Objective: To examine metabolic factors among overweight/obese individuals with binge-eating disorder (BED) and non-binge-eating overweight/obese (NBO) patients recruited from primary care and to examine and compare medication use by these groups.
Method: Participants were 102 adults recruited for a weight loss study within primary care centers who were assessed for BED (28 [38%] met DSM-5 BED criteria). Participants completed a medication log, had physiologic measurements taken, and were evaluated for the presence of metabolic syndrome using 2 methods. Data were collected between February 2012 and October 2012.
Results: The BED group had a higher mean body mass index (BMI), a higher pulse, and a larger waist circumference than the NBO group. Of the sample, 65% reported current medication use (prescription and/or over-the-counter medications): 19.6% took 3 to 4 medications and 15.7% took ≥ 5 medications. Aside from vitamin and over-the-counter allergy pill use, there were no differences in medication use between BED and NBO patients. Full metabolic syndrome (≥ 3 criteria met) was present in 31.5% of the sample when using objective measurement alone, and 39.1% of the sample when defined by objective measurement and pharmacologic management. No significant differences were observed regardless of definition.
Conclusions: Despite higher BMI, pulse, and waist circumference, the current sample of BED patients in primary care did not present with poorer metabolic health than NBO patients.
Medication Use and Metabolic Syndrome Among
Overweight/Obese Patients With and Without
Binge-Eating Disorder in a Primary Care Sample
ABSTRACT
Objective: To examine metabolic factors among overweight/obese individuals with binge-eating disorder (BED) and non–binge-eating overweight/obese (NBO) patients recruited from primary care and to examine and compare medication use by these groups.
Method: Participants were 102 adults recruited for a weight loss study within primary care centers who were assessed for BED (28 [38%] met DSM-5 BED criteria). Participants completed a medication log, had physiologic measurements taken, and were evaluated for the presence of metabolic syndrome using 2 methods. Data were collected between February 2012 and October 2012.
Results: The BED group had a higher mean body mass index (BMI), a higher pulse, and a larger waist circumference than the NBO group. Of the sample, 65% reported current medication use (prescription and/or over-the-counter medications): 19.6% took 3 to 4 medications and 15.7% took ≥ 5 medications. Aside from vitamin and over-the-counter allergy pill use, there were no differences in medication use between BED and NBO patients. Full metabolic syndrome (≥ 3 criteria met) was present in 31.5% of the sample when using objective measurement alone, and 39.1% of the sample when defined by objective measurement and pharmacologic management. No significant differences were observed regardless of definition.
Conclusions: Despite higher BMI, pulse, and waist circumference, the current sample of BED patients in primary care did not present with poorer metabolic health than NBO patients.
Prim Care Companion CNS Disord 2015;17(5):doi:10.4088/PCC.15m01816
© Copyright 2015 Physicians Postgraduate Press, Inc.
aDepartment of Psychology, VA Connecticut Healthcare System, West Haven
bDepartment of Psychiatry, Yale School of Medicine, New Haven, Connecticut
cDepartment of Psychiatry, University of Maryland School of Medicine, Baltimore, Maryland
*Corresponding author: Jessica A. Barber, PhD, VA Connecticut Healthcare System, 950 Campbell Ave, Psychology 116B, West Haven, CT 06516
([email protected]).
Binge-eating disorder (BED), previously a provisional diagnosis, has officially been incorporated into the DSM-51 and has been the subject of research as a clinical syndrome independent of other eating disorders for years.1,2 BED is defined by the presence of recurrent, discrete periods of eating an unusually large amount of food, accompanied by a subjective sense of loss of control over eating without regular compensatory behaviors.1 Kessler et al3 estimate that the lifetime prevalence of BED in the United States is 2.6%.
Overweight/obesity is common in patients with BED, with 34.9% classified as overweight (body mass index [BMI] = 25–29.9 kg/m2) and 38.1% classified as obese (BMI ≥ 30 kg/m2). The prevalence of overweight and obesity has risen dramatically in the United States, with estimates now at 69.2%.4 Obesity is a heterogeneous clinical syndrome associated with significant medical and psychiatric comorbidity and higher levels of health care system utilization.5–9 Given the high comorbidity of overweight/obesity with BED, the current study sought to understand differences in metabolic factors and medication use between overweight/obese patients with BED compared with non–binge-eating overweight/obese (NBO) patients.
Regardless of weight, BED status is associated with increased medical comorbidity and health care utilization.5,10–14 However, many of these comparisons have been done using patient self-report of physical health or measures of health-related quality of life.5,11,14,15 There are limited data about the impact of BED on objectively assessed chronic illness (vs self-reported symptoms), including metabolic syndrome.
Metabolic syndrome is defined by the presence of 3 of 5 of the following risk factors: elevated fasting blood glucose, low serum high-density lipoprotein (HDL) cholesterol level, hypertriglyceridemia, hypertension, and central adiposity (Table 1).16–18 Metabolic syndrome significantly increases the risk for cardiovascular disease, including major coronary events, diabetes, and all-cause mortality.19–21

Binge-eating episodes may be related to metabolic abnormalities,10,22,23 but the findings are mixed. Some studies have observed a positive association between binge eating and dysregulation of blood lipids and glucose,10,12,13,24–26 but this finding is not consistent.27–29 Similarly, discrepancies exist as to whether or not overweight/obese individuals with BED differ metabolically from their NBO counterparts.11,13,20,25 The studies reporting no differences,11,20 however, did not examine full metabolic syndrome but rather elements thereof, and there are limited data on objectively measured components of metabolic syndrome (vs self-report data). Thus far, only 1 study10 appears to have examined metabolic health in BED with objectively measured physiologic data for all components of metabolic syndrome. Abraham and colleagues10 found that BED was associated with increased triglycerides, poorer fasting glucose, lower HDL, hypertension, and increased risk of obesity and type 2 diabetes. However, this was a population-based study that did not specifically compare BED to NBO.10 The current study sought to examine these factors in a sample of overweight/obese individuals with BED and an NBO comparison group.
Long-term medication management of obesity, metabolic syndrome, and related health sequelae has become routine, but to our knowledge, no previous research has explored the use of medications for metabolic dysregulation and other common, chronic health conditions among individuals with BED nor have previous studies compared these variables between individuals with BED versus NBO. Therefore, it is difficult to fully appreciate the burden of chronic illness among this group and how it may be different from that of nonbingeing obese patients.
Greater clarity regarding the relationship between BED and chronic health conditions, particularly metabolic syndrome, and medication use is important for several reasons. Metabolic syndrome, being associated with several other chronic health problems, has implications for long-term health, health care system resource utilization, and cost.30–36 Concerns regarding cost and utilization are particularly relevant when individuals are prescribed long-term pharmacologic management. The primary care setting is ideal for examining these questions, as primary care providers have regular contact with their patients and frequent opportunities to screen for disordered eating patterns and are the first line in terms of treatment for the sequelae of overweight/obesity.
In this article, we aim to (1) examine metabolic factors in a primary care sample of overweight/obese BED and NBO patients including presence of metabolic syndrome and (2) compare the use of medications by these 2 groups. We hypothesized that the BED group would be more likely to have metabolic measurements outside the normal range and to meet full metabolic syndrome criteria. We also hypothesized that the BED group would report taking a greater number of medications than the NBO group. Our findings have implications for the understanding of BED as a clinical syndrome, the impact of BED on health above and beyond the impact of overweight/obesity alone, and the impact of BED on health care utilization.
METHOD
Participants
Participants were 102 adult (80 women, 22 men) overweight/obese (BMI ≥ 25 kg/m2) individuals recruited for weight loss treatment from local primary care centers. Participants were recruited through primary care provider referrals and flyers placed in waiting/patient rooms for a behavioral weight loss treatment study being performed in primary care centers in a large university-based medical health care center in an urban setting. Based on a structured clinical interview, 28 (38%) participants met DSM-5 BED criteria.1 The mean age of the sample was 47.27 years (SD = 10.75). Ethnicity was white not Hispanic (n = 73, 71.6%), black (n = 21, 20.6%), white Hispanic (n = 9, 8.9%), and other/multiracial/biracial (n = 8, 7.8%). Most participants were married (n = 59, 57.8%) and had at least some college education (n = 89, 88.2%). Exclusion criteria included BMI ≥ 55 kg/m2; over 65 years of age; current/planned pregnancy; breastfeeding; schizophrenia, bipolar disorder; and uncontrolled medical illnesses including uncontrolled liver disease, uncontrolled hypertension (> 160/95), uncontrolled thyroid disease (thyroid-stimulating hormone > 6.75), and uncontrolled diabetes (HbA1c > 8.0%). These exclusion criteria are consistent with other studies on metabolic syndrome and BED.37
Measures
Medication list. Participants completed a paper and pencil medication log that elicited the name, dose, and frequency of all current medications. This measure was completed at home for optimal accuracy.
Physiologic measurements. Height was measured. Participants were weighed in light clothing with shoes removed using a large-capacity digital scale. Blood pressure and pulse were measured using automated blood pressure monitors in a standardized procedure, and recorded readings were an average of 2 measurements. Blood was drawn and analyzed by Quest Diagnostics (Madison, New Jersey).
BMI. BMI was calculated based on participant weight and height using the following conversion formula (weight [lb] ÷ height2 [in2] × 703).
Metabolic syndrome. Metabolic syndrome was defined in 2 ways: (1) based on objective measurements only and (2) based on objective measurements and pharmacologic management as outlined by the Mayo Clinic18 and National Institutes of Health National Heart, Lung, and Blood Institute.17 Because previous BED and metabolic syndrome literature is not consistent in terms of which definition is used, with both appearing with similar frequency,25,38,39,40 and to further examine explanations for inconsistencies in the literature, we present results based on both definitions.
Procedures
Study procedures were reviewed and approved by the Yale University School of Medicine Institutional Review Board, New Haven, Connecticut. Data were collected between February 2012 and October 2012. Participants provided informed consent. They were assessed for BED by master- or doctoral-level psychology clinicians trained in eating and weight disorders using gold standard semistructured interviews edited for DSM-5 criteria (ie, Eating Disorder Examination41 and the Structured Clinical Interview for DSM-IV-TR Axis I Disorders42). Participants’ height, weight, blood pressure, and pulse rate were recorded by trained clinicians. The medication log, which was completed at home, was collected. Blood was drawn.
Data Analysis
Descriptive statistics were computed to examine participant characteristics. We compared clinical characteristics, metabolic syndrome, and medication use between the BED and NBO groups; χ2 tests were used for categorical variables, and Student t tests were used for continuous variables.
RESULTS
Clinical Characteristics
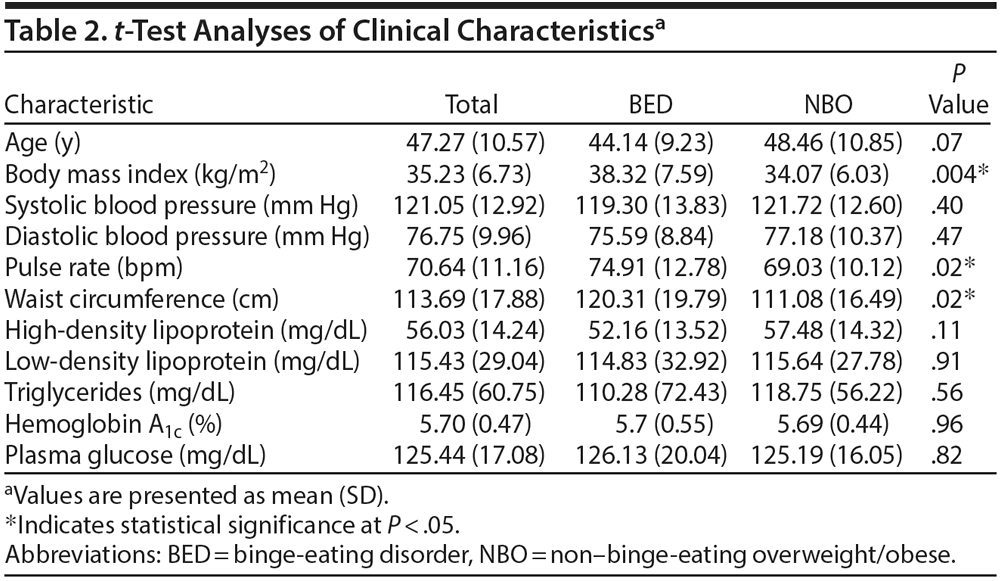
Clinical characteristics of the sample are presented in Table 2. Mean BMI was 35.23 (SD = 6.73; range, 25–55), indicating that, on average, participants in this sample were obese. Mean waist circumference was 113.69 cm (SD=17.88). Both systolic (mean = 121.05 mm Hg, SD = 12.92) and diastolic (mean = 76.75 mm Hg, SD=9.96) blood pressure were well-controlled, and pulse rate averaged 70.64 bpm (SD = 11.16). Both HDL (mean = 56.03 mg/dL, SD = 14.24) and low-density lipoprotein (mean = 115.43 mg/dL, SD = 29.04) cholesterol, as well as triglycerides (mean = 116.45 mg/dL, SD = 60.75), were well controlled. Average hemoglobin A1c was 5.70% (SD = 0.47) and average fasting blood glucose was 125.44 mg/dL (SD = 17.08), both of which fall below the diagnostic criteria for diabetes but are within the range of impaired fasting glucose, suggesting individuals with either impaired fasting glucose or diabetes were represented in this sample. As can be seen in Table 2, the BED group had a higher mean BMI (P = .004), a higher pulse (P = .02), and a larger waist circumference (P = .02) compared with the NBO group. There were no other differences between the groups in terms of demographics (age, gender, education) or physiologic variables.
Medication Use
The 15 most commonly reported medications by category are listed in Table 3. The most commonly used medication was over-the-counter (OTC) vitamins, with 24% of the sample reporting usage. A significantly larger proportion of the NBO group reported using OTC vitamins compared with the BED group (28% vs 11%, P = .048). Blood pressure medications were also commonly used in this sample (22% of the sample, with no differences between the BED and NBO groups). Sleep aids (3%) and arthritis medications (2%) were the least commonly reported medications in this sample. Only 2 medications were used differently by the BED and NBO groups: OTC vitamins and OTC allergy medications, with greater use reported in both cases by the NBO group.
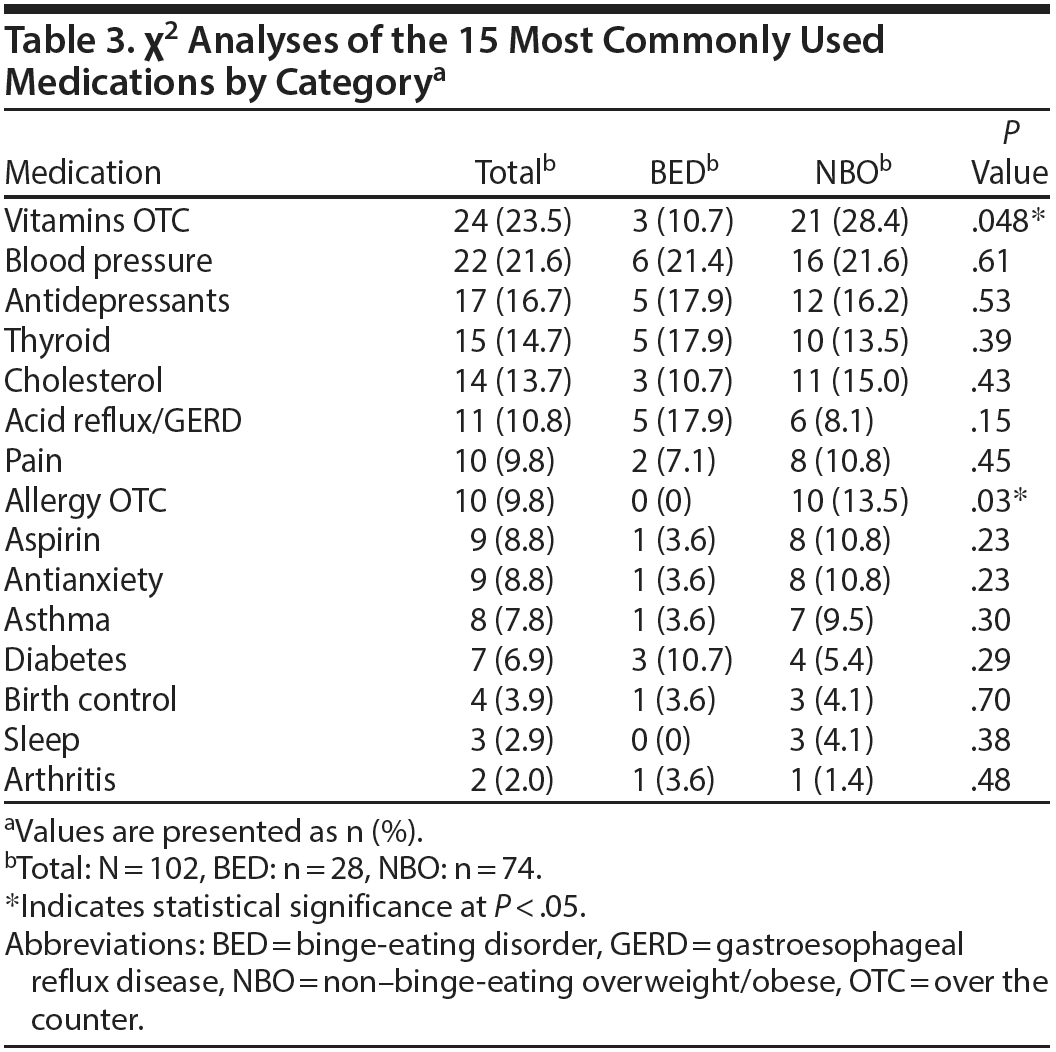
Sixty-five percent of the sample reported taking at least 1 medication (prescription or over-the-counter medication, excluding OTC vitamins), with no significant differences in the rates of medication use between BED and NBO individuals (46% vs 30%, respectively). Prescription usage for the BED group was none = 35.3%, 1 to 2 = 29.4%, 3 to 4 = 19.6%, and ≥ 5 = 15.7%. Rates for the NBO group were none = 31.1%, 1 to 2 = 32.4%, 3 to 4 = 20.3%, and ≥ 5 = 16.2%. Differences observed were nonsignificant.
Metabolic Syndrome
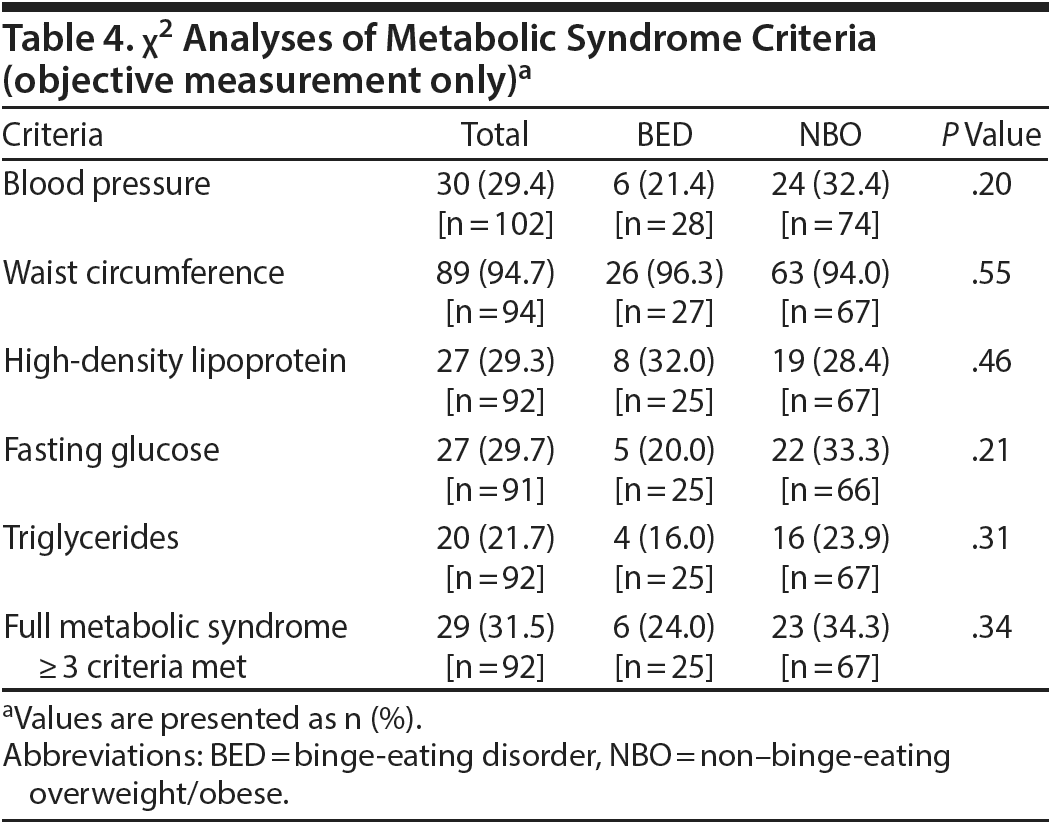
Objective measurement alone. Table 4 presents the percentage of participants meeting each of the diagnostic criteria for metabolic syndrome based on objective measurement for each of the criteria, as well as the percentage of participants meeting the cutoff for full metabolic syndrome (≥ 3 criteria met). In the full sample, 29.4% met criteria for elevated blood pressure, 94.7% met criteria for large waist circumference, 29.3% met criteria for low HDL, 29.7% met criteria for elevated fasting glucose, and 21.7% met criteria for high triglycerides. Full metabolic syndrome (≥ 3 criteria met) was present in 31.5% of the sample, with no significant differences in rates of metabolic syndrome in the BED (24.0%) versus NBO (34.3%) groups.
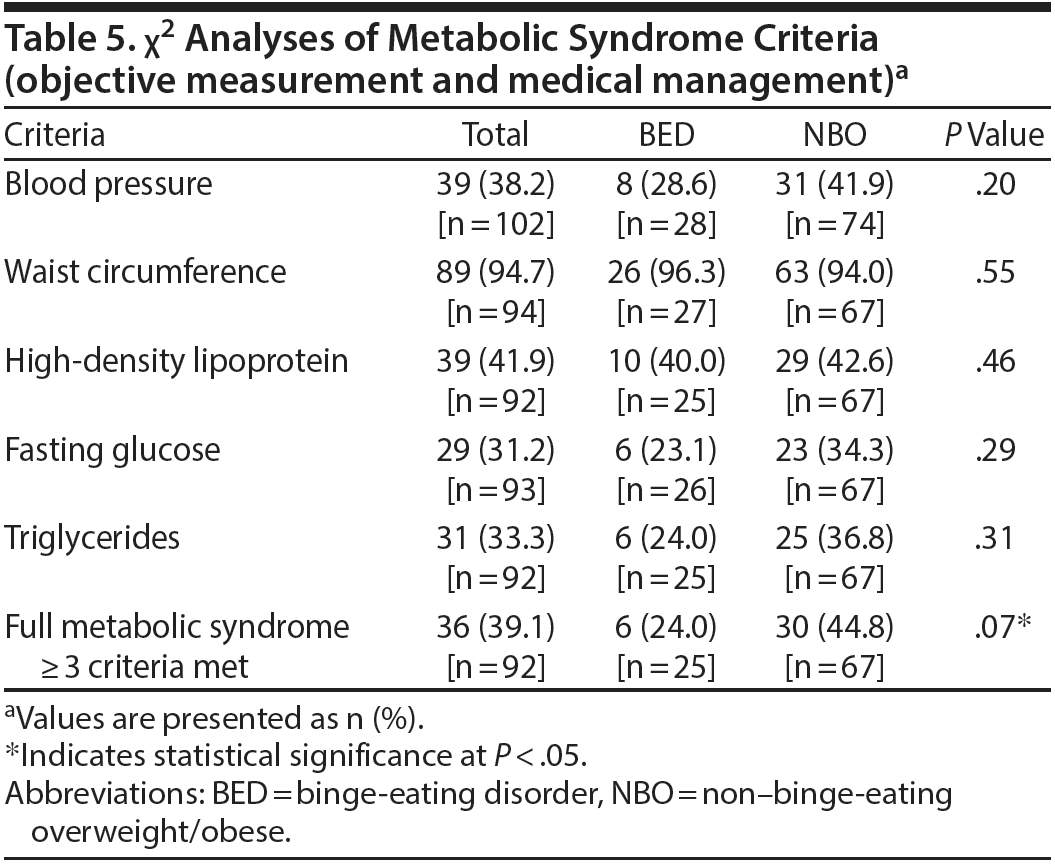
Objective measurement and medical management for metabolic syndrome criteria. Table 5 presents the percentage of participants meeting each of the diagnostic criteria for metabolic syndrome based on the more inclusive Mayo Clinic and National Institutes of Health National Heart, Lung, and Blood Institute definition (objective measurement and medical management for each of the criteria), as well as the percentage of participants meeting the cutoff for full metabolic syndrome (≥ 3 criteria met). In the full sample, 38.2% met criteria for elevated blood pressure (ie, ≥ 130/85), 94.7% met criteria for large waist circumference (≥ 35 in for women, ≥ 40 in for men), 41.9% met criteria for low HDL (< 50 mg/dL for women, < 40 mg/dL for men), 31.2% met criteria for elevated fasting glucose (≥ 100 mg d/L), and 33.3% met criteria for high triglycerides (≥ 150 mg/dL). Full metabolic syndrome (≥ 3 criteria met) was present in 39.1% of the sample, with no significant group differences.
DISCUSSION
In this treatment-seeking, primary care sample, overweight/obese individuals with BED were surprisingly similar to NBO patients on many objectively measured physiologic indicators of health. Although waist circumference, BMI, and pulse were higher for the BED group, prevalence of metabolic syndrome based on objectively measured physical criteria alone (vs objective measurement and/or medication management criteria) did not differ. Despite no differences in medication use between the groups, metabolic syndrome was more prevalent in NBO patients when the more inclusive definition was used.
To our knowledge, no other study has compared prescription medication use in BED versus NBO. This is important because there may be differences in this element of chronic disease management between the groups that would suggest differential disease risk/health outcomes. One study found sleep, tranquilizer, and pain medication use to be higher in BED versus non-BED,43 but the researchers did not focus on overweight/obese individuals (ie, BED vs NBO) and did not include the range of medications examined here. With the exception that the NBO group used more OTC allergy medications and vitamins, we found no significant differences between the groups in terms of medication use. It is unclear why the NBO group reported higher rates of OTC allergy medication and vitamin use. Perhaps differences exist between these groups in terms of health self-management with NBO individuals taking a more active role, but future research is needed to explore this hypothesis.
We found little evidence that overweight/obese individuals with BED have poorer health than NBO individuals. The BED group had higher waist circumference, BMI, and pulse than the NBO group, which is consistent with our hypothesis and previous literature. Yet, we found no statistically significant differences in other metabolic indicators of health including cholesterol, blood lipids, and blood glucose. Prevalence of metabolic syndrome was statistically equivalent between the groups. Medication use, including type and number of medication used, and pharmacologic treatment for components of metabolic syndrome specifically, was equivalent across groups. In other samples of overweight/obese individuals with BED, metabolic syndrome has been found to range from 2% to 60%,25,27,38,44 with US estimates at nearly 30% for overweight and 65% for obese adults,44 whereas the approximations are closer to 3% for their NBO counterparts. Our prevalence for both groups fell within the midrange of previous estimates, although BED group rates were at the lower end of this range. The way in which metabolic syndrome is defined may play a role in the varying rates as some studies citing the ATPIII criteria as their definition of metabolic syndrome specify the inclusion of pharmacologic management as meeting the definition of metabolic syndrome 25,39,40 and others do not.27,38 It is unclear, therefore, if researchers included pharmacologic management in their definition. Since the metabolic syndrome definitions used may influence observed rates, it is difficult to compare findings across studies, which is compounded by the relative lack of research comparing metabolic syndrome between BED and NBO.
Measurement is another factor. In the present study, we directly measured the metabolic parameters of metabolic syndrome, but many studies rely on patients’ self-reported diagnoses/symptoms. Of the 8 articles5,11,14,15,25,45,46,47 we reviewed that presented comparisons of health between overweight/obese individuals with BED versus NBO, one47 utilized objective measurement of a health index other than BMI. They studied both BMI and serum lipids, and found no difference between the BED and NBO groups on either.
How BED is defined, including the differences between the DSM-IV-TR and DSM-5, is also important to consider. We present some of the first research on metabolic syndrome and BED using DSM-5 criteria, which differs from DSM-IV criteria in terms of frequency and duration of binges: once weekly for 3 months (DSM-5)1 versus twice weekly for 6 months (DSM-IV-TR).48 It is possible that using DSM-5 criteria resulted in inclusion of individuals with less severe BED,49 thereby influencing the metabolic syndrome prevalence in our BED group; however, recent data suggest that DSM-5 criteria result in similar rates of diagnoses as DSM-IV.50 Continued metabolic syndrome research with the new BED criteria is necessary.
Finally, we studied a primary care sample, which differs in important ways from community, psychiatric, or other samples of overweight/obese individuals with and without BED. For example, individuals seen in primary care may be healthier in general.51 Despite this possibility, 20% of our sample was taking 3 to 4 medications with nearly 16% taking 5 or more, suggesting notable disease burden. The results offer clear evidence that more data are needed before conclusions can be drawn on the differences in health between overweight/obese BED and NBO individuals.
This study is not without limitations. We utilized a cross-sectional design. We studied a treatment-seeking sample of patients seen within 1 geographical area, and we recruited small numbers of BED patients who were somewhat younger than the NBO group (although this difference was not statistically significant). Finally, patients were required to be less than 65 years of age, and while uncontrolled hypertension and uncontrolled diabetes were rather broadly defined, patients were excluded if their conditions were outside of the aforementioned limits.
Our findings suggest that despite higher BMI, overweight/obese BED patients in primary care may not be at risk for poorer overall health than NBO patients, but multiple measurement factors must be considered in interpreting these findings. Future research should compare larger clinical and community BED and NBO samples controlling for psychiatric symptoms, include objectively measured diagnoses, and utilize standardized diagnostic criteria for both BED and metabolic syndrome.
Submitted: April 2, 2015; accepted June 15, 2015.
Published online: October 29, 2015.
Author contributions: All authors contributed equally to the literature review and manuscript preparation.
Potential conflicts of interest: None reported.
Funding/support: This study was supported, in part, by a National Institutes of Health career development grant to Dr Barnes (K23 DK092279). No additional funding was obtained for submitting this article.
Role of the sponsor: The National Institutes of Health were not involved in study design, collection, analysis, interpretation of data, the writing of the report, or in the decision to submit this article for publication.
Disclaimer: The article does not represent the views of the National Institutes of Health. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the US government.
Acknowledgment: The work referenced in this manuscript was conducted at Yale School of Medicine.
REFERENCES
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Arlington, VA: American Psychiatric Publishing; 2013.
2. Wilfley DE, Wilson GT, Agras WS. The clinical significance of binge eating disorder. Int J Eat Disord. 2003;34(suppl 1):S96–S106. doi:10.1002/eat.10209 PubMed
3. Kessler RC, Berglund PA, Chiu WT, et al. The prevalence and correlates of binge eating disorder in the World Health Organization World Mental Health Surveys. Biol Psychiatry. 2013;73(9):904–914. doi:10.1016/j.biopsych.2012.11.020 PubMed
4. Health, United States, 2012: With Special Feature on Emergency Care. Hyattsville, MD: National Center for Health Statistics; 2013.
5. Hsu LK, Mulliken B, McDonagh B, et al. Binge eating disorder in extreme obesity. Int J Obes Relat Metab Disord. 2002;26(10):1398–1403. doi:10.1038/sj.ijo.0802081 PubMed
6. Marques L, Alegria M, Becker A, et al. Comparative prevalence, correlates of impairment, and service utilization for eating disorders across US ethnic groups: implications for reducing ethnic disparities in health care access for eating disorders. Int J Eat Disord. 2011;44(5):412–420. doi:10.1002/eat.20787 PubMed
7. Mitchell JE, Mussell MP. Comorbidity and binge eating disorder. Addict Behav. 1995;20(6):725–732. doi:10.1016/0306-4603(95)00095-X PubMed
8. Telch CF, Agras WS. Obesity, binge eating and psychopathology: are they related? Int J Eat Disord. 1994;15(1):53–61. doi:10.1002/1098-108X(199401)15:1<53::AID-EAT2260150107>3.0.CO;2-0 PubMed
9. Specker S, de Zwaan M, Raymond N, et al. Psychopathology in subgroups of obese women with and without binge eating disorder. Compr Psychiatry. 1994;35(3):185–190. doi:10.1016/0010-440X(94)90190-2 PubMed
10. Abraham TM, Massaro JM, Hoffmann U, et al. Metabolic characterization of adults with binge eating in the general population: the Framingham Heart Study. Obesity (Silver Spring). 2014;22(11):2441–2449. doi:10.1002/oby.20867 PubMed
11. Bulik CM, Sullivan PF, Kendler KS. Medical and psychiatric morbidity in obese women with and without binge eating. Int J Eat Disord. 2002;32(1):72–78. doi:10.1002/eat.10072 PubMed
12. Johnson J, Spitzer R, Williams J. Health problems, impairment and illnesses associated with bulimia nervosa and binge eating disorder among primary care and obstetric gynecology patients. Psychol Med. 2001;31(8):1455–1466. doi:10.1017/S0033291701004640 PubMed
13. Kenardy J, Mensch M, Bowen K, et al. Disordered eating behaviors in women with type 2 diabetes mellitus. Eat Behav. 2001;2(2):183–192. doi:10.1016/S1471-0153(01)00028-9 PubMed
14. Rieger E, Wilfley DE, Stein RI, et al. A comparison of quality of life in obese individuals with and without binge eating disorder. Int J Eat Disord. 2005;37(3):234–240. doi:10.1002/eat.20101 PubMed
15. Bruce B, Agras W. Binge eating in females: a population-based investigation. Int J Eat Disord. 1992;12(4):365–373. doi:10.1002/1098-108X(199212)12:4<365::AID-EAT2260120404>3.0.CO;2-M
16. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA. 2001;285(19):2486–2497. doi:10.1001/jama.285.19.2486 PubMed
17. What is metabolic syndrome? National Heart, Lung, and Blood Institute Web site. http://www.nhlbi.nih.gov/health/dci/Diseases/ms/ms_all.html. Updated November 3, 2011. Accessed June 6, 2014.
18. Metabolic syndrome. Mayo Clinic Web site. http://www.mayoclinic.org/diseases-conditions/metabolic-syndrome/basics/ definition/con-20027243. Updated August 22, 2014. Accessed June 6, 2014.
19. Ford ES. Risks for all-cause mortality, cardiovascular disease, and diabetes associated with the metabolic syndrome: a summary of the evidence. Diabetes Care. 2005;28(7):1769–1778. doi:10.2337/diacare.28.7.1769 PubMed
20. Girman CJ, Rhodes T, Mercuri M, et al; 4S Group and the AFCAPS/TexCAPS Research Group. The metabolic syndrome and risk of major coronary events in the Scandinavian Simvastatin Survival Study (4S) and the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS). Am J Cardiol. 2004;93(2):136–141. doi:10.1016/j.amjcard.2003.09.028 PubMed
21. Grundy SM, Brewer HB Jr, Cleeman JI, et al; National Heart, Lung, and Blood Institute. Definition of metabolic syndrome: report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation. 2004;109(3):433–438. doi:10.1161/01.CIR.0000111245.75752.C6 PubMed
22. Taylor AE, Hubbard J, Anderson EJ. Impact of binge eating on metabolic and leptin dynamics in normal young women. J Clin Endocrinol Metab. 1999;84(2):428–434. PubMed
23. Jenkins DJ, Ocana A, Jenkins AL, et al. Metabolic advantages of spreading the nutrient load: effects of increased meal frequency in non–insulin-dependent diabetes. Am J Clin Nutr. 1992;55(2):461–467. PubMed
24. Herpertz S, Albus C, Lichtblau K, et al. Relationship of weight and eating disorders in type 2 diabetic patients: a multicenter study. Int J Eat Disord. 2000;28(1):68–77. doi:10.1002/(SICI)1098-108X(200007)28:1<68::AID-EAT8>3.0.CO;2-R PubMed
25. Hudson JI, Lalonde JK, Coit CE, et al. Longitudinal study of the diagnosis of components of the metabolic syndrome in individuals with binge eating disorder. Am J Clin Nutr. 2010;91(6):1568–1573. doi:10.3945/ajcn.2010.29203 PubMed
26. Mannucci E, Tesi F, Ricca V, et al. Eating behavior in obese patients with and without type 2 diabetes mellitus. Int J Obes Relat Metab Disord. 2002;26(6):848–853. PubMed
27. Barnes RD, Boeka AG, McKenzie KC, et al. Metabolic syndrome in obese patients with binge eating disorder in primary care clinics: a cross-sectional study. Prim Care Companion CNS Disord. 2011;13(2):doi:10.4088/PCC. 10m01050. PubMed
28. Crow S, Kendall D, Praus B, et al. Binge eating and other psychopathology in patients with type II diabetes mellitus. Int J Eat Disord. 2001;30(2):222–226. doi:10.1002/eat.1077 PubMed
29. Barnes RD, White MA, Martino S, et al. A randomized controlled trial comparing scalable weight loss treatments in primary care. Obesity (Silver Spring). 2014;22(12):
2508–2516. PubMed
30. Narbro K, Agren G, Jonsson E, et al; Swedish Obese Subjects Intervention Study. Pharmaceutical costs in obese individuals: comparison with a randomly selected population sample and long-term changes after conventional and surgical treatment: the SOS intervention study. Arch Intern Med. 2002;162(18):2061–2069. doi:10.1001/archinte.162.18.2061 PubMed
31. Raebel MA, Malone DC, Conner DA, et al. Health services use and health care costs of obese and nonobese individuals. Arch Intern Med. 2004;164(19):2135–2140. doi:10.1001/archinte.164.19.2135 PubMed
32. Segal L, Carter R, Zimmet P. The cost of obesity: the Australian perspective. Pharmacoeconomics. 1994;5(suppl 1):45–52. doi:10.2165/00019053-199400051-00009 PubMed
33. Sullivan PW, Ghushchyan V, Wyatt HR, et al. The medical cost of cardiometabolic risk factor clusters in the United States. Obesity (Silver Spring). 2007;15(12):3150–3158. doi:10.1038/oby.2007.375 PubMed
34. Swinburn B, Ashton T, Gillespie J, et al. Health care costs of obesity in New Zealand. Int J Obes Relat Metab Disord. 1997;21(10):891–896. doi:10.1038/sj.ijo.0800486 PubMed
35. Thompson D, Brown JB, Nichols GA, et al. Body mass index and future health care costs: a retrospective cohort study. Obes Res. 2001;9(3):210–218. doi:10.1038/oby.2001.23 PubMed
36. World Health Report 2000–Health systems: improving performance. World Health Organization Web site. http://www.who.int/whr/2000/en/. Accessed August 27, 2015.
37. Grilo CM, Masheb RM, White MA, et al. Treatment of binge eating disorder in racially and ethnically diverse obese patients in primary care: randomized placebo-controlled clinical trial of self-help and medication. Behav Res Ther. 2014;58:1–9. doi:10.1016/j.brat.2014.04.002 PubMed
38. Blomquist KK, Milsom VA, Barnes RD, et al. Metabolic syndrome in obese men and women with binge eating disorder: developmental trajectories of eating and weight-related behaviors. Compr Psychiatry. 2012;53(7):1021–1027. doi:10.1016/j.comppsych.2012.02.006 PubMed
39. Assmann G, Guerra R, Fox G, et al. Harmonizing the definition of the metabolic syndrome: comparison of the criteria of the Adult Treatment Panel III and the International Diabetes Federation in United States American and European populations. Am J Cardiol. 2007;99(4):541–548. doi:10.1016/j.amjcard.2006.08.045 PubMed
40. Tan CE, Ma S, Wai D, et al. Can we apply the national cholesterol education program adult treatment panel definition of the metabolic syndrome to Asians? Diabetes Care. 2004;27(5):1182–1186.
41. Fairburn CG, Cooper Z. The Eating Disorder Examination. In: Fairburn CG, Wilson GT, eds. Binge Eating: Nature, Assessment, and Treatment. 12th ed. New York, NY: Guilford Press; 1993:317–360.doi:10.2337/diacare.27.5.1182 PubMed
42. First M, Spitzer R, Gibbon M, et al. Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Patient Edition. (SCID-I/P). New York, NY: Biometrics Research, New York State Psychiatric Institute; 2002.
43. Reichborn-Kjennerud T, Bulik CM, Sullivan PF, et al. Psychiatric and medical symptoms in binge eating in the absence of compensatory behaviors. Obes Res. 2004;12(9):1445–1454. doi:10.1038/oby.2004.181 PubMed
44. Ervin RB. Prevalence of metabolic syndrome among adults 20 years of age and over, by sex, age, race and ethnicity, and body mass index: United States, 2003–2006. Natl Health Stat Rep. 2009;13(13):1–7. PubMed
45. Busetto L, Segato G, De Luca M, et al. Weight loss and postoperative complications in morbidly obese patients with binge eating disorder treated by laparoscopic adjustable gastric banding. Obes Surg. 2005;15(2):
195–201.doi:10.1381/0960892053268327 PubMed
46. Grucza RA, Przybeck TR, Cloninger CR. Prevalence and correlates of binge eating disorder in a community sample. Compr Psychiatry. 2007;48(2):124–131. PubMed
47. Wadden TA, Foster GD, Letizia KA, et al. Metabolic, anthropometric, and psychological characteristics of obese binge eaters. Int J Eat Disord. 1993;14(1):17–25. PubMed
48. American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
49. Smink FR, van Hoeken D, Hoek HW. Epidemiology of eating disorders: incidence, prevalence and mortality rates. Curr Psychiatry Rep. 2012;14(4):406–414. doi:10.1007/s11920-012-0282-y PubMed
50. Thomas JJ, Koh KA, Eddy KT, et al. Do DSM-5 eating disorder criteria overpathologize normative eating patterns among individuals with obesity? J Obes. 2014;2014:320803. doi:10.1155/2014/320803 PubMed
51. Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83(3):457–502. doi:10.1111/j.1468-0009.2005.00409.x PubMed
Enjoy this premium PDF as part of your membership benefits!


