Apical Ballooning Syndrome: The “Broken Heart” Syndrome
Have you ever wondered about the “broken heart syndrome”? When a patient presents with cardiac symptoms in the context of acute stress, are you aware of the most important conditions in the differential diagnosis? Have you ever wanted to know more about how an acutely stressful situation can impact cardiac physiology? If so, this case and the resulting discussion may provide insight into the complex interplay of the heart and mind.
CASE PRESENTATION
While riding the subway to work, Ms A, a 36-year-old woman, developed the acute onset of shortness of breath associated with dull, aching pain radiating down her left arm. Minutes later, she developed crushing substernal chest pain of 10/10 intensity; paramedics arrived and sublingual nitroglycerin led to prompt reduction of her pain to 4/10 intensity. In the emergency department, an electrocardiogram (ECG) revealed ST-segment elevations in the inferior leads, and a screening troponin value was positive. She was taken promptly for cardiac catheterization; angiography revealed patent coronary arteries, and an echocardiogram measured an ejection fraction of 35% with hypokinesis of the mid and apical segments of the left ventricle. Ms A was admitted for observation due to new-onset cardiomyopathy of unknown origin.
On evaluation in the hospital, Ms A was tearful, with her husband and daughter at her bedside. She reported never having similar symptoms in the past and was now free of chest pain; her physical examination was unremarkable. Further interview revealed a history of mild hypertension; her blood pressure had been well controlled with propranolol (40 mg twice daily), prescribed for “blood pressure and anxiety.” She was also prescribed paroxetine 10 mg daily for anxiety. Family history was significant for her father having hypertension and a stroke at age 39 years; there was no family history of cardiomyopathy or sudden death. Social history revealed no history of illicit drug use, and she specifically denied use of cocaine, stimulants, opiates, or intravenous drug, or abuse of alcohol. She was employed as a librarian and had been living with her 4-year-old daughter and husband of 10 years. However, when asked about stress, Ms A endorsed significant emotional distress and depressed mood since learning 4 days before that her husband had been unfaithful; on the day prior to admission, her husband had moved into a nearby hotel.
During the admission, she received treatment with β-blockers for blood pressure control. Her troponin T peaked at 2.45 ng/mL and subsequently trended down with no return of her chest pain. A subsequent transthoracic echocardiogram on hospital day 3 showed an improved ejection fraction of 60% without hypokinesis. Due to the spontaneous resolution of her symptoms, the classical apical ballooning pattern seen on the echocardiogram, and the significant emotional stressor preceding the onset of her symptoms, this event was consistent with apical ballooning syndrome (ABS), also known as Takotsubo’s cardiomyopathy.
HOW DO PSYCHIATRIC SYMPTOMS AFFECT CARDIAC HEALTH?
A variety of psychiatric symptoms and syndromes appear to have significant effects on cardiovascular outcomes. Among these, depressive disorders and anxiety have been the best studied. Depression has been established as a risk factor for negative cardiac outcomes across the spectrum of cardiac disease. Initially healthy adults who have depression are more likely to eventually develop heart disease, independent of standard cardiovascular risk factors.1 Furthermore, among those with known coronary artery disease, being depressed is associated with inferior cardiac health.2 Among patients hospitalized with an acute coronary syndrome or congestive heart failure, or those having coronary artery bypass graft surgery, depression is an independent risk factor for recurrent cardiac events and mortality.3,4
Anxiety has also been associated with the development of coronary artery disease, with 2 large recent studies finding a strong and independent association between anxiety and coronary artery disease.5 With respect to anxiety among patients with recent coronary events, several studies have found that anxiety after myocardial infarction (MI) is associated with elevated rates of in-hospital complications, independent of other medical factors.6,7 More specific anxiety states have been associated with cardiac events as well. Phobic anxiety, an agoraphobia-like condition, has been associated with cardiac disease, particularly sudden cardiac death.8 Panic disorder also appears to be more common in patients with coronary artery disease than in the general population9 and has been associated with the onset of MI.10 Finally, other serious mental illnesses, such as bipolar disorder and schizophrenia, have also been associated with elevated rates of mortality from cardiovascular disease.11,12
In sum, it appears that psychiatric symptoms and conditions appear to be consistently associated with the onset and progression of cardiovascular illness. The mechanism by which this link exists is likely a combination of the physiologic effects of these states (eg, inflammation) and the impact on health maintenance behaviors such as smoking, exercise, and medication adherence.
WHAT IS APICAL BALLOONING SYNDROME?
Apical ballooning syndrome is a form of acute dilated cardiomyopathy. It was first described in the Japanese literature in 1991 by Dote and colleagues13 and was originally named Takotsubo cardiomyopathy. Tako-tsubo is the Japanese word for octopus trap, and, in this disorder, the heart has this distinctive shape when viewed on end-diastolic ventriculogram, with a wide base and a long thin neck. In the literature, the terms apical ballooning syndrome and Takotsubo cardiomyopathy are used interchangeably, while broken heart syndrome is commonly used in the popular press. For the purposes of this article, ABS will be used to refer to the clinical syndrome.
Apical ballooning syndrome occurs most commonly in women, especially postmenopausal women. Patients with ABS classically present with anginal chest pain, often at rest and with associated dyspnea,14,15 following the experience of a stressful event (often within minutes or hours of the onset of symptoms).16 However, one-third of patients ultimately diagnosed with ABS do not have an identified preceding emotional or physical stressor, and, therefore, the absence of a precipitating stressor is insufficient to rule out ABS when other findings suggest its presence.
In addition to chest pain, patients with ABS usually manifest ECG changes, especially ST-segment elevations in the anterior leads, with subsequent development of T-wave inversions; patients may have mildly elevated levels of cardiac enzymes (troponin I, troponin T, and CK-MB)3 and brain natriuretic peptide, a marker of congestive heart failure.14,17 Finally, a hallmark of ABS is the presence of wall-motion abnormalities on echocardiograms, with abnormalities of the middle segments and apex of the left ventricle that extend beyond a single vascular distribution.
Although rare, more complications of ABS may occur; these include arrhythmia, hypotension, pulmonary edema, and cardiogenic shock.15,16 Wall-motion abnormalities may cause left ventricular outflow obstruction and mitral regurgitation, leading to hypertrophic obstructive cardiomyopathy. There are also rare reports of ventricular free wall rupture and ventricular septal defects, and thromboembolic events related to left ventricular hypokinesis or dyskinesis may also occur. Overall, in-hospital mortality rates from ABS are much lower than from other acute cardiac illnesses (approximately 1%-3%)18; the most likely causes of mortality from ABS are cardiogenic shock and systemic embolization.19
In sum, ABS is an acute cardiomyopathy associated with anginal chest pain (often at rest), dyspnea, ST-segment elevations on the ECG, and characteristic wall-motion abnormalities on the echocardiogram. Such symptoms very often—but not invariably—occur in the context of an acute emotional stressor.
HOW CAN APICAL BALLOONING SYNDROME BE DISTINGUISHED FROM OTHER CARDIAC CONDITIONS?
ABS is virtually indistinguishable from acute MI, on the basis of presenting symptoms and routine laboratory testing. Accordingly, per the 2007 guidelines from the American College of Cardiology and the American Heart Association,2 ABS should be included in the differential for an acute coronary syndrome.
How can one differentiate ABS from an acute coronary syndrome? There are several factors that can help distinguish ABS from other acute cardiac conditions. First, ABS often presents following the occurrence of a stressful event. Common emotional stressors include unexpected death of a loved one, devastating financial loss, intense argument with friends or relatives, and receipt of a catastrophic medical diagnosis.16,20 ABS may also be immediately preceded by physical stressors, such as intense pain, noncardiac surgery, and intense exercise.15
However, as noted, the presence or absence of a stressful event cannot be used reliably to diagnose ABS, as a substantial minority of patients with ABS cannot identify a physical or emotional stress preceding the symptoms. In addition, acute cardiac events, including MI, congestive heart failure, and arrhythmia, can all be preceded by a stressful event. Therefore, although emotional or physical stress is characteristic of ABS and can be a clue to the presence of this condition, recent stress is neither necessary nor sufficient to differentiate ABS from other cardiac pathology. Structural brain lesions, cerebrovascular accidents, subarachnoid hemorrhage, and other neurologic conditions can also lead to cardiac events, although it is not clear whether these events typically lead to apical ballooning.21
In contrast, the results of cardiac catheterization can much more convincingly distinguish ABS from MI or other syndromes associated with coronary artery disease. In general, patients with ABS have normal cardiac catheterizations, with minimal evidence of vessel narrowing or blockage, and most patients with MI have substantial abnormalities that require acute intervention. However, there are also caveats when using catheterization results to distinguish ABS from other syndromes. For example, symptoms of a coronary event could have been caused by the transient rupture of a plaque, which then spontaneously lysed prior to catheterization, giving the appearance that coronary artery disease was not the cause of symptoms. Conversely, some patients suffering from ABS may have baseline coronary disease (given the high base rate of this condition in the population), leaving the appearance that a lesion seen on catheterization caused the presenting symptoms. Despite these caveats, however, catheterization results are quite helpful in distinguishing ABS from other conditions when they are added to the remainder of the clinical picture.
Perhaps the most convincing finding that distinguishes ABS from MI is seen most clearly on the echocardiogram: the characteristic set of wall-motion abnormalities associated with ABS. Classically, one observes hypokinesis or akinesis of the mid and apical segments of the left ventricle. This is what gives the apical ballooning appearance: narrowing at the ventricular neck and ballooning at the apex, with the basal left ventricle often spared.15,18 Of note, up to 40% of patients with ABS may have only midventricle hypokinesis (the so-called “apical-sparing variant”).22 There is a corresponding reduction in left ventricular ejection fraction, generally ranging from 20%-49%.14 Even more suggestive of the ABS diagnosis is a normalization of the wall-motion abnormalities and ejection fraction several days later. The natural course of ABS is one of rapid restoration of cardiac function, and normalization of the echocardiogram (after characteristic abnormalities on echocardiogram acutely) strongly suggests ABS.
There are no universally accepted diagnostic criteria for ABS, in part, because the syndrome has only recently been described, and more information needs to be gathered. However, Prasad and colleagues15 have developed proposed criteria for the condition that focus on the objective findings of this condition that distinguish it from other cardiovascular syndromes. The proposed criteria include characteristic wall-motion abnormalities seen on the ECG; the absence of coronary disease or acute plaque rupture, ECG abnormalities, or elevated cardiac troponins; and the absence of acute head trauma, intracranial bleeding, pheochromocytoma, myocarditis, or hypertrophic cardiomyopathy.
HοW CAN APICAL BALLOONING SYNDROME BE DISTINGUISHED FROM PSYCHIATRIC CONDITIONS THAT OCCUR IN THE CONTEXT OF ACUTE STRESS SUCH AS PANIC ATTACKS?
Just as it is important to differentiate ABS from other cardiac diseases, it is important to distinguish ABS from an acute presentation of psychiatric illness. As described earlier, there is evidence to support the interplay between chronic psychiatric disorders and the chronic course of medical illnesses. Conversely, acute symptoms of psychiatric disorders can mimic the presentation of other acute medical problems.
In this case, the most pertinent example is the similarity between a panic attack and acute coronary syndrome or ABS. As defined by the DSM-IV-TR,23 a panic attack is an intense fear or discomfort in which 4 (or more) symptoms develop abruptly and reach a peak within 10 minutes. Symptoms can include palpitations, pounding heart, or accelerated heart rate; sweating; trembling/shaking; sensation of shortness of breath or smothering; feeling of choking; chest pain or discomfort; nausea or abdominal distress; feeling unsteady, lightheaded, or faint; derealization or depersonalization; fear of losing control or going crazy; fear of dying; paresthesias; and chills or hot flushes.23(p432) Panic attacks frequently occur in the context of psychosocial stressors and can also at times occur in the setting of physical discomfort or exercise.
On the basis of diagnostic criteria, it is evident that a person presenting with symptoms of a panic attack might appear similar to someone experiencing ABS (or MI). The acuity of the patient’s distress, the constellation of associated symptoms, and the association with stressful events make panic attacks and ABS quite similar with respect to their initial presentation. Indeed, for any patient presenting with such symptoms, it would be necessary to rule out all other relevant medical causes before making the diagnosis of a panic attack.
There are, however, some hallmark features (though not pathognomonic) that suggest a panic attack as opposed to MI or ABS. For instance, panic attacks are characterized by a rapid crescendo-decrescendo pattern of symptom onset. Furthermore, panic symptoms usually resolve completely within 1 hour, often sooner. Additionally, the experience of depersonalization and/or derealization may suggest a panic attack because both are less common in acute coronary syndrome. Panic attacks can occur at rest and are not necessarily precipitated by an acute stressor. Finally, panic attacks most often have an age at onset during the third decade of life.23 This is especially relevant to differentiating between ABS and a panic attack. The classic patient with ABS is a postmenopausal woman. If she has no past history of panic attacks, it would make the diagnosis less likely. Ultimately, although the clinical presentation of ABS and panic attacks is quite similar, the diagnosis is clarified by ECG, cardiac enzymes, and echocardiogram. Patients with ABS typically have characteristic abnormalities on these diagnostic evaluations, while such tests should be normal in a patient having a panic attack.
Finally, it is important to consider the potential for coexisting panic attacks and cardiac ischemia. Among patients with panic disorder who are predisposed to cardiac events, the catecholamine surge associated with a panic attack could lead to vasoconstriction, hypertension, and increased myocardial demand, and, thus, the panic attack may lead to the subsequent development of a true cardiac event. Such a connection could also go in the other direction—the organically driven shortness of breath, chest pain, and diaphoresis of a myocardial event could serve as a nidus for a superimposed panic attack.
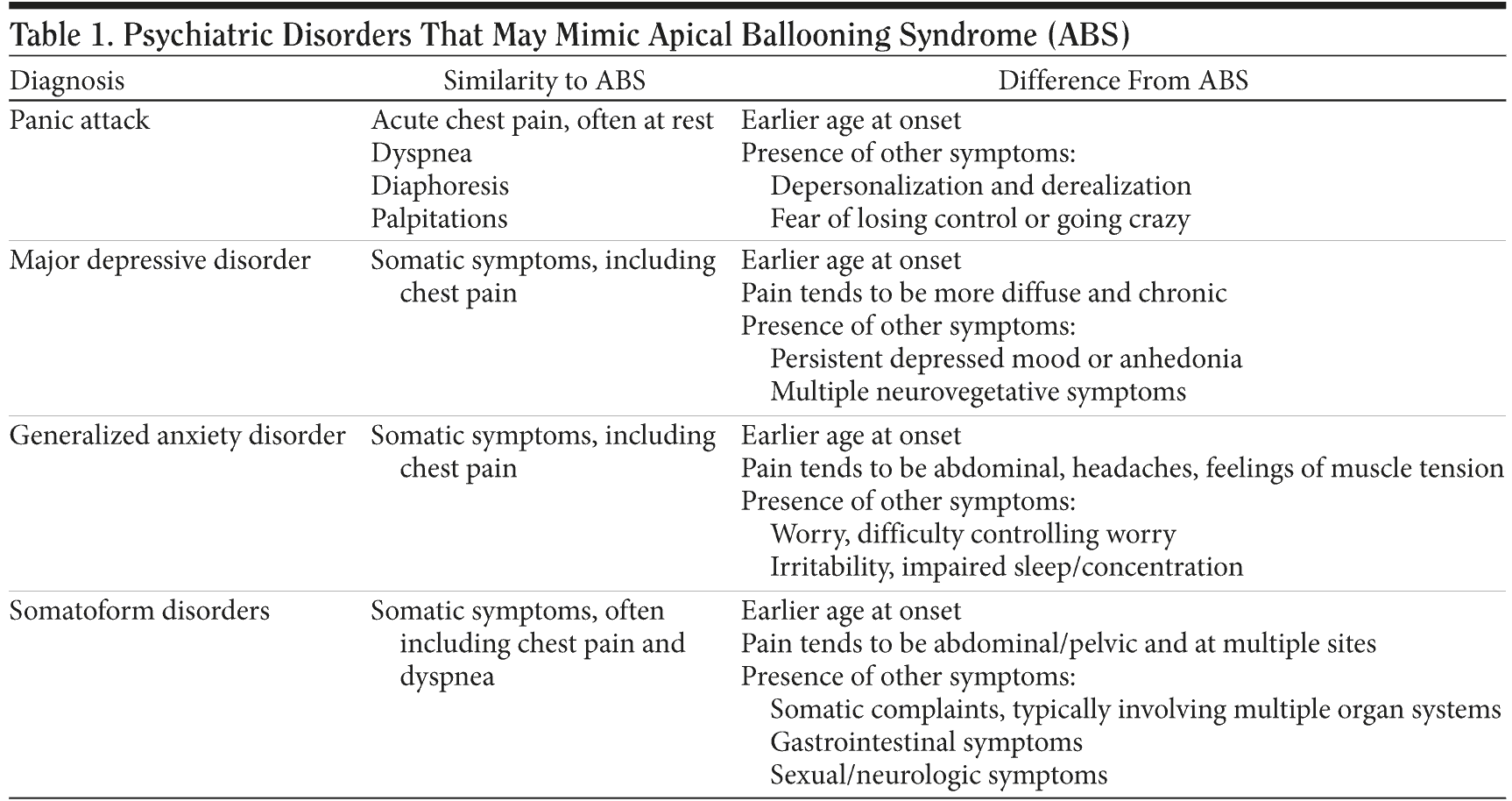
In addition to panic attacks, there are other psychiatric illnesses worth noting because of their propensity to present with physical complaints. Major depressive disorder, generalized anxiety disorder, and the somatoform disorders are the most common psychiatric disorders to share overlapping physical symptoms. However, unlike panic attacks, the physical manifestations in these other disorders tend to be chronic, diffuse, and associated with symptoms of decreased energy and/or fatigue. Moreover, a diagnosis of a somatization disorder, by definition, requires the manifestation of somatic complaints involving multiple organ systems. As with panic disorder, the age at onset of symptoms or diagnosis usually occurs much earlier in life compared to the initial presentation of ABS. Table 1 outlines the ways in which these disorders are similar to, and distinct from, ABS.
WHAT IS THE PATHOPHYSIOLOGY BEHIND APICAL BALLOONING SYNDROME?
The pathophysiology of ABS is not yet clearly understood. One proposed mechanism involves transient multivessel epicardial spasm causing the characteristic features of ABS; however, this has only been demonstrated in a small percentage of cases,18 and a study by Wittstein and colleagues24 of 19 patients with ABS failed to demonstrate epicardial spasm. A mechanism proposed by Ibanez and colleagues25 is plaque rupture of the left anterior descending artery; however, this fails to explain the characteristic left ventricular apical ballooning pattern.
Another proposed mechanism implicates microvascular ischemia, and researchers have documented abnormal myocardial perfusion in the left ventricular areas in which wall-motion abnormalities are found.22,26 However, imaging modalities have never been able to detect myocardial necrosis, despite the finding of troponin leaks in ABS, making it unlikely that this is the mechanism in all cases.
Perhaps the most likely of the proposed mechanisms involves abnormal levels of catecholamines.18 Animal models have found that physical immobilization leads to ABS-like findings in the heart, and blockade of catecholamines prevents these events. Furthermore, myocardial biopsies in some ABS patients show findings consistent with catecholamine-associated cardiotoxicity. Finally, the study by Wittstein and colleagues24 found that ABS patients had 2 to 3 times higher levels of catecholamines compared to matched controls with acute coronary syndrome, and the catecholamine hypothesis was further supported by findings of contraction band necrosis and mononuclear infiltrates in 4 of the 5 patients in the study who underwent endomyocardial biopsy. Still, for all of the proposed mechanisms, it has been difficult to determine whether these findings regarding catecholamines, spasm, plaque rupture, and ischemia are causative or epiphenomena, and further work is needed.
HOW SHOULD PATIENTS WITH APICAL BALLOONING SYNDROME BE MANAGED?
Perhaps the most important aspect of educating patients with ABS is reassurance. By definition, ABS is a self-limited syndrome; unlike MI and other acute cardiac illnesses, the mortality from ABS is low.18 Since stress plays a role in triggering these episodes, it seems prudent to limit additional stress by assuring patients that cardiac contractility typically recovers quickly, with complete normalization of left ventricular function within 1-3 months, often sooner.
All patients suspected of having ABS should have an echocardiogram (or cardiac magnetic resonance imaging) acutely, as well as an echocardiogram before discharge to ensure that left ventricular dysfunction is improved. Monitoring via cardiac telemetry is indicated to observe for arrhythmias. If coronary artery disease is suspected as a cause of the presenting symptoms, cardiac catheterization is indicated to aid diagnosis and to allow intervention if needed.
On occasion, acute anticoagulation with heparin may be considered to prevent embolization if there is apical involvement and no contraindications to anticoagulation; for those patients with persistent depression of left ventricular function at discharge, transition to warfarin could be considered. Considering the stress/catecholamine hypothesis, β-blockers may provide protection from a recurrence; however, the rate of ABS recurrence is low, estimated at 2%-10%, and, therefore, the use of β-blockers is not standard.16,27 Finally, follow-up echocardiograms performed 1 and 3 months postdischarge may be indicated for those patients without complete in-hospital restoration of ventricular function.
Given that symptoms of ABS frequently occur in the setting of emotional distress, it is often useful to assess such patients for treatable psychiatric conditions, such as panic disorder, generalized anxiety disorder, and major depressive disorder. Indeed, a recent study found that over one-half of 25 ABS patients had a prior history of anxiety or depression, far greater than a comparison group of MI patients.28 If such anxiety or depressive syndromes exist, they should be treated. Furthermore, even if patients do not meet criteria for a formal psychiatric syndrome, the extent to which their body’s physiology has responded to an emotional stressor suggests that they should be counseled about stress reduction and means of coping with distressing life events.
Drug names: nitroglycerin (Minitran, Nitro-Dur, and others), paroxetine (Paxil, Pexeva, and others), warfarin (Coumadin, Jantoven, and others).
Author affiliations: Department of Psychiatry, Massachusetts General Hospital, Boston.
Potential conflicts of interest: None reported.
Funding/support: None reported.
REFERENCES
1. Rugulies R. Depression as a predictor for coronary heart disease: a review and meta-analysis. Am J Prev Med. 2002;23(1):51-61. PubMed doi:10.1016/S0749-3797(02)00439-7
2. Barefoot JC, Helms MJ, Mark DB, et al. Depression and long-term mortality risk in patients with coronary artery disease. Am J Cardiol. 1996;78(6):613-617. PubMed doi:10.1016/S0002-9149(96)00380-3
3. Barth J, Schumacher M, Herrmann-Lingen C. Depression as a risk factor for mortality in patients with coronary heart disease: a meta-analysis. Psychosom Med. 2004;66(6):802-813. PubMed doi:10.1097/01.psy.0000146332.53619.b2
4. Rutledge T, Reis VA, Linke SE, et al. Depression in heart failure: a meta-analytic review of prevalence, intervention effects, and associations with clinical outcomes. J Am Coll Cardiol. 2006;48(8):1527-1537. PubMed doi:10.1016/j.jacc.2006.06.055
5. Dimsdale JE. What does heart disease have to do with anxiety? J Am Coll Cardiol. 2010;56(1):47-48. doi:10.1016/j.jacc.2010.04.013
6. Huffman JC, Smith FA, Blais MA, et al. Anxiety, independent of depressive symptoms, is associated with in-hospital cardiac complications after acute myocardial infarction. J Psychosom Res. 2008;65(6):557-563. PubMed doi:10.1016/j.jpsychores.2008.08.001
7. Moser DK, Riegel B, McKinley S, et al. Impact of anxiety and perceived control on in-hospital complications after acute myocardial infarction. Psychosom Med. 2007;69(1):10-16. PubMed doi:10.1097/01.psy.0000245868.43447.d8
8. Watkins LL, Blumenthal JA, Davidson JR, et al. Phobic anxiety, depression, and risk of ventricular arrhythmias in patients with coronary heart disease. Psychosom Med. 2006;68(5):651-656. PubMed doi:10.1097/01.psy.0000228342.53606.b3
9. Huffman JC, Pollack MH. Predicting panic disorder among patients with chest pain: an analysis of the literature. Psychosomatics. 2003;44(3):222-236. PubMed doi:10.1176/appi.psy.44.3.222
10. Smoller JW, Pollack MH, Wassertheil-Smoller S, et al. Panic attacks and risk of incident cardiovascular events among postmenopausal women in the Women’s Health Initiative Observational Study. Arch Gen Psychiatry. 2007;64(10):1153-1160. PubMed doi:10.1001/archpsyc.64.10.1153
11. Roshanaei-Moghaddam B, Katon W. Premature mortality from general medical illnesses among persons with bipolar disorder: a review. Psychiatr Serv. 2009;60(2):147-156. PubMed doi:10.1176/appi.ps.60.2.147
12. Brown S. Excess mortality of schizophrenia: a meta-analysis. Br J Psychiatry. 1997;171(6):502-508. PubMed doi:10.1192/bjp.171.6.502
13. Dote K, Sato H, Tateishi H, et al. Myocardial stunning due to simultaneous multivessel coronary spasms: a review of 5 cases. J Cardiol. 1991;21(2):203-214. PubMed
14. Gianni M, Dentali F, Grandi AM, et al. Apical ballooning syndrome or takotsubo cardiomyopathy: a systematic review. Eur Heart J. 2006;27(13):1523-1529. PubMed doi:10.1093/eurheartj/ehl032
15. Prasad A, Lerman A, Rihal CS. Apical ballooning syndrome (Tako-Tsubo or stress cardiomyopathy): a mimic of acute myocardial infarction. Am Heart J. 2008;155(3):408-417. PubMed doi:10.1016/j.ahj.2007.11.008
16. Sharkey SW, Lesser JR, Zenovich AG, et al. Acute and reversible cardiomyopathy provoked by stress in women from the United States. Circulation. 2005;111(4):472-479. PubMed doi:10.1161/01.CIR.0000153801.51470.EB
17. Nef HM, Möllmann H, Weber M, et al. Release pattern of cardiac biomarkers in left ventricular apical ballooning. Int J Cardiol. 2007;115(1):128-129. PubMed doi:10.1016/j.ijcard.2006.01.034
18. Bybee KA, Prasad A. Stress-related cardiomyopathy syndromes. Circulation. 2008;118(4):397-409. PubMed doi:10.1161/CIRCULATIONAHA.106.677625
19. Donohue D, Movahed MR. Clinical characteristics, demographics and prognosis of transient left ventricular apical ballooning syndrome. Heart Fail Rev. 2005;10(4):311-316. PubMed doi:10.1007/s10741-005-8555-8
20. Akashi YJ, Goldstein DS, Barbaro G, et al. Takotsubo cardiomyopathy: a new form of acute, reversible heart failure. Circulation. 2008;118(25):2754-2762. PubMed doi:10.1161/CIRCULATIONAHA.108.767012
21. Samuels MA. The brain-heart connection. Circulation. 2007;116(1):77-84. PubMed doi:10.1161/CIRCULATIONAHA.106.678995
22. Kurowski V, Kaiser A, von Hof K, et al. Apical and midventricular transient left ventricular dysfunction syndrome (tako-tsubo cardiomyopathy): frequency, mechanisms, and prognosis. Chest. 2007;132(3):809-816. PubMed doi:10.1378/chest.07-0608
23. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
24. Wittstein IS, Thiemann DR, Lima JA, et al. Neurohumoral features of myocardial stunning due to sudden emotional stress. N Engl J Med. 2005;352(6):539-548. PubMed doi:10.1056/NEJMoa043046
25. Ibanez B, Navarro F, Cordoba M, et al. Tako-tsubo transient left ventricular apical ballooning: is intravascular ultrasound the key to resolve the enigma? Heart. 2005;91(1):102-104. PubMed doi:10.1136/hrt.2004.035709
26. Bybee KA, Murphy J, Prasad A, et al. Acute impairment of regional myocardial glucose uptake in the apical ballooning (takotsubo) syndrome. J Nucl Cardiol. 2006;13(2):244-250. PubMed
27. Elesber AA, Prasad A, Lennon RJ, et al. Four-year recurrence rate and prognosis of the apical ballooning syndrome. J Am Coll Cardiol. 2007;50(5):448-452. PubMed doi:10.1016/j.jacc.2007.03.050
28. Summers MR, Lennon RJ, Prasad A. Pre-morbid psychiatric and cardiovascular diseases in apical ballooning syndrome (tako-tsubo/stress-induced cardiomyopathy): potential pre-disposing factors? J Am Coll Cardiol. 2010;55(7):700-701. PubMed doi:10.1016/j.jacc.2009.10.031