Objective: To describe the prevalence of vitamin D deficiency in psychiatric inpatients with serious mental illness. Associated clinical and sociodemographic factors are also explored.
Method: Data were collected using a retrospective review of medical records. Eligible subjects were individuals aged ≥ 18 years who were consecutively newly admitted to an adult inpatient teaching unit of a state psychiatric hospital from July 2012 through August 2013. The main outcome measure was prevalence rate of vitamin D deficiency in the target population. Vitamin D deficiency was defined as a level < 20 ng/mL. Psychiatric diagnoses were established using DSM-IV-TR criteria.
Results: Of 85 subjects, approximately two-thirds (67%) had a vitamin D level < 20 ng/mL. The mean vitamin D level was 18.4 ng/mL. Among the sociodemographic and clinical factors analyzed, only total serum protein (odds ratio = 0.33; CI, 0.12-0.88; P < .05) was associated with vitamin D deficiency.
Conclusions: The high prevalence of vitamin D deficiency with all the attendant physical and mental health burdens in vulnerable populations such as individuals with serious mental illness requires further large research studies. In the meantime, it seems prudent to institute routine screening for vitamin D deficiency in individuals with mental illness, especially those who are hospitalized.
Vitamin D Levels and Sociodemographic
and Clinical Correlates in Individuals With
Serious Mental Illness Admitted to an Acute Psychiatry Unit
ABSTRACT
Objective: To describe the prevalence of vitamin D deficiency in psychiatric inpatients with serious mental illness. Associated clinical and sociodemographic factors are also explored.
Method: Data were collected using a retrospective review of medical records. Eligible subjects were individuals aged ≥ 18 years who were consecutively newly admitted to an adult inpatient teaching unit of a state psychiatric hospital from July 2012 through August 2013. The main outcome measure was prevalence rate of vitamin D deficiency in the target population. Vitamin D deficiency was defined as a level < 20 ng/mL. Psychiatric diagnoses were established using DSM-IV-TR criteria.
Results: Of 85 subjects, approximately two-thirds (67%) had a vitamin D level < 20 ng/mL. The mean vitamin D level was 18.4 ng/mL. Among the sociodemographic and clinical factors analyzed, only total serum protein (odds ratio = 0.33; CI, 0.12–0.88; P < .05) was associated with vitamin D deficiency.
Conclusions: The high prevalence of vitamin D deficiency with all the attendant physical and mental health burdens in vulnerable populations such as individuals with serious mental illness requires further large research studies. In the meantime, it seems prudent to institute routine screening for vitamin D deficiency in individuals with mental illness, especially those who are hospitalized.
Prim Care Companion CNS Disord 2015;17(2):doi:10.4088/PCC.14m01726
© Copyright 2015 Physicians Postgraduate Press, Inc.
Submitted: September 30, 2014; accepted November 11, 2014.
Published online: April 30, 2015.
Corresponding author: Lakshminarayana Chekuri, MD, MPH, Department of Psychiatry, University of Arkansas for Medical Sciences, 4301 W. Markham, #554, Little Rock, AR 72205 ([email protected]).
The role of vitamin D in regulating calcium homeostasis and bone mineralization has long been well established.1 Only recently has there been a growing recognition of its potentially important physiologic role in maintaining general physical and mental health. Findings from studies suggest that vitamin D may have a beneficial role in cardiovascular health, the immune system, and the endocrine system and in prevention of certain types of cancers and infections.2–4
An emerging body of literature also suggests a plausible role for vitamin D in brain function and mental health.5–9 This role is supported by several lines of evidence. The presence of nuclear receptors for vitamin D in the brain and vitamin D’s involvement in the biosynthesis of neurotrophic factors, synthesis of nitric oxide synthase, and increased glutathione levels suggest an important role of vitamin D in brain function. Animal data indicate that tyrosine hydroxylase, the rate-limiting enzyme for the brain monoamines, is increased by vitamin D, and rats born to vitamin D–deficient dams have significant brain abnormalities.5 Several epidemiologic studies, mostly correlational in design, suggest that low levels of vitamin D are associated with mental illness, such as autism,6 cognitive impairment,7 depression,8 and schizophrenia.9 A recent large, prospective study that followed 1,658 elderly ambulatory subjects for a mean of 5.6 years found that individuals with baseline vitamin D deficiency had a nearly 2-fold statistically significant increase in the risk of development of new-onset dementia compared to those with normal vitamin D levels.10 Although not definitive, some intervention studies have shown that supplementation with vitamin D improves symptoms of depression.11,12 However, a 2011 Institute of Medicine report2 on vitamin D concluded that, other than its effects on bone health, there was insufficient evidence to support a “causal” role on other health outcomes and that further research was needed.2 However, vitamin D deficiency is a recognized medical condition that needs to and can easily be corrected.13,14
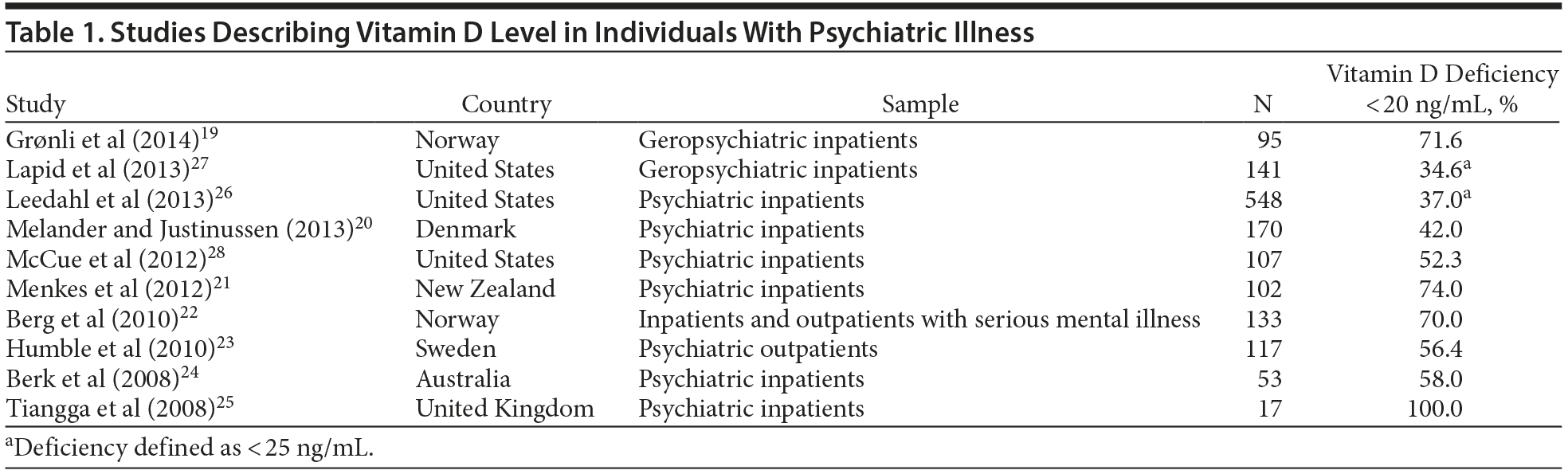
This is of concern because approximately 42% of adult Americans are estimated to have vitamin D levels < 20 ng/mL.15 Institutionalized individuals, such as the elderly living in nursing homes,16 postmenopausal women,17 and general medicine inpatients,18 are particularly at risk. Blacks and Hispanics tend to be at higher risk than whites.15 There is also increasing evidence that individuals with mental illness, especially those admitted to inpatient psychiatric units, are at higher risk of vitamin D deficiency than the general population. Several published studies suggest wide-spread vitamin D deficiency in individuals with mental illness in many countries (Table 1). Seven studies of individuals with mental illness conducted outside the United States reported vitamin D deficiency ranging from 42% to 100% of study subjects, higher than the prevalence in their respective general populations.19–25 Of 3 US studies26–28 of vitamin D deficiency in psychiatric inpatients, 226,27 reported a prevalence level lower than that in the general population. However, the prevalence cited in those 2 studies26,27 does not reflect the true prevalence of vitamin D deficiency in this population. Estimates in the first study26 were based on convenience sampling wherein only subjects who had a vitamin D level measured for unspecified reasons within 7 days of admission to the psychiatric unit over a 6-year period were included in the study with no mention of the total number of admissions during the same period. The second study27 was limited to a subset of subjects of the first study with similar limitations. Furthermore, the authors also acknowledge that about 17% of the study subjects had vitamin D supplementation prior to admission, which would have skewed their prevalence rate.26 The third US study by McCue and colleagues28 found that about 52% of 107 patients admitted consecutively to a large psychiatric facility in New York City within a 4-month period had vitamin D deficiency.
Based on the fact that vitamin D may potentially play an important role in general physical and mental health, that its deficiency can easily be corrected, and that individuals with serious mental illness on average have more physical problems and die 25 years earlier than the general population,29 correcting vitamin D deficiency in such a high-risk population seems reasonable. For these reasons, we have been screening for vitamin D deficiency in patients with serious mental illness admitted to an acute adult psychiatric unit of a state hospital and treating those who have vitamin D deficiency as part of routine clinical care. The objective of the present study is to describe the prevalence of vitamin D deficiency in this population and to evaluate associated clinical and sociodemographic factors.
METHOD
This study was a retrospective review of medical records limited to subjects who were consecutively newly admitted from July 2012 through August 2013 in the teaching unit of the Arkansas state hospital in Little Rock, which is the only public psychiatric hospital in Arkansas. Eligible subjects were individuals aged ≥ 18 years. If a subject was admitted more than once during the study period, only the first admission was included. There were no exclusion criteria. This study was approved by the institutional review board at University of Arkansas for Medical Sciences, Little Rock.
Data were collected from existing hospital and medical records. Patients admitted to the unit during the study period were identified through the hospital admissions database. Medical records of eligible subjects were reviewed, and relevant data items were directly entered into a Microsoft-Excel database. Data items collected included sociodemographic characteristics (age, gender, race), psychiatric (based on DSM-IV-TR criteria) and medical diagnosis, and serum biochemical markers (alkaline phosphatase, total protein, albumin, serum calcium, total cholesterol, high-density lipoprotein [HDL], and low-density lipoprotein [LDL]).
Vitamin D levels were estimated using serum 25-hydroxy vitamin D (25[OH]D). There is no general consensus on recommended cutoff serum levels of vitamin D to diagnose deficiency. The Institute of Medicine2 recommends that a level < 12 ng/mL indicates vitamin D deficiency, and levels between 12 ng/mL and 20 ng/mL indicate inadequacy. The Endocrine Society Clinical Practice Guidelines13 recommend a level < 20 ng/mL as indicative of vitamin D deficiency, and levels between 21 ng/mL and 29 ng/mL as indicative of vitamin D insufficiency. For the present study, we considered vitamin D levels < 20 ng/mL as vitamin D deficiency, and levels ≥ 20 ng/mL as normal. This categorization is clinically meaningful since it includes the Institute of Medicine’s recommendations on vitamin D deficiency and insufficiency levels. It also allowed comparison of our findings with existing reports on vitamin D levels in psychiatric patients. In an attempt to replicate the findings from previously published studies,23,28 subjects were divided into 2 age groups, 18–34 years and ≥ 35 years, and age was analyzed as a categorical variable.
Descriptive analysis of the sociodemographic and clinical characteristics of the study subjects, including mean levels of vitamin D and percent with deficiency, was performed. To identify factors associated with vitamin D deficiency, all study variables were compared between subjects with and without vitamin D deficiency. The χ2 statistic for categorical variables and independent t tests for continuous variables were used to evaluate this association. Using vitamin D as the dependent variable, a stepwise logistic regression analysis, which included any independent variable associated with vitamin D deficiency with a P value < .10, was performed. The analysis was conducted using IBM SPSS-22 software (Armonk, New York).
RESULTS
There were 144 total admissions to the teaching unit during the study period. Of these, 24 were excluded because they were repeat admissions (n = 14), transferred to another unit soon after admission (n = 3), or had a significant amount of missing information (n = 7). Of the 120 eligible subjects, 35 did not have a documented vitamin D level. Comparison of the 85 subjects with vitamin D data and the 35 subjects with missing vitamin D data revealed no significant differences in sociodemographic or clinical characteristics. The present study is limited to the 85 subjects with vitamin D data.
Study subjects were relatively young (mean age = 36.5 years, SD = 13 years), predominantly white (n = 48, 56.5%), and male (n = 50, 58.8%). Approximately 81% of subjects had a diagnosis of severe mental illness (schizophrenia [n = 21, 24.7%], schizoaffective disorder [n = 21, 24.7%], psychosis not otherwise specified [n = 6, 7.1%], bipolar disorder [n = 21, 24.7%]). Approximately 1 in 5 subjects had a diagnosis of hypertension (n = 19, 22.4%) and diabetes mellitus (n = 16, 18.8%). The mean body mass index was 28.9 kg/m2 with an SD of 6.4 kg/m2.
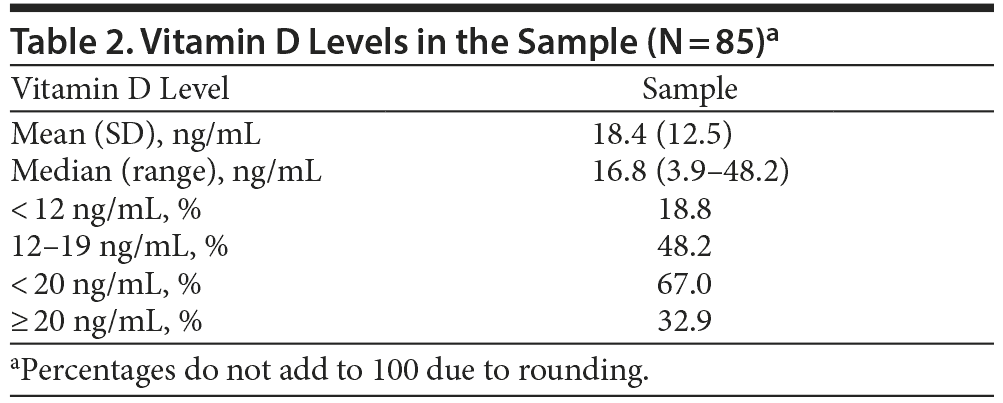
Vitamin D levels in study subjects are presented in Table 2. Approximately two-thirds (67%) of subjects had a vitamin D level < 20 ng/mL. Of these, 18.8% (n = 16) had vitamin D levels < 12 ng/mL, and 48.2% (n = 41) had vitamin D levels between 12 ng/mL and 19 ng/mL. Thirty-three percent of subjects (n = 28) had vitamin D levels ≥ 20 ng/mL. The median vitamin D level was 16.8 ng/mL, with a wide range of 3.9–48.2 ng/mL.
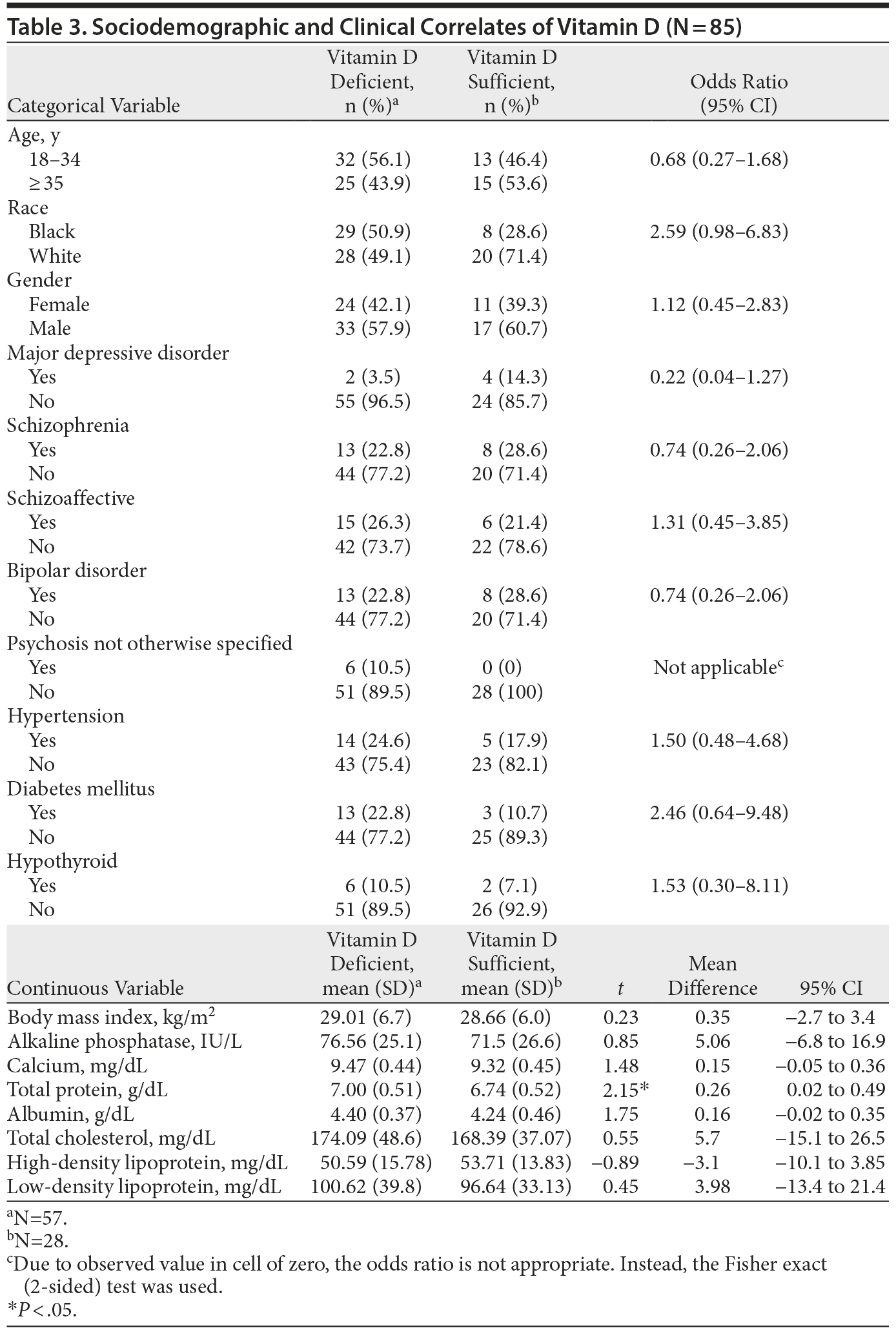
The bivariate analyses for association between sociodemographic and clinical characteristics with Vitamin D levels are presented in Table 3. Sociodemographic variables (age, gender, and race) were not significantly associated with vitamin D levels. Among the clinical variables analyzed, only total protein level had a significant modest inverse relationship with vitamin D level (P < .05). Four independent variables (race, diagnosis of major depressive disorder, total protein level, and albumin level) that were associated with vitamin D levels in the bivariate analysis with P values < .10 were included in a logistic regression model to evaluate the association with vitamin D deficiency (dependent variable) using a backward elimination method. Only higher total protein level was significantly predictive of vitamin D deficiency (odds ratio = 0.33; 95% CI, 0.12–0.88).
DISCUSSION
Data from the National Health and Nutrition Examination Survey (NHANES) 2005 to 200615 estimate that 41.6% of American adults had vitamin D deficiency (< 20 ng/mL). The present study found that 67% of subjects with a serious mental illness admitted to an acute psychiatric unit had vitamin D deficiency. Although our finding is higher than the 52.3% prevalence rate reported in a recently published US study of psychiatric inpatients with serious mental illness,28 it is consistent with a pattern supporting higher vitamin D deficiency states in this vulnerable population compared to the general population. Studies of vitamin D deficiency in individuals with mental illness conducted in subjects from other countries support a similar trend (Table 1).
Of the 3 recent studies of vitamin D deficiency in the United States, 2 were from the Midwest with subjects comprised of primarily white, female, geropsychiatric inpatients and did not estimate true prevalence. The third US study was from the Northeast, and its subjects comprised predominantly young, nonwhite male inpatients with serious mental illness. To our knowledge, our study is the first report on the prevalence of vitamin D deficiency in predominantly young white males with a serious mental illness admitted to a psychiatric unit located in the southern United States.
Factors, such as institutionalization,16 age,23,28 gender,17 and race,15 have been reported to be associated with vitamin D deficiency, but the published results are inconsistent.15,18 Since vitamin D levels in our subjects were drawn at admission, the high prevalence of deficiency cannot be attributed to institutionalization. While large studies involving the general population (such as the NHANES15) and inpatient medical patients18 did not find any association between age, gender, and vitamin D levels, studies in psychiatric patients found the younger age group (18–34 years) to be significantly associated with lower vitamin D levels.23,28 Our study did not show any significant association of age and gender with vitamin D deficiency. Similarly, nonwhites (such as blacks and Hispanics) are reported to have significantly lower vitamin D levels compared to whites.15 Even though it did not reach statistical significance (P = .05), blacks in our study had a higher prevalence rate of vitamin D deficiency. A low vitamin D level in dark-skinned people could partly be explained by higher melanin content in the nonwhite skin that absorbs some of the ultraviolet radiation from the sun. Another reason for this observation could also be due to genomic polymorphism that results in lower vitamin D–binding protein levels in blacks compared to whites.30
Several cross-sectional observational studies have reported significant association between low vitamin D levels and risk of schizophrenia,9 psychosis,31 poorer cognitive function and higher risk of Alzheimer’s dementia,7 and depression.8,32 However, recent studies in psychiatric inpatients found no significant association between vitamin D levels and cognitive function, depressive symptoms, and psychiatric diagnoses.19,26,27 Likewise, findings from our study also did not show any association between vitamin D deficiency and schizophrenia spectrum disorders, major depressive disorder, or psychosis. This finding may reflect the fact that vitamin D through its antioxidant effect33 plays a long-term role in the neurodevelopmental6,9 and neurodegenerative10 processes and impacts global brain function instead of specific cognitive, mood, or psychotic domains.
The role of vitamin D deficiency in the metabolic syndrome has received considerable attention in the literature.15,18,34,35 Low levels of vitamin D are reported to be associated with insulin resistance, elevated blood pressure, and high total cholesterol and triglyceride levels. Vitamin D is implicated in insulin action and its pathways, and vitamin D deficiency is reported to have a correlation with development of type 2 diabetes mellitus.36 Findings from the NHANES15 suggest that, in the general population, vitamin D deficiency is more prevalent in individuals with obesity, hypertension, and low levels of HDLs. Our study showed no significant association between vitamin D deficiency and a diagnosis of hypertension, diabetes mellitus, hypothyroidism, body mass index, total cholesterol, or HDL and LDL levels. In a study involving general medical inpatients,18 low serum concentrations of ionized calcium and albumin and higher serum concentrations of alkaline phosphatase were predictive of vitamin D deficiency. Results from our study showed no significant association between these variables and vitamin D deficiency. Only serum total protein levels were inversely correlated with vitamin D deficiency. Clinical and biological relevance of such serum biomarkers in vitamin D deficiency need to be explored in future studies.
Our study had several limitations. The study was limited to subjects from 1 psychiatric unit of a state hospital, which may limit its generalizability. However, the prevalence rate of vitamin D deficiency is consistent with the extant literature and is a matter of concern. Vitamin D data were missing for about 30% of the subjects and that could have influenced our results. However, analyses of the sociodemographic and clinical characteristics of those with missing and nonmissing vitamin D data were essentially similar, suggesting that the missing data may not have affected the results significantly. Finally, the study had a relatively small sample size with limited statistical power to detect associations reported in larger studies. However, the primary objective of the study was to estimate the prevalence of vitamin D deficiency in this population.
The high prevalence rate of vitamin D deficiency with all the attendant physical and mental health burdens in this vulnerable population requires further large research studies. In the meantime, it seems prudent to institute routine screening for vitamin D deficiency in individuals with mental illness, especially those who are hospitalized. Psychiatrists routinely screen patients for general medical conditions with a battery of tests. Adding a vitamin D screen to this battery of tests does not pose unnecessary burden to the patient. This screening is important for the following reasons. Vitamin D deficiency is a recognized clinical condition that can be easily corrected with supplementation. Although not definitive, there is growing evidence that vitamin D appears to play an important role in maintaining general physical and mental health. Findings from our study along with other studies show a high prevalence of vitamin D deficiency in this population. Finally, individuals with serious mental illness on average have more physical problems and die 25 years earlier than the general population.29
Author affiliations: Department of Applied Gerontology, University of North Texas, Denton (Dr Chekuri); Department of Psychiatry, University of Arkansas for Medical Sciences, Little Rock (Drs Chekuri, Thapa, Mittal, and Messias); Department of Psychiatry, Arkansas State Hospital, Little Rock (Dr Thapa); Gerontology Program, School of Social Work, University of Arkansas at Little Rock (Dr Turturro); and Veterans Administration South Central (VISN 16) Mental Illness Research, Education, and Clinical Center, Central Arkansas Veterans Healthcare System, North Little Rock, Arkansas (Dr Mittal).
Potential conflicts of interest: None reported.
Funding/support: None reported.
REFERENCES
1. Anderson PH, Turner AG, Morris HA. Vitamin D actions to regulate calcium and skeletal homeostasis. Clin Biochem. 2012;45(12):880–886. doi:10.1016/j.clinbiochem.2012.02.020 PubMed
2. Ross AC, Taylor CL, Yaktine AL, et al, eds. Dietary Reference Intakes for Calcium and Vitamin D. Institute of Medicine Committee to Review Dietary Reference Intakes for Vitamin D and Calcium. Washington, DC: National Academies Press; 2011.
3. Glade MJ. Vitamin D: health panacea or false prophet? Nutrition. 2013;29(1):37–41. doi:10.1016/j.nut.2012.05.010 PubMed
4. Garland CF, Garland FC, Gorham ED, et al. The role of vitamin D in cancer prevention. Am J Public Health. 2006;96(2):252–261. doi:10.2105/AJPH.2004.045260 PubMed
5. McCann JC, Ames BN. Is there convincing biological or behavioral evidence linking vitamin D deficiency to brain dysfunction? FASEB J. 2008;22(4):
982–1001. doi:10.1096/fj.07-9326rev PubMed
6. Kočovská E, Fernell E, Billstedt E, et al. Vitamin D and autism: clinical review. Res Dev Disabil. 2012;33(5):1541–1550. doi:10.1016/j.ridd.2012.02.015 PubMed
7. Balion C, Griffith LE, Strifler L, et al. Vitamin D, cognition, and dementia:
a systematic review and meta-analysis. Neurology. 2012;79(13):
1397–1405. doi:10.1212/WNL.0b013e31826c197f PubMed
8. Parker G, Brotchie H. ‘D’ for depression: any role for vitamin D? ‘food for thought’ II. Acta Psychiatr Scand. 2011;124(4):243–249. doi:10.1111/j.1600-0447.2011.01705.x PubMed
9. McGrath JJ, Burne TH, Féron F, et al. Developmental vitamin D deficiency and risk of schizophrenia: a 10-year update. Schizophr Bull. 2010;36(6):
1073–1078. doi:10.1093/schbul/sbq101 PubMed
10. Littlejohns TJ, Henley WE, Lang IA, et al. Vitamin D and the risk of dementia and Alzheimer disease. Neurology. 2014;83(10):920–928. doi:10.1212/WNL.0000000000000755 PubMed
11. Jorde R, Sneve M, Figenschau Y, et al. Effects of vitamin D supplementation on symptoms of depression in overweight and obese subjects: randomized double blind trial. J Intern Med. 2008;264(6):599–609. doi:10.1111/j.1365-2796.2008.02008.x PubMed
12. Khoraminya N, Tehrani-Doost M, Jazayeri S, et al. Therapeutic effects of vitamin D as adjunctive therapy to fluoxetine in patients with major depressive disorder. Aust N Z J Psychiatry. 2013;47(3):271–275. doi:10.1177/0004867412465022 PubMed
13. Holick MF, Binkley NC, Bischoff-Ferrari HA, et al; Endocrine Society. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):
1911–1930. doi:10.1210/jc.2011-0385 PubMed
14. Rosen CJ. Clinical practice: vitamin D insufficiency. N Engl J Med. 2011;364(3):248–254. doi:10.1056/NEJMcp1009570 PubMed
15. Forrest KY, Stuhldreher WL. Prevalence and correlates of vitamin D deficiency in US adults. Nutr Res. 2011;31(1):48–54. doi:10.1016/j.nutres.2010.12.001 PubMed
16. Kennel KA, Drake MT, Hurley DL. Vitamin D deficiency in adults: when
to test and how to treat. Mayo Clin Proc. 2010;85(8):752–757, quiz
757–758. doi:10.4065/mcp.2010.0138 PubMed
17. Gaugris S, Heaney RP, Boonen S, et al. Vitamin D inadequacy among postmenopausal women: a systematic review. QJM. 2005;98(9):667–676. doi:10.1093/qjmed/hci096 PubMed
18. Thomas MK, Lloyd-Jones DM, Thadhani RI, et al. Hypovitaminosis D in medical inpatients. N Engl J Med. 1998;338(12):777–783. doi:10.1056/NEJM199803193381201 PubMed
19. Grønli O, Kvamme JM, Jorde R, et al. Vitamin D deficiency is common in psychogeriatric patients, independent of diagnosis. BMC Psychiatry. 2014;14(1):134. doi:10.1186/1471-244X-14-134 PubMed
20. Melander KR, Justinussen K. Vitamin D plasma levels during summer in a psychiatric population are comparable to the winter levels of healthy individuals. Dan Med J. 2013;60(3):A4598. PubMed
21. Menkes DB, Lancaster K, Grant M, et al. Vitamin D status of psychiatric inpatients in New Zealand’s Waikato region. BMC Psychiatry. 2012;12(1):68. doi:10.1186/1471-244X-12-68 PubMed
22. Berg AO, Melle I, Torjesen PA, et al. A cross-sectional study of vitamin D deficiency among immigrants and Norwegians with psychosis compared to the general population. J Clin Psychiatry. 2010;71(12):1598–1604. doi:10.4088/JCP.09m05299yel PubMed
23. Humble MB, Gustafsson S, Bejerot S. Low serum levels of 25-hydroxyvitamin D (25-OHD) among psychiatric outpatients in Sweden: relations with season, age, ethnic origin and psychiatric diagnosis. J Steroid Biochem Mol Biol. 2010;121(1–2):467–470. doi:10.1016/j.jsbmb.2010.03.013 PubMed
24. Berk M, Jacka FN, Williams LJ, et al. Is this D vitamin to worry about? vitamin D insufficiency in an inpatient sample. Aust N Z J Psychiatry. 2008;42(10):874–878. doi:10.1080/00048670802345516 PubMed
25. Tiangga E, Gowda A, Dent JA. Vitamin D deficiency in psychiatric inpatients and treatment with daily supplements of calcium and ergocalciferol. Psychiatr Bull. 2008;32(10):390–393. doi:10.1192/pb.bp.107.019109
26. Leedahl DD, Cunningham JL, Drake MT, et al. Hypovitaminosis D in psychiatric inpatients: clinical correlation with depressive symptoms, cognitive impairment, and prescribing practices. Psychosomatics. 2013;54(3):257–262. doi:10.1016/j.psym.2012.07.005 PubMed
27. Lapid MI, Drake MT, Geske JR, et al. Hypovitaminosis D in psychogeriatric inpatients. J Nutr Health Aging. 2013;17(3):231–234. doi:10.1007/s12603-012-0383-7 PubMed
28. McCue RE, Charles RA, Orendain GC, et al. Vitamin D deficiency among psychiatric inpatients. Prim Care Companion CNS Disord. 2012;14(2):doi:10.4088/PCC.11m01230. PubMed
29. Parks J, Svendsen D, Singer P, et al. Morbidity and Mortality in People With Serious Mental Illness. Alexandria, VA: National Association of State Mental Health Program Directors (NASMHPD) Medical Directors Council; 2006.
30. Holick MF. Bioavailability of vitamin D and its metabolites in black and white adults. N Engl J Med. 2013;369(21):2047–2048. doi:10.1056/NEJMe1312291 PubMed
31. Belvederi Murri M, Respino M, Masotti M, et al. Vitamin D and psychosis: mini meta-analysis. Schizophr Res. 2013;150(1):235–239. doi:10.1016/j.schres.2013.07.017 PubMed
32. Anglin RE, Samaan Z, Walter SD, et al. Vitamin D deficiency and depression in adults: systematic review and meta-analysis. Br J Psychiatry. 2013;202(2):
100–107. doi:10.1192/bjp.bp.111.106666 PubMed
33. Noyan T, Balaharoğlu R, Kömüroğlu U. The oxidant and antioxidant effects of 25-hydroxyvitamin D3 in liver, kidney and heart tissues of diabetic rats. Clin Exp Med. 2005;5(1):31–36. doi:10.1007/s10238-005-0061-8 PubMed
34. Ford ES, Ajani UA, McGuire LC, et al. Concentrations of serum vitamin D and the metabolic syndrome among US adults. Diabetes Care. 2005;28(5):1228–1230. doi:10.2337/diacare.28.5.1228 PubMed
35. Lind L, Hänni A, Lithell H, et al. Vitamin D is related to blood pressure and other cardiovascular risk factors in middle-aged men. Am J Hypertens. 1995;8(9):894–901. doi:10.1016/0895-7061(95)00154-H PubMed
36. Ozfirat Z, Chowdhury TA. Vitamin D deficiency and type 2 diabetes. Postgrad Med J. 2010;86(1011):18–25, quiz 24. doi:10.1136/pgmj.2009.078626 PubMed
Enjoy this premium PDF as part of your membership benefits!


