italy), who identified relevant literature. None of the acknowledged individuals have conflicts of interest related to the subject of this article.
REFERENCES
1. World Health Organization. The ICD-10 Classification of Mental and Behavioral Disorders: Clinical Descriptions and Diagnostic Guidelines. Geneva, Switzerland: World Health Organization; 1992.
2. Drummond DC. Theories of drug craving, ancient and modern. Addiction. 2001;96(1):33-46. PubMed doi:10.1046/j.1360-0443.2001.961333.x
3. Park MS, Sohn JH, Suk JA, et al. Brain substrates of craving to alcohol cues in subjects with alcohol use disorder. Alcohol Alcohol. 2007;42(5):417-422. PubMed
4. Boggio PS, Sultani N, Fecteau S, et al. Prefrontal cortex modulation using transcranial DC stimulation reduces alcohol craving: a double-blind, sham-controlled study. Drug Alcohol Depend. 2008;92(1-3):55-60. PubMed doi:10.1016/j.drugalcdep.2007.06.011
5. Olbrich HM, Valerius G, Paris C, et al. Brain activation during craving for alcohol measured by positron emission tomography. Aust N Z J Psychiatry. 2006;40(2):171-178. PubMed doi:10.1080/j.1440-1614.2006.01765.x
6. Goetzel RZ, Hawkins K, Ozminkowski RJ, et al. The health and productivity cost burden of the “top 10” physical and mental health conditions affecting six large US employers in 1999. J Occup Environ Med. 2003;45(1):5-14. PubMed doi:10.1097/00043764-200301000-00007
7. Hoff RA, Rosenheck RA. The cost of treating substance abuse patients with and without comorbid psychiatric disorders. Psychiatr Serv. 1999;50(10):1309-1315. PubMed
8. Westermeyer J, Eames SL, Nugent S. Comorbid dysthymia and substance disorder: treatment history and cost. Am J Psychiatry. 1998;155(11):1556-1560. PubMed
9. Weissman MM, Leaf PJ, Bruce ML, et al. The epidemiology of dysthymia in five communities: rates, risks, comorbidity, and treatment. Am J Psychiatry. 1988;145(7):815-819. PubMed
10. Iovieno N, Tedeschini E, Bentley KH, et al. Antidepressants for major depressive disorder and dysthymic disorder in patients with comorbid alcohol use disorders: a meta-analysis of placebo-controlled randomized trials. J Clin Psychiatry. 2011;72(8):1144-1151. PubMed doi:10.4088/JCP.10m06217
11. O’ Brien CP. Anticraving medications for relapse prevention: a possible new class of psychoactive medications. Am J Psychiatry. 2005;162(8):1423-1431. PubMed doi:10.1176/appi.ajp.162.8.1423
12. Mishra BR, Nizamie SH, Das B, et al. Efficacy of repetitive transcranial magnetic stimulation in alcohol dependence: a sham-controlled study. Addiction. 2010;105(1):49-55. PubMed doi:10.1111/j.1360-0443.2009.02777.x
13. Politi E, Fauci E, Santoro A, et al. Daily sessions of transcranial magnetic stimulation to the left prefrontal cortex gradually reduce cocaine craving. Am J Addict. 2008;17(4):345-346. PubMed doi:10.1080/10550490802139283
14. Lang N, Hasan A, Sueske E, et al. Cortical hypoexcitability in chronic smokers? a transcranial magnetic stimulation study. Neuropsychopharmacology. 2008;33(10):2517-2523. PubMed doi:10.1038/sj.npp.1301645
15. Amiaz R, Levy D, Vainiger D, et al. Repeated high-frequency transcranial magnetic stimulation over the dorsolateral prefrontal cortex reduces cigarette craving and consumption. Addiction. 2009;104(4):653-660. PubMed doi:10.1111/j.1360-0443.2008.02448.x
16. Barr MS, Fitzgerald PB, Farzan F, et al. Transcranial magnetic stimulation to understand the pathophysiology and treatment of substance use disorders. Curr Drug Abuse Rev. 2008;1(3):328-339. PubMed doi:10.2174/1874473710801030328
17. Feil J, Zangen A. Brain stimulation in the study and treatment of addiction. Neurosci Biobehav Rev. 2010;34(4):559-574. PubMed doi:10.1016/j.neubiorev.2009.11.006
18. Kobayashi M, Pascual-Leone A. Transcranial magnetic stimulation in neurology. Lancet Neurol. 2003;2(3):145-156. PubMed doi:10.1016/S1474-4422(03)00321-1
19. Staroverov AT, Vil’ yanov VB, Raigorodskii YM, et al. Transcranial magnetotherapy in the complex treatment of affective disorders in patients with alcoholism. Neurosci Behav Physiol. 2009;39(5):507-511. PubMed doi:10.1007/s11055-009-9149-z
20. Staroverov AT, Zhukov OB, Raigorodskii YM. Efficacy of transcranial magnetotherapy in the complex treatment of alcohol withdrawal syndrome. Neurosci Behav Physiol. 2009;39(9):891-895. PubMed doi:10.1007/s11055-009-9206-7
21. De Ridder D, Vanneste S, Kovacs S, et al. Transient alcohol craving suppression by rTMS of dorsal anterior cingulate: an fMRI and LORETA EEG study. Neurosci Lett. 2011;496(1):5-10. PubMed doi:10.1016/j.neulet.2011.03.074
22. Herremans SC, Baeken C, Vanderbruggen N, et al. No influence of one right-sided prefrontal HF-rTMS session on alcohol craving in recently detoxified alcohol-dependent patients: results of a naturalistic study. Drug Alcohol Depend. 2012;120(1-3):209-213. doi:10.1016/j.drugalcdep.2011.07.021 PubMed
23. Staroverov AT, Zhukov OB, RaÄgorodskiÄ IuM. Effectiveness of transcranial magnetic therapy in the complex treatment of alcohol abstinent syndrome [Russian]. Zh Nevrol Psikhiatr Im S S Korsakova. 2008;108(9):57-61. PubMed
24. Levkovitz Y, Harel EV, Roth Y, et al. Deep transcranial magnetic stimulation over the prefrontal cortex: evaluation of antidepressant and cognitive effects in depressive patients. Brain Stimulat. 2009;2(4):188-200. PubMed doi:10.1016/j.brs.2009.08.002
25. Rosenberg O, Shoenfeld N, Zangen A, et al. Deep TMS in a resistant major depressive disorder: a brief report. Depress Anxiety. 2010a;27(5):465-469. PubMed doi:10.1002/da.20689
26. Rosenberg O, Zangen A, Stryjer R, et al. Response to deep TMS in depressive patients with previous electroconvulsive treatment. Brain Stimulat. 2010b;3(4):211-217. PubMed doi:10.1016/j.brs.2009.12.001
27. Rosenberg O, Isserles M, Levkovitz Y, et al. Effectiveness of a second deep TMS in depression: a brief report. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35(4):1041-1044. PubMed doi:10.1016/j.pnpbp.2011.02.015
28. Isserles M, Rosenberg O, Dannon P, et al. Cognitive-emotional reactivation during deep transcranial magnetic stimulation over the prefrontal cortex of depressive patients affects antidepressant outcome. J Affect Disord. 2011;128(3):235-242. PubMed doi:10.1016/j.jad.2010.06.038
29. Harel EV, Zangen A, Roth Y, et al. H-coil repetitive transcranial magnetic stimulation for the treatment of bipolar depression: an add-on, safety and feasibility study. World J Biol Psychiatry. 2011;12(2):119-126. PubMed doi:10.3109/15622975.2010.510893
30. Bersani FS, Girardi N, Sanna L, et al. Deep transcranial magnetic stimulation for treatment-resistant bipolar depression: a case report of acute and maintenance efficacy. [published online ahead of print July 24, 2012]. Neurocase.d
31. First MB, Spitzer RL, Gibbon M, et al. Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Patient Edition. New York, NY: Biometrics Research, New York State Psychiatric Institute; 2002: A1-A46.
32. First MB, Gibbon M, Spitzer RL, et al. Structured Clinical Interview for DSM-IV Axis II Personality Disorders, (SCID-II). Washington, DC: American Psychiatric Press, Inc, 1997.
33. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56-62. PubMed doi:10.1136/jnnp.23.1.56
34. Anton RF, Moak DH, Latham P. The Obsessive Compulsive Drinking Scale: a self-rated instrument for the quantification of thoughts about alcohol and drinking behavior. Alcohol Clin Exp Res. 1995;19(1):92-99. PubMed doi:10.1111/j.1530-0277.1995.tb01475.x
35. Roth Y, Amir A, Levkovitz Y, et al. Three-dimensional distribution of the electric field induced in the brain by transcranial magnetic stimulation using figure-8 and deep H-coils. J Clin Neurophysiol. 2007;24(1):31-38. PubMed doi:10.1097/WNP.0b013e31802fa393
36. Bates ME, Bowden SC, Barry D. Neurocognitive impairment associated with alcohol use disorders: implications for treatment. Exp Clin Psychopharmacol. 2002;10(3):193-212. PubMed doi:10.1037/1064-1297.10.3.193
37. Bazargan-Hejazi S, Bazargan M, Hardin E, et al. Alcohol use and adherence to prescribed therapy among under-served Latino and African-American patients using emergency department services. Ethn Dis. 2005;15(2):267-275. PubMed
38. Harwood R, Fountain D, Livermore G. The Economic Costs of Alcohol and Drug Abuse in the United States, 1992. Bethesda, MD: National Institute on Alcohol Abuse and Alcoholism and National Institute on Drug Abuse; 1998.
39. Hasin DS, Goodwin RD, Stinson FS, et al. Epidemiology of major depressive disorder: results from the National Epidemiologic Survey on Alcoholism and Related Conditions. Arch Gen Psychiatry. 2005;62(10):1097-1106. PubMed doi:10.1001/archpsyc.62.10.1097
40. Grant BF, Stinson FS, Dawson DA, et al. Prevalence and co-occurrence of substance use disorders and independent mood and anxiety disorders: results from the National Epidemiologic Survey on Alcohol and Related Conditions. Arch Gen Psychiatry. 2004;61(8):807-816. PubMed doi:10.1001/archpsyc.61.8.807
41. Flensborg-Madsen T. Alcohol use disorders and depression—the chicken or the egg? Addiction. 2011;106(5):916-918. PubMed doi:10.1111/j.1360-0443.2011.03406.x
42. Fergusson DM, Boden JM, Horwood LJ. Tests of causal links between alcohol abuse or dependence and major depression. Arch Gen Psychiatry. 2009;66(3):260-266. PubMed doi:10.1001/archgenpsychiatry.2008.543